Abstract
Acute reperfusion despite persistent arterial occlusion may occur in up to 30% of ischemic stroke patients. Recruitment of leptomeningeal collaterals may explain this phenomenon. Using dynamic susceptibility-contrast perfusion imaging (DSC-PI), we assessed acute changes in collateral flow among patients without recanalization. From a multicenter prospective database (I-KNOW), 46 patients with magnetic resonance angiography visible occlusion in whom both reperfusion and recanalization were assessed within 6 h of onset were identified. Maps of collateral flow at arterial, capillary and late venous phases were generated from DSC-PI through inter-frame registration, baseline signal subtraction and temporal summation, and graded blind to all other relevant clinical and radiological data using the Higashida scale. Flow direction and the acute evolution of collaterals were evaluated against the reperfusion status. Among patients without recanalization (n = 33), flow direction remained retrograde. Collateral grades significantly improved between admission and acute follow-up in patients who reperfused (OR: 4.57; 95% CI: 1.1–22.7; p = 0.048), but not in those without reperfusion (OR: 1.34; 95% CI: 0.4–4.5; p = 0.623). Our study confirmed that acute reperfusion without recanalization is associated with a significant improvement of retrograde collateral flow. DSC-PI can detect acute changes in collateral flow, and may help evaluate novel treatments targeting leptomeningeal collaterals.
Introduction
Early reperfusion (i.e. reinstatement of blood flow at the capillary level) is the main therapeutic objective in acute ischemic stroke. As demonstrated in recent trials of intravenous tissue plasminogen activator (tPA) and thrombectomy, recanalization of the primary arterial occlusion is an effective way of achieving reperfusion and improving clinical outcome. 1 Still, although reperfusion and recanalization are closely related, these two phenomena can evolve independently over time. Recanalization without reperfusion (‘no-reflow’) may occur in cases with severely damaged microcirculation. 2 Conversely, significant reperfusion without recanalization can be observed in up to 30% of patients within the first 6 h of symptoms onset. 3 Available data suggest that reperfusion is a stronger predictor of tissue and clinical outcome than recanalization.3–6
We previously hypothesized that efficient retrograde collateral flow from leptomeningeal anastomoses likely underlies reperfusion without recanalization. 3 Cortical collaterals can mitigate the lack of anterograde flow and prolong the survival of, or even revert the ischemic penumbra. Good collaterals are strongly associated with slower infarct growth, lower risk of hemorrhagic transformation, increased likelihood of reperfusion and favorable clinical outcome.7–11
Because assessing collateral status may assist in patient triage and acute treatment decisions, and help evaluate novel treatments aiming at enhancing collateral flow, non-invasive imaging of collaterals by means of computed tomography (CT)-angiography and CT or MR-based perfusion imaging has gained increasing attention lately.7,12–16 Collateral grades at baseline, generated from dynamic susceptibility-contrast perfusion imaging (DSC-PI), were found to be well correlated with digital subtraction angiography (DSA) collateral grades, 14 subsequent lesion growth13,14 and clinical outcome. 14 In our previous work, lower hypoperfusion intensity ratio (proportion of voxels with Tmax ≥7 s within the Tmax ≥2 s lesion), an indirect marker of good collaterals, was observed at baseline in patients showing subsequent reperfusion without recanalization. 3
Still, only limited data so far have related the evolution of collateral flow to infarct growth 13 or clinical outcome, 17 and these studies explored late follow-up time points only (i.e. ≥24 h). In the present unique study of acute ischemic stroke patients in whom both presence of arterial occlusion and perfusion were assessed twice, acutely and again within 6 hours of onset, we determined whether acute reperfusion despite no recanalization is associated with: (1) good collaterals at baseline, as defined by DSC-PI-derived collateral maps; and (2) improved retrograde collateral flow over time. Our ancillary objective was to assess whether MRI-based collateral maps can detect acute changes in collateral flow.
Material and methods
Patients
We analyzed a multicenter, prospective database (I-KNOW) of patients who underwent both admission and serial follow-up MRI.18,19 Inclusion criteria for I-KNOW were: (1) NIHSS ≥4; (2) diffusion-weighted imaging (DWI) and DSC-PI consistent with an acute anterior circulation ischemic stroke; (3) admission MRI completed within 6 h for patients treated with intravenous tPA, or within 12 h for those managed without tPA. Patients with lacunar or posterior circulation stroke, unknown time of onset or intracerebral hemorrhage were excluded. No patient received intra-arterial therapy. For the present study, we added the following inclusion criteria: (i) both admission MRI (MRI-1) and acute follow-up (MRI-2, obtained 3 h after MRI-1 to assess acute reperfusion and recanalization) were available and assessable, and included DSC-PI, DWI and MR-angiography (MRA); and (ii) visible occlusion on baseline MRA.
