Abstract
Type 2 diabetes has been linked to cognitive impairment, but its potential metabolic mechanism is still unclear. The present study aimed to explore neuron–astrocyte metabolic cooperation in the brain of diabetic (db/db, BKS.Cg-m+/+ Leprdb/J) mice with cognitive decline using 13C NMR technique in combination with intravenous [2-13C]-acetate and [3-13C]-lactate infusions. We found that the 13C-enrichment from [2-13C]-acetate into tricarboxylic acid cycle intermediate, succinate, was significantly decreased in db/db mice with cognitive decline compared with wild-type (WT, C57BLKS/J) mice, while an opposite result was obtained after [3-13C]-lactate infusion. Relative to WT mice, db/db mice with cognitive decline had significantly lower 13C labeling percentages in neurotransmitters including glutamine, glutamate, and γ-aminobutyric acid after [2-13C]-acetate infusion. However, [3-13C]-lactate resulted in increased 13C-enrichments in neurotransmitters in db/db mice with cognitive decline. This may indicate that the disturbance of neurotransmitter metabolism occurred during the development of cognitive decline. In addition, a reduction in 13C-labeling of lactate and an increase in gluconeogenesis were found from both labeled infusions in db/db mice with cognitive decline. Therefore, our results suggest that the development of cognitive decline in type 2 diabetes may be implicated to an unbalanced metabolism in neuron–astrocyte cooperation and an enhancement of gluconeogenesis.
Introduction
Type 2 diabetes (T2D) is a metabolic disorder characterized by hyperglycemia due to impaired insulin secretion or insulin resistance. T2D and its complications are increasingly becoming considerable public health problems. In addition to complications related to kidneys, eyes, heart, and blood vessels, T2D has also been associated with central nervous system (CNS) damage and cognitive impairment.1–4 Several potential mechanisms relating T2D-associated cognitive decline have been proposed. Hyperglycemia can induce neuronal apoptosis and dysfunction through augmented oxidative stress, increased deposition of advanced glycation end-products, excessive release of cytokines, activation of protein kinase C, increased flux of the polyol pathway, reduced level of neurotrophins, and pro-inflammatory processes. 5 An elevated insulin level was also found to adversely affect cognitive function.6,7 Manschot et al. 8 reported that T2D-associated cognitive impairment may be associated with white matter lesion, atrophy, and infarct. In addition, magnetic resonance imaging data showed that a reduction in hippocampal volumes of T2D patients may result in memory impairment. 9 However, brain metabolic mechanisms underlying T2D-associated cognitive decline are still far from being fully understood.
Since normal brain function requires a stable energy supply, disturbance of brain energy metabolism has been associated with accelerated cognitive impairment. 10 Glucose has long been considered as the main substrate for neuronal energy metabolism in the mammalian brain. 11 However, this conventional view has been challenged by the astrocyte–neuron lactate shuttle (ANLS) hypothesis first proposed by Pellerin and Magistretti. 12 According to the ANLS hypothesis, glucose is predominantly metabolized to lactate in astrocytes and lactate released from astrocytes is subsequently transported to neurons as the primary metabolic fuel. 12 After this, lactate as a neuronal energy source was also verified both in vitro 13 and in vivo. 14 Relative to glucose, lactate was found to be equivalent regarding its access to the tricarboxylic acid (TCA) cycle 15 and also serve as a precursor for the synthesis of neurotransmitters, γ-aminobutyric acid (GABA) and glutamate, and maintain normal glutamatergic and GABAergic function. 16 Thus, further understanding of brain energy metabolism in neurons and astrocytes is of great interest and importance for exploring the pathogenesis of neurological disorders. 17
13C nuclear magnetic resonance (NMR) technique is a promising tool for investigating the metabolism in specific astrocytes and neurons and their interactions. 18 Astrocytes and neurons possess different enzyme and transport systems, resulting in different metabolic processes between them. For instance, acetate can be selectively taken up by a specific transport system in astrocytes, yet glucose enters the TCA cycle more in neurons than in astrocytes. 19 Therefore, information about neuronal and astrocytic metabolism can be recorded by 13C NMR spectroscopy combined with injection of 13C-labeled glucose or acetate. 20 Recently, we used 13C NMR with [1-13C]-glucose and [2-13C]-acetate infusion to study the changes in brain energy metabolism during type 1 diabetes (T1D) development. 21 The 13C incorporation into glutamine, glutamate, and GABA was increased from [2-13C]-acetate and reduced from [1-13C]-glucose in one-week T1D rats, suggesting that mitochondrial metabolism was enhanced in astrocytes and impaired in neurons. In addition, we also found that pyruvate recycling pathway was enhanced in one-week T1D rats, but inhibited in 15-week T1D rats. 21 However, Tyson et al. 22 reported that lactate was primarily metabolized in neurons but glucose was ubiquitous. Therefore, for investigating neuronal metabolism lactate had a significant improvement as compared with glucose. 22 In the present study, we used 13C NMR spectroscopy in combination with intravenous [2-13C]-acetate and [3-13C]-lactate infusions to explore neuron–astrocyte metabolic cooperation in the brain of db/db mice with cognitive decline.
Materials and methods
Animals
Twelve-week-old male diabetic (db/db; BKS.Cg-m+/+ Leprdb/J; body weight = 46.91 ± 5.56; n = 20) and wild-type (WT; C57BLKS/J; body weight = 24.65 ± 1.34; n = 20) mice were purchased from the Mode Animal Research Center of Nanjing University. db/db mice are homozygous mutant (leptin receptor deficient) mice originated from C57BLKS/J mice. Thus, these two types of mice used in the present study have the same genetic background of C57BLKS. Relative to WT mice, db/db mice show typical characteristics of T2D, such as polyphagic, polydipsic, and polyuric. Moreover, db/db mice are always obese and hyperglycemia. All mice were housed in a specific pathogen-free colony with regulated temperature and humidity and a 12:12 h light–dark cycle (lights-on at 8:00 a.m.), and given free access to standard rat chow and tap water at the Laboratory Animal Center of Wenzhou Medical University (Wenzhou, China). This study was conducted on the basis of the Guide for the Care and Use of Laboratory Animals and approved by the Institutional Animal Care and Use Committee of Wenzhou Medical University. Experiments were reported according to the ARRIVE guidelines.
