Abstract
Previous comparative reviews of add-on therapies for patients with major depressive
disorder (MDD) with an inadequate response to antidepressants have not used meta-analytic
techniques to compare different drug classes and have included non-licensed therapies.
This meta-analysis reviewed all published peer-reviewed evidence for the efficacy of
EU-licensed therapies in patients with MDD and an inadequate response to antidepressant
monotherapy. Papers concerning randomized clinical trials (RCTs) were identified using
criteria from the Cochrane Handbook for Systematic Reviews of Interventions. Add-on
therapies reviewed were antidepressants, quetiapine XR, lithium, and
S-adenosyl-
Keywords
Introduction
The 12-month prevalence of major depressive disorder (MDD) in the European Union (EU) is estimated to be 6.9%; this equates to approximately 30 million individuals with MDD (Wittchen et al., 2011). Unsurprisingly, the highly prevalent and chronic nature of MDD creates a substantial economic and societal burden. The cost of MDD in Europe in 2004 was €118 billion, €42 billion of which was attributable to direct costs such as hospitalization (€10 billion), outpatient care (€22 billion), and drug costs (€9 billion), while €76 billion was attributable to indirect costs such as morbidity and mortality (Sobocki et al., 2006). Moreover, absence from work and lost productivity make up a substantial proportion of the overall costs associated with depression (Chisholm et al., 2003), with almost one-third of patients with severe MDD reporting unemployment or work disability (Birnbaum et al., 2010).
Patients with MDD experience reduced quality of life in terms of psychological, physical, and social functioning, and this impairment increases with greater disease severity (Daly et al., 2010). Furthermore, patients with MDD demonstrate high rates of comorbidities, which further impact on treatment costs and can compromise patients’ daily functioning (Chisholm et al., 2003). In addition, the rate of suicide is increased in patients with MDD compared with the general population (2.2% versus 0.5%, respectively) (Bostwick and Pankratz, 2000). Moreover, a recent survey of clinical practice in the UK has reported that patients may have to wait a number of years between being prescribed their first treatment (usually a monotherapy) and receiving effective treatment that controls their symptoms if a combination of medications is required (AstraZeneca data on file).
Antidepressants, such as the selective serotonin-reuptake inhibitors or serotonin norepinephrine reuptake inhibitors, are generally recommended as first-line treatments for MDD; however, approximately 50% of patients with MDD do not experience a response to first-line antidepressant therapy and the proportion of patients achieving a response decreases to approximately 30% with second-line treatment (Rush, 2007). In addition, patients who experience an inadequate response to first-line antidepressant treatment have increased disease chronicity and greater associated economic costs compared with those experiencing a response (Judd et al., 2000; Knoth et al., 2010).
A number of strategies can be utilized for patients with an inadequate response. Current EU treatment guidelines recommend that patients with MDD who experience an inadequate response to antidepressant treatment should have their dose of medication increased, be switched to an alternative antidepressant, use a combination of two antidepressants, or augment their treatment with an atypical antipsychotic or lithium (Anderson et al., 2008; Bauer et al., 2007; National Institute for Health and Clinical Excellence, 2009). In addition, these guidelines variously recommend augmentation with triiodothyronine, thyroxine, lamotrigine, tryptophan, modafinil, stimulants, estrogen (in perimenopausal women), antiglucocorticoids and nonpharmacotherapeutic strategies such as psychotherapy (Anderson et al., 2008; Bauer et al., 2007; National Institute for Health and Clinical Excellence, 2009).
Several meta-analyses have focused on single drugs/drug classes as add-on therapies for inadequate response in MDD. A meta-analysis of placebo-controlled, randomized, double-blind studies in patients with MDD and an inadequate response to first-line antidepressant therapy demonstrated that augmentation with atypical antipsychotics significantly improved response and remission rates compared with placebo augmentation (p<0.001 for both endpoints) (Nelson and Papakostas, 2009). However, rates of discontinuation were higher in patients receiving an active agent (Nelson and Papakostas, 2009). Augmentation with lithium in patients with MDD and an inadequate response was analysed in a systematic review of placebo-controlled, double-blind trials and findings suggested that lithium has a large evidence base for effective use as an augmentation therapy (Bauer et al., 2010).
A number of recent systematic reviews have gone further in their evaluation of a broad range of treatment options in patients with MDD and an inadequate response to first-line therapy (Connolly and Thase, 2011; Fleurence et al., 2009). These reviews concluded that although many second-line strategies were used widely, their use was not validated empirically, so additional evidence is required of their efficacy versus placebo or standard therapies. Importantly, many of the therapies described were not licensed for the treatment of inadequate response in MDD. A number of factors must be considered when prescribing unlicensed medications. For example, issues arising as a result of the use of unlicensed treatments are legally the responsibility of the prescriber (General Medical Council, 2008). In addition, the absence of evaluation by an independent regulatory body makes the risks and benefits of a medication being used in an unlicensed indication difficult to establish and monitor. Furthermore, agencies of medical governance generally recommend that physicians use licensed pharmacotherapies over unlicensed medications (General Medical Council, 2008). A recent online interview study of 500 patients and 250 physicians in the UK emphasized the importance of pharmaceutical licenses in physician prescribing practices and patients’ acceptance of treatment (Chisholm, 2012). Overall, 81% of patients said that they would be worried about safety if they were prescribed unlicensed medications, and 76% of physicians had concerns about associated legal risks and the lack of pharmacovigilance with unlicensed prescribing (Chisholm, 2012).
In light of the findings that both patients and physicians are most comfortable with using and prescribing licensed therapies (Chisholm, 2012), it is important to provide clear guidance in this regard for patients with MDD and an inadequate response to antidepressant monotherapy. However, to our knowledge, there are currently no published systematic reviews from a EU perspective that evaluate the efficacy of licensed therapies when used as an adjunct for patients with MDD and an inadequate response to treatment. Such an evaluation is needed to objectively and critically appraise the evidence for current EU-approved treatment options and to guide best practice. Here we report the results of a systematic review and meta-analysis that included all published peer-reviewed evidence for EU-licensed therapies when patients with MDD have had a suboptimal response to antidepressant monotherapy. This study aimed to assess whether any of the add-on therapies provided a clinical benefit over placebo and to also compare the efficacy of the add-on therapies.
