Abstract
Cerebral white matter hyperintensities are believed the consequence of small vessel disease and are associated with risk and progression of Alzheimer's disease. The ɛ4 allele of the APOE gene is the major factor accountable for Alzheimer's disease heritability. However, the relationship between white matter hyperintensities and APOE genotype in healthy subjects remains controversial. We investigated the association between APOE-ɛ4 and vascular risk factors with white matter hyperintensities, and explored their interactions, in a cohort of cognitively healthy adults (45–75 years). White matter hyperintensities were assessed with the Fazekas Scale from magnetic resonance images (575 participants: 74 APOE-ɛ4 homozygotes, 220 heterozygotes and 281 noncarriers) and classified into normal (Fazekas < 2) and pathological (≥2). Stepwise logistic regression was used to study the association between pathological Fazekas and APOE genotype after correcting for cardiovascular and sociodemographic factors. APOE-ɛ4 homozygotes, but not heterozygotes, bear a significantly higher risk (OR 3.432; 95% CI [1.297–9.082]; p = 0.013) of displaying pathological white matter hyperintensities. As expected, aging, hypertension and cardiovascular and dementia risk scales were also positively associated to pathological white matter hyperintensities, but these did not modulate the effect of APOE-ɛ4/ɛ4. In subjects at genetic risk of developing Alzheimer's disease, the control of modifiable risk factors of white matter hyperintensities is of particular relevance to reduce or delay dementia’s onset.
Keywords
Introduction
Small vessel cerebrovascular pathology constitutes one of the most common findings in neuroimaging studies carried out in late adulthood and elderly individuals.1,2 The most common of these lesions can be observed through computed tomography or magnetic resonance imaging (MRI) as areas of increased signal in the white matter, commonly in periventricular locations, and are referred to as white matter hyperintensities (WMH). The Fazekas scale 3 rates WMH on a 0–3 point scale in the periventricular and subcortical regions combined. The Fazekas scale was designed for cross-sectional rating of WMHs, shows good intra- and inter-observer agreement and closely correlates with volumetric assessments.4,5
It is widely accepted that the etiology of these WMH is of ischemic origin, but the precise underlying pathology is not completely understood. 6 In this way, several alterations have been described in brain samples of deceased patients ranging from local blood brain barrier alterations with leakage of plasmatic proteins to major myelin destruction and axonal loss.7–9 Both the prevalence and extension of WMH are strongly associated with aging and cardiovascular risk factors (CRF), particularly hypertension. 10 The reported prevalence of WMH is highly variable, ranging from below 10% to over 90% of a given study’s participants. This variability can be accounted for by differences in the age of participants included in these cohorts and their comorbidities.11,12 Additional factors could also contribute to these differences, such as ethnicity and lifestyles of the evaluated samples as well as technical differences in the MRI protocols.13–15
From a clinical perspective, severe affectation of white matter could lead to vascular dementia and the presence of these lesions may also contribute to the development of cognitive impairment in other causes of dementia.16,17 For instance, it has been suggested that the presence of WMH could have an additive deleterious effect to amyloid pathology in Alzheimer’s disease (AD), facilitating the progression of cognitive deterioration.18,19 The contribution of midlife vascular risk factors in future brain function deterioration is further supported by risk scores for vascular disease like those from the Framingham study, which predict the development of cognitive deterioration. 20 Similarly, the CAIDE (Cardiovascular risk factors, Aging, and Incidence of DEmentia) risk score not only correlates with the extension of WMH, but also predicts the development of dementia after a follow-up of three decades.21,22
Although the etiology of sporadic AD remains elusive, the Apolipoprotein E gene (APOE) genotype is the major known factor accountable for the heritability of the disease. 23 In this regard, the presence of one APOE-ɛ4 allele increases the risk of developing AD by a factor of 3 approximately, and homozygotes for this allele have nearly 14 times higher risk than APOE-ɛ3/ɛ3 individuals. By contrast, the APOE-ɛ2 allele seems to confer resistance towards developing the disease. 24 In addition to its role in sporadic AD, the APOE-ɛ4 allele has also been associated with atherosclerosis, a risk factor for the development of cerebral vascular lesions and, eventually, vascular dementia. 25 In this regard, it has been proposed that the APOE genotype could be an independent risk factor for the development of WMH. 26 Thus, the association of the APOE-ɛ4 allele with the development of AD could be explained, at least in part, by the major presence of WMH and other vascular lesions in these patients, which, in turn, could facilitate the onset of cognitive impairment and, ultimately, lead to dementia. In this way, it has been suggested that APOE-ɛ4 carriers could be more vulnerable to environmental factors that facilitate cognitive deterioration including some vascular risk factors such as fat dietary intake. 21
Several authors have described a positive association between the APOE-ɛ4 allele and the prevalence of WMH.27–30 However, these works frequently included patients with relevant comorbidities or advanced age that increased the prevalence of WMH and could mask the effect of the APOE genotype. Moreover, the number of subjects homozygous for the APOE-ɛ4 allele recruited in previous studies was, in general, too low to confidently evaluate whether the number of alleles could have an additive effect on WMH in the same way that occurs with the development of AD: the largest of them only included 21 homozygotes from a total of 1779 recruited subjects. 28
In the present work, we sought to investigate the association between the presence of the APOE-ɛ4 allele with WMH in a cohort of cognitively healthy adults aged between 45 and 75 years. In addition, we aimed to recruit a high number of APOE-ɛ4 homozygotes, with the aim of assessing whether the number of APOE-ɛ4 alleles could have an additive effect on WMH load. Finally, we aimed to study the association of vascular risk factors and their potential interaction with the APOE-ɛ4 allele.
