Abstract
Background:
Major depressive disorder (MDD) is a globally prevalent psychiatric condition associated with significant morbidity and often suboptimal treatment outcomes. Probiotics have emerged as a promising adjunctive therapy targeting the gut–brain axis in MDD.
Aims:
This systematic review aimed to critically evaluate the efficacy of probiotics in the treatment of clinically diagnosed MDD, assess methodological quality and therapeutic readiness, and identify research gaps relevant to clinical application.
Methods:
A systematic search of MEDLINE, Web of Science, and PubMed was conducted in June 2023 in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analysis guidelines. Double-blind, placebo-controlled, randomised controlled trials (RCTs) involving adults with MDD were included. Risk of bias was assessed using the Cochrane Risk of Bias tool.
Results:
Thirteen RCTs involving 437 adults aged 18–75 years with MDD were included. Most trials reported improvements in depressive symptoms, particularly in outpatients with mild-to-moderate MDD treated with multi-strain formulations containing Lactobacillus and Bifidobacterium over 4–8 weeks. Secondary outcomes across metabolic, inflammatory, cognitive, neurotrophic, and gastrointestinal domains showed variable improvements. Probiotic benefits were most consistent in Iranian outpatients, more modest in European inpatients, and positive but variable in East Asian outpatients. Risk of bias was predominantly low, though attrition, selective reporting, and methodological heterogeneity remained sources of bias.
Conclusions:
Probiotics appear safe and well-tolerated, with modest adjunctive benefit in MDD, most evident for multi-strain formulations in younger, outpatient populations with mild-to-moderate symptoms, supporting an individualised, context-dependent approach. Future trials should stratify patients, extend intervention durations, and integrate mechanistic endpoints to refine patient-tailored recommendations.
Introduction
Major depressive disorder (MDD) is a heterogeneous disorder with a range of associated psychosocial and neurobiological underpinnings (Johnson et al., 2023). In a typical psychiatric setting, MDD remains a clinical diagnosis that benefits from the clinician’s expertise and overall judgement, integrating various information derived from structured clinical interviews, validated diagnostic manuals and rating scales, and baseline laboratory investigations to rule out organic causes of depression (Bilello et al., 2015; Herrman et al., 2022; Kennedy, 2022; Maj et al., 2020; Otte et al., 2016). Antidepressants are widely prescribed as the first-line pharmacologic treatment for MDD; however, they are often associated with suboptimal outcomes, poor adherence, and adverse effects.
A landmark multicentre trial reported that only about one-third of patients achieved remission after an initial adequate course, with a first treatment step remission rate of 36.8%, and that the cumulative remission rate after up to four sequential steps reached 67%, while relapse occurred in approximately half of those who remitted after completing all four steps within 1 year (Rush et al., 2006). Similar patterns emerged in a U.S. insurance-claims cohort of over 84,000 adults: approximately one-third relapsed within 2 years, often requiring treatment escalation and generating higher healthcare costs (Touya et al., 2022). Primary-care data show that a third of more than 20,000 adults discontinued antidepressants within 3 months and half within 6 months, frequently citing adverse effects such as gastrointestinal symptoms, sedation, sexual dysfunction, and weight gain (Garcia-Marin et al., 2023). These limitations underscore the need for additional, well-tolerated therapeutic strategies that can improve outcomes beyond standard treatment, especially amid the rising global prevalence of MDD (Mahmud et al., 2023; Li et al., 2021; Proudman et al., 2021).
Probiotics appear to be one such promising option with therapeutic potential in the treatment of depression, with initial hypothetical insights on their use in MDD dating back to 2005 (Logan and Katzman, 2005). Since then, an upsurge of pre-clinical studies has followed to identify potential anti-depressive mechanisms of probiotics in depression (Alli et al., 2022; Goh et al., 2019; Huang et al., 2016; Johnson et al., 2023; Nikolova et al., 2019). Emerging evidence suggests that probiotics, particularly multi-strain formulations containing strains of Lactobacillus and Bifidobacterium, are associated with significant reductions in depressive symptom severity, modulation of inflammatory and metabolic biomarkers, and improvement in gastrointestinal comorbidities (Johnson et al., 2023; Lee, 2025; Rahmannia et al., 2024). Numerous systematic reviews and meta-analyses have further supported the clinical use of probiotics as an adjunct to antidepressants (Alli et al., 2022; Goh et al., 2019; Huang et al., 2016; Lin et al., 2023; Musazadeh et al., 2022; Ng et al., 2023; Nikolova et al., 2019, 2021).
In keeping with an evidence-based medicine (EBM) approach, reliable reference to research within the clinically defined cohort is necessary to ascertain the true potential of probiotics as a therapeutic option in MDD. Careful, rigorous scrutiny of the body of scientific evidence is crucial to balance the claims of the marketing industry with the clinical needs of the intended target population. This responsibility becomes even more remarkable when the product is promoted for a therapeutic potential within the healthcare sector, targeting a specific patient population. Developing robust scientific research methodologies that serve as reference points for clinicians and bridge the translational gaps to promote practical implementation in real-world medicine is pivotal. Randomised controlled trials (RCTs) have been traditionally considered the gold standard of interventional studies and EBM (Ahuja, 2019). Therefore, in this systematic review, we aim to examine RCTs evaluating the efficacy of probiotics in MDD to understand their therapeutic readiness within the clinical domain of depression management. Following this, we will propose recommendations for future research directives of the intended new therapy.
Materials and Methods
Eligibility criteria
As part of an imperative primary step in identifying suitable studies for this review, only RCTs of probiotics involving clinical subjects with a diagnosis of MDD, with depression symptomology or depression-related measures as primary outcome measures, were considered. Studies with probiotics as either an adjunct to conventional depression treatment or a stand-alone intervention with a placebo-controlled arm were included. Detailed characteristics of the included studies are presented in “Study Characteristics.”
Study characteristics
Studies with subjects that were not clinically diagnosed MDD patients were excluded. Studies that assessed probiotics’ efficacy on depressive symptoms, general mental health, and stress parameters in subjects without a clinical diagnosis of MDD were also excluded. Depression secondary to a medical condition or drug-induced, depression as a comorbid condition, treatment-resistant, and subthreshold depression were excluded. Paediatric, adolescent, and elderly (above 65 years old) subjects were excluded. Co-administration of probiotics with prebiotics, minerals, or other supplements without at least one probiotic-as-a-stand-alone intervention arm in depressed subjects was excluded. Non-RCT interventional studies were excluded.
Search strategy
This systematic review was performed per the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA; Page et al., 2021). A literature search was conducted in MEDLINE, Web of Science, and PubMed electronic databases for articles published since the establishment of the first record in the databases until June 2023. Two independent reviewers performed the literature search using search formulas adjusted according to the databases. In MEDLINE, the following keywords and Medical Subject Heading terms were used: (“depression” OR “major depressive disorder” OR “depressive symptoms”) AND (“probiotics” OR “Bifidobacterium” OR “Lactobacillus” OR “psychobiotics”). In PubMed and Web of Science, the following search terms were used: (“depression” OR “major depressive disorder” OR “depressive symptoms”) AND (“probiotics” OR “Bifidobacterium” OR “Lactobacillus” OR “psychobiotics”) AND (“clinical trials” OR “clinical trials as a topic” OR “randomised controlled trial” OR “Randomised Controlled Trials as Topic” OR “controlled clinical trial” OR “Controlled Clinical Trials as Topic” OR “random allocation” OR “randomly allocated”). The search results were narrowed by selecting “RCT” and “humans” in MEDLINE and PubMed, whereas in the Web of Science, “article” was selected. Search strategies followed the Population, Intervention, Comparison, and Outcome (PICO) framework, including adults (⩾18 years) with a clinical diagnosis of MDD, receiving probiotics as adjunct or stand-alone therapy, with outcomes of depressive symptoms or related measures.
