Abstract
An agent for visualizing cells by positron emission tomography is described and used to label red blood cells. The labeled red blood cells are injected systemically so that intracranial hemorrhage can be visualized by positron emission tomography (PET). Red blood cells are labeled with 0.3 µg of a positron-emitting, fluorescent multimodal imaging probe, and used to non-invasively image cryolesion induced intracranial hemorrhage in a murine model (BALB/c, 2.36 × 108 cells, 100 µCi, <4 mm hemorrhage). Intracranial hemorrhage is confirmed by histology, fluorescence, bright-field, and PET ex vivo imaging. The low required activity, minimal mass, and high resolution of this technique make this strategy an attractive alternative for imaging intracranial hemorrhage. PET is one solution to a spectrum of issues that complicate single photon emission computed tomography (SPECT). For this reason, this application serves as a PET alternative to [99mTc]-agents, and SPECT technology that is used in 2 million annual medical procedures. PET contrast is also superior to gadolinium and iodide contrast angiography for its lack of clinical contraindications.
Keywords
Introduction
Hemorrhage is an important symptom of many diseases. Its rapid identification and repair can greatly improve patient prognosis. Unfortunately, it can be difficult to accurately image hemorrhage, especially in situations of internal bleeding. The accurate diagnosis of hemorrhage is especially vital in emergencies. This is clear in the case of stroke1,2 where 40,000 patients per year will experience intracranial hemorrhage, and a third will die within 30 days from its complications3,4 in the US alone. Direct, real-time monitoring of hemorrhage can improve patient outcomes, and could entail a shift in its emergency management 5 from one that is dependent on an exclusion-of-pathology process to one that capitalizes on precise and efficient diagnostic methods to inform treatment decision-making. Accurate, direct imaging of hemorrhage would help guide the surgical evaluation of a hemorrhage and prevent deterioration, brainstem compression, hydrocephalus, and supratentorial hematoma. In the case of ischemic stroke, accurate and rapid direct imaging that can exclude intracerebral hemorrhage (ICH) could play a clinically important role, and potentially expand the utilization of tissue plasminogen activator (tPA) to reverse symptoms of ischemia.6–8
ICH in the brain is best imaged with contrast. 9 Both cerebral computed tomography angiography (CTA) 10 and magnetic resonance angiography (MRA) 11 have been used to image ICH, as they enable clear visualization of the vessels and blood pool associated with hemorrhage. Unfortunately, both CTA and MRA have clear weaknesses for emergency management. Morbidity and risk accompany angiography. 12 Patients must first be evaluated for contraindications, which take time, and inevitably, not all patients are candidates for angiography. For these reasons, CTA/MRA imaging is a foregone option in emergencies, 13 and the current standard for determining ICH and indicating intravenous tPA administration during emergencies is non-contrast CT. Unfortunately, the low sensitivity and spatial resolution of non-contrast CT can make the diagnosis of hemorrhage problematic.
Currently, most hospitals already have positron emission tomography (PET) scanners, making PET a highly promising but undervalued tool, particularly in general hemorrhage situations including acute stroke, 14 traumatic brain injury, and hematoma. There are no known life-threatening contraindications for 18F-PET. PET is also directly compatible with CT, and scanners that can simultaneously acquire PET and CT are now clinically commonplace, due to the popularity of the [18F]-fluorodeoxyglucose ([18F]-FDG) contrast agent. 15 For imaging general hemorrhage by PET, the best contrast agent is, unironically, blood. Unlike [15O]-water, and other small PET molecules, a labeled blood cell can never incorrectly indicate hemorrhage, and trigger unnecessary endoscopy/intervention, by occupying a blood-free space.16,17 Changes in blood flow occur before metabolic and oxidative changes in tissue, making red blood cells (RBC) superior to other agents, such as [18F]-FDG 18 and [18F]-hypoxia sensors, 19 for imaging ICH. In addition, PET also offers a solution to a spectrum of problems that complicate single photon emission computed tomography (SPECT). [99mTc]-RBCs 20 and [99mTc]-leukocytes (exametazime)21,22 have been applied to the imaging of stroke and intestinal/renal bleeding by SPECT. But [18F]-PET is superior to SPECT because it can be used at lower doses, acquired at superior resolutions,23,24 and is compatible with new PET/CT and PET/MR equipment. Finally, [18F]-PET tracers are backward compatible with SPECT equipment,25,26 while SPECT tracers cannot be imaged by PET.
