Abstract
The glymphatic system plays a key role for clearance of waste solutes from the rodent brain. We recently found evidence of glymphatic circulation in the human brain when using magnetic resonance imaging (MRI) contrast agent as cerebrospinal fluid (CSF) tracer in conjunction with multiple MRI acquisitions (gMRI). The present study explored the hypothesis that reduced glymphatic clearance in entorhinal cortex (ERC) may be instrumental in idiopathic normal pressure hydrocephalus (iNPH) dementia. gMRI acquisitions were obtained over a 24–48 h time span in cognitively affected iNPH patients and non-cognitively affected patients with suspected CSF leaks. The CSF tracer enrichment was determined as changes in normalized MRI T1 signal units. The study included 30 patients with iNPH and 8 individuals with suspected CSF leaks (i.e. reference individuals). Compared to reference individuals, iNPH patients presented with higher medial temporal lobe atrophy score and Evan's index and inferior ERC thickness. We found delayed clearance of the intrathecal CSF tracer gadobutrol from CSF, the ERC and adjacent white matter, suggesting impaired glymphatic circulation. Reduced clearance and accumulation of toxic waste product such as amyloid-β may be a mechanism behind dementia in iNPH. Glymphatic MRI (gMRI) may become a tool for assessment of early dementia.
Keywords
Introduction
The entorhinal cortex (ERC) receives direct projections from the neocortex and constitutes the major convergent input to the hippocampus, thus playing a key role for memory function such as memories for locations and events. 1 The entorhinal-hippocampal circuit is affected at an early point of Alzheimer's disease (AD) development, which may be visualized with magnetic resonance imaging (MRI) as thinning of the ERC. 2 Profound degeneration of ERC layer II has been found early in AD. 3 It was recently demonstrated that ERC thinning was associated with increased postmortem neurofibrillary tangle burden and amyloid-β (Aβ) load, 4 and the authors suggested that ERC thickness may become a biomarker of early AD. The observations highlight the need for elucidating mechanisms behind ERC pathology and Aβ accumulation.
In early AD, one possible pathogenic factor behind ERC abnormality may be impaired clearance of waste solutes such as hyperphosphorylated tau (HPτ) and soluble Aβ, leading to deposition of Aβ plaques. In 2012, a brain-wide para-vascular system for transport and clearance of solutes and waste metabolites was described and denoted the glymphatic (glia-lymphatic) system. 5 The authors suggested that glymphatic failure may play a crucial role in the development of AD due to reduced glymphatic clearance of Aβ. 5
We recently described findings suggestive of a glymphatic system in man by utilizing intrathecal injection of an MRI contrast agent (gadobutrol), rendering this the first in-vivo CSF tracer study carried out in humans. 6 Gadobutrol has proper molecular size to enter the glymphatic system (604 Da), and clearance of gadobutrol should therefore resemble glymphatic clearance of other interstitial solutes transported through the same pathway, such as Aβ. In this previous study, repetitive MRI acquisitions following CSF tracer administration (glymphatic MRI, gMRI) gave evidence of impaired clearance of tracer from CSF and brain parenchyma in patients with idiopathic normal hydrocephalus (iNPH). INPH is one subtype of dementia that may be improved by diversion of cerebrospinal fluid (CSF) even though the clinical effect declines over time. 7 While the mechanisms behind dementia in iNPH remain unclear, knowledge about this condition may also help understand AD since an increasing body of evidence suggests an association between iNPH dementia and AD. 8 For example, cortical biopsies from iNPH patients show accumulation of HPτ and Aβ in proportion of patients, which is associated with an unfavorable outcome of CSF diversion surgery.9–13
Studies of the glymphatic system in rodents,5,14 as well as humans, 6 suggest that vascular pulsations mediated via CSF are the main force behind glymphatic flow. It has further been provided evidence that restricted artery pulsations may reduce glymphatic clearance of Aβ from the brain. 5 The proximity between ERC in the medial temporal lobe and large artery trunks residing at the base of the brain, particularly the circle of Willis, posterior cerebral- and superior cerebellar arteries, renders for a large impact from pulsations on ERC glymphatic flow. Based on our recent study, 6 we hypothesized that impaired glymphatic circulation within the ERC of iNPH patients may be one important mechanism behind dementia. The present study tested the hypothesis that glymphatic clearance, assessed by utilization of in-vivo CSF tracer, is delayed from the ERC and ERC subcortical white matter, in iNPH versus reference (REF) individuals, and that impaired clearance is related to anatomical biomarkers of neurodegeneration.
Materials and methods
Study design and ethical approvals
This study was approved by The Regional Committee for Medical and Health Research Ethics (REK) of Health Region South-East, Norway (2015/96), the Institutional Review Board of Oslo university hospital (2015/1868), and the National Medicines Agency (15/04932-7). The conduct of the study was governed by ethical standards according to the Helsinki Declaration of 1975 (and as revised in 1983). Patients were included after written and oral informed consent.
The study design was prospective and observational, and included consecutive iNPH patients and patients with suspected CSF leaks, and as from here referred to as reference (REF) patients. The iNPH and REF patients were imaged with MRI before (Pre), and at regular time points after intrathecal injection of the MRI contrast agent gadobutrol (gMRI). Exclusion criteria were: history of hypersensitivity reactions to contrast agents, history of severe allergy reactions in general, evidence of renal dysfunction, pregnant or breastfeeding women, and age <18 or > 80 years.
Participants
The iNPH patients were referred to the Department of Neurosurgery, Oslo University Hospital - Rikshospitalet, Oslo, Norway based on clinical symptoms and findings indicative of iNPH, combined with imaging findings of ventriculomegaly. Clinical assessment, including assessment of cognitive impairment, was performed by the referring neurological departments. Within the department of neurosurgery, clinical severity was graded based on a previously described NPH grading scale (15 possible scores ranging from 3 to 15), which assesses the combined severity of gait disturbance (scores ranging from 1 to 5), urinary incontinence ((scores ranging from 1 to 5) and dementia (scores ranging from 1 to 5).7,15
The REF patients were in parallel with iNPH patients recruited prospectively and consecutively from referrals to clinical work-up of tentative idiopathic intracranial hypotension due to suspected CSF leakage. MRI with intrathecal gadobutrol was done with the primary indication to define site of CSF leakage, but also included a complete gMRI time series.
Determination of ERC thickness, medial temporal atrophy-score and Evan's index
ERC thickness
Accurate prediction of ERC borders from in-vivo MRI constitutes a major challenge when whole ERC volumetric measurements are applied, both with concern to delineation against the parasubiculum (upper medial), perirhinal cortex (lower lateral), apex of intralimbic gyrus (posterior) and white matter of limen insula (anterior). 16 However, ERC thickness can be measured more readily and robustly, and ERC thickness has previously been among regions to provide the best discrimination between cognitively normal subjects and patients with mild cognitive impairment or AD. 17 ERC thickness was measured in coronally reconstructed T1 volume acquisitions with 1 mm slice thickness at level of the hippocampal sulcus and measured from the ERC surface to the grey/white matter interface, and midway between the tentative location of parasubiculum and perirhinal cortex. Supplementary Figure 1 illustrates the determination of ERC thickness.
Medial temporal atrophy
The degree of medial temporal atrophy (MTA) was categorized using the Scheltens score, 18 which is based on a visual rating of the width of the choroid fissure, the width of the temporal horn, and the height of the hippocampal formation: Score 0 (no atrophy), score 1 (only widening of choroid fissure), score 2 (also widening of temporal horn of lateral ventricle), score 3 (moderate loss of hippocampal volume, decrease in height), and score 4 (severe volume loss of hippocampus).
