Abstract
Background:
Preclinical animal and preliminary human studies indicate that 5-HT7 antagonists have the potential as a new treatment approach for mood and anxiety disorders. In this systematic review, we aimed to review the relationship between the 5-HT7 receptor system and mood and anxiety disorders, and to explore the pharmacology and therapeutic potential of medications that target the 5-HT7 receptor for their treatment.
Methods:
Medline, Cochrane Library, EMBASE, PsycINFO databases, the National Institute of Health website Clinicaltrials.gov, controlled-trials.com, and relevant grey literature were used to search for original research articles, and reference lists of included articles were then hand searched.
Results:
Sixty-four studies were included in the review: 52 animal studies and 12 human studies. Studies used a variety of preclinical paradigms and questionnaires to assess change in mood, and few studies examined sleep or cognition. Forty-four out of 47 (44/47) preclinical 5-HT7 modulation studies identified potential antidepressant effects and 20/23 studies identified potential anxiolytic effects. In clinical studies, 5/7 identified potential antidepressant effects in major depressive disorder, 1/2 identified potential anxiolytic effects in generalized anxiety disorder, and 3/3 identified potential antidepressant effects in bipolar disorders.
Conclusion:
While there is some evidence that the 5-HT7 receptor system may be a potential target for treating mood and anxiety disorders, many agents included in the review also bind to other receptors. Further research is needed using drugs that bind specifically to 5-HT7 receptors to examine treatment proof of concept further.
Introduction
The 5-hydroxtryptamine (5-HT or serotonin) system has been widely implicated in the pathophysiology of mood and anxiety disorders, with a main treatment focus on the serotonin transporter (SERT), 5-HT1A, 5-HT1B, and 5-HT2A receptor subtypes (Fakhoury, 2016; Thase and Denko, 2008). More recently, this has extended to the 5-HT7 receptor, which was first identified in 1993 (Bard et al., 1993; Lovenberg et al., 1993; Ruat et al., 1993). The 5-HT7 receptor is widely distributed across the brain, including the cortex, hippocampus, thalamus, and hypothalamus (Horisawa et al., 2013; Martin-Cora and Pazos, 2004; Thomas et al., 2003; To, 1995) and has been implicated in a variety of brain functions such as mood, sleep, learning and memory, stress, seizures, and circadian rhythm regulation. Moreover, the 5-HT7 receptor may have a significant role in mediating cognition, especially in people with mood disorders (Gasbarri and Pompili, 2014). Given these associations, the 5-HT7 receptor may be a promising treatment target for mood and anxiety disorders (Canese et al., 2015; Cates et al., 2013; Hedlund and Sutcliffe, 2007).
Many second-generation antipsychotic drugs currently available show high affinity to the 5-HT7 receptor and two medications with 5-HT7 antagonist properties, vortioxetine and lurasidone, have been found to enhance cognitive functioning in people with major depressive disorder (MDD) and schizophrenia (Harvey, 2015; McIntyre et al., 2013). Furthermore, preclinical animal studies have indicated the potential use of 5-HT7 antagonists for the treatment of anxiety disorders (Hedlund and Sutcliffe, 2007).
In this paper, we aimed to systematically review the relationship between 5-HT7 receptors and mood and anxiety disorders, and to further explore the pharmacology and therapeutic potential of medications that target the 5-HT7 receptor for the treatment of mood and anxiety disorders.
Methods
The systematic review study protocol was registered on the International Prospective Register for Systematic Review (PROSPERO) database (registration number CRD42019138174). All study procedures are documented and were reported following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting guidelines (Page et al., 2021).
Search methods
The systematic search was conducted using Medline, Cochrane Library, EMBASE, PsycINFO, the National Institute of Health website Clinicaltrials.gov, controlled-trials.com, and relevant grey literature) from 1993 inception to March 2021. The following search string was used:
[(5-HT7 OR serotonin receptor 7 OR 5-hydroxytryptamine 7) AND (depress* OR bipolar disorder OR anxiety disorder)] OR [(5-HT7 OR serotonin receptor 7 OR 5-hydroxytryptamine 7) AND (animals OR humans OR preclinical study OR clinical trial OR experimental medicine)] OR [(5-HT7 OR serotonin receptor 7 OR 5-hydroxytryptamine 7) AND (lurasidone OR vortioxetine)] OR (5-HT7 antagonists OR 5-HT7 agonists)
Reference lists of included articles were further searched for eligible studies. If papers were not written in English, we attempted to obtain a translated version.