Methods
Data sources and search strategy
A protocol was written for this meta-analysis prior to the initiation of the literature search which was conducted in the Cochrane Central Register of Controlled Trials (CENTRAL), the Excerpta Medica Database (EMBASE), and the Index Medicus database (MEDLINE). The search was restricted to English-language papers and abstracts, and all searching was completed in November 2011 with no restriction on date of publication.
A comprehensive search strategy was developed and adapted to comply with the
functionality of the individual databases; however, in each case the generic drug names of
selected licensed medications (mianserin, mirtazapine, phenelzine, isocarboxazid,
tranylcypromine, moclobemide, amitriptyline, clomipramine, dosulepin, doxepin, imipramine,
lofepramine, nortriptyline, trimipramine, desvenlafaxine, duloxetine, venlafaxine,
flupentixol, citalopram, escitalopram, fluoxetine, paroxetine, sertraline, trazodone,
agomelatine, reboxetine, quetiapine and lithium) and MDD were included as search terms. In
order to retrieve only papers reporting data from randomized controlled trials (RCTs), the
highly sensitive search strategies for identifying reports of RCTs described in the
Cochrane Handbook for Systematic Reviews of Interventions were used (Cochrane Collaboration, 2011). Following initial
review, the authors noted an absence of information on the licensed drug
S-adenosyl-
Inclusion criteria
Titles and/or abstracts of articles identified during our preliminary searches were screened to see if they met a set of predetermined inclusion criteria, followed by a second review stage of the full-text publications. The selection criteria for inclusion of trials in this review were: study design was a double-blind, placebo-controlled RCT; trial intervention was any drug in use as an adjunct that was licensed in the EU as an antidepressant or as an adjunct to an antidepressant in adult patients with MDD who had an inadequate response to antidepressant monotherapy. For the purpose of this review, EU-licensed drugs were defined as medications licensed in one or more countries listed as part of the European license process (European Medicines Agency, 2012). Only trials where patients were treated with the same antidepressant before and after the addition of adjunct therapy were included. Given that there are very few data in the published literature regarding augmentation, it was expected that a number of trials for licensed drugs would be against unlicensed comparators. In order to encompass a sufficient data set in this analysis, and to ensure the results achieved some external validity without compromising internal validity, trials were included where up to 20% of patients on comparator medications were treated off-label. No attempts were made to contact the authors of the identified studies for patient level/summary level data.
Assessment of trial quality
The quality of trials identified in the database searches was assessed according to their methods of treatment randomization and drug-allocation blinding. These aspects of study design are reportedly most likely to introduce bias into a RCT (Schulz et al., 1995). Two reviewers independently assessed trial quality using the risk of bias tool developed by the Cochrane Collaboration (Cochrane Collaboration, 2011: Chapter 8) and any differences in opinion were adjudicated by a third party.
Data extraction
Data regarding response and remission were extracted from the identified studies as these were the only outcomes measured consistently across all studies (using either the Hamilton Depression Rating Scale (HAM-D) or the Montgomery Åsberg Depression Rating Scale (MADRS)). Change from baseline was not reported consistently across all trials. Again, two reviewers independently extracted these data whilst a third party adjudicated their decisions. For each selected trial, the intention-to-treat (ITT) population was included in the overall analysis. For the purposes of this review, the ITT population was defined as patients who were enrolled and randomized to treatment, regardless of whether they discontinued from the study or received incorrect treatment (i.e. not in accordance with their randomized drug allocation). If data were presented in an alternative format in these publications, they were recalculated for the ITT population where possible. Patients were reinstated into the ITT population based on a worst-case assumption if they had received at least one dose of study medication.
No attempts were made to standardize the definition used for the outcomes considered (response and remission) as data were limited in the studies identified in this systematic review.
Data analysis
Where the data were deemed sufficient, traditional pair-wise Mantel–Haenszel random effects meta-analyses were performed for placebo versus each category of add-on agent. The software used was Review Manager (RevMan) Version 5.0. (Copenhagen: The Nordic Cochrane Centre, the Cochrane Collaboration, 2008).
Adjusted indirect comparisons were performed to yield estimates of the relative differences in the incidences of response and remission, using placebo as a data bridge. The Bucher method, a commonly used and well-validated technique, was used to perform adjusted indirect comparisons with a third therapy as a common comparator (Bucher et al., 1997; Edwards et al., 2009; Song et al., 2003).
Comprehensive Meta-Analysis Version 2 (Biostat Inc., Englewood, NJ, USA) was used to conduct the adjusted indirect comparison for each outcome of interest. The Bucher method was employed to convert the summary estimates (log odds ratios) and measures of uncertainty (variances) from the meta-analyses into an odds ratio and confidence interval representing the difference between the treatments of interest (Bucher et al., 1997).
Assessment of heterogeneity and small-study effects
I2 statistics were used to assess heterogeneity within each pair-wise comparison. Values of 25%, 50%, and 75% were used for low, moderate, and high levels of heterogeneity, respectively, as recommended by the Cochrane Collaboration (2011).
It was planned that funnel plots would be used to check for small-study effects for outcomes where a minimum of 10 data points were available, as this was considered sufficient data to allow meaningful statistical calculations (Cochrane Collaboration, 2011). Small-study effects and funnel plot asymmetry would highlight possible publication bias in the studies identified.
Sensitivity analyses and subgroup analyses
Although it is relatively common practice for a fixed-effect model to be run as a sensitivity analysis if a random-effect model was used for the primary analysis, or vice versa, the Bucher method was validated for the random-effect model only (Song et al., 2003), therefore no such sensitivity analyses were conducted for this review.
Sensitivity analyses were conducted to ensure the robustness of the study’s findings where heterogeneity was present or suspected, or where there was doubt as to the effect of specific studies on the overall results of the meta-analysis (for example, in the case of studies with very small patient populations).
