Abstract
A characteristic feature of complex healthy biological systems is the ability to react and adapt to minute changes in the environment. This ‘complexity’ manifests itself in highly irregular patterns of various physiological measurements. Here, we apply Multiscale Entropy (MSE) analysis to assess the complexity of systemic and cerebral near-infrared spectroscopy (NIRS) signals in a cohort of 61 critically ill preterm infants born at median (range) gestational age of 26 (23–31) weeks, before 24 h of life. We further correlate the complexity of these parameters with brain injury and mortality. Lower complexity index (CoI) of oxygenated haemoglobin (HbO2), deoxygenated haemoglobin (Hb) and tissue oxygenation index (TOI) were observed in those infants who developed intraventricular haemorrhage (IVH) compared to those who did not (P = 0.002, P = 0.010 and P = 0.038, respectively). Mean CoI of HbO2, Hb and total haemoglobin index (THI) were lower in those infants who died compared to those who survived (P = 0.012, P = 0.004 and P = 0.003, respectively). CoI-HbO2 was an independent predictor of IVH (P = 0.010). Decreased complexity of brain signals was associated with mortality and brain injury. Measurement of brain signal complexity in preterm infants is feasible and could represent a significant advance in the brain-oriented care.
Introduction
Brain injury in the preterm infant is associated with an increased risk of poor developmental outcome and death.1,2 The preterm brain is particularly susceptible to intraventricular haemorrhage and haemorrhagic parenchymal infarction, usually occurring within the first 3 days of life. This results from bleeding in the germinal layer surrounding the lateral ventricles; this is an area of high cellular proliferation in the developing foetal brain, where neuron and glial cells precursors arise. The rich vasculature of the germinal matrix is formed by thin vessels, which are very sensitive to hypoxia and changes in cerebral perfusion pressure. 2 Several studies have shown that fluctuations in systemic and cerebral blood flow have been associated with the pathophysiology of ischemic and haemorrhagic brain lesions in preterm infants.3–5 In clinical practice, strategies for preventing brain injury in the preterm population have emphasized the importance of measurements of systemic blood flow and cardiac function; more recently, early monitoring of cerebral blood flow and oxygenation has been investigated, but has yet to be established as the standard of care.6,7 Near-infrared spectroscopy (NIRS) has been applied as a non-invasive monitor to measure cerebral oxygenation. Conventional NIRS measures the changes in the concentration of oxygenated haemoglobin (HbO2) and deoxygenated haemoglobin (Hb). Spatially resolved NIRS is able to provide a quantitative index of cerebral oxygen saturation – the tissue oxygenation index (TOI) and a semi-quantitative index of cerebral blood volume – the tissue haemoglobin index (THI). 8 Conventional and spatially resolved NIRS have been used to investigate cerebral haemodynamics in both adults and newborn infants.9–11 Assuming a low and constant cerebral metabolic rate for oxygen (CMRO2), TOI can also be interpreted as a surrogate measurement of cerebral blood flow and has been extensively used in neonatal research to investigate brain perfusion or cerebral autoregulation.12–16 More recently, studies have demonstrated that it is possible to stabilize cerebral oxygenation and monitor cerebrovascular reactivity using NIRS, supporting the idea that early monitoring of cerebral blood flow could have a positive impact on outcome of preterm infants.6,17
Most studies using cerebral NIRS or systemic measurements of blood flow use linear analysis. However, a complex biological system, such as the human brain, includes many complex regulatory mechanisms that interact in a non-linear way, resulting in effects that cannot be understood wholly through the analysis of its individual constituents. Therefore, non-linear methods to assess physiological signals may reveal more accurately the magnitude of changes over time. A complex biological system has the ability to react and adapt to minute changes in its environment. When the complexity of a system is reduced or lost, a pathological status is potentially present. Loss of complexity has been observed in several diseases and ageing.18,19 It has also been associated with poor outcome in severely sick adults and children.20,21 A suggested method of assessment of the complexity of biological signals in healthy and pathological systems is the Analysis of Entropy, a non-linear measurement of system randomness and unpredictability. In the early 1990s, Approximate Entropy was used first to assess the irregularity of biological time series and has been extensively used in physiological and medical studies. 22 A decade later, Sample Entropy was proposed as a modified method, which was less dependent on the length of the time series. In neonates, Sample Entropy analysis of heart rate variability (HRV) was used to predict the onset of neonatal sepsis 24 h before clinical diagnosis. 23 More recently, Multiscale Entropy (MSE) has been used to provide a more meaningful measure of complexity, exploring calculations of Sample Entropy over multiple time scales. MSE shows that correlated random signals are more complex than uncorrelated random signals (white noise). Moreover, MSE describes pathological physiology better than simple entropy measures, doing a better differentiation between healthy, complex systems and deranged or random interactions. 24 MSE has been used to assess HRV and electroencephalogram (EEG) changes in patients with different neurological conditions. 25 Complexity of intracranial pressure (ICP) measured using MSE has been correlated with clinical outcome. In adults with traumatic brain injury, reduced complexity of mean ICP was associated with poor outcome and increased mortality. 26 In infants, MSE was significantly reduced in infants with neonatal seizures and later diagnosis of epilepsy when compared to control infants. 27
To date, there are no previous studies using MSE to assess complexity of NIRS signals in preterm infants. Here, we hypothesize that MSE can be applied to study the complexity of cerebral NIRS signals and the complexity of systemic physiological signals in a cohort of preterm infants undergoing intensive care. We further hypothesize that the complexity index (CoI) of cerebral NIRS signals and systemic signals, measured using MSE, correlate with intraventricular haemorrhage and mortality in this population.