Systematic searches of the preselected databases were carried out independently by two researchers (TYL and NG), using the predetermined search string. Results were compiled using Rayyan QCRI software (Ouzzani et al., 2016), and titles and abstracts were independently screened by both researchers. Any studies that appeared eligible, or if there was any uncertainty about eligibility, underwent a full-text review. Final inclusion lists were compared, and any disagreements were discussed until a consensus was reached. An additional (PRS) reviewer was consulted as needed.
Study selection
Study and participant type
Only full-text original research articles using an appropriate control group (sham or placebo) were included. Animal studies that used a mood or anxiety disorder model and a relevant genetic or pharmacological manipulation to the 5-HT7 system were included.
For human experimental medicine studies or clinical trials, randomized control trials (RCTs) using males and females over the age of 18 fulfilling International Classification of Diseases (ICD) or Diagnostic and Statistical Manual of Mental Disorders (DSM) criteria diagnosis for an MDD, major depressive episode (MDE), bipolar affective disorder (BD) or an anxiety disorder were included. All subtypes of MDD or MDE (mild, moderate, severe, with/without psychotic features) and bipolar disorder (rapid cycling, type I, type II and other) were included. Participants who only met criteria for dysthymia or cyclothymia were excluded.
Intervention type
Any studies that used an appropriate genetic or pharmacological manipulation to the 5-HT7 system were included in the present review. Pharmacological agents, in both preclinical and human studies, that have substantial selectivity for 5-HT7 receptors were included, such as selective agonists (AS-19, LP-44, LP-12, LP-211, E55888) (Brenchat et al., 2009; Godínez-Chaparro et al., 2011; Leopoldo et al., 2004, 2008; Sanin et al., 2004) and antagonists (SB-238719, SB-269970, SB-656104, DR-4004, DR-4446, PZ-766, JNJ-18038683, asenapine, amisulpride, imipramine, and desipramine) (Azima and Vispo, 1958; Bonaventure et al., 2012; Brodie et al., 1961; Canale et al., 2016a; Forbes et al., 2002; Frånberg et al., 2008; Hagan et al., 2000; Kikuchi et al., 1999; Puech et al., 1998; Thomas et al., 1999; Zhang et al., 2002; Zajdel et al., 2012a). Lurasidone and vortioxetine, antipsychotic and antidepressant medications, respectively, were also included due to their partial 5-HT7 receptor antagonism (Adell, 2010; Ohno et al., 1997). Studies that did not use pharmacological agents with reasonable 5-HT7 affinity were excluded.
Outcome measures
The primary outcome measure of interest was changes in mood and anxiety behaviors after pharmacological interventions with agents that bind to, or alter, 5-HT7 receptor function. In preclinical animal studies, these behavioral changes were evaluated by use of validated functional tests that may measure depressive or anxiety symptomatology (e.g. light dark transfer test, elevated plus-maze tests, forced swim tests, and tail suspension tests). Secondary outcome measures of interests were changes in sleep or cognition measures.
For human studies, changes from baseline to endpoint in mood status, assessed by change in mood-related symptoms measured by validated rating scales such as the Hamilton Depression Rating Scale (HAM-D) or Quick Inventory of Depressive Symptomology (QIDS-CR) was the primary outcome measure (Hamilton, 1960; Rush et al., 2003). Any included data on change in sleep, cognition, or other experimental medicine markers (e.g. neuroimaging) were also collected.
Data collection and analysis
Data extraction was conducted after individual articles were assessed against inclusion criteria and discussed further where needed. All data extraction was completed independently by two authors (NG and TYL).
Quality measures
Study quality was measured by using the “Quality Assessment Tool for Quantitative Studies,” which was designed by Effective Public Health Practice Project (Armijo-Olivo et al., 2012; Thomas et al., 2004). Included studies were assessed in the following domains: selection bias, study design, confounders, blinding, data collection methods, withdrawal, and dropout. A global quality rating was scored independently, and any disagreements discussed by reviewers.
Results
Systematic search results
The initial search identified 8097 papers. After removal of duplicates, 4989 studies underwent initial title and abstract screening leaving 109 studies for full-text review. Review of full-text articles excluded 49 articles for the following reasons: article type (7 review or meta-analysis papers and 18 conference abstracts or posters), wrong disorder or outcome measure (20), data reported elsewhere (2), not a randomized control trials study (1), and article language (1). Two further studies were added from hand searching after the original search. In total, 64 studies were included (52 animal studies and 12 human studies), and details are outlined in Figure 1.
