Abstract
Electroconvulsive therapy (ECT), a direct form of brain stimulation, is an effective antidepressant. We hypothesized that the beneficial effects of ECT are mediated by increased dopaminergic neurotransmission, in which the baseline activity of D1 receptors may predict the response to ECT. We established a novel model of brain stimulation in Göttingen minipigs based on the protocol of ECT applied in humans. With positron emission tomography (PET), we determined a measure of dopaminergic neurotransmission with the dopamine D1 receptor antagonist [11C]SCH23390. Seven minipigs were anesthetized and completed PET at baseline, prior to the onset of ECT treatment, and at 24–48 h and 8–10 days after the end of a clinical course of ECT, consisting of 10 ECT sessions over a 3.5-week period. In all pigs, the binding of [11C]SCH23390 to striatal D1 receptors had increased by 24–48 h after ECT, and in most, binding returned towards baseline at 8–10 days. Increased binding was observed in inverse proportion to baseline binding rates. Increased binding to dopamine D1 receptors suggests facilitation of dopaminergic neurotransmission, which may contribute to the therapeutic effects of ECT. Importantly, the baseline binding capacity of D1 receptors predicts the magnitude of increased binding, up to a maximum binding capacity.
Introduction
Electroconvulsive therapy (ECT) is an effective and safe antidepressant treatment, with well-established indications, few contraindications, and low morbidity and mortality.1–3 The treatment is associated with transient memory disturbances, which in rare cases persist after treatment. 4 The effectiveness and relative benign side effect profile of ECT notwithstanding, little is known of the treatment’s mechanism of antidepressant action. Therefore, ECT usually is a late treatment option in depressive or psychotic disorders, or other conditions in which depression is common, such as Parkinson’s disease (PD).
Increased serotonin and noradrenaline release over a three- to four-week time period after initiation of therapy are hallmarks of positive antidepressant response and occur in a majority (70–80%) of patients receiving ECT, assumed to be associated with restored cortical inhibitory mechanisms.5–9 The role of the dopamine (DA) system in the therapeutic effects is less understood, despite a large number of preclinical studies showing increased monoaminergic neurotransmission. Studies of non-human primates in vivo by the present group of researchers reveal increased DAergic signaling, linked to increased receptor availability after ECT.10,11 In these studies, DAergic stimulation is the habitual response to ECT or electroconvulsive stimulation (ECS, the rodent equivalent of ECT), independently of the antidepressant effect. At first, the response of the DA system was observed in patients with PD, in whom the antiparkinsonian effect occurs almost immediately following ECT initiation.12–19 While it is held that DA plays a less prominent role than other monoamines in depression, DA stimulation may be of benefit, especially in patients with poor response to serotonin and noradrenaline-targeted antidepressant medication.
Pigs were tested extensively at the onset of ECT in pioneering work of Ugo Cerletti in the 1930s, but were replaced by rodents in more recent investigations. In the current study, we used the Göttingen minipig to test the hypothesis of (1) increased DA D1 receptor availability in response to ECT in large mammals subjected individually to clinical ECT, and (2) that baseline DA D1 binding levels may predict the individual response to ECT.
As a test of the hypothesis, with repeated tomographies, we determined the effects of a clinical course of ECT 24–48 h, 8–10 days, and 6–8 weeks after the end of the course, on accumulation of the labeled dopamine D1 receptor antagonist [11C]SCH23390 in tracer dose in vivo. Dopamine D1 receptors are confined to dendritic spines, 20 in which case the binding of the radioligand is an index of the number of dendritic spines available.
Methods
Animals
We studied seven adult Göttingen minipigs (female, 16 months old, 23–29 kg) from Ellegaard Minipigs ApS (Dalmose, Denmark) in accordance with a protocol approved by the Danish Animal Experiments Inspectorate and their guidelines. We reported data in compliance with the ARRIVE guidelines. Minipigs were fed a restricted pellet diet (Special Diets Services (SDS), Aarhus, Denmark) and fasted prior to the study with continuous access to tap water. Environmental conditions in the animal facility were 20℃ and 50–55% relative humidity, the photo-period was not controlled (12 h light), and the air was changed eight times every hour. All minipigs were clinically healthy prior to the study.