Methods
Ethics
This prospective observational study was authorized by The Research and Development Department of Cambridge University Hospitals NHS Foundation Trust and approved by The East of England Research Ethics Committee (12/EE/0524), in accordance with the declaration of Helsinki. The data included in this study were collected at the Neonatal Intensive Care Unit (NICU) at The Rosie Hospital, a busy tertiary unit that receives the most complex antenatal and postnatal cases from across the east of England region. Data were collected from September 2010 to May 2015. All infants were studied following signed informed parental consent.
Population
Demographic data.

Characteristics of enrolled infants and differences between no-IVH (intraventricular haemorrhage) and IVH groups and between infants who survived and died. Gestational age, birth weight and CRIBII (clinical risk index for babies II) are presented as median (range). Gender is presented as absolute numbers. Remaining variables are presented as frequencies.
P < 0.05.
Measurements
Following enrolment, a NIRS sensor from a NIRO 200NX near-infrared spectrophotometer (Hamamatsu Photonics, KK, Japan) was placed on one side of the infant’s head in the temporoparietal area. The NIRO 200NX is a bedside NIRS monitor that measures changes in the concentration of HbO2 and Hb using conventional NIRS, calculated using the modified Beer–Lambert Law, as well as providing measurements of TOI and THI using spatially resolved NIRS, calculated using an approximation of the light diffusion equation. The NIRO 200NX uses three light-emitting diodes with wavelengths of 735, 810 and 850 nm, respectively, and two detecting photodiodes, to measure light attenuation at different distances from the source. A sensor holder was used to fix the light source at 3 cm away from the receiver diodes and adhesive paper secured the sensor to the infant’s skin. A light-proof cover was also used. The sensor was changed to the opposite temporo-parietal side of the infant’s head every 6 to 8 h to avoid skin marks.
The following systemic physiological signals were recorded simultaneously and continuously from the neonatal intensive care monitors (Solar 8000; Carescape B850, GE Healthcare, Milwaukee, Wisconsin): full resolution of arterial blood pressure waveform (120 Hz, Solar, or 100 Hz, Carescape) peripheral oxygen saturation (SaO2) and heart rate (HR) (1 Hz). NIRS signals were sampled at the rate of 1 Hz. Both NIRS and physiological signals were collected, synchronized and stored using ICM+ software (Cambridge Enterprise Ltd, Cambridge, UK). 28
Clinical data within the study period and data on outcome were collected from the medical notes. Clinical decision-making was at the sole discretion of the attending neonatal consultant and included the management of blood pressure and treatment of sepsis. All infants were screened for sepsis and antibiotics started within the first hour after admission to the neonatal unit. Those with no clinical signs of sepsis, normal c-protein reactive (CRP), negative blood culture or antibiotics given only for the first 48 h of life were considered to have a negative sepsis screen. Those with presumed sepsis (CRP moderately high, negative culture and antibiotics for 5–7 days), clinical sepsis (high CRP, clinically unwell, negative culture and antibiotics for 7–14 days) or confirmed sepsis (clinical sepsis with positive culture) were considered to have a positive screen. CRIB II (Clinical Risk Index for Babies II) score of mortality and morbidity was calculated for all infants in the study. 29 Cranial ultrasonography scans were performed at the start of the study and repeated every 12–24 h until third of life and then every 1–3 weeks until corrected gestational age at term. IVH was defined according to Papile et al. and the greatest grade of haemorrhage during the admission period was used for analysis. 30
Data pre-processing
Data collected at the full frequency resolution supported by respective monitors was then retrospectively analysed using ICM+. 28 Artefacts had been identified and manually removed. Most of the artefacts were the result of umbilical arterial line sampling, infants being handled or movements. Nurses used a separate study chart to record date and time of events like cares, examination, procedures, changes on drug infusions or any other intervention. These annotations allowed experienced investigators to exclude manually periods of data with invalid measurements or with transients induced by external interventions. To date there is no universal, validated method for automatic robust removal of data artefacts.
Subsequently, all the recorded physiological variables were treated with the moving average filter of 10-s length and downsampled to frequency of 0.1 Hz. This has effectively removed all the components associated with pulse and respiratory activities while retaining waves in the low and very low frequency range (<0.02 Hz), thus focusing further analysis entirely on the cerebrovascular and systemic regulatory responses. These time series were then entered into the MSE analysis in order to produce complexity indices of the studied variables.
MSE analysis
MSE analysis was performed using algorithm published originally by Costa et al.24,25 Briefly, the analysis can be divided into three steps: constructing coarse-grained time series, computing sample entropy for each of them and calculating overall complexity. 31 Coarse-grained time series for scale τ is derived from original time series by averaging its τ samples in a moving (non-overlapping) window. Thus, time series for scale τ is τ times shorter than original time series. Particularly, for scale 1, we get simply original time series. For each coarse-grained time series, sample entropy was computed. Sample entropy estimates the probability that similar sequences of m consecutive points in time series will also be similar when sequences of the length m + 1 are considered. To be precise, two sequences containing the same number of points are considered similar if the absolute differences between their corresponding points are smaller than a specified tolerance value r. Sample entropy is calculated as the negative natural logarithm of the ratio of the total number of (m + 1)-point template matches to the total number of corresponding m-point template matches. 32 Higher values of sample entropy indicates that time series is more irregular and less predictable. We assumed m = 2 and r = 0.15 SD, where SD denotes standard deviation of analysed time series, as originally proposed by Costa et al. 24
MSE was calculated up to scale 20.25,26 It can be presented as a function of scale factor (Figure 2). The area under MSE curve, or in other words, the sum of sample entropy values for all scales, was defined as the CoI.26,31
Statistical analysis
Data were assessed for normality using the Shapiro–Wilk test and analysed using parametric or non-parametric statistical methods as appropriate. Comparisons between groups of infants with IVH and no-IVH, and between infants who died or survived, were made using the Student t-test, Mann–Whitney or Chi-square test, depending on data distribution. Binary logistic regression analysis was used to ascertain independent effects of the complexity of NIRS signals on the likelihood of the infants developing IVH. Spearman’s rank correlation coefficient (rs) was used to correlate the complexity of brain and systemic signals. A statistical test was considered significant if P value was < 0.05 (two-tailed). Statistical analysis was performed using the SPSS software package version 23 (SPSS, Inc., Chicago, IL, USA).
