Abstract
Retinal microvascular changes can be visualized noninvasively and have been associated with cognitive decline and brain changes in relation to aging and vascular disease. We systematically reviewed studies, published between 1990 and November 2012, on the association between retinal microvascular changes and dementia, cognitive functioning, and brain imaging abnormalities, in the context of aging and vascular risk factors. In cross-sectional studies (k = 26), retinal microvascular changes were associated with the presence of dementia (range of odds ratios (ORs) 1.17;5.57), with modest decrements in cognitive functioning in nondemented people (effect sizes -0.25;0.03), and with brain imaging abnormalities, including atrophy and vascular lesions (ORs 0.94;2.95). Longitudinal studies were more sparse (k = 9) and showed no consistent associations between retinal microvascular changes and dementia or cognitive dysfunctioning 3 to 15 years later (ORs and hazard ratios 0.77;1.55). However, there were indications of prospective associations with brain imaging abnormalities ((ORs) 0.81;3.19). In conclusion, particularly in cross-sectional studies there is a correlation between retinal microvascular changes and dementia, cognitive impairment, and brain imaging abnormalities. Associations are strongest for more severe retinal microvascular abnormalities. Retinal microvascular abnormalities may offer an important window on the brain for etiological studies.
Keywords
INTRODUCTION
Vascular disease is an important cause of dementia and cognitive decline in older people. 1 Vascular damage in the brain often co-occurs with vascular disease elsewhere in the body, probably reflecting common underlying pathophysiological processes. 2 This concerns damage to both large and small vessels. Hence, markers of microvascular or macrovascular disease outside the brain may convey information about vascular abnormalities in the brain and, consequently, about late-life cognitive functioning. In this context, the vessels of the retina are of particular interest for two reasons: they can be directly visualized noninvasively and they share anatomic, embryologic, and physiologic characteristics with the cerebral microvasculature.3,4
Microvascular changes in the retina are associated with hypertension, inflammation, diabetes, stroke, and cardiovascular disease, as previously described in review articles.5,6 Also, associations between retinal microvascular changes and dementia risk, cognitive functioning or brain imaging abnormalities have been reviewed.7,8 However, to our knowledge, no integrated, comprehensive, and quantitative overview of these associations has been published yet.
We hypothesized that retinal microvascular changes are a marker of cerebral abnormalities, in particular vascular lesions and atrophy, and as such may be linked to dementia or cognitive impairment. Therefore, the aim of this paper is to provide a systematic review of the literature on the association between retinal microvascular changes and dementia or cognitive functioning and brain imaging abnormalities, in the context of aging and vascular risk factors.
MATERIALS AND METHODS
Identification of Studies
This systematic review was performed according to the methodology of the PRISMA statement.9,10 We aimed to include all published studies that provided analyses of the relation between retinal microvascular changes and the following outcome measures: dementia or cognitive functioning and brain imaging abnormalities. We applied the following inclusion criteria: the study (1) reported on measures of retinal microvascular changes in relation to dementia or cognitive functioning or brain imaging abnormalities, (2) was published after 1990, (3) had a population-based design or consisted of participants selected on the presence of specific vascular risk factors or vascular disease, (4) reported on a sample size of ≥ 250 for population-based cohorts and ≥ 50 for patient-based cohorts, (5) reported on participants with a mean age of ≥ 50 and adjusted for age in their analyses. A minimum sample size was chosen because in small studies the statistical power may be insufficient to reliably evaluate the relation between retinal microvascular changes and the outcome measures. A minimum age was required because this review focuses on the relation between retinal microvascular changes and the outcomes in the context of aging. Any method that was used to assess retinal vasculature, cognition, or brain imaging abnormalities was accepted. We excluded studies that selected participants on the presence of retinal microvascular changes or the outcomes (i.e., dementia and cognitive functioning or brain imaging abnormalities). We also excluded studies that investigated type 1 diabetes, because in these patients the role of vascular risk factors and aging is different from the general population or patients with type 2 diabetes.
Medline (1990 to 22 November 2012) and cross references from included papers were used to identify relevant papers. The search was limited to papers that were written in English. Searches were performed separately for cognitive and imaging outcome measures. We used the following search terms: ('retina’ and ('microvascular', ‘arteriolar', ‘venular', or ‘vascular') or ‘retinopathy') in combination with ('cognition', ‘dementia', or ‘memory') and, separately, in combination with (('brain’ or ‘cerebral') and ('mri’ or ‘ct’ or ‘imaging’ or ‘lesion')) and ('volume', ‘atrophy', ‘white matter', ‘hemorrhage', ‘bleeding', ‘microbleeds', ‘infarct', ‘stroke', or ‘subcortical'), all terms in full or truncated versions. Titles and abstracts were scanned and potentially eligible papers were collected in full-text versions. SMH and WHB independently judged all eligible papers according to the inclusion criteria.
Included Studies
For dementia and cognitive functioning, the search yielded 278 hits, of these 16 were included.11-26 One additional study 27 was added after search of reference lists. For brain imaging abnormalities, the search yielded 228 hits, of these 15 were included.4,23,26,28-40 Multiple papers from the same study population were included only if different types of retinal microvascular changes or outcome measures were examined in separate papers.
We excluded two population-based studies because they reported on a sample size of 207 49 or 96. 50 All patient-based studies had a sample size of more than 50.
Data Extraction
We extracted study characteristics, the ways in which retinal microvascular changes and the outcomes were measured, and factors that were adjusted for in analyses (Table 1). We compiled retinal, cognitive, and brain imaging variables into categories that optimally accommodated the data extracted from the included studies.