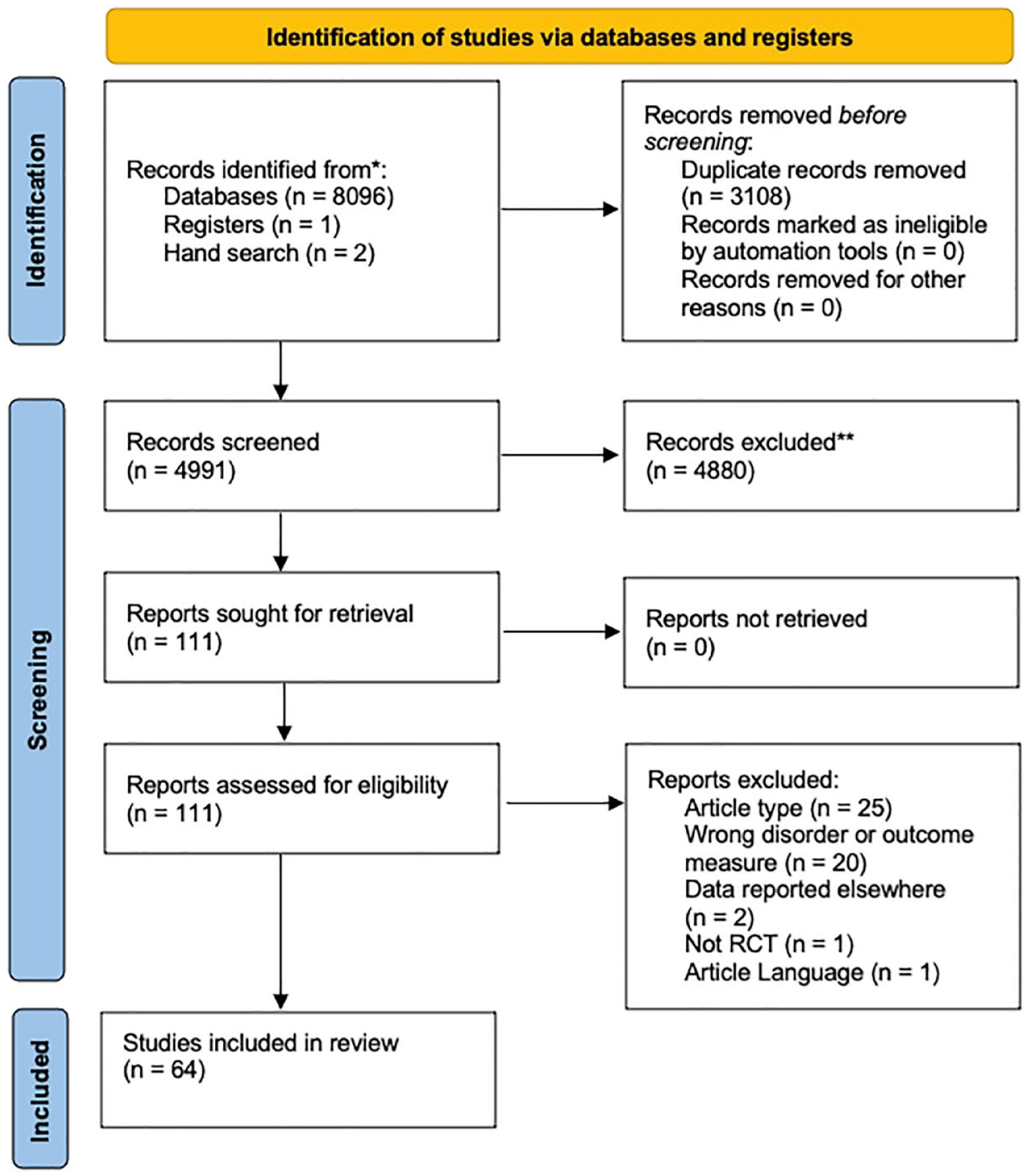
PRISMA diagram.
Preclinical study results
Study characteristics
Fifty preclinical animal studies were included in the review. Most of the studies (48/52) examined 5-HT7 drug or knockout (KO) effects on preclinical models of depression, with a further 23 examining models of anxiety. Study details and summarized findings are listed in Table 1.
