Abstract
Early detection of hemorrhagic transformation after reperfusion therapy is crucial in acute stroke treatment. Here, we evaluated the relationship between hemorrhagic transformation and post-reperfusion hyperperfusion using pulsed arterial spin labeling (ASL) perfusion MRI and 123I-iodoamphetamine single-photon emission-computed tomography. Patients who developed hemorrhagic transformation showed significantly higher cerebral blood flow in the affected lesion after thrombolysis and/or endovascular intervention. Focal hyperperfusion (ipsilateral to contralateral ratio >1.5) was associated with hemorrhagic transformation after reperfusion (odds ratio, 9.3; 95% confidence interval, 1.4–64.0). Our findings suggest that post-reperfusion hyperperfusion on ASL could represent a reliable marker of hemorrhagic transformation.
Keywords
Introduction
Hemorrhagic transformation is a major concern in acute stroke patients treated with intravenous thrombolysis or endovascular intervention. Early detection of hemorrhagic transformation after reperfusion therapy is crucial in determining subsequent treatment strategies, including blood pressure management and antithrombotic therapy initiation. Cerebral hyperperfusion after carotid endarterectomy or carotid artery stenting has been considered as a risk factor for hemorrhagic transformation and poor prognosis. 1 To date, however, there is a lack of data on the significance of cerebral hyperperfusion after acute reperfusion therapy. In this study, we evaluated the relationship between hemorrhagic transformation and post-ischemic hyperperfusion after reperfusion therapy using pulsed arterial spin labeling (ASL) perfusion MRI. In addition, we compared images from ASL perfusion with those from single-photon emission-computed tomography (SPECT) in patients who underwent both ASL and SPECT after reperfusion therapy.
Materials and methods
This observational study was performed at National Cerebral and Cardiovascular Center, Suita, Japan. The local ethics committee of National Cerebral and Cardiovascular Center approved the study. The Japanese Ethical Guidelines for Medical and Health Research Involving Human Subjects 2 were followed throughout the study. The requirement of informed consent was waived in accordance with the guidelines and the local ethics committee. We reviewed the prospectively collected database of acute stroke patients with middle cerebral artery (MCA) occlusion who underwent intravenous thrombolysis or endovascular intervention between November 2015 and June 2016. MRI examinations, including ASL, were recommended to all patients without contraindications who had undergone reperfusion therapy for the purpose of evaluating the therapeutic efficacy and complications. In addition, in order to examine late-onset complications, all patients received additional follow-up CT before discharge. All MRIs were performed on a 3.0 Tesla MRI scanner (Spectra, Siemens, Erlangen, Germany). All ASL images using second version of a system for the quantitative imaging of perfusion with thin-slice TI1 periodic saturation (Q2TIPS), flow-sensitive alternating inversion recovery (FAIR) labeling scheme with single shot three-dimensional gradient and spin echo (3D-GRASE) readout (TR/TE, 5000/37 ms; TI, 1800 ms; voxel dimensions, 1.5 × 1.5 × 3 mm3; matrix, 63 × 64; 1 average; acquisition time, 2 min 45 s) and N-isopropyl-p-123I-iodoamphetamine (IMP) SPECT images after reperfusion therapy were analysed using fully automated three-dimensional stereotactic region-of-interest template (3DSRT) software (Fujifilm RI Pharma, Japan), as described previously.3,4 In brief, non-labelled (control) ASL images before subtraction were registered to the baseline FLAIR images of each patient. The calculated transformation matrix was then applied to the corresponding ASL subtraction perfusion images, and then normalized into the Montreal Neurological Institute (MNI) space via registration of the FLAIR images to the MNI template. Normalized ASL images were analysed using 3DSRT template. 3 Arterial transit artifact was identified as tubular hyperintense signal on ASL perfusion images in reference to FLAIR images. 4 The ipsilateral-to-contralateral cerebral blood flow ratio (IC ratio) of each MCA branch territory (precentral, central, parietal, angular, temporal, and basal ganglia) and the whole MCA territory was calculated. Follow-up CT images were independently assessed for hemorrhagic transformation by two stroke neurologists (SO and TY), according to the Second European-Australasian Acute Stroke Study (ECASS II) criteria. 5 Patients with subarachnoid hemorrhage induced by endovascular perforation were excluded. There was consistent agreement between the two observers and any discrepancies were resolved by consensus. The relationship between ASL and SPECT IC ratio was assessed by linear regression analysis. Significance was determined by Student’s t-test for continuous variables, and Fisher’s exact test for categorical variables.