The main analysis used the primary outcome measures as reported in the trials. The only trials to use MADRS as a primary outcome measure were the quetiapine-related ones, with the others using HAM-D. MADRS and HAM-D have been shown to correlate well (Cusin et al., 2010). We deemed it consistent to use the primary outcome measure as reported given that the MADRS is more sensitive to change in symptoms as a result of treatment and this approach has been used in another meta-analysis in this area (Spielmans et al., 2013). However, HAM-D was also measured in the quetiapine trials so we obtained response and remission data using this outcome measure and reran the analyses to assess whether the results significantly changed.
Results
Trial flow
The literature searches initially identified 8984 papers (6965 papers after the removal of duplicates) that were potentially relevant to this systematic review (Figure 1). Of these papers, only 12 were found to report RCTs of EU-licensed add-on treatments for patients with MDD with an inadequate response to their index antidepressant treatment; seven of these trials reported response and remission in such a way that allowed quantitative analysis. Medications in the identified studies were approved for the treatment of depression (including MDD, recurrent depression, or depressive syndromes) in the UK (and other EU countries: lithium, mirtazapine, and quetiapine XR), Ireland (and other EU countries: mianserin), and Italy (SAMe (licensed as Samyr® for depressive syndromes).
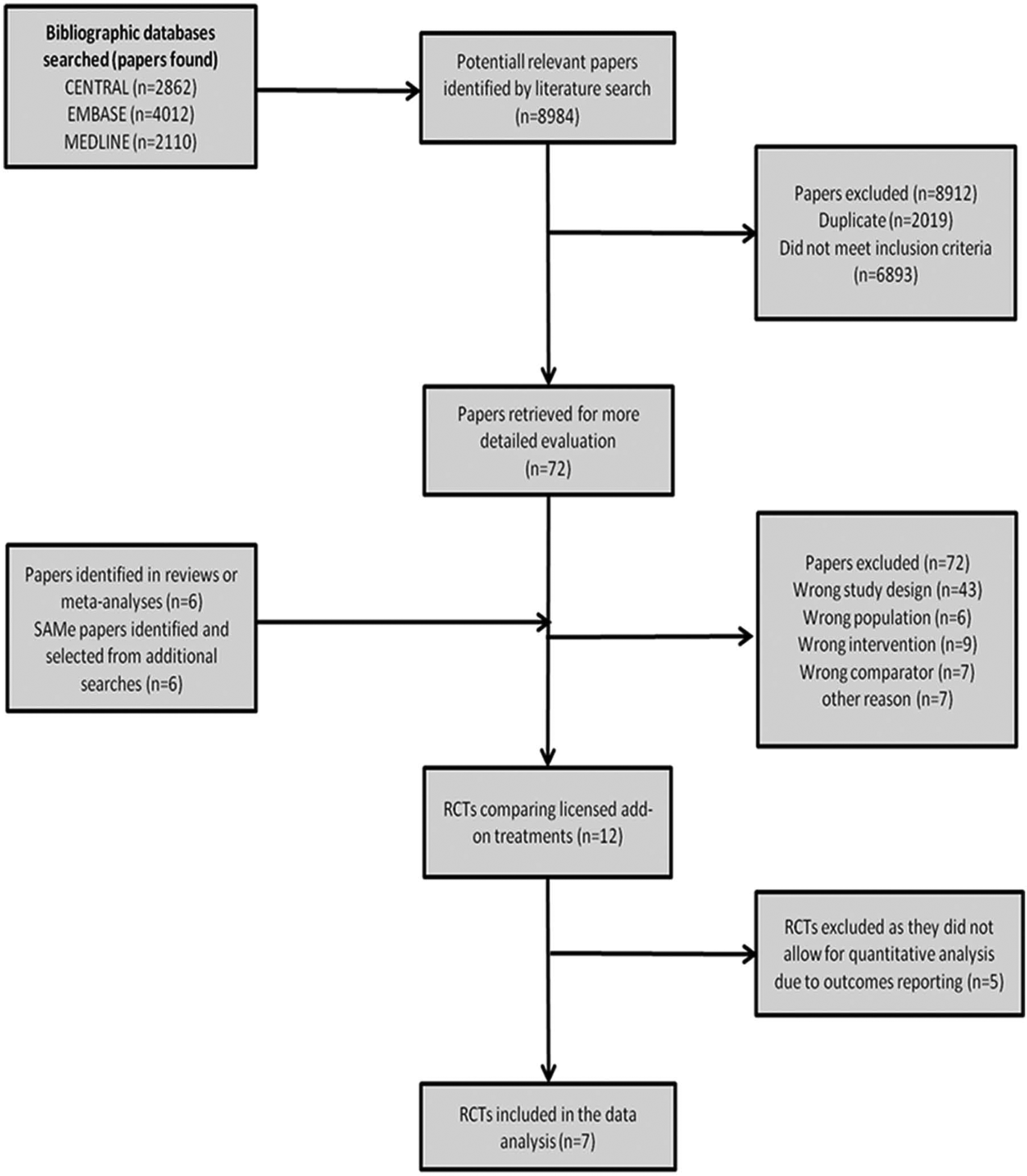
Flow of papers through the systematic searches.
Trial characteristics
Two trials compared add-on lithium with placebo, one trial compared add-on mianserin with placebo, one trial compared add-on mirtazapine with placebo, two trials compared add-on extended release (XR) quetiapine fumarate with placebo, and one trial compared add-on SAMe with placebo (Bauer et al., 2009; Carpenter et al., 2002; El-Khalili et al., 2010; Licht and Qvitzau, 2002; Nierenberg et al., 2003; Papakostas et al., 2010; Stein and Bernadt, 1993). Table 1 summarizes the main characteristics of these trials, including the antidepressants that were augmented in each study and the baseline disease severity and age of the patients, where available.
Study designs of trials included in the meta-analysis.