Evans's index
To objectively assess ventricular size, the EI 19 was measured in axially reconstructed T1 scans (1 mm slice thickness) by dividing maximal width of the frontal horns with the largest internal diameter of the cranium in the same slice. EI ≥ 0.3 is typically considered pathologically increased in adults 20 and can be considered to reflect underlying neurological disease in an individual. 21
gMRI
The MRI protocol followed a strict and standardized protocol, which has been described recently in detail. 6 Sagittal 3D T1-weighted gradient echo volume scans were obtained using a 3 Tesla Philips Ingenia MRI scanner (Philips Medical systems, Best, The Netherlands) with equal imaging protocol settings before and at multiple time points up to 48 h after intrathecal injection of gadobutrol, for details see Ringstad et al. 6
Using X-ray guided lumbar puncture, an interventional neuroradiologist injected 0.5 ml of 1.0 mmol/ml gadobutrol (Gadovist™, Bayer Pharma AG, Berlin, Germany) to the intrathecal space, as previously described. 6
While the MRI exams, for practical reasons, could not be obtained at identical time points for every study subject, all exams were categorized into the following time intervals for this present study: Before contrast (Pre), 40–60 min, 2–4 h, 6–9 h, 24 h, and 48 h.
For each time point, a neuroradiologist (G.R.) with 11 years' experience in neuroradiology placed circular regions of interest (ROIs) inside ERC and ERC subcortical white matter and CSF nearby ERC directly on 1 mm thick, coronally reconstructed T1-weighted images at level of the hippocampal sulcus. All measurements were done directly in the hospital PACS (Sectra IDS7®, Sectra, Sweden), where each ROI provides the mean T1 signal unit from the image greyscale and were then normalized against a reference ROI by dividing any measured T1 signal unit from CSF or brain parenchyma with the value of the reference ROI (the ratio from hereby referred to as normalized T1 signal unit) to correct for any baseline changes of image grey scale due to image scaling. The reference ROI was placed within the posterior part of the superior sagittal sinus in axially reconstructed images from the same T1 volume scan. Previous observations indicate no measureable enhancement of contrast agent at MRI in this location after intrathecal injection of gadobutrol in the current amount. 6 All ROIs were manually fitted to local anatomy to avoid partial volume effects from neighboring tissue or CSF. For the CSF, a single region of interest was placed within the subarachnoid space nearby ERC. For all pairwise locations, bilateral ROIs were determined and the average calculated.
Statistical analysis
Statistical analyses were performed using the SPSS software version 22 (IBM Corporation, Armonk, NY). Differences between categorical data were determined using Pearson Chi-square test, and differences between continuous data were determined using linear mixed models with a random intercept, and independent samples t-tests. Correlations were determined by Pearson correlation coefficient. Statistical significance was accepted at the .05 level (two-tailed).
Results
Patient cohorts
The study cohort consists of 30 iNPH patients and 8 reference (REF) patients were included consecutively during the period October 2015 – February 2017. The 8 REF patients had been referred for work-up of possible CSF leakage; only 2/8 patients had verified CSF leakage. No serious adverse events related to the intrathecal administration of gadobutrol were registered.
Demographic, clinical and imaging data of the iNPH and REF cohorts.
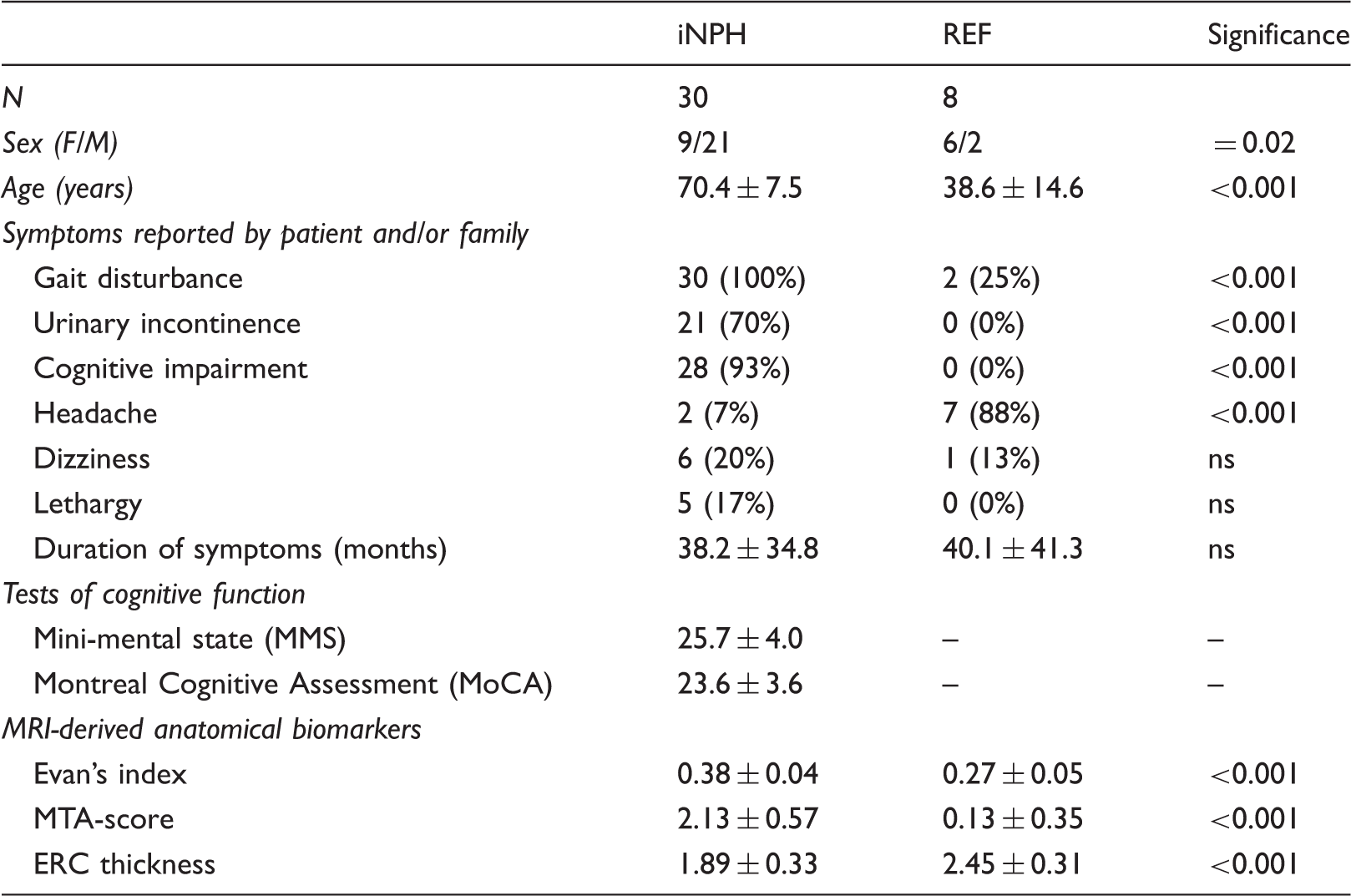
Note: Categorical data presented as numbers; continuous data presented as mean ± standard deviation. Significant differences between groups were determined by Pearson Chi-square test for categorical data and by independent samples t-test for continuous data.
iNPH: idiopathic normal pressure hydrocephalus; REF: reference patients; MTA-score: medial temporal lobe atrophy score; ERC: entorhinal cortex.
