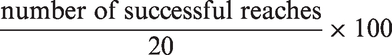Abstract
The prevalence of subcortical white matter strokes in elderly patients is on the rise, but these patients show mixed responses to conventional rehabilitative interventions. To examine whether cortical electrical stimulation can promote motor recovery after white matter stroke, we delivered stimulation to a small or wide region of sensory-parietal cortex for two weeks in a rodent model of circumscribed subcortical capsular infarct. The sham-operated group (SOG) showed persistent and severe motor impairments together with decreased activation in bilateral sensorimotor cortices and striatum. In contrast, sensory-parietal cortex stimulation significantly improved motor recovery: final recovery levels were 72.9% of prelesion levels in the wide stimulation group (WSG) and 37% of prelesion levels in the small stimulation group (SSG). The microPET imaging showed reversal of cortical diaschisis in both groups: in both hemispheres for the WSG, and in the hemisphere ipsilateral to stimulation in the SSG. In addition, we observed activation of the corpus callosum and subcortical corticostriatal structures after stimulation. The results from the c-Fos mapping study were grossly consistent with the microPET imaging. Sensory-parietal cortex stimulation may therefore be a useful strategy for overcoming the limits of rehabilitative training in patients with severe forms of subcortical capsular infarct.
Introduction
White matter is commonly affected by ischemic injury in the central nervous system. Damage to the white matter often results in stroke-related functional disabilities, ranging from transient ischemic attacks to vascular dementia. The prevalence of white matter strokes in elderly people has increased recently, and this subtype now represents 30% of all strokes.1–3 Among the various white matter anatomical structures, the internal capsule is a key structure because it contains pyramidal tract fibers. When the internal capsule is disrupted by an infarct lesion, it causes severe motor impairments and patients have a poor recovery rate compared with patients with cortical infarcts.4,5 Therefore, the promotion of quality motor recovery is the most relevant issue in the management of patients with subcortical capsular infarct (SCI).
Invasive and noninvasive cortical stimulation have both been reported to be effective in improving the functional deficits resulting from cortical gray matter stroke in animal models and in human survivors.6–8 The mechanisms underlying this functional recovery include increased synaptic plasticity and functional reorganization of the neural substrates, which depend on multiple factors such as type of stimulation, parameters of stimulation, timing and pathological conditions;9,10 depending on the polarity, transcranial direct current stimulation (tDCS) enhances or suppresses the excitability of cortical tissue; 11 low-frequency transcranial magnetic stimulation (TMS) or cathodal tDCS inhibits the contralesional activity, whereas high-frequency TMS or anodal tDCS enhances the ipsilesional cortical activity;6,12 there is a difference in recovery between groups with cortical stimulation versus no stimulation early in training.13,14 Anatomical, electrophysiological, and imaging studies have demonstrated the various changes underpinning neuroplasticity in regions of cortical stimulation: higher dendritic density, greater synaptogenesis and enhanced synaptic efficacy, long-term-potentiation-like changes, and white matter change.13,15–17 However, there is lack of evidence that a similar strategy could be effective in treating deficits from SCI.
We have previously demonstrated that a circumscribed SCI in the posterior limb of the internal capsule (PLIC) in rats produces depression of the resting-state brain activity in various cortical and subcortical areas in the absence of direct damage to those areas, consistent with diaschisis. 18 We also found that reaching training helped restore motor function in animals with incomplete destruction of the internal capsule; however, reaching training never contributed to motor recovery in animals with complete destruction of the internal capsule, suggesting that an alternative strategy is required to augment motor recovery in severe cases.18,19 We hypothesized that cortical stimulation may overcome the limitations of conventional rehabilitation. However, current information on the effects of cortical stimulation in subcortical stroke models is poor: there have been several human studies but these produced conflicting results, probably because of variations in the type of stimulation method, timing of the intervention, pattern of cortical activation, and integrity of the corticospinal tract.20–23 In addition, the optimal spatial extent of cortical stimulation is an open question. Here, we report the efficacy of sensory-parietal cortex (SPC) stimulation in a chronic SCI model with persistent motor impairments, and whether the stimulation-induced motor recovery depends on the size of the electrical field. In addition, we measured brain activity using longitudinal microPET and c-Fos mapping to elucidate the potential mechanism and network through which cortical stimulation aids motor recovery.
Materials and methods
Experimental animals
Twenty-two male Sprague Dawley rats (300–350 g, nine weeks old) were used for this experiment. Animals were housed three per plastic cage with water ad libitum in a controlled animal husbandry unit at 21 ± 1℃, under a 12-h light–dark cycle with lights on at 7:00 a.m. Animal experiments were conducted according to the ARRIVE guidelines and the institutional guidelines of the Gwangju Institute of Science and Technology (GIST), and all procedures were approved by the Institutional Animal Care and Use Committee at GIST. Animals were included in the present study if they showed persistent impairment in a reaching task more than two weeks after circumscribed photothrombotic SCI lesioning in the dominant hemisphere, despite daily training on the task (n = 22). Forty-one animals underwent infarct lesioning and 32 (78%) showed persistent impairment. Additional 10 animals were excluded because they could not be longitudinally followed-up with microPET due to technical failures. Animals were divided into three groups: the wide stimulation group (WSG: n = 8), which received cortical stimulation via a large electrode, the small stimulation group (SSG: n = 8), which received stimulation via a small electrode, and the sham-operated group (SOG: n = 6). An a priori power analysis with a predicted effect size of 2.0 (α = 0.05, β = 0.05, power = 0.95, allocation ratio = 1.2) was used to determine that a sample size of 14 rats (eight in the test group and six in the SOG) would be sufficient to detect a statistically significant difference between groups.