Materials and methods
Participants
The ALFA (for
Research ethics and patient consent
The study was approved by the Ethics Committee of the “Parc de Salut Mar” (Barcelona, Spain; MRI/FBB2014v1.0) and conducted in accordance to the directives of the Spanish Law 14/2007, of 3 July, on Biomedical Research. All participants accepted the study procedures by signing an informed consent.
Cardiovascular and dementia risk factors
At ALFA baseline assessment, participants were enquired about their familiar and personal medical history and chronic medication use was recorded. Participants’ weight, height, blood pressure, and waist and hip circumference were measured. Participants were considered as hypertensive if their measured systolic blood pressure was ≥160 mmHg and/or if they were on antihypertensive medication and/or if they self-reported to suffer current hypertension. Participants were classified as having diabetes or hypercholesterolemia if they were on anti-diabetic or anti-hypercholesterolemic medication, respectively. In addition, the presence of these comorbidities was also taken as positive if self-reported. Participants’ weight and height measurements were used to calculate their body mass index (BMI). Smoking habits were collected using a simplified version of the 2009 Spanish Ministry of Health National Plan on Drugs Questionnaire. Finally, participants’ level of physical activity was measured with the Spanish short version of the Minnesota Leisure Time Physical Activity Questionnaire 32 and they were categorized as active if exercised at least 150 min per week of moderate workout or 75 min per week of vigorous workout as recommended by the current guidelines.
Cardiovascular and dementia risk scores
Clinical and lifestyle-related variables gathered were used to estimate participants’ risk of suffering coronary disease events at 10 years, using the REGICOR cardiovascular risk function (an adaptation of the Framingham function validated in a Spanish sample 33 ), and the probability of dementia in 20 years, using the CAIDE dementia risk score, a well-established approach to assess the probability of dementia in late life according to the risk score categories in middle age. 34 Participants’ risk scores were estimated using both CAIDE statistical models that differ in whether they do not include (model I) or include (model II) the APOE status into consideration.
The estimated variables were as follows: (i) For the risk of suffering coronary disease events. Total cholesterol: if a participant did not self-report to suffer hypercholesterolemia and they were not on anti-hypercholesterolemic medication, the assigned cholesterol value was ≥160 and <200 mg/dl, corresponding to a coefficient of 0 in the REGICOR Cox model. If they self-reported hypercholesterolemia and/or were on anti-hypercholesterolemic medication, the assigned value was ≥240 and <280 mg/dl, corresponding to a coefficient of 0.5054 and 0.2439 for men and women, respectively, in the same model. HDL-cholesterol: Participants were assigned a ≥45 to <50 mg/dl value, corresponding to a coefficient of 0 and 0.1979 for men and women, respectively, in the REGICOR Cox model. (ii) For the probability of dementia in 20 years. Total cholesterol: if a participant did not self-report to suffer hypercholesterolemia and they were not on anti-hypercholesterolemic medication, the assigned cholesterol value was 6.5 mmol/L, corresponding to a coefficient of 0 in the CAIDE Cox model. If they self-reported hypercholesterolemia and/or were on anti-hypercholesterolemic medication, the assigned value was 6.6 mmol/L, corresponding to a coefficient of 0.631 and 0.460 in model I and II, respectively.
Apolipoprotein E genotyping
Participants’ APOE genotype was determined as described in Molinuevo et al. 31
MRI acquisition
Scans were acquired using a 3.0-T scanner (GE Discovery MR750 W 3T). The MRI protocol was identical for all subjects and included high-resolution T1-weighted 3D structural images with an isotropic voxel size of 1 mm3 (TR/TE/TI = 8.0/3.7/450 ms, NSA = 1, Flip angle = 8° and a matrix size of 256 × 256 × 160), as well as three T2-weighted pulse sequences (256 × 256, 1 × 1 × 3 mm matrix): (i) Fluid attenuation inversion recovery (FLAIR: TR/TE/TI = 11000/90/2600 ms, Flip angle = 160°), (ii) fast spin echo (FSE: TR/TE = 5000/85 ms, Flip angle = 110°) and (iii) gradient-recalled echo (GRE: TR/TE = 1300/23 ms, Flip angle = 15°).