CGI: Clinical Global Impression; DSM-III-R: Diagnostic and Statistical Manual of Mental Disorders, Third Edition, Revised; DSM-IV-TR: Diagnostic and Statistical Manual of Mental Disorders Fourth Edition Text Revision; HAM-D: Hamilton Depression Rating Scale; HAM-D-17: 17-item Hamilton Depression Rating Scale; HATH: Harvard Antidepressant History Form; MADRS: Montgomery–Å sberg Depression Rating Scale; MAOI: monoamine oxidase inhibitor; MDD: major depressive disorder; MDE: major depressive episode; RDC: Research Diagnostic Criteria; SAMe: S-adenosyl methionine; SCID-P: Structured Clinical Interview for DSM-IV Axis I Disorders Patient Edition; SNRI: serotonin norepinephrine reuptake inhibitor; SSRI: selective serotonin-reuptake inhibitor; TCA: tricyclic antidepressant; tid: three times daily; XR: extended release.
The duration of add-on treatment with lithium ranged from 3–6 weeks (Nierenberg et al., 2003; Stein and Bernadt, 1993). In general, the lithium studies were older than those for other agents, had small patient populations, and did not assess remission; therefore there was more uncertainty in terms of the information that these studies reported than with the other medications assessed. Our quality assessment process using a risk of bias tool developed by the Cochrane Collaboration (2011) (data not shown) revealed that only one out of the two lithium trials was well conducted (Nierenberg et al., 2003). However, this trial included a very small number of patients (n=35). Stein and Bernadt (1993) carried out a 9-week trial of lithium as an add-on treatment. For the purpose of this review, only data from Weeks 1–3 were included as this encompassed the double-blind phase of the study; in subsequent weeks, each patient received open-label lithium treatment.
Only two trials that combined antidepressants were included in this systematic review; one trial each of mianserin and mirtazapine in combination with the index antidepressant (Carpenter et al., 2002; Licht and Qvitzau, 2002). The mirtazapine trial (Carpenter et al., 2002) had fewer than 20 patients in each treatment arm.
Quetiapine XR is currently the only licensed atypical antipsychotic in the EU for add-on treatment in patients with MDD and an inadequate response to their index antidepressant. Two trials of quetiapine XR met the inclusion criteria for this systematic review (Bauer et al., 2009; El-Khalili et al., 2010); both studies were judged to be well conducted with large patient populations (~500 patients in each study).
One trial included in this systematic review assessed add-on therapy with SAMe (Papakostas et al., 2010).
All of the included trials used similar definitions of response and remission (Table 1). One should note though that not all definitions of inadequate response to an initial antidepressant were the same.
Comparison of response and remission rates with add-on therapies versus placebo
Data from the trials included in this systematic review were used to run nine pair-wise meta-analyses (Figures 2 and 3). Across the studies, patients treated with quetiapine XR 150 mg (p=0.06), quetiapine XR 300 mg (p=0.003), and SAMe (p=0.02) as add-on therapies were significantly more likely to achieve response than patients administered placebo. Antidepressants (p=0.47) and lithium (p=0.37) as add-on therapies did not separate from placebo in terms of response (Figure 2).
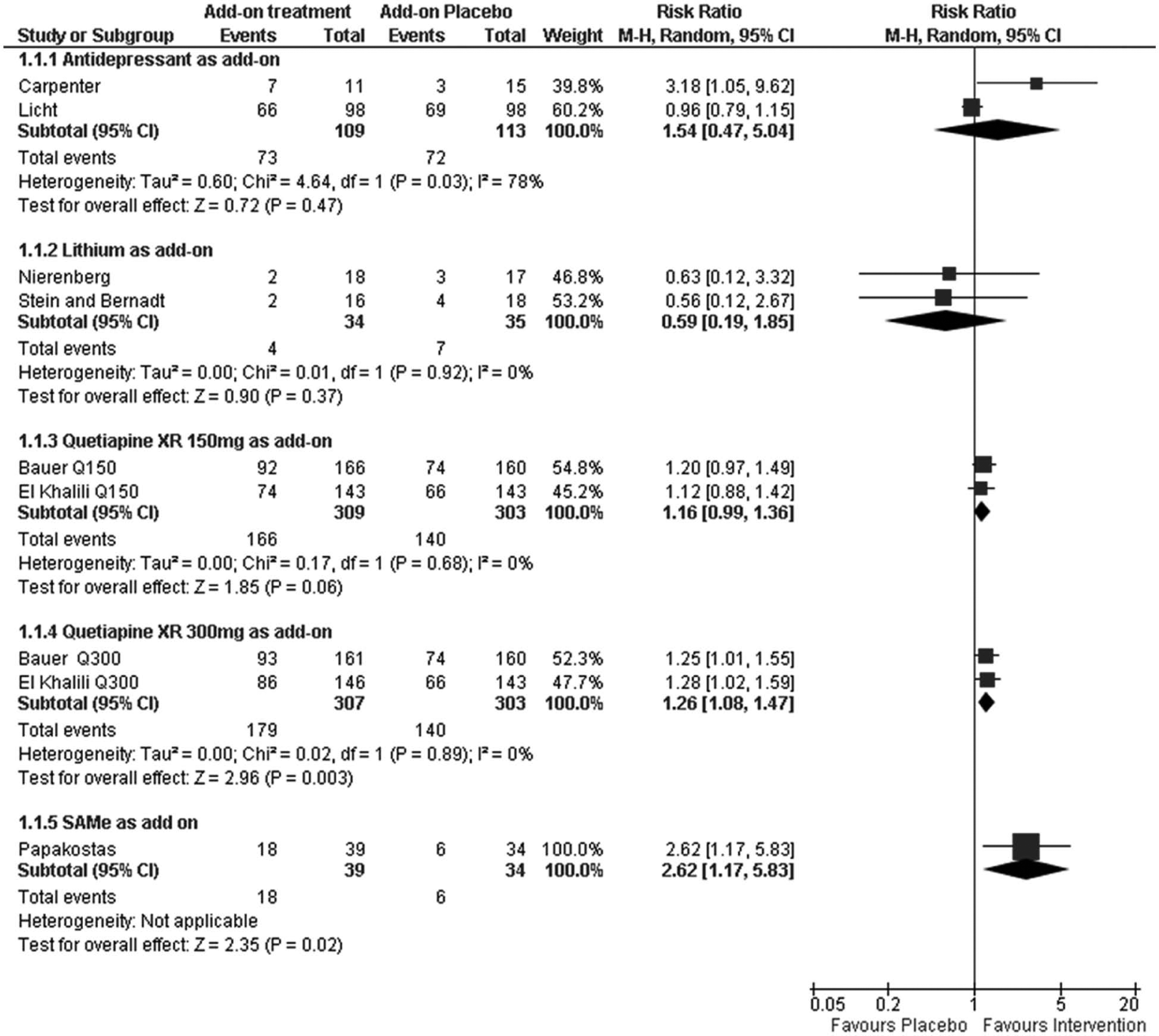
Forest plots for response for the comparisons of add-on antidepressants, add-on lithium, add-on quetiapine XR and add-on SAMe versus placebo.