Behavioral training
All rats were trained on the Single Pellet Reaching Task (SPRT)
24
daily throughout the experiment, except during a one-week recovery period after electrode implantation (Figure 1). The animals were food-restricted to 90% of their initial body weight to motivate them to perform the task. The SPRT was conducted in a box constructed from clear Plexiglas (40 cm × 45 cm × 13.1 cm wide) with a 1 cm wide slit in the middle of the front wall. A small shelf was attached behind the slit. A sucrose pellet (Bio-Serve, Frenchtown, NJ, USA) was placed obliquely on the shelf contralateral to the preferred forelimb. The positioning of the pellet prevented the rats from using the nondominant paw. The rats were given 20 pellets in each daily session. Reaching performance was calculated as the percentage of successful reaches according to the following formula:
Experimental design. The single pellet reaching task (SPRT) was carried daily throughout the experiment except one week of post-operative recovery period. One week after post-operative recovery, the SSG and the WSG received two weeks of electrical cortical stimulation. MicroPET images were acquired for control scan after electrode placement (PS0), seven days post-stimulation (PS7) and 14 days post-stimulation (PS14) for the SSG and the WSG. For SOG, an additional scan was performed in the prior to the lesion (pre-lesion scan, PL), as well as the three scans on PS0, PS7, and PS14.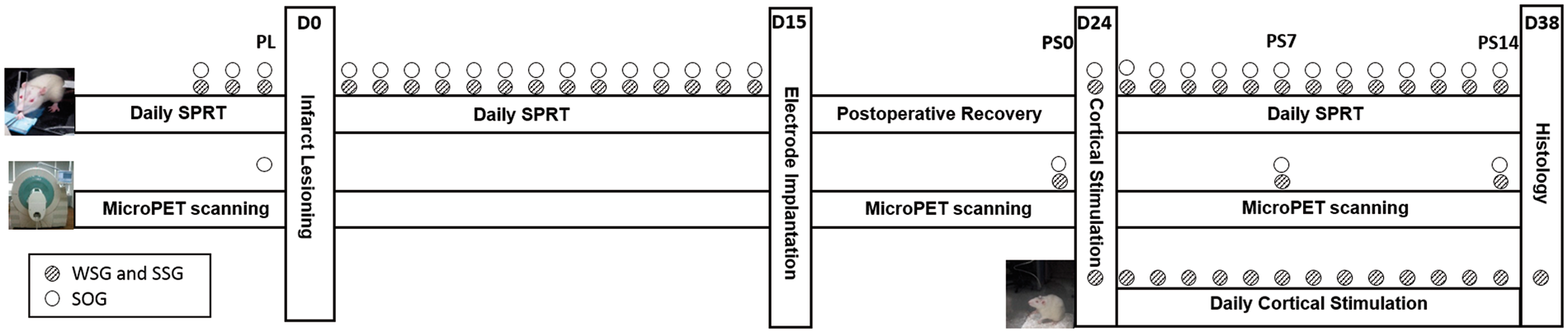
Prelesional baseline performance was defined as the average of SPRT scores from three training sessions prior to the induction of photothrombotic SCI. Post-lesional reaching performance was assessed by daily testing on the task for two weeks, which confirmed a persistent reaching impairment. As shown in our previous studies, persistent impairment likely indicates complete capsular infarction. 19 After electrode placement and a one-week post-operative recovery period, SPRT was carried out daily for another two weeks, during which time cortical electrical stimulation was applied.
Induction of photothrombotic capsular infarction
Photothrombotic SCI was induced as described in our previous studies.18,19 Briefly, rats were anesthetized with a mixture of ketamine hydrochloride (100 mg/kg) and xylazine (7 mg/kg), then fixed in a small animal stereotactic frame. Body temperature was maintained at 37.5 ± 0.5℃ with a thermocouple blanket. Twenty-two rats, 13 with a left-paw preference and 9 with a right-paw preference, underwent a photothrombotic capsular infarction contralateral to the preferred forelimb. Following a midline scalp incision and alignment of bregma and lambda in a flat plane, a small craniotomy was made 2 mm posterior to bregma and 3.1 mm lateral to the midline. An optical neural interface (ONI) composed of an optical fiber (core: 62.5 µm; outer diameter: 125 µm) contained in a steel cannula (27 gauge) was stereotactically inserted into the PLIC (7.8 mm ventral to skull). The other end of the ONI was connected to a green laser source (532 nm, Changchun New Industries Optoelectronics Tech. Co., Ltd, Jilin, China). Rose Bengal dye (20 mg/kg) was injected through the tail vein, followed by 1.5 min of laser irradiation with a laser light intensity of 3.7±0.2mW at the PLIC target.
Cortical stimulation in SPC
All rats (n = 22) were subject to electrode placement in the affected hemisphere. Two types of electrode were used to vary the extent (effective volume) of neural excitation 25 : a custom-made, 4 mm× 2 mm stainless-steel electrode for the WSG and a 0.7-mm-diameter screw electrode for the SSG. After making a small craniectomy, stimulating electrode was implanted epidurally over SPC and a reference electrode was positioned in the motor cortex. In the WSG, the electrode extended from 0.5 mm to 4.5 mm posterior and 2 mm to 4 mm lateral to bregma (8 mm2), covering parietal cortex and hindlimb, trunk, and forelimb areas of sensory cortex. In the SSG, the electrode was centered at 4.0 mm posterior and 3 mm lateral to bregma (0.38 mm2), covering only parietal cortex. Electrodes were connected to a pedestal on the skull, and to the stimulating equipment via a swivel adaptor at the top of the reaching task box. Rats in the SOG were implanted with a screw electrode, as described above for the SSG but did not receive electrical stimulation.
Voltage stimulation was delivered continuously to targeted areas in the WSG and the SSG via a programmable stimulator (Cybermedic, Iksan, Korea) for 24 h/day for two weeks, beginning one week after electrode placement (Figure 1). Continuous-mode stimulation (24 h/day) was selected for this study because preliminary data from our laboratory indicated that intermittent-mode stimulation (2 × 1 h/day) was not effective in enhancing motor recovery (Supplementary Figure 1). We chose anodal stimulation to selectively activate the pyramidal neurons. 26 Every other day during the stimulation period, we measured the individual threshold voltage required to evoke movement of the forelimb and/or face contralateral to the lesion by delivering stimulation to the reference electrode in motor cortex in each animal. We then set the experimental stimulation intensity equal to the half of individual movement threshold. Experimental stimulation intensities ranged from 1.0 V to 2.5 V; frequency (50 Hz) and pulse duration (194 ms) were the same for all animals in the experimental groups (WSG and SSG). These parameters were well tolerated in our previous study. 7 All rats received rehabilitative SPRT training throughout the stimulation period, in the same manner as described above for preoperative behavioral training.