Results
Age at death and causes related to death.
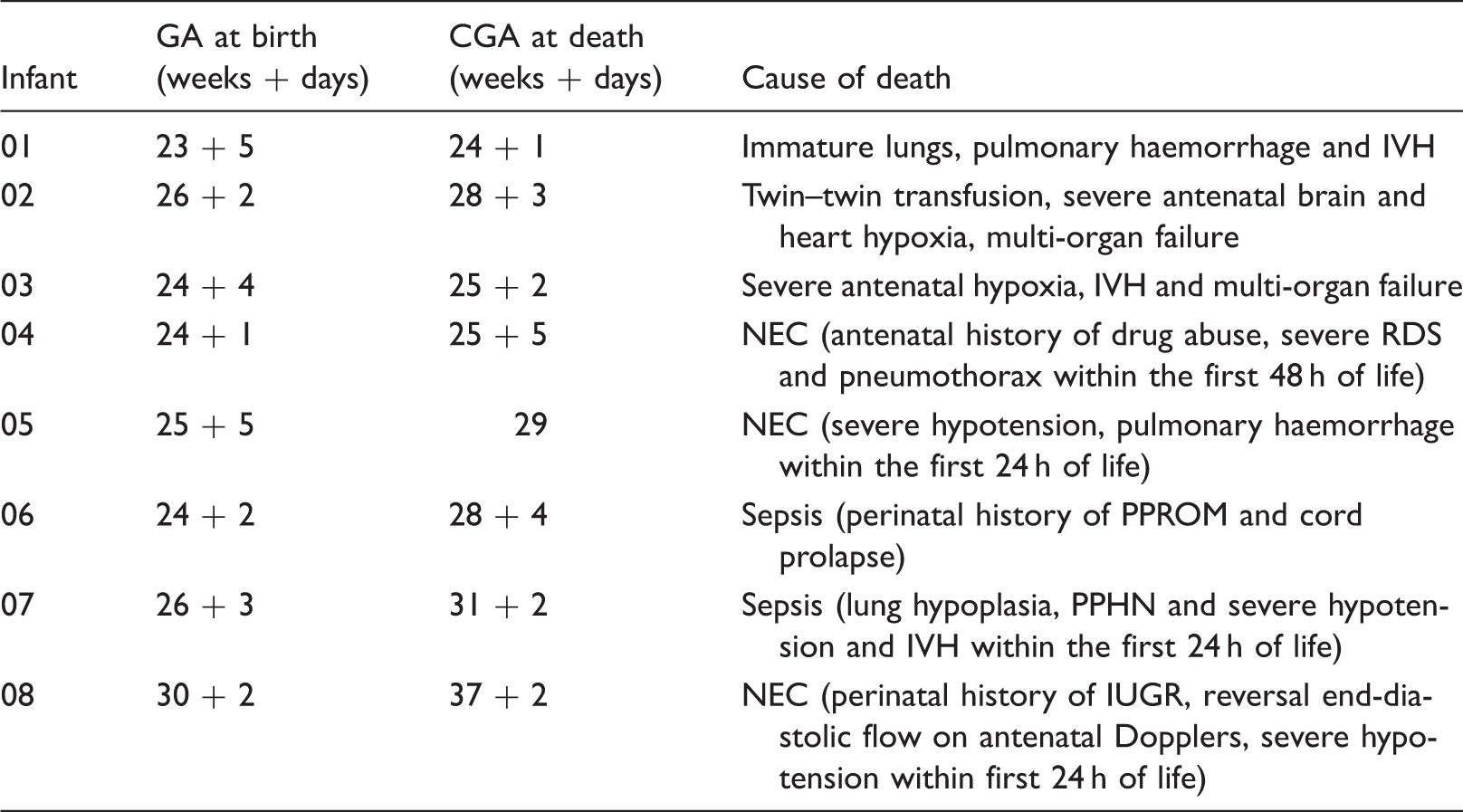
IVH: intraventricular haemorrhage; NEC: necrotizing enterocolitis; PPROM: preterm prolonged rupture of membranes; PPHN: persistent pulmonary hypertension of the newborn.
Examples of monitoring of brain and systemic signals are presented in Figure 1(a) (infant who died) and Figure 1(b) (infant who survived).