Radiological reporting of white matter hyperintensities
Scans were evaluated by a trained neuroradiologist within the following week after MRI acquisition and blind to the APOE-ɛ4 status of the participants. The grades of WMH were evaluated using modifications of the Fazekas Scale,
3
which separately categorizes the severity of deep and periventricular lesions, on a scale from 0 to 3 (0: None or a single punctate WHM lesion, 1: Multiple punctate lesions, 2: Beginning confluency of lesions [bridging] and 3: Large confluent lesions) (Figure 1). For data analyses, the variable was dichotomized, classifying participants into two groups, namely Fazekas score <2 and score ≥2, the latter usually considered as pathological in individuals younger than 75 years old.35–37
T2-FLAIR images illustrating the Fazekas Scores (A, 0; B, 1; C, 2 and D, 3).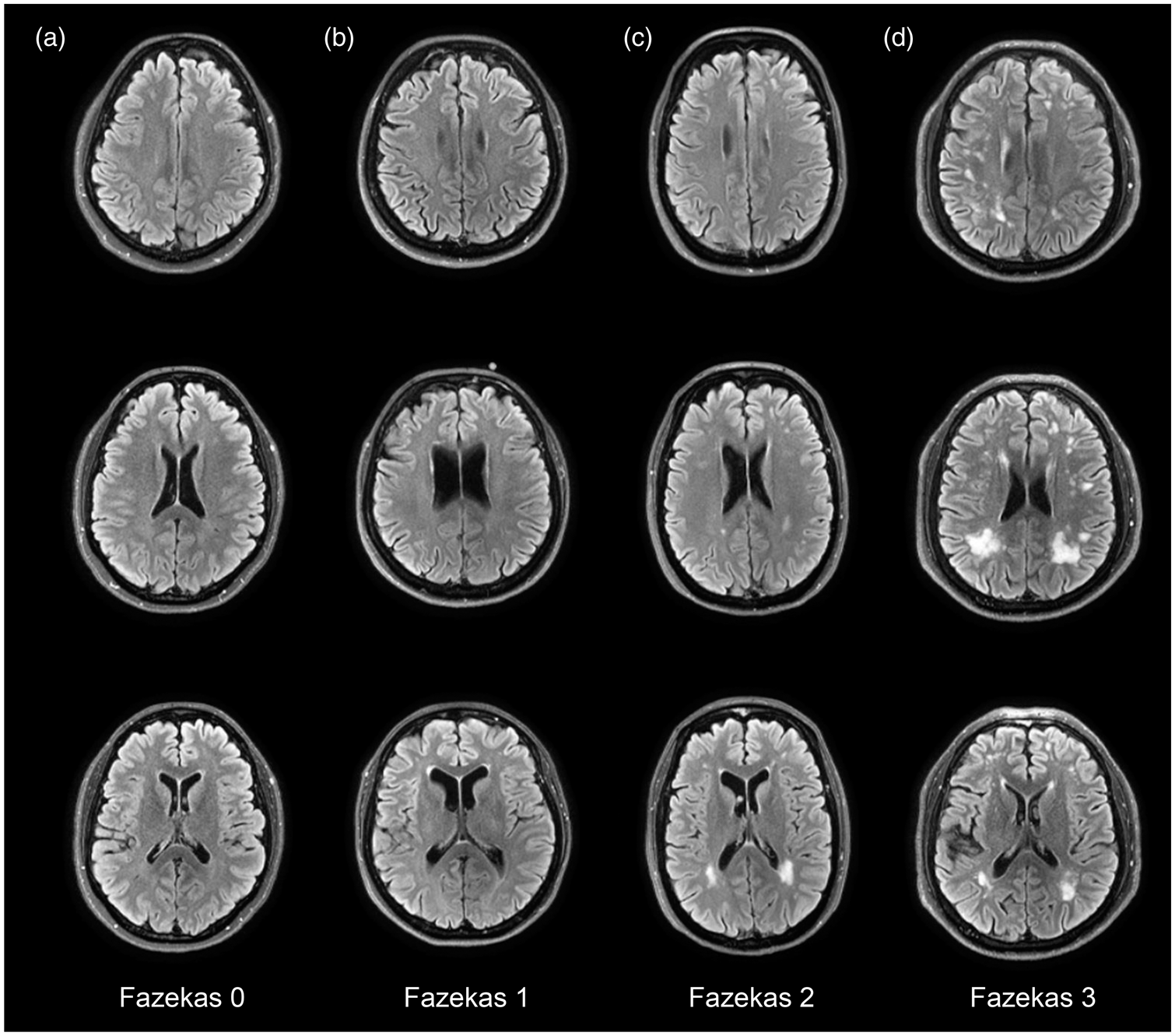
Statistical analyses
An initial descriptive analysis of the study sample was made and participants were divided into three groups according to the number of APOE-ɛ4 alleles of their APOE genotype. A between-group one-way ANOVA or a Kruskal–Wallis test (depending on the normality of the variables as assessed by a Kolmogorov-Smirnoff test; p < 0.05) was conducted to assess the effect of each of the variables.
