Abstract
Despite the plethora of published studies on intracranial aneurysms (IAs) hemodynamic using computational fluid dynamics (CFD), limited progress has been made towards understanding the complex physics and biology underlying IA pathophysiology. Guided by 1733 published papers, we review and discuss the contemporary IA hemodynamics paradigm established through two decades of IA CFD simulations. We have traced the historical origins of simplified CFD models which impede the progress of comprehending IA pathology. We also delve into the debate concerning the Newtonian fluid assumption used to represent blood flow computationally. We evidently demonstrate that the Newtonian assumption, used in almost 90% of studies, might be insufficient to describe IA hemodynamics. In addition, some fundamental properties of the Navier–Stokes equation are revisited in supplementary material to highlight some widely spread misconceptions regarding wall shear stress (WSS) and its derivatives. Conclusively, our study draws a roadmap for next-generation IA CFD models to help researchers investigate the pathophysiology of IAs.
Introduction
With 50–60% mortality after rupture and 30–40% dependence rate amongst survivors, 1 intracranial aneurysms (IAs) are one of the most feared cerebrovascular pathologies. While treatment options are very well established in the medical field, either in the form of surgical clipping, with its modifications based on the location, geometry, complexity, etc., or endovascular treatment with its various forms,2,3 there exists a dilemma that is yet to be solved. Approximately 50–80% of IAs do not rupture in the individual's lifetime. 4 While it is the standard to treat all ruptured aneurysm cases upon detection, 3 unruptured aneurysms make up for a difficult decision making. Unruptured aneurysms can be followed for certain period of time, especially smaller sized ones. 5 However, while changes in aneurysm morphology or size are warning signs against impending rupture, there is no telling how fast such changes will occur6–8 and treating unruptured aneurysms is certainly not a risk-free option.9–11 Therefore, taking chances in IA care do not only risk the livelihood of patients, but it also multiplies the cost of healthcare delivery as well as burden of the healthcare services. 12 Given the aforementioned factors, the cerebrovascular research into IA pathobiology needs not only to answer questions regarding the formation and growth of IAs, but also seeks to answer why and how aneurysms rupture. A question that may seem direct at the first glance, yet it is made complex by the intertwining relationships between biological and physical factors that are yet to be fully understood.
Therefore, to understand the pathobiology of IAs, it is imperative to understand the different flow regimes in the intracranial vessels and inside the IAs and how they affect the ECs and consequently the vessel wall. To understand the blood flow dynamics, researchers had to turn to the field of fluid mechanics, modifying their tools to be used for the study of blood flow in various vessels, connecting the dots and ultimately searching for a holistic understanding of the pathology of IAs. The most famous and ubiquitous of these tools used nowadays is computational fluid dynamics (CFD) analysis.
It is a very well established fact that endothelial cells (ECs) sense the blood flow via a variety of cell surface and intracellular mechano-sensors, 13 and they transmit signals of the blood flow into cellular response that derives the vascular homeostasis.13,14 Any abnormality in the blood flow induces pathologic responses in the ECs, causing vascular diseases such as atherosclerosis.15–17 ECs also were shown to transmit flow signals to the underlying cells in the vascular wall, such as vascular smooth muscle cells, via exosomes and microRNA transfer. 17 Moreover, ECs were shown to respond to wall shear stress (WSS) via several mechanobiological mechanisms.18,19 From such observation, it is safe to base the model of IAs formation, growth and rupture on an interaction between blood flow and ECs. Even in ruptured aneurysms where the ECs are lacking, 20 such a finding must be linked with ECs death and shedding, possibly leading to an accelerated aneurysmal wall weakening and eventual rupture from the loss of the normal buffer between the flow and the wall (i.e. the ECs and their intercellular junctions15,21,22). ECs damage is associated with internal elastic lamina pathophysiologic changes and damage, as well as inflammatory cell recruitment, via cytokines and other molecules, and aneurysm wall degeneration or sclerosis.23,24 Moreover, abnormal collagen fibers in the aneurysm wall indicate a failed wall remodeling. 25 Indeed, differences in wall inflammation and internal elastic lamina were observed between ruptured and unruptured aneurysms. 24 This indicates that the pathophysiology of IA, while might start at the level of ECs, is in fact spanning all the layers of the wall and should be studied with this in mind.
Early works on IA hemodynamics
There is almost a consensus in literature on the prime role of wall shear stress (WSS) as the most profound hemodynamic parameter affecting aneurysm fate. Such consensus was established following to the early work of Ferguson et al.26–28 in the early 1970s, in which they constructed in vitro flow models of IA geometries guided by craniotomy observations. Their work was revolutionary at the time as they suggested that transition to turbulence occurs in IA and lead to degeneration of internal elastic lamina and rapid weakening of the aneurysm wall. 27 Their work was followed by a series of studies by Steiger et al., 29 Liepsch et al., 30 Gonzalez et al. 31 and Kim et al. 32 which settled the consensual WSS theory in IA hemodynamics. 33 Steiger's group work was the first recorded in vitro measurements of IA flow fields using laser techniques.34,35 Until that time in the late 1970s to early-mid 1980s, in vitro works postulated that blood flow in IA exhibits turbulence and transitional features which contribute to the IA growth and wall deterioration,26,36,37 which is in line with recent biological findings. 15 Moreover, such early works considered the non-Newtonian effects of IA hemodynamics influential.34,38–40 Wall shear stress in these studies was only a measure of the flow at the IA wall, not an independent variable that thought to control IA pathophysiology and mechanotransduction.
CFD and current controversies
In the past two decades, CFD has been an effective research tool for investigating numerous aspects of IA,10,41–43 most importantly growth and rupture mechanisms and their relation with vascular hemodynamics.44–46 IA CFD simulations mostly aim to evaluate the morphologic factors and hemodynamic forces on the aneurysm walls 47 which trigger a set of vascular remodeling mechanisms48,49 leading to IA progression and possible rupture. 50 Moreover, CFD is used to study the growth and rupture of IAs, as well as assessing different endovascular treatment modalities 51 and validating new endovascular devices.8,10,11 While CFD and flow studies have significantly improved our understanding of vascular pathologies such as atherosclerosis and that the flow perturbations at bifurcations were linked to evident pathobiological processes,15,52 such an understanding is yet to be reached in the field of IAs research, and a large gap of knowledge still exist. This gap of knowledge is in part due to the complexity of blood flow in IAs as well as controversies regarding the actual flow regime causing rupture.10,53 These controversies are related to the actual inducer of aneurysm rupture, whether it is high or low WSS 54 and to the actual viscosity model that should be adopted to model the blood flow, whether Newtonian or non-Newtonian. 55 Moreover, there are controversial issues pertaining to the validity of the some of the parameters used in the analysis of the flow regimes, such as the vector calculations of WSS, that need to be addressed. 56 Thus, we believe it is vital to evaluate the tools we are using to understand IAs, highlighting the controversies and unveiling the shortcomings in our approaches and tools to be able to progress and improve.