Micro-PET/CT imaging acquisition and analysis
All animals in the WSG (n = 8) and SSG (n = 8) were scanned three times to evaluate the changes in regional glucose metabolic activity during the period in which cortical stimulation was being delivered: a control scan after electrode placement (PS0), seven days post-stimulation (PS7), and 14 days post-stimulation (PS14). To assess SCI-induced changes of regional glucose metabolic activity in the absence of cortical stimulation, we performed an additional scan in the SOG (n = 6) prior to the lesion (pre-lesion scan, PL), as well as the three scans on PS0, PS7, and PS14 (Figure 1). The animals were deprived of food for 12 h and then injected with F18-FDG (100 mCi/100 g) through the tail vein during brief isoflurane anesthesia, followed by a 30-min uptake period. The stimulation groups received cortical stimulation during the uptake period, whereas the SOG did not receive any cortical stimulation. After the uptake period, animals were again anesthetized with isoflurane and their heads fixed in a custom-made head holder. Respiration, heart rate, and body temperature were monitored in real time throughout the scanning procedure (BioVet System, m2m Imaging Corp., OH, USA). Static PET scans (25 min) and attenuation-correction computerized tomography (5 min) were performed with an Inveon PET/CT scanner (Siemens Medical Solutions, TN, USA).
After scanning, image data were corrected for attenuation and reconstructed using the iterative OSEM3D/MAP algorithm, achieving a spatial resolution of 1.4 mm full width at half maximum. MINC tool kit (McConnell Brain Imaging Centre, Montreal Neurological Institute, Montreal, Canada) and AFNI packages (National Institutes of Health, MD, USA) were used to analyze the images. 27 Each image was normalized using an intensity scaling approach and the images were coregistered to a standard histological template. 28 Coregistered data were spatially smoothed using an isotropic Gaussian kernel with 1.2 mm full width at half maximum.
To determine the stimulation effect, the subtraction of PS7 and PS14 to PS0 (PS7−PS0 and PS14−PS0) was performed in the WSG and SSG. In addition, to elucidate diaschisis throughout the experiment in absent of stimulation after the SCI, PS7, and PS14 were compared to PL for the SOG (PS7-PL and PS14-PL), using a group-level linear mixed-effect model. 29 Statistical maps were thresholded at the significance level (p < 0.0025, false discovery rate (FDR) q < 0.05), and overlaid on the histological template. The region of interest (ROI) was defined over anatomic brain regions that showed significant differences in regional glucose metabolic activity to quantify changes after cortical stimulation. In addition, the regional glucose metabolic activity of the ROIs that were activated or deactivated were compared with SPRT score using Pearson’s correlation to elucidate the relationship between the ROIs and motor recovery (p < 0.03, FDR q < 0.05).
Neurohistolology and c-Fos mapping
After two weeks of cortical stimulation, three rats in each group were sacrificed and processed for c-Fos immunohistochemistry. Briefly, rats were perfused with 4% paraformaldehyde (PFA), and post-fixed in 4% PFA overnight. Then, the brains were transferred to 30% sucrose for cryoprotection. Brain blocks were mounted in OCT compound (Cell Path, Newtown, UK) and coronal brain sections (40 µm) were cut on a freezing microtome (Cryocut 3000, Leica Biosystems, Nussloch, Germany). The brain sections were treated with 3% H2O2 in TBS and 1% normal goat serum, and incubated in c-Fos antibody (C-Fos 9F6 rabbit antibody, Cell Signaling Technology, Boston, MA, USA). In the following day, the sections were incubated in Polink-1 HRP detection system for rabbit antibody (GBI labs, Mukilteo, WA, USA). A color reaction was obtained by incubating sections in diaminobenzidine/peroxidase solution (DAB; 0.02%; 0.08% nickel sulfate) in TBS, and brain sections were mounted on gelatin-coated slides. Microscope images were reconstructed to show the extent of c-Fos expression in the whole brain. The resulting images were fast Fourier transform (FFT)-bandpass filtered in ImageJ (NIH, Bethesda, USA) and cell-density maps were created using a custom Matlab-based program (MathWorks, Natick, MA, USA). 30 ROIs were drawn over motor cortex, sensory cortex, the striatum, and the thalamus, and the c-Fos cells in each ROI were counted. Cells were counted automatically by calculating the mean image pixel intensity and applying a threshold. Automated counts were validated by microscopic counting. In addition, histological evaluation of capsular infarct lesion was performed with glial fibrillary acidic protein (GFAP), Luxol fast blue-PAS (LFB-PAS), and neurofilament protein L staining (NF). Capsular infarct volume and cortical damage after electrode implantation were measured using ImageJ software (NIH, Bethesda, MD, USA).
Statistical analysis
Data were analyzed using the statistical analysis software (OriginPro version 9.1, OriginLab, Northampton, MA, USA). Skilled reaching performances were analyzed using repeated measure analysis of variances for the effect of groups and times. One-way analyses of variance were used to test for differences between groups in the number of cells expressing c-Fos. Tukey’s post hoc comparison was performed to assess the significant differences between groups for each ROI.
Results
Neurohistological finding of capsular infarct lesion
Neurohistological analysis showed that the SCI lesion had losses of axons and myeline in the central cavity (LFB-PAS and NF), which was surrounded by reactive gliosis (GFAP), and infiltrating macrophages (Figure 2) show the losses of axons and myelin (LFB-PAS) in the cavity as well as destruction of capsular fibers (NF). The mean infarct volume was 0.78 ± 0.4 mm3 without significant group difference. However, the amount of cortical loss due to electrode compression was 0.53 ± 0.2 mm3 in the SSG and SOG and 2.70 ± 0.4 mm3 in the WSG, respectively. However, further derangement of the cortical neuronal layer, degenerative changes in individual neurons, and reactive gliosis in the vicinity of the area of cortical loss were not observed in the SSG or the WSG compared with the SOG.