Good functional outcome was defined as modified Rankin scale 0 or 1 at one month. Local ethics committee approved the protocol (Comité Consultatif de Protection des Personnes dans la Recherche Biomédicale – Lyon B; approval QH1/2006) and written informed consent was obtained from all patients and/or relatives. I-KNOW followed the ethical guidelines of the Declaration of Helsinki. The same cohort was the basis of a previous study that compared the prognostic impact of acute reperfusion and recanalization. 3
MRI
All patients underwent DWI (repetition time 6000 ms, field of view 24 cm, matrix 128 × 128, slice thickness 5 mm), Fluid-attenuated-inversion-recovery, T2-weighted gradient echo, MRA and DSC-PI (echo time 40 ms, repetition time 1500 ms, field of view 24 cm, matrix 128 × 128, 18 slices, slice thickness 5 mm; gadolinium contrast at 0.1 mmol/kg), both for MRI-1 and MRI-2.
Image analysis
Lesion segmentation and DSC-PI processing
Baseline DWI lesions (b = 1000 images) were semi-automatically outlined using an in-house developed software. After motion correction, maps of the time-to-maximum of the residue function (Tmax) were computed by circular singular value decomposition of the tissue concentration curves with an arterial input function from the contralateral middle cerebral artery (Penguin software, MATLAB 2010b; MathWorks, USA). 20 All images were coregistered within subjects to the baseline DSC-PI using statistical parametric mapping 8 (SPM; http://www.fil.ion.ucl.ac.uk/spm).
Tmax ≥6 s was used to define the perfusion lesion on MRI-1 and MRI-2. A voxel was deemed reperfused if Tmax was ≥6 s on MRI-1 and <6 s on MRI-2. Acute reperfusion was defined by a reperfusion ratio (volume of reperfused voxels on MRI-2/perfusion lesion volume on MRI-1) of ≥50%. 21 Acute recanalization was evaluated on MRI-2 MRA using the arterial occlusive lesion (AOL) classification: 0 = no recanalization of the primary occlusive lesion; 1 = incomplete or partial recanalization of the primary occlusive lesion with no distal flow; 2 = incomplete or partial recanalization of the primary occlusive lesion with any distal flow; and 3 = complete recanalization of the primary occlusive lesion with any distal flow. 22 A score ≥2 indicated acute recanalization.
MR-based collateral flow maps
Collateral maps were automatically generated from the raw DSC-PI according to the methodology of Kim et al.
14