The references of all the search results were imported into the web-based online software Covidence for screening and quality assessment by two independent reviewers. Any reviewer discrepancies were resolved through discussions and cross-checking with a third independent reviewer. Title and abstract screenings were performed to identify relevant studies. Full texts were obtained for all the relevant studies and reviewed to finalise eligible studies that met the predefined inclusion and exclusion criteria.
Data extraction and quality assessment
Data extraction was performed based on a tailored form gathering information on authorship, publication year, country, study design, study population, diagnosis establishment, intervention and comparison, duration, and primary outcome measures. This was counter-checked by the third independent reviewer.
Quality assessment was done with reference to the built-in Cochrane Risk of Bias template on Covidence and Cochrane Handbook for Systematic Reviews of Interventions version 6.3, upon consensus by all three independent reviewers (Higgins et al., 2022). The eligible studies were evaluated based on seven key domains: sequence generation (selection bias), allocation concealment (selection bias), blinding of participants and personnel for all outcomes (performance bias), blinding of outcome assessors for all outcomes (detection bias), incomplete outcome data for all outcomes (attrition bias), selective outcome reporting (reporting bias), and other sources of bias. The risk of bias for each domain was reported as either high, low, or unclear. Refer to “Risk of Bias Assessment” for more details.
Results and Discussions
Study Selection
The PRISMA flow chart presents the study selection process, as shown in Figure 1. Thirteen studies were included out of 333 obtained from database searches. One hundred and sixty four studies were gathered from MEDLINE, 91 from Web of Science, and 78 from PubMed. These studies were imported into the Covidence software for further screening. In all, 56 duplicates were identified and removed, leaving 277 studies for abstract/title screening. Of 277 studies, 235 were irrelevant based on the search criteria. Studies conducted on animal models, review articles, non-interventional studies, study protocols, maternal subjects, and those not meeting the PICO criteria were considered irrelevant. Full texts were retrieved for the remaining 42 studies and extensively reviewed for eligibility. In total, 29 studies were excluded. Studies investigating probiotic efficacy in subjects with subthreshold depression (n = 1) and non-RCT interventional studies (n = 2) were excluded. Studies that co-administered probiotics with supplements that lacked at least one probiotic-as-a-stand-alone intervention arm (n = 4), and studies that did not have depression or depression-related measures as the primary outcome measure (n = 2) were also excluded. Studies (n = 21) with clinical diagnosis of MDD that were neither established nor clearly indicated in the study population, that is, self-referral, depression establishment based on single-handed questionnaires, depressive symptoms, and generalised mental health assessment. Finally, the remaining 13 studies consisting of 7 primary studies (Akkasheh et al., 2016; Kazemi et al., 2019; Reininghaus et al., 2020; Rudzki et al., 2019; Schaub et al., 2022; Tian et al., 2022, 2023), 4 post hoc analyses (Heidarzadeh-Rad et al., 2020; Kazemi et al., 2020; Schneider et al., 2023; Yamanbaeva et al., 2023) of prior studies (Kazemi et al., 2019; Schaub et al., 2022), and preliminary and extended studies (Kreuzer et al., 2022; Reiter et al., 2020) of one of the primary studies (Reininghaus et al., 2020) were deemed eligible and included in this review. The post hoc analyses, preliminary and extended studies, were included due to varying depression-related outcome assessments and reporting from their respective primary studies. The detailed study characteristics of the included studies are presented in Table 1.
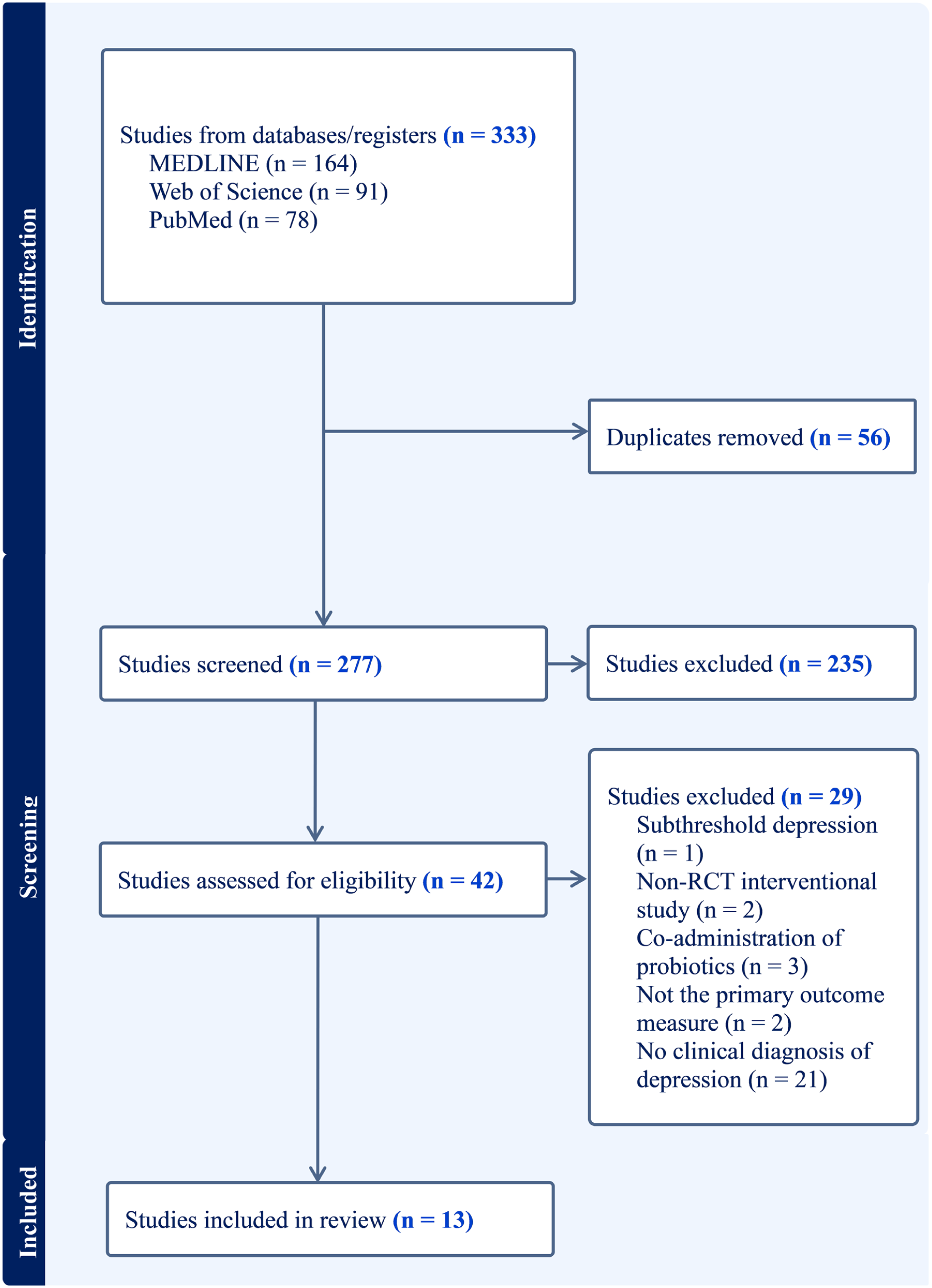
PRISMA flowchart outlining the article screening process.
Study characteristics of the 13 included studies.