Morris water maze (MWM) test
After five weeks, the MWM test was performed according to a previously published method, 23 with a minor modification. In brief, the test was conducted in a circular pool (diameter=110 cm, height=30 cm) filled with opaque water and kept at 26 ± 1 ℃. The circular escape platform (diameter=7 cm) was submerged 1 cm below the surface of the water. Mice were trained to reach the escape platform for four continuous days (four trials per day). If mice cannot find the platform within 60 s, they were guided to be there by the operator. On the last day, trained mice were tested in a single 90 s probe trial without the platform. The escape latency, swimming path length, and the number of the original platform location crossings were recorded by a computer system.
Infusion techniques
Prior to infusion, mice were fasted overnight (12 h) and given free access to tap water. The db/db and WT mice were randomly assigned to [2-13C]-acetate and [3-13C]-lactate infusion groups (n=10 for each group), respectively. The animals were anesthetized and then the left jugular veins were cannulated for infusion of labeled substrates. In this study, [2-13C]-acetate and [3-13C]-lactate were infused at a continuous rate of 0.18 and 0.11 ml/kg/min using a syringe pump, respectively. During infusion, mice were kept under a glow lamp to maintain rectal temperature at 36.8–37.5 ℃.
Sample collection and preparation
Owing to the low abundance of 13C, a substantial amount of samples was required to achieve sufficient sensitivity in a 13C NMR spectrum. Thus, whole brain tissues were collected for 13C NMR analysis. In this study, at 15 min after infusion, mice were sacrificed by decapitation and whole brains were dissected immediately, frozen in liquid nitrogen, and stored at -80 ℃ until use. The preparation of brain tissue extracts was conducted using the methanol–chloroform method as described in our previous work. 21 In brief, the tissue sample was weighed into a centrifuge tube, added with ice-cold methanol (4.00 ml/g) and distilled water (0.85 ml/g), and homogenized by vortex at 4 ℃. Then, the mixture was added with chloroform (2.00 ml/g) and distilled water (2.00 ml/g), mixed by vortex, and kept on ice for 15 min. Subsequently, the final mixture was centrifuged at 1000g for 15 min at 4 ℃ and the supernatant was extracted and lyophilized for about 24 h. The lyophilized sample was resuspended in 500 μl D2O and transferred to a 5 mm tube for NMR measurement.
Acquisition of 13C NMR spectra
13C NMR spectra of brain tissue extracts were carried out on a Bruker AVANCE III 600 MHz NMR spectrometer operating at 150.92 (C) MHz at 298 K. An inverse-gated decoupling sequence (INVGATE) was applied to avoid the Nuclear Overhauser effect. In addition, the main parameters included: flip angle, 30 °; spectral width, 33,333 Hz; data points, 64 K; acquisition time, 1 s; delay time, 2 s; number of scans, 32,768.
Data analysis
In this study, all mice were randomly assigned to the experimental procedures including housing and feeding, the MWM test, infusion, and sample collection. Data acquisition was performed by masking the group of the animals. The specific 13C-enrichment of metabolites after [2-13C]-acetate and [3-13C]-lactate infusions was calculated on the basis of the natural abundance of 13C (1.1%). Prior to statistical analysis, data were evaluated for normal distribution using the Anderson−Darling test (PROC UNIVARIATE; P value < 0.05) in SAS software (SAS 9.2, SAS Institute Inc., Cary, NC, USA), and log-transformed if nonnormality was obtained. Then, the statistical difference between two groups was analyzed by Student's t-test (PROC ANOVA) and a P<0.05 was considered as a statistically significant difference.
Results
Impaired learning and memory in db/db mice
Compared with age-matched WT mice, 17-week-old db/db mice displayed obvious changes of characteristics, such as shiny hair, inactivity, dullness, low pain sensitivity, and poor wound healing ability. In the hidden-platform trials, a longer escape latency was observed in db/db mice throughout the four-day training period than WT mice; however, there was no significant difference between different training days (Figure 1(a)). In WT mice, the escape latency on day 3 was significantly reduced relative to that on day 1, and on day 4 the escape latency was significantly shorter than on days 1 and 2 (Figure 1(a)). Figure 1(b) shows the swimming path of WT and db/db mice in the probe test. It can be seen from Figure 1(c) that db/db mice had significantly longer swim length to cross the original platform location compared with WT mice. Moreover, during the probe test, db/db mice crossed the original platform location fewer times than WT mice, as shown in Figure 1(d). Taken together, results based on the MWM test indicated impairment of learning and memory in db/db mice. In the present study, all db/db mice were accompanied with cognitive decline. Therefore, we examined the metabolic cooperation between astrocytes and neurons in the brain of db/db mice with cognitive decline using 13C NMR spectroscopy combined with intravenous [2-13C]-acetate and [3-13C]-lactate infusions.