Data on the cognitive and brain imaging outcomes are presented in Tables 2a through c. When applicable, results are presented that were adjusted for demographics and vascular risk factors. For dichotomous outcome variables, results are presented as odds ratios (ORs) or hazard ratios (HRs). With regard to dementia, data are presented for any dementia, rather than for a subtype such as Alzheimer's disease or vascular dementia. For continuous variables, standardized regression coefficients (betas) were extracted, or means and standard deviations were converted into Cohen's d 41 to provide estimates of effect sizes. Median ORs or effect sizes are presented in case multiple results were reported for the same measure. In Tables 2a through c, negative effect sizes or betas indicate worse cognition or more brain imaging abnormalities in the group with (more) retinal microvascular changes. Effect sizes <0.2 are considered as small, 0.2 to 0.8 as medium, and >0.8 as large. 41
The results of studies that did not present data that could be converted into effect sizes or ORs/HRs are presented by means of direction of effect. Associations of elevated levels of retinal microvascular changes with worse cognition or more brain imaging abnormalities were indicated by ‘-', association with better cognition or less brain imaging abnormalities were indicated by ’ + ‘, and no statistically significant association between retinal microvascular changes and cognition or brain imaging abnormalities was indicated by ’ = ‘. Results from cross-sectional and longitudinal studies are presented separately, as well as population-based studies and studies with patients with vascular risk factors.
Results are presented separately for population-based studies and patient-based studies.
We intended to perform meta-analyses, using random effect models, to pool the findings of studies that were sufficiently homogeneous with regard to study design, retinal measures, and cerebral outcome measures. Pooled data are presented only when effect sizes between studies were not heterogeneous (examined with I 2 statistic, values near 75% or greater indicate high heterogeneity 42 ). Such meta-analyses only proved to be possible for the cognitive outcome measure of any retinopathy.
RESULTS
Measurements
Retinal measurements
Photographs of the retina were taken after pharmacological pupil dilation, except in the Atherosclerosis Risk in Communities (ARIC) and Cardiovascular Health Study (CHS) studies, in which dark adaptation was used.12,13,15,29-33,40 In the Japanese Elderly Diabetes Intervention Trial (J-EDIT) study, 27 no photographs were taken, but fundoscopy was performed after dilation. Studies reported on the following retinal measures: any retinopathy, arteriovenous nicking, focal narrowing, arteriolar narrowing, and venular widening. Retinopathy, focal narrowing, and arteriovenous nicking were graded visually from photographs. Authors defined any retinopathy as present if any of the following lesions were observed: blot or flameshaped hemorrhages, microaneurysms, soft exudates (cotton wool spots), hard exudates, optic disc swelling, intraretinal microvascular abnormalities, venous beading, new vessels at the disk or elsewhere, vitreous hemorrhage, or laser photocoagulation scars. Three population-based studies (Blue Mountains Eye Study (BMES), 14 Singapore Malay Eye Study (SiMES), 22 and Women's Health Initiative (WHI) 23 ) used a grading scale for diabetic retinopathy that was derived from the ETDRS (Early Treatment Diabetic Retinopathy Study). 43 The definition of any retinopathy in studies comprising patients with diabetes was extended to include fibrous proliferations, preretinal hemorrhage, and retinal detachment.24,27