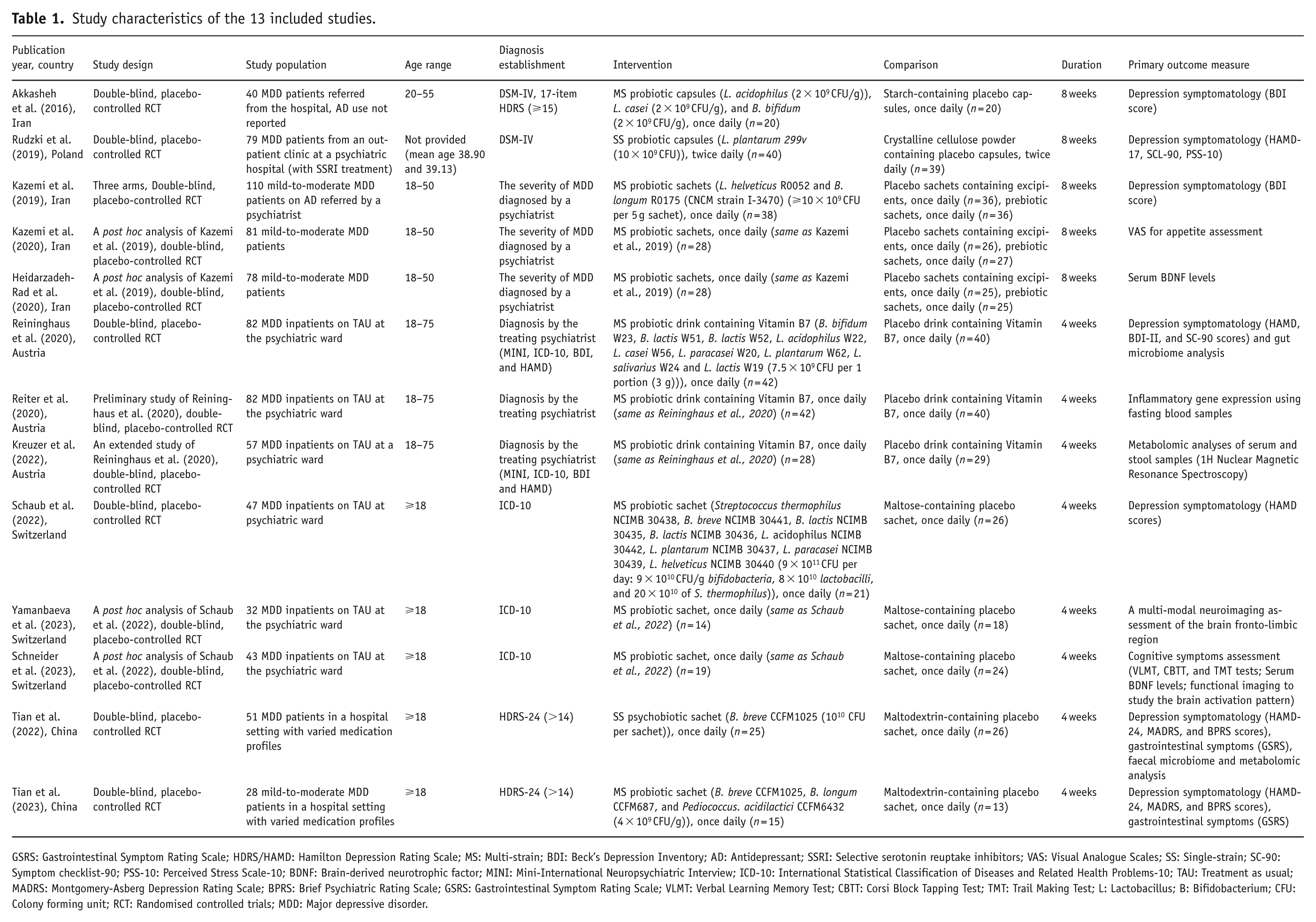
GSRS: Gastrointestinal Symptom Rating Scale; HDRS/HAMD: Hamilton Depression Rating Scale; MS: Multi-strain; BDI: Beck’s Depression Inventory; AD: Antidepressant; SSRI: Selective serotonin reuptake inhibitors; VAS: Visual Analogue Scales; SS: Single-strain; SC-90: Symptom checklist-90; PSS-10: Perceived Stress Scale-10; BDNF: Brain-derived neurotrophic factor; MINI: Mini-International Neuropsychiatric Interview; ICD-10: International Statistical Classification of Diseases and Related Health Problems-10; TAU: Treatment as usual; MADRS: Montgomery-Asberg Depression Rating Scale; BPRS: Brief Psychiatric Rating Scale; GSRS: Gastrointestinal Symptom Rating Scale; VLMT: Verbal Learning Memory Test; CBTT: Corsi Block Tapping Test; TMT: Trail Making Test; L: Lactobacillus; B: Bifidobacterium; CFU: Colony forming unit; RCT: Randomised controlled trials; MDD: Major depressive disorder.
Study characteristics
Risk of bias assessment
Among the included studies, only one trial demonstrated an overall low risk of bias across all domains (Kazemi et al., 2019), while the remainder were rated as unclear due to concerns in specific domains, most commonly attrition bias, selective reporting, and other methodological issues, as shown in Table 2.
Summary of risk of bias assessment.
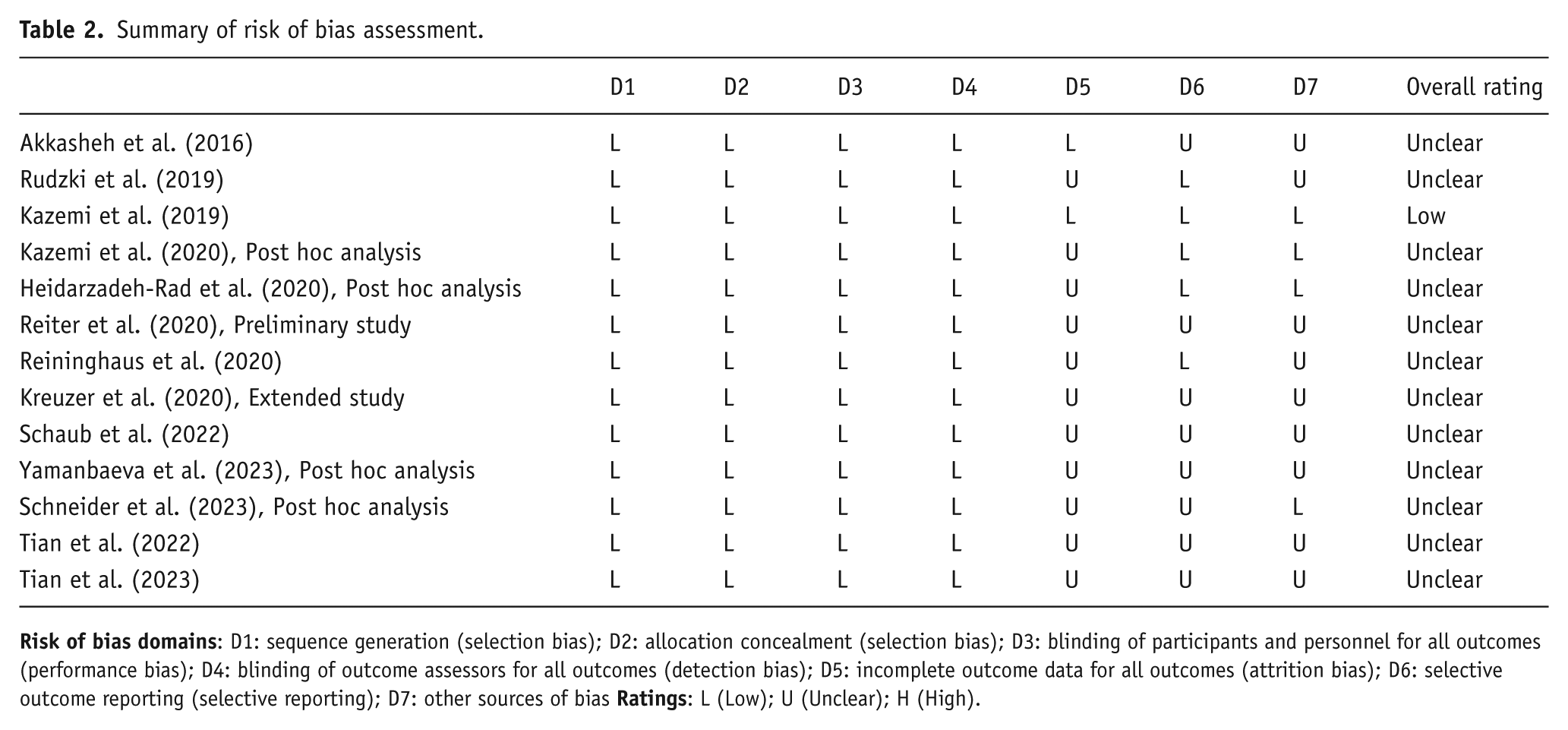
Attrition bias was a recurring concern, particularly in trials where discrepancies were noted between the number randomised and the number analysed, or where compliance-based exclusions reduced the modified intention-to-treat population. For instance, the PROVIT study in Austria (Reininghaus et al., 2020) reported prespecified clinical outcomes transparently, yet uncertainties remained regarding the handling of incomplete data. Similarly, the Swiss inpatient trial (Schaub et al., 2022) excluded non-compliant participants, thereby reducing the effective sample size and raising the possibility that observed benefits reflected a more adherent subgroup. The Chinese outpatient trials (Tian et al., 2022, 2023) demonstrated positive clinical effects but provided limited detail on attrition, creating some uncertainty about the robustness of effect estimates. Such patterns may compromise validity by introducing systematic differences between completers and non-completers, reduce reliability by diminishing statistical power, and constrain generalisability, particularly in inpatient or moderate-to-severe cohorts where dropout was more pronounced. By contrast, outpatient studies conducted in Iran (Akkasheh et al., 2016; Kazemi et al., 2019) generally reported lower attrition, lending greater confidence to their findings and supporting applicability to similar populations.