A stepwise logistic regression analysis was conducted to predict the presence of a pathological Fazekas score (≥2). Participants’ APOE status was codified in three different ways that are based on (i) Model 1, the APOE haplotype (6 groups: “2/2”, “2/3”, “3/3”, “2/4”, “3/4”, “4/4”), (ii) Model 2, the number of APOE-ɛ4 alleles [3 groups: “0” (2/2, 2/3, 3/3); “1” (2/4, 3/4); “2” (4/4)] and (iii) Model 3, APOE-ɛ4 carriers/non-carriers [2 groups: “Non-carriers” (2/2, 2/3, 3/3); “Carriers” (2/4, 3/4, 4/4)]. Aside from the APOE-ɛ4 status, all models included the following predictors: hypertension, hypercholesterolemia, BMI and smoking habits, as well as sex, age and years of education as sociodemographic factors. A multinomial regression model to assess for the association of the Fazekas score as an ordinal variable was used (collapsing score = 3 with score = 2).
Regarding dementia risk estimates (CAIDE), a stepwise logistic regression analysis was also conducted, using the percentage of risk as a continuous variable. The analysis was performed for both CAIDE model I (model 4) and model II (model 5) and APOE status categorized as APOE-ԑ4/4 homozygote or not. The predictors introduced in Models 1–3 were not included here since they are already considered in the CAIDE function.
Finally, Model 6 represents a stepwise logistic regression analysis in which the estimated 10-year cardiovascular percentage of risk was used as a continuous variable. APOE status was categorized as APOE-ԑ4/4 homozygote or not. Education, the only predictor not included in the REGICOR function, was included in the analysis.
Initially, all predictors were introduced in the models along with all two-way interactions. Progressively, factors showing p values >0.1 were removed from the model except for sociodemographic factors. SPSS 17.0 for Windows was used for all the statistical analyses. Differences were considered to be statistically significant at p < 0.05 in all analyses.
Results
Characteristics of participants according to number of APOE-ɛ4 alleles.
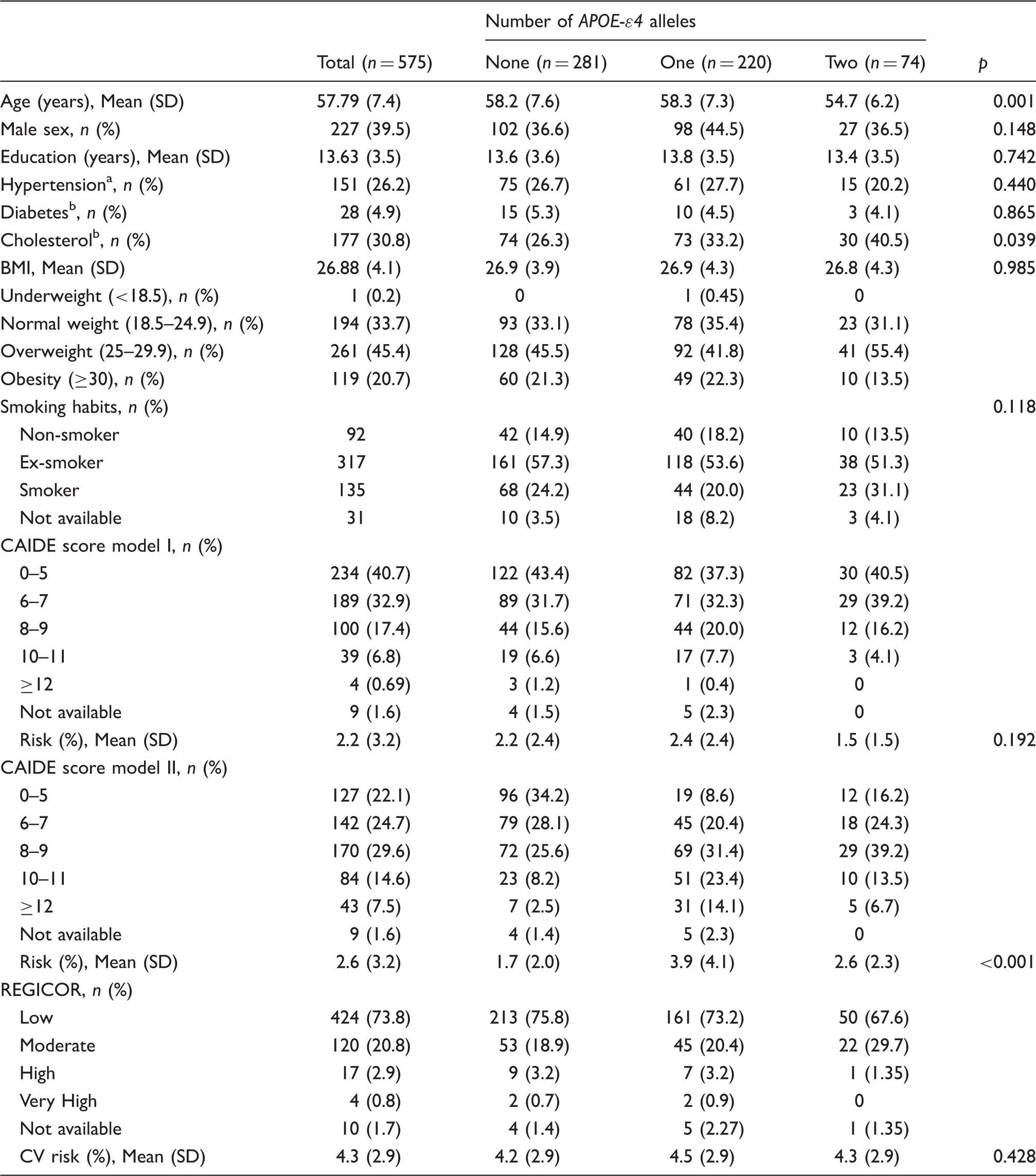
BMI: body mass index; CV: cardiovascular.
