Abstract
This is the first study to assess the influence of sex on the evolution of ischaemic injury and penumbra. Permanent middle cerebral artery occlusion was induced in male (n = 9) and female (n = 10) Sprague-Dawley rats. Diffusion-weighted imaging was acquired over 4 h and infarct determined from T2 images at 24 h post-permanent middle cerebral artery occlusion. Penumbra was determined retrospectively from serial apparent diffusion coefficient lesions and T2-defined infarct. Apparent diffusion coefficient lesion volume was significantly smaller in females from 0.5 to 4 h post permanent middle cerebral artery occlusion as was infarct volume. Penumbral volume, and its loss over time, was not significantly different despite the sex difference in acute and final lesion volumes.
Introduction
The ischaemic penumbra is a well-established concept in stroke, defined as a region of hypoperfused, metabolically active tissue surrounding the ischaemic core. 1 If blood flow is not restored, this tissue becomes incorporated into the ischaemic core and progresses to infarction.2,3 Therefore, it is crucial that the spatiotemporal evolution of ischaemic damage and penumbra is characterised in animal models of focal cerebral ischaemia to determine if there are differences in the optimal time for therapeutic intervention with regard to sex and in the presence of known stroke risk factors.
Magnetic resonance (MRI) diffusion-weighted imaging (DWI) identifies tissue where diffusion is restricted as a result of cytotoxic oedema leading to a reduction in the apparent diffusion coefficient (ADC). Assessment of penumbral tissue can be determined retrospectively from the growth of the DWI or ADC lesion over time and the final infarct, a method used previously for both pre-clinical and clinical data sets. 4
In pre-clinical research, across the biological disciplines, there is still a considerable sex bias with studies on male animals outnumbering those in females and in particular this bias is most prominent in neuroscience research. 5 Reasons often cited for the prevalence of males over females are due to the concerns over contributions of the oestrus cycle; however, a recent meta-analysis in female mice demonstrated that they were no more variable when tested throughout their oestrus cycle than males. 6 Sex is a key factor influencing various aspects of human stroke including stroke outcome and response to treatment. 7 Likewise, in animal models of stroke, differences in molecular mechanisms of ischaemic cell death, stroke outcome and response to therapy have been observed between males and females. 8 To date, the acute spatiotemporal progression of ischaemic damage and fate of penumbra has not been characterised in female rats.
The study aims were: (1) to establish MRI diffusion thresholds of abnormality in male and female rats following permanent middle cerebral artery occlusion (MCAO); (2) apply these thresholds to assess differences in the spatiotemporal evolution of ischaemic damage; (3) using retrospective analysis of ADC lesion and infarct volume to calculate penumbra, and compare penumbra volume and lifespan in males and females over the first 4 h post-stroke.
Materials and methods
All experiments were performed in age-matched (12–16 weeks) Sprague-Dawley rats (males: 300–350 g, females: 250–280 g, Harlan, UK) under licence from the UK Home Office, were subject to the Animals (Scientific Procedures) Act, 1986. The report was carried out in accordance with the ARRIVE guidelines (http://www.nc3rs.org.uk/arrive). Rats were randomly assigned to surgery using a random list generator (www.random.org).
Model of middle cerebral artery occlusion
Rats were intubated, ventilated and permanent MCAO was carried out under isoflurane anaesthesia (5% induction, 2–2.5% maintenance in 70% N2O 30% O2). Body temperature was maintained at 37℃ and both femoral arteries were cannulated for mean arterial blood pressure (MABP) measurement and blood gas analysis. MABP and heart rate were continuously recorded under general anaesthesia, including scanning (AcqKnowledge, Biopac Systems, CA, USA). Permanent MCAO was induced with an intraluminal filament (diameter: 0.31–0.35 mm, tip length: 5–6 mm, Doccol, CA, USA) as previously described. 9
MRI scanning
MRI was performed on a Bruker Biospec 7-T/30-cm system with a gradient insert (121 mm ID, 400 mT/m) and a 72-mm birdcage resonator. A four-channel phased-array rat head surface coil was used for brain imaging. Arterial blood gas analysis was determined hourly (0–4 h) during scanning.
DWI was performed at 0.5 h and hourly for 4 h post MCAO to generate quantitative ADC maps and allow assessment of ischaemic injury.