Selective reporting bias was evident, particularly in secondary analyses derived from larger trials. The Austrian PROVIT study (Reininghaus et al., 2020) reported a positive outcome but no superiority of probiotics over placebo on depressive symptoms, with follow-up analyses from the same cohort highlighted mechanistic findings, including reduced IL-6 gene expression (Reiter et al., 2020) and stool metabolomic alterations (Kreuzer et al., 2022). Similarly, the Swiss inpatient trial (Schaub et al., 2022), which reported clinical improvements, generated secondary analyses that emphasised selective neuroimaging changes in fronto-limbic networks (Yamanbaeva et al., 2023) and cognitive and biomarker outcomes (Schneider et al., 2023). These insights are valuable for elucidating potential biological pathways; however, selective emphasis on positive secondary endpoints reduces transparency and limits reproducibility. Even trials that reported favourable primary outcomes left gaps in the reporting of secondary measures, including adverse events and microbiome outcomes, which constrain interpretation of mechanisms and safety profiles (Akkasheh et al., 2016; Kreuzer et al., 2022; Reininghaus et al., 2020; Reiter et al., 2020; Schaub et al., 2022; Schneider et al., 2023; Tian et al., 2023; Yamanbaeva et al., 2023).
Additional sources of bias included insufficient reporting of baseline depression severity and treatment status (Akkasheh et al., 2016; Tian et al., 2022, 2023), variability in diagnostic procedures for confirming MDD, and limited transparency regarding randomisation methods in smaller or post-hoc analyses. Some studies also applied broadened inclusion criteria of MDD patients in terms of severity and antidepressant treatment profile (Akkasheh et al., 2016; Rudzki et al., 2019; Tian et al., 2022, 2023), introducing heterogeneity that complicates attribution of observed changes solely to probiotic supplementation.
Overall, attrition and selective reporting emerged as the most consistent sources of bias across the included trials. These factors can compromise validity by distorting effect estimates, undermine reliability by reducing statistical power and reproducibility, and restrict generalisability by limiting the applicability of findings beyond the study samples. Nevertheless, the risk of bias was consistently low for sequence generation, allocation concealment, and blinding of both participants and outcome assessors (D1–D4), indicating that randomisation methods, concealment strategies, and blinding procedures were generally robust. Therefore, most trials were considered methodologically rigorous, with the main concerns confined to attrition, outcome reporting, and other methodological heterogeneity.
Key methodological characteristics of the included studies
Patient selection
All the included seven primary studies recruited adult subjects (⩾18 years old) from either a pool of outpatients or inpatients from hospital settings, matched to the pre-determined inclusion criteria (Akkasheh et al., 2016; Kazemi et al., 2019; Reininghaus et al., 2020; Rudzki et al., 2019; Schaub et al., 2022; Tian et al., 2022, 2023). Among these, two studies recruited inpatients receiving Treatment as usual (TAU) in psychiatric care settings (Reininghaus et al., 2020; Schaub et al., 2022). A clinical diagnosis of MDD by a psychiatrist using a structured diagnostic interview, Mini-International Neuropsychiatric Interview (MINI), diagnostic tool ICD-10, and rating scales Beck’s Depression Inventory (BDI) and Hamilton Depression Rating Scale (HAMD) reported by two studies were deemed appropriate (Kreuzer et al., 2022; Reininghaus et al., 2020). Only two studies reported baseline severity of MDD, which ranged from mild to moderate depression (Kazemi et al., 2019; Tian et al., 2023). Standard exclusion criteria include pregnant and breastfeeding women, and the existence of psychiatric or other severe medical comorbidities. Three studies explicitly mentioned the use of probiotics and/or antibiotics prior to participation in the trial, commonly 2 months, as exclusion criteria (Akkasheh et al., 2016; Kazemi et al., 2019; Rudzki et al., 2019). The most comprehensive list of stringent exclusion criteria encompassing possible organic causes of depression, neurological and immune-related co-morbidities, suicidality, drug dependency, history of head trauma or surgery, and the regular intake of probiotics in the last year before participation in the trial was provided by Reiter et al. (2020). The characteristics of the study populations in the post hoc analyses, preliminary, and extended studies were as defined in their respective primary studies.
Probiotic intervention
All the primary studies compared probiotic strains of Lactobacillus, Bifidobacterium, and placebos. Probiotics used in the studies were primarily supplied by pharmaceutical companies or obtained from local drug stores. All the probiotics used were reported with their respective strain designations. The most commonly studied probiotic strains were B. breve, B. bifidum, B. lactis, L. plantarum, L. acidophilus, and L. helveticus to examine their anti-depressive effect on MDD patients. Two studies included probiotic strains of Streptococcus and Pediococcus (Schaub et al., 2022; Tian et al., 2023). Only one study used the term psychobiotic, indicating the use of a probiotic strain, B. breve CCFM1025, that has been proven to exert anti-depressant-like effects in pre-clinical models (Tian et al., 2022). Two studies used single-strain probiotic intervention using L. plantarum 299v and B. breve CCFM1025, respectively (Rudzki et al., 2019; Tian et al., 2022). Schaub et al. (2022) used the greatest number of probiotic strains with the highest dosage of 900 billion colony-forming unit (CFU) bacteria daily.
Most studies used probiotics as an adjunct to depression treatment instead of a stand-alone intervention. One study had no information on the medication status of the study participants (Akkasheh et al., 2016), while two studies had no restrictions on medication use (Tian et al., 2022, 2023). In another study, antidepressant (AD) selective serotonin reuptake inhibitors (SSRI) were commenced for the included participants, and those already on SSRI were continued (Rudzki et al., 2019). Reininghaus et al. used a probiotic drink and a placebo drink containing Vitamin B7 (biotin) for ethical reasons to benefit all the study participants as a health supplement as it presumably does not influence the mood outcome. However, one of their findings of the upregulated biotin metabolism in the probiotic group alone argued that it could be due to an augmented biotin availability or production by either the probiotic species, native gut microbiota, or dietary means (Kreuzer et al., 2022; Reininghaus et al., 2020; Reiter et al., 2020).
During intervention phases, participants were generally instructed to keep a 3-day (weekly) record of their dietary intake and physical activities. The compliance was checked by asking the participants to return their product containers at every follow-up visit and recording the remaining products. Intervention phases lasted 4–8 weeks at varying dosages and probiotic strain combinations.