Example of brain and systemic signal monitoring for an infant who died (a) and survived (b).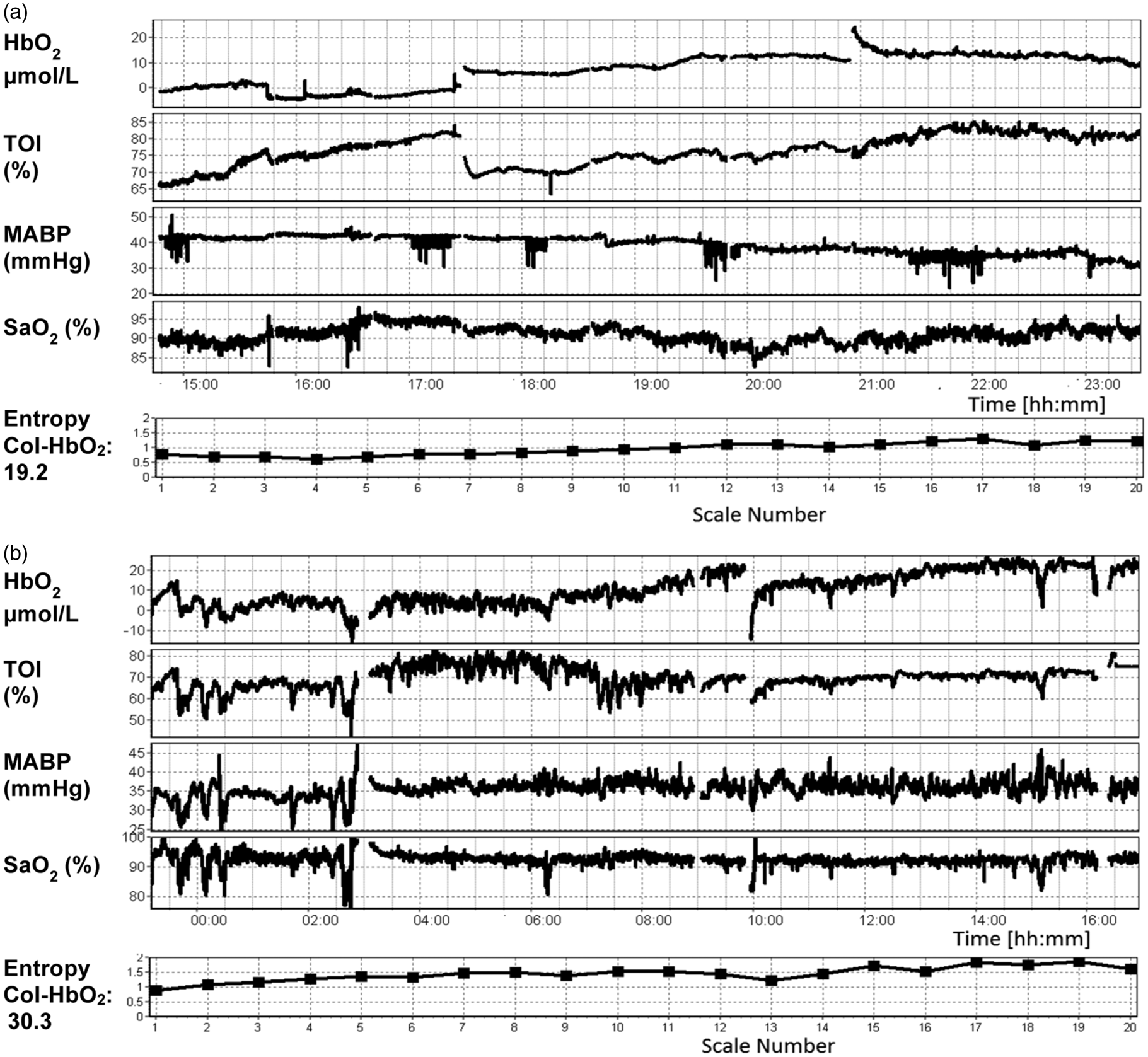
Figure 2 shows average MSE plots of HbO2 and Hb for the whole cohort of infants. Sample Entropy values were averaged individually for each scale and plotted against the scale. This was done separately in groups of those infants who had IVH and did not have IVH, as well as those infants who died and survived. It can be observed through the lower value of sample entropy at each scale factor, resulting in a decrease in overall CoI, that infants who developed IVH or died had consistently lower entropy in the NIRS signals.
Multiscale entropy. Results are mean ± 1.96 standard error of mean. Oxygenated haemoglobin (HbO2) and deoxygenated haemoglobin (Hb).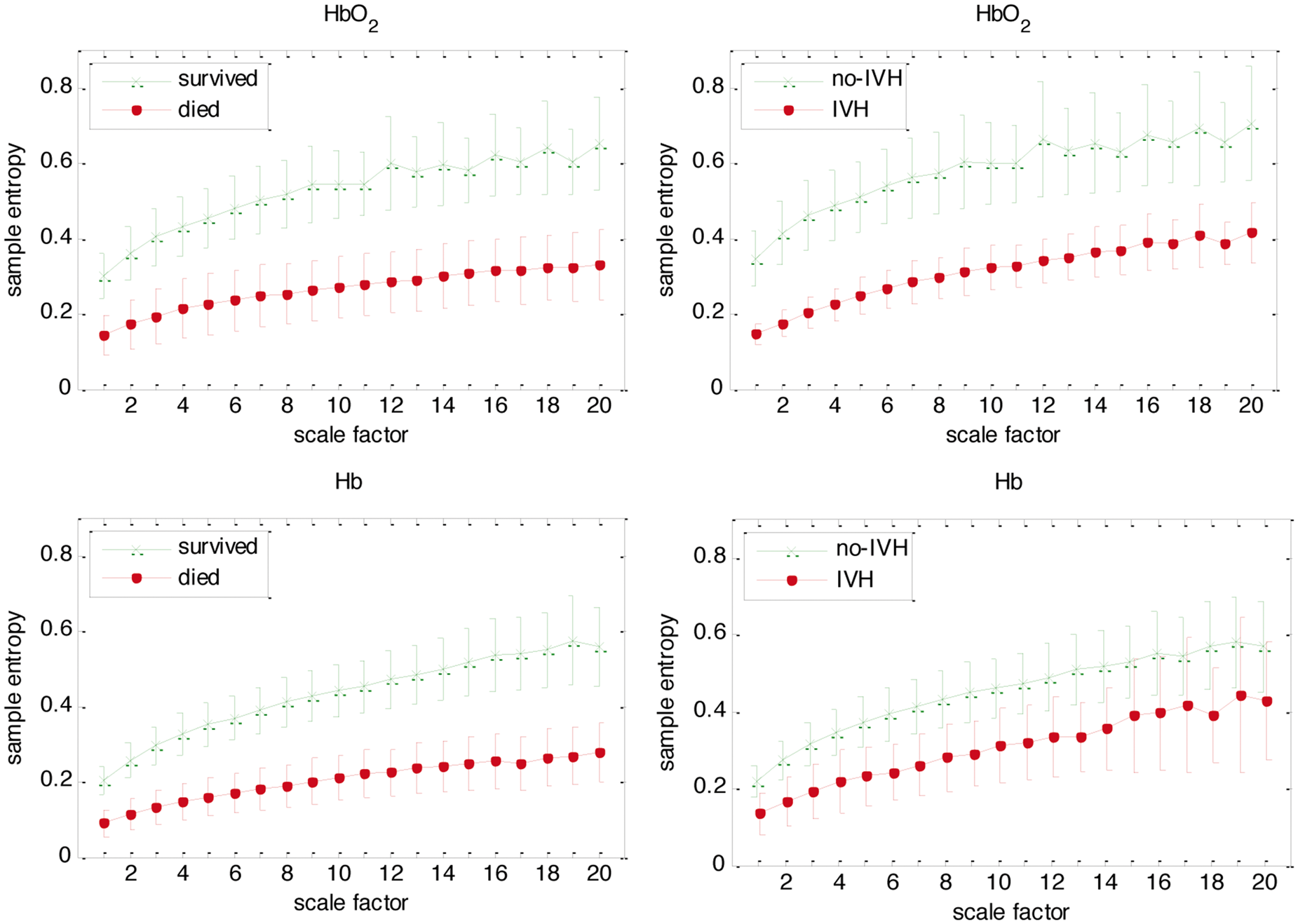
Association between brain and systemic signals with outcome
Differences in complexity of brain and systemic signals between outcome groups.
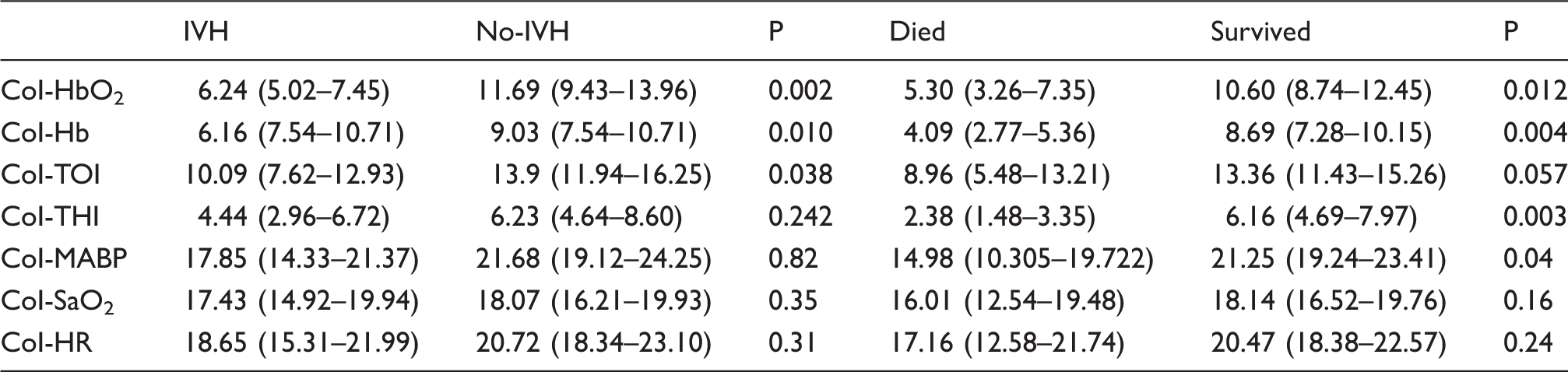
Mean and 95% confidence interval for complexity index (CoI) of NIRS and systemic physiological signals.
Decreased complexity of brain signals was observed in infants who died. Mean CoI-HbO2, CoI-Hb and CoI-THI were lower in those infants who died compared to those who survived (Table 3). CoI-TOI just failed to reach statistical significance (P = 0.057). There was no difference in mean TOI between these two groups (P = 0.52).
There was no difference between mean CoI of mean arterial blood pressure (MABP), mean CoI-HR and mean CoI-SaO2 between those infants who developed IVH and those who did not (Table 3). Mean MABP, mean SaO2 and mean HR were not different between these two groups (P = 0.52, P = 0.46 and P = 0.91, respectively). In contrast, mean CoI-MABP was lower in those infants who died compared to those who survived. However, there was no difference in mean CoI-SaO2 or mean CoI-HR between these two groups (Table 3). There was no difference between MABP, SaO2 and HR between those infants who survived or died (P = 0.79, P = 0.38 and P = 0.20, respectively).
Complexity of brain signals as an independent predictor of IVH
Binary logistic regression for CoI-HbO2, sepsis and CRIB II as predictors of IVH.