Inter-frame rigid registration was performed to correct for patient motion, and non-brain tissue was removed from each time frame (FSL v5.0, FMRIB, Oxford, UK). As in DSA, to enhance the visualization of the contrast agent, the first frame was used to subtract the anatomical structures from all consecutive frames. Next, each time-frame was collapsed into a single summated signal intensity value for the whole brain. To partition the DSC-PI into physiologically driven phases, the summated signal intensity was plotted against time and a set of six consecutive frames with the lowest signal intensity was identified. As in Kim et al., we assumed that these frames corresponded to the mid phase of the collateral flow map. This phase was used as reference to generate the rest of the collateral flow maps by summing up the adjacent images. Thus, the DSC-PI was divided into an early, mid and late phase by summing an increasing number of frames (four, six, and eight, respectively). The early phase demonstrated the arrival of the contrast agent in the sylvian fissure; the mid phase was characterized by the maximal parenchymal ‘staining’ in the normal hemisphere; the late phase showed a delayed washout of gadolinium in the ischemic hemisphere. The processing of the collateral maps is summarized in Figure 1. Image computations were performed with an in-house program (MATLAB).
Processing of collateral flow maps (see text for details). Time elapsed since the start of image acquisition was as follows: early phase, 18.48–24.64 s; mid phase: 24.64–33.88 s; late phase: 33.88–46.20 s.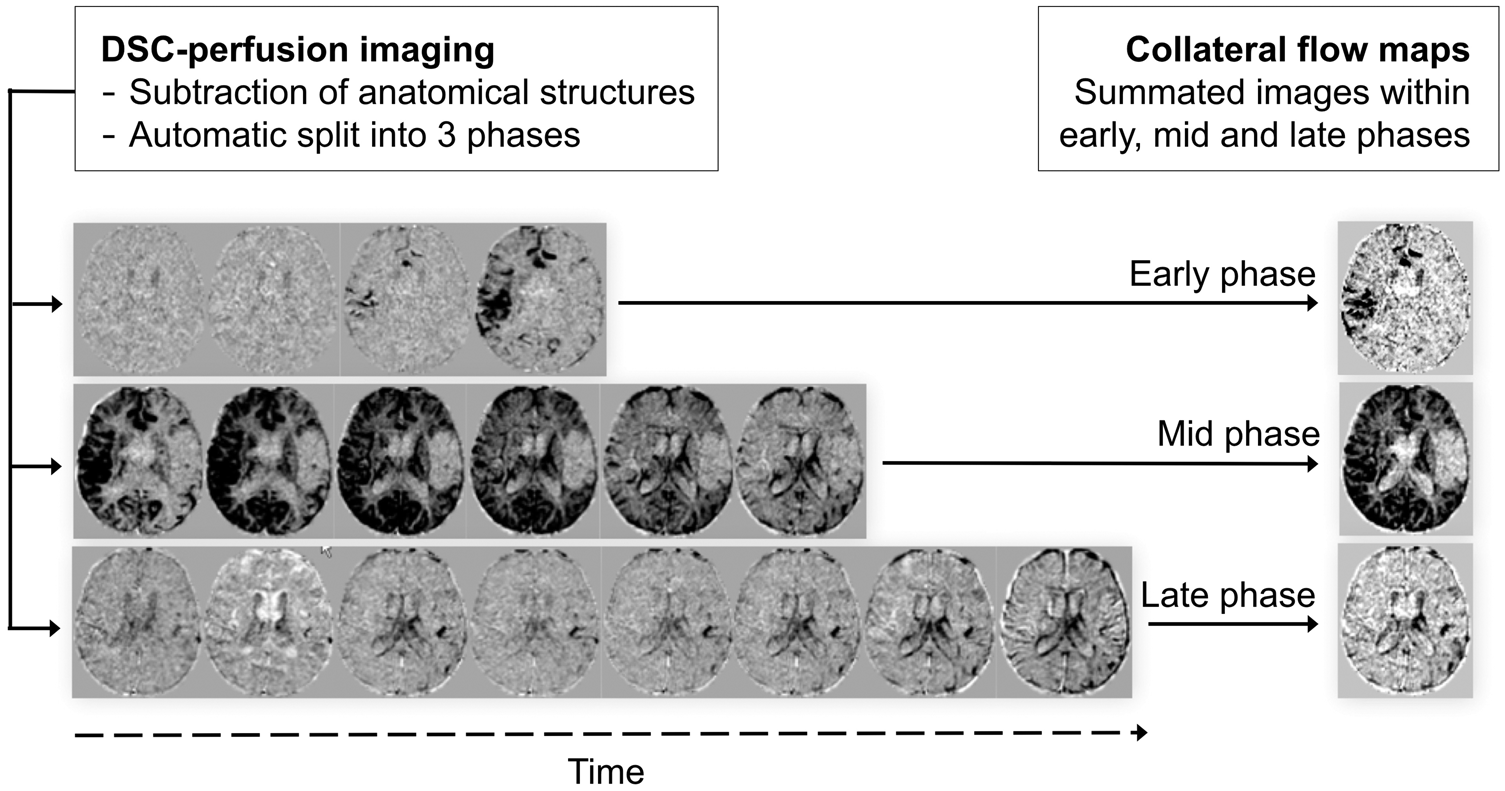
Collateral grades on MR-based maps
Collateral flow maps were graded by analyzing the extent of the contrast ‘staining’ over the early, mid and late phases. We adapted the Higashida (or American Society of Interventional and Therapeutic Neuroradiology/Society of Interventional Radiology) system: no visible collateral to the ischemic site (grade 0); slow collaterals to the periphery of the ischemic site with persistence of some of the defect (grade 1); rapid collaterals to the periphery of the ischemic site with persistence of some of the defect and to only a portion of the ischemic territory (grade 2); collaterals with slow but complete angiographic blood flow of the ischemic bed by the late venous phase (grade 3); complete and rapid collateral blood flow to the vascular bed in the entire ischemic territory by retrograde perfusion (grade 4).
23
Specifically, grades 1 and 2 were differentiated by a contrast staining of <50% and 50% to <100% of the ischemic territory, respectively (Figure 2). All slices covering the ischemic area were analyzed.