Results
Of the 47 consecutive patients with MCA occlusion who received acute reperfusion therapy during the study period, 31 patients undergoing ASL after treatment (median 3 days from reperfusion) were analysed. IMP SPECT was performed in 11 patients (4 days from reperfusion). Intravenous thrombolysis was performed in 19 patients and endovascular intervention in 23. Eleven patients received both intravenous thrombolysis and endovascular intervention. In the comparison of ASL and IMP SPECT images, the IC ratio of each MCA branch territory, measured by ASL, was well correlated with that acquired by IMP SPECT (Figure 1(a), R = 0.71, P < 0.01).
(a) Correlation between ASL and 123I-IMP SPECT of regional cerebral blood flow. The IC ratio indicates ipsilateral-to-contralateral cerebral blood flow ratio. (b) Relationship between regional cerebral blood flow in the whole MCA territory and the development of hemorrhagic transformation after acute reperfusion therapy.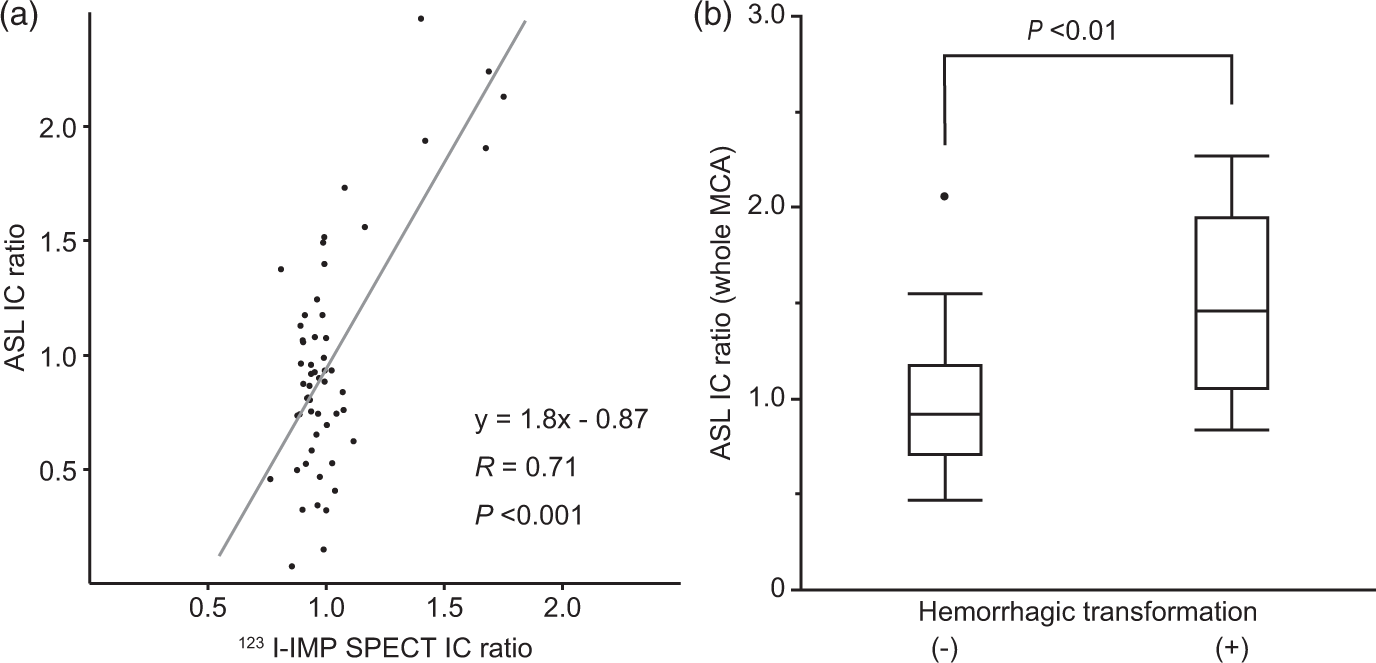
Hemorrhagic transformation was observed in seven patients (six HI and one PH), all asymptomatic. Among these seven patients, five showed hemorrhagic change in the affected lesion on T2*-weighted image at the time of ASL examination.