For these reasons, we focus on developing fast, simplified chemistry for synthesizing [18F]-RBCs to successfully image internal bleeding, specifically intracranial hemorrhage, by PET/CT in vivo and ex vivo.
Materials and methods
Macromolecular syntheses and characterization are described in accompanying supporting material.
RBCs isolation
Whole blood was collected from an anesthetized BALB/c mouse (3–4 week old, Charles River) by cardiac puncture in the presence of heparin as an anti-coagulant. Blood (∼0.6 mL per mouse) was diluted with sterile PBS at 1:1 ratio and then layered gently onto 1.2 mL of Ficoll Paque (GE Healthcare, 17-1440-03). The mixture was then subjected to density gradient centrifugation at 190 g for 20 min at 20℃. The top and middle layers were removed by pipette carefully, leaving only the bottom, dark red, layer of RBCs. At least 10 volumes of PBS were added and the resulting mixture was centrifuged at 190 g for 30 min at 20℃ to wash the RBCs. This step was repeated 6 times and was required to remove small molecules and free protein from RBCs. Supernatant was removed carefully after every centrifuge and the volume of the final RBCs solution was approximately 1 mL.
18F labeling of NHS ester 1 and 2
In a typical radiosynthesis, an aliquot of cyclotron-proton bombarded, pure [18O]-water was received and flushed with nitrogen at 28 psi in a 5.0 mL glass v-vial (Thermo Scientific Reacti-Vial #13223). The dry vial contained 67 mCi of dry activity (t = 0 min, otherwise referred to as the start of synthesis, SOS. To recover maximal activity in the referenced glass v-vial, it is important that activity is concentrated to a reduced volume but not fully evaporated). To the concentrate, ∼ 5 µL volume of water, 0.8 µL of 100 mM HF (80 nmol) fluoride pH 3.0, and 4 µL of a 1 mM (20 nmol) DMSO solution of NHS esters
RBC labeling and workup
RBCs were labeled at room temperature with [18F]-
Cryolesion model
All procedures conducted in mice were approved by the Weill Cornell Medical Center Institutional Animal Care and Use Committee (#2014-0030) and are consistent with the recommendations of the American Veterinary Medical Association and the National Institutes of Health Guide for the Care and Use of Laboratory Animals. This work is in compliance with ARRIVE guidelines for how to report animal experiments. Sixty BALB/c mice were used for this project.
A cylinder described by Raslan et al. 27 was machined for this project. The cylinder is modified to bear a tip diameter of 2.0 mm and is machined from aluminum instead of copper. The cylinder is secured in a holder fixed on an optical breadboard via a screw that allows a user to raise or lower the cryoprobe to precisely adjust contact time and force over the cryolesion-focal point. To quantitate the applied force, mice were anesthetized (2% isolfluorane/oxygen) and positioned on a scale using either 3M micropore surgical tape, or a Kopf model 900 stereotaxic instrument (images of the apparatus are shown in Supporting Figure S14).