Retinal arteriolar and venular caliber measurements were performed via computer-assisted techniques and summarized into CRAE (central retinal artery equivalents) or CRVE (central retinal venular equivalents). 44 Some studies used caliber measurements as continuous measures,17–19,34 whereas others categorized these into quartiles,13,14,17 quintiles,4,12,15,29,30,33,37 or deciles. 40 Narrowest arteriolar and widest venular calibers were set against their reference groups. Any retinopathy was assessed in most studies (eight out of nine population-based cohorts and all six patient-based cohorts). Arteriovenous nicking or focal narrowing was assessed in three population-based cohorts and one patient-based cohort. Measures of vessel caliber were assessed in seven population-based and two patient-based cohorts.
Cognitive outcome measures
The relation between retinal microvascular changes and dementia or cognitive functioning was assessed in nine population-based cohorts: Age, Gene/Environment Susceptibility (AGES), 11 ARIC,12,13 BMES, 14 CHS,15,40 Los Angeles Latino Eye Study (LALES), 17 Lothian Birth Cohort 1921, 18 Rotterdam Study,19,20 SiMES,21,22 and WHI, 23 and in three cohorts of patients with diabetes: ET2DM,24,25 J-EDIT, 27 and UDES. 26 Dementia as an outcome measure was used in three population-based cohorts. Diagnostic procedures for dementia assessment consisted of cognitive screening of all participants and a subsequent elaborate examination for the presence of dementia in those who screened positive. A multidisciplinary team of physicians (neurologist, psychiatrist, geriatrician, and neuroradiologist) and psychologists determined final dementia diagnosis, based on standardized criteria for dementia, 45 Alzheimer's disease, 46 or vascular dementia. 47 Four population-based cohorts and one cohort of patients with diabetes reported on cognitive impairment as an outcome measure. Impairment was considered as present if performance was below a predefined cutoff or deviated from normal within the studied population (≤ 2 SD below the mean, or < 10th decile). Five population-based studies and two type 2 diabetes studies reported on one to four of the following cognitive domains: memory, information processing speed, executive functioning, or abstract reasoning 48 (see Appendix for cognitive tests per domain). Three population-based studies and one diabetes study reported on change in cognitive functioning over time. In one study, cognitive domains were measured at four time points, outcome measures were rates of decline per domain and risk of belonging to the 10% most rapid decliners. 13 In another study, risk of incident dementia was rated19,20 and in yet another Modified MMSE (3MSE) scores of each annual time point were compared between groups. 23 One study in patients with diabetes expressed cognitive decline as a regression-based index, controlling for baseline demographics, baseline cognition, and decline in control participants. 26
Description of included studies
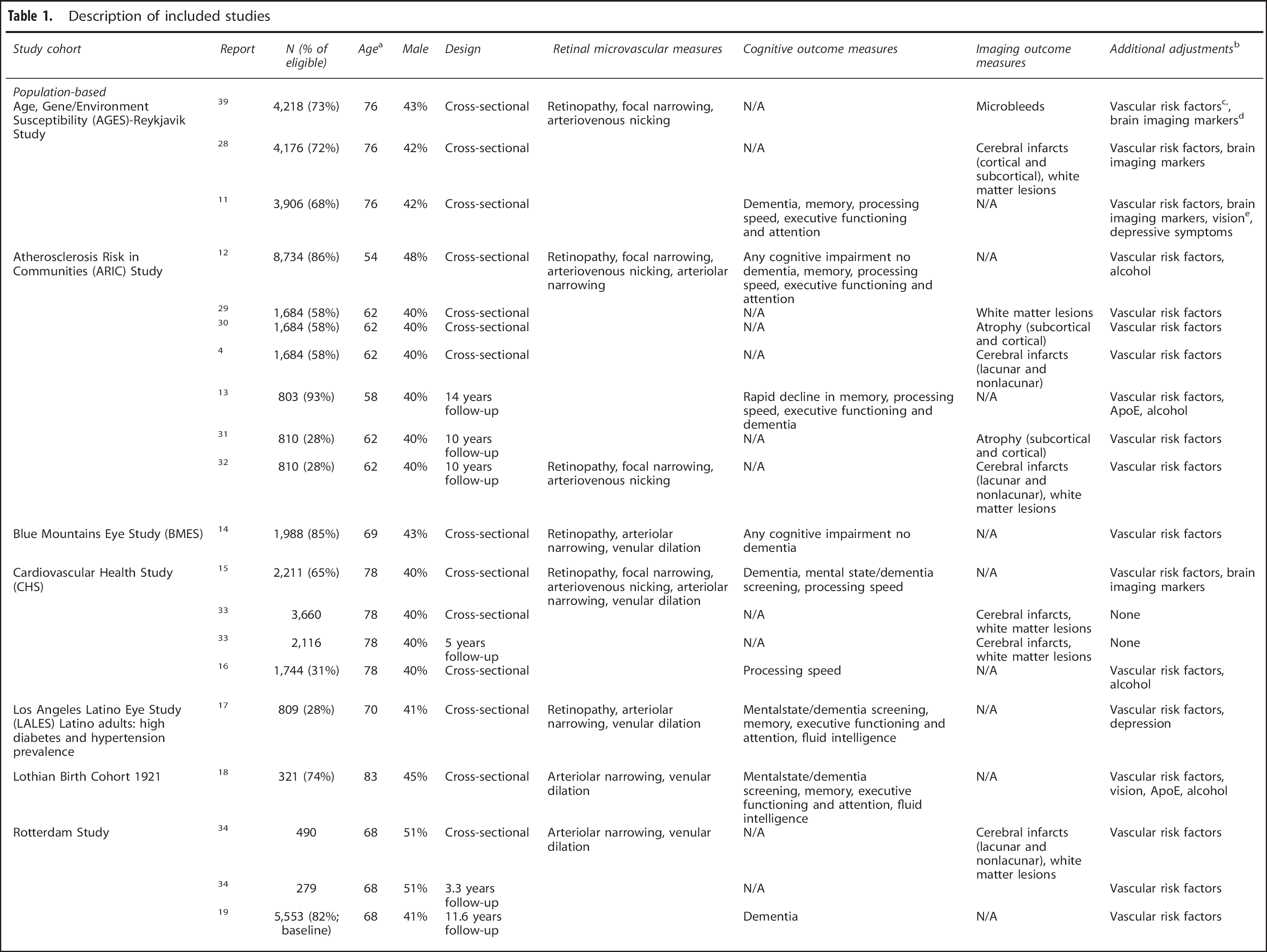
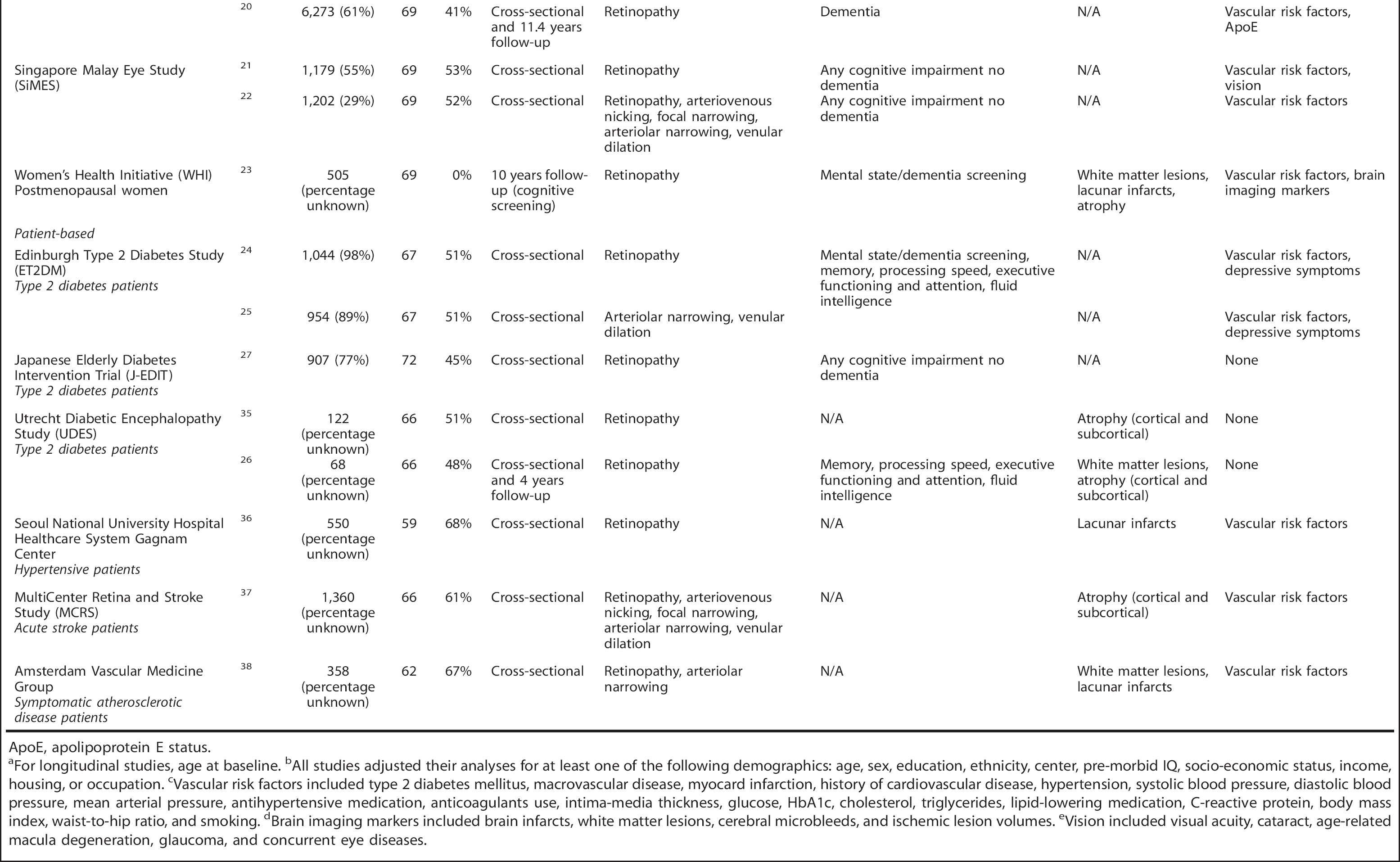
ApoE, apolipoprotein E status.