The optimality principle, proposed by Murray, 57 which is often used in CFD simulations, correlates arterial hemodynamics to the dimensions of individual arteries via a power-law correlation. The major consequence of such principle is that any vascular remodeling due to hemodynamic changes should eventually be attributed to deviation from the optimal geometrical parameters. However, Murray's principle was derived assuming steady laminar flow in straight vessels, and no direct communication between arteries of the same rank. This is not the case in the circle of Willis or any intracranial vessel. 58 In vivo evidence showed that Murray's law could be valid on the scale of the system; however, it is incapable of accurately predicting in vivo WSS measurements in intracranial arteries.59–61 This leaves the coupled interaction between IA and hemodynamics, on different scales, an open question beyond any geometrical characterization attempts. Consequently, this coupled interaction inherits multi-layered complexity from the morphological sophistication of the circle of Willis, 62 let alone the fluid dynamics intricacies driven by the multi-harmonic blood flow waveform which exhibits phase shifts,63,64 non-linear dynamics, 65 and a multitude of biochemical, biophysical and mechanobiological processes. 66
Aim and motivation
The aim of this work was to evaluate the current stand-point of the CFD research on IAs by performing a meta-analysis of all the literature concerning IA hemodynamics and critically reviewing it. The ultimate aim is to evaluate the tools and techniques used in order to provide a recipe for improvement. Such an improvement will enhance the outcome of CFD research and provide a better link between the physics of the blood flow and the biological processes observed in the IAs. Through our meta-analysis, we came across several open-ended questions that we will try to either answer or highlight and present to the readers. These questions include:
What are the current trends in IA hemodynamics research? Does the Newtonian assumption, widely used in CFD models, accurately represent physiological conditions? What is the actual flow regime of blood in intracranial aneurysm? In the light of current controversies in CFD literature, what is the clinical and biological relevance of such models with respect to the initiation, growth and rupture of intracranial aneurysm?
We aim to tackle these questions based on the contemporary paradigm driven from published studies in the field. The final target of this meta-analysis and critical review is to stimulate debate, critical analysis and falsification of such paradigm to advance IA CFD models towards future theoretical and applied relevance.
Methods and data
Meta-analysis on IA CFD studies
We obtained the literature records and references from Scopus bibliographic database using TITLE-ABS-KEY search function with the following search terms:
– “Cerebral aneurysm” OR “Intracranial aneurysm” – AND “Hemodynamics” OR “Flow dynamics” OR “Blood flow”
Then we classified the references corpus, which contained 1733 published articles, manually through reading the abstracts and conclusions and grouping the articles into groups based on the technique of study (CFD or other techniques including in vivo, in vitro studies and animal models). Then, the CFD references, which counted 795 papers were analyzed in detail for technique, methods and models used. All the references obtained from the meta-analysis search records were classified according to their relevance to the topic of this study. The details and findings of such classification and analysis are presented in the Results and Discussion section. The meta-analysis was conduced in line with the PRISMA guidelines. 67
Meta-analysis criteria and mathematical model for WSS comparison