At 24 h post-MCAO, animals were re-anaesthetised and scanned with a RARE T2-weighted sequence for assessment of infarct volume. For full details of imaging protocol see supplementary file.
Image analysis
Quantitative ADC maps, ( × 10−3 mm2/s) generated from raw DWI images using Paravision 5 software (Bruker, Germany) were subsequently processed using Image J software (http://rsb.info.nih.gov/ij/).
Infarct volume was calculated by manually delineating the hyperintense region on T2 slices, which corresponded anatomically to the ADC slices acquired acutely. Area of infarct was summed over all slices and multiplied by slice thickness to calculate total infarct volume which was corrected for brain swelling. 10 ADC thresholds were calculated as previously described. 11
Penumbral tissue was assessed retrospectively from ADC-derived lesion growth from 30 min and the oedema-corrected infarct volume at 24 h. ADC lesion growth at each time point was also expressed as a percentage of final infarct volume to determine the potential impact of intervention at each time point. All analyses were undertaken blind to the sex of rats.
Statistical analysis
Data are presented as mean ± SD. Infarct volume and ADC threshold values were compared between males and females using a Student’s unpaired t-test. Changes in ADC lesion volume and penumbra over time and between sexes were assessed by a two-way ANOVA (between-subjects factor: sex; within-subjects factor: time) with Bonferroni’s post-test. P < 0.05 was considered statistically significant and statistical tests were performed using GraphPad Prism v6.
Results
Mortality and physiological parameters
Two of 12 female rats were excluded from analysis due to incomplete MCA occlusion. Three of 12 male rats died overnight before T2-weighted images could be acquired. Physiological variables remained within normal levels during stroke surgery and throughout MRI scanning and were comparable between the sexes.
Temporal evolution of ADC lesion and final infarct
Oedema-corrected infarct volume at 24 h post-MCAO was significantly larger in males (males 217 ± 49 mm3, females 151 ± 56 mm3; P < 0.05). Brain swelling was not significantly different between groups (males: 18.2 ± 3.0%, females: 17.3 ± 1.9% increase of ipsilateral hemisphere, P = 0.3).
Brain volume at 4 h post-MCAO calculated across eight coronal slices, was not statistically different between sexes (males: 901 ± 38 mm3, females: 892 ± 35 mm3, P = 0.6). Similarly, there were no differences in brain volume at 24 h post MCAO between sexes (males: 976 ± 45 mm3, females: 951 ± 33 mm3). There was no evidence of acute brain swelling as hemisphere volume did not change significantly over the 0–4 h time course. Mean contralateral ADC values were similar between the sexes (males: 0.78 ± 0.02, females: 0.77 ± 0.02 × 10−3 mm2/s, P = 0.7) and did not change significantly throughout the 4 h scanning protocol. The absolute ADC threshold of abnormality for male rats was 0.58 ± 0.05 × 10−3 mm2/s, a 25 ± 2% reduction from mean contralateral ADC values at 4 h post MCAO. In female rats, the equivalent ADC threshold was 0.53 ± 0.03 × 10−3 mm2/s, a 29 ± 2% reduction from mean contralateral ADC values. Both the absolute and relative ADC thresholds were similar between the sexes (P > 0.05).
From the two-way ANOVA, the main effect of sex was significant (P = 0.0037, F (1, 19) = 10.97) as was the main effect of time (P < 0.0001, F (4, 76) = 112.1). The interaction between the two factors was not significant (P = 0.3757). Using the ADC thresholds for each sex, the ADC-derived lesions increased significantly over time in males (from 98 ± 64 mm3 at 30 min to 223 ± 50 mm3 at 4 h; P < 0.001) and females (45 ± 28 mm3 at 30 min to 147 ± 50 mm3; P < 0.001). ADC lesions were significantly smaller in female rats from as early as 60 min post-MCAO, and this effect was maintained throughout the 4 h time course (Figure 1(a)).