Performance of WT and db/db mice in the Morris water-maze test: (a) escape latency to reach the hidden platform in the water maze during the four-day training period, (b) swimming path, (c) swimming length, and (d) number of the original platform location crossings in the probe test. Significant level: *P < 0.05; **P < 0.01; ***P < 0.001.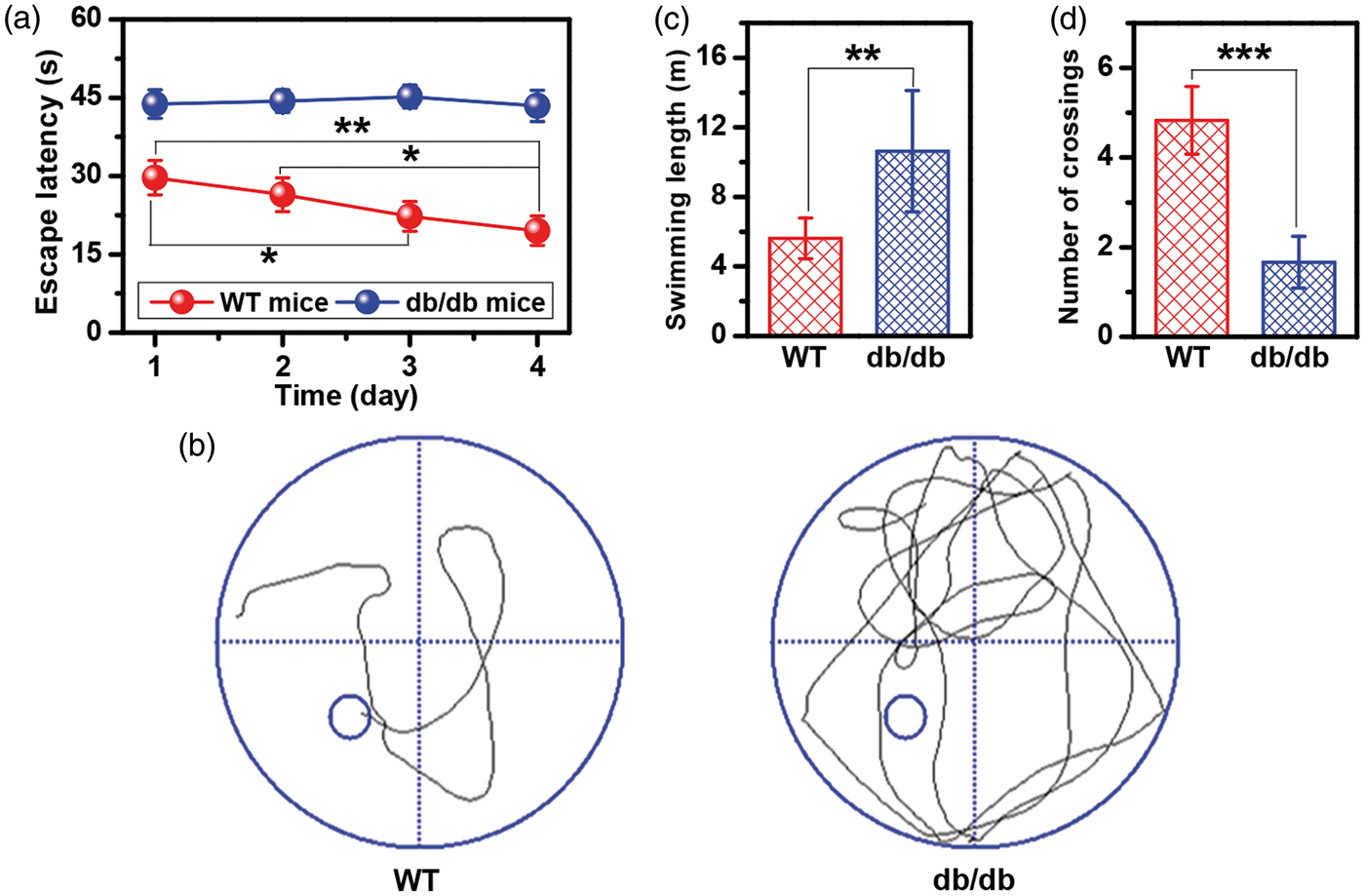
13C NMR spectra in the brain extracts of WT mice and db/db mice with cognitive decline
Figure 2 illustrates typical 13C NMR spectra obtained from brain tissue extracts of WT mice and db/db mice with cognitive decline infused with [2-13C]-acetate and [3-13C]-lactate. It can be seen that the 13C isotope has been successfully incorporated into the specific carbon sites of metabolites including aspartate (Asp: C2, δ53.07; C3, δ37.36), succinate (Suc: C2/C3, δ35.05), glutamine (Gln: C2, δ54.98; C3, δ27.05; C4, δ31.65), glutamate (Glu: C2, δ55.50; C3, δ27.77; C4, δ34.33), γ-aminobutyric acid (GABA: C2, δ35.23; C3, δ24.42; C4, δ40.13), and lactate (Lac: C2, δ69.33; C3, δ20.87). However, acetate (Ace: C2, δ24.30) and alanine (Ala: C3, δ16.95) were only derived from [2-13C]-acetate and [3-13C]-lactate infusions, respectively.
Typical 13C NMR spectra of brain tissue extracts in mice infused with [2-13C]-acetate (a, wild-type; b, db/db) and [3-13C]-lactate (c, wild-type; d, db/db). Assignments: acetate (Ace): C2, 24.30 ppm; alanine (Ala): C3, 16.95 ppm; aspartate (Asp): C2, 53.07 ppm, C3, 37.36 ppm; succinate (Suc): C2/C3, 35.05 ppm; glutamine (Gln): C2, 54.98 ppm, C3, 27.05 ppm, C4, 31.65 ppm; glutamate (Glu): C2, 55.50 ppm, C3, 27.77 ppm, C4, 34.33 ppm; γ-aminobutyric acid (GABA): C2, 35.23 ppm, C3, 24.42 ppm, C4, 40.13 ppm; lactate (Lac): C2, 69.33 ppm, C3, 20.87 ppm.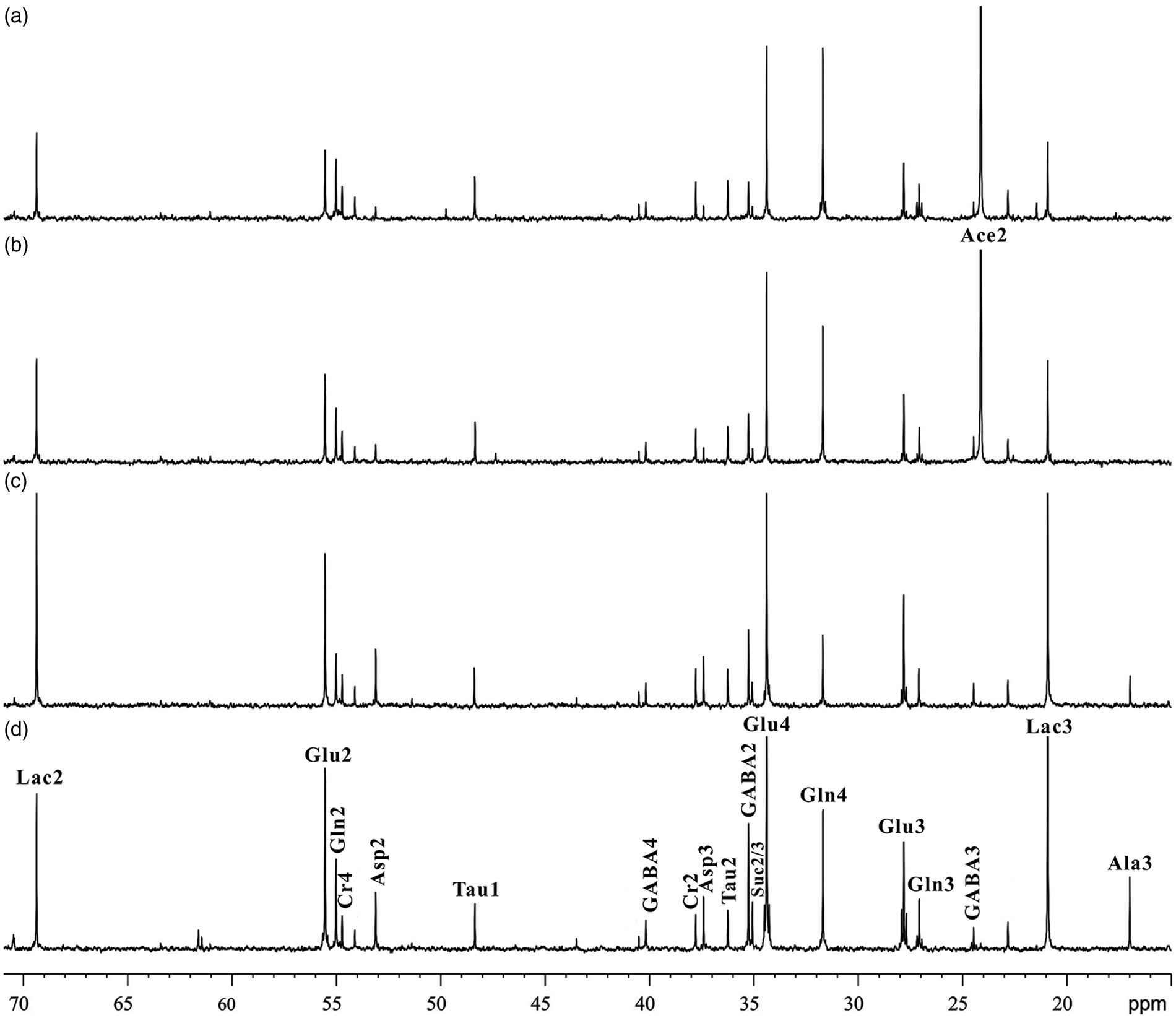
Metabolism of [2-13C]-acetate in the brain of WT mice and db/db mice with cognitive decline