Collateral flow grading with the Higashida scale, applied on subtracted dynamic susceptibility-contrast perfusion imaging. All illustrated patients had an M1 occlusion; patient with grade 1 collaterals also had an intracranial internal carotid and A1 occlusions. The lesions were flipped on the same side for better legibility. Grade 0: no collateral flow was visible within the middle cerebral artery (MCA) territory. Grade 1: limited collateral flow was seen in the deep MCA territory. Grade 2: rapid collateral flow was present, staining >50% of the MCA territory (persistent defect in the anterior part). Grade 3: complete but slow (i.e. late venous phase) collateral flow in the entire MCA territory. Grade 4: complete and fast staining of the ischemic bed, with a delayed venous wash-out.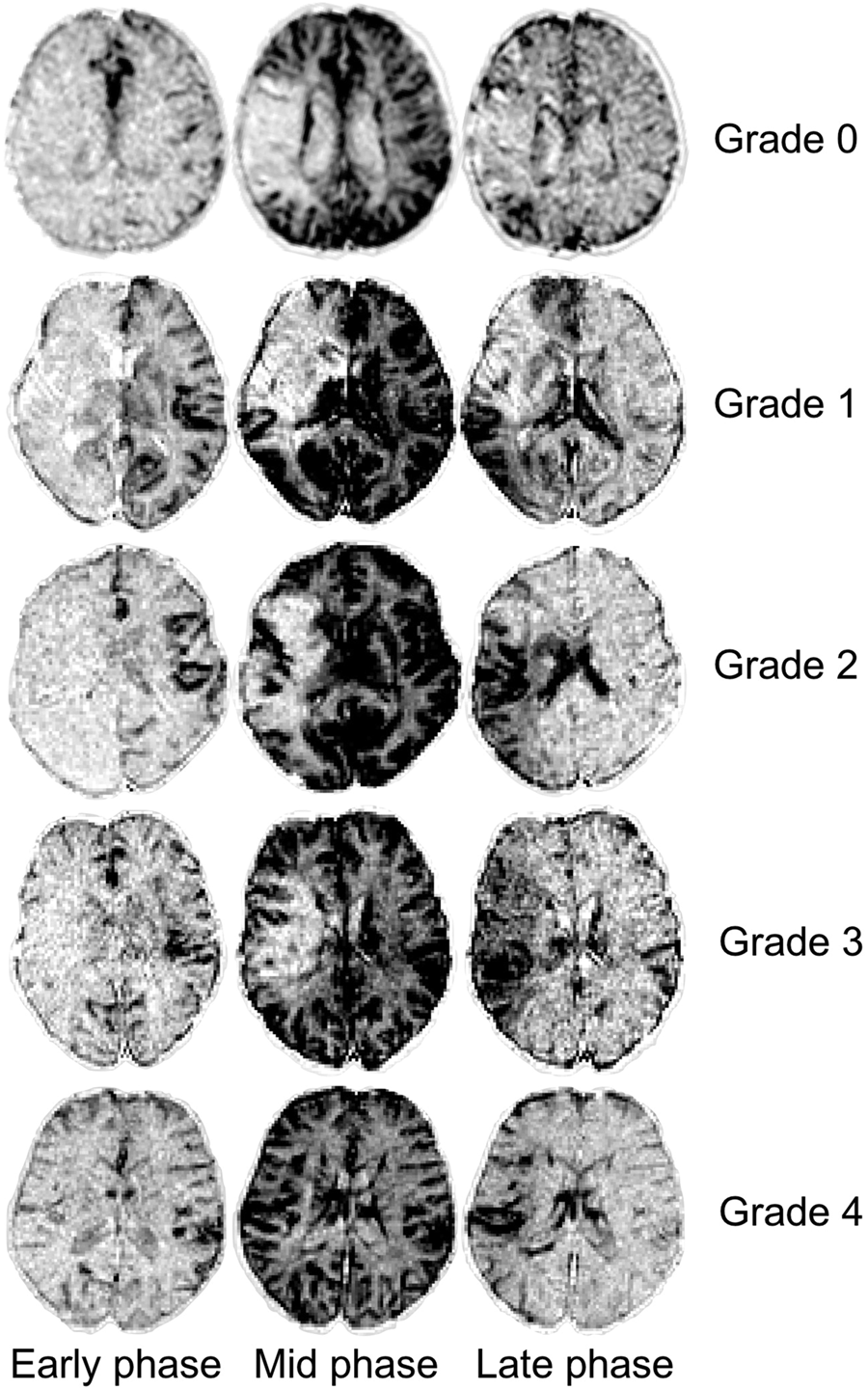
Flow direction
The direction of collateral blood flow was assessed using all time-frames from the DSC-PI, after inter-frame registration and subtraction of anatomical structures (i.e. after the first steps of DSC-PI processing as mentioned above). Flow direction was rated as anterograde when contrast staining followed an antero-posterior direction, as typically observed in non-occluded hemispheres, given the orientation of the middle cerebral artery (MCA) and its branches. Retrograde flow was characterized by a centripetal pattern (i.e. from the watershed areas to the center of the ischemic area).
After a common preliminary training session (using five randomly chosen cases), two raters (LC and THC), blinded to all clinical and radiological data except for the site and side of the baseline occlusion, independently assessed the collateral grades and flow direction. In discrepant cases, a final rating was made by consensus.
Statistical analysis
Clinical and imaging variables were described as median and interquartile range (IQR) or proportions as appropriate. Inter-rater agreement for collateral maps and flow direction was evaluated with Cohen’s kappa coefficient (weighted to the squared distance from perfect agreement). 24 The association between recanalization status and flow direction on MRI-2 was tested with McNemar’s test. 25
Our main analyses focused on patients without acute recanalization. Firstly, the impact of baseline collateral grades on reperfusion was evaluated by comparing: (1) the reperfusion ratio of patients with poor versus good collaterals (defined by grades 0–2 and grades 3–4, respectively), using the Wilcoxon rank-sum test; (2) the distribution of baseline collateral grades between patients with or without reperfusion and failed recanalization, using ordinal logistic regression (shift analysis). Secondly, ordinal logistic regressions assessed whether changes in collateral grades occurred between MRI-1 and MRI-2 according to the reperfusion status. The functional outcome of patients showing reperfusion without recanalization and those with both reperfusion and recanalization was compared in an exploratory analysis using Fisher’s exact test. 26
All statistical analyses were performed using R (MASS package, R Foundation for Statistical Computing, Vienna, Austria, http://www.R-project.org/).