For longitudinal studies, age at baseline.
All studies adjusted their analyses for at least one of the following demographics: age, sex, education, ethnicity, center, pre-morbid IQ, socio-economic status, income, housing, or occupation.
Vascular risk factors included type 2 diabetes mellitus, macrovascular disease, myocard infarction, history of cardiovascular disease, hypertension, systolic blood pressure, diastolic blood pressure, mean arterial pressure, antihypertensive medication, anticoagulants use, intima-media thickness, glucose, HbA1c, cholesterol, triglycerides, lipid-lowering medication, C-reactive protein, body mass index, waist-to-hip ratio, and smoking.
Brain imaging markers included brain infarcts, white matter lesions, cerebral microbleeds, and ischemic lesion volumes.
Vision included visual acuity, cataract, age-related macula degeneration, glaucoma, and concurrent eye diseases.
Brain imaging measures. Studies on five population-based cohorts (AGES, 28 ARIC,4,29–32 CHS, 33 Rotterdam Study, 34 and WHI 23 ) and four patient-based cohorts (patients with diabetes J-EDIT, 27 UDES,26,35 hypertension, 36 acute stroke, 37 or symptomatic atherosclerosis 38 ) reported on retinal microvascular changes and brain imaging measures. All studies used magnetic resonance imaging (MRI) scans, one study used computed tomography scans in a subgroup of participants. 38 Reported brain imaging abnormalities were infarcts, white matter lesions, and atrophy. In some studies, these were subdivided by type (whole brain or lacunar infarcts) or by location (subcortical or periventricular white matter lesions;subcortical or cortical atrophy). All population-based studies assessed cerebral infarcts and white matter lesions; in two population-based and two patient-based cohorts atrophy was reported.
Several definitions for cerebral infarcts were used, based on lesion size, location, and appearance on different MRI sequences.4,11,28,33,36,51 In Rotterdam 51 and ARIC,4,32 a subset of infarcts was classified as lacunar if they had a diameter of <20 mm and were located in the basal ganglia, internal capsule, thalamus, or deep cerebral white matter. In AGES, 11 infarcts were subdivided into cortical and subcortical (i.e., lacunar) infarcts, whereas in CHS 33 no distinction in types of infarcts was made. In WHI, 23 lacunar infarctions were defined as ischemic lesions located in basal ganglia, as determined by automatic segmentation.
White matter lesions were defined as hyperintense lesions on T2-weighted and FLAIR images,11,23,26,35 on T2-weighted and proton-density images without prominent hypointensity on T1-weighted images, 34 or as white matter signal abnormality on spin-density images; 29 two studies did not specify MRI characteristics of white matter lesions.33,38 In WHI 23 and UDES, 26 ischemic white matter disease volume was determined by automatic segmentation, whereas AGES, ARIC, CHS, and Rotterdam used an ordinal scale for white matter lesion grading. Two studies19,20,28 subdivided white matter lesions into periventricular or subcortical.
For assessing atrophy, ARIC investigators30,31 used a visual grading scale for sulcal widening and ventricular enlargement on T1-, T2-, and proton density-weighted images, independently of focal abnormalities and white matter lesions. One study in hypertensive patients used an atrophy grading scale for computed tomography and MRI scans. 37 In two studies,23,26 T1-weighted volumetric MRI scans were automatically segmented and regional brain volumes were calculated.
Only one study examined microbleeds, 39 defined as a focal area of signal void within the brain parenchyma.
In three population-based cohorts and one cohort of patients with diabetes, changes in brain imaging abnormalities over time were examined (ARIC,31,32 CHS, 33 Rotterdam, 34 and UDES). 26 Incidence of infarcts were measured, as well as changes in white matter lesions defined as progression on a ordinal grading scale. Progression of atrophy was measured in the ARIC study, where it was defined as an increase in ventricular size and sulcal size on a visual ordinal grading scale, and in the UDES study where brain volume changes were calculated.
Associations Between Retinal Microvascular Changes and Dementia or Cognition
Cross-sectional studies: any retinopathy
Nine population-based studies reported on the relation between any retinopathy and cognitive outcomes. Six studies reported on a clinical diagnosis of dementia or impaired cognitive functioning (see Table 2a), all of which found an increased risk (range ORs 1.17 to 5.57), which was statistically significant in two studies. Four studies reported on the relation between any retinopathy and cognitive performance as a continuous measure. These studies found that overall, persons with any retinopathy tended to perform worse on the cognitive tasks, for all cognitive domains. However, effect sizes were small (range -0.25 to 0.03, Table 2a) and associations were statistically significant in only one study.
Pooled random effect models showed that across studies, any retinopathy was significantly associated with the presence of dementia (pooled OR 1.51, 95% confidence interval (CI) 1.14 to 1.99) and cognitive impairment (pooled OR 1.97, 95% CI 1.27 to 3.06), as well as with decreased processing speed (pooled effect size 0.07, 95% CI -0.11 to -0.03).