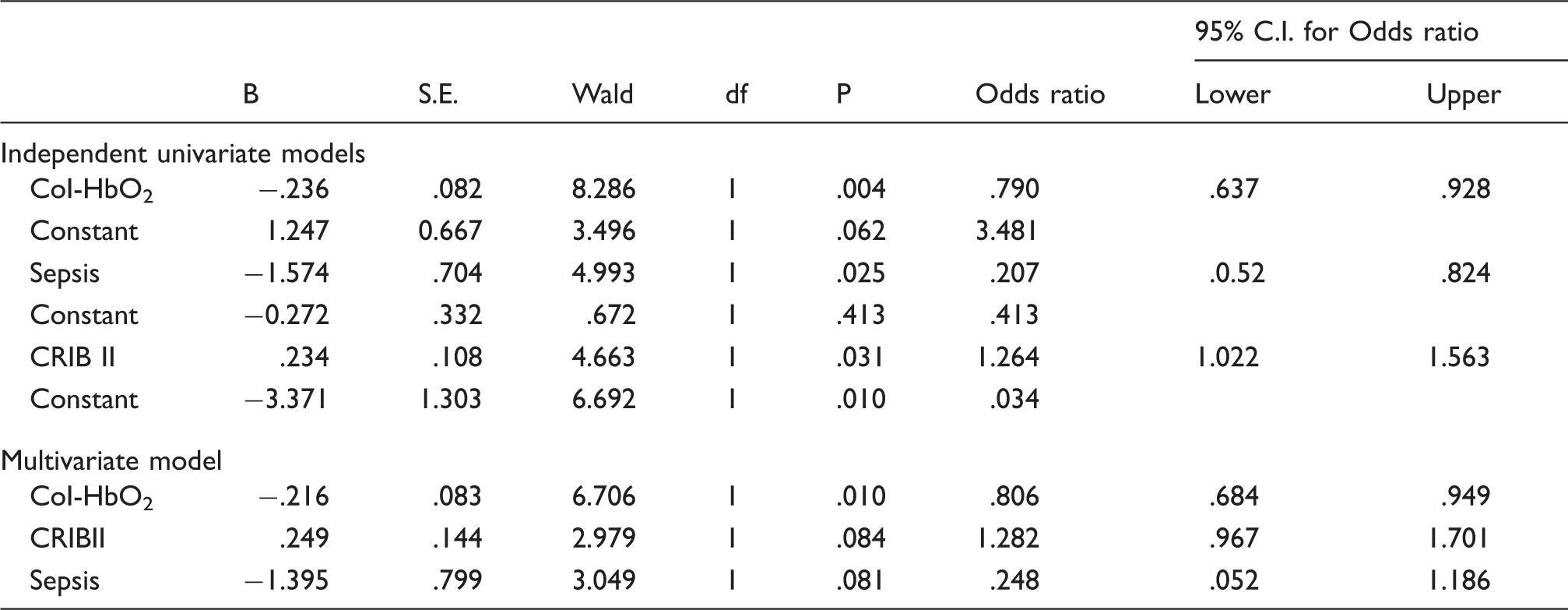
Sepsis is for presence of sepsis compared to no sepsis.
CoI-HbO2: complex index of oxygenated haemoglobin; CRIBII: clinical risk index for babies II.
Correlation between complexity index of brain and systemic signals.
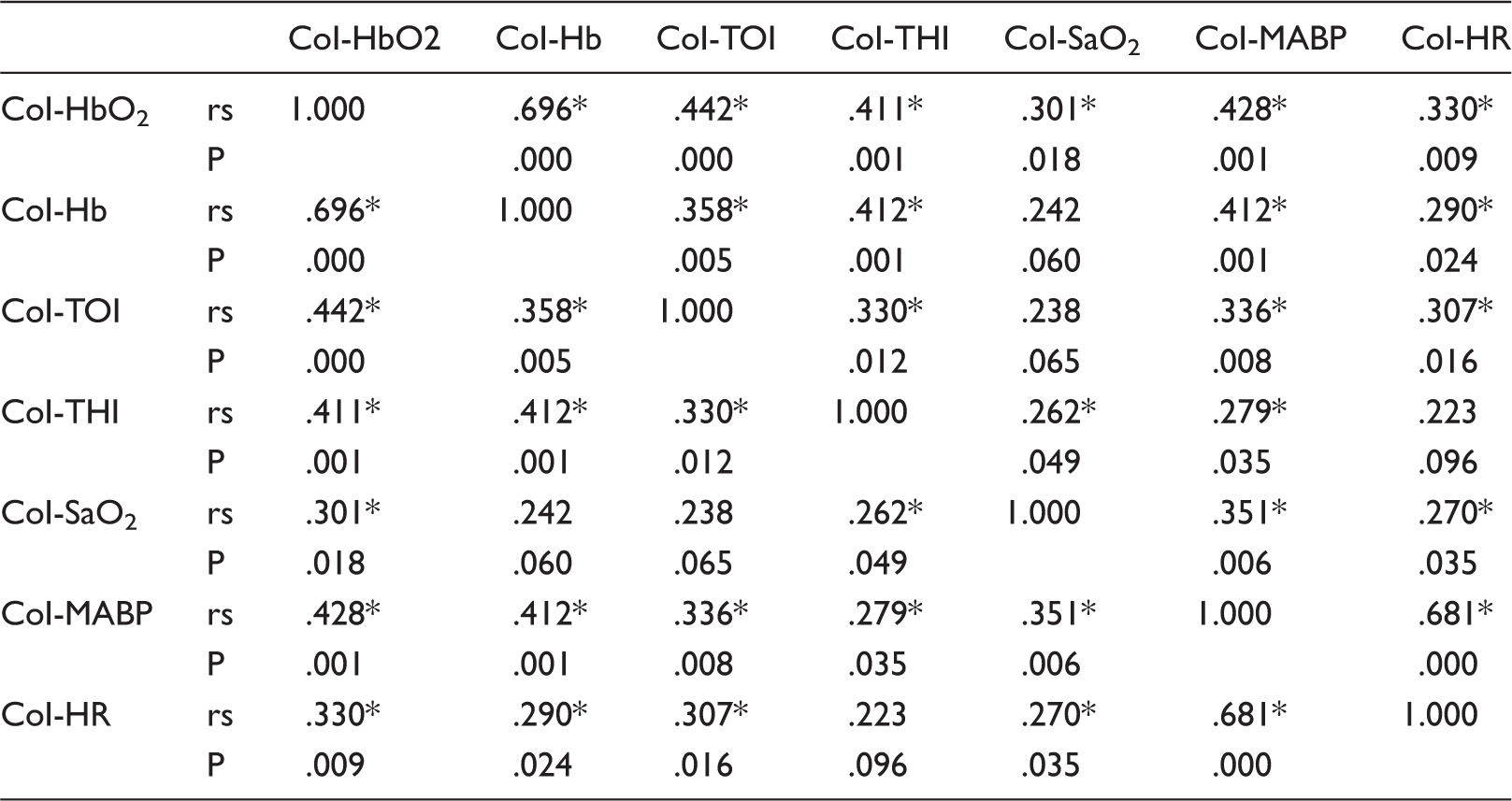
P values are not adjusted for multiple comparisons.