Outcome measures
Depression symptomatology was evaluated using a self-rated scale, BDI (Akkasheh et al., 2016; Kazemi et al., 2019), and/or a clinician-rated scale, HAMD (Reininghaus et al., 2020; Rudzki et al., 2019; Schaub et al., 2022; Tian et al., 2022, 2023), as the primary outcome measure. Post hoc analyses focused on other depression-related measures, including appetite, serum brain-derived neurotrophic factor (BDNF) level, cognitive function, and specific brain regions, mainly to elucidate the possible mechanism of actions of probiotics in depression (Heidarzadeh-Rad et al., 2020; Kazemi et al., 2019; Schneider et al., 2023; Yamanbaeva et al., 2023). A preliminary and extended study by Reininghaus et al. focused on gene expression of inflammatory genes and metabolomic profiles, respectively (Reiter et al., 2020; Schneider et al., 2023). Tian et al. (2022) indicated gastrointestinal symptoms, faecal microbiome, and tryptophan metabolism analyses as primary outcome measures. Secondary outcome measures in primary studies include faecal microbiome analysis, gastrointestinal-related parameters, inflammatory markers, metabolomic profiles, cognitive function, and brain functional and structural assessments. These measures were inferred to be related to the pathomechanism of depression. Gastrointestinal symptoms were inferred as a common co-morbidity in MDD. Blood and stool samples of the participants were the common investigation tools. Brain imaging using magnetic resonance imaging has been used to provide a more extensive plausible role of probiotics in influencing activation patterns in specific brain regions, that is, the putamen and hippocampus, which were related to depression (Schaub et al., 2022; Schneider et al., 2023; Yamanbaeva et al., 2023).
Compliance rates and adverse events
Compliance rates were measured based on the count of the returned number of investigation products. Three studies reported reasonably high compliance rates; 90% of capsules were taken in both probiotic and placebo groups, a mean of 91.9% with a standard deviation of 5.53% in the probiotic group and 88% in both groups, respectively (Akkasheh et al., 2016; Kazemi et al., 2019; Schaub et al., 2022). The dropout rate was reported to be 30% in the probiotics and 13% in the placebo groups (Schaub et al., 2022). Tian et al. (2022) predicted a 25% dropout rate in the probiotic group. Regarding adverse events, only two studies reported adverse events possibly attributable to the intervention. Adverse events include gastrointestinal disturbances, nausea, fever and body aches, increased appetite, and worsened mental status symptoms unrelated to depression (Kazemi et al., 2019). Another study reported no adverse events among the study participants (Rudzki et al., 2019).
Evaluation of probiotic efficacy in MDD
Depression symptomatology
Most studies demonstrated significant positive results favouring improvement in depression symptomatology in a minimum of 4 weeks (Reininghaus et al., 2020; Schaub et al., 2022; Tian et al., 2022, 2023) and a maximum of 8 weeks of varying trial durations (Akkasheh et al., 2016; Kazemi et al., 2019) compared to placebo arms (Table 3). All the studies that demonstrated significant improvement in depression symptomatology used multi-strain probiotics of largely Lactobacillus and Bifidobacterium genera (Akkasheh et al., 2016; Kazemi et al., 2019; Reininghaus et al., 2020; Schaub et al., 2022; Tian et al., 2023). Among these, Schaub et al. (2022) used the highest dose of probiotics at 900 billion CFU/day with the most bacterial strain combinations, including S. thermophilus, to evaluate probiotics’ clinical and neural effects in 4 weeks. One study that administered single-strain probiotic L. plantarum 299v did not find improvement in depression symptomatology, but saw significant improvement in other depression-related measures (Rudzki et al., 2019). Another study that administered a single-strain probiotic B. breve CCFM1025 demonstrated significant improvement in depression symptomatology (Tian et al., 2022). Probiotics were administered as an adjunct to depression treatment in most studies (Kazemi et al., 2019; Reininghaus et al., 2020; Rudzki et al., 2019; Schaub et al., 2022). Among these, three studies showed improvement in depression symptomatology (Kazemi et al., 2019; Reininghaus et al., 2020; Schaub et al., 2022), whereas one showed no changes (Rudzki et al., 2019). Two other studies did not restrict medication use (Tian et al., 2022, 2023), while no medication status was reported in one study (Akkasheh et al., 2016) with favourable outcomes in depression symptomatology.
Outcome assessments and global findings in the included studies.
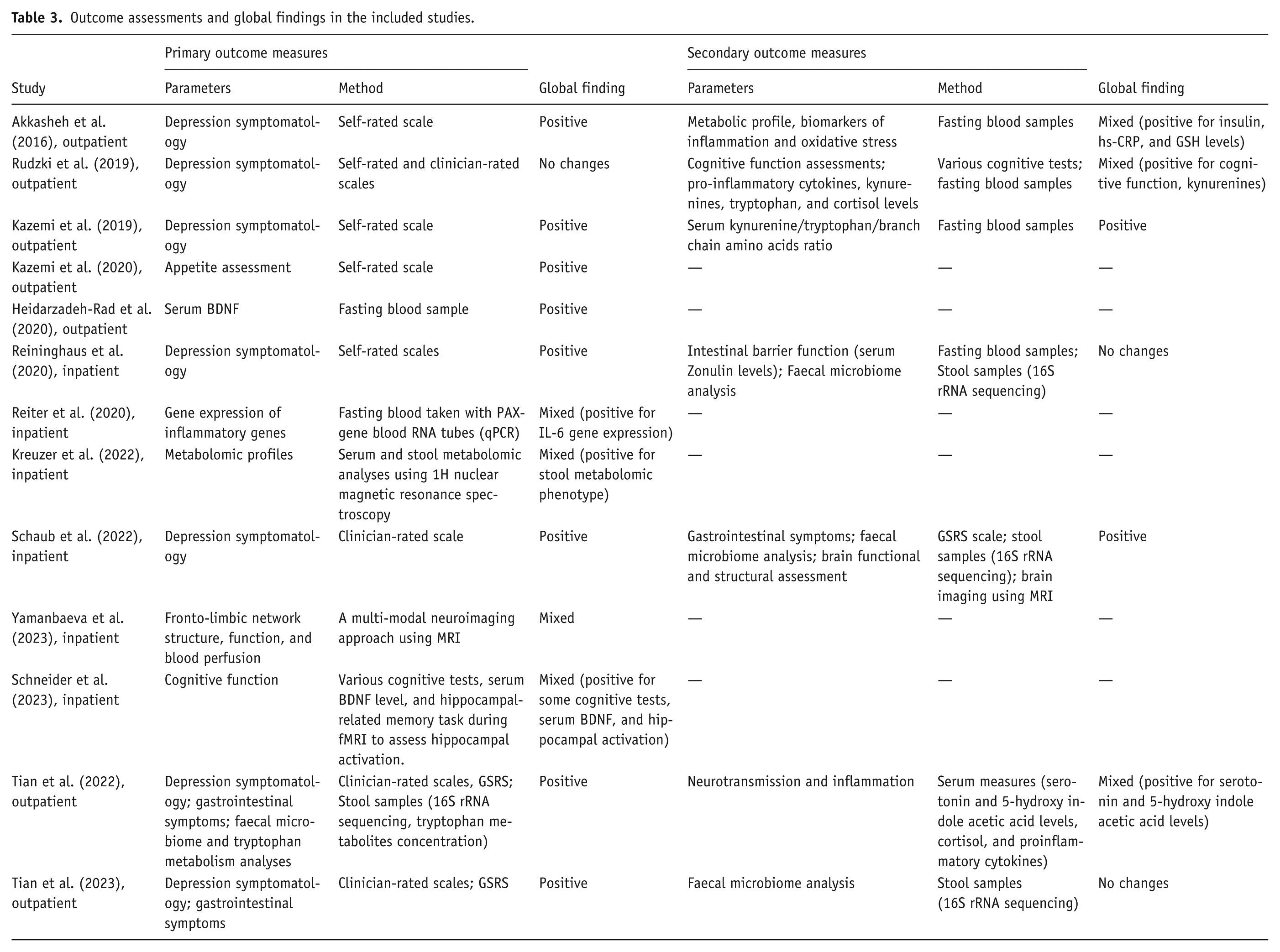
Other depression-related measures
Other depression-related measures were presented as secondary outcome measures in primary studies or the post hoc analyses, preliminary and extended studies of primary studies mostly complementing improved depression symptomatology. Akkasheh et al. studied metabolic profiles and various inflammation and oxidative stress biomarkers. Decreased serum insulin level and insulin resistance for metabolic profile, decreased high-sensitivity C-reactive protein (hs-CRP) for inflammatory markers, and increased glutathione (GSH) levels for oxidative stress were significant findings correlated to improved depression symptomatology (Akkasheh et al., 2016). Rudzki et al. (2019) showed an improved cognitive function and reduced kynurenine levels, but no significant changes were seen in the kynurenine to tryptophan ratio, levels of the proinflammatory cytokines; tumour necrosis factor-alpha (TNF-α), interleukin 6 (IL-6), interleukin 1-beta (IL-1b), and cortisol with a single-strain probiotic; Lactobacillus Plantarum 299v administration over 8 weeks. Similarly, another study that administered a single-strain probiotic, Bifidobacterium breve CCFM1025, over 4 weeks did not find significant changes in the proinflammatory cytokines; TNF-α, IL-1b, and cortisol levels, but showed significant elevation of serum serotonin (5-HT) and tryptophan metabolites (Tian et al., 2022). Kazemi et al. (2019) showed a significant reduction in kynurenine to tryptophan and tryptophan to branch chain amino acids ratios following 8-week administration of multi-strain probiotics. In the post hoc analyses of Kazemi et al., significant improvement in appetite and increased serum BDNF levels were reported in the probiotic group compared to the placebo group (Heidarzadeh-Rad et al., 2020; Kazemi et al., 2020). Another