In eight studies, the modulating role of vascular risk factors was explored by adjusting for a range of factors or by stratifying for hypertension and diabetes. In two studies, the relation between any retinopathy and dementia 15 or cognitive impairment 14 was stronger in participants with hypertension than in those without. One study only found a statistically significant relation between any retinopathy and dementia in persons without diabetes. 15 The highest risk of dementia or cognitive impairment was found in a population of Latinos with a high diabetes and hypertension prevalence. 17 Three studies found no differences according to hypertension or diabetes status.12,20 All three studies that examined dementia found stronger results for vascular dementia or mixed pathology than for ‘pure’ Alzheimer's disease.11,15,20
Regarding patient-based cohorts, studies in patients with type 2 diabetes observed associations within the range of the population-based studies (Table 2b). One study in patients with diabetes found no association between retinopathy and cognitive functioning at baseline or cognitive decline after 4 years.26,35
Cross-sectional studies: focal narrowing, arteriovenous nicking, or retinal vascular caliber
Eight studies describing seven different population-based cohorts reported on the relation between focal narrowing, arteriovenous nicking, or vessel caliber (i.e., generalized arteriolar narrowing or generalized venular widening) and dementia or cognitive functioning. For all measures, an increased risk of dementia or cognitive impairment was found, but ORs were lower compared with any retinopathy (range 1.08 to 2.04; Table 2a). Associations between focal vascular lesions or vascular caliber and domains of cognitive functioning were found in both directions and were mostly not statistically significant. In one study, suboptimal retinal vascular networks, but not focal retinal lesions or retinal caliber, were significantly associated with cognitive functioning. 22
Stratifying for hypertension or diabetes status revealed in one study that the association between retinal caliber and cognitive impairment was driven by persons with hypertension. 17 Another study found that focal narrowing was associated with dementia only in persons with diabetes. 15 Patient-based data were available of one study, the authors found no relation between retinal caliber and cognitive functioning 25 (Table 2b).
Longitudinal studies
In three population-based cohorts, retinal microvascular changes in relation to incident dementia or to accelerated cognitive decline 10 to 14 years later were examined (see Table 2c). Any retinopathy was not related to risk of dementia (Rotterdam study 20 ), but associations were found with an increased risk of decline in processing speed and executive functioning and attention (OR 2.18 and 1.33, respectively) (ARIC 13 ), and with poorer performance on a cognitive screening instrument (3MSE) throughout follow-up (WHI 23 ). Focal narrowing was associated with a decline in executive functioning (ARIC 13 ). Arteriolar narrowing was unrelated to dementia risk or cognitive decline.13,19 Venular widening was associated with incident dementia in the Rotterdam study (HR 1.11), 19 but unrelated to cognitive decline in ARIC. 13 One study in patients with diabetes found no association between diabetic retinopathy and cognitive change after 4 years. 26
Associations between retinal microvascular changes and outcome measures of cognitive functioning and MRI abnormalities in cross-sectional population-based studies
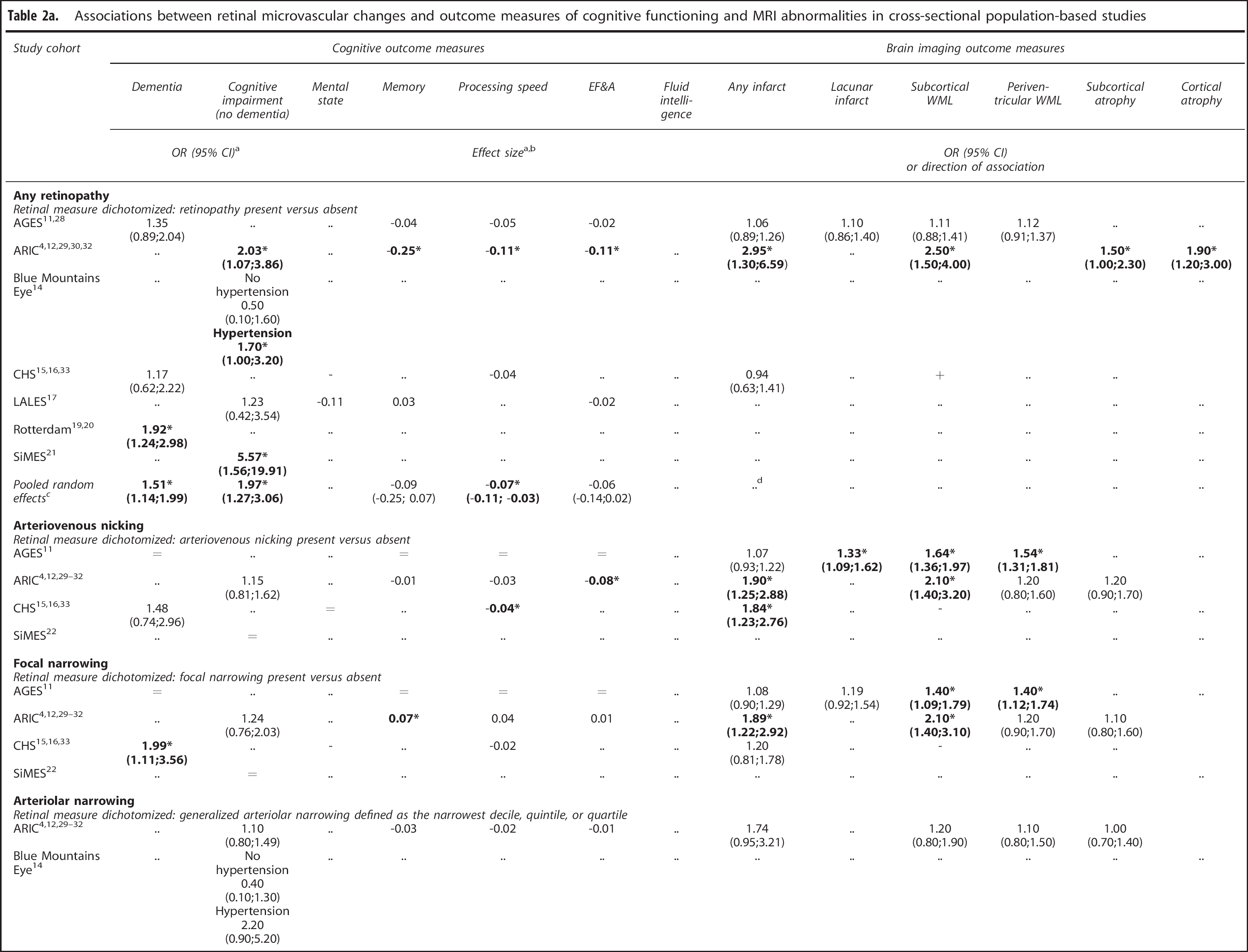

EF&A, Executive functioning and attention; WML, white matter lesions; OR, odds ratio; CI, confidence interval; MRI, magnetic resonance imaging; AGES, Age, Gene/Environment Susceptibility; ARIC, Atherosclerosis Risk in Communities; CHS, Cardiovascular Health Study; LALES, Los Angeles Latino Eye Study; SiMES, Singapore Malay Eye Study. *P<0.05; .., outcome measure not evaluated; -, outcome measure evaluated, presence of retinal microvascular changes associated with worse cognition or more white matter lesions; +, outcome measure evaluated, presence of retinal microvascular changes associated with better cognition or less white matter lesions; =, no association between presence of retinal microvascular changes and outcome measure.