Results
Patients’ characteristics and revascularization endpoints
Baseline parameters.
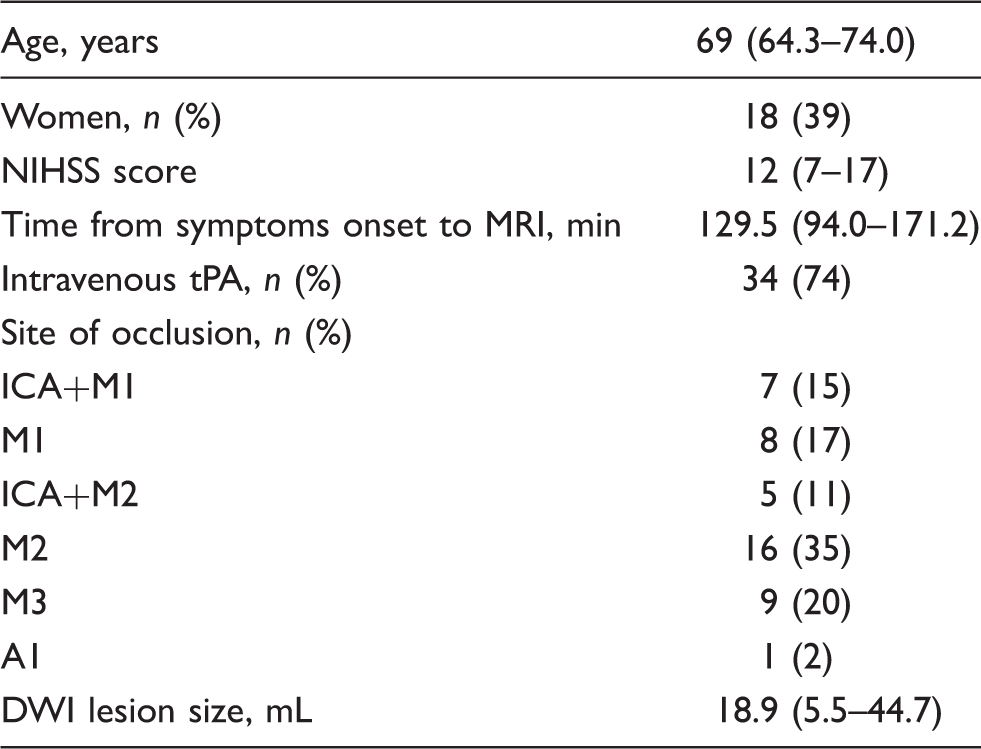
Note: Variables are given as median (interquartile range) or number (%).
DWI: diffusion-weighted imaging.
Collateral maps and flow direction
At baseline, grade 0 collaterals were found in 4 (8.7%) patients, grade 1 in 11 (23.9%), grade 2 in 11 (23.9%), grade 3 in 16 (34.8%) and grade 4 collaterals in 4 (8.7%). The kappa value for inter-rater agreement in grading MRI-based collateral maps was 0.85 (95% CI: 0.79–0.92).
Inter-rater agreement for flow direction had a kappa value of 0.69 (95% CI: 0.46–0.92). At baseline, flow was retrograde in all patients. On MRI-2, there was no significant discrepancy between flow direction and recanalization status (McNemar’s test, p = 0.32): i.e. flow remained retrograde in patients without recanalization, while it became anterograde in those with acute recanalization (Supplemental Movie).
Patients without recanalization (n=33)
Impact of baseline collateral grades
Poor and good collaterals on admission were observed in 22 (67%) and 11 (33%) patients, respectively. The reperfusion ratio was significantly higher in patients with good collaterals compared to those with poor collaterals at baseline (Wilcoxon rank-sum test, p = 0.004, Supplemental Figure). Patients with acute reperfusion despite persistent arterial occlusion had better collateral grades at baseline compared to those without reperfusion (OR: 37.2; 95% CI: 6.97–313.74; p = 0.0001, Supplemental Figure).
Acute evolution of collaterals
Among patients without recanalization, those who experienced acute reperfusion (i.e. reperfusion without recanalization) had a significant improvement of collateral grades from MRI-1 to MRI-2 (OR: 4.57; 95% CI: 1.1–22.7; p = 0.048; Figures 3(a) and 4). Conversely, patients without recanalization who did not reperfuse exhibited no change in collateral flow over time (OR: 1.34; 95% CI: 0.4–4.5; p = 0.623; Figure 3(b)).