While none of the REF individuals had cognitive deficits, 28/30 of the iNPH had cognitive deficits, reported by the patient and/or family. The cognitive score of the NPH score was available in all individuals, while MMS and/or MoCA had been obtained in 20/30 iNPH patients by referring neurologists. There was a highly significant positive correlation between the NPH cognitive score and the MMS (Pearson correlation 0.74, P = 0.001).
MRI-derived anatomical biomarkers
As compared with the REF individuals, iNPH patients presented with lower ERC thickness, as well as higher MTA-score and EI (Table 1).
Regarding ERC thickness and cognitive function, we found a significant positive correlation between ERC thickness and MMS (Pearson correlation 0.52, P = 0.04), but not for NPH cognitive score (P = 0.07).
We found no association between ERC thickness and age in the iNPH or REF cohorts (P > 0.3; data not shown). In comparison, there was a significant positive correlation between age and MTA-score in the iNPH cohort (age range 46–80 years; Pearson correlation 0.43, P = 0.02), while not in the REF cohort (age range 22–59 years; P = 0.23).
The Evan's index correlated positively with the MTA-score (Pearson correlation 0.42, P = 0.02) and with the duration of disease (Pearson correlation 0.39, P = 0.04).
Delayed clearance of CSF tracer from the ERC and subcortical white matter of iNPH patients
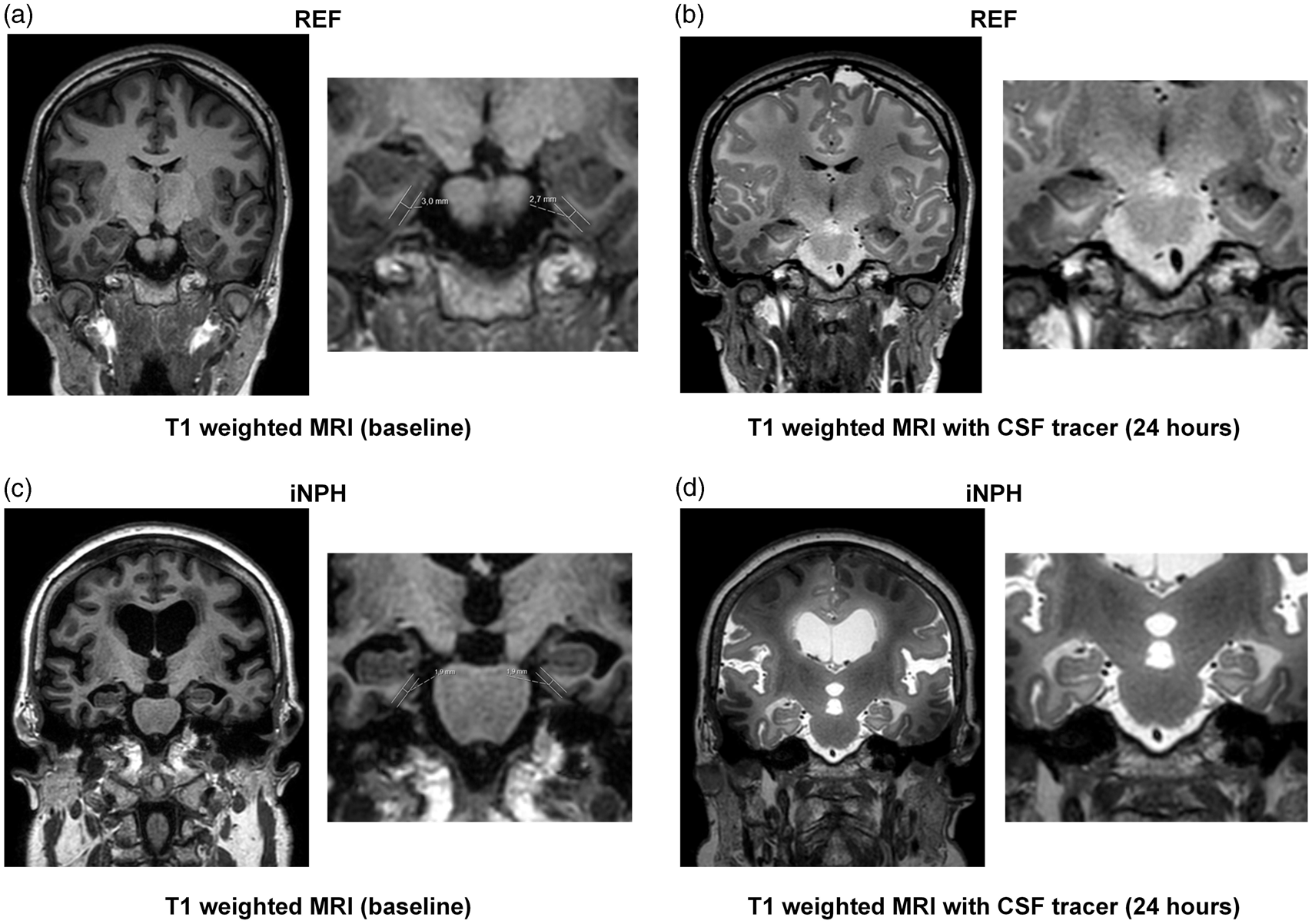
Figure 1(a) and (c) (pre contrast) and (b) and (d) (post contrast) illustrates the effect of MR image enhancement by the CSF tracer gadobutrol in one REF (Figure 1(a) and (b)) and in one iNPH (Figure 1(c) and (d)) individual, respectively.
In an REF individual (a, b) and an iNPH patient (c, d) T1-weighted MRI in coronal sections at ERC level at baseline (before contrast agent administration) (a and c) and after 24 h (b and d) illustrate time-dependent enrichment of CSF by increased T1 image intensity at the image greyscale. Ventricular reflux of contrast agent is a typical feature of iNPH (d) as described previously.
6
On unenhanced images, the normalized T1 signal unit was lower within the ERC (Figure 2(a)), and ERC subcortical white matter in iNPH (Figure 2(b)). The lower signal unit ratio within the ERC grey and white matter of iNPH patients indicates higher water content within the brain tissue. Figure 2(c) illustrates manual fitting of ROIs for assessment of signal units within the ERC and ERC subcortical white matter.