Median ORs or effect sizes are presented in case multiple results were reported for the same measure.
Expressed as Cohen's d or standardized regression coefficients.
Data were pooled for studies investigating any retinopathy and cognitive outcome measures, because data were available from multiple studies that were similar in design, weighted ORs or effect sizes are presented for fully adjusted data.
Studies investigating any retinopathy in relation to infarcts were too heterogeneous (I 2 69%, P = 0.04).
Results for arteriolar caliber were recoded in such a way that a negative coefficient means arteriolar narrowing is associated with worse cognition or more brain imaging abnormalities.
Bold values indicate significant associations
Associations between retinal microvascular changes and outcome measures of cognitive functioning in patient-based studies

EF&A, Executive functioning and attention; WML, white matter lesions; OR, odds ratio; CI, confidence interval; MCRS, MultiCenter Retina and Stroke Study; UDES, Utrecht Diabetic Encephalopathy Study; J-EDIT, Japanese Elderly Diabetes Intervention Trial; white matter lesions. *P<0.05;.., outcome measure not evaluated; -, outcome measure evaluated, presence of retinal microvascular changes associated with worse cognition or more white matter lesions; +, outcome measure evaluated, presence of retinal microvascular changes associated with better cognition or less white matter lesions; =, no association between presence of retinal microvascular changes and outcome measure.
Expressed as Cohen's d or standardized regression coefficients.
Results for arteriolar caliber were recoded in such a way that a negative coefficient means arteriolar narrowing is associated with worse cognition.
Associations between retinal microvascular changes and outcome measures of cognitive functioning and MRI abnormalities in longitudinal population-based studies
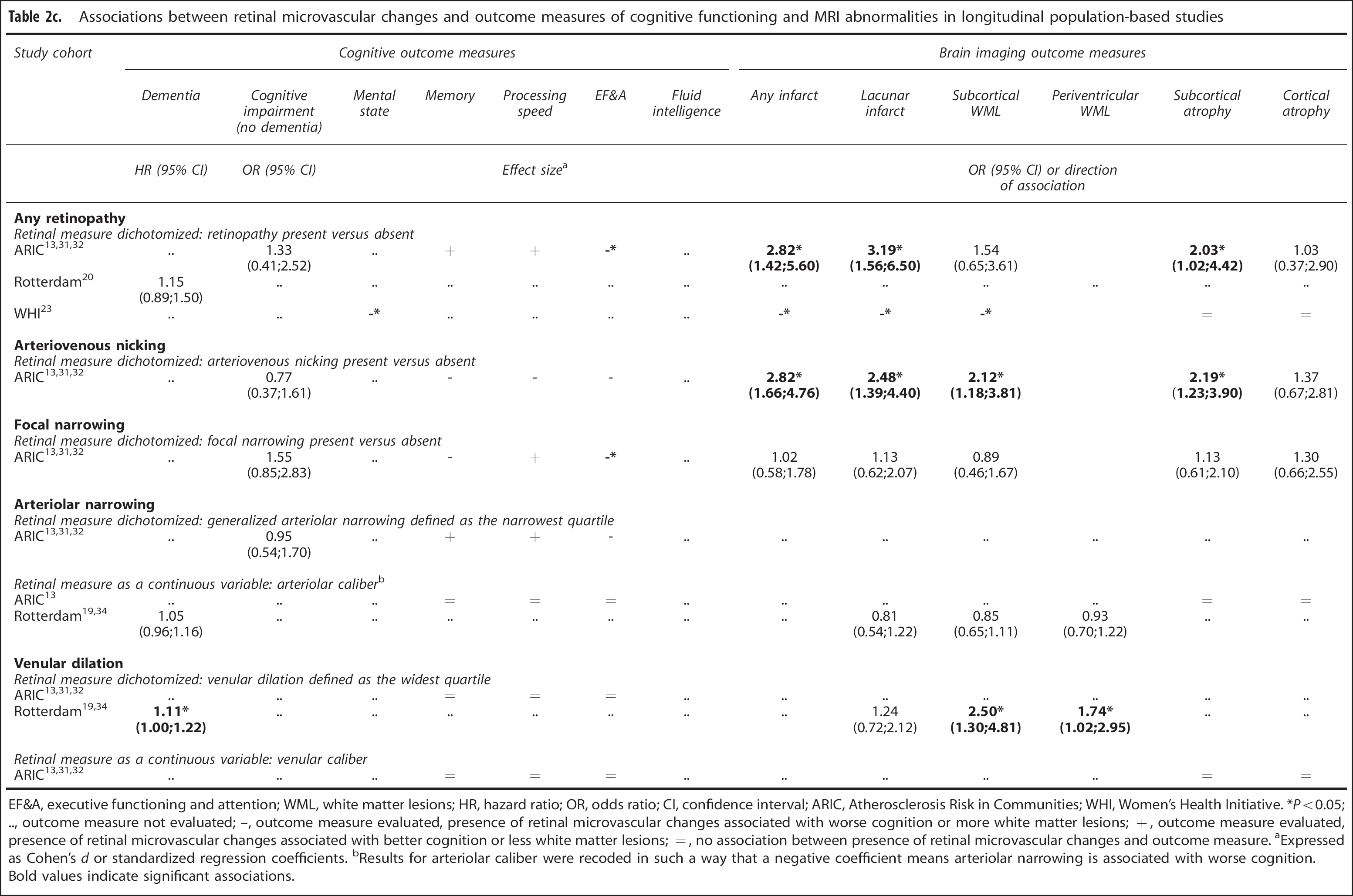
EF&A, executive functioning and attention; WML, white matter lesions; HR, hazard ratio; OR, odds ratio; CI, confidence interval; ARIC, Atherosclerosis Risk in Communities; WHI, Women's Health Initiative. *Po0.05; .., outcome measure not evaluated; –, outcome measure evaluated, presence of retinal microvascular changes associated with worse cognition or more white matter lesions; +, outcome measure evaluated, presence of retinal microvascular changes associated with better cognition or less white matter lesions; =, no association between presence of retinal microvascular changes and outcome measure. aExpressed as Cohen's d or standardized regression coefficients. bResults for arteriolar caliber were recoded in such a way that a negative coefficient means arteriolar narrowing is associated with worse cognition.