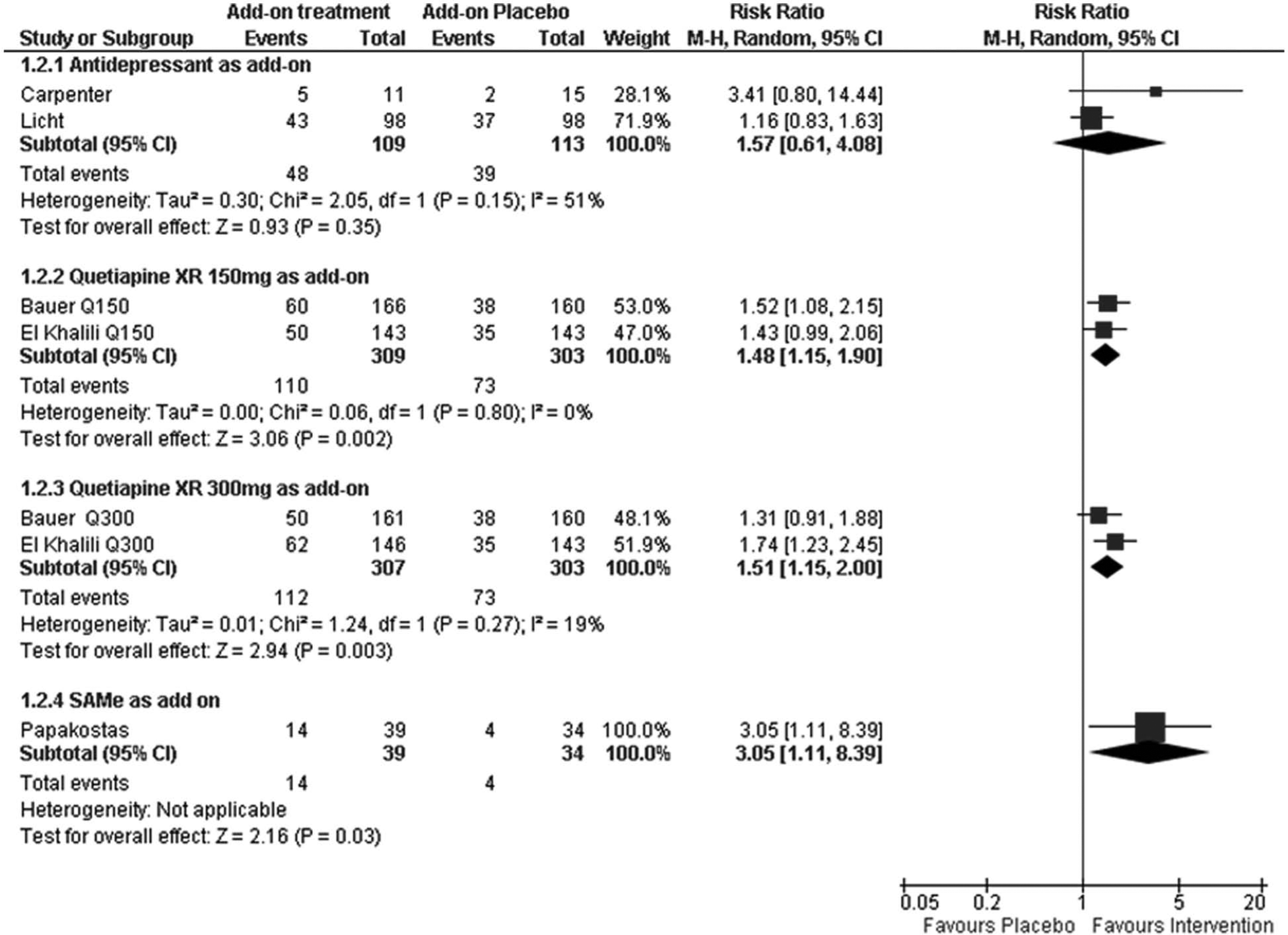
Forest plots for remission for the comparisons of add-on antidepressants, add-on quetiapine XR and add-on SAMe with placebo.
Comparison of the add-on therapies with placebo in terms of rates of remission yielded similar results to those for response. Patients treated with either dose of quetiapine XR (150 mg, p=0.002; 300 mg, p=0.003) or SAMe (p=0.03) were significantly more likely to achieve remission than patients treated with placebo; antidepressant as add-on therapy did not separate from placebo (p=0.35) (Figure 3). Remission was not an endpoint in any of the lithium studies included in this meta-analysis.
Adjusted indirect comparison results
The results of the adjusted indirect comparisons between classes of add-on intervention (i.e. antidepressant, lithium, atypical antipsychotic, and SAMe) are shown in Table 2. Comparison of the different classes of licensed add-on therapies indicated that there was an equal likelihood of achieving response or remission with add-on quetiapine XR (150 mg or 300 mg) versus an antidepressant, lithium, or SAMe, or with an add-on antidepressant versus lithium or SAMe. However, when compared with add-on lithium, patients treated with add-on SAMe were significantly more likely to achieve response (as indicated by a confidence interval entirely below 1).
Results of the indirect comparisons between classes of add-on intervention using the Bucher method.