ECT treatment
Here, we used the term ECT to differentiate the chosen clinically inspired design from ECS applied daily to rodents by ear clips, without anesthesia. With the ECT design, we pre-anesthetized the seven minipigs with increasing doses of s-ketamine ranging from 10 to 20 mg/kg intramuscularly. We induced deep anesthesia with intravenous thiopental (4.5 ug/kg), followed by administration of succinyl choline (suxameton, 1 mg/kg) into an ear vein catheter (21G Venflon) for muscle relaxation immediately prior to application of ECT. ECT treatments were given with a clinical ECT device (Thymatron, frequency: 70 Hz, pulse width: 0.5 msec, current: 0.9A). We placed electrodes bilaterally (with electrogel) as this provides the most effective response in human subjects. In this feasibility study, we measured the duration of generalized seizures with a stopwatch by visual observation of the animal, as previously used to evaluate seizure duration in rhesus monkey, 21 since repeated accurate measurement of EEG activity with surface electrodes in pigs is complicated by skull thickness, a large sinus, and significant fat and muscle mass above the skull. The clinical course consisted of 10 treatments every other weekday morning (Monday, Wednesday and Friday), over a three- to four-week period, thus mimicking the treatment given to humans. Seven animals underwent PET before the onset of ECT treatment and at 24–48 h and 8–10 days after the completion of ECT. Three of the pigs were scanned again 6–8 weeks after ECT.
Tomography
Minipigs were scanned as previously described. 22 We pre-medicated the minipigs with a mixture of 1.25 mg/kg midazolam and 6.25 mg/kg s-ketamine IM. After placement of an ear vein catheter (21G Venflon), we induced anesthesia with a mixture of 1.25 mg/kg midazolam and 3.13 mg/kg s-ketamine IV. We then intubated the minipigs and maintained the anesthesia at 2% isoflurane. The minipigs were mechanically ventilated with approximately 8 mL/kg/min of a mixture of 1 part O2 and 2.2 parts medical air. Pulse, arterial oxygen (SaO2) and body temperature were monitored throughout the tomography session and maintained at normal values for minipig 23 . Hydration was maintained with a saline drip. We positioned the minipigs with the brain in the center of the field of view of the High-Resolution Research Tomograph (HRRT, CTI/Siemens) in a dorsal recumbent position, with the head immobilized by a custom device. A heating blanket set to maintain a stable body temperature was used to cover the pigs.
Average (standard deviation) of injected dose, specific activity and injected mass of [11C]SCH23390.

Data analysis
A summed PET image of the entire scan was co-registered to an average magnetic resonance image (MRI) atlas of the minipig brain
24
using Montreal Neurological Institute PET imaging software. We chose striatum as the region of interest and the cerebellum as the reference region devoid of D1 receptors. Because of the linear increase of the target-to-reference ratio of the accumulation of [11C]SCH23390 in the minipigs over the acquisition time of 90 min,
25
we analyzed the binding by a version of the Gjedde-Patlak analysis of the accumulation of radioligands26,27 and used the cerebellum as a reference region. The target-to-reference ratio analysis as function of time yielded the unidirectional binding rate constant
Statistics
We tested the baseline, 1–2-day, and 8–10-day data, with one-way repeated measures analysis of variance (ANOVA) using GraphPad Prism 6.07, followed by post-hoc Dunnet tests, comparing the means of the two post-ECT datasets to baseline values with control for multiple comparisons. The six-week data were not included in the analysis and are shown only for information. The percentage changes between 1–2 days post-ECT and baseline were calculated as 100×(Post-baseline-Baseline)/Baseline and evaluated as a function of baseline.