Acute evolution of (a) ADC-defined ischaemic injury and T2–derived final infarct volume and (b) penumbra volume as defined by ADC lesion expansion into final infarct in male and female rats. (c) The amount of penumbral tissue expressed at each time point as a percentage of the respective final infarct in male and female SD rats. (*P < 0.05, Two-way ANOVA with Bonferroni post test, n = 10 for both groups). Data are displayed as mean ± SD.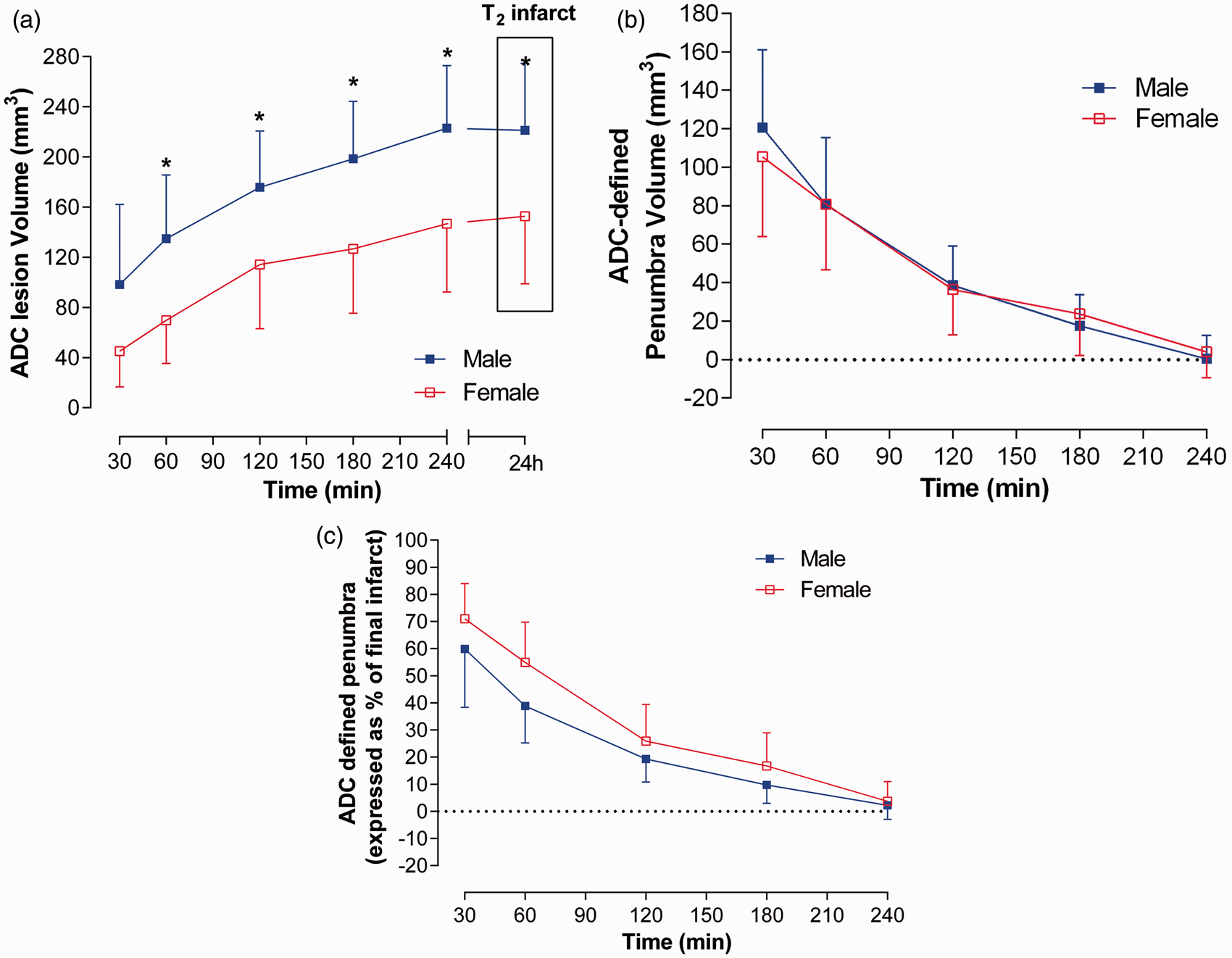
Temporal change of the ischaemic penumbra
Penumbral tissue was assessed retrospectively from the growth of the ADC lesion into the final infarct at 24 h post-stroke. Figure 2 illustrates the growth of the ADC lesion at each time point in the median animal. The absolute volume of penumbra determined by comparing the ADC lesion at each time point with 24 h infarct decreased significantly over time in both male (from 121 ± 41 mm3 at 0.5 h to 0 ± 12 mm3 at 4 h, P < 0.0001) and female rats (from 105 ± 44 mm3 at 0.5 h to 4 ± 14 mm3 at 4 h, P < 0.0001) with no significant differences between sexes (F (1, 17) = 0.1312, P = 0.7216, Figure 1(b)). Figure 1(c) shows the amount of penumbral tissue at each time point expressed as a percentage of the final infarct in males and females reflecting the potential impact of intervention at these time points. For instance, at 30 min following MCAO, 59 ± 23% of the final infarct in males was potentially salvageable while in females this equated to 70 ± 13% (Figure 1(c)).