Description of preclinical studies.
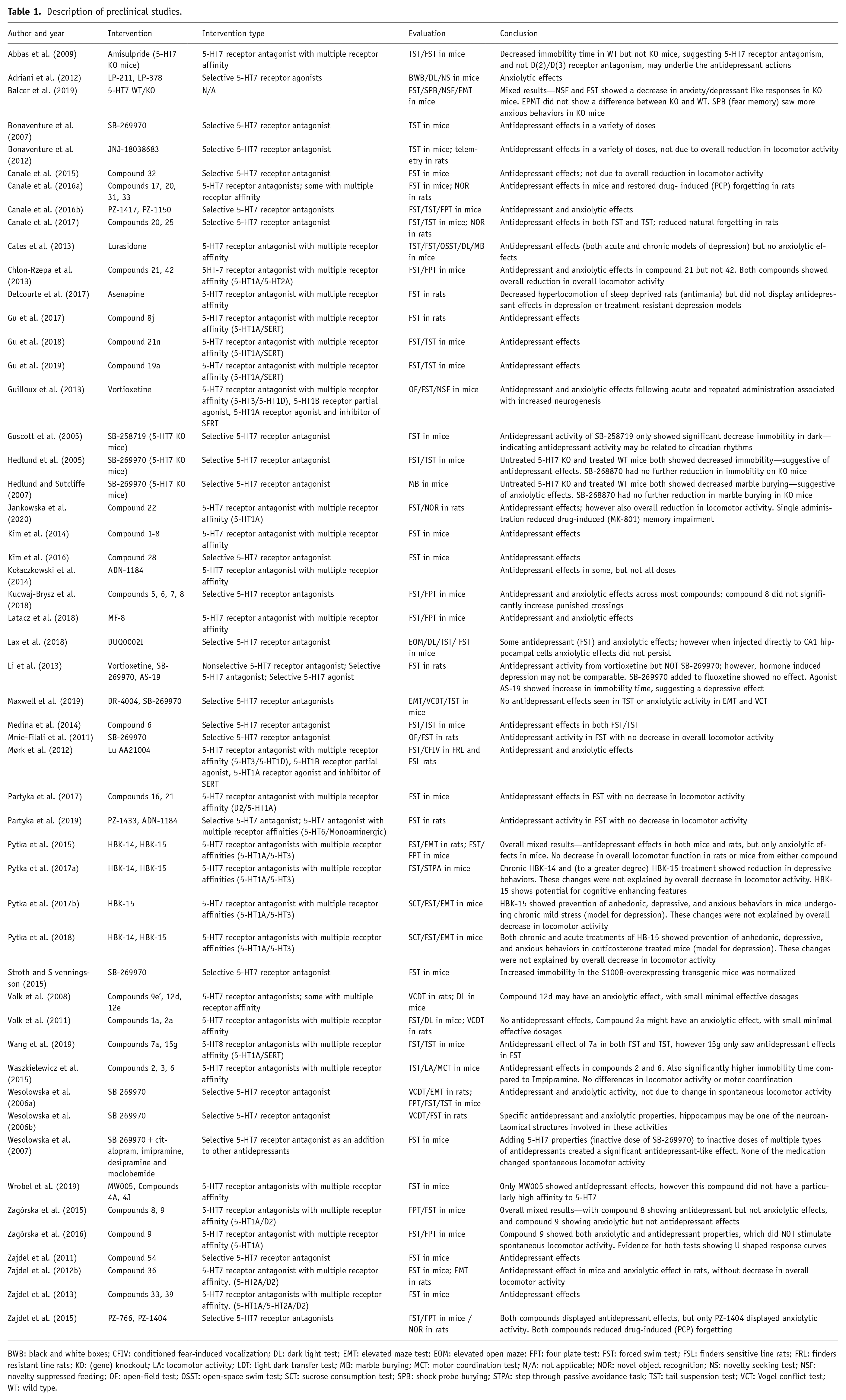
BWB: black and white boxes; CFIV: conditioned fear-induced vocalization; DL: dark light test; EMT: elevated maze test; EOM: elevated open maze; FPT: four plate test; FST: forced swim test; FSL: finders sensitive line rats; FRL: finders resistant line rats; KO: (gene) knockout; LA: locomotor activity; LDT: light dark transfer test; MB: marble burying; MCT: motor coordination test; N/A: not applicable; NOR: novel object recognition; NS: novelty seeking test; NSF: novelty suppressed feeding; OF: open-field test; OSST: open-space swim test; SCT: sucrose consumption test; SPB: shock probe burying; STPA: step through passive avoidance task; TST: tail suspension test; VCT: Vogel conflict test; WT: wild type.