Figure 3 illustrates [2-13C]-acetate metabolism in the brain of WT mice and db/db mice with cognitive decline. Firstly, [2-13C]-acetate was converted into acetyl coenzyme A in astrocytes, which labeled astrocytic TCA cycle intermediates, such as Suc C2, and subsequently Asp C2, Lac C2, Glu C4, and Gln C4. Then, neuronal Glu C4 and GABA C2 were subsequently labeled by astrocytic Gln C4. In addition, the C3 of Asp, the C3 of Lac, the C2 and C3 of Glu, the C2 and C3 of Gln, and the C3 and C4 of GABA were labeled after the second turn of astrocytic TCA cycle. Relative to WT mice, a significantly reduced flux of 13C from [2-13C]-acetate into the C2 and C3 of Suc was observed in the brain of db/db mice with cognitive decline (1.80 ± 1.30 versus 3.22 ± 0.94, P = 0.02), indicating that cognitive decline may be associated with a down-regulation of astrocytic TCA cycle (Figure 3). For db/db mice with cognitive decline, the 13C-enrichment in all carbon positions of Gln was significantly reduced as compared with WT mice (C2, 3.15 ± 1.11 versus 5.19 ± 0.88, P = 0.0005; C3, 2.40 ± 0.98 versus 3.89 ± 0.55, P = 0.0007; C4, 7.26 ± 3.75 versus 13.06 ± 2.13, P = 0.0007). The 13C-enrichment in the C2 position of Glu was decreased significantly in db/db mice with cognitive decline relative to WT mice (2.26 ± 0.65 versus 2.94 ± 0.55, P = 0.03). Moreover, db/db mice with cognitive decline had a lower 13C labeling percentage in the C2 of GABA than WT mice (2.80 ± 1.45 versus 4.21 ± 0.88, P = 0.02). In WT mice, the 13C-enrichment of Lac was increased in the C2 and C3 positions (C2, 2.30 ± 0.48 versus 1.64 ± 0.33, P = 0.006; C3, 2.12 ± 0.25 versus 1.39 ± 0.36, P = 0.0001) compared with db/db mice with cognitive decline (Figure 3). No significant differences were found in Asp C2 and C3 enrichments.
Metabolism of [2-13C]-acetate in the brain of wild-type (red bar) and db/db (blue bar) mice. Metabolites: Ace, acetate; Asp, aspartate; GABA, γ-aminobutyric acid; Gln, glutamine; Glu, glutamate; Lac, lactate; OAA, oxaloacetate; Pyr, pyruvate; Suc, succinate; α-KG, α-ketoglutarate. Significant level: *P < 0.05; **P < 0.01; ***P < 0.001.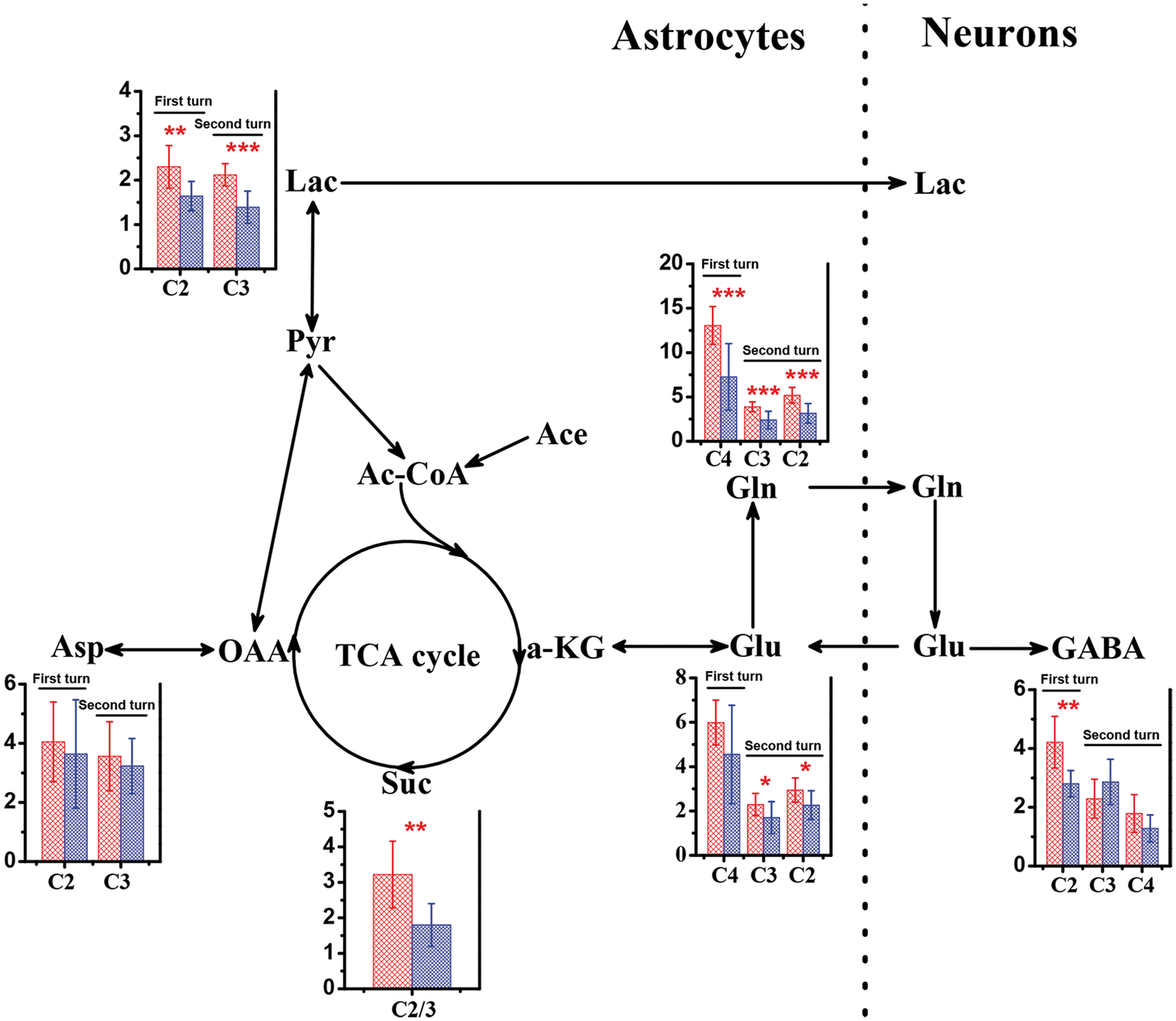
Metabolism of [3-13C]-lactate in the brain of WT mice and db/db mice with cognitive decline