study reported mild increases in serum BDNF levels and improvements in cognitive function after 4 weeks of probiotic intervention (Schneider et al., 2023). Strains of L. helveticus were the common probiotics used in both studies (Heidarzadeh-Rad et al., 2020; Schneider et al., 2023). Reininghaus et al. (2020) measured Zonulin levels to assess the intestinal barrier function following probiotic intervention, but no significant results were obtained. In its preliminary study, an assessment of gene expression of inflammatory genes showed decreasing expression of IL-6 in the probiotic group compared to increasing levels in the placebo group within 4 weeks (Reiter et al., 2020). A significant change in stool metabolites in the probiotic group was presented in its extended study. However, no change in serum metabolites was observed (Kreuzer et al., 2022). Three studies assessed gastrointestinal symptoms using the Gastrointestinal Symptom Rating Scale and reported beneficial gastrointestinal effects of probiotics (Schaub et al., 2022). Brain imaging outcomes demonstrating functional and structural changes related to depression were shown following the short-term, high-dose probiotic intervention (Schaub et al., 2022; Schneider et al., 2023; Yamanbaeva et al., 2023). Reduced putamen activation in response to neutral face processing (Schaub et al., 2022), consistent mean diffusivity in uncinate fasciculus and altered fronto-limbic resting-state functional connectivity (Yamanbaeva et al., 2023), and improvement in hippocampus-related memory task and reduced hippocampal activation (Schneider et al., 2023) were the significant findings reported in probiotic groups. Four studies performed faecal microbiome analyses using the 16S ribosomal ribonucleic acid sequencing method (Reininghaus et al., 2020; Schaub et al., 2022; Tian et al., 2022, 2023). Two studies reported significantly increased beta diversity and specific bacterial taxa abundances but no changes in terms of alpha diversity (Reininghaus et al., 2020; Schaub et al., 2022). Another single-strain probiotic study showed no significant differences in beta diversity but increased alpha diversity and taxa-specific abundance of bacteria (Tian et al., 2022). Another study using multi-strain probiotics over 4 weeks observed increased alpha diversity in the probiotic group post- and pre-intervention, but there was no difference between the two study groups. No significant difference in beta diversity was observed. Increased taxa-specific bacterial abundance was observed in the probiotic group (Tian et al., 2023).
Potential mechanisms of probiotics for depression
Evidence from the included trials indicates that probiotics may alleviate depressive symptoms through several interconnected biological pathways. One of the findings relates to modulation of immune and oxidative processes, with reductions in inflammatory markers such as hs-CRP and favourable changes in antioxidants such as GSH (Akkasheh et al., 2016). These findings are in line with broader evidence implicating systemic inflammation and oxidative stress in the pathophysiology of MDD (Hassamal, 2023; Nady et al., 2024; Peruzzolo et al., 2022).
Another pathway under investigation is the kynurenine–tryptophan axis. Several trials reported changes in kynurenine, tryptophan, or their ratios, suggesting possible modulation of this pathway (Kazemi et al., 2019; Rudzki et al., 2019; Tian et al., 2022). In parallel, some studies observed increased serotonin and 5-hydroxyindoleacetic acid and variable cortisol changes (Rudzki et al., 2019; Tian et al., 2022). These observations suggest the possible probiotic modulation of the tryptophan–kynurenine–serotonin pathway and hypothalamic–pituitary–adrenal (HPA) neuroendocrine signalling, which have been implicated in the pathophysiology of depression (Jia et al., 2024; Johnson et al., 2023; Tiwari and Paramanik, 2025).
Neuroplasticity has also emerged as a possible target, with reported increases in serum BDNF and hippocampal activation (Heidarzadeh-Rad et al., 2020; Schneider et al., 2023). These findings are consistent with broader evidence implicating BDNF and hippocampal plasticity in antidepressant response, but direct causal links remain to be demonstrated (Cubillos et al., 2022; Li et al., 2025; Rosas-Sánchez et al., 2024).
A few trials have reported improvements in gastrointestinal (GI) symptoms and stool metabolomic profiles, often without significant microbiome compositional changes accompanying positive changes in depressive outcome (Kreuzer et al., 2022; Reininghaus et al., 2020; Schaub et al., 2022; Tian et al., 2022, 2023). This pattern suggests that functional modulation may precede detectable compositional shifts, aligning with gut–brain axis involvement and the recognised burden of GI comorbidities in MDD (Cantarero-Prieto and Moreno-Mencia, 2022; Chen et al., 2024; Huang et al., 2021; Ng et al., 2023). The lack of compositional changes may possibly reflect methodological limitations or the need for prolonged probiotic exposure (Ng et al., 2023; Nourizadeh et al., 2022).
Finally, additional outcomes have been reported, including improved insulin sensitivity (Akkasheh et al., 2016), appetite regulation (Kazemi et al., 2020), and selective cognitive improvements (Rudzki et al., 2019; Schneider et al., 2023). These findings suggest downstream effects of probiotics on metabolic, systemic, and neurocognitive functions, which have been closely linked to depression risk and progression (Berk et al., 2023; Cui et al., 2022).
Collectively, the evidence indicates that probiotics may interact with multiple interconnected domains, including immune-inflammatory and oxidative-stress pathways, metabolic regulation, the tryptophan–kynurenine–serotonin pathway, HPA neuroendocrine signalling, and central neuroplasticity, with downstream effects on appetite regulation and cognitive function, most of which converge through the gut–brain axis as the central integrative pathway to exert antidepressive potential of probiotics (Figure 2). However, these interpretations should be viewed as preliminary, since current trials do not conclusively establish causal mechanisms; rather, they highlight plausible biological pathways through which probiotics may exert antidepressant effects, and most trials were not specifically designed to establish causal mechanisms. Nevertheless, the observed patterns provide a valuable framework for hypothesis generation and highlight the importance of incorporating biomarker-integrated and stratified trial designs in future research.