Literature records have been searched on Scopus® database for studies reporting the dimensions and flow rates or velocities of intracranial and cerebral arteries in healthy subjects. Patients suffering from intracranial aneurysm are not necessarily suffering from other disorders that could alter their blood viscosity. In addition, blood flow velocity inside aneurysms is often much less than such of the parent artery. Hence, it is valid to assume that the blood viscosity behavior in aneurysm patients is similar to such in healthy patients for the scope of the present study. The studies were selected such that each study provide synchronized readings for the blood velocity (m/s) or flow rate (m3/s) with the vessel diameter (m) or cross-sectional area (m2) during specific instants of the cardiac cycles or as mean values for one cardiac cycle. The details of such studies are summarized in Supplementary Table 1. A summary of the measurements is plotted in Supplementary Figure 1. The collected measurements from selected studies were used to compute the shear rate (s−1) based on the exact Hagen-Poiseuille solution of Newtonian incompressible flow68–70 as following
The rise of simplified CFD models of IA hemodynamics during the past two decades. (a) Exponential growth in the use of CFD as a research tool in aneurysm hemodynamics based on Scopus® database. Search query available in the supplementary materials. (b) Comparison of the citation counts (2000–2018) from Scopus® bibliographic database between the early in vitro works reporting complex IA flow physics (black column indicates total annual citations for references34,38,39) and subsequent CFD models adopting simplified assumed flow physics (blue outlined column indicates total annual citations of references85,87,234). Hypothesis testing of the Newtonian assumption based on WSS calculations from in vivo radiological measurements. Note: The sample population consists of the WSS values from all the arteries in the meta-analysis.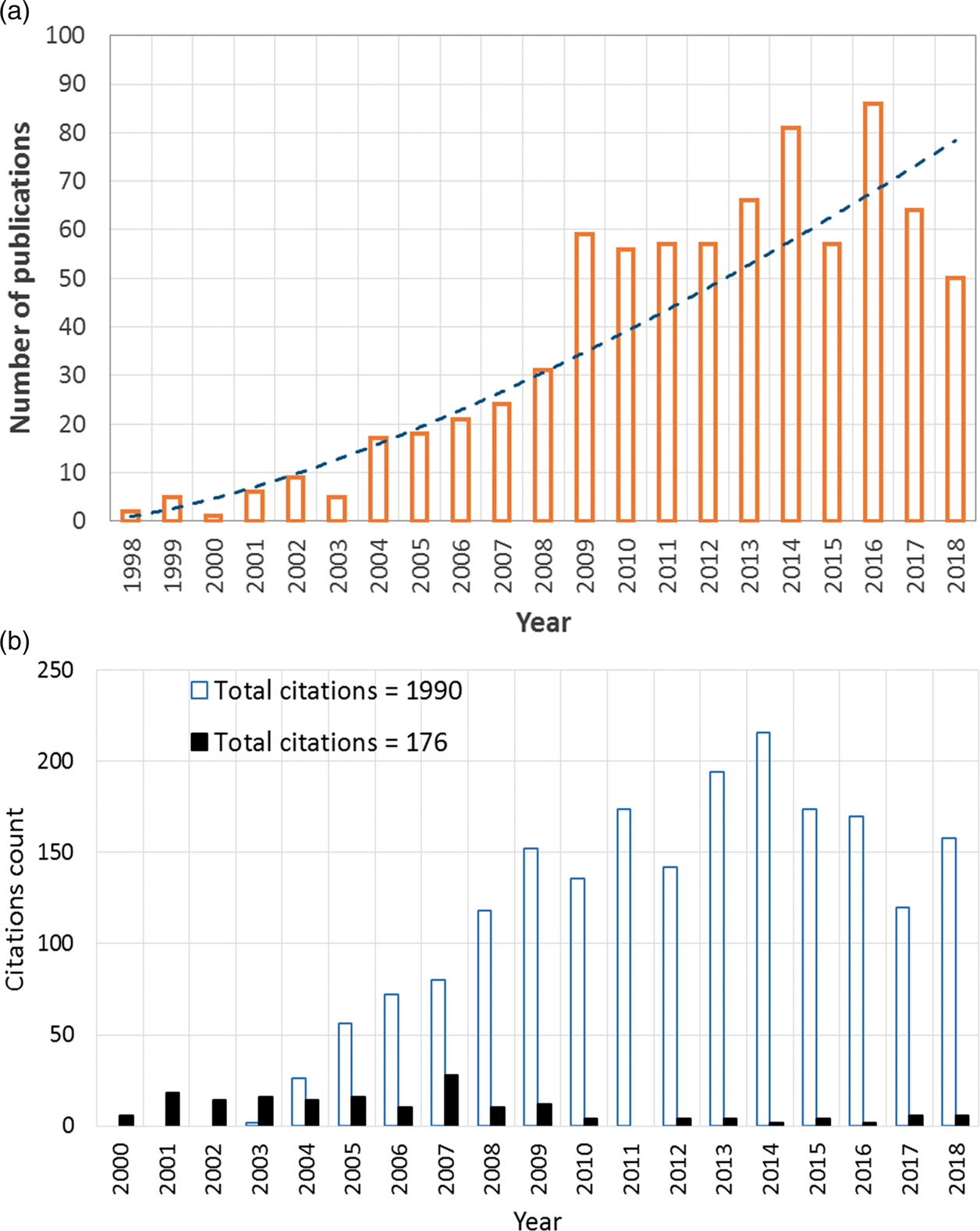
For shear thinning fluids, where velocity profiles deviate from the parabolic profile shown by the Hagen-Poiseuille solution, shear rate values should be corrected using the Weissenberg–Rabinowitsch (W-R) shear-thinning correction71,72



Power-law (PL)
73
:

Carreau (C)74–76:

Carreau-Yasuda (C-Y)
77
:

Casson (CS)78,79:

Cross (CR)
81
:

Hypothesis testing and statistical analysis
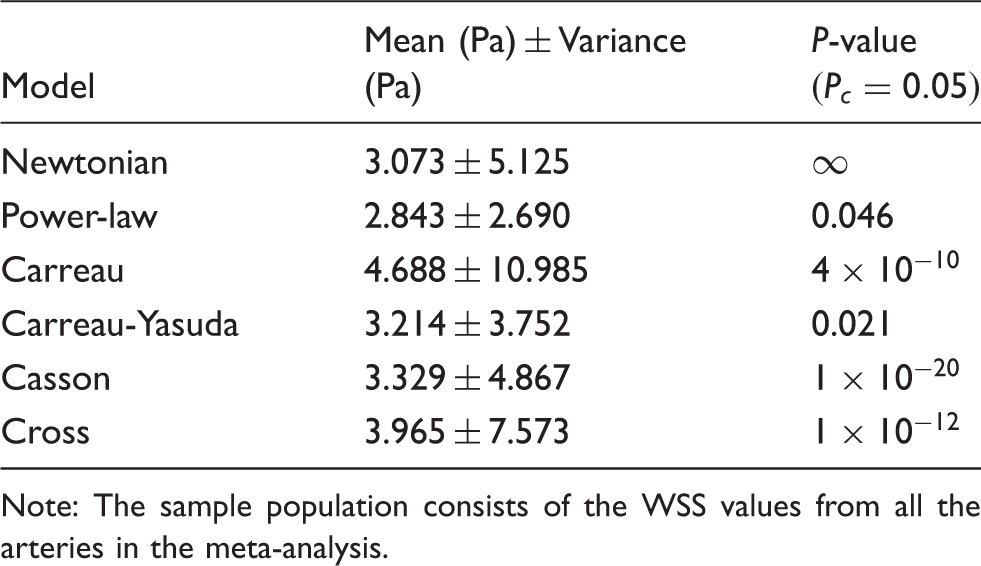
The null hypothesis of the WSS comparative study was that the Newtonian and non-Newtonian calculations of WSS are similar with no significant differences. This is the essence of using Newtonian viscosity in CFD simulations. Paired t-tests were conducted to compare the Newtonian (N) model with each of the five non-Newtonian rheological models that are commonly used in literature, given by equations (6) to (10).
In vivo calculations of WSS in IAs
The validation of CFD models against in vivo measurements is a crucial task, yet not commonly practiced in the IA hemodynamics research community. Here, we explored the studies which reported in vivo estimations of WSS in IA. In principle, different radiological techniques such as MRI or transcranial color Doppler (TCCD) are used to measure the velocity time-series in the region of interest. Then, post-measurement calculations are conducted to estimate WSS as function of the measured velocity series and geometry. A comparative analysis based on six studies, as summarized in Table 2, which reported in vivo post-measurement calculations of WSS in IAs was conducted. The criteria for selecting the six studies were as following:
– Usage of high-fidelity multidimensional radiological technique (3D/4D MR) – Study must report quantitative and qualitative results – The method of computing WSS sufficiently explained in each study in terms of adequate use of measurements data and fully explained mathematical model. Summary of in vivo estimations of hemodynamic parameters in IA. Note: WSSrel is the ratio between average WSS in the aneurysm and parent artery, respectively, while WSSIA is the average value of WSS in IA. Uavg is the spatial range of maximum velocity inside the aneurysm per study and
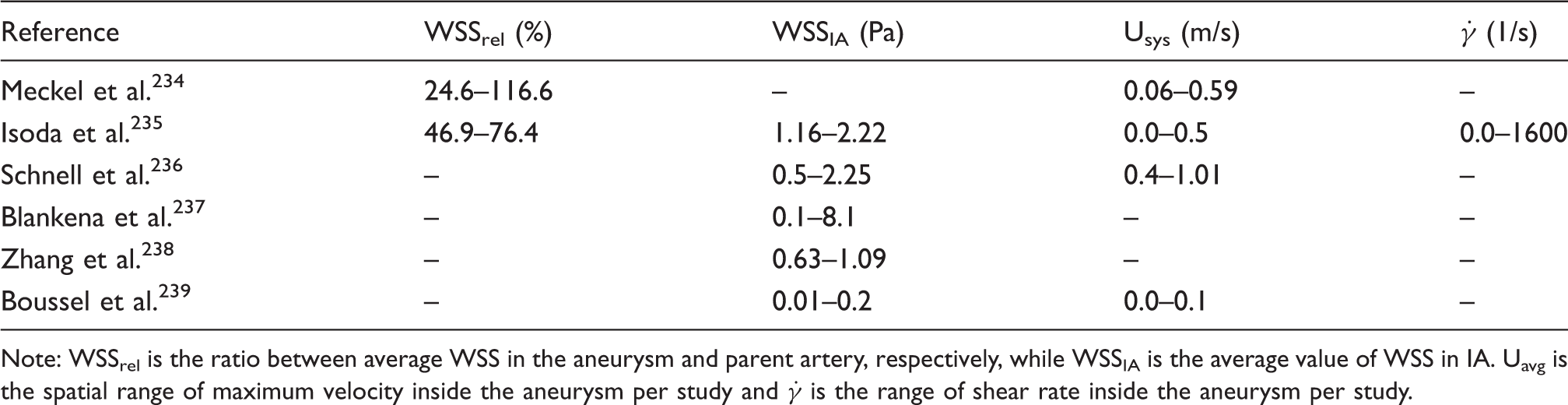
The values of WSS presented in such studies are reviewed and analyzed. The purpose of this analysis is to explore the hemodynamics of IA as detected in vivo to provide benchmark to discuss the current hypothesis proposed by CFD studies.