Images from the median rat from each group (a: male; b: female) showing the spatial location of ADC lesion growth (in white) from 0.5 to 1, 1 to 2, 2 to 3 and 3 to 4 h following MCAO. The T2-weighted infarct (hyperintense region outlined in red) at 24 h post MCAO is shown on the bottom row.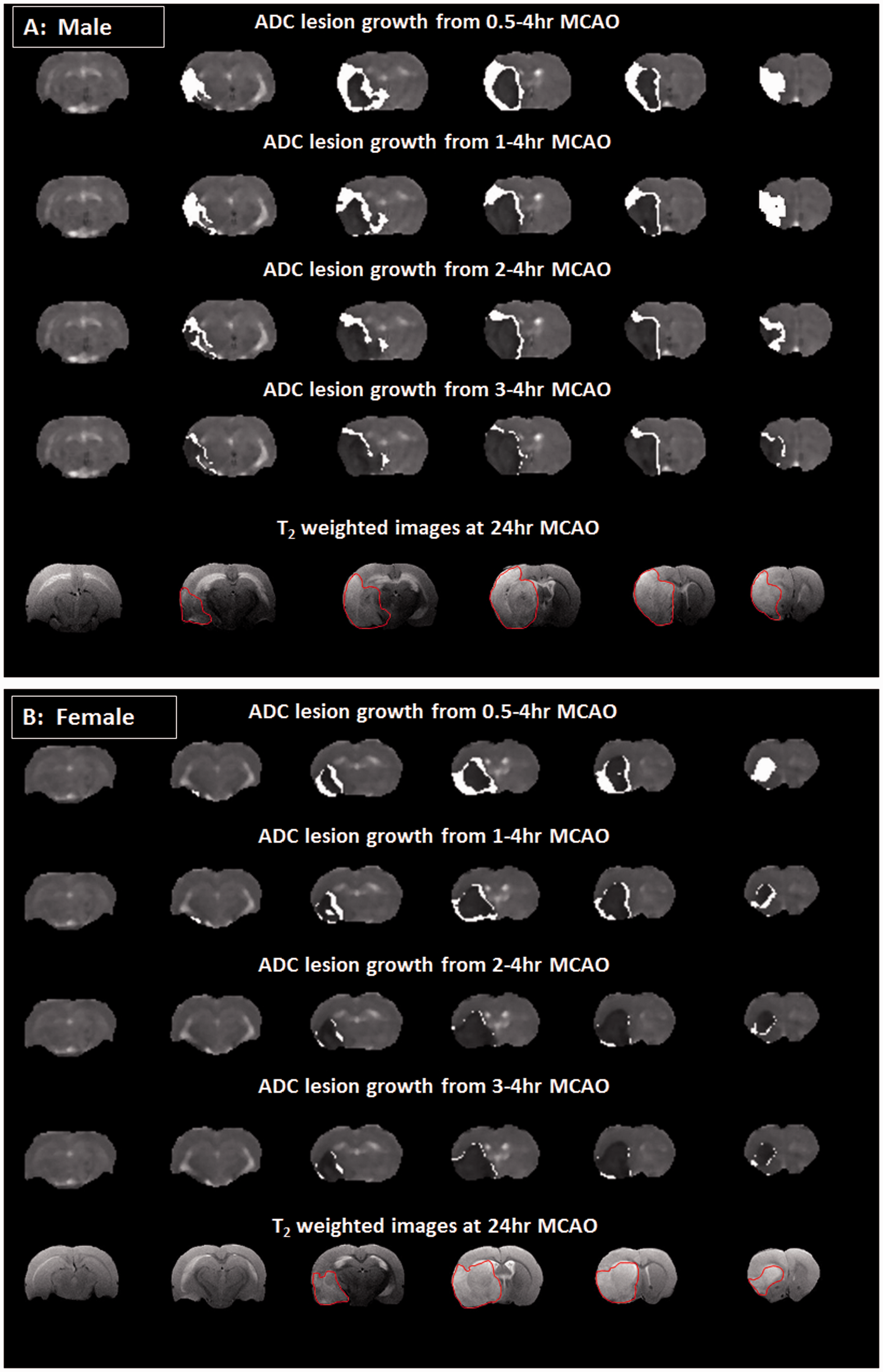
Discussion
The STAIR recommendations highlight the importance of investigating sex differences in experimental stroke studies. 12 It is well established that sex differences exist in relation to infarct size following experimental stroke with females exhibiting smaller infarcts and improved behavioural outcome compared to males. 8 However, to our knowledge, sex differences in the amount of salvageable penumbra and its lifespan have not been studied. This is the first study to (1) define sex-specific diffusion viability thresholds and (2) investigate if sex influences the acute evolution of ischaemic damage, penumbra volume and its loss during the critical first hours following stroke. The major findings are (1) calculated diffusion thresholds of tissue abnormality were comparable between the sexes; (2) the acute ADC lesion volume and final infarct were smaller in female rats; (3) penumbra volume, and its loss over time, was not significantly different between sexes; (4) Our ADC thresholds of 0.58 and 0.53 × 10−3 mm2/s (25% and 29% reduction) for male and female rats, respectively, were similar to previously published thresholds for male Sprague Dawley rats 13–15 and co-morbid strains such as the spontaneously hypertensive stroke-prone (SHRSP) rat (21% reduction) and its normotensive control the WKY (23% reduction). 11 This suggests that rat strain and sex do not have a significant influence on the ADC threshold following MCAO.
Using these ADC thresholds, although the absolute ADC lesion volume was smaller in females, the growth in ADC lesion volume (and consequent loss of penumbra over time) was similar in males and females. Previous studies have established sex-specific differences in infarct volume following MCAO with female sex hormones, oestrogen and progesterone having neuroprotective influences.8,16 Our data show that ischaemic damage was smaller in female rats from as early as 30 min after MCAO and this was maintained throughout the acute scanning protocol and similarly reflected in the final infarct at 24 h.