The IC ratio in the whole MCA territory of patients with hemorrhagic transformation was significantly higher than those without (Figure 1(b), 1.50 ± 0.53 vs. 0.98 ± 0.37, P < 0.001). Cerebral hyperperfusion in the ipsilateral MCA territory (IC ratio > 1.5) was observed in 7 of 31 patients (23%), and 2 showed diffuse hyperperfusion in the whole MCA territory; 4 of these 7 patients (57%) developed hemorrhagic transformation, while 3 of the other 24 patients without hyperperfusion (13%) showed hemorrhagic transformation (P = 0.03). This cerebral hyperperfusion was observed inside or adjacent to hyperintensity on diffusion-weighted images. The presence of cerebral hyperperfusion (IC ratio > 1.5) was associated with the development of hemorrhagic transformation after reperfusion therapy (odds ratio 9.3, 95% confidence interval 1.4–64.0). A representative case is shown in Figure 2.
Representative case. A 52-year-old woman on hormone replacement therapy presented with aphasia and right hemiplegia. Initial diffusion-weighted MRI showed acute cerebral infarction in the distribution of the left internal carotid artery (a). She had the left internal carotid artery occlusion (b) and received successful endovascular intervention (c). Post-interventional ASL on Day 4 (d) and IMP-SPECT on Day 9 (e) revealed marked hyperperfusion in the left basal ganglia. T2*-weighted MRI on Day 4 (f) showed asymptomatic hemorrhagic transformation in the corresponding area.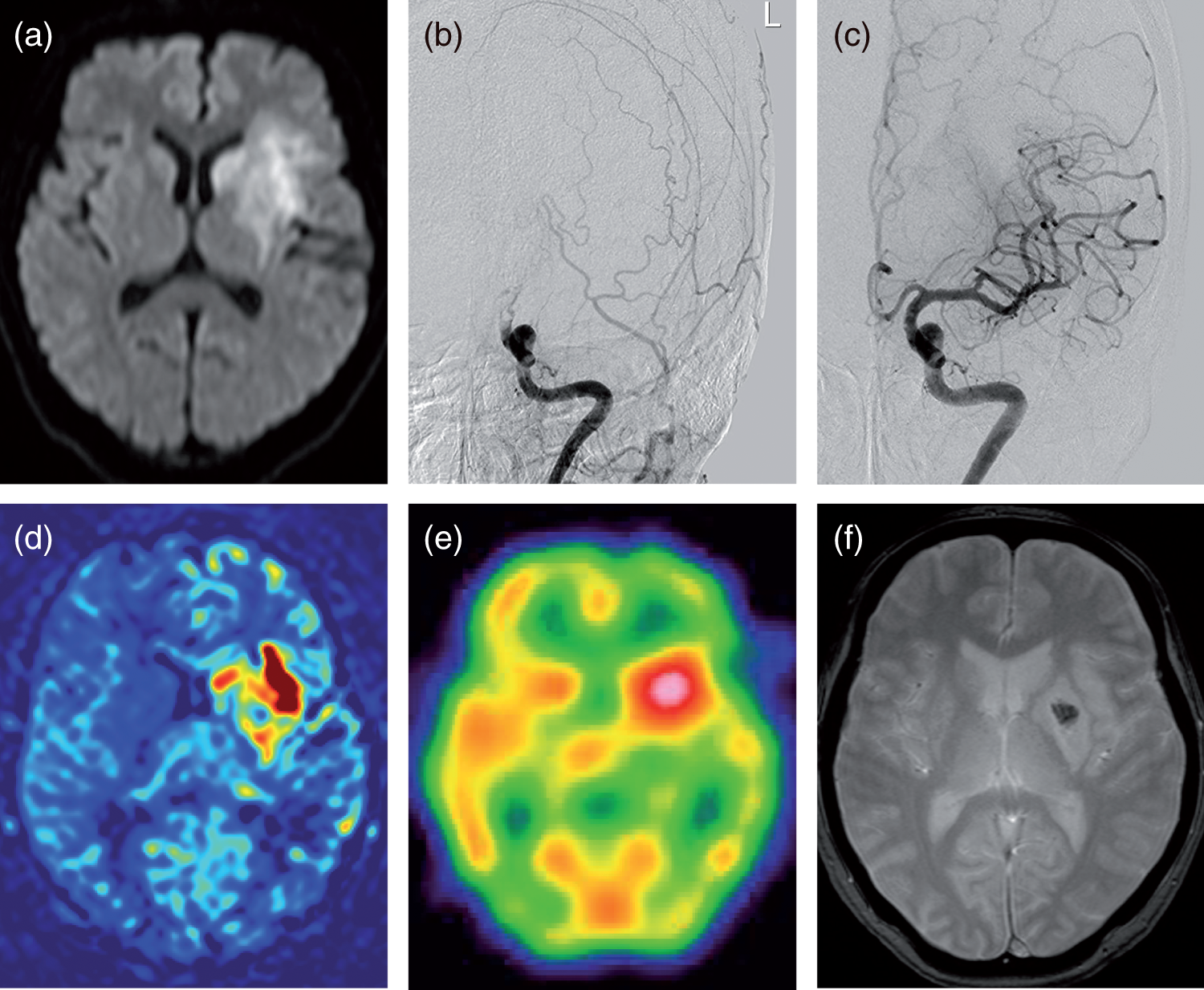
Discussion
This study shows that acute stroke patients with ASL hyperperfusion in the ischemic lesion after intravenous thrombolysis or endovascular intervention had a high risk of hemorrhagic transformation. Cerebral hyperperfusion in the chronically ischemic brain after revascularization has been well investigated in patients with carotid stenosis or moyamoya disease.1,6,7 Impaired cerebrovascular autoregulation caused by ischemia-induced endothelial dysfunction, which can occur inside and adjacent to the acute infarct lesions, is involved in the pathogenesis of cerebral hyperperfusion. 6 Hyperperfusion can contribute to the development of hemorrhagic transformation by causing blood–brain barrier disruption. 