BALB/c mice are ideal for RBC imaging because blood can be interchanged between BALB/c mice. Anesthetized BALB/c mice were disinfected twice with 70% isopropyl alcohol and 10% povidone-iodine (Betadine® Scrub). Surgical instruments were sterilized before surgery by steam sterilization and between animals using a glass bead sterilizer. An incision was made to expose the parietal bone through a 1.5 cm vertical, mid-line, cutaneous incision between the ears to create soft tissue flaps that were retracted for skull exposure in order to prepare the contact area for the cryoprobe. The cryoprobe was filled with liquid nitrogen and equilibrated for 5 min before it contacted the bare, exposed skull over the right parietal cortex of the mice (target coordinate from bregma: 1.5 mm posterior and 1.5 mm lateral). The tip of the liquid nitrogen filled cryoprobe is applied to the coordinate with a weight of 100 g (applied force 7.9 g/mm2, over a 12.6 mm2 surface area) as measured by scale displacement. An application time between 35 to 55 s was used. One drop of 0.25% bupivacaine solution was applied to the cryolesion site, and the incision was then closed using one or two stainless steel wound clips. The animal can be immediately transferred to an Inveon PET/CT for imaging. However, in our initial experiments, we noted that 8/30 mice perished before the end of a 40 min PET scan (55 s lesion) when we proceeded with a direct transfer from the operating table to the PET scanner. To limit mortality, we implemented a recovery step post-cryolesion where the animal was transferred from the operating table to a cage (with access to food and water) that is uniformly warmed to 25℃ using a space heater. Even with longer survivals (5–6 h post cryo-lesion), no mice were lost when this recovery step is employed (over 30 mice bearing 35 s to 55 s cryolesions). This step must be performed to prevent mice from dying from hypothermia as a result of cryolesion and immediate, subsequent scanning in an Inveon PET/CT with a bore that is calibrated and run at 21℃.
[18F]-PET/CT imaging of intracranial hemorrhage and scintillated biodistribution
In order to visualize intracranial bleeding, 200 µL of labeled RBC (3.76 × 108 cells, <200 µCi) were injected into the tail vein, at different time points pre- or post-cryolesion. Intracranial hemorrhage could be imaged if [18F]-
PET/CT were processed with Amide v1.0.4 and Inveon Research Workplace. Brains were frozen in Optimal Cutting Temperature (OCT) Compound (Sakura Tissue-Tec, #4585) or refrigerated in 10% paraformaldehyde PBS solution for fluorescent or histological analyses. Preservation in OCT compound is recommended if histological processing is performed after prolonged storage, i.e. >2 days, to minimize deterioration during storage (Supporting Figure S10).
Biodistribution
Acute biodistribution was determined in a separate cohort (n = 6 mice) that underwent cryolesion and RBC injection, but were not PET imaged. After cryolesion and contrast agent injection (<5 min following lesion), blood samples were collected from the left ventricle before tissues were collected for biodistribution. Scintillation measurements were taken with a Wallac Wizard 3.0 gamma counter. Mice were sacrificed by cervical dislocation before tissue was harvested. PBS or saline perfusion was not performed prior to tissue harvesting.
Biodistribution was determined by scintillated (gamma) counting. The majority of the injected [18F]-
Sample size/replicates
Approximately 60 cryolesion bearing mice were used to evaluate the utility of [18F]-
Data blinding
The depth of a hemorrhage is difficult to know during and following cryolesion, especially since the process of cryolesion opaquely colors the skull. This phenomenon effectively randomizes our study, as the exact volume of hemorrhage becomes impossible to know until after brain excision and ex vivo PET/Brightfield/histological analysis.
Results
The primary objective of this study is the development of an imaging agent for the general observation of internal hemorrhage; specifically, intracranial hemorrhage. The chemistry chosen had to be general and applicable to all cell types in order for the imaging agent to have application to many different future cell types. For this reason, [18F]-cell labeling strategies cannot employ organic co-solvents, which are cytotoxic. In recent work, we observed that the displacement of a dioxaborolane with [18F]-fluoride is gentle, and that trifluoroborate synthesis proceeds in aqueous media without hydrolyzing an appended, activated N-hydroxysuccinide ester, 29 making this chemistry is uniquely suited to labeling cells. Additionally, the RBC made an ideal choice for imaging intracranial hemorrhage because, by definition, RBCs cannot occupy a blood-free space. For this reason, RBCs could be used to accurately image the porosity of the blood–brain barrier to cells and additionally distinguish hemorrhage from non-hemorrhagic edema, unlike small molecule, protein, and antibody surrogates for hemorrhage.