Results
The accumulation of [11C]SCH23390 increased significantly in the striatum of healthy animals at 1–2 days after ECT. Representative images from one animal are shown in Figure 1(a). Figure 1(b) shows the average binding rate constant (a) Representative parametric maps of [11C]SCH23390 accumulation at baseline vs. at 24 h post ECT in pig 4. (b) Longitudinal changes in uptake rate for individual animals in response to ECT. The data from each animal are represented in a different color.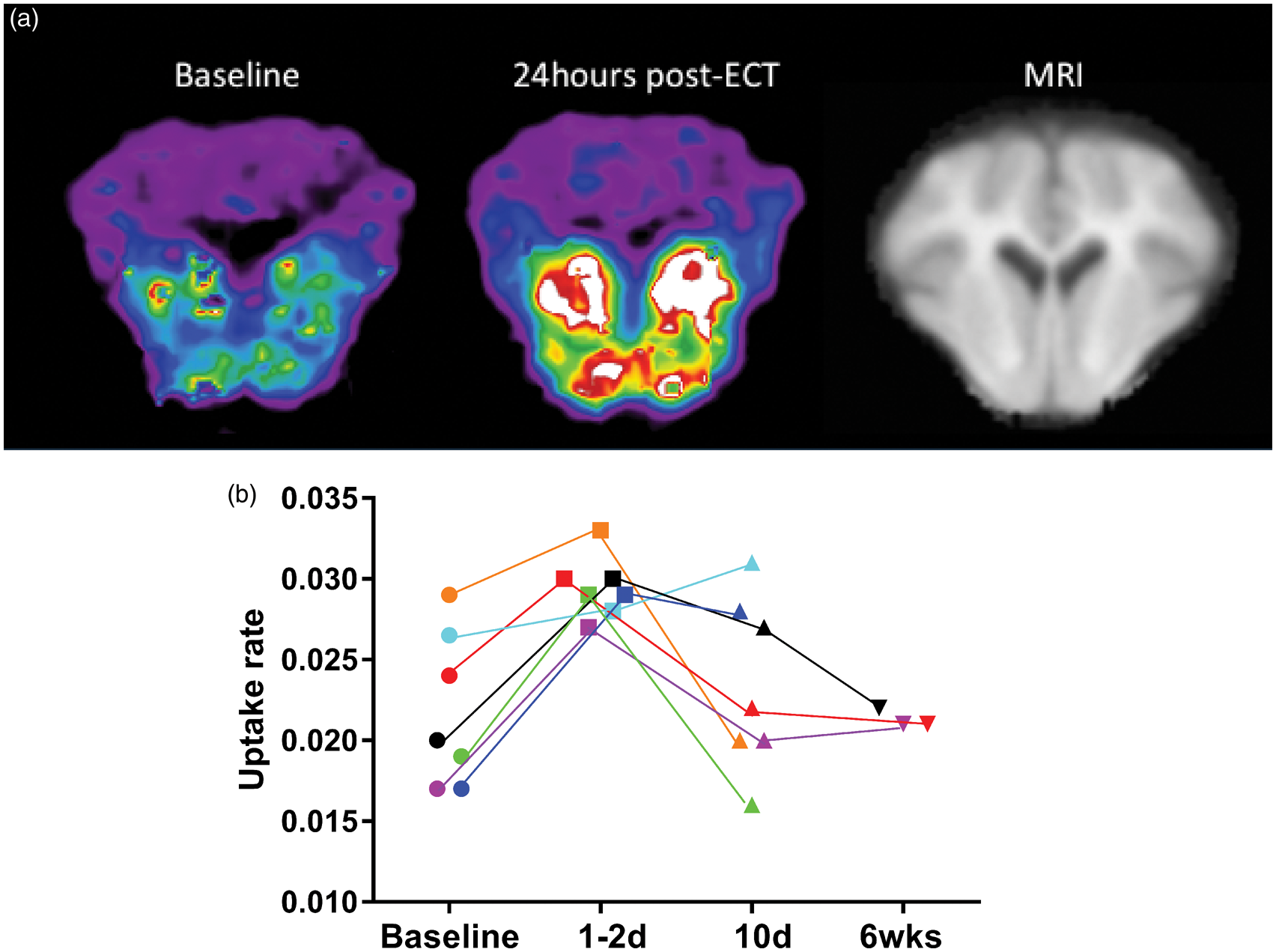
There was an inverse linear relationship between the percentage change at 24–48 h and the baseline value (r2 = 0.92). The animals with the lowest baseline striatal binding rates of [11C]SCH23390 were associated with the greatest increase after ECT, as shown in Figure 2. In Figure 2, the abscissa intercept identifies the theoretical baseline binding rate at which no further increase of binding capacity would be expected.