Histological confirmation of circumscribed subcortical capsular infarct and the pattern of motor recovery during two weeks of cortical stimulation in sensory-parietal cortex. Glial fibrillary acidic protein (GFAP), Luxol fast blue-PAS (LBP-PAS), and neurofilament (NF) stainings show the circumscribed infarct lesion in the posterior limb of the internal capsule.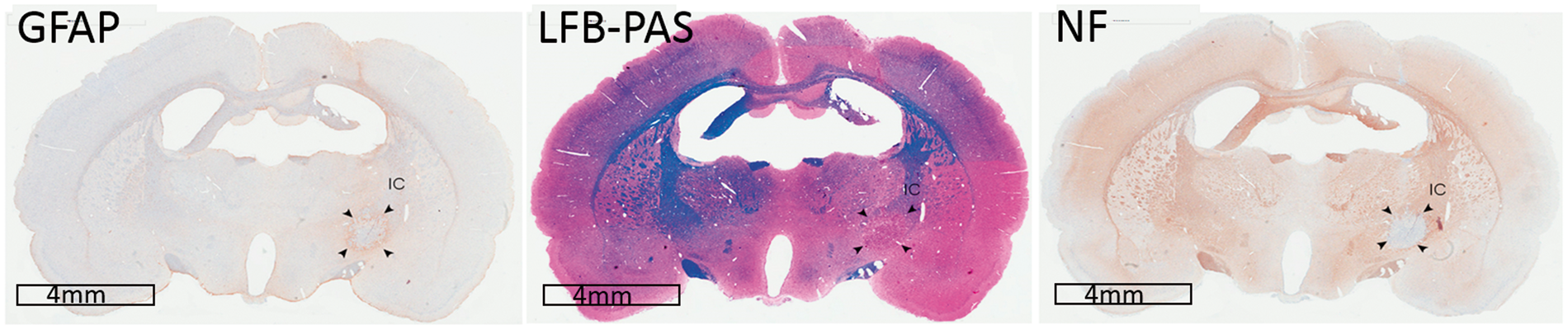
Behavioral recovery in animals with SCI after cortical stimulation of SPC
Figure 3 shows the behavioral patterns in three different groups. All experimental animals showed a marked decrease in SPRT performance immediately after photothrombotic lesioning in the PLIC. The SOG did not receive cortical stimulation and had persistently impaired reaching performance throughout the experiment. However, both cortical stimulation groups demonstrated a significant improvement in reaching scores, starting on the second day of stimulation for the WSG (p < 0.01) and the sixth day of stimulation for the SSG (p < 0.05). Recovery continued until the end of stimulation in the SSG but reached a plateau around the eleventh day in the WSG. Post hoc analysis showed that recovery in the WSG was significantly greater than the SSG between the second and fifth and between ninth and 14th day of stimulation, inclusive (p < 0.05) (Figure 3(a)). The WSG animals recovered to 72.9% of prelesion performance levels whereas the SSG animals recovered to 37% of prelesion levels (Figure 3(b)).