The normalized T1 signal units before intrathecal administration of gadobutrol in REF and iNPH patients within (a) ERC, and (b) subcortical white matter of ERC. Differences between REF and iNPH individuals are indicated for (a) ERC and (b) ERC subcortical white matter, and may be attributed to increased brain water content in iNPH. In (c) is shown how ROIs for measurement of MRI T1 signal units within the ERC and subcortical white matter of ERC were manually fitted in an REF subject. All T1 signal unit measurements were normalized against a reference value retrieved from the superior sagittal sinus, which does not enhance with intrathecal contrast agent.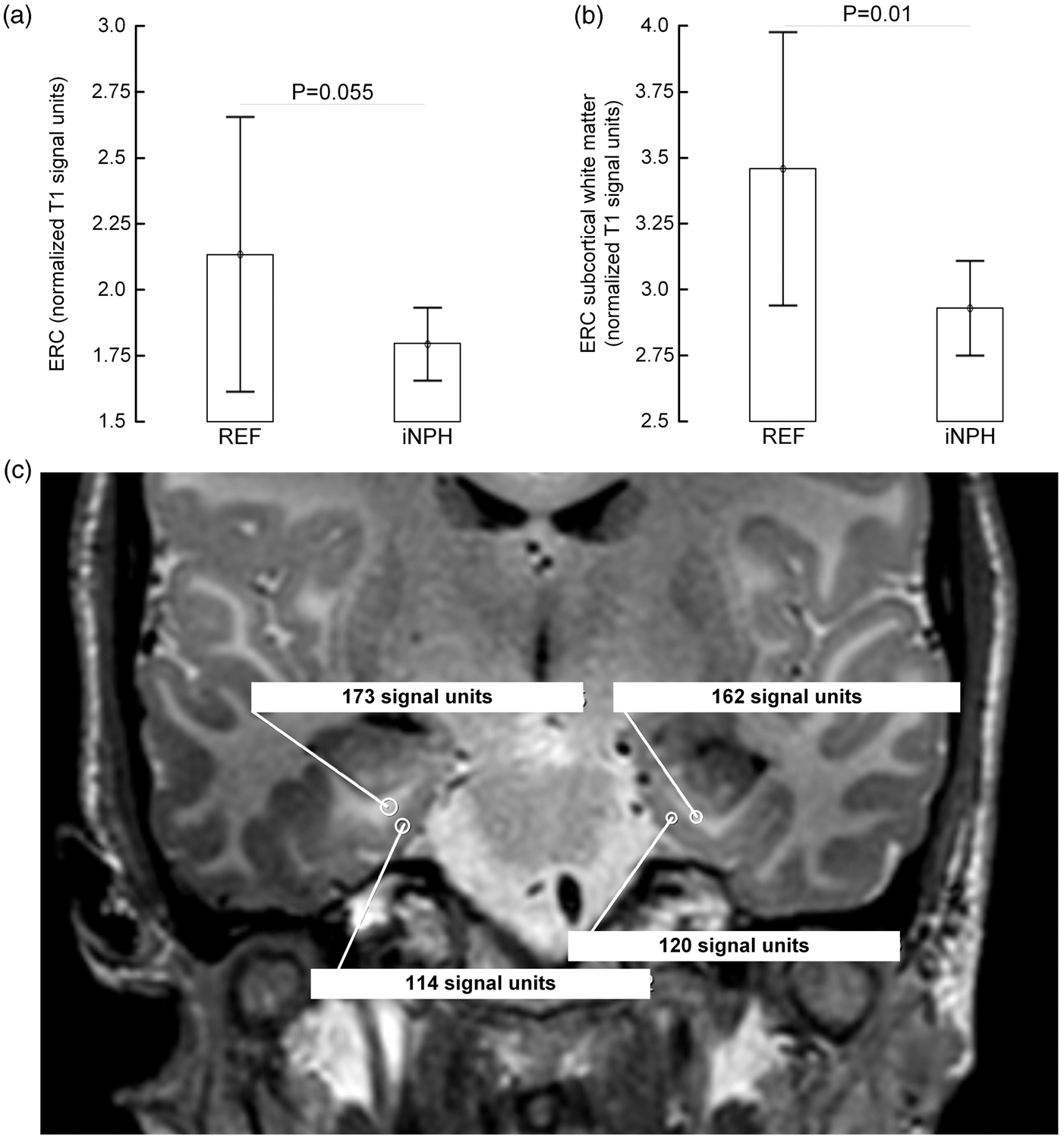
Figure 3 presents the time-dependent percentage increase in normalized T1 signal units within the CSF nearby ERC (Figure 3(a)), ERC (Figure 3(b)) and ERC subcortical white matter (Figure 3(c)) in REF and iNPH individuals. In iNPH, presence of tracer was significantly higher in CSF after 6–9 and 24 h, indicating reduced tracer clearance, as well as after 24 h in the ERC and subcortical white matter. The increased normalized T1 signal units within all three compartments in iNPH patients at 24 h may be interpreted as reduced ability of the system to clear the tracer from the respective locations and the intracranial compartment in general. The percentage change in normalized T1 signal units from baseline at different time points within CSF, ERC, and ERC subcortical white matter is presented in Table 2.
Trend plots of time-dependent CSF tracer (gadobutrol) uptake in REF (continuous lines) and iNPH (dotted lines) patients revealed as percentage change in normalized T1 signal unit from baseline at three locations: (a) CSF nearby ERC, (b) ERC and (c) subcortical white matter of ERC. Error bars refer to 95% CI. Differences between groups at individual time points were determined by mixed model analysis (*p < 0.05, **p < 0.01, ***p < 0.001). In iNPH cases, the CSF tracer enhancement was significantly higher at all locations at 24 h, indicative of delayed tracer clearance. Peak enhancement in ERC (b) precedes peak enhancement