Systolic blood pressure ≥160 mm Hg and/or use antihypertensive medication and/or self-reported.
Medications and/or self-reported.
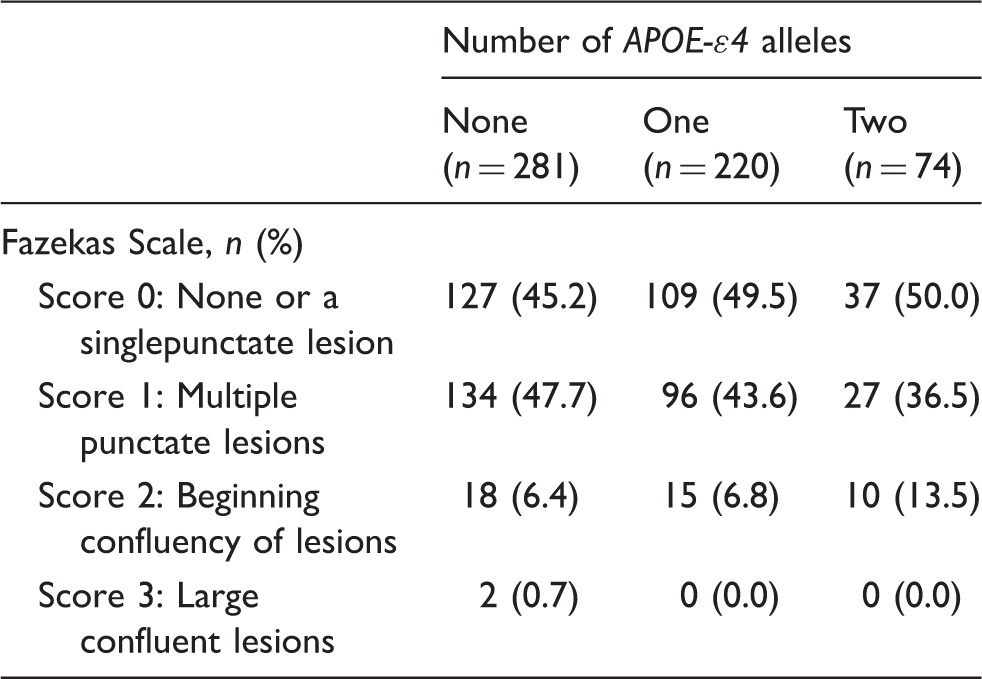
Distribution of Fazekas Scale score according to number of APOE-ɛ4 alleles.
Logistic regression models for the association of Fazekas score with APOE-ԑ4.