Timeline
The general timeline for RBC labeling was as follows: [18F]-
RBC labeling: In vitro studies
To demonstrate definitively that RBCs could be stably labeled with an [18F]-bearing compound, it was necessary to first synthesize two small-molecule reagents, compounds Structure of [18F]-PET/fluorescent probes, RBC reactivity, and a scheme for demonstrating [18F]-RBC stability. (a) The amine reactive 18F-PET/NIRF precursor, 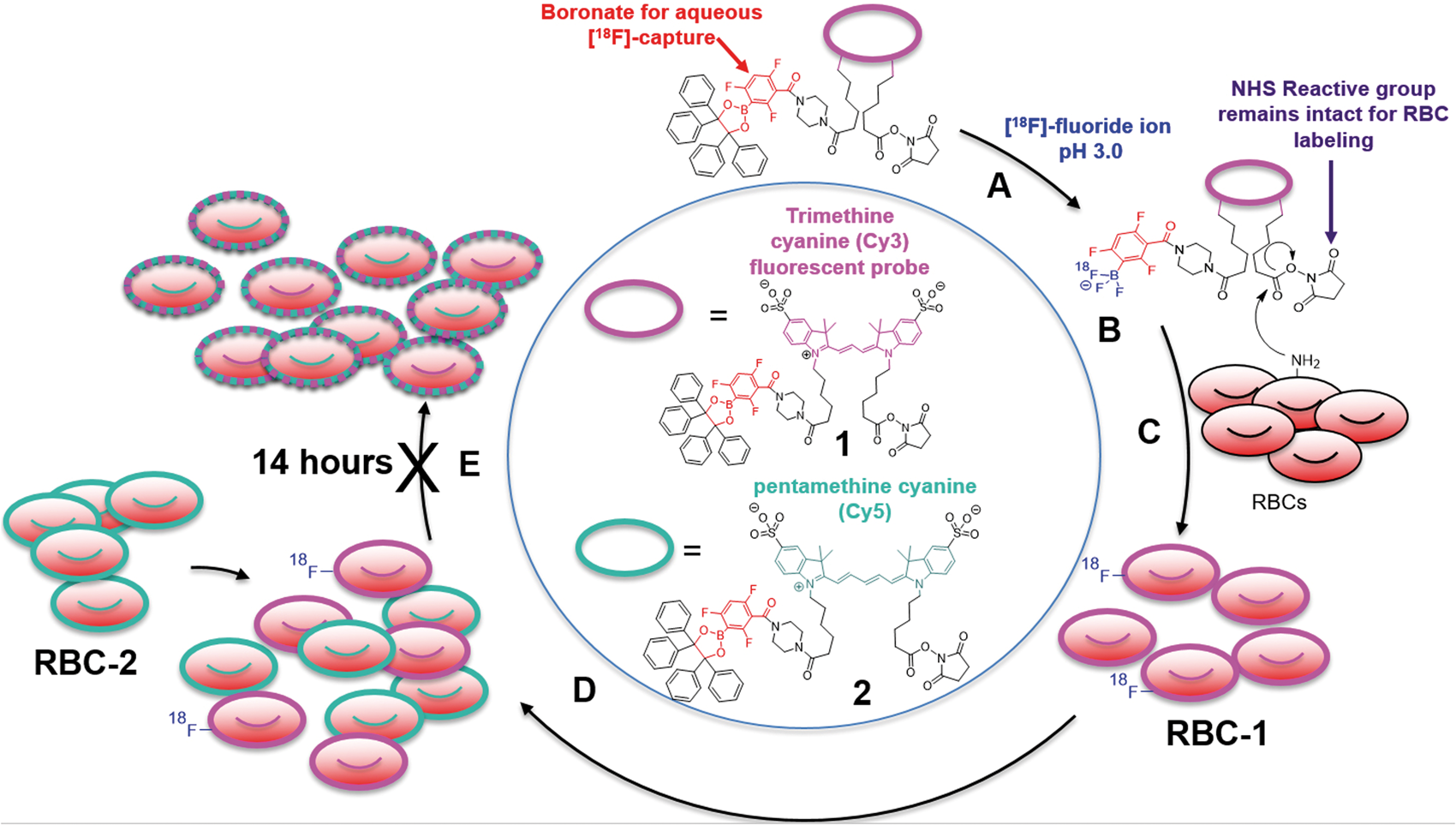
Once an RBC is labeled with [18F]-PET/fluorescent probes 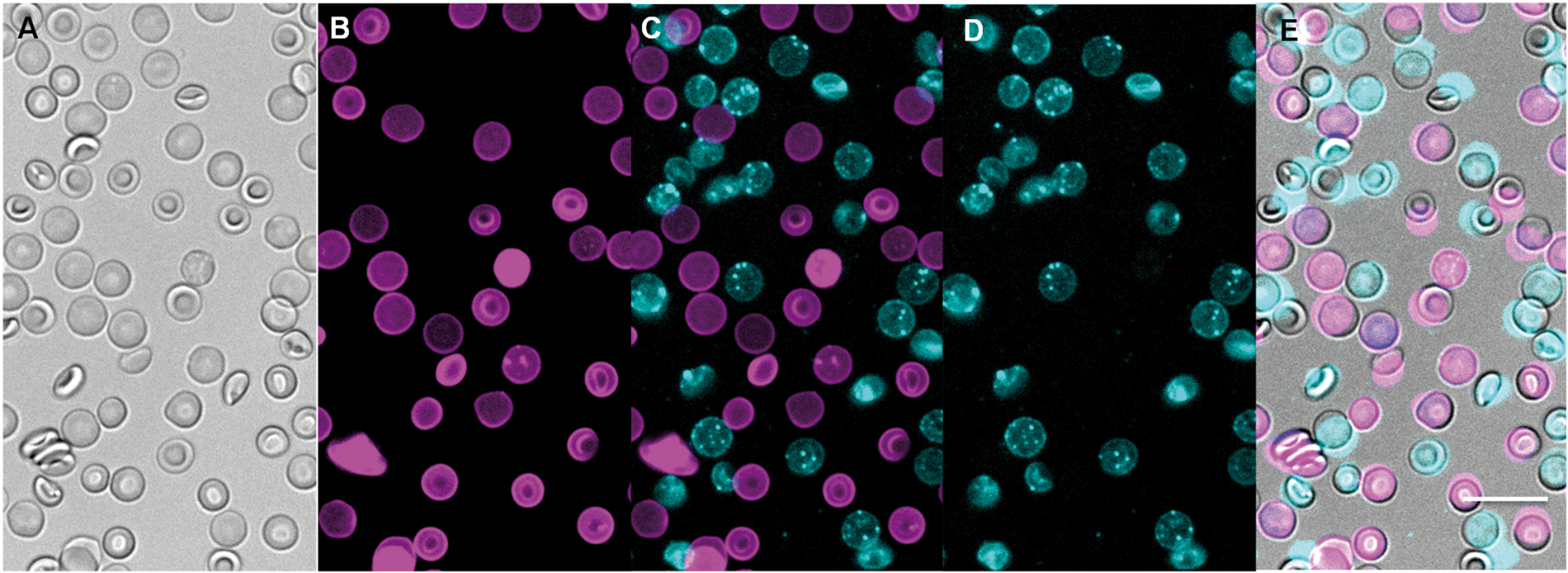
These data prove that fluorophores do not degrade, excrete, or exchange between cells in timeframes that are relevant to [18F]-PET imaging. Additionally, these data suggest that RBC bearing
The labeling of RBCs with
[18F]-RBC radiochemistry
[18F]-RBCs suitable for PET imaging were synthesized in a one-vessel, two-step aqueous reaction without the use of cytotoxic organic co-solvent. In prototypical radiolabeling, compound
The lower specific activity (SA) limit per molecule of
Internal hemorrhage model
Klatzo et al.
36
reports a closed-skull, cryolesion-based model for generating traumatic brain injury (TBI) in feline models. Raslan et al.