in ERC subcortical white matter (c), demonstrating a centripetal pattern of brain enrichment by CSF tracer. Percentage change in normalized T1 signal units from baseline of CSF tracer enrichment at different time points. Note: Significant differences between iNPH and REF groups were determined by independent samples t-test. CSF: cerebrospinal fluid; iNPH: idiopathic normal pressure hydrocephalus; REF: reference patients; ERC: entorhinal cortex. P < 0.05, P < 0.001, P = 0.008.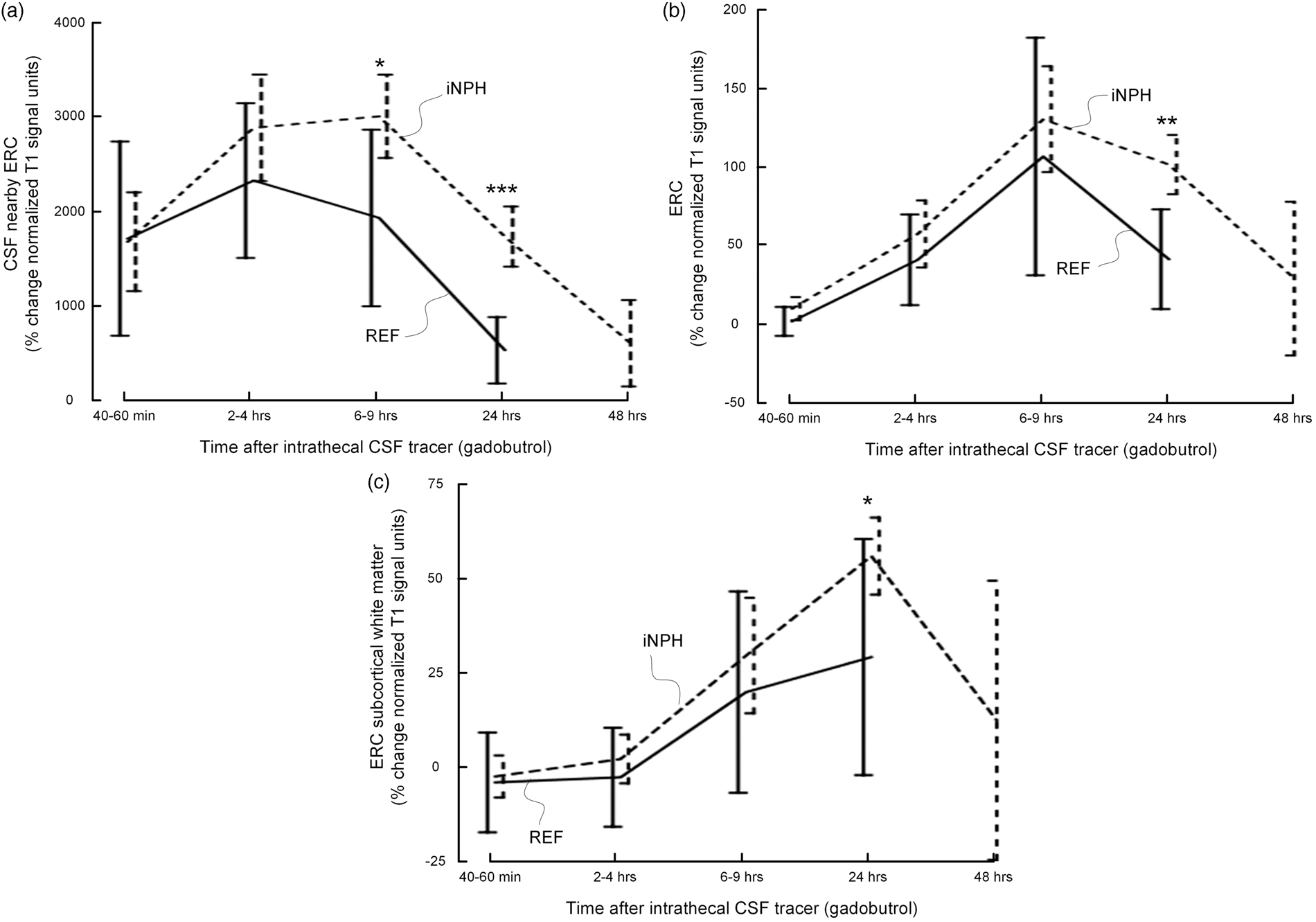
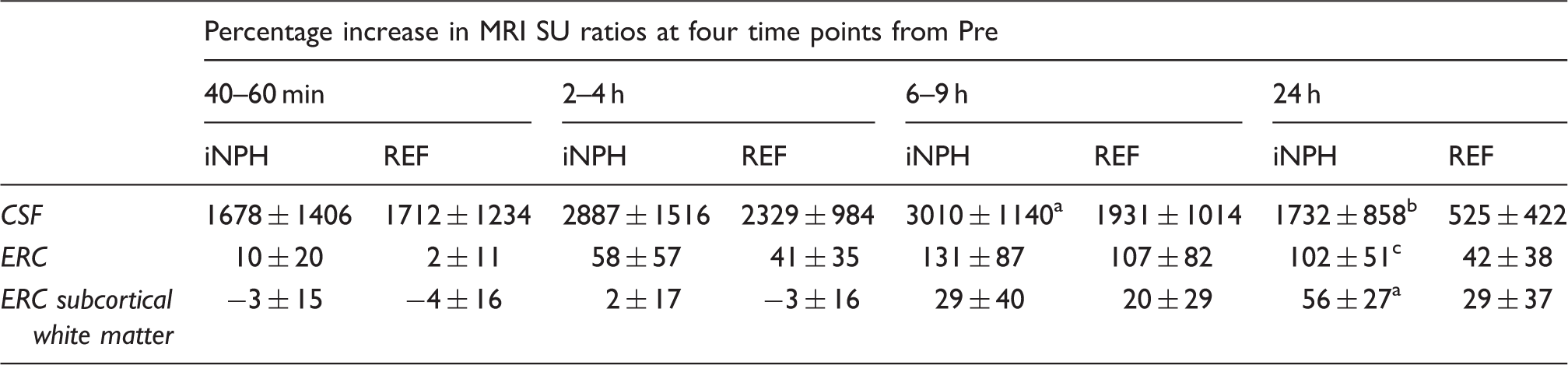
The enrichment of CSF tracer within the ERC and ERC subcortical white matter was dependent on the availability of tracer within nearby CSF. Hence, there was a strongly positive correlation between maximum tracer enrichment in CSF (i.e. maximum percentage increase in normalized T1 signal units) and ERC (Figure 4(a)), and further a highly significant positive correlation between maximum tracer enrichment in CSF and subcortical white matter of ERC (Figure 4(b)). Finally, tracer enrichment of ERC and subcortical white matter was strongly associated (Figure 4(c)).
CSF tracer uptake within ERC is a function of tracer enhancement within CSF nearby ERC. There was a highly significant positive correlation between CSF tracer availability within the CSF space nearby ERC and (a) ERC and (b) subcortical white matter of ERC, as well as (c) between ERC and subcortical white matter of ERC. For each plot is presented the fit line and the Pearson correlation coefficient (R) with P-value.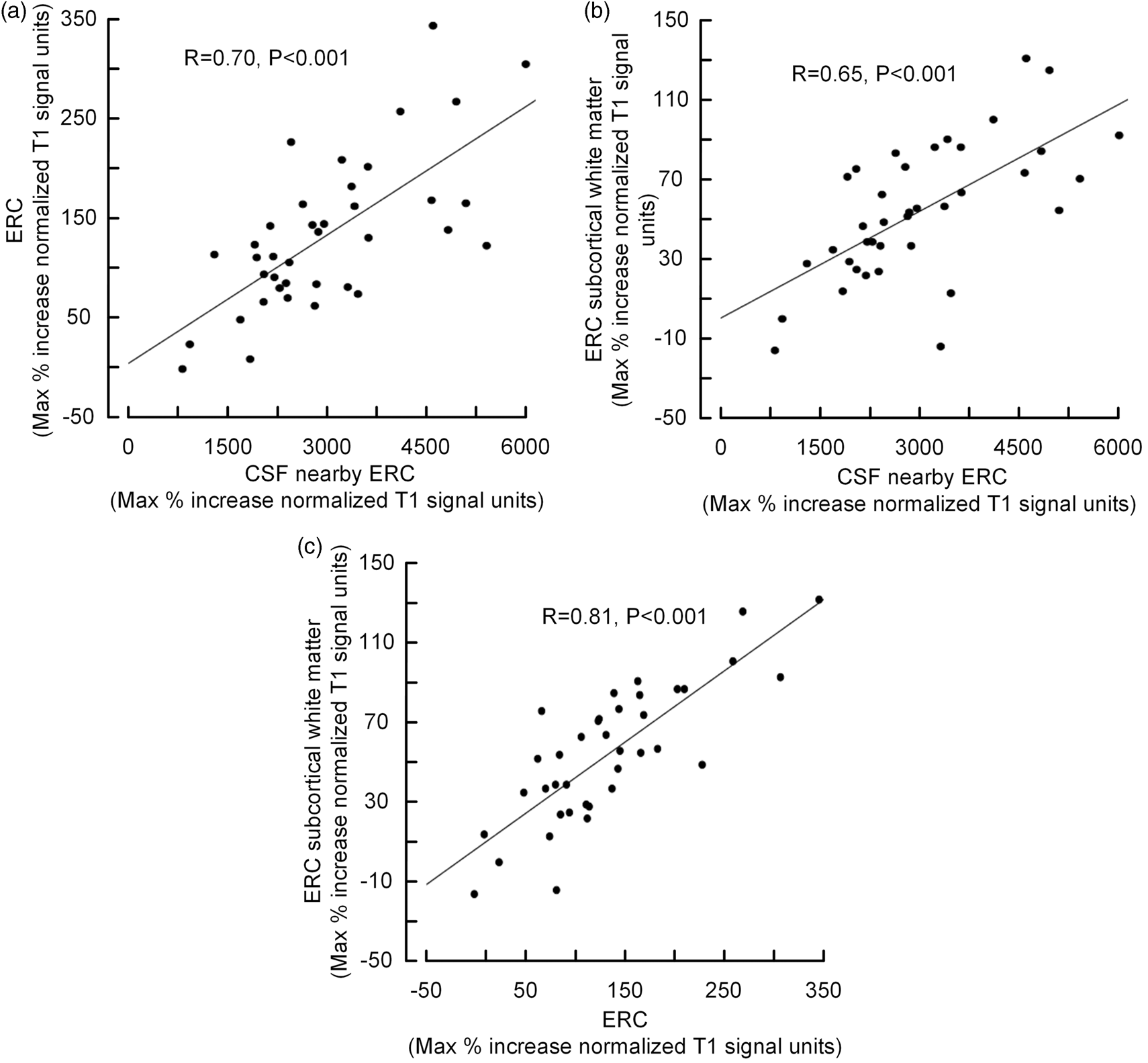
Comparing tracer enrichment between the 2/8 REF individuals with verified CSF leakage and the 6/8 REF individuals without showed lower tracer enrichment within ERC (max tracer enrichment 78 ± 5% versus 119 ± 83%) and ERC subcortical white matter (max tracer enrichment −1 ± 19% versus 46 ± 32%). The results could not be attributed to reduced availability of tracer within the CSF since tracer enrichment within the CSF nearby ERC was higher in the two individuals compared to the other six (max tracer enrichment 3462 ± 112% versus 2101 ± 855%).