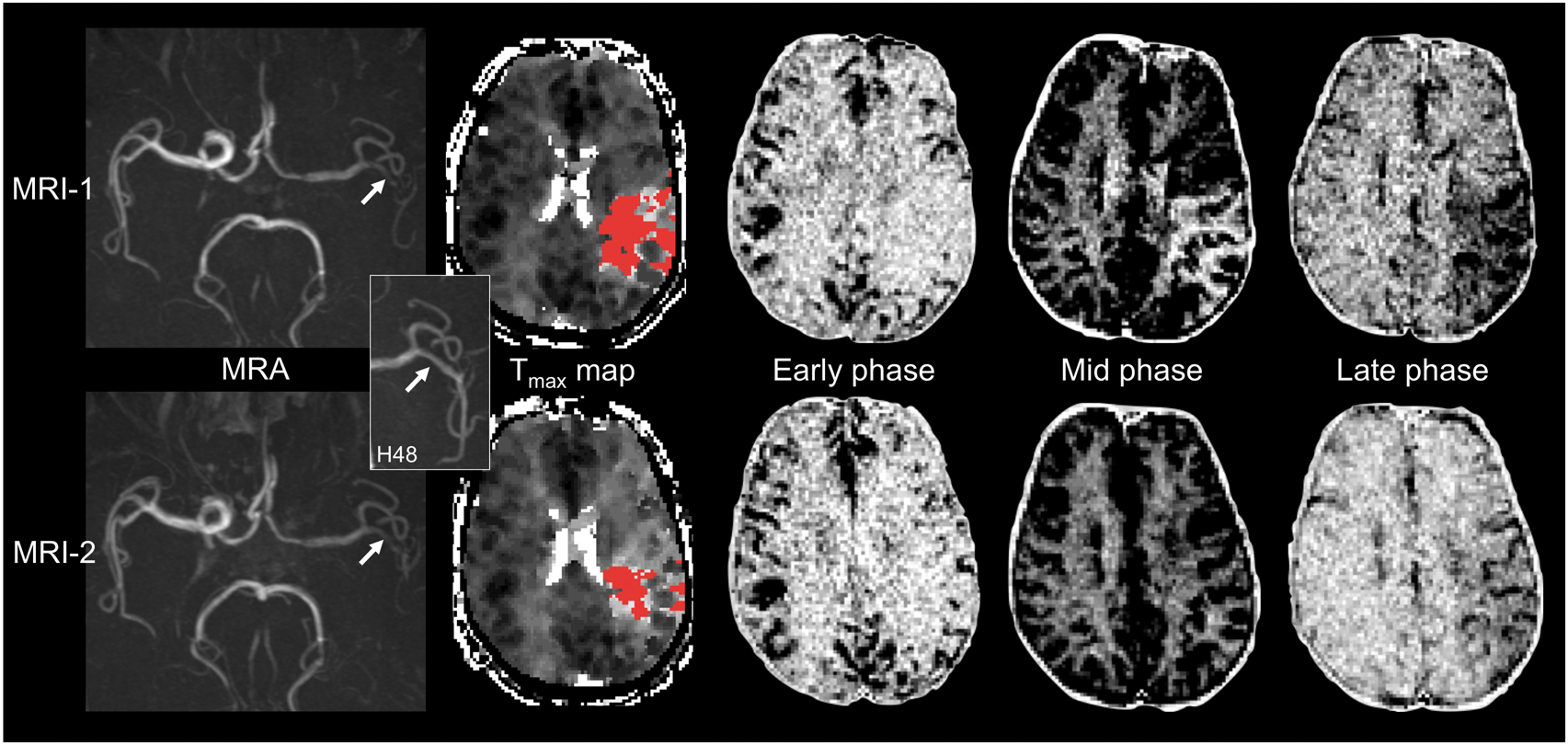
Distribution of collateral grades at baseline (MRI-1) and acute follow-up (MRI-2) in non-recanalized patients with (a) or without (b) early reperfusion. Fifty-six-years-old male with left internal carotid artery and M2 occlusion (upper row, magnetic resonance angiography (MRA), arrow). No recanalization was observed on MRI-2 (lower row, MRA, arrow). The inset shows a late M2 recanalization at 48 h (arrow). Acute reperfusion (reperfusion ratio: 85%) was found on Tmax maps (red overlay indicates Tmax ≥ 6 s). An improvement of collateral flow was observed between admission (MRI-1, upper row; Higashida grade: 3) and acute follow-up (MRI-2, lower row; Higashida grade: 4).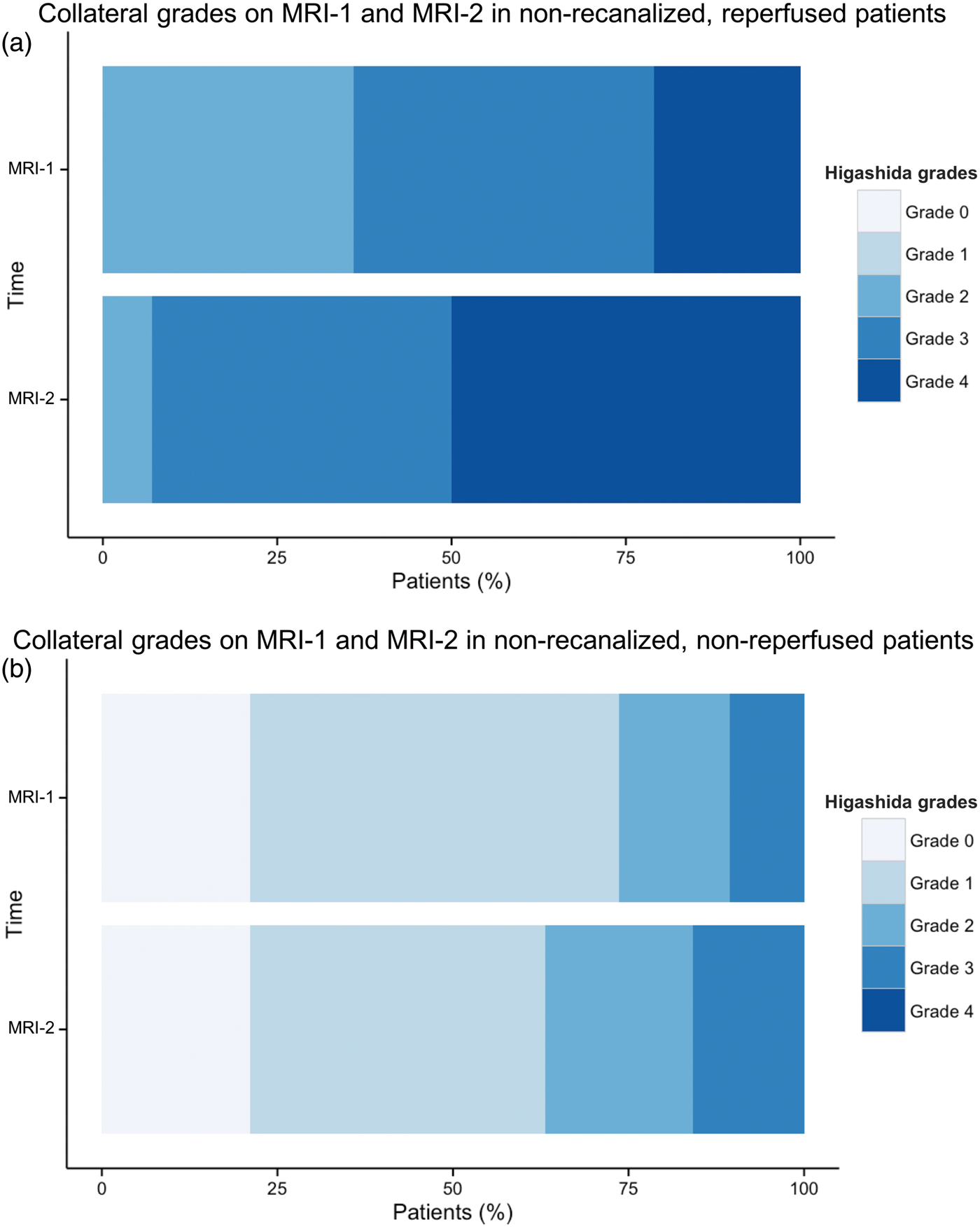
Among patients with acute reperfusion, no difference in functional outcome at one month was found between those with or without recanalization, i.e. patients with anterograde or retrograde reperfusion, respectively (p = 0.71).
Discussion
In this unique study of hyperacute stroke patients in whom perfusion MRI was obtained at baseline and again a few hours later, we investigated the relationship between reperfusion, recanalization and cortical collaterals. In agreement with our hypotheses, acute reperfusion without recanalization was significantly related to better collaterals at baseline, and subsequently, improved retrograde flow through cortical collaterals resulting in retrograde tissue reperfusion.
This is the first study to assess the hyperacute (<6 h) dynamics of leptomeningeal collateral flow in ischemic stroke. Only two previous studies reported on the evolution of collaterals and its impact on tissue or clinical outcome. Campbell et al. 13 found that deterioration of collaterals was associated