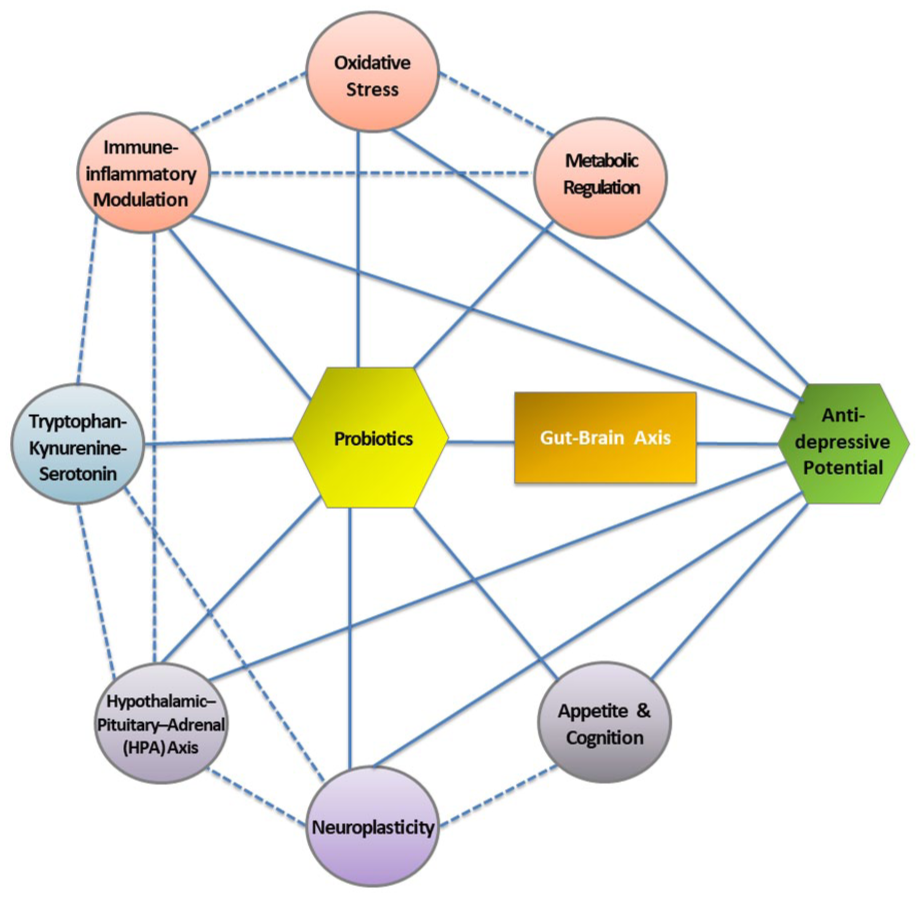
Probiotic mechanisms in MDD.
Study heterogeneity and interpretability
Across the included primary studies, adult patients with MDD were consistently recruited and diagnosed using structured clinical tools (DSM-IV, ICD-10, HDRS, BDI, or MINI), and all trials employed double-blind, placebo-controlled designs with clearly defined Lactobacillus and Bifidobacterium probiotics, often as adjunctive therapy over 4–8 weeks (Akkasheh et al., 2016; Kazemi et al., 2019; Reininghaus et al., 2020; Rudzki et al., 2019; Schaub et al., 2022; Tian et al., 2022, 2023). Depression symptomatology was uniformly assessed using validated scales, with secondary outcomes including biomarkers, gut microbiome, cognitive function, and neuroimaging measures; compliance was generally high and adverse events minimal (Akkasheh et al., 2016; Kazemi et al., 2019; Rudzki et al., 2019; Schaub et al., 2022). Heterogeneity arose in demographics, clinical presentations (baseline depression severity, treatment setting, concurrent antidepressant use), and intervention characteristics, possibly influencing study outcomes.
Demographically, age and geographic factors appeared to influence both probiotic efficacy and outcome variability. Iranian trials predominantly enrolled younger adults aged 18–55 with mild-to-moderate MDD, and consistently demonstrated positive primary depressive symptoms outcomes as well as beneficial secondary effects on metabolic parameters (insulin, hs-CRP, GSH), kynurenine-to-tryptophan ratios, and BDNF levels (Akkasheh et al., 2016; Heidarzadeh-Rad et al., 2020; Kazemi et al., 2019, 2020). By contrast, Austrian, Swiss, and Chinese studies included broader age ranges (18–75) and, despite consistently reporting improvements in primary depressive symptom outcomes, they demonstrated more variable effects on secondary domains, particularly metabolic and gut-related parameters (Kreuzer et al., 2022; Reininghaus et al., 2020; Reiter et al., 2020; Schaub et al., 2022; Schneider et al., 2023; Tian et al., 2022, 2023; Yamanbaeva et al., 2023). Notably, the Polish trial was the only study that did not observe improvements in primary depressive symptoms, though selective benefits were seen in secondary outcomes, including kynurenine metabolism and cognition (Rudzki et al., 2019). Age-related changes in gut microbial diversity, neuroplasticity, and systemic inflammation could potentially explain these differences. Additionally, regional dietary patterns and baseline microbiome composition could modulate probiotic colonisation and functionality, potentially contributing to the more consistent improvements observed in Iranian populations relative to European and East Asian cohorts.
In terms of clinical presentation, baseline depression severity, treatment setting, and concurrent AD use were also critical determinants of outcome variability. Outpatients with mild-to-moderate MDD in Iranian trials (Akkasheh et al., 2016; Heidarzadeh-Rad et al., 2020; Kazemi et al., 2019, 2020) demonstrated consistent improvements in depressive symptoms and secondary biomarkers. Conversely, Polish outpatients on SSRIs did not exhibit improvements in primary depressive symptoms and showed mixed secondary outcomes, suggesting that pharmacotherapy may modulate the observable effects of probiotics (Rudzki et al., 2019). Inpatients in Austria, Switzerland, and hospital-based outpatient cohorts in China generally included participants with more severe depression and concurrent treatments. These studies reported modest or inconsistent improvements in depressive symptoms and variable secondary outcomes, with several reporting little or no change in metabolic, inflammatory, or microbiome markers (Kreuzer et al., 2022; Reininghaus et al., 2020; Reiter et al., 2020; Schaub et al., 2022; Schneider et al., 2023; Tian et al., 2022, 2023; Yamanbaeva et al., 2023). These observations highlight that baseline severity, hospitalisation, and pharmacological heterogeneity can possibly attenuate detectable probiotic effects.
Intervention characteristics, including probiotic formulation, strain diversity, dosage, and duration, emerged as important predictors of efficacy. Multi-strain interventions, particularly those frequently containing Lactobacillus acidophilus, L. casei, L. plantarum, L. helveticus, Bifidobacterium bifidum, B. longum, B. lactis, and B. breve, generally produced broader improvements in both primary and secondary outcomes (Akkasheh et al., 2016; Kazemi et al., 2019; Schaub et al., 2022; Tian et al., 2023). Effective interventions typically delivered ⩾10⁹ CFU per strain per day, with 8-week durations in outpatient trials (Akkasheh et al., 2016; Kazemi et al., 2019) and 4-week durations in inpatient studies (Schaub et al., 2022; Tian et al., 2023). Single-strain probiotics, L. plantarum 299v and B. breve CCFM1025, exhibited selective or inconsistent effects, often limited to certain secondary biomarkers such as kynurenine or serotonin metabolites, underscoring the importance of strain diversity for systemic and neurocognitive outcomes (Rudzki et al., 2019; Tian et al., 2022).
Overall, demographic factors (age, geography), clinical presentation (baseline depression severity, treatment setting, concurrent antidepressant use), and intervention characteristics (strain diversity, dosage, duration) interact to possibly explain the observed variability in study outcomes. Probiotics showed consistent benefits in Iranian outpatients, more modest effects in European inpatients, and positive but variable secondary outcomes in East Asian outpatients. Younger, mild-to-moderate outpatient populations receiving multi-strain probiotics consistently demonstrated improvements in both primary depressive symptoms and secondary metabolic, neurotransmission, cognitive, and gut-related outcomes (Akkasheh et al., 2016; Kazemi et al., 2019; Schaub et al., 2022). By contrast, older, hospitalised, or pharmacologically heterogeneous populations exhibited more modest or selective effects (Reininghaus et al., 2020; Rudzki et al., 2019; Tian et al., 2022). These findings highlight the importance of tailoring probiotic interventions based on patient demographics, clinical context, and treatment parameters.