Retrospective assessment of penumbra volume revealed no significant sex difference in the absolute volume of penumbra nor in loss of penumbra to the ischaemic core suggesting that the therapeutic time window for intervention is similar. However, absolute volumes do not inform on potential impact of an intervention on final outcome, given that final infarct volumes are different. Therefore, we calculated penumbra volume as a percentage of the respective final infarct. Within the first hour of stroke onset, an intervention (i.e. thrombolysis or thrombectomy to induce reperfusion) would have potentially have greater benefit in females in terms of the reduction in final infarct. As a comparison, our previous data from normotensive WKY and stroke-prone spontaneously hypertensive rats (SHRSP) demonstrated that early intervention in SHRSP has much less of an impact on the final infarct volume compared to other strains. One limitation of the present study was that we did not know which stage of the oestrus cycle female rats were within at the time of stroke surgery and this may account for increased variability in this group; however, a recent meta-analysis challenged this assumption. 6
In conclusion, although the amount of brain damage is significantly smaller in females during the acute phase following stroke, there was no difference in the ADC viability threshold, or the amount or lifespan of potentially salvageable penumbral tissue.
Footnotes
Funding
This work was supported by an award (Ref: NS-GU-122) from the Translational Medicine Research Collaboration – a consortium of the Universities of Aberdeen, Dundee, Edinburgh and Glasgow, Grampian, Tayside, Lothian and Greater Glasgow & Clyde NHS Health Boards, Scottish Enterprise and Wyeth Pharmaceuticals.
Acknowledgements
The authors thank Mr. Jim Mullin and Mrs. Lindsay Gallagher for technical assistance involving MRI scanning.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors' contributions
Tracey Baskerville contributed to the execution of experiments, MRI scanning, data analysis and preparation of manuscript. I Mhairi Macrae contributed to the writing and editing of manuscript, experimental design and interpretation of data. William M Holmes developed the MRI sequences and contributed to editing of manuscript. Christopher McCabe contributed to the execution of experiments, MRI scanning, data analysis, preparation of manuscript and experimental design.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