with increased hypoperfusion severity and infarct growth. However, causality was uncertain given the delayed follow-up assessment (day 3–5), when extensive infarction and edema can severely impede retrograde (or anterograde) flow. Interestingly, using CT-angiography, Yeo et al. 17 recently reported that in patients without recanalization or with poor collaterals at baseline, late (i.e. 24 h) recruitment of cortical collaterals was associated with worse clinical outcome and higher mortality. Because these patients are likely to experience faster lesion growth and larger infarcts, a delayed increase in collateral flow may foster harmful reperfusion injury. Thus, acute and delayed changes in collaterals may have entirely distinct causes and effects.
Three main mechanisms account for the positive effects of collaterals: (i) prolonged survival of the ischemic tissue via improved tissue perfusion, 10 and hence extended therapeutic time window; (ii) increased rate of recanalization, 8 possibly related to an improved delivery of exogenous and endogenous thrombolytics to the distal side of the clot; and (iii) reduced damage to the microcirculation implying a lesser risk of subsequent no-reflow in the event of recanalization, be it therapeutic or spontaneous. Our results suggest that even in patients without recanalization, increasing collateral flow within the first 6 h after vessel occlusion may promote reperfusion and in turn a favourable clinical outcome. In the same cohort, we previously reported that acute reperfusion was an independent predictor of good functional outcome, regardless of recanalization. 3 The present study suggests that retrograde reperfusion via pial collaterals may be of similar clinical benefit as is anterograde reperfusion.