Results and discussion
CFD and deviation towards simplification
In the 1990s, and with the large leap in CPU speed and radical increase in PC availability, CFD has emerged as a research tool to investigate IA hemodynamics.82,83 Figure 1(a) shows the trend of using CFD as a research tool in cerebral aneurysm hemodynamics based on Scopus® database. Nevertheless, IA CFD simulations, attempting to provide clinically relevant analyses, had adopted assumptions far from the experience of the earlier in vitro works. Now, the highest citations of CFD studies goes to works that have assumed Newtonian blood viscosity84,85 and laminar flow with no transition to turbulence.86–88 Figure 1(b) shows a comparison between the citations of early in vitro laser measurements encompassing complex physics (i.e. non-Newtonian effects, transition to turbulence) compared with the early CFD works with simplified physical assumptions (i.e. Newtonian viscosity, laminar flow). As a result of such relaxed assumptions in early CFD simulations, the argument favoring the role of high WSS in aneurysm rupture84,89 challenged an opposing argument correlating low WSS with IA wall disintegration and degeneration leading to rupture.85,90–92 While in some other CFD studies, low 93 as well as high 94 WSS were controversially correlated to the growth of IA. Several articles95–99 have highlighted such controversies along the past two decades and discussed the discrepancy of findings, however without providing enough reasoning to their root causes. The recent works of Can et al., 100 Zhou et al., 101 and Meng et al. 102 are exemplary endeavors to find coherence among the large IA CFD corpus, however, without definitive and conclusive findings, especially with relevance to IA mechanobiology and clinical applications.
In addition to the attempts of unfolding the physics of IA hemodynamics using CFD, CFD has been a very useful tool in the design, development, and evaluation of endovascular management methods. The term virtual stenting has attracted many research groups around the world to empower endovascular management with CFD decision-making tools.103–106 Adopting simplified flow physics as the mainstream IA CFD works, many research groups have evaluated the embolization efficacy of flow diverters and stents107,108 and IAs endovascular coiling. 11 In the matter of fact, much of the development in endovascular devices can be attributed to the availability of CFD as an optimization tool for the design, testing and synthesis of such devices. However, the incomplete understanding of complex blood flow dynamics has maintained the concept of such devices with minimal progress along the years. Devices used to treat IAs depend on one concept: flow isolation.109–112 Stents, coils and flow diverters aim at isolating the aneurysm cavity from the main flow, hence allowing ECs to recover and the vascular wall to heal. In addition to the design complexity of such devices,113,114 their efficacy depends on IA geometry, morphology, location and other inherent factors of the IA.115,116 It can safely be argued that a better understanding of IA hemodynamics, and blood dynamics at large, is necessary to radically improve the current design theory of endovascular devices.
Linking hemodynamics to IA mechanobiology
Well-established in vitro studies of endothelial cells response to WSS, which is relevant to IA mechanobiology, 117 depend on the Newtonian assumption.118–120 In such studies, EC cultures are subjected to different levels and regimes of WSS, created by simple Poiseuille-type Newtonian fluid flow,119,121–123 to investigate the cells response to each WSS level and regime. It has been established, through the vast majority of these studies, that endothelial dysfunction (ED) associated with cerebral aneurysm occurs under WSS regime which can be best classified under disturbed/turbulent flow rather than uniform/laminar flow.118,124–126 Chiu and Shien 118 showed that low values of WSS, often associated with distrubed/turbulent flow, are associated with long-term ED and remodeling processes similar to those encountered in IA growth. In a comprehensive and extensive meta-analysis, Zhou et al. 127 has also showed that IA rupture is often associated with low WSS values inside the aneurysm. Now, it has been established that EC respond to laminar flow by supressing pro-inflammatory transcription agents and respond to distrubed (i.e. turbulent-like) flow by upregulating inflammatory and chemotactic mediators. 128 However, these findings have not been reflected on the published CFD studies of IA hemodynamics, as shown in Figure 3(c) and (d) and discussed in the next section, such that only 31% of the studies considered the pulsatile nature of blood flow, while 4% of such studies attempted to explore turbulent-like structure that might exist in IA hemodynamics, as referred to from EC research.
Classification of IA CFD models
CFD models of IA can be generally classified according to the level of complexity to two major categories: ideal and patient-specific models. Figure 2 shows a schematic of the different parameters determining the level of IA CFD model complexity, as explained henceforth. Ideal models are often based on assumed geometry with anatomically relevant dimensions used to study the morphological patterns129–133 and Neuroendovascular device efficacy.134,135 Some of the ideal model geometries are actually constructed from averaging of patient-specific dimensions across a specific population to compare the relevance of gender,
136
morphology
137
or complex cases of IA.
138
Patient-specific models are often constructed from DSA images,
139
and then converted to volume meshing suitable for any adequate CFD solver.