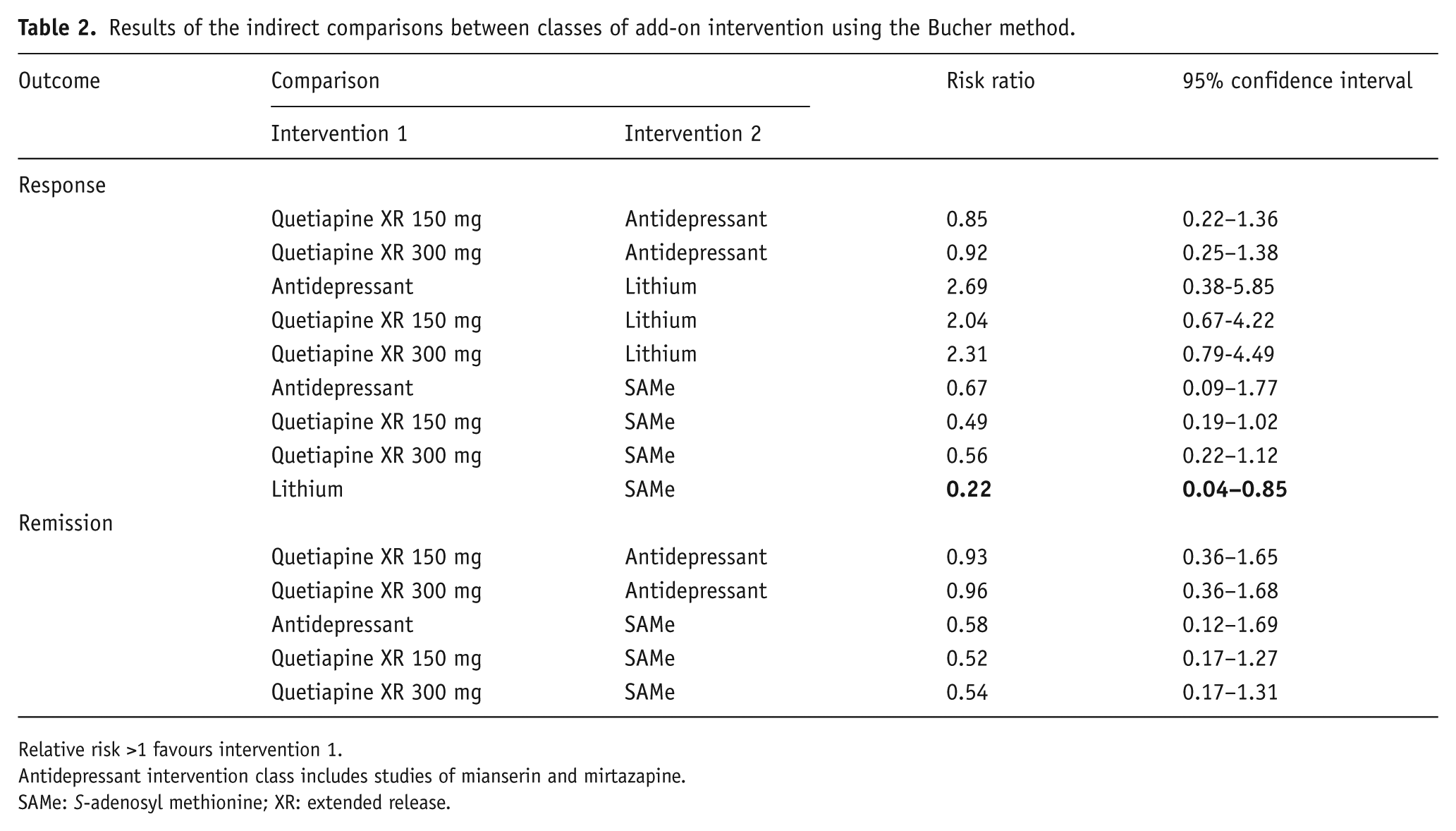
Relative risk >1 favours intervention 1.
Antidepressant intervention class includes studies of mianserin and mirtazapine.
SAMe: S-adenosyl methionine; XR: extended release.
Sensitivity analyses
Running an analysis using HAM-D scores only produced largely similar results, the only noticeable differences being that the response risk ratios for quetiapine XR compared with SAMe changed from 0.49 (95%CI 0.19–1.02) to 0.44 (95%CI 0.17–0.94) and from 0.56 (95%CI 0.22–1.12) to 0.46 (95%CI 0.18–0.97) for quetiapine XR 150 mg and quetiapine XR 300 mg, respectively. In addition, the remission risk ratio for quetiapine XR 150 mg versus mianserin changed from 1.39 (95%CI 1.00–1.73) to 1.23 (95%CI 0.87–1.58). The slight changes may be as a result of the HAM-D scale weighting domains that are less sensitive to changes with treatment, but overall do not change the findings of this analysis and show that SAMe appears to offer greater efficacy than the other compounds.
Assessment of heterogeneity
Assessment of heterogeneity of the different add-on therapy classes showed an I2 of 79% for the add-on antidepressant group. This is a high level of heterogeneity, thereby throwing doubt on the validity and appropriateness of pooling results from the antidepressant (mirtazapine and mianserin) trials. The I2 value was 0% for the meta-analyses of the other medication types. As a consequence of the high level of heterogeneity in the add-on antidepressant group, the indirect comparison analysis was repeated without pooling the results of the antidepressant trials (Table 3).
Indirect comparisons of individual treatments using the Bucher method.