Figure 4 shows [3-13C]-lactate metabolism in the brain of WT mice and db/db mice with cognitive decline. [3-13C]-lactate was firstly metabolized to [3-13C]-pyruvate via glycolysis and then converted to Ala C3 by amino-transferase or introduced to neuronal TCA cycle via pyruvate carboxylase and pyruvate dehydrogenase, resulting in the specific labeling of Suc C2, Asp C2, Lac C2, GABA C2, Glu C4, and Gln C4. After the second turn of neuronal TCA cycle, the C3 of Asp, the C2 and C3 of Glu, the C2 and C3 of Gln, and the C3 and C4 of GABA were labeled. By contrast with [2-13C]-acetate metabolism, [3-13C]-lactate infusion resulted in a significant increase of 13C flux through TCA cycle in db/db mice with cognitive decline as indicated by increased flux into the C2 and C3 of Suc (14.29 ± 3.70 versus 6.02 ± 2.28, P = 0.0001), relative to that in WT mice (Figure 4). Figure 4 also shows that db/db mice with cognitive decline had significantly higher 13C labeling in all carbon positions of Glu (C2, 10.37 ± 2.80 versus 4.76 ± 1.13, P = 0.009; C3, 7.77 ± 2.01 versus 3.68 ± 0.78, P = 0.009; C4, 27.39 ± 5.96 versus 11.91 ± 2.80, P = 0.002) and Gln (C2, 5.16 ± 0.86 versus 3.12 ± 0.61, P = 0.0001; C3, 3.29 ± 0.56 versus 2.20 ± 0.47, P = 0.002; C4, 6.99 ± 1.83 versus 3.95 ± 1.29, P = 0.002) than WT mice. Moreover, as compared with WT mice, increased 13C fluxes into all carbon positions of GABA were found in db/db mice with cognitive decline, but significant differences between them were obtained only in the C2 and C4 positions (C2, 14.51 ± 2.87 versus 6.72 ± 1.61, P1 < .0001; C4, 3.99 ± 0.57 versus 2.28 ± 0.69, P = 0.0004). For db/db mice with cognitive decline, the 13C-enrichment of Asp was significantly increased in the C2 and C3 positions, relative to WT mice (C2, 22.14 ± 2.82 versus 11.06 ± 2.81, P<.0001; C3, 17.72 ± 3.02 versus 9.25 ± 2.35, P<.0001). It is worth noting that the 13C flux from [3-13C]-lactate into Lac C2 was significantly reduced in db/db mice with cognitive decline relative to WT mice (3.67 ± 0.82 versus 5.67 ± 1.90, P = 0.01). In addition, we found that the 13C-enrichment in the C3 position of Ala was significantly enhanced in the brain of db/db mice with cognitive decline compared with WT mice (28.00 ± 3.89 versus 12.45 ± 3.77, P<.0001).