So far, the majority of research on non-invasive imaging of collaterals has used static images from single-phase CT-angiography.7,12,27,28 More recent studies highlighted the superior prognostic value of dynamic data, using either multiphase CT-angiography 29 or various post-processing of CT-perfusion.30–32 Subtracted DSC-PI inherently combines an assessment of both the anatomical extent and the velocity of collateral flow. This approach was first described by Campbell et al. 13 Kim et al. 14 modified this approach by merging multiple time frames, producing summary collateral maps at early, mid and late phases. Two other recent reports evaluated collaterals with subtracted DSC-PI.16,33 One can visualize the entire dynamic series (∼50 time-frames amounting to ∼1000 images),13,16,33 or as in the present study, merge multiple frames into three main flow phases. 14 Using temporally summated maps, our inter-rater agreement (weighted kappa: 0.85) was higher than in reports using non-summated maps (weighted kappa: 0.5816 and 0.7113). This could suggest that summated maps may be easier to interpret, but this hypothesis will require an independent comparison. Our study also indicates that applying the Higashida scale on subtracted DSC-PI is an effective, non-invasive method for detecting changes in collateral flow during the first 6 h, i.e. the most critical period for tissue and clinical outcome.
Do subtracted DSC-PI collateral maps have an added value to routinely available diffusion and perfusion MRI? Previous data have shown that MRI-based collaterals are strongly correlated with the baseline DWI lesion size,13,14,16 an established marker of outcome, as well as with hypoperfusion severity on Tmax maps.13,16,34 Accordingly, the baseline DWI lesion size has been found independently associated with infarct growth and clinical outcome, whereas collateral grade did not improve these predictive models.13,14,16 Recently, Potreck et al. 15 introduced yet another DSC-PI based method of assessing cortical collaterals: the extent of severe Tmax delay (≥10 s) within the subarachnoid space was determined and found to correlate with DSA. Fast pial collaterals (Tmax ≥10 s in < 1% of the subarachnoid volume) were associated with favorable clinical outcome, and appeared to add to the prognostic value of the standard DWI-Tmax mismatch ratio. However, the baseline DWI lesion volume was not considered in the predictive model. Thus, additional studies are required to determine whether the evaluation of cortical collaterals increases the predictive information of standard core and penumbra imaging. Still, collateral maps present potential advantages over the latter, namely simpler post-processing steps compared to those required to derive core, penumbra and mismatch maps from MRI or CT-perfusion, and a straightforward grading system well-correlated with the Higashida DSA scale, which could translate into easier implementation and improved reproducibility between multiple centers.
Our study has some limitations. Our analyses were based on a limited population with mostly distal (>M1) occlusions. The frequency and clinical impact of retrograde reperfusion will require further evaluation in homogenous cohorts with proximal occlusions. The evaluation of our patients did not include DSA. Although MRI-defined collaterals were well correlated with DSA in a previous study, 14 our results would benefit from an independent validation using catheter angiography. Admittedly, iterative examinations with DSA may be limited to specific settings (e.g. trials of invasive collateral therapeutics such as partial aortic occlusion). Intravenous tPA was not associated with improved reperfusion rates or clinical outcome. Although it may suggest that tPA has no significant effect on collaterals and retrograde reperfusion, limitations inherent to a non-randomized study of limited size preclude any definite conclusion.
In conclusion, we documented that acute reperfusion without recanalization was related to improved retrograde collateral flow. Perfusion-weighted MRI can detect acute changes in collateral circulation, and may help evaluate novel treatments targeting leptomeningeal collaterals.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the European Commission’s Sixth Framework Program (grant 027294).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Nikolaos Makris: analysis and interpretation of the data, drafting of the manuscript and revisions of the manuscript. Leila Chamard: analysis and interpretation of the data, and revisions of the manuscript. Irene Klærke Mikkelsen: data analysis and interpretation, revisions of the manuscript. Marc Hermier: acquisition of data and revisions of the manuscript. Laurent Derex: acquisition of data and revisions of the manuscript. Salvador Pedraza: acquisition of data and revisions of the manuscript. Götz Thomalla : acquisition of data and revisions of the manuscript. Leif Østergaard : concept and design, acquisition of data, and revisions of the manuscript. Jean-Claude Baron : concept and design, acquisition of data, data analysis and interpretation, drafting and revisions of the manuscript. Norbert Nighoghossian: concept and design, acquisition of data, data analysis and interpretation, drafting and revisions of the manuscript. Yves Berthezène: concept and design, acquisition of data, data analysis and interpretation, drafting and revisions of the manuscript. Tae-Hee Cho: concept and design, acquisition of data, data analysis and interpretation, drafting and revisions of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