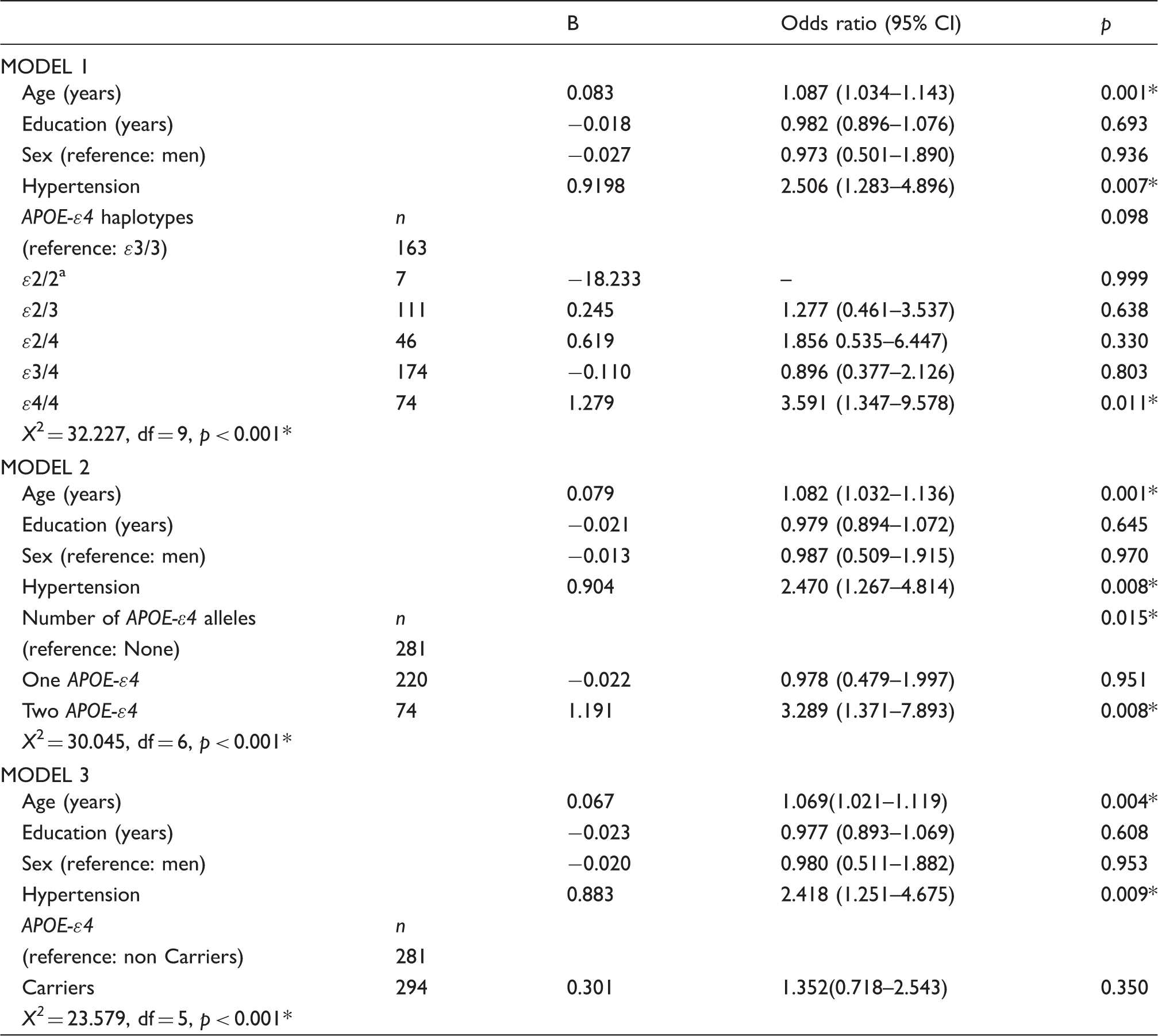
Was excluded due to small n.
p ≤ 0.005.
Logistic regression models for the association of Fazekas score with cardiovascular and dementia risk scores and APOE-ԑ4.
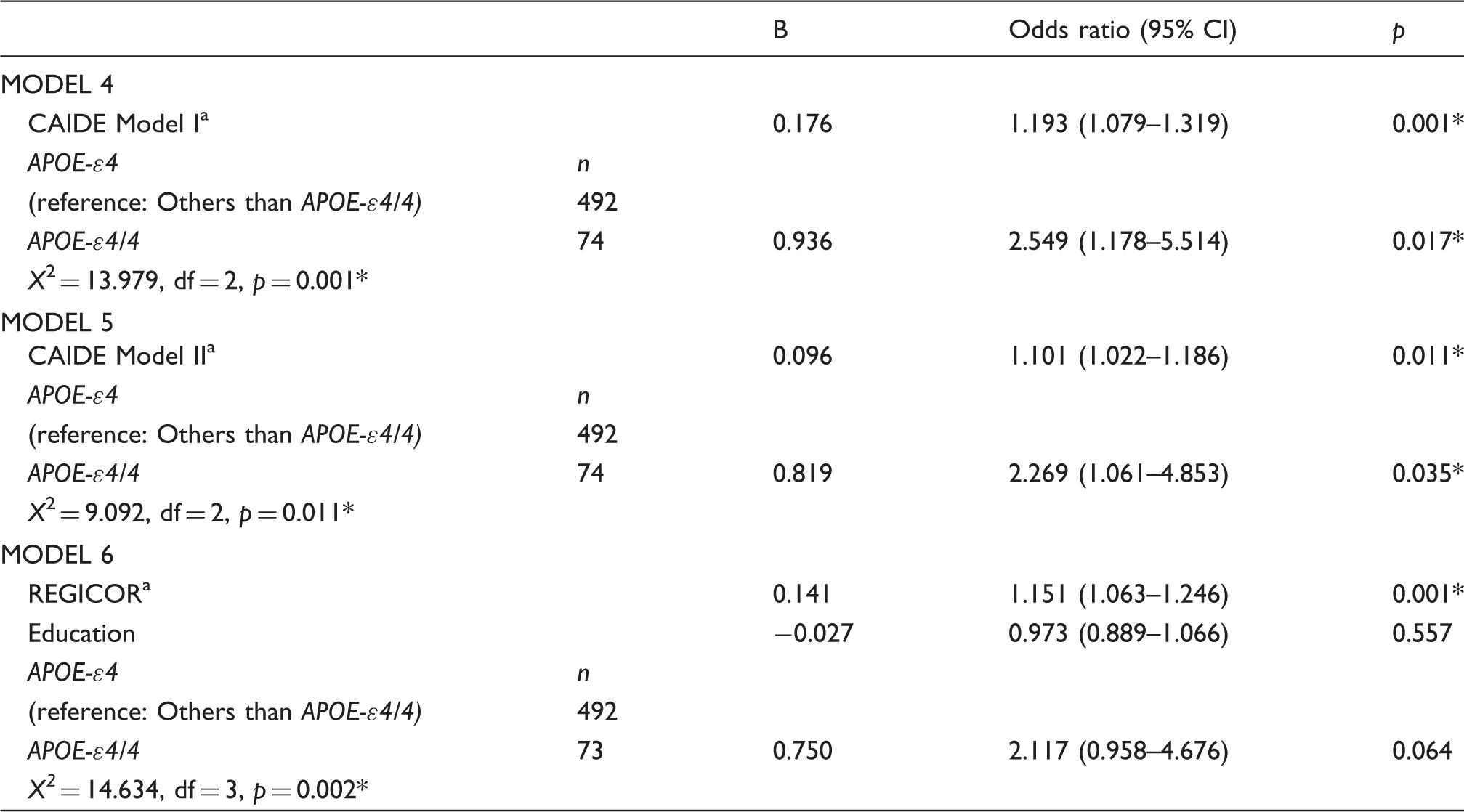
Percentage of cardiovascular risk.