Metabolism of [3-13C]-lactate in the brain between wild-type (red bar) and db/db (blue bar) mice. Metabolites: Ala, alanine; Asp, aspartate; GABA, γ-aminobutyric acid; Gln, glutamine; Glu, glutamate; Lac, lactate; OAA, oxaloacetate; Pyr, pyruvate; Suc, succinate; α-KG, α-ketoglutarate. Significant level: *P<0.05; **P<0.01;***P<0.001.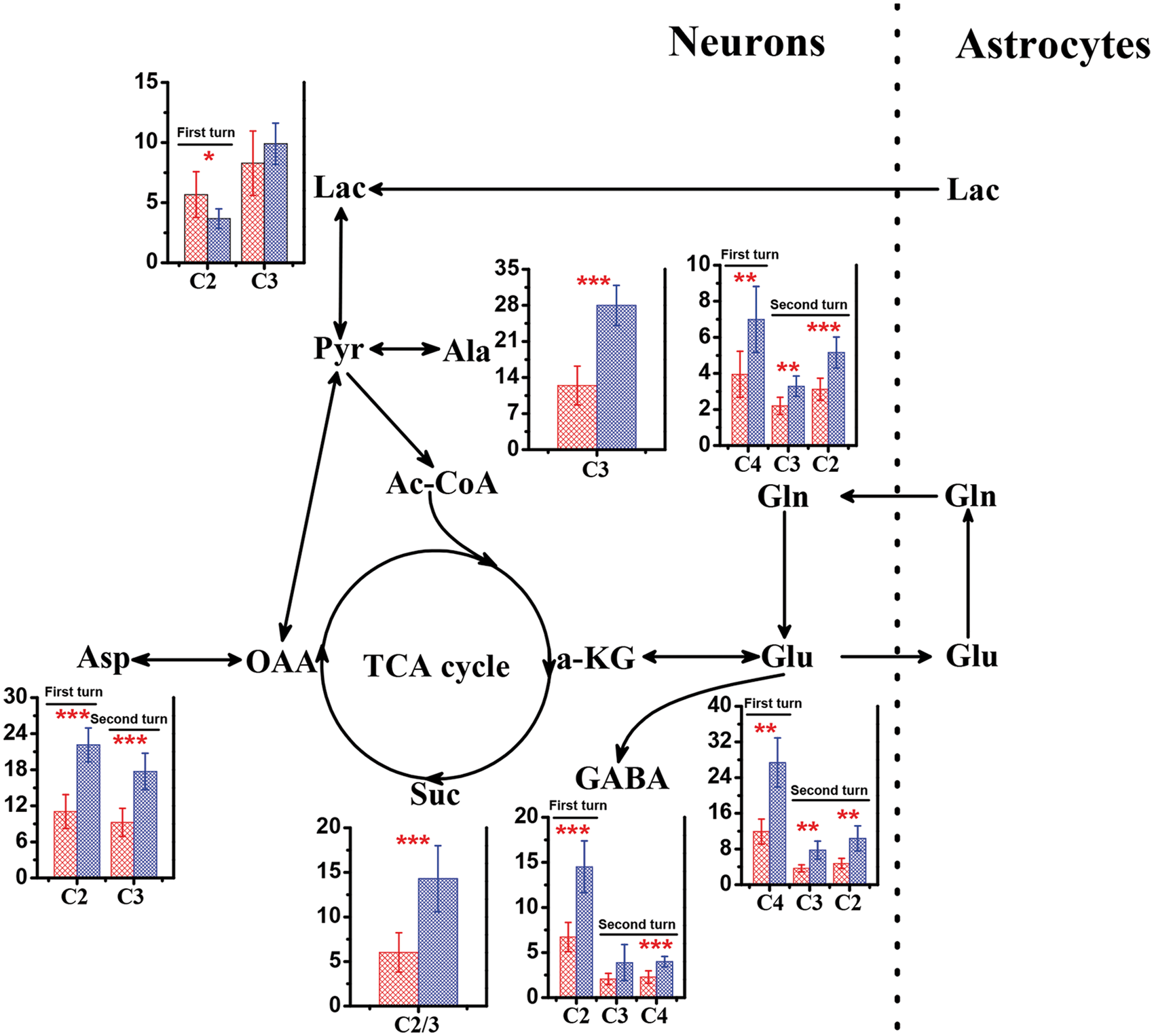
Gluconeogenesis in WT mice and db/db mice with cognitive decline
Figure 5 shows typical 13C NMR spectra of brain glucose in WT mice and db/db mice with cognitive decline infused with [2-13C]-acetate and [3-13C]-lactate. Interestingly, db/db mice with cognitive decline displayed a noticeable enhancement of the 13C flux from both [2-13C]-acetate and [3-13C]-lactate into glucose in the brain as compared with WT mice, which may be attributed to an increased gluconeogenesis.
Typical 13C NMR spectra of brain glucose in mice infused with [2-13C]-acetate (a, wild-type; b, db/db) and [3-13C]-lactate (c, wild-type; d, db/db). Assignments: α-glucose: C1, 93.05/72.50 ppm; C2, 75.10/74.35 ppm; C3, 75.72 ppm; C5, 74.35 ppm; C6, 61.45 ppm. β-glucose: C1, 96.85 ppm; C3, 76.70 ppm; C4, 72.50 ppm; C5, 76.83 ppm; C6, 61.70 ppm.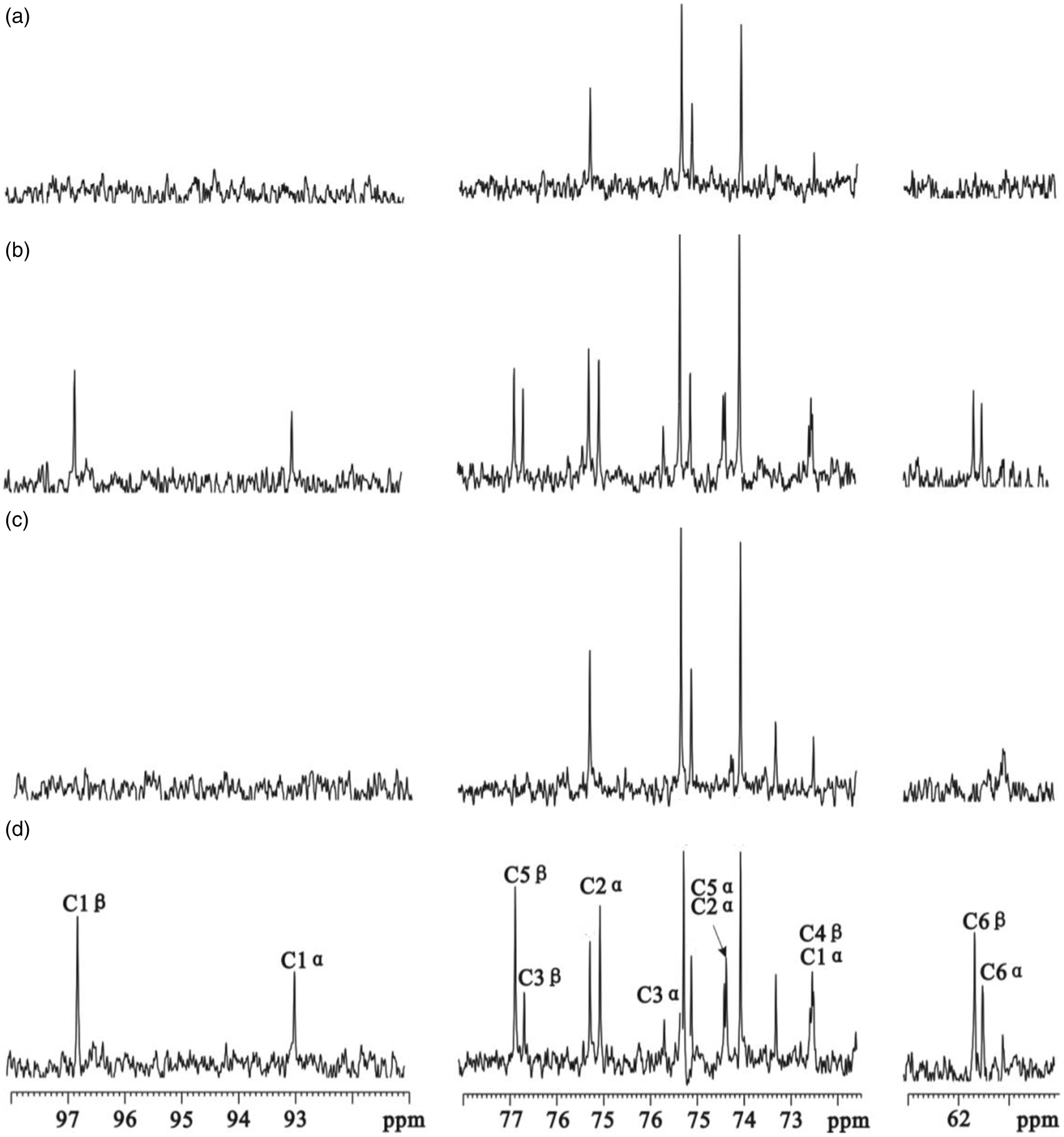
Discussion
T2D is a complex metabolic disorder that causes a series of complications in various organs and tissues in human body. Currently, db/db mouse, a leptin receptor-deficient diabetic model, has been widely used as a preclinical rodent model in T2D research. Leptin has been reported to involve in the differentiation of astrocytes and neurons in the prenatal and neonatal periods of mice 24 and rats.25,26 Thus, cognitive decline was found even in seven-week-old db/db mice. 27 We expectedly found that learning and memory in 17-week-old db/db mice were impaired as compared with age-matched WT mice. In clinical practice, however, cognitive impairment generally occurs in elderly patients with T2D. In addition, diet-induced diabetic model, as an alternative T2D model, can be reversible by reducing dietary fat. 28 Therefore, it is still a challenge to develop adequate models for exploring the relationship between T2D and cognition. 29 Although leptin receptor deficiency of db/db mice as an influence factor cannot be completely excluded in our study, Stranahan et al. 30 and Dinel et al. 31 reported that leptin may not play a critical role in cognitive changes of db/db mice. Moreover, mice fed a high-fat diet for 19 weeks were also accompanied by a decrease in leptin sensitivity. 32 Therefore, taken together, exploring metabolic variations in the brain of db/db mice with cognitive decline can still provide valuable information on metabolic mechanisms underlying diabetic encephalopathy, whereas leptin receptor deficiency of db/db mice should also be taken into account.