MTA-score associates with clearance of ERC, ERC thickness, and Evan's index
Considering all the 38 patients, the combined CSF tracer enrichment after 24 hours in ERC (Figure 5(a)) and ERC subcortical white matter (Figure 5(b)), indicating uncleared tracer, correlated with MTA-score. Further, MTA-score was significantly negatively correlated with ERC thickness (i.e. higher MTA-score was associated with thinning of the ERC; Figure 5(c)). In addition, MTA-score was significantly positively associated with Evan's index (Figure 5(d)).
Association between MTA-score and percentage increase of normalized T1 signal units (i.e. CSF tracer enrichment) at 24 h as compared to before contrast agent administration within (a) ERC, and within (b) subcortical white matter of ERC. Further, the associations between MTA-score and (c) ERC thickness and (d) Evans index are indicated. For each plot is presented the fit line and the Pearson correlation coefficient (R) with significance P-value.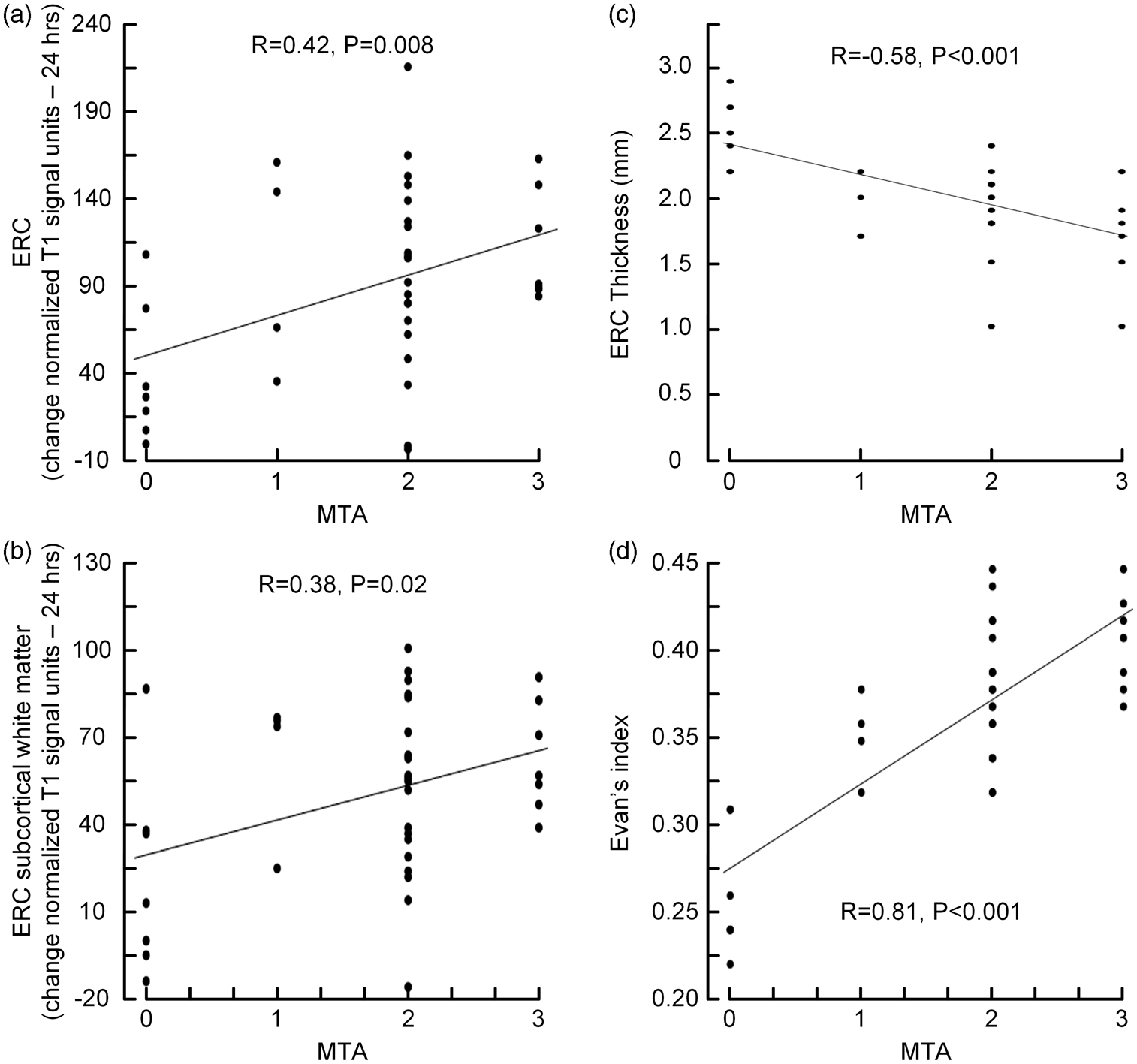
Association between cognitive measures and clearance of CSF tracer from the ERC and subcortical white matter of iNPH patients
There were no significant association between MMS or NPH cognitive sub-scores and clearance of CSF tracer from CSF, ERC or ERC subcortical white matter, neither when clearance was measured as maximum percentage increase in normalized signal units or as percentage increase in normalized signal units after 24 h (data not shown).
Discussion
The major observation of this present study was delayed clearance of CSF tracer from the ERC, subcortical white matter of ERC and from CSF nearby the ERC in individuals with iNPH. ERC thickness was smaller and MTA score was higher in iNPH patients compared to REF individuals. Further, the MTA score correlated significantly with the signs of reduced CSF tracer clearance after 24 h within ERC and subcortical white matter of ERC.
Patient cohorts and iNPH dementia
INPH constitutes one subtype of dementia. 22 Its prevalence increases markedly with advancing age 23 and has been estimated to 5.9% in people aged >80 years. 24 Several authors have pointed at a close association between iNPH and AD.8,25 In line with this view, a significant proportion of iNPH patients present with HPτ and amyloid-β deposition within the brain.9–13,26,27
While none of the REF individuals reported cognitive impairment, cognitive impairment was reported by the patient or their family in 28 of 30 iNPH patients. In these patients, the cognitive decline was moderate, as revealed by the cognitive tests (Table 1). INPH patients with severe dementia will normally not be candidates for CSF diversion surgery (i.e. shunt surgery) and were therefore not part of this study. The present iNPH subjects had a shunt response rate in line with our previous reports.7,15 The high proportion of shunt responders in this study implies that the study cohort may be considered representative for “true,” or definite iNPH. 28
The MRI biomarkers MTA-score, ERC thickness and Evan's index were significantly worse in iNPH than REF cases, providing an anatomical basis for cognitive decline in iNPH. Others have previously found evidence that cognitive decline as well as early dementia development is accompanied with higher MTA-score 18 and thinning or volume loss of the ERC.2,4,29–32 The reason for giving attention to these particular anatomical regions is the well-established knowledge that the ERC-hippocampus circuit plays a fundamental role in learning and orientation.1,33–35
The present differences in MRI biomarkers between iNPH and REF individuals may probably to some extent be attributed to ageing. It has been reported that the ERC thickness declines with age from the age of 44 years. 36 We found reduced MTA-score with increasing age within the iNPH cohort. It seems, however, unlikely that differences in these MRI biomarkers between iNPH and REF can be attributed solely to age, and there could for the entire cohort not be revealed any significant correlation between age and ERC thickness.