p ≤ 0.005.
Discussion
In this work, we assessed the impact of the APOE genotype on the prevalence of pathological levels of WMH in a sample of cognitively healthy, late-middle-aged subjects and their potential interactions with vascular risk factors. To evaluate whether the number of APOE-ɛ4 alleles could have an additive effect on WMH, participants in this study were selected according to their APOE genotype, preferentially including APOE-ԑ4 allele carriers, among members of the ALFA parent cohort. This strategy resulted in a highly enriched sample for the APOE-ɛ4/4 genotype which allowed us to distinctly study the impact on WMH of APOE-ɛ4 heterozygosity and homozygosity as compared to non-carriers. Our main finding is that cognitively healthy late-middle-aged APOE-ɛ4 homozygous bear a significantly higher risk (OR 3.591; 95% Confidence Interval [1.347–9.578]; p = 0.011) of displaying pathological levels of WMH than non-carriers. On the other hand, the risk of APOE-ɛ4 heterozygous participants is not significantly different from that of non-carriers. As expected, age and hypertension were also positively associated to a pathological degree of WMH. Similarly, the observed association between presence of WMH with cardiovascular and dementia risk score estimates was also expected, in view of the existing literature.22,38,39 Interestingly, none of the CRF analyzed here (namely diabetes, hypertension, hypercholesterolemia, BMI and smoking habits) showed an interaction with APOE status, indicating that the effect of APOE on WMH is independent of these factors.
Several studies have evaluated the association between APOE genotype and the prevalence and severity of WMH. In fact, some articles could not find any differences in WMH load between APOE-ɛ4 allele carriers and non-carriers,40,41 but others have observed a clear correlation.29,42 These recruited subjects from general population, patients with cerebrovascular disease 43 or patients affected of probable AD. 44 There is, therefore, a great variability on the characteristics of subjects evaluated among studies, including age, ethnicity, comorbidities and other demographic factors that could have an impact on WMH. Consequently, it is not surprising that previous published works have reported contradictory results regarding the impact of the APOE genotype on WMH prevalence and severity. In general, those suggesting a possible role of the APOE-ɛ4 isoform in WMH studied large cohorts recruited from the general population. 30 Recently, the results of a meta-analysis that addressed this topic have also reinforced the idea that, at least in general population, APOE-ɛ4 allele carriers presented a higher risk of developing WMH. 26 By contrast, several studies that did not find any correlations were carried out in cohorts of patients affected from cerebrovascular disease or AD. Under these conditions, other factors that participate in the development of WMH, such as cardiovascular disease, could prevail over the effect of the APOE genotype and contribute to the observed lack of association. In our study, subjects with relevant medical pathology or neurologic disease were excluded. In consequence, our sample is healthier than could be expected from an age-matched cohort selected from the general population. Moreover, participants are younger in comparison with those in previous studies. Since comorbidities and age are risk factors to develop WMH, our selection criteria produced a cohort with a lower WMH load than others reported in the literature. The relatively low level of WMH pathology in our cohort might account for the lack of correlation in heterozygosis in our results. It could be speculated that an increased risk may be found in older APOE-ɛ4 heterozygotes.