Single pellet reaching scores following capsular infarct and cortical stimulation over time. (a) Single pellet reaching scores dropped immediately after capsular infarct lesioning. Sensory-parietal cortical stimulation improved reaching scores in both the small stimulation group (SSG, p < 0.05) and the wide stimulation group (WSG, p < 0.01), whereas reaching scores were unchanged in the sham-operated group (SOG). Data represent the mean ± SEM. (b) The WSG showed greater improvement in reaching performance than the SSG.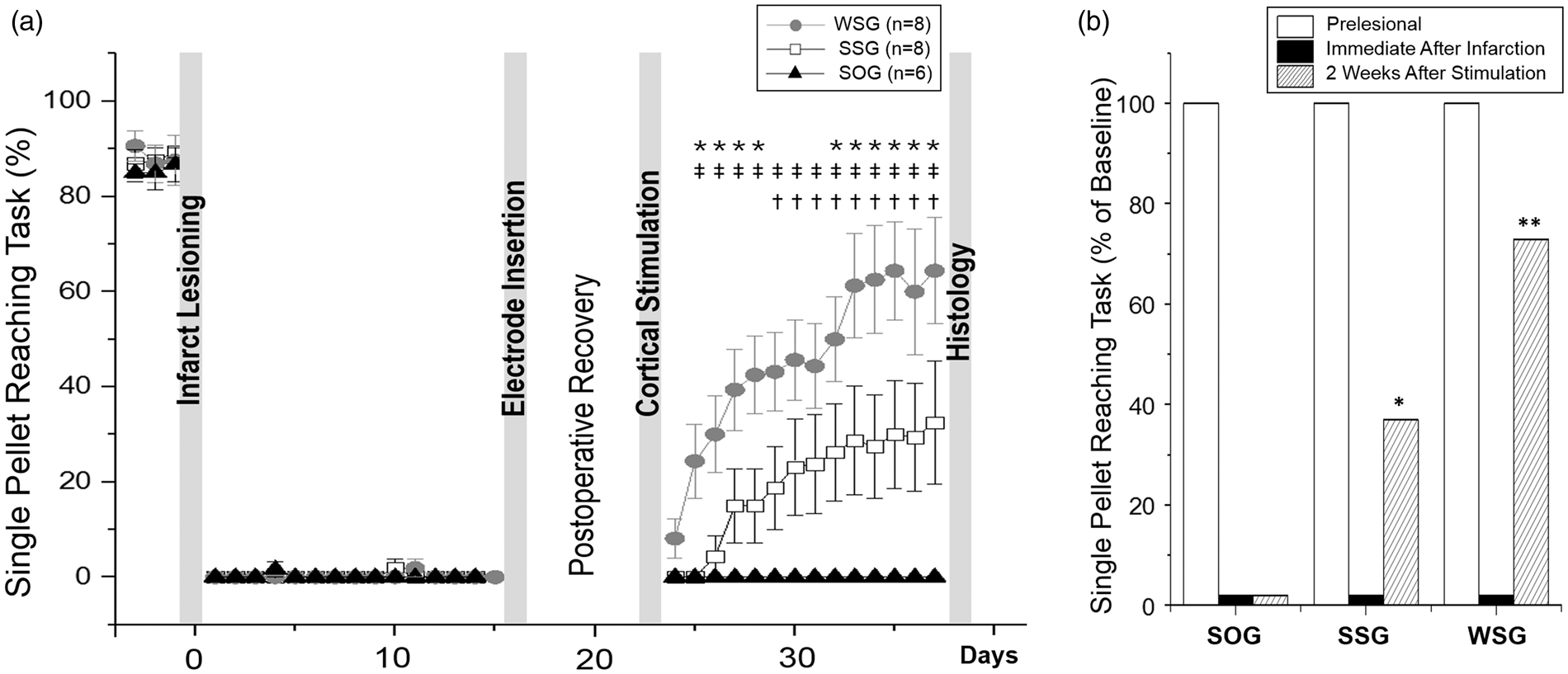
Longitudinal changes of regional glucose metabolism
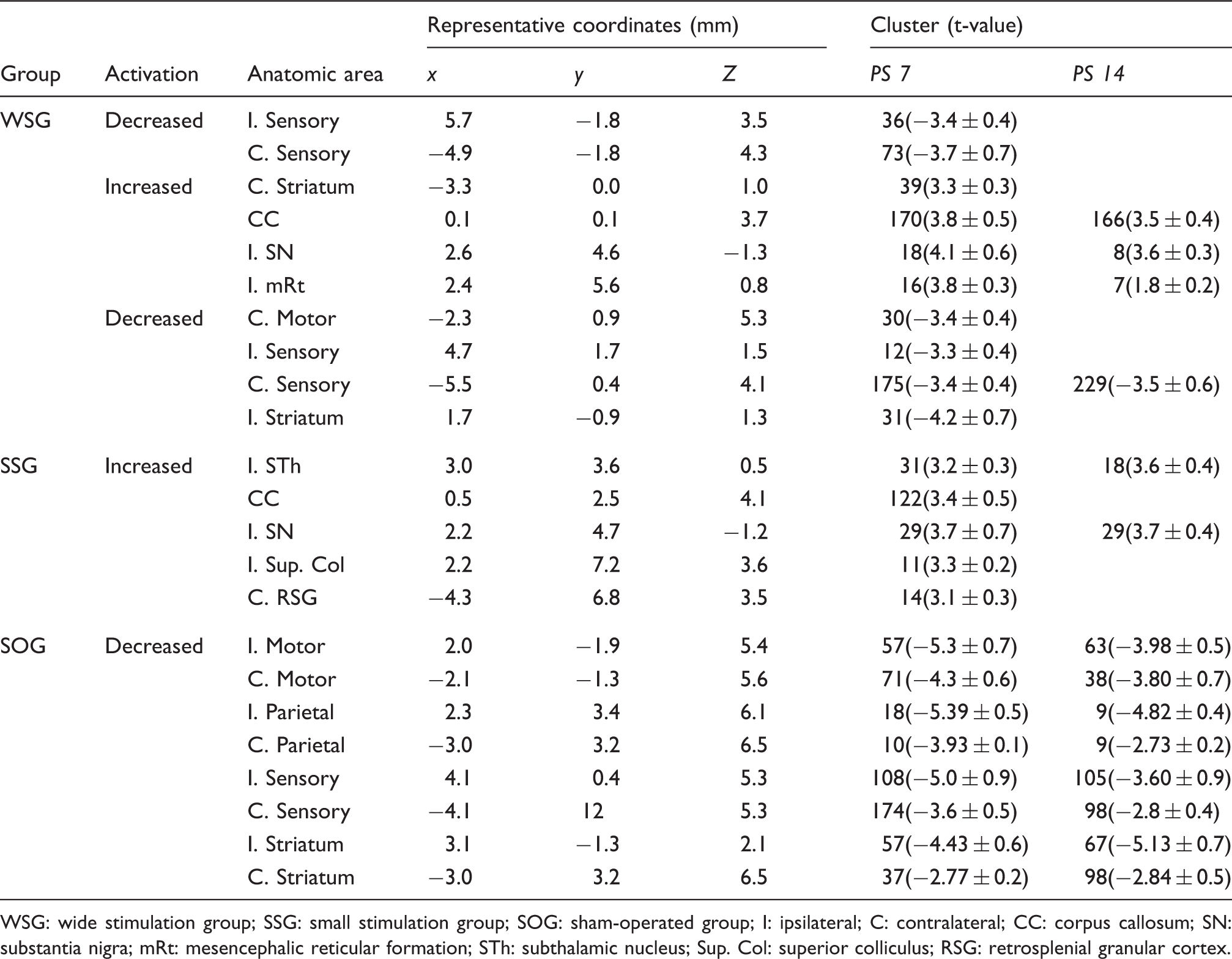
Figure 4 shows the longitudinal change of regional glucose metabolic activity in three different groups. The SOG animals showed persistent bilateral depression of regional glucose metabolic activity in sensorimotor cortices and striatum on PS7 and PS14, compatible with functional diaschisis. However, the WSG and SSG animals showed a reduction in depression of regional glucose metabolic activity, bilaterally in the WSG and predominantly on the stimulation side in the SSG (Figure 4(a)). The reversal of depression was more evident by PS14 than PS7. In addition, several subcortical structures showed the time-dependent activations in the stimulation groups. In both the SSG and the WSG, cortical stimulation induced activation in the corpus callosum (in both hemispheres) and in structures of the corticostriatal pathway, including the ipsilateral subthalamic nucleus, substantia nigra and mesencephalic reticular formation. In addition, activation of the pontine nucleus and contralateral striatum was observed in the WSG on PS7. In general, the same structures were activated in both groups, although to a lesser extent in the SSG. The WSG also showed more persistent activation of the corpus callosum and the substantia nigra (Figure 4(a), Table 1).