8 Recent studies have revealed that ASL is very sensitive to pathologic states, manifesting as focal and regional hyperperfusion. 9 Postischemic hyperperfusion on ASL is detected in about 30% of acute ischemic patients, and is very common in patients receiving intravenous thrombolysis. 10 In accordance with the current study, Yu et al. 10 reported ASL hyperperfusion was associated with hemorrhagic transformation, at any time point.
Our study has limitations. First, ASL and follow-up CTs were not acquired at regular intervals after reperfusion therapy. Consequently, a causal relationship between ASL hyperperfusion and hemorrhagic transformation could not be established. About 70% of patients with hemorrhagic transformation had already hemorrhage at the time of ASL imaging. There is no denying that hyperperfusion on ASL was influenced by the hemorrhage itself. Second, in the comparison between ASL and SPECT of regional cerebral blood flow (Figure 1(a)), multiple plots were collected from a patient. This can cause to overestimate the correlation in the linear regression analysis. Third, no cases with symptomatic hemorrhage were included in the study. It is therefore unclear whether the hemorrhagic transformation observed in patients affected the functional prognosis. Larger prospective studies are needed to establish the validity of ASL imaging after reperfusion therapy.
In conclusion, our data suggest that ASL hyperperfusion after intravenous thrombolysis or endovascular intervention could represent a reliable marker of hemorrhagic transformation. Early detection of ASL hyperperfusion after reperfusion therapy may make it possible to reduce hemorrhagic risk through more careful treatment, such as the intensive lowering of blood pressure and withholding of antithrombotic therapy.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by The Grant-in-Aid for Community Health and Medical Care from the Osaka University Medical School Alumni.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
SO, HY, TY, YM conceived and designed the work. SO, TY, YM were involved in the analysis and interpretation of the data. SO wrote the manuscript and HY, KT, MI provided critical revisions.