27
has recently transformed this model in mice. By contacting the skull with a machined, liquid-nitrogen-filled aluminum probe, non-lethal, intracranial hemorrhage could be directed to chosen regions of the brain. The model does not penetrate the skull, and is superficial, allowing us to immediately corroborate [18F]-
We identify the ideal time and contact weight that should be used with the 2.0 mm cryoprobe in 3 - to 4-week-old BALB/c mice. A contact weight of 100 g should be applied to the skull at 1.5 mm posterior and 1.5 mm lateral vs. bregma
27
for a time between 35 and 55 s. In studies where insufficient contact is applied (less than 35 s) mice frequently do not develop hemorrhage, develop low-volume lesions that scab, or develop hemorrhage does not penetrate further than 1 mm in depth (determined ex vivo by macroscopic dissection). Non-hemorrhage and low volume hemorrhage cannot be imaged by [18F]-
A contact time that approaches 55 s has never failed to generate intracranial hemorrhage, and for this reason, is the preferred contact time for [18F]-
[18F]-RBC animal imaging
Six mice were imaged with [18F]-
Pairs of BALB/c mice were injected with 100 µCi (3.78 × 108 RBCs) of [18F]-
Hemorrhage at the site of cryolesion was clearly visible by whole body PET, showing that [18F]- Intracranial hemorrhage is visualized with [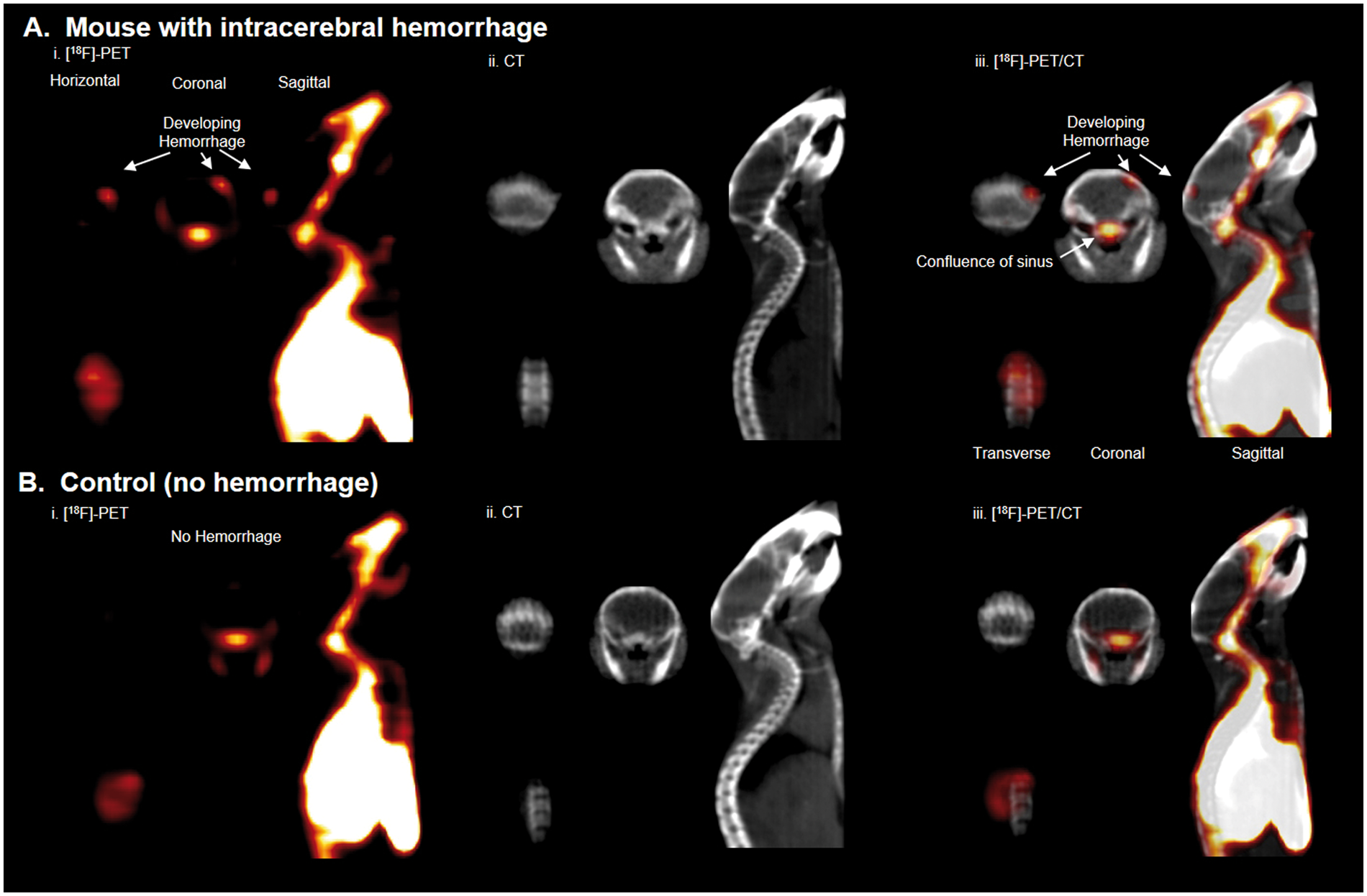
To confirm that the activity of interest is indeed due to hemorrhage, mice were sacrificed following scanning, and the brains of both cryolesion and control mice were excised, photographed (white light), and imaged again, ex vivo by PET/CT. Bright field imaging showed hemorrhage near the surface of the brain at the site of cryolesion (Figure 