MicroPET imaging changes and its correlation with motor recovery after sensory-parietal cortical stimulation in the capsular infarct model. (a) Longitudinal changes in regional glucose metabolism in the capsular infarct model shows the persistence of diaschisis in SOG (PS7-PL and PS14-PL); however, stimulation groups show the reversal of diaschisis in the ipsilateral hemisphere in the small stimulation group (SSG) and in both hemispheres in the wide stimulation group (WSG) at post-stimulation days 7 (PS7-PS0) and 14 (PS14-PS0) (p < 0.0025, q < 0.05). Activation of subcortical structures was also observed (p < 0.0025, q < 0.05). The y coordinates represent distance from the anterior commissure. (b) Correlation analysis of changes in regional glucose metabolism (for PS7 and PS14) and reaching task performances across all groups (n = 22 animals). Five regions of interest showed positive correlations: the corpus callosum, contralateral striatum, ipsilateral and contralateral sensory cortex, and contralateral motor cortex. Brain areas with significant differences in regional glucose metabolism (rGM) between baseline and the 7th (PS7) or 14th (PS14) day post-stimulation (p < 0.0025, q < 0.05). WSG: wide stimulation group; SSG: small stimulation group; SOG: sham-operated group; I: ipsilateral; C: contralateral; CC: corpus callosum; SN: substantia nigra; mRt: mesencephalic reticular formation; STh: subthalamic nucleus; Sup. Col: superior colliculus; RSG: retrosplenial granular cortex.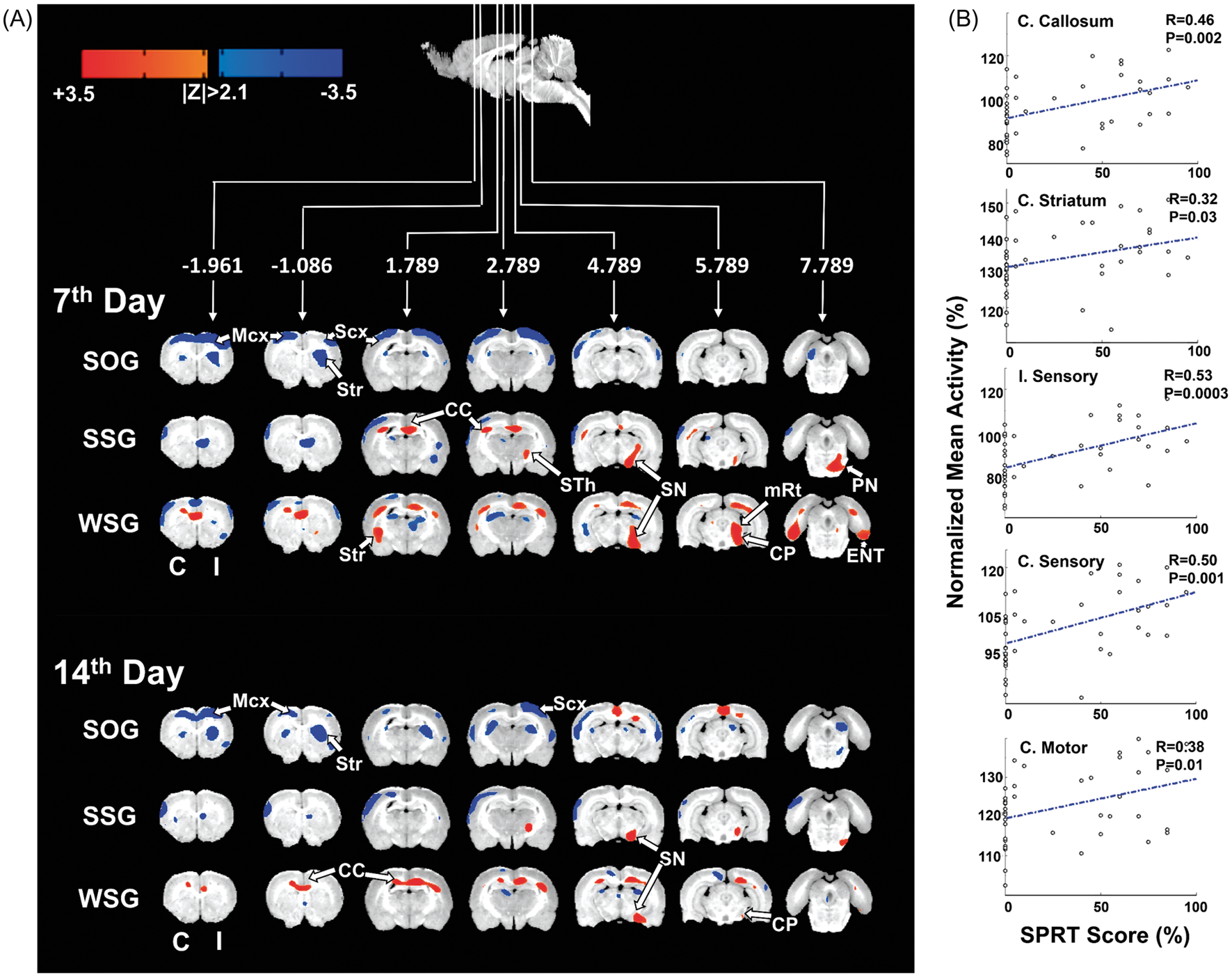
To determine the contribution of individual structures to promote the motor recovery, SPRT scores and changes of regional glucose metabolic activity in ROIs were correlated. Positive correlations were found in five ROIs, indicating that motor recovery was associated with the disappearance of depressed metabolic activity (ipsilateral sensory cortex, r = 0.53, p = 0.0003; contralateral sensory cortex, r = 0.50, p = 0.001; contralateral motor cortex, r = 0.38, p = 0.01), compatible with the reversal of diaschisis. In addition, activation of subcortical structures such as corpus callosum and contralateral striatum (corpus callosum, r = 0.46, p = 0.002; contralateral striatum, r = 0.32, p = 0.03) (Figure 4(b)) was positively correlated, indicating that these structures contribute to the motor recovery.
Patterns of c-Fos expression after chronic cortical stimulation
To further investigate which areas were activated by SPC stimulation, we performed immunostaining for c-Fos, a biomarker of neuronal activity, after two weeks of cortical stimulation. We observed significantly increased c-Fos expression in sensorimotor cortices bilaterally in both the SSG and WSG compared with the SOG (p < 0.05, one-way ANOVA), consistent with results from the microPET studies. However, c-Fos expression was also observed in both striata, whereas the microPET study showed activation of the contralateral striatum only in the WSG (Figure 5(a)). Motor cortex, sensory cortex, thalamus, and striatum in both hemispheres of the SSG and the WSG, except the ipsilateral thalamus of the SSG, showed the significant differences in number of c-Fos when compared to the SOG (p < 0.05, post hoc Tukey’s test). In addition, c-Fos count in contralateral sensory cortex and bilateral striatum of the WSG showed the significant differences to the SSG (p < 0.05, post hoc Tukey’s test) (Figure 5(b)).