The ERC thickness we measured in REF individuals is at the low end of what has been reported as normal values throughout life span in a large cohort of healthy individuals. 36 While that study utilized segmentation software (FreeSurfer) to provide average cortical thickness from the entire ERC, we measured ERC thickness from direct visual analysis and manual measurement at one standardized level. It is therefore likely that these differences may be referred to different measurement techniques.
Assessment of glymphatic clearance with MRI
We recently described gMRI as an imaging modality to assess glymphatic circulation in humans.6,37 The present data extend our previous results by showing delayed clearance of CSF tracer from ERC and ERC subcortical white matter and from CSF nearby ERC.
The glymphatic system refers to a paravascular pathway within the brain for transport of fluids and solutes, and for removal of waste products from the brain.38,39 Its function depends on sleep, 40 and becomes impaired with increasing age. 41 It has been suggested that glymphatic failure has a role in dementia development. 42 However, the vast current knowledge is pre-clinical and based on studies in animals, primarily rodents. Several aspects of the concept of glymphatic circulation remain controversial. For example, contrary to the original hypothesis, 5 a modeling study concluded that transport of solutes within the brain interstitial space occurs by diffusion 43 though not addressing mechanisms behind solute transport paravascular along vessels. Further, while the glymphatic concept relies on antegrade transport along arteries, others have provided evidence that fluid transport occurs retrograde along cerebral arteries. 44
At present, there is a knowledge gap regarding the role of glymphatic circulation within the human brain 38 since in vivo CSF tracer studies have largely been limited to animals.
In the current study, manually fitted ROIs of suitable size were placed in a part of ERC where the cortex can be assessed in a plane that is almost parallel to the orientation of the image slice and thus should any partial averaging from CSF be minimized. These measurements demonstrate that glymphatic enhancement in cortex precedes enhancement in subcortical white matter and with peak enhancement at 6–9 and 24 h, respectively (Figure 3). The additional observation of strong association between enhancement in CSF and adjacent parenchyma (Figure 4(a) and (b)), and that CSF enhancement precedes parenchymal enhancement (Figure 3), clearly suggest glymphatic enhancement derives from CSF tracer at the surface of the brain, and then propagates through the parenchyma in a centripetal direction. These in vivo observations directly contradict previous electron microscopy studies of fixed human and mouse brain tissue,45,46 which have laid ground for a prevailing concept that cortical perivascular spaces do not exist neither in human nor rodent brain. 47 The cortical enrichment of a CSF tracer in humans is well in line with a recent tracer study of rats, where it was concluded that the CSF compartment forms a continuous and extensive network that surrounds and penetrates the brain to allow for exchange between interstitial fluid and CSF. 48 Further, the arterial pulsations seem to be the main driving force for CSF tracer anterograde along arteries. 14
We used an MRI contrast agent gadobutrol that is well suited as a CSF tracer due to its low molecular weight (605 DA) and its hydrophilic, non-ionic properties causing it to distribute freely in water. 49 We administered only 0.5 ml of gadobutrol into the spinal subarachnoid space, which would not impact intracranial pressure or CSF flow.
The fact that the ERC is located close to large vessels, in particular the posterior cerebral artery (Suppl. Figure 3), may be of particular significance for glymphatic clearance in the ERC. In a previous study, we found that CSF tracer distributed freely within subarachnoid sulci and cisternal spaces that were traversed by the major cerebral leptomeningeal arteries (anterior, middle and posterior). 6 Our interpretation of the MRI findings is that the CSF tracer gadobutrol propagates along the arteries in antegrade direction, which may support the experimental evidence that arterial pulsatility drives paravascular flow.14,50 Interestingly, vascular pathology 51 and co-morbidity52,53 are frequent in iNPH. Moreover, several studies have suggested that cardiovascular disease risk factors can be linked to sporadic AD.54–57 As the connection between cardiovascular disease and dementia is still not fully established, it can from the present observations be hypothesized an impact from vascular disease and restricted arterial pulsations on glymphatic function, and thereby compromised clearance of interstitial macromolecules such as Aβ.
A significant observation of the study was that after 24 h, CSF tracer enrichment was significantly higher in ERC as well as adjacent subcortical white matter, and CSF in iNPH patients. As the destiny of a CSF tracer is to be cleared from the intracranial compartment with a certain rate, the observations can hardly be explained in another way than that clearance of CSF tracer was delayed in iNPH. This was a robust and consistent observation within all three ROIs studied, and in line with our previous observations in other human brain regions. 6
It should be emphasized that CSF tracer enrichment in ERC subcortical white matter peaked as late as at 24 h, i.e. after sleep. In anesthetized rodents (mice and rats), cortical glymphatic CSF tracer enhancement typically peaked about 1–2 h. 58 The observed penetration of tracer substance to the subcortical white matter of ERC in humans extends knowledge from studies on mice using two-photon electron microscopy, where imaging was limited to 240 µm below the cortical surface 5 and adds to an in vivo MRI study of rats, where MRI contrast agent distributed widely, however, not in the entire brain parenchyma. 58 The ability of a tracer substance to propagate in brain parenchyma over distances that by far exceeds the distance over which diffusion within synapses and cells occur, favors the existence of so-called “volume transmission” or “paracrine transmission” in the brain. Such information exchange between brain cells, other than what is mediated by synapses and the neuronal circuitry, has previously been limitedly characterized and remained an elusive concept, 59 but has more recently been integrated conceptually with the CSF compartment and suggested important for learning and memory. 60 In light of the study by Xie et al. 40 who demonstrated that natural sleep or anesthesia was associated with a 60% increase of the interstitial space, and which increased glymphatic clearance of Aβ two-fold, our observation of over-night peak enhancement at the farthest distance from the surface (ERC subcortical white matter) strongly suggests an important role of sleep for glymphatic function.
Impaired CSF clearance and the pathophysiology of iNPH dementia
How can delayed clearance of CSF tracer be related to the pathophysiology of iNPH dementia? The cause(s) of dementia in iNPH has been poorly understood, and attributed to a number of factors such as ventriculomegaly associated stretching and compression of neuronal pathways involved in cognition, interstitial edema, impaired cerebral blood flow, intracranial pressure changes and accumulation of waste such as Aβ and HPτ. 61 The neurodegenerative aspect of the disease is illustrated by the fact that the beneficial effect of shunt surgery on cognitive function declines over years. 7
In particular, the role of impaired clearance of waste solutes such as soluble Aβ leading to Aβ plaques has emerged as an important mechanism behind dementia. 42 The present imaging data demonstrate for the first time in patients with cognitive deficits delayed clearance of an in vivo CSF tracer from ERC and subcortical white matter of ERC with its trajectories towards the hippocampus. A model for a possible pathogenic pathway linking AQP4 loss and restricted pulsations to ERC neurodegeneration is provided in Supplementary Figure 2(b). Further, evidence suggesting affection of this particular anatomical region in iNPH was indicated by reduced ERC thickness as well as increased MTA-score. With regard to cognitive function level, measures of CSF tracer clearance were not correlated with MMS or NPH dementia sub-score. This may not be surprising, as reduced clearance function is expected to be deteriorated already in the pre-symptomatic phase of dementia. Ability of solute clearance from the parenchyma may therefore be completely decoupled from cognitive function level.