Implications for clinical utility
When considering probiotics for the management of MDD, clinicians should prioritise multi-strain formulations, particularly those frequently containing Lactobacillus acidophilus, L. casei, L. plantarum, L. helveticus, Bifidobacterium bifidum, B. longum, B. Lactis, and B. Breve, as these have demonstrated more consistent efficacy across diverse patient populations. Single-strain interventions may yield targeted mechanistic effects but are less consistently associated with symptomatic improvement and should be considered mainly in adjunctive or exploratory contexts (Rudzki et al., 2019; Tian et al., 2022). Evidence suggests that structured settings, such as inpatient care, where diet and daily routines are more stable, may be particularly conducive to achieving robust treatment responses (Schaub et al., 2022; Tian et al., 2023).
Probiotics should be regarded as adjunctive strategies rather than replacements for standard antidepressant therapies. Their utility lies in offering safe, tolerable, and context-dependent benefits, with the greatest improvements reported in depressive symptomatology and select secondary outcomes such as cognition, inflammatory markers, and gut-related symptoms. However, their effects are often modest, and improvements in mood may occur without parallel changes in microbiome composition, underscoring the need for realistic expectations about achievable outcomes.
Tailoring probiotic interventions to patient-specific factors remains critical as age, baseline depression severity, comorbidities, concurrent medications, and habitual dietary patterns can possibly influence treatment response. Therefore, patient-specific factors likely shape response variability and should guide clinical decision-making. Clinicians are advised to monitor treatment response using validated depression scales and consider mechanistic outcomes such as inflammation, cognition, and gut function as exploratory endpoints rather than definitive markers of efficacy.
Overall, probiotics may be considered low-risk adjunctive options in the management of MDD, with the most consistent benefits observed in younger, outpatient populations with mild-to-moderate symptoms. Their use should be individualised, integrated with established antidepressant strategies, and closely monitored, given that evidence in more severe or pharmacologically heterogeneous populations remains mixed.
Conclusions, clinical applicability, and future research recommendations
The current review supports the progressive expansion of probiotics as a potential therapeutic within the clinical management of patients with MDD. The selection of probiotics in clinical research is mainly from the pool of probiotics with generally recognised as safe status (Elshaghabee et al., 2017; Li et al., 2016), most commonly Lactobacillus and Bifidobacterium, which are human-derived commensals with favourable properties for therapeutic use (Dinan and Quigley, 2011; Nowak et al., 2019). The primary outcome in most trials was improvement in depressive symptomatology, while secondary measures examined metabolic, inflammatory, cognitive, neurotrophic, and gastrointestinal domains (Akkasheh et al., 2016; Kazemi et al., 2019; Reininghaus et al., 2020; Rudzki et al., 2019; Schaub et al., 2022; Tian et al., 2022, 2023). Although brain imaging remains underutilised, early evidence linking probiotic efficacy to neuroimaging changes is encouraging (Schaub et al., 2022; Schneider et al., 2023; Yamanbaeva et al., 2023). Other observed benefits, including improvements in metabolic profile, inflammatory markers, cognitive function, and serum BDNF, further support the therapeutic potential of probiotics in MDD and promote the customised use of probiotics (Akkasheh et al., 2016; Heidarzadeh-Rad et al., 2020; Reiter et al., 2020; Rudzki et al., 2019; Schneider et al., 2023).
Probiotics, thus, represent a promising adjunctive strategy for managing MDD, with the most consistent clinical benefits observed in younger, outpatient populations with mild-to-moderate symptoms, particularly when multi-strain formulations are used. Inpatient or more structured settings may support adherence and provide mechanistic insights, but clinical outcomes in these populations have been more variable. Evidence suggests that probiotics may improve depressive symptomatology directly while also modulating secondary outcomes such as inflammatory and metabolic markers, tryptophan–kynurenine balance, and gut-related symptoms. This positions probiotics as a safe, tolerable, and potentially effective adjunct to conventional antidepressant therapy. However, their clinical utility remains conditional and context-dependent. Effects are less robust in older, pharmacologically heterogeneous inpatient cohorts and inconsistent when relying on single-strain formulations or short treatment durations. Furthermore, improvements in depressive symptoms often occur without parallel shifts in microbiome composition, highlighting that symptomatic relief may precede measurable microbial changes. Accordingly, probiotics should be integrated as adjuncts, not stand-alone treatments, with expectations set for modest to moderate benefit rather than full remission. Clinicians should therefore adopt an individualised approach, carefully considering patient context and ongoing therapies while systematically monitoring depressive symptoms to maximise clinical benefit.
We underscore the contribution of existing interventional studies while advocating the need for continued high-quality trials to more definitively establish probiotics’ therapeutic potential in MDD. Overall, most of the included trials were well designed, with core procedures reported transparently, and several demonstrated consistent symptomatic improvements. The main concerns, reflected in the “unclear” ratings, related to attrition, selective reporting, and methodological heterogeneity. However, these limitations generally reflected insufficient methodological detail rather than fundamental flaws. Taken together, the evidence base can be considered reasonably robust, with multiple trials consistently reporting symptomatic benefits. Future research should prioritise strategies to minimise attrition, register protocols with clearly prespecified outcomes, provide comprehensive reporting including safety data, and implement intention-to-treat analyses to further strengthen validity, enhance reliability, and improve generalisability. To strengthen the field, research questions should be clearly formulated, eligibility criteria rigorously defined, and clinical experts engaged throughout the design process. Methodological approaches should be standardised to reduce translational gaps and support applicability in real-world settings. Eligibility and diagnostic criteria should be reported transparently, with structured clinical interviews and validated depression scales used to confirm diagnosis and severity. Ethical oversight is especially critical in inpatient settings, where psychiatrists should determine whether probiotics may be trialled as a stand-alone intervention. Comorbidities and concurrent medication use should also be carefully documented to improve the interpretability of outcomes. Integration of biological measures is essential to bridge preclinical and clinical evidence. Neuroimaging protocols should be precisely specified to avoid fragmented or inconsistent reporting. Public trial registration remains essential to ensure transparency and credibility. To advance mechanistic understanding, future trials consider incorporating standardised biomarker panels, functional omics, and neuroimaging endpoints, as discussed in detail in the mechanisms section of this review. These approaches will enable the field to move beyond documenting symptomatic benefit towards clarifying the biological pathways through which probiotics may exert therapeutic effects in depression. We, therefore, encourage the pursuit of additional interventional studies across diverse geographical and clinical contexts, to build on current RCTs and advance the evidence base for probiotics as a promising therapeutic adjunct in MDD.
Footnotes
ORCID iDs
Author Contributions
D.J.: Conceptualisation, Formal analysis, Methodology, Writing—Original draft preparation, Writing—Review and editing. D.S.R.: Proofreading, Writing—Review and editing. J.D.: Proofreading, Writing—Review and editing. G.C.: Conceptualisation, Writing—Review and editing. K.-G.C.: Writing—Review and editing. S.T.: Conceptualisation, Proofreading, Supervision, Writing—Review and editing. L.-H.L.: Conceptualisation, Proofreading, Supervision, Writing—Review and editing. V.L.: Conceptualisation, Funding Acquisition, Proofreading, Supervision, Writing—Review and editing. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Jeffrey Cheah School of Medicine and Health Sciences (JCSMHS) Early Career Researcher Grant 2021 ECR000021 (awarded to V.L.).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.C. has received honoraria from Janssen, Probi and Apsen as an invited speaker, is in receipt of research funding from Pharmavite, Reckitt, Tate and Lyle, and Nestle Fonterra, and has received payments as a consultant from Yakult, Zentiva and Heel Pharmaceuticals. APC Microbiome Ireland has received research support from Mead Johnson, Cremo, 4D Pharma, Suntory Wellness, and Nutricia. The authors declare no conflict of interest.