41
The discretization of the reconstructed 3D models into computational grid dictates the flow regime and limits the underlying physics in the solution. If the computational grid and time marching scheme are sufficiently accurate, it would be possible to capture the flow specific physics taking place at higher frequencies
140
such as flow instabilities141,142 or transition.143,144 It is also essential to define the solution method, boundary conditions, and the viscosity model to solve the Navier-Stokes equations (NSE) for the blood flow in the aneurysm geometry of interest. There are three main methods for solving NSE numerically for IA blood flow scenarios: the generic finite volume method,
145
adapted finite element methods,146–148 or the lattice Boltzmann method149–151 which was recently introduced to the field after success in other numerous applications. The boundary conditions required by any NSE solver for an IA CFD simulation are either steady, pulsatile (i.e. sinusoidal) or Womersley waveform. Steady flow boundary conditions are usually used to provide qualitative assessment of the aneurysm flow field and WSS;
152
however, they are considered to be a form of idealization even if used with patient-specific geometries as they neglect some of the main phenomena in aneurysm hemodynamics and reduce the clinical relevance of the CFD models.153–155
The parameters and assumptions which define the complexity of CFD models. The meshing resolution of the flow domain, boundary conditions as well as the viscosity models and solver numerical settings define the flow physics predicted by the CFD model and whether or not it could model transitional and turbulent features of the flow. Classification of published studies on intracranial anereurysm hemodynamics with respect to physical assumptions in the CFD models, as revealed by Scopus® bibliographic databases. A total number of 1733 publications was found by searching Scopus® on 14 October 2018. This classification was conducted using sequential searches via Scopus® analytic tools. Classification methodology is explained in the methods section. Articles using CFD as a method of investigation comprised 47% of the literature on aneurysm hemodynamics (a). Of these CFD-based articles, only 10% used the non-Newtonian viscocity assumption to investigate aneurysm hemodynamic features (b), 31% used unsteady and pulsatile models (c) and only 4% investigated turbulence and transition to turbulence (d).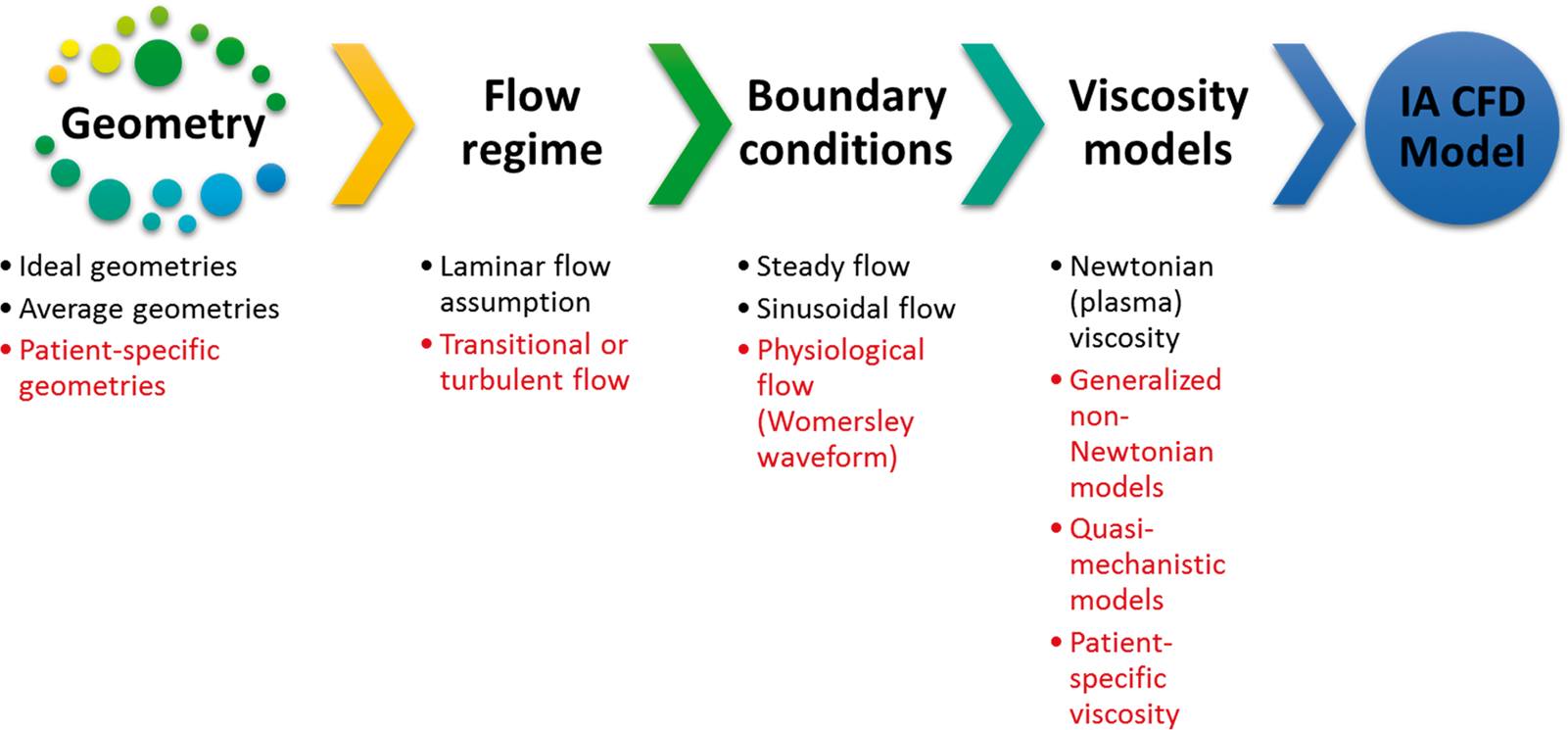
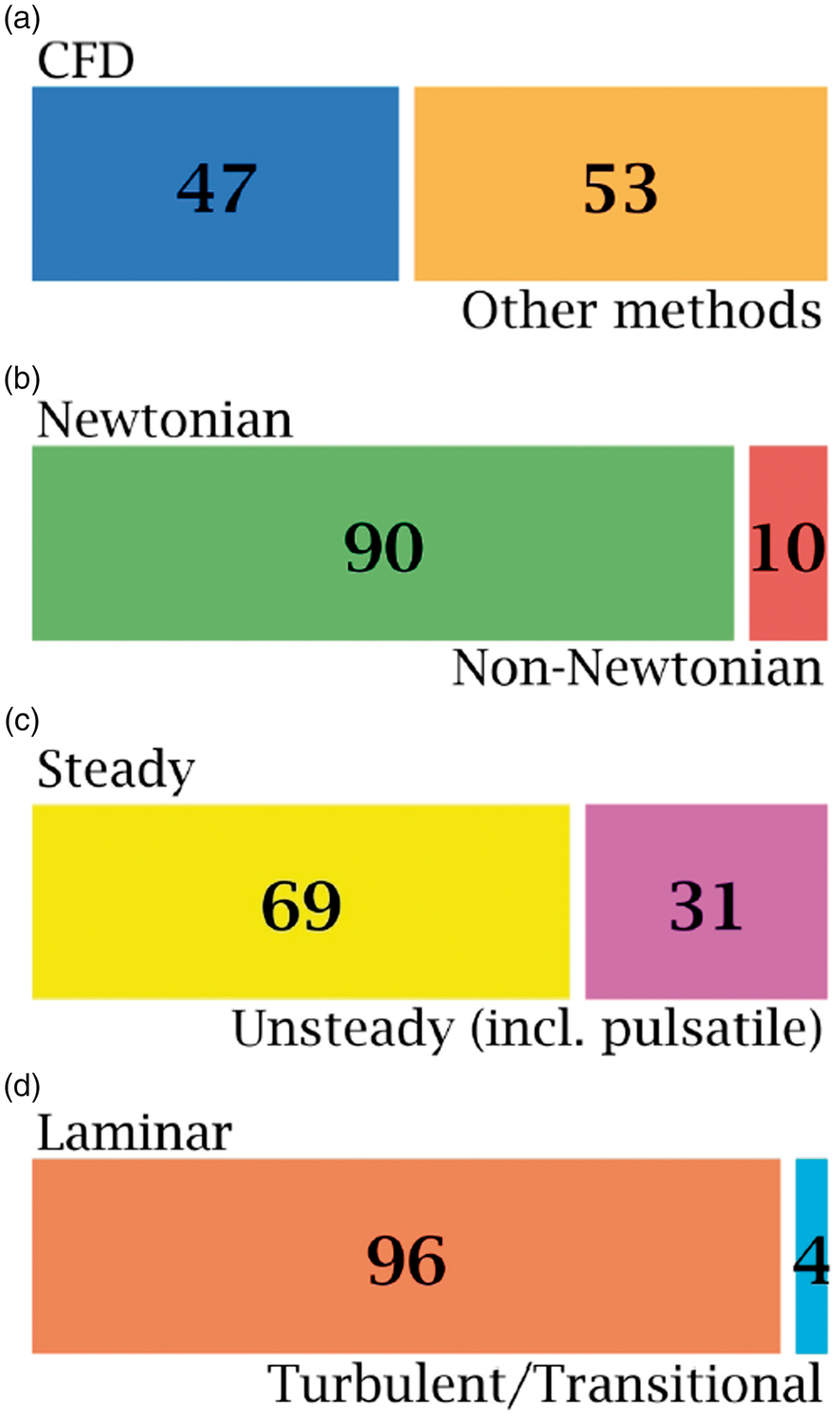
As to the blood viscosity models, the majority of IA CFD simulations have adopted Newtonian viscosity assumption when solving the NSE for either steady or pulsatile flows.10,45,75,84,156,157 Bibliographic meta-analysis from Scopus® database revealed interesting insights about the current paradigm of IA CFD models. The most important aspects of any IA CFD model are the viscosity, transient nature, and flow regime as resolved by the computational grid. Figure 3 summarizes such aspects as revealed by surveying the IA hemodynamics literature. It was found that 47% of published literature studying IA hemodynamics was based on CFD models, while the remaining 53% was shared by in vitro, in vivo, pathological and other research methods in the field. Only 10% of the published CFD studies to date have considered the non-Newtonian viscosity of blood in modelling intracranial aneurysm hemodynamics. In fact, most of the former studies relied on the assumption that blood viscosity follows a Newtonian behavior in cerebral arteries since the shear rates are presumed to be higher than the range required for the non-Newtonian properties to become effective.158–161 Tracing the origins of such assumption was quite difficult, and a considerable number of the highly cited studies in the field have not discussed such assumption in details,87,162,163 or just refer to the parent artery of the aneurysm to be large enough to neglect the non-Newtonian properties of blood.164,165 While some other studies have arbitrarily used this assumption with some sort of basic physical reasoning,84,166 some studies simply cite classic publications on erythrocytes167,168 or plasma169,170 viscosity to be dominantly Newtonian in in vitro measurements, hence, justifying the use of their values in complex in silico CFD simulations.