Percent change at 1–2 days post-ECT as a function of baseline uptake rate.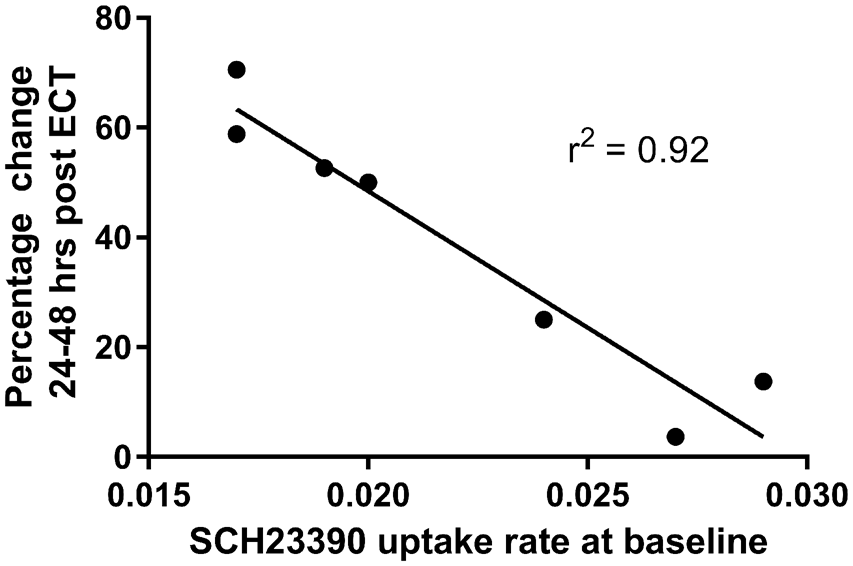
Discussion
We tested the hypothesis that longitudinal effects of ECT are associated with changes of the numbers of postsynaptic DA D1 receptors on dendritic spines. We determined an index of the binding capacity of the receptors with PET of the binding rate of the tracer [11C]SCH23390 that is specific for postsynaptic DA D1 receptors. We previously demonstrated significant increases in pre-synaptic re-uptake and vesicle transporter (DAT and VMAT2) availabilities in the striatal areas in ECT-treated non-human primates as well as increases in striatal SCH23390 binding. 10 In the present study, we tested the effect of the treatment on the binding capacity of post-synaptic dopamine D1 receptors in another mammalian species, the Göttingen minipig. In pigs, the binding kinetics of the tracer differs from that of primates: while the tracer binds reversibly to the receptors during the tomography duration chosen for primates, we previously demonstrated that, in pigs, the binding of SCH23390 is relatively irreversible over the tomography duration. 25 This different kinetics in pigs allowed us to obtain an index of the binding rate and hence of the receptor numbers in brain regions. The findings indicate that minipigs treated with a clinical course of ECT had significant increase of the rate of binding of [11C]SCH23390 in the striatum, and hence of the D1 receptor binding capacity, at 24–48 h after the last ECT session.