Expression of c-Fos after sensory-parietal cortical stimulation. (a) Cell-density maps for c-Fos positive cells at three different bregma levels ( + 0.96, −2.04, and −4.68 mm) for each group, together with the atlas reference section (right). (b) Automated cell counts in four different regions of interest: motor cortex, sensory cortex, thalamus, and striatum.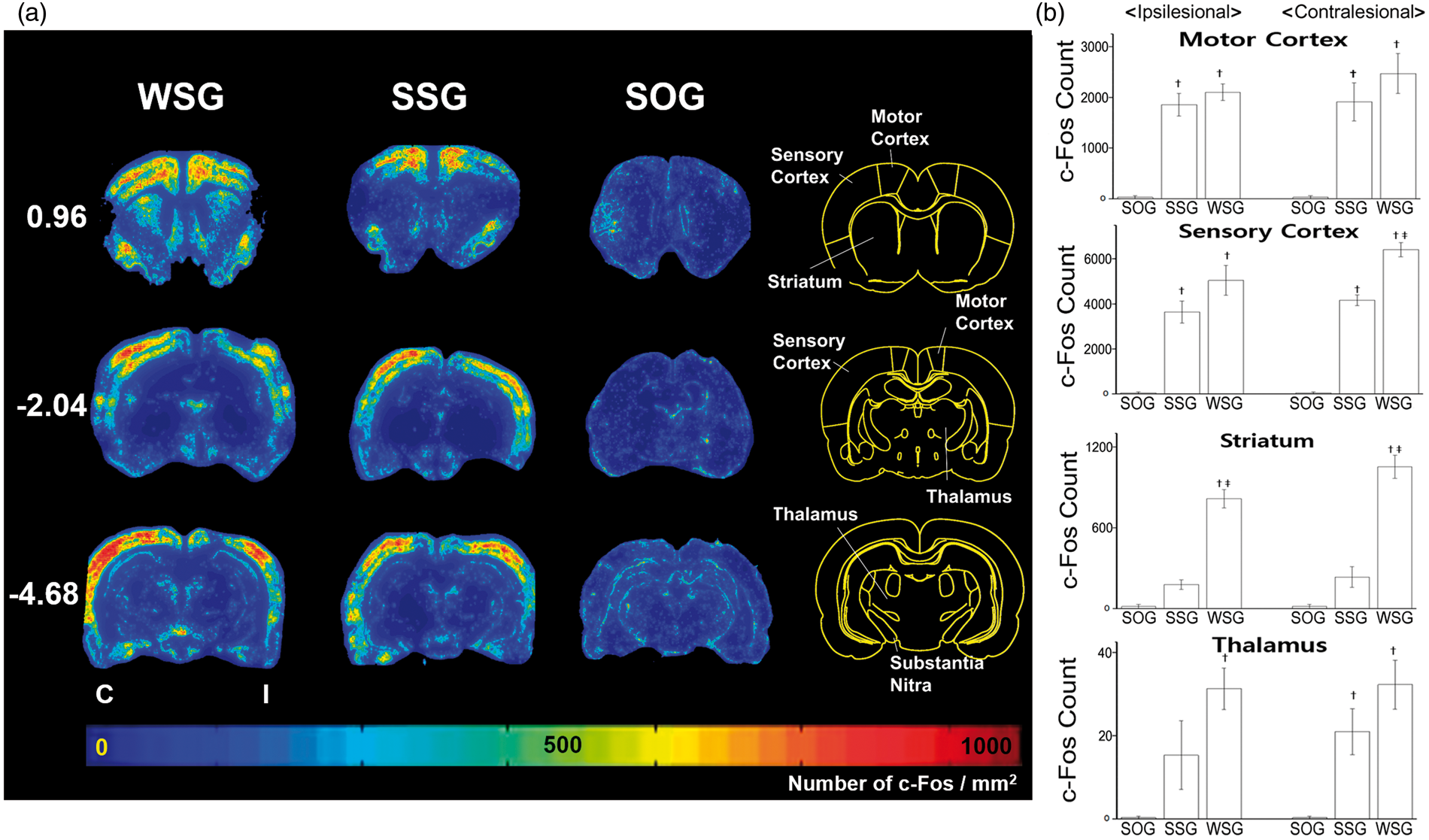
Discussion
In this study, we found that cortical stimulation in SPC could enhance the motor recovery even in the rats whose PLIC was completely disrupted. 19 The motor recovery was more successful in the WSG than the SSG, suggesting that the recruitment of wide cortical areas is mandatory for better recovery. The SPC stimulation contributed to the disappearance of diaschisis, and activation of corpus callosum, and contralateral striatum, leading to motor recovery.
Traditionally, the preferred target for cortical stimulation is generally motor and premotor cortex in the vicinity of the damaged cortex.31–33 Recent reports have revealed that multiple cortical areas are organized into a set of widely distributed motor networks that not only show post-stroke neurological deficits but also demonstrate functional reorganization during recovery.18,34–36 The SPC may be considered an attractive target for post-stroke neuromodulation for several reasons. First, the SPC plays a critical role in action planning, sensory guidance of movement, motor intention, and multisensory processing of the space surrounding the body, in the context of reciprocal fronto-sensory-parietal circuits.37,38 Second, the SPC has been targeted to drive functional reorganization and improve neuroplasticity in rehabilitative interventions. 39 Third, activity in sensory cortex is depressed during the early phase of stroke (diaschisis), which is likely strongly related to the neurological deficits.18,40 Reversing this depression through electrical stimulation may therefore be therapeutic. Fourth, previous trials with stimulation in sensory cortex achieved improvement of hand function in patients with chronic cerebral lesions. 41 Based on these reports, the SPC is likely to be a promising alternative target for post-stroke cortical stimulation. This study demonstrated that SPC stimulation promoted recovery of motor function after infarct lesioning of PLIC for both the SSG and the WSG.