P < 0.05.
rs: Spearman’s rank correlation coefficient.
Discussion
This study is the first to assess the complexity of cerebral NIRS signals in critically ill preterm infants. We applied MSE to study the complexity of brain and physiological systemic signals in a cohort of preterm infants in the first 24 h of life. Our results revealed an association between complexity of brain signals and outcome.
Several studies have suggested that decreased variability in physiological signals is a sign of failure in complex regulatory system. In perinatal medicine, the ‘silent pattern’ in foetal ECG is a well-known example of loss of ‘natural’ variability of vital signs and has been associated with poor neonatal outcome. 33 However, in clinical practice, there is no consensus on which method of intrapartum monitoring is the most effective. 34 Moreover, the continuous monitoring of neonatal vital signs does not take in consideration the non-stationarity characteristic of the biological signals. Entropy analysis has been extensively studied to investigate physiological systems. According to the ‘decomplexification’ theory of illness, loss of complexity is usually associated with pathological states like diseases or ageing. However, despite being an old concept, assessment of complexity of physiological signals in the neonatal unit is not performed routinely. Moreover, clinicians persist to correlate lack of variability as a sign of clinical stability. In our study, decreased complexity of brain signals was observed in those infants who developed IVH and who died, supporting the ‘decomplexification’ theory.22,35 MSE of HbO2 and Hb were consistently lower in all scale factors for groups of infants with IVH compared to those who did not have IVH, and for groups of infants who died compared to those who survived. These results are similar to recently published work using MSE to investigate the association between complexity of EEG and neurological outcome in a cohort of term infants with refractory neonatal seizures. 26 Lower values of MSE were observed in most scales in the group of infants who later developed epilepsy, compared to normal controls, and to those who did not develop epilepsy. In adults with traumatic brain injury, a decrease in entropy of ICP signals across all MSE scales was observed during the development of intracranial hypertension and loss of cerebrovascular reactivity. In this cohort, the CoI was significantly lower in those patients who had moderate and severe disability compared to those who had good outcome. 26
High complexity reflects strong, non-linear interactions between different healthy control systems responding to transient stressors or adapting to changes in the environment. The impairment of those regulatory mechanisms or the breakdown in their interactions is characterized by low complexity of various relevant physiological measurements. This may manifest itself in the total breakdown of (auto and cross) correlations of the measured signals (e.g. instantaneous change in heart rate in atrial fibrillation) or patterns that are overly regular and deterministic in nature (e.g. flat heart rate dynamic seen in chronic heart failure).19,36 Assuming that the impairment of certain physiological control systems occurs before the onset of clinical symptoms of the developing pathology, or even assuming that such impairment could be the potential cause of diseases, a decrease in complexity could serve as an alert of complications like sepsis or poor outcome.26,37 In our study, complexity of HbO2 was an independent predictor of IVH and decreased complexity of brain signals within the first 24 h of life was observed in those infants who died later during admission, suggesting that the loss of interaction between control systems may be present even before the occurrence of brain injury and death. This could potentially, from early hours of life, divide those infants who are at more risk of poor outcome to those who are more likely to have less morbidity. While the ultimate goal of early analysis of physiological signals would be to identify the ‘high risk’ infants and develop therapies to prevent IVH and death, identifying those who are ‘healthy’ could help clinicians to be less invasive and differentiate between normal physiological to pathological adaptation.
The lower complexity of HbO2, Hb and TOI in those infants who developed IVH may reflect, at least partially, the underlining loss of cerebrovascular reactivity that precedes the occurrence of IVH, as most infants developed IVH after the first 24 h of life. Changes in cerebrovascular reactivity and loss of autoregulation within the first hours of life have been associated with the incidence of IVH in preterm infants.15,38,39 Several studies have shown that cerebral hypo-perfusion is associated with the pathophysiology of IVH and death.3,40 In our study, most infants who died were extremely preterm and all of them had some degree of either antenatal or early postnatal hypo-perfusion or hypoxia. Even those infants who died due to NEC or sepsis were severely sick within the first days of life. Decreased oxygenation has been associated with death as shown in a large cohort of infants included in the BOOST-II trial. In this study, infants with lower oxygen saturation target had higher rate of NEC requiring surgery or causing death. 41 In our cohort, target SaO2 was kept the same (between 91%–95%) in all groups and there was no difference between mean SaO2 and CoI-SaO2 between those infants who died or survived or between those who developed IVH and who did not. However, cerebral hypo-perfusion and loss of cerebrovascular reactivity and may be associated with reduced complexity of cerebral NIRS signals. This hypothesis should be tested in a larger cohort where other factors related with changes in cerebral blood flow, like PaCO2, are also taken into consideration.