4(a)). The dimensions and asymmetric, anatomical location of ICH were corroborated by PET imaging (Figure 4(b), Supporting Video S3). Fluorescence confirmed intracranial hemorrhage in the brain ex vivo (Supporting Figure S4). Tissue disintegration and major RBC washout were observed in attempts at preserving brain for confirmative MRI (Supporting Figure S5); however, hemorrhage was confirmed in formalin-fixed paraffin sections of brain with RBC-specific, TER-119 immunohistochemical staining (Figure 4(c)). Tissue disintegration can be avoided by storing tissue in OCT compound at −78℃ (Supporting Figure S10).
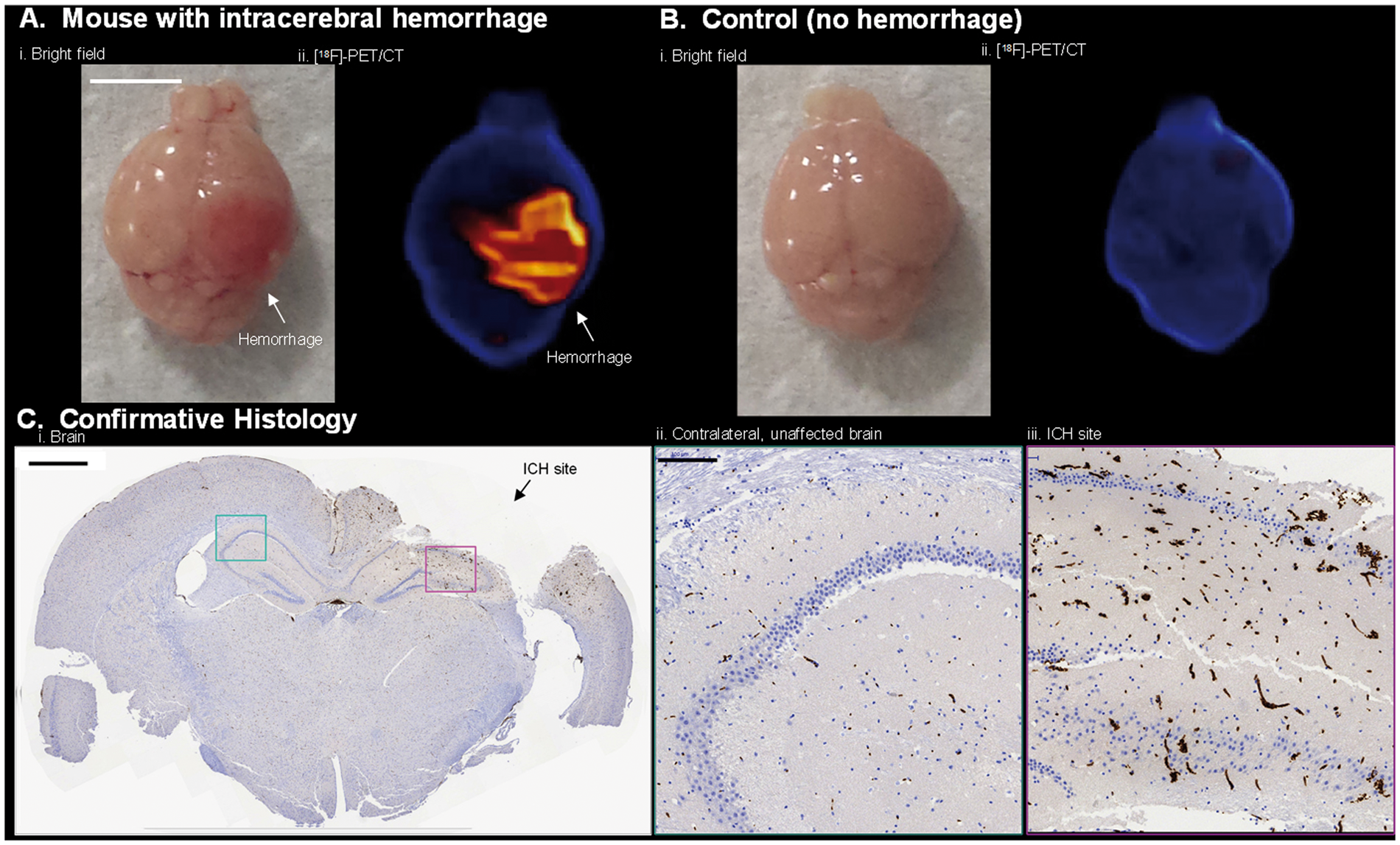
[18F]-
Discussion
Experimental design
The primary objective of this study was the development of a universal imaging agent for observing active hemorrhage. The chemistry chosen must be general and applicable to all cell types in order for the imaging agent to have application to many diseases, and to be of interest to a general scientific community. It is preferred that [18F]-cell labeling strategies that do not employ organic co-solvents, which are cytotoxic, and are preferred by biologists and clinicians. The RBC made an ideal choice for imaging hemorrhage because, by definition, RBCs cannot occupy a blood-free space. RBCs could be used to additionally distinguish hemorrhage from edema, unlike [18F]-small molecules, proteins, and antibodies.
A disease model had to be chosen in order to demonstrate an imaging sensitivity that is superior to existing SPECT technology. Of all internal bleeding models, the ICH model produces the maximum degree of morbidity and mortality from minimal volumes of bleeding and is therefore more difficult to image than other models of internal bleeding. When significant delays are inserted between cryolesion and [18F]-