It is well known that neuron–astrocyte metabolic cooperation plays a critical role in brain function. 33 Therefore, 13C NMR spectroscopy in combination with intravenous [2-13C]-acetate and [3-13C]-lactate infusions was used to explore potential metabolic relationships between astrocytes and neurons in db/db mice with cognitive decline on the basis of the fact that acetate is primarily utilized by astrocytes and lactate by neurons. Our results reveal that the development of cognitive decline in db/db mice may be associated with the changes of energy metabolism, neurotransmitter metabolism, lactate–alanine shuttle, pyruvate recycling, and gluconeogenesis.
Energy metabolism in the brain of db/db mice with cognitive decline
The brain has high energy demand to maintain its normal function. The cooperation between astrocytes and neurons has been reported to play a central role in brain energy metabolism instead of a “neurocentric” view. 34 In brain, astrocytes outnumber neurons and their functions have been closely linked to normal neuronal activity. Thus, disturbances of neuron–astrocyte metabolic interactions are responsible for neurodegenerative disorders. 34 In the present study, as compared with WT mice, we found that the 13C-enrichment of the C2 and C3 of Suc as a TCA cycle intermediate was significantly decreased in the brain of db/db mice with cognitive decline after [2-13C]-acetate infusion, while an opposite result was obtained after [3-13C]-lactate infusion. This may suggest that TCA cycle was down-regulated in astrocytes and up-regulated in neurons in db/db mice with cognitive decline relative to WT mice. In our previous study, the 13C-enrichment of Suc was significantly reduced in 15-week-old rats with T1D after [2-13C]-acetate infusion, suggesting that astrocytic mitochondrial function was impaired. 21 It has been well known that neurons are dependent on a constant supply of TCA cycle intermediates from astrocytes due to a lack of pyruvate carboxylase. 35 Therefore, the reduction of astrocytic TCA cycle cannot meet high demand of TCA cycle intermediates in neurons, which may be responsible for the development of cognitive decline in db/db mice. In our study, variations of energy metabolism in astrocytes and neurons of the whole brain in db/db mice with cognitive decline were examined. However, the brain site-specific alteration in energy metabolism was unavailable. Sickmann et al. 36 investigated variations in energy metabolism in the cerebral cortex, hippocampus, and cerebellum of diabetic and obese rats using [1-13C]-glucose administration. They found that TCA cycle was down-regulated more than glycolysis in all brain regions of T2D rats. In obese rats, however, the activity of TCA cycle was lower in the hippocampus than in the cortex. Thus, it might be interesting to explore the specific difference in energy metabolism between astrocytes and neurons in different brain regions of db/db mice with cognitive decline.