The underlying molecular mechanisms need to be further elucidated. We recently reported that in iNPH patients, the expression of the water channel aquaporin-4 (AQP4) and its anchor molecule dystrophin-71 (Dp-71) were reduced at astrocytic perivascular endfeet. 62 Hence, in iNPH, reduced perivascular AQP4 may hamper transport of fluid and solutes along micro vessels. Postmortem assessment of frontal cortex from healthy humans and AD individuals gave evidence that loss of perivascular AQP4 localization was accompanied with increased Aβ burden. 63 In rodents, evidence has been provided that glymphatic circulation depends on proper AQP4 function. 5 In AQP4 knock-out mice, glymphatic function and clearance of metabolites from the extracellular space are reduced. 5 Animal experiments suggest that AQP4 failure may be accompanied with reduced clearance of Aβ. 5 Further, recently it was reported that variations in the AQP4 gene were accompanied with different rates of cognitive decline in AD. 64 However, Smith et al. 65 recently reported that glymphatic transport was independent of the AQP4 status. Therefore, the role of AQP4 for glymphatic function needs to be further explored.
In this study, there were signs of increased water content of ERC and subcortical white matter in iNPH, by means of lower normalized T1 signal units in these regions. Water content is in general a main contributor to reduced T1 signal intensity, and signs of increased water content have previously also been demonstrated in other brain regions of iNPH subjects. 6
Limitations
One limitation with regard to our patient cohorts is the significant age differences between iNPH and of REF individuals. Experimental data provide evidence that glymphatic function becomes impaired with increasing age. 41 Therefore, we cannot exclude that some of the differences in tracer clearance observed between iNPH and REF individuals may be attributed to age. Future studies should preferably include age-matched controls.
It should also be noted that the REF individuals may not in all respects be considered as healthy individuals with normal CSF flow pattern. A CSF leakage was identified in 2/8 individuals. However, in the 6/8 individuals, no apparent cause of the complaints was found. Therefore, we consider our REF individuals as a group to be close to healthy. In these two, parenchymal tracer enrichment was inferior as compared to the other six REF individuals (in whom no apparent cause of the complaints was found). This observation underlines the important role of unrestricted CSF pulsations for parenchymal entry of tracer. Leakage of CSF and intracranial hypotension might impair the efficacy of arterial pulsations as removal of CSF alters the intracranial pulsatility, measured by intracranial pressure monitoring. In line with this reasoning, CSF drainage by a shunt markedly reduced the intracranial pulsatility. 66 It should be noted, however, that tracer levels within the CSF were not lower in the two individuals with verified leakage. Further, clearance of tracer was comparable in the patients with CSF leakage, and their contributed data to the REF cohort as whole did not change the study conclusions.
Another potential limitation is that MR image analysis of CSF tracer enrichment at different time points was carried out manually from the grey and white matter of the ERC and the nearby CSF space. The ROI had to be fitted to adjust for local anatomy to avoid partial volume averaging effects. Given the small size of the ERC, this represented a challenge, as also was the case for ERC thickness measurements. An improved method would be to perfectly align and co-register images from different time points, incorporating segmentation techniques to differentiate brain sub-regions, and CSF from parenchyma. We are currently elaborating such post-processing algorithms.
Finally, MR images have resolution in the order of mm, while paravascular and interstitial spaces are of sizes in the order of µm and nm. MRI can therefore not discriminate tracer enhancement in paravascular and interstitial spaces. While detection of tracer beneath cortical structures would strongly suggest bulk flow rather than strict diffusion, the study does not reveal whether bulk flow occurs along low-resistance pathways, such as along vessels or axons, or convectively through the interstitial space. Use of the term “glymphatic” thus leans towards previous observations made at a more basic level.5,48,67
Conclusions
Taken together, the present data provide evidence of delayed clearance of CSF tracer substance from the ERC and ERC subcortical white matter in iNPH. We suggest that the present data support the hypothesis that impaired glymphatic function in these pivotal structures for cognitive function hampers removal of toxic waste solutes, which may lead to neurodegeneration, and eventually dementia in iNPH.
Future prospects
The observations made in this study carry potentially profound implications for understanding, diagnosing and treating dementia. If reduced glymphatic function has a major role in neurodegeneration and dementia, as indicated in previous animal studies, and that glymphatic function can be assessed by means of long-term in vivo imaging of CSF tracer metabolism, as done here with gMRI, gMRI could serve as a pre-clinical test to measure dementia susceptibility, not at least AD risk, before pathologic Aβ deposition and irreversible neuronal loss occurs. While this study explored transit of CSF tracer in the ERC due to the previously suggested pivotal role of ERC in dementia, a brain-wide approach could well be applied in adjunction with the use of sophisticated segmentation tools.
Further, previous knowledge about intrathecal drug administration advocates that drug penetration into brain parenchyma from CSF is minimal, 68 and typically not more than 2 mm beneath the brain surface. 69 Our present and previous 6 observations demonstrate clearly the ability of a substance administered in CSF to enter parts of the brain far deeper than 2 mm, at least for a substance with (given) similar molecular properties as the CSF tracer we used (gadobutrol). It has also been shown that conjugation between a drug and a suitable carrier may enhance tissue penetration in the brain. 70 In AD, several attempts at therapeutically targeting pathological Aβ aggregates have been unsuccessful,71–73 perhaps due to inability of monoclonal antibodies to engage their target outside the blood–brain barrier. 74 In future developments, drugs with the ability to both target disease and enter the extravascular compartment (i.e. glymphatic pathways) may be considered attractive.
Supplemental Material
Supplemental material for Delayed clearance of cerebrospinal fluid tracer from entorhinal cortex in idiopathic normal pressure hydrocephalus: A glymphatic magnetic resonance imaging study
Supplemental material for Delayed clearance of cerebrospinal fluid tracer from entorhinal cortex in idiopathic normal pressure hydrocephalus: A glymphatic magnetic resonance imaging study by Per K Eide and Geir Ringstad in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article. We sincerely thank the Intervention Centre for providing valuable support with MR scanning and Department of Neurosurgery at Oslo University Hospital Rikshospitalet for care-taking of all study subjects throughout the study.
Acknowledgements
We thank Dr. Øivind Gjertsen, Dr. Bård Nedregaard and Dr. Ruth Sletteberg from the Department of Radiology, Oslo University Hospital – Rikshospitalet, who performed the intrathecal gadobutrol injections in all study subjects. Further, the author thanks Are Hugo Pripp, PhD, Department of Biostatistics, Epidemiology and Health Economics, Oslo University Hospital, Oslo, for statistical help during preparation of the paper. Finally, we sincerely thank the Nurse Staff at the Hydrocephalus outward clinic, Department of neurosurgery Oslo University Hospital – Rikshospitalet.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors' contributions
Both authors (PKE and GR) contributed to study design, data analysis, acquisition and interpretation, and have approved the final version for publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