Another important consideration to take into account is the difference in the ethnicity of the subjects between studies. The association between carrying APOE-ɛ4 alleles and WMH has been clearly observed in white Caucasians, thus it cannot be a priori generalized to other populations. 27 In this way, other studies carried out in Japanese patients did not observe a correlation between APOE-ɛ4 and WMH.40,45
Aside of the demographic differences between the studied cohorts, the low number of APOE-ɛ4/4 homozygotes undoubtedly represents the most relevant limitation in previous studies exploring the relationship between APOE-ɛ4 status and WMH. Very few studies have analyzed the correlation between WMH and APOE-ɛ4/4 homozygosity and the largest of them only included 21 homozygotes from a total of 1779 recruited subjects. In this work, APOE-ɛ4/4 homozygotes showed more WMH and an increased progression of the lesions during a five-year follow up period. In the same way, it was also found that WMH load of APOE-ɛ4/4 individuals is higher than that of their APOE-ɛ3/4 counterparts. 28 A recent report on more than 2000 participants aged between 20 and 90 years did not discover any association with the APOE genotype, purportedly because they were not able to analyse homozygotes separately (the number of homozygotes is not reported in the paper). 41 Therefore, the major strength of our study is the number of recruited APOE-ɛ4/4 homozygous (n = 74) which is, to our knowledge, considerably higher than any other single center study published to date. This enabled us to characterize the association between APOE-ɛ4 genetic load and WMH. In fact, no correlation with pathological WMH could be detected in our sample in association with APOE-ɛ4 when we pooled all subjects presenting at least one ɛ4 allele together. Nevertheless, it is worth mentioning that our sampling strategy was designed to strengthen the ability to detect associations for APOE-ɛ4 homozygotes. The limitation of this strategy is that the allele frequencies in our study are not representative of an unselected population, thus increasing the risk of bias and preventing us from drawing any epidemiological conclusions. In addition, even though the vast majority of the study participants are white Caucasians and, consequently, ethnically homogeneous, specific genotyping of ancestry informative markers in our study population has not been performed. Given that APOE alleles are highly stratified by ethnicity 46 even within white European populations, unmeasured social, cultural or genetic factors that vary by ethnicity and increase WMH could confound the observed associations.
On the other hand, a few studies have suggested an association between the APOE-ɛ2 allele and increased levels of WMH as well as with other vascular cerebral alterations. 47 Our observations do not support this hypothesis since no correlation was found between WHM and this allele. However, other vascular lesions such as microbleeds or lacunar infarcts were too scarce in our cohort to allow us to perform correlational analyses.
The increased WMH observed in APOE-ɛ4/4 individuals could be related to an increased tendency towards vascular pathology, a higher sensibility to risk factors, a reduced resilience to brain injury and a reduced recovery of lesions. 48 In any case, the accumulation of WMH could decrease the efficiency of subcortical white matter to perform its function and may have additive or even synergistic effects with other noxious processes acting in the brain like neurodegeneration in the cortical grey matter. 16 In the last instance, accumulation of WMH could facilitate cognitive deterioration and the onset of AD dementia.49,50 Hence, controlling WMH load in AD susceptible individuals could be a useful preventive strategy to reduce or delay cognitive decline. 51 In contrast with age or APOE genotype, other factors associated with WMH, like hypertension, are modifiable with changes in life style or pharmacological treatment. There is no doubt that the control of vascular risk factors is the way to go for all patients not only for these with increased risk of developing WMH. However, it could be theorized that a more strict control of these factors could particularly benefit the APOE-ɛ4/4 homozygotes. The same rationale could be considered for the development of treatments focused on reducing WMH load and progression. Such interventions could reduce the likelihood of cognitive deterioration and eventually the onset of dementia in APOE-ɛ4/4 homozygotes, which are at a higher risk of developing AD. 28
Cognitively healthy APOE-ɛ4 homozygotes have also been reported to show a significantly higher prevalence of cerebral amyloid pathology. At the age of 55, approximately 50% of APOE-ɛ4/ɛ4 individuals display abnormal levels of amyloid biomarkers, as compared to only 10% of APOE-ɛ3/ɛ3 persons and about 20% of carriers of a single APOE-ɛ4 allele. 52 Cerebral amyloid pathology has also been pinpointed as a relevant factor in the development of WMH in cognitively normal elderly individuals.53,54 In this regard, the main limitation of our work is that we could not assess the impact in the prevalence of WMH of cerebral amyloid pathology independently of that of APOE-ɛ4 status, hypertension and age.
Conclusions
Cognitively healthy, late-middle-aged APOE-ɛ4 homozygotes show a higher risk of presenting pathological levels of WMH as compared to their heterozygous and non-carrier counterparts. This association was found after controlling for well-established risk factors for the development of WMH such as age and hypertension and was also independent from other CRF. Given the known association between WMH and future cognitive decline, the control of modifiable risk factors in individuals at higher risk of developing WMH appears as a useful preventive strategy to reduce or delay the onset of dementia.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research leading to these results has received funding from “la Caixa” Foundation. Additional funding was obtained from Fondo de Investigación Sanitaria (FIS), Instituto de Salud Carlos III (ISC-III) under grant PI12/00326 and Barcelona city council under agreement #0724/13. Juan D Gispert holds a ‘Ramón y Cajal’ fellowship (grant no. RYC-2013-13054).
Acknowledgements
This publication is part of the ALFA study (ALzheimer and FAmilies). The authors would like to express their most sincere gratitude to the ALFA project volunteers, without whom this research would have not been possible.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
SR and AB-R contributed equally to this work. All authors listed (SR, AB-R, NB, CM, AT, CF, AC, SM, ME, NG, KF, JC, JLM and JDG) made a substantial contribution to the concept and design, acquisition of data or analysis and interpretation of data, drafted the article or revised it critically for important intellectual content and approved the version to be published.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