Hemodynamic instabilities and transitional features of aneurysmal flow
In the past few years, some Direct Numerical Simulation (DNS) studies171–173 were conducted to provide detailed physical insight onto aneurysm hemodynamics. Such studies were performed using the Newtonian viscosity assumption. According to these studies, blood flow in cerebral and intracranial aneurysms exhibits turbulent-like structures and shows subcritical transitional behavior (Reynolds < 103, Womersley < 10) which was shown by monitoring the power spectrum of the velocity time-series at different locations of patient-specific aneurysms. Another study, by the same research group, reported patient-specific CFD simulations of intracranial aneurysm hemodynamics to examine the impact of a non-Newtonian viscosity model in comparison to the effect of solution methods on the results of WSS and Oscillatory Shear Index (OSI). 174 The simulation results, deemed to represent a DNS solution, showed that the impact of non-Newtonian assumption is insignificant in comparison to the impact of solution methods. However, the study has not provided sufficient information on the impact of non-Newtonian viscosity on the turbulence-like structures found in aneurysmal flow as reported previously by the same group of researchers.171–173 It is well known that non-Newtonian shear thinning fluids, such as blood, are more asymptotically and monotonically stable than Newtonian fluids, such that subcritical transition to turbulence becomes unlikely to take place.175–177
The credibility of the DNS results comes from the fact that in DNS, the Navier-Stokes equation is solved to the highest possible resolution of length and time scales.178,179 Beside its theoretical rigor,
180
DNS has been reproducibly validated in numerous works.181,182 On the other hand, in commonly used CFD solvers and settings among the community, only the laminar scales are resolved. Hence, the latter solvers dictate in priori simplified physics in the solution. Therefore, and despite their expensive computational cost, DNS solvers have been extensively used for four decades in all areas of fluid dynamics research,183–185 except in hemodynamics until recently.
186
In DNS settings, the smallest grid cell in the fluid domain should be smaller than the smallest vortex in the flow. The total number of grid cells is often proportional to
It must be noted that the classical theory of hydrodynamic stability has been established to study steady flows subjected to finite perturbations in space and time. 188 The physics underlying transition to turbulence in viscous incompressible flow, as in simple geometries such as straight pipes or under shear conditions, remains in question until today.189–192 Previous investigations of transition to turbulence in pulsatile pipe flow are limited and have not established a consensus regarding this critical condition.193–196 It must also be noted that most of such works considered monoharmonic pulsatile flow 196 with very few works that aimed at discussing multiharmonic pulsatile flow which is the nature of the blood flow. 197 Blood flow in IA exhibits several fluid dynamics phenomena depending on the morphology of parent and daughter arteries, as well as the size and location of the IA. Shear layers, 198 precessing vortices,199,200 jet impingement,201,202 recirculation and separation zones203,204 are inherently found in IAs. IA hemodynamics, therefore, are quite different from pulsatile pipe flows investigated in literature for turbulence transition criteria.194–196,205,206 It is impossible to apply the criteria of laminar-turbulence transition, classically devised for flows in straight pipes, to characterize IA flows. Therefore, fundamental fluid dynamics research is needed to characterize the transition to turbulence in IA flow.
Non-Newtonian properties of intracranial blood flow
Here, we concisely reviewed the previous studies aimed at evaluating the significance of the Newtonian assumption on WSS predictions. It was shown that the use of Newtonian viscosity results in overpredictions of aneurysm wall shear stress (WSS) 207 which could compromise rupture risk prediction (RRP) efficacy. 208 Frolov et al. 209 conducted in vitro comparative study between Newtonian and non-Newtonian blood surrogate fluids to investigate WSS distribution in patient-specific internal carotid artery aneurysm (ICAA) using 1D Laser Doppler Anemometry (LDA) with micrometer-scale spatial resolution. Their results showed that the use of Newtonian fluid over-predicts WSS by 11.7–19.7% at the aneurysm neck and dome, respectively. Hippelheuser et al. 210 investigated the hemodynamics of 26 patient-specific cerebral aneurysm using Newtonian and non-Newtonian (Carreau) viscosity models. Their comparative analysis showed significant differences in time-averaged wall shear stress (TAWSS) between the Newtonian and Carreau models. Such difference impacts the predicted rupture criteria of the aneurysm via CFD. Sano et al. 211 conducted comparative analysis on cerebral aneurysm hemodynamics using CFD. They used patient-specific viscosity data in comparison with the generalized Newtonian assumption. They found up to 25% difference in the normalized wall shear stress (NWSS) between the non-Newtonian and Newtonian simulations. They concluded that any rupture risk criteria, irrespective from aneurysm size, should be significantly affected by the viscosity model used in the CFD simulation. Otani et al. 212 simulated the hemodynamics of a coiled patient-specific cerebral aneurysm using two viscosity models. They used the Carreau-Yasuda non-Newtonian model in comparison with the generalized Newtonian viscosity assumption. They evidently showed that the use of non-Newtonian viscosity model in CFD produces flow structure and hemodynamic parameters that are different from such produced by the Newtonian assumption. More specifically, they identified that the Newtonian assumption underestimates the low shear-rate regions which could possibly skip thrombus formation features in the aneurysm. Moreover, Xiang et al., 213 Morales et al., 214 and Owen et al. 215 showed noteworthy differences between the Newtonian and various non-Newtonian blood viscosity models resulting in significant differences in predicted hemodynamics of intracranial aneurysm.