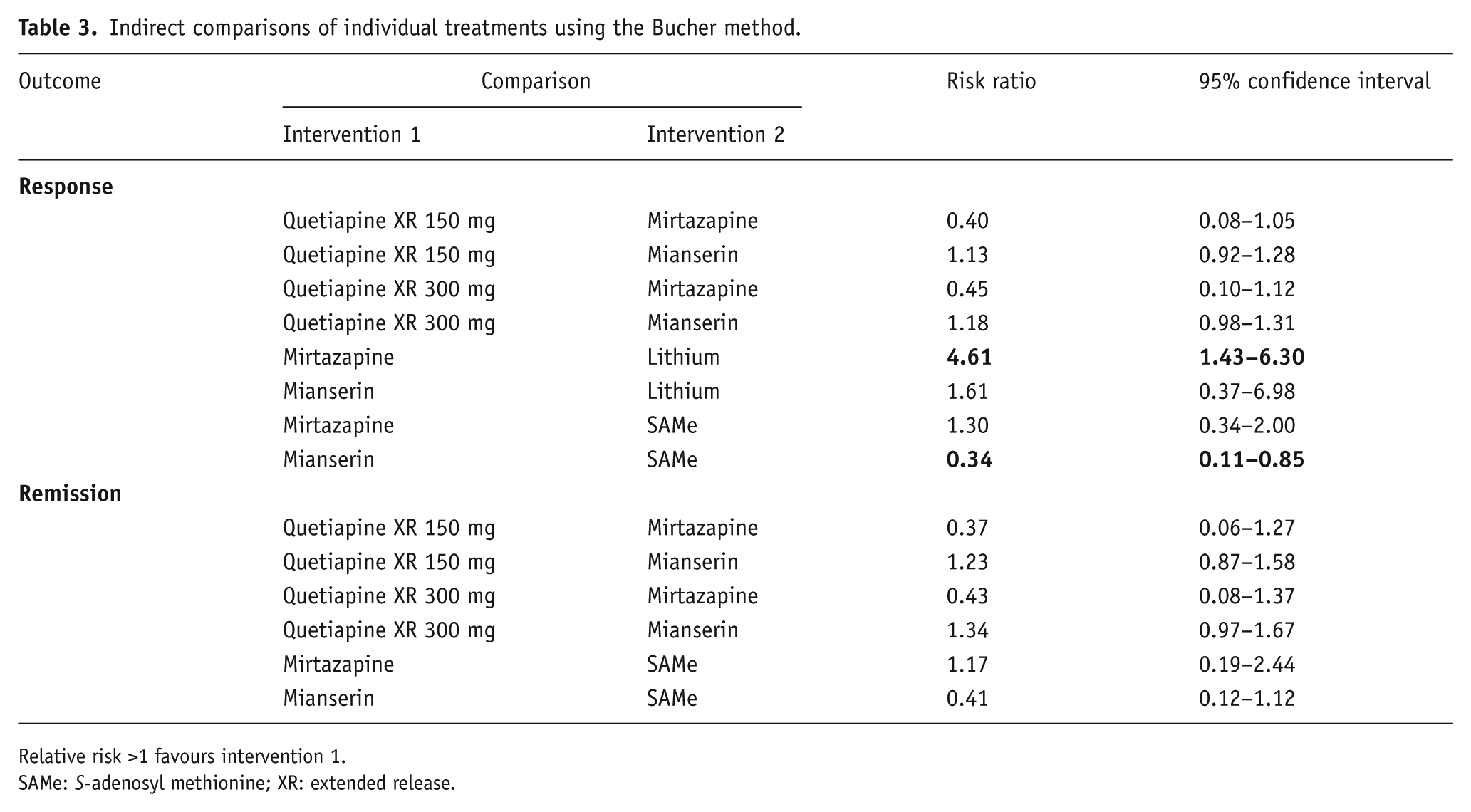
Relative risk >1 favours intervention 1.
SAMe: S-adenosyl methionine; XR: extended release.
Compared with patients treated with lithium, there was a significantly greater likelihood of patients treated with mirtazapine achieving response. However, this result was based on a single study of each add-on agent and confidence intervals were broad, and hence this finding should be viewed with caution. Patients treated with SAMe were significantly more likely to achieve a response than patients treated with mianserin (again this finding was based on a single study).
Assessment of small-study effects
None of the types of add-on treatment (lithium, atypical antipsychotic, antidepressant, or SAMe) were assessed in more than three trials in this systematic review. The prespecified cut-off for generating funnel plots was 10 trials for each adjunct treatment type and therefore no testing for small-study effects was possible.
Discussion
Despite the high rates of patients with MDD who experience an inadequate response to the antidepressant monotherapy, the quantity of good-quality RCT evidence in this regard is low. Only seven papers out of almost 9000 articles initially identified were deemed to be of sufficient quality (and had comparable definitions of response and remission) to be included in this systematic review.
The results of this systematic review showed that, in the main, all classes of add-on interventions were similar in terms of rates of response and remission. The only statistically significant difference in efficacy was between SAMe and lithium, with SAMe demonstrating higher rates of response; remission was not measured in the studies of lithium included in this analysis (Papakostas et al., 2010). However, the clinical relevance of this finding is not clear and it should be borne in mind that SAMe is used as a nutritional supplement in a number of countries and is a licensed drug in Italy only. A question that warrants further study is whether SAMe should be used more often in the treatment of patients with MDD. Although statistical comparison of the sole trial of SAMe versus the lithium studies indicates that SAMe has clinical benefits, further evidence is required to understand its place in therapy compared with other licensed medications. Furthermore, as only one of the identified lithium studies was judged to be well conducted and, in general, the identified lithium studies were old and had small patient populations, the robustness of the comparison between SAMe and lithium requires further corroboration.
Of note is the efficacy of quetiapine XR compared with placebo in patients with MDD and an inadequate response to antidepressant treatment. Quetiapine XR is approved in a number of EU countries for the treatment of amongst other things, an adjunct therapy for patients with MDD and an inadequate response to antidepressant treatment. The results of the high-quality studies of quetiapine XR reported herein (Bauer et al., 2009; El-Khalili et al., 2010) demonstrate its usefulness as a second-line agent for the treatment of MDD alongside more traditional agents, such as antidepressants. Unlike the antidepressants analysed herein, quetiapine XR separated from placebo in terms of both response and remission.