Bold values indicate significant associations.
Two studies with longitudinal data adjusted or stratified for vascular risk factors;the results did not change.13,20
Associations Between Retinal Microvascular Changes and Brain Imaging
Cross-sectional studies: any retinopathy
Three population-based studies reported on the relation between any retinopathy and brain imaging abnormalities. Only ARIC31,32 found significant associations for infarcts, white matter lesions, and atrophy (ORs from all studies ranged from 0.94 to 2.95, Table 2a). One study found an association between retinopathy and multiple microbleeds (OR 1.75; CI 1.25 to 2.45). 39
After correction for vascular risk factors, associations with all brain imaging outcomes attenuated slightly. Stratifying for hypertension or diabetes status showed that, overall, associations between any retinopathy and imaging abnormalities were stronger in persons with hypertension or diabetes than in those without.
In patient-based studies (see Table 2b), retinopathy was associated with infarcts in a cohort of patients with hypertension; 36 with lacunar infarcts and white matter lesions in a cohort of patients with symptomatic atherosclerotic disease; 38 with cortical atrophy in a cohort of patients with diabetes;26,35 and with subcortical atrophy in a cohort of patients with acute stroke 15 (ORs within range of population-based results).
Cross-sectional studies: focal narrowing, arteriovenous nicking, or retinal vascular caliber
Focal narrowing and arteriovenous nicking were significantly associated with brain imaging abnormalities in three out of three population-based cohorts for infarcts (whole brain or lacunar) and white matter lesions (subcortical or periventricular), ORs ranged from 1.07 to 2.10 (Table 2a). Associations with atrophy were not significant. One study found an association between focal narrowing and multiple microbleeds (OR 1.45; CI 1.01 to 2.09), and between arteriovenous nicking and multiple microbleeds (OR 1.44; CI 1.06 to 1.95). 39
Associations of arteriolar narrowing and venular dilation with imaging abnormalities were generally weaker compared with the other retinal measures. In three out of three population-based cohorts, effects were very small and not significant, for all imaging abnormalities (OR range 1.05 to 1.74).
As with any retinopathy, associations with brain imaging outcomes attenuated slightly after correction for vascular risk factors. In a cohort of patients with symptomatic atherosclerotic disease, arteriolar narrowing was associated with lacunar infarcts and white matter lesions. 38 No significant associations with atrophy were found in acute stroke patients (ORs range 0.70 to 1.40;Table 2b).
Longitudinal studies
In three population-based cohorts, retinal microvascular changes were examined in relation to progression of brain imaging abnormalities 3 to 10 years later. Any retinopathy at baseline was related to an increased risk of (lacunar) infarcts, white matter lesions or subcortical atrophy at follow-up, in one or two of two study cohorts (median OR 2.03;Table 2c). Focal narrowing and arteriovenous nicking were examined only in ARIC. Arteriovenous nicking was significantly related to incident (lacunar) infarcts, white matter lesions, or subcortical atrophy (median OR 2.19), whereas focal narrowing was unrelated to progression of brain imaging abnormalities (median OR 1.13). Regarding measures of caliber, only venular dilation was related to white matter lesion progression, and, in unadjusted analyses only, to lacunar infarcts (arteriolar narrowing: median OR 0.85;venular dilation: median OR 1.74). For none of the retinal microvascular changes, a relation with cortical atrophy after follow-up was found.
In a cohort of patients with diabetes, no associations between retinopathy and brain imaging changes after follow-up were found.26,35
Four longitudinal studies adjusted or stratified for vascular risk factors and found varying results. After adjusting, the relation attenuated in some cases (between venular widening and incident infarcts 34 ) became stronger in others (between retinopathy and arteriovenous nicking and subcortical atrophy 31 ) or did not change.23,31 One study found that any retinopathy was more strongly associated with incident infarcts and white matter lesion progression 10 years later in participants with diabetes and without hypertension, whereas for arteriovenous nicking this was the case in participants without diabetes and with hypertension. 32
DISCUSSION
The relations between microvascular changes in the retina and dementia or cognitive functioning and between these microvascular changes and brain imaging abnormalities have been examined in several large cohort studies in the last decade. Despite considerable heterogeneity in measures of both retinal microvascular changes and the cerebral outcomes, cross-sectional studies found consistent and moderately strong associations with dementia, with modest decrements in cognitive functioning in nondemented people, as well as with brain imaging abnormalities. This supports a role for vascular disease as the underlying pathophysiology of cognitive decline and brain changes. The few available longitudinal studies found associations with progression of brain imaging abnormalities, but only marginally with incident dementia or cognitive decline.