Newtonian vs non-Newtonian WSS calculation
Recently, we 55 provided the first in vivo evidence of the large discrepancy between non-Newtonian and Newtonian blood viscosity assumptions in major cerebral arteries from Doppler ultrasound measurements. The following section employs somehow similar methodology, 55 however, with measurements collected from a large pool of radiological studies using different techniques. The measurements and analytical solutions of the Hagen-Poiseuille and Weissenberg-Rabinowitsch equations are comparatively assessed using statistical paired t-tests to evaluate the null hypothesis representing the Newtonian assumption.
The calculations of WSS based on the mathematical model presented in methods section showed considerable variations between the Newtonian and non-Newtonian models, as shown in Figure 4. Different non-Newtonian models over-predict and under-predict Newtonian WSS by values corresponding to 2 and 4 Pa, respectively, as shown in Figure 5. The statistical t-test results presented in Table 1 show statistically significant differences between WSS calculation using Newtonian and different non-Newtonian models. In CFD calculations, the Weissenberg-Rabinowitsch (W-R) shear-thinning correction is not applied since the non-Newtonian closure of the shear stress term in Navier-Stokes equation is solved explicitly with the velocity equation. The coupling parameter is therefore the shear rate tensor. Here, the W-R correction is applied in order to ensure that the non-Newtonian calculations of WSS mimic such that from CFD simulations in terms of its proportionality with the Newtonian calculations. The W-R correction has been extensively verified and validated.216,217 Therefore, the statistical t-tests applied here represent meaningful comparison between WSS calculated from Newtonian and non-Newtonian models.
Box plots of WSS as calculated by Newtonian and five different non-Newtonian models in different intracranial arteries which are known for harboring IAs. Box plots were computed using m means for a total of n measurements by artery. The red midline in the box plots represents the median of WSS per artery per model for all studies. Models compared: Newtonian (N), Power-law (PL), Carreau (C), Carreau-Yasuda (C-Y), Casson (CS) and Cross (CR). WSS estimation difference 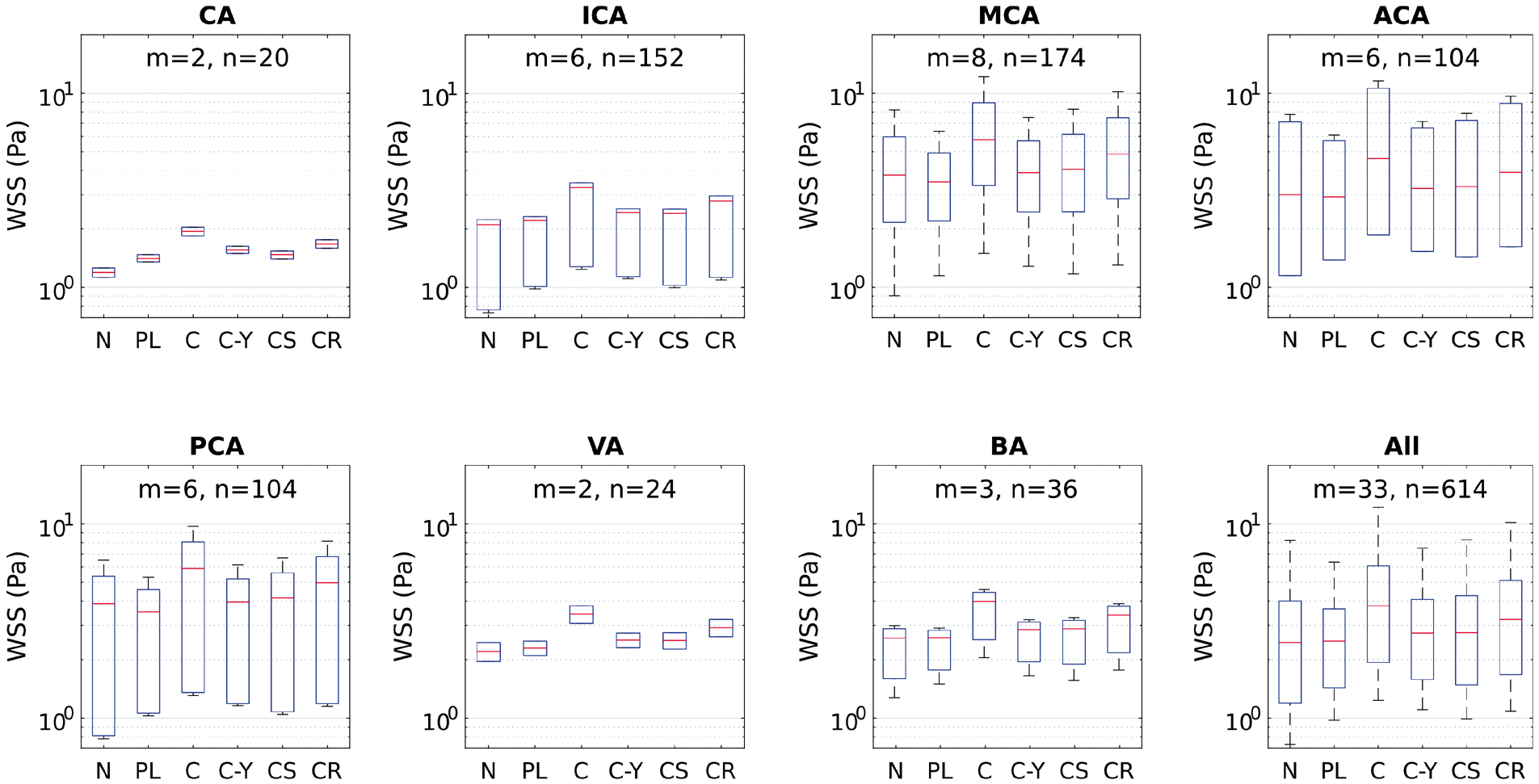
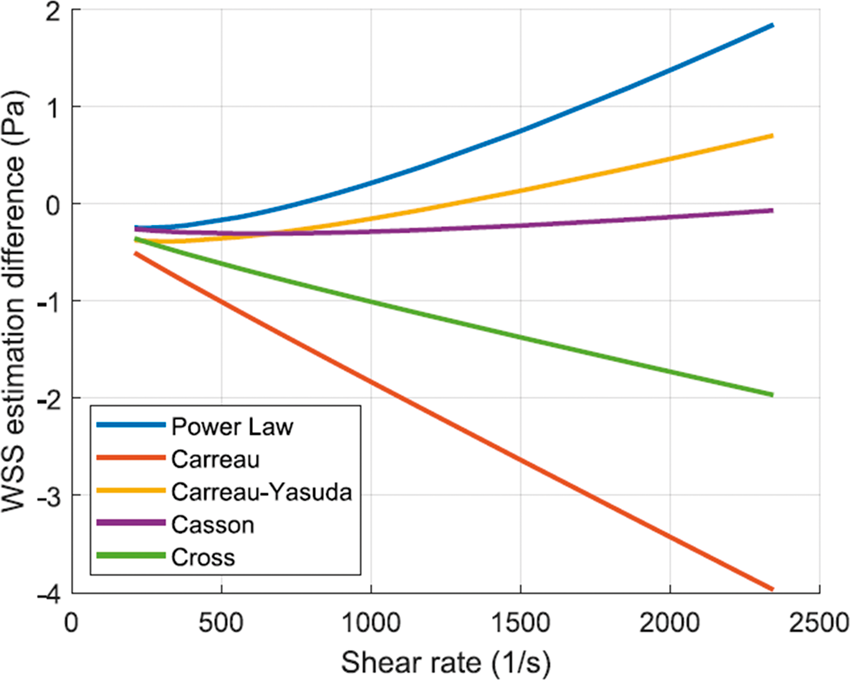
The range of WSS in IAs spans values that correspond to stagnant, creeping, and transitional flows. The order of magnitude of WSS, as depicted in Table 2 by in vivo methods, is between 10−2 and 10 Pa, depending on numerous factors including size, shape, morphology and location of IA. This range corresponds to shear rates in the order of 100 to 103 S−1. From such in vivo measurements, it is clear that blood flow inside the aneurysm manifests the non-Newtonian properties of blood. On the other hand, even in healthy intracranial arteries, the viscous behavior of blood shows considerable variation from the Newtonian simplification. This has been evidently proved from the meta-analysis calculations plotted in Figure 5. The wide range of values of shear rate and corresponding WSS in intracranial arteries clearly proves that the Newtonian assumption is inaccurate both qualitatively and quantitatively. This is quite evident from the hypothesis tests summarized in Table 1. All non-Newtonian models showed differences with the Newtonian model, with the most significant differences attributed to Carreau, Casson and Cross models. Figure 5 shows that Cross and Carreau models consistently produced WSS that were always higher than the Newtonian assumption. The remaining models produced both negative and positive differences showing varying response to different ranges of shear rate.
Is WSS enough to bridge hemodynamics and aneurysm pathology?
Choosing wall shear stress (WSS) as the discriminant variable to link blood flow (i.e. hemodynamics) to cellular response, hence physiological changes in blood vessels, was an arbitrary choice based on modest physical reasoning. Simply, early works chose WSS since it is the only parameter that depends on the flow (i.e. function of the near-wall shear rate) and shows multicomponent mechanical stress on the wall. Early researches in vascular biomechanics considered blood vessels as elastic or rigid tubes which respond mechanically to blood flow.218–221 The concept of mechanical response of arteries to blood flow propagated in literature and scientific community in the following decades.222,223 Following to the discovery of endothelial mechanosensors,224,225 it was found that endothelial cells respond to flow direction226,227 and local structures in disturbed flow.118,228 Flow with inherent vorticity field and vortex structures was found to affect endothelial cells differently than fully developed uniform flow. 118 When any fluid dynamicist thinks of flow structures and directionality, a number of flow field variables arise, excluding WSS. In the supplementary material to this article, we have explained why WSS is insufficient to justify the response of ECs to flow directionality and local structures in transitional and turbulent flows.