This meta-analysis highlights the lack of high-quality RCT evidence for the common practice of combining antidepressant agents, a finding reported previously in an earlier systematic review (Connolly and Thase, 2011). Although we tried to pool the results of the two included studies of mirtazapine and mianserin (both noradrenergic and specific serotonergic antidepressants), statistical analysis showed them to be too heterogeneous for this approach. Mirtazapine, in particular, is commonly used in combination with other antidepressants (Alvarez and Vinas, 2010); however, it appears that alternative classes of medication may be just as effective. Given that relatively few patients respond to their first-line antidepressant therapy (Rush, 2007) and that patients being treated with mirtazapine (and indeed any other agent) may experience unwanted side effects (Masand and Gupta, 2002), options from other drug classes are especially welcome. Budgetary pressures imposed on commissioners and payers pose a challenge to prescribing higher-cost drugs instead of generic mirtazapine. However, given that hospitalization for MDD is extremely expensive, choosing a higher-cost treatment alternative with greater efficacy and a more benign safety profile may offset the cost of a potential stay in hospital. Health economics studies comparing combination treatment with mirtazapine and treatment with other add-on therapies would be instructive.
Two large-scale initiatives have assessed the efficacy of different antidepressant treatment strategies in patients with MDD. The Sequenced Treatment Alternatives to Relieve Depression (STAR*D) study was initiated to assess the effectiveness of a variety of treatment steps after inadequate response in patients with MDD. The results of STAR*D have highlighted the low proportion of patients who respond to first-line treatment (and later treatment strategies) and have been used to improve treatment algorithms in patients with MDD (Rush, 2007). The Combining Medication to Enhance Depression Outcomes (CO-MED) study was designed to compare the effects of treatment with a combination of antidepressants against treatment with a single antidepressant agent. To date, results from CO-MED have indicated that combining traditional antidepressants does not result in improved outcomes compared with antidepressant monotherapy (Morris et al., 2012; Rush et al., 2011).
Previous articles reporting on this topic have tended to focus on summarizing the results of placebo-controlled trials of single drugs used in the adjunct setting in patients with MDD and an inadequate response to antidepressants (Bauer et al., 2010). Papers that have discussed multiple medications in this indication have used descriptive statistics rather than meta-analyses to report on the differences in efficacy between drugs in the same or different classes, have included drugs that are not licensed for depression, or have conducted meta-analyses of studies concerned with a single drug class only (Connolly and Thase, 2011; Fleurence et al., 2009; Nelson and Papakostas, 2009). Thus the findings of the current meta-analysis, which compares the results of high-quality RCTs of multiple EU-licensed therapies, provide a useful addition to the evidence base in terms of the relative efficacy of each of these medications in patients with MDD with an inadequate response to antidepressants. Guidance regarding the most efficacious use of licensed therapies is important in light of a recent study which has indicated that both physicians and patients are most comfortable prescribing and receiving treatment with medications that have been approved in the apposite setting (Chisholm, 2012).
Strengths and limitations
The current study employed a robust design and stringent inclusion and exclusion criteria. Furthermore, sensitivity analyses were employed to confirm the study’s findings. For the main analysis we used reported primary outcomes from trials to obtain response and remission data, and this meant including response and remission data based on two outcome measures (HAM-D and MADRS) in the primary analysis. We were, however, able to obtain response and remission data based on HAM-D for the two trials that reported their primary outcome with MADRS, and reran the analyses. Although this meant that response and remission were consistently reported using the same index it also meant that the response and remission analysis used data from both primary and secondary outcomes (as HAM-D was recorded as secondary outcome in the two quetiapine XR trials). Neither method is ideal, but running the sensitivity analysis increases confidence in the results.
Limitations of this meta-analysis include a low overall number of selected studies, and those trials that were included had small patient populations (n values ranged from 26–73) (apart from the two quetiapine XR studies and the mianserin study). Furthermore, there was a high degree of heterogeneity in the studies of antidepressant combinations, which led to uncertainty concerning the robustness of the pooled results. Moreover, the studies included in this meta-analysis also varied widely in terms of their duration, had differing response and remission criteria, and also had different levels of MDD severity at baseline. Another limitation of note with all trials of the nature included in this meta-analysis relates to the rare likelihood that some of the patients included in trials where resistant depression is suspected could in fact be undiagnosed bipolar disorder patients. With this risk present, interpreting results involving trials that include lithium becomes more difficult; however, due to effective standardized screening, we deemed this risk to be low. Given that this review included only licensed therapies, therapies such as T3 or aripiprazole were excluded even though an evidence base does exist for these agents.
Conclusions
Current data indicate that the add-on therapies analysed in this study have equivalent efficacy for the treatment of patients with MDD and an inadequate response to their index antidepressant. There is a paucity of high-quality evidence in this area, with the most comprehensive evidence base belonging to quetiapine XR. As a result, quetiapine XR is currently the only atypical antipsychotic in the EU licensed as an add-on therapy in patients with an inadequate response to their initial antidepressant. Overall the results of the indirect comparisons showed that all therapies had comparable efficacy. There was one exception to this finding; the comparison of one study of SAMe and two studies of lithium indicated that patients treated with an antidepressant plus SAMe were significantly more likely to achieve a response than patients treated with an antidepressant plus lithium. These findings are supported by only a small number of high-quality clinical studies. There is clearly a requirement for further high-quality research regarding the use of add-on treatment in patients with MDD and an inadequate response to antidepressant therapy.
Footnotes
Acknowledgements
The authors thank Jane Bryant, PhD, from Complete Medical Communications (Macclesfield, UK), who provided medical writing support funded by AstraZeneca UKMC.
Conflict of interest
Pauline Turner and Rakesh Kantaria are both employees of AstraZeneca. Allan Young has: served on advisory boards for AstraZeneca, BCI, Bristol-Myers Squibb, Eli Lilly, Lundbeck, Pfizer, and sanofi-aventis; received grant support from AstraZeneca, BCI, Eli Lilly, GlaxoSmithKline, Novartis, sanofi-aventis, and Wyeth; and has received honoraria for giving lectures from AstraZeneca, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen, Lundbeck, Novartis, Otsuka Pharmaceutical Co., Pharmaceutica, Pfizer, sanofi-aventis, Servier Laboratories, and Wyeth.
Funding
Sources of financial and material support this systematic review was funded by AstraZeneca UKMC, Luton, UK.