In adults, early assessment of complexity of HR and HRV measured by MSE has been used to predict outcome in patients with traumatic brain injury and acute stroke.21,42 In neonates, early monitoring of systemic blood flow and cerebral oxygenation has been used to understand the pathophysiology of brain injury in preterm infants.3–5,43 Although the incidence of severe IVH and mortality in this population has decreased in the last 20 years, with improved survival, the prevalence remains high, as does the degree of disability and morbidity in survivors. 44 Preterm infants who develop IVH are at risk of higher morbidity during the neonatal period and later in childhood, including the development of post-haemorrhagic ventriculomegaly, recurrent hospital admissions and mortality. In addition, they may present long-term neurodevelopment and cognitive delays, resulting in increased workload for the health and educational services.1,45 Studies using cerebral NIRS have attempted to define limits of cerebral autoregulation and thresholds of cerebral oxygenation, in order to prevent the occurrence of IVH and improve outcome.7,46,47 However, there is no established threshold of cerebral perfusion and the continuous assessment of cerebrovascular reactivity and oxygenation has not yet been proved to change the outcome of preterm infants. In this context, the analysis of complexity of cerebral NIRS signals may offer a new approach – perhaps an alternative if not a complementary one to the cerebrovascular reactivity assessment – in order to understand the physiological mechanisms involved in the developing brain and to identify those infants who are more likely to be severely sick or even die during the neonatal period.
Complexity of brain signals demonstrated stronger association with outcome than complexity of systemic signals. In our study, the complexity of MABP was the only complexity of systemic physiological signals that correlated with outcome. Decreased complexity of MABP was associated with mortality but not with IVH. In clinical practice, the assessment of organ perfusion in preterm infants still relies on indirect measures of systemic blood flow, such as MABP. In adults, the regulation of cerebral perfusion is independent of systemic perfusion. The healthy adult brain has mechanisms that protect its tissue against ischaemic damage. The complexity of cerebral NIRS signals may represent a more direct measure of cerebral perfusion and regulation of blood flow than the complexity of systemic signals. On the other hand, the positive correlation between complexity of systemic and brain signals may suggest that factors associated with loss of cerebral and systemic regulatory mechanisms are related to each other. Further studies are needed to explore the significance of this relationship. Nevertheless, our findings support the idea that the monitoring of brain signals in addition to systemic signals would provide more comprehensive grounds for understanding the physiological mechanisms related to mortality and brain injury in preterm infants.
Limitations
MSE analysis requires a large data set to be able to estimate complexity in a plausible way. The minimum number of samples from time series that allows reasonable MSE calculation rises with the number of scales. It can be estimated that at least 2000 data samples are required to perform analysis up to scale 20. 48 In our study, the analysed time series had one sample every 10 s, so at least a 6-h long monitoring period was required to produce meaningful values. Furthermore, MSE analysis is relatively sensitive to outliers and artefacts. Removing a small percentage of them is allowed, however, if the structure of time series is grossly altered due to artefact removal, the value of entropy may also change. 24 The amount of data containing artefacts from our cohort was small, but the artefacts were manually and retrospectively removed. Real-time bedside analysis would require appropriate automatic artefacts detection and removal as well as a criterion for automatic rejection of data segments with too high artefacts content.
Our results of the mortality analysis have to be treated with some caution. Although the association between complexity and mortality was significant, we acknowledge that our sample was small and unbalanced, as only eight infants died. Moreover, most infants in our cohort died later during the neonatal period due to factors, which were not necessarily directly related to changes in cerebral perfusion within the first days of life, as sepsis and NEC. However, all infants who died had signs of antenatal or perinatal hypoxia and remained very sick throughout the neonatal admission. Further studies with a larger sample size should be conducted, in order to understand the factors associated with decreased complexity of brain signals in sick preterm infants. However, early recruitment should be taken in consideration to better understand the pathophysiology of brain injury. We aimed to recruit infants before 12 h of age, but this may have contributed to our limited our sample size, in view of the challenges of getting informed consent from parents soon after the delivery of a sick preterm infant.
Conclusion
We have demonstrated that the assessment of complexity of brain and systemic signals using MSE in preterm infants undergoing intensive care is feasible. Decreased complexity of cerebral signals was more strongly associated with outcome of IVH and mortality than the complexity of systemic signals. Furthermore, the complexity of brain signals was an independent predictor of brain lesions in this preterm population. Real-time bedside complexity signal analysis could represent a significant advance in the brain oriented care of critically ill newborn infants.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SPARKS charity (11CUH02); Cambridge Trust and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) scholarship to da Costa CS; National Science Centre, Poland (no. DEC-2013/10/E/ST7/00117) scholarship to Placek MM and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPQ) (203792/2014-9) post-doctoral scholarship for Cabella B.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The ICM+ software used for data monitoring and analysis is licensed by Cambridge Enterprise Limited (University of Cambridge); P.S. and M.C. have interest in a fraction of the licensing fee. The other authors declare no conflicts of interest.
Authors’ contributions
da Costa CS co-conceptualized and designed the study, was responsible for identifying infants who met the inclusion criteria and for getting parental consent, collected the data, collaborated with data analysis, drafted the initial manuscript and approved the final submitted manuscript. Placek MM co-conceptualized the study, was responsible for the Multiscale Entropy analysis of the data and collaborated with statistical analysis. Czosnyka M co-conceptualized and designed the study, run exploratory data analysis, supervised data analysis. Cabella B supervised data analysis. Kasprowicz M supervised the data analysis. Austin T co-conceptualized and designed the study, supervised data collection and data analysis, revised the results. Smielewski P designed the study, supervised data analysis and revised statistical analysis. All authors contributed to writing the manuscript and approved the submitted manuscript.