Multiple lines of evidence suggest that impaired DA neurotransmission plays a role in depression, as acutely depressed patients display a 13% reduction in D1 receptor binding in the striatum, 28 and neuroimaging with SPECT reveals lower baseline levels of DA in depression. 29 In humans, DA facilitating drugs such as bupropion and moclobemide, in combination with classical antidepressants, improve clinical outcomes, 30 while DA antagonists accentuate depressive symptoms. 31 Transcranial magnetic stimulation, another form of brain stimulation with potential antidepressant actions, modulates DA release. 32
Previous studies support the hypothesis that ECT stimulates DA function.8,33–35 Microdialysis studies in rodent following ECS show increased interstitial striatal DA release. 33 Notably, ECT also produces improvements in the motor deficits of PD. In depressed PD patients, a rapid amelioration of the motor symptoms is observed almost immediately after initiation of a clinical course of ECT prescribed for the treatment of the depressive comorbidity and well before any antidepressant effects.15,38 Consistent with this, a course of ECS in rats improved gross motor function 8 and induced DA stereotypic behaviors, 9 suggesting a very rapid and early effect of ECS on the DA system. We found an increase in D1 binding in the days following the end of the ECT course, which is in agreement with several studies in rodents and one in non-human primates showing increases in DA D1 receptor binding in the normal striatum in response to ECT or ECS regardless of administration parameters.8,10,37–40
With the currently available ligands (SCH23390 and NCC112), the D1 receptor system is characterized by low affinity of D1 receptors and thus is not affected measurably by the DA concentrations encountered in this experiment. The reason for the changes in D1 receptor expression in response to ECT/ECS is not completely understood but is believed to result from increases in DA transmission due to plasticity associated with changes of the dendritic spines where the D1 receptors reside, as compensation for the evidence of decreased D1 binding in DA depleted animals and vice versa.41–43–45. The dopaminergic fibres in striatum are now held to specifically support the function of the dendritic spines, including the receptors that change with the spines.
Increased serotonergic and noradrenergic transmission is a widely accepted hallmark of effective antidepressant treatment in about two-thirds of ECT-treated depressed patients as well as in healthy non-human primates 21 and Göttingen minipigs (Doudet et al., 2016, in preparation). A comparable proportion of minipigs demonstrated increased noradrenergic transmission in response to another antidepressant brain stimulation therapy, vagal nerve stimulation. 46 We consistently observed stimulation also of DA transmission in every animal of the current study of ECT-treated pigs and in non-human primates, 10 independently of the serotonergic or adrenergic responses.
The effects of ECT on the DA system waned after six weeks in three animals studied at that length. The biphasic response raises the issue of the relationship between the neurochemical effects of ECT and the persistence of the therapeutic effect. Similarly, transient effects of ECT on 5HT2 receptor binding were observed in non-human primates. 21 It is well known that both antidepressant and antiparkinsonian effects tend to be transient, lasting in most cases for weeks to months, with relapse in most patients within six months, prompting the addition of pharmacological or ECT maintenance therapy for long-term efficacy. Individual differences of the neurochemical state to which ECT is applied may contribute to the large variations of the therapeutic effect and its persistence among individual subjects.
The inverse proportionality between effect size and baseline in terms of the binding capacity in this study appears to be a common finding in studies of dopaminergic neurotransmission, evident in measures of changes of general receptor availability in response to stimulation. While acute changes of receptor availability are likely to be the result of changes of monoamine release, the more protracted changes, such as the ones seen in the present study, are more likely to represent an actual change of the number of receptors, in part or in whole due to changes of dendritic spine sizes or numbers or both. The maximum binding capacity to which the changes due to the ECT appeared to aim in the present study, suggests that the plasticity induced by the ECT has a ceiling effect that limits the response. It also appears that the increase of DA D1 receptor binding capacity is a consequence of plasticity mechanisms that are the result of release of one or more other monoamines.10,11,21,46
In summary, a clinical course of bilateral ECT in ECT-naïve minipigs increases dopamine D1 binding capacity, a response which may be associated with antidepressant effects in patients with depression or PD. We provided evidence that the baseline binding capacity of D1 receptors in the minipigs predict the magnitude of increased binding, up to a maximum binding capacity.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Danish Medical Research Council and Parkinson Society Canada provided postdoctoral fellowships to AML.
Footnotes
Acknowledgements
We are grateful for the technical support from the staff at the Aarhus University Hospital PET Centre. Special thanks to Kim Vang for his invaluable assistance with image analysis, and the staff at the Aarhus University Farm for help with the treatment of the animals.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
AML, GW, and DD designed the study. PV, GW, and DD participated in the initial ECT setup and AML, AKOA, MS and AM performed the ECT. HA and SJ synthesized the tracers for the PET study while AML and AKOA scanned the animals. AML, AG, and DD performed the PET analysis, and AG and DD did the statistical analyses. AML wrote the first draft of the manuscript with support from AG and DD, and all authors have approved the final version of the manuscript.