[18F]-
There are no major foreseeable barriers to having [18F]-
In this application, the fluorescent component on [18F]-
In conclusion, we report a small-molecule multimodality PET/fluorescent cell labeling reagent that is used to label RBCs. The fluorescent component of this probe is used to show that labeled RBCs do not transfer their probe to other tissues (read other RBCs) even after 14 h of incubation. The PET component of the probe is used to successfully image active intracranial hemorrhage, in vivo, in live mice. This is confirmed by ex vivo PET, fluorescence, and histology.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project is supported by grants to R.Y.T. from the US National Institutes of Health (CA158448) and the Howard Hughes Medical Institute. R.T. is funded by the National Institute of Biomedical Imaging and Bioengineering K99/R00 (EB013904). O.A. is funded in part through the NIH/NCI Cancer Center Support Grant P30 CA008748.
Acknowledgments
This study is dedicated to the memory of Roger Y Tsien.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship and or publication of this article.
Authors’ contributions
RYT, OA, BF, and RT designed the study. BF, YW, FA, and RT generated animal models. FA and MC performed chemical syntheses. YW and EAR performed in vitro cell labeling studies. YW, FA, and RT performed RBC isolation. Radiolabeling was done by YW, FA, and RT. OA acquired histology. RYT, OA, RT, EAR, and BF contributed to the editing and writing of the manuscript. All authors contributed to the discussion of the methods and animal models.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