Due to the aforementioned shortcomings of WSS, there has been a recently growing trend to link IA growth and rupture events with other flow variables such as vorticity, coherent structures and turbulence identifying parameters. Valencia et al. 229 investigated the vortex ring structures and recirculation zones in IA models by inspecting the three-dimensional instantaneous velocity vector fields. They identified regimes of flow stability in the IA as function of the aneurysm tilt angle and showed the stabilizing effect of non-Newtonian blood viscosity. The local structures and energy cascade phenomena in IA were also investigated by means of DNS in several studies.173,191,230 These efforts are promising to link the biology and mechanotransducive responses of ECs to aneurysm pathology. In a pioneering attempt to probe such complex interaction, Varble et al. 231 investigated the vortex structure in 204 patient-specific IAs using high resolution DNS and patient-specific boundary condition waveforms. They evidently shown that IA rupture is significantly correlated with the near-wall vortex structures as expressed in terms of the surface vortex fraction. The outcome of their study highlights a new correlation between aneurysm rupture and higher levels of rotationality and vorticity in the IA flow. This finding is evidently supported by ECs mechanotransduction, where flow disturbance initiates pro-inflammatory EC responses,66,232 degenerative progress and EC misalignment. 118
Conclusion: towards next generation IA CFD models
The main challenges facing IA CFD currently have been summerized in the questions proposed in the aim and motivation section. Through the review and meta-analysis, it is evident that tackling such questions is impossible with the present IA CFD models which incorporates inappropriate simplifications, reliance on physically meaningless parameters, and loose computational representation of physiologic blood flow. Next generation IA CFD models must strive towards achiving the following objectives:
Capture more physics underlying IA hemodynamics Provide meaningful parameters to distinguish the pathobiology-hemodynamics interaction in IA genesis, growth and rupture Guide EC mechanotransduction studies by providing detailed quantitative and qualitative flow descriptions that can be used to trace biological factors influencing IA
In order to achieve such objectives, next generation IA CFD models must at least incorporate all of the following elements:
Patient-specific viscosity and blood flow waveforms Direct numerical simulation or robust large eddy simulation scheme Investigate the flow via one or more of the vector fields (such as vorticity) instead of scalar-tensor field (WSS or OSI)
In the end, modeling a complex biological system as IAs, which involves countless factors from the molecular and cellular levels to the complexities of the interacting blood flow, is never going to be a simple task. Indeed, in such a case some degree of simplification is always warranted, and of course justified. However, over-simplification in the modeling would eventually hinder us from capturing phenomena that could correlate the physics with the biology and ultimately enhace our understanding of the pathology of IAs.
Supplemental Material
Supplemental material for What does computational fluid dynamics tell us about intracranial aneurysms? A meta-analysis and critical review
Supplemental Material for What does computational fluid dynamics tell us about intracranial aneurysms? A meta-analysis and critical review by Khalid M Saqr, Sherif Rashad, Simon Tupin, Kuniyasu Niizuma, Tamer Hassan, Teiji Tominaga and Makoto Ohta in Journal of Cerebral Blood Flow and Metabolism
Footnotes
Funding
This work was supported in part by a grant-in-aid for Young Scientists (A) (#17H04745, to KN) from the Japan Society for the Promotion of Science.
Acknowledgement
The authors acknowledge the support through the Collaborative Research Grant from the Institute of Fluid Science, Tohoku University.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
KMS conceived and designed the study, contributed data, developed the mathematical models and wrote the paper. SR contributed data and wrote the paper. ST was responsible for the Software, statistics and visualization. KN, TH and TT critically revised the manuscript. MO was involved in funding acquisition, project management, and critical review of the paper. All authors approved the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