In this study, the extent of cortical stimulation was associated with the degree of functional recovery. The larger electrode presumably recruited more neural structures, which resulted in better functional recovery. Further, after two weeks of cortical stimulation, the WSG recovered to 72.9% of prelesion SPRT scores, while the SSG recovered to 31% of prelesion levels. Once motor function was restored, it was maintained without decline. A motor-recovery follow-up conducted in a subset of the rats at 12 weeks after the start of stimulation showed that recovery lasted at least 12 weeks (data not shown). Previously, we demonstrated that circumscribed SCI induced the depression of resting-state brain activity in bilateral cortices and striatum, and this observation was compatible with ‘functional diaschisis’ likely related to the neurological deficits.18,19 Given that motor recovery was much better in the WSG than in the SSG, it is plausible that the extent of stimulation influenced both the degree and the speed of diaschisis reversal, leading to the difference in motor recovery. The microPET images support this result by showing wider activation of sensorimotor cortex and subcortical structures in the WSG than in the SSG. Recently, van Meer et al. showed that the degree of functional recovery after stroke is associated with the extent of restoration of ipsilesional corticospinal tracts, in combination with reinstatement of interhemispheric synchronization of neuronal signals and normalization of cortical network organization. 42 In line with this, the wide electrode appears to recruit a larger area for functional restoration of corticospinal tract.
Our results show that SPC stimulation produced time-dependent changes in regional glucose metabolic activity in cortical and subcortical structures, coupled with behavioral recovery. Notably, behavioral recovery was established over a longer period of time in the WSG than in the SSG. In the SSG, motor recovery reached a plateau on about the sixth day after the start of cortical stimulation, whereas motor recovery continued to rise in the WSG until about the eleventh day of stimulation, after which recovery plateaued. In gross accordance with this behavioral recovery, the microPET study showed that the progressive disappearance of diaschisis was more marked in the WSG. In addition, activation of the corpus callosum was prominent in the WSG on both PS7 and PS14, whereas the SSG showed partial disappearance of diaschisis and the absence of callosal activation on PS14. This suggests that the time limit of responsiveness to cortical stimulation is likely to differ depending on the extent of stimulation. Taken together, these data indicate that stimulation across a wide area of SPC can prolong the duration of motor recovery by altering activity in cortical and subcortical structures over the course of two weeks.
Signal changes in the white matter are rarely reported, despite playing an important connecting role during neuronal activation or deactivation. This is likely because white matter shows relatively small alterations in metabolism and blood flow compared with gray matter. Nonetheless, we observed that glucose metabolism in the white matter increased during cortical stimulation, suggesting increased interhemispheric communication and the possibility of white matter reorganization. 43 Additionally, activations of subcortical structures including the ipsilateral subthalamic nucleus, substantial nigra, and mesencephalic reticular formation in the corticostriatal pathway, the pontine nucleus and the contralateral striatum are thought to contribute to plasticity and functional reorganization to promote motor recovery.
The expression of c-Fos protein grossly matched the area of diaschisis—bilateral SPCs and striatum—supporting the suggestion that diaschisis may be reversed by SPC stimulation. It has previously been shown that chronic electrical stimulation of motor cortex results in c-Fos expression in contralateral as well as ipsilateral sensorimotor cortex. 44 The inconsistency in striatal activation between c-Fos immunostaining and microPET is probably because the latter is represented as statistical differences between the pre- and post-stimulation conditions, and the temporal summation of positive neural activity may have been different in the two hemispheres.
There is currently no specific strategy to promote motor recovery in patients with severe forms of SCI; furthermore, these patients show conflicting results with conventional rehabilitative interventions and have a poor clinical prognosis.22,45,46 This study therefore has important clinical implications because the results suggest an alternative means to accelerate motor recovery in patients with SCI. Cortical stimulation through epidural electrodes placed in motor cortex or the perilesional cortex has been used to promote neurological recovery in chronic stroke patients with motor impairments. 47 However, cortical stimulation for recovery from capsular stroke has rarely been reported. 48 Given that the target of stimulation is important for cortical stimulation-induced recovery, we propose SPC in the affected hemisphere as an attractive stimulation target for patients with chronic capsular stroke. We have observed that continuous-mode (24 h/day) cortical stimulation is markedly more effective than intermittent-mode stimulation, so we would advise implantation of neural stimulators equipped with the ability to deliver continuous-mode stimulation.
This study has some limitations. In particular, we observed loss of cortical structures underneath the electrode placements because of compression. However, this cortical loss from the electrodes did not change the resting-state brain activity, which was comparable to the activity observed without electrode placement in our previous study of longitudinal microPET changes after capsular infarct lesioning. 18 Furthermore, electrical stimulation over the areas of cortical loss did not interfere with the level of motor recovery observed in our study, indicating that the cortical loss was not a confounding factor for either conduction of the stimulation current or for the degree of motor recovery. However, parallel wire or ring electrodes would be a desirable alternative to avoid damage to the cortical structure, if such electrodes are capable of generating the same extent and strength of electrical field. 49
In conclusion, we have shown that continuous SPC stimulation in the affected hemisphere significantly improves motor recovery of severe deficits in a rodent model of SCI. We ascribe the recovery to reversal of cortical diaschisis and activation of the corpus callosum and subcortical structures in the corticostriatal pathway as demonstrated by microPET and c-Fos mapping study. The SPC stimulation may therefore be a useful strategy for overcoming the limits of rehabilitative training in patients with severe forms of SCI.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Institute of Medical System Engineering (iMSE; K04804) & GIST-Caltech Collaborative Fund (K04592) in GIST and by Basic Science Research Program through NRF of Korea funded by Ministry of Science, ICT and future Planning (NRF-2013R1A2A2A01067890).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
RGK performed experiment and wrote the manuscript. JR and HSK performed experiment. JC and PRN analyzed PET data. JMK and MCL analyzed and interpreted histological data. HIK, MCL and PRN designed and supervised experiment and entire work of manuscript. All authors read and approved the final manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.