Neurotransmitter metabolism in the brain of db/db mice with cognitive decline
Owing to glutamine synthetase exists in astrocytes but not in neurons, 37 astrocytes have been considered as the major source of uptaking and metabolizing Glu to form Gln. Then, Gln can be transferred from astrocytes to neurons and reconverted to Glu. Moreover, Gln can also be converted to GABA via Glu. This is called the Gln-Glu/GABA cycle. Glu and GABA are the major excitatory and inhibitory neurotransmitters in the CNS, respectively. The Gln-Glu/GABA cycle between astrocytes and neurons can regulate neurotransmitter homeostasis and maintain normal CNS function. 38 In our previous study, we have found a disturbance of the Gln-Glu/GABA cycle in the brain of T1D rats using 13C NMR method. 21 For instance, [2-13C]-acetate infusion in 15-week-old rats with T1D resulted in increased labeling of Gln C4 but unchanged Glu C4, which may be attributed to a decreased Gln transfer from astrocytes to neurons. 21 However, in the present study, the 13C-enrichments of the C2, C3, and C4 of Gln and the C2 and C3 of Glu were decreased in db/db mice with cognitive decline after [2-13C]-acetate infusion, which may be due to an inhibition of astrocytic TCA cycle. On the contrary, increased enrichments of the C2 and C3 of Suc, the C2, C3, and C4 of Gln, and the C2, C3, and C4 of Glu after [3-13C]-lactate infusion may indicate activated neuronal activity in db/db mice with cognitive decline, relative to WT mice. In addition, for db/db mice with cognitive decline, the enrichment of the C2 of GABA was significantly reduced after [2-13C]-acetate infusion, while significantly increased enrichments of the C2, C3, and C4 of GABA after [3-13C]-lactate infusion were observed as compared with WT mice. Thus, overall, our data reveal that the Gln-Glu/GABA cycle between astrocytes and neurons was disordered in the whole brain of db/db mice with cognitive decline. However, Sickmann et al. 36 found a brain regional variation in the Gln-Glu/GABA cycle, which was less affected by T2D in the cortex and cerebellum than in the hippocampus.
Aspartate is another excitatory neurotransmitter that was derived directly from a TCA cycle intermediate, oxaloacetate, by transamination. In this study, we found that the 13C enrichment of the C2 and C3 of Asp was significantly increased in db/db mice with cognitive decline after [3-13C]-lactate infusion. Thus, the alteration of brain Asp level may be attributed to cognitive decline-induced changes of neuronal energy metabolism.
Lactate–alanine shuttle in the brain of db/db mice with cognitive decline
Complementary to the Glu-Gln/GABA cycle, a lactate–alanine shuttle would account for the nitrogen exchange from neurons to astrocytes in mammalian brain.39,40 We observed that Ala in the brain was only labeled after [3-13C]-lactate infusion but not after [2-13C]-acetate infusion. This is in agreement with the in vitro finding reported by Waagepetersen et al. 41 that alanine was highly enriched with 13C in neurons but not in astrocytes. In addition, db/db mice with cognitive decline had a higher labeled brain Ala level than WT mice, indicating that an increased transfer from Lac to Ala in neurons may be linked to the development of cognitive decline in db/db mice.
Pyruvate recycling in the brain of db/db mice with cognitive decline
Pyruvate recycling pathway in the brain was firstly found by Cerdan et al. 42 using 13C NMR method. When energy substrates are insufficient in the brain, this pathway can maintain pyruvate functional activity for energy metabolism. 42 In the present study, we found that pyruvate recycling occurred in the brain of db/db mice with cognitive decline as indicated by the formation of labeled Lac C2 and C3 from [2-13C]-acetate infusion and labeled Lac C2 from [3-13C]-lactate infusion. However, the 13C labeling percentage in Lac by [3-13C]-lactate infusion was higher than that by [2-13C]-acetate infusion. In T1D rat brain, we have observed that the C2 and C3 of Lac were labeled by [2-13C]-acetate due to pyruvate recycling and the C3 of Lac was heavily labeled by [1-13C]-glucose because of glycolysis. 21 Moreover, it is worth noting that db/db mice with cognitive decline had significantly lower 13C-enrichments in the Lac C2 and C3 from [2-13C]-acetate infusion and Lac C2 from [3-13C]-lactate infusion than WT mice, suggesting that an inhibition of pyruvate recycling pathway may be associated with the development of cognitive decline. In our previous study, a decrease in pyruvate recycling was also observed in 15-week T1D rats. 21 In addition, the change of Lac level in the brain of diabetic mice also showed a regional variation, where Lac was decreased in the hippocampus but not in the cortex. 43 Therefore, we speculate that pyruvate recycling pathway may vary between different brain regions in db/db mice with cognitive decline.
Gluconeogenesis in db/db mice with cognitive decline
The increase in hepatic gluconeogenesis has been associated with increased whole body glucose level in T2D patients. 44 In this study, we found that labeled glucose was enhanced in the brain of db/db mice with cognitive decline relative to WT mice after both [2-13C]-acetate and [3-13C]-lactate infusions. Hyperglycemia as a common diabetic symptom plays an important role in the development of diabetic neuropathy. 45 Therefore, our results suggest that gluconeogenesis increases glucose level in the brain and thereby may cause cognitive impairment. However, we cannot elucidate specific variations in glucose level between different brain regions. Interestingly, Sickmann et al. 36 found that a decrease in glycogen level was caused by T2D in the cerebellum but not in the cortex and hippocampus.
Limitations
Several limitations to consider in the present study are that (1) whole brain tissues were analyzed due to sample amount limitation, but it could be more robust to analyze metabolic changes in the hippocampus relative to hippocampal-based memory tasks, and of great interest to explore specific metabolic differences between different brain regions; (2) the effect of leptin on cognitive decline cannot be completely excluded due to leptin receptor deficiency in db/db mice; (3) limited metabolites were measured by a single analytical technique (e.g. NMR), so more sensitive methods such as LC–MS and GC–MS are recommended to achieve a more detailed analysis of metabolic pathways; and (4) the results were only obtained from the metabolic level, yet coupling with genomic or proteomic data will advance comprehensively understanding of the pathogenesis of cognitive decline.
Conclusions
In the present study, we found that astrocytic TCA cycle was inhibited in the brain of db/db mice with cognitive decline relative to WT mice. Thus, the demand of TCA cycle intermediates in neurons and glutamine synthesis and glutamate uptake in astrocytes were impaired due to the reduction of astrocytic TCA cycle. As compared with WT mice, an increased transfer from Lac to Ala in neurons was observed in db/db mice with cognitive decline. A reduced 13C labeling percentage in Lac in db/db mice with cognitive decline may indicate an inhibition of pyruvate recycling pathway. In addition, gluconeogenesis was increased in db/db mice with cognitive decline. Thus, the development of cognitive decline in T2D may be attributed to the disturbance of neuron–astrocyte metabolic cooperation and an increase in gluconeogenesis.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (Nos.: 21175099, 21575105, 81501303) and Zhejiang Provincial Natural Science Foundation (Nos.: LY13H070005, LY14H090014, LY15H180010).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors' contributions
HG and LZ contributed to experimental design. YZ, DW, AC, QL, MC, MD and XY contributed to animal experiment data acquisition. HZ and HG contributed to data analysis, result interpretation, and writing. All authors have read, revised, and approved the final manuscript.