In the light of the results of this systematic review, some methodological issues warrant consideration. First, a formal meta-analysis was not performed, because of the heterogeneity of study designs and outcome measures. Second, studies with negative results may have been underrepresented due to the effects of publication bias. Third, no information was available on the cognitive domains language or perception, because these were tested in none of the studies.
Moreover, compared with the cross-sectional studies, longitudinal studies were fewer in number and tended to have lower power to find associations between the retinal microvascular changes and the cerebral outcomes. Nevertheless, in longitudinal studies somewhat weaker associations were found than in cross-sectional studies, especially with regard to dementia and cognitive decline. If this is a real effect, then it might be caused by competing risk effects of mortality. It is also conceivable that retinal vascular pathology occurs at the same time as clinical symptoms of dementia or cognitive impairment and is a relatively late phenomenon in the etiological cascade. 20 At present, in our view, no definite conclusions with regard to the prospective associations between retinal microvascular changes and the cerebral outcomes should be drawn and a key target for future studies is to obtain reliable longitudinal data from large data sets.
The reported association between retinal vascular changes and adverse cerebral outcomes conveys an important etiological message. Abnormalities in the retinal vasculature can be considered as a marker of a disadvantageous vascular risk factor profile and of an individual's vulnerability to adverse outcomes. Indeed, the measures of the retinal vessels that are described in this review reflect increasing exposure to high blood pressure. Arteriolar narrowing and venular dilation are early consequences of a rise in blood pressure. Focal narrowing and arteriovenous nicking develop later, in the phase of chronic hypertension. When blood pressure rises further and requires acute treatment, the blood-retinal barrier breaks down and signs of retinopathy occur, such as hemorrhages, hard exudates, and cotton-wool spots.5,34 A similar gradient in severity of pathologies is reflected in associations between retinal vascular changes and systemic vascular disease. Milder retinal vascular changes such as generalized arteriolar narrowing, focal narrowing, and arteriovenous nicking are weakly related to an increased risk of cerebrovascular and cardiovascular disease, whereas this relation is stronger for moderately severe forms such as retinopathy signs.52-54 These findings corroborate the results of this review in that the strength of the association with cognition and brain imaging abnormalities increases as the vascular disease process progresses: associations with adverse outcomes were relatively weak for vascular caliber measurements, intermediate for focal narrowing and arteriovenous nicking, and stronger for any retinopathy. The role of vascular disease is also illustrated by the fact that associations with cognitive impairment and brain infarcts were stronger in persons with hypertension. A similar finding is reported for diabetes mellitus: in persons with diabetes, retinopathy signs were more strongly associated with adverse outcomes than in persons without diabetes.
Another finding of etiological significance is the fact that retinal vascular changes are associated with multiple types of brain imaging abnormalities, including atrophy, several manifestations of small vessel disease, as well as large vessel infarcts. This may reflect the fact that retinal and cerebral pathologies share common risk factors and etiologies. It is also possible that retinal vascular changes and brain disease are the result of several etiological pathways developing simultaneously in the context of aging and an adverse vascular risk factor profile.
The prognostic value of a risk marker depends on its prevalence and on the strength of the association with the outcomes. Associations found for any retinopathy, which reflects moderately severe microvascular disease, were strongest, but the prevalence of retinopathy is only 2% to 15% in the general population. 55 Changes in retinal vascular caliber are milder and more common, but were only weakly related to cerebral outcomes. Consequently, only a small proportion of cases of dementia, cognitive impairment, or brain changes in the population is attributable to variations in retinal vasculature. Therefore, at the population level, retinal vascular assessment is not considered to be a useful predictor for adverse cerebral outcomes by itself. At best it can be incorporated in prediction models together with other predictors, although there is doubt whether this can provide vital prognostic information in addition to traditional risk factors. 56 Nevertheless, presence of more severe retinal microvascular abnormalities can still be used to identify individuals at elevated risk.
Some contrasts in strengths of associations deserve attention because they provide insight in the nature of the relation between retinal microvascular changes and the outcomes. First, stronger cross-sectional associations were found in the middle-aged ARIC population (mean baseline ages ranging from 54 to 62 years), than in other populations with an age range from 69 to 83 years. These findings are in line with an age-related decrease in the strength of associations that have been found for vascular risk factors and blood pressure 57 and cerebrovascular and cardiovascular disease 53 in relation to retinopathy signs, as well as for vascular risk factors such as hypertension, diabetes, dyslipidemia, and obesity in relation to dementia. 58 Second, associations with retinal microvascular changes were stronger for dementia and other definitions of severe cognitive impairment, than for milder forms of cognitive dysfunction. This differential relation most likely reflects the fact that subtle cognitive decrements that can be found in nondemented populations do not necessarily evolve into frank cognitive decline in all individuals. Within the population of older persons with microvascular damage, severe cognitive decline probably occurs only in a subgroup of persons, possibly in interaction with other risk factors. 59 Third, concerning brain infarcts, relations between retinal microvascular changes and lacunar infarcts were slightly stronger than with any brain infarcts, which supports the idea that small vessel disease is one of the underlying pathophysiological mechanisms.
In conclusion, there is a consistent relation between the presence of retinal microvascular changes and dementia, cognitive impairment, and brain imaging abnormalities, although more prospective data are needed. Associations are strongest for more severe retinal vascular abnormalities, supporting a role for vascular disease as the underlying pathophysiology of cognitive decline and brain changes. The strength of the relation is modest, particularly for more common retinal microvascular abnormalities. This limits the prognostic value at the population level, however, retinal abnormalities may identify individuals at risk and do offer an important window on the brain for etiological studies.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
Footnotes
APPENDIX
Classification of cognitive domains and included tests