A comprehensive listing of all relevant study results, with corresponding mood-related behavioral paradigms and mood models, comparators, individual results and p-values can be found in Supplemental Information—Preclinical Behavioral Results.
While all included studies used compounds or genetic manipulations that reasonably targeted 5-HT7 receptors, some compounds had higher selectivity than others.
Thirty used nonspecific 5-HT7 antagonist agents, which also have moderate to high affinity to other receptors, including other 5-HT and dopaminergic receptors, while 25 studies used compounds selective to 5-HT7 receptors.
For the purpose of this review, selective compounds are considered those which had preferential binding defined as a 5-HT7 inhibition constant (Ki) at least fourfold less than all other receptors of interest. A complete list of compounds considered in this review, with their chemical (IUPAC) name and receptor binding profiles, can be found in Supplemental Information—Receptor Activity. Receptor bindings are described by inhibition constants (Ki in nanomoles) unless otherwise noted (e.g. IC50 or pKi values). The Psychoactive Drug Screening Program Ki database was used to reference all compounds (Helmuth, 2000; Roth et al., 2000). If compounds were not listed in the database, this is noted in the Supplemental Information along with source data for receptor bindings.
Antidepressant effects
The forced swim test (FST) and the tail suspension test (TST) are often used to evaluate antidepressant agents in preclinical rodent studies, and these tests measure immobility time as a correlate of negative mood, or hopelessness (Can et al., 2011; Yankelevitch-Yahav et al., 2015). Even though the FST and TST are not recognized as effective models of depression, they are still used for screening potential antidepressants in preclinical studies (Bourin et al., 2001; Sewell et al., 2021). Within our systematic review, 43 studies used the FST, and 15 studies used the TST.
Twenty-five studies that used compounds nonspecific to the 5-HT7 receptor identified potential antidepressant effects depression such as immobility time, in both the FST or TST (Abbas et al., 2009; Cates et al., 2013; Chlon-Rzepa et al., 2013; Gu et al., 2019, 2018, 2017; Guilloux et al., 2013; Jankowska et al., 2020; Kim et al., 2014; Kołaczkowski et al., 2014; Latacz et al., 2018; Li et al., 2013; Mørk et al., 2012; Partyka et al., 2017, 2019; Pytka et al., 2015, 2017a, 2017b, 2018; Wang et al., 2019; Waszkielewicz et al., 2015; Wróbel et al., 2019; Zagórska et al., 2015, 2016; Zajdel et al., 2013).
Furthermore, 19 studies using agents with selective 5-HT7 antagonist properties found significant improvements in potential markers of depression; for example, SB-269970 was found to decrease animals’ immobility time both in water (FST) and land (TST) (Bonaventure et al., 2012, 2007; Canale et al., 2015, 2016b, 2016a, 2017; Guscott et al., 2005; Hedlund et al., 2005; Kim et al., 2016; Kucwaj-Brysz et al., 2018; Lax et al., 2018; Medina et al., 2014; Partyka et al., 2019; Stroth and Svenningsson, 2015; Wesolowska et al., 2006a, 2006b, 2007; Zajdel et al., 2011, 2015). An additional study found decreased immobility time in 5-HT7 gene KO mice, compared to wild type (WT) mice, in the FST (Balcer et al., 2019). Lastly, one study using a highly selective 5-HT7 agonist, AS-19, reported increased immobility (i.e. more depressed-like behavior) in the FST (Li et al., 2013).
Of note, the antipsychotic medication amisulpride and antidepressants with 5-HT7 antagonist properties, such as imipramine and desipramine, were found to have potential antidepressant effects in these tests (Abbas et al., 2009; Wesolowska et al., 2007). Abbas and colleagues found that amisulpride, an antipsychotic that acts primarily as a dopaminergic receptor antagonist but also has potent 5-HT7 receptor antagonism (Abbas et al., 2009), showed potential antidepressant effects in both the TST and FST in WT mice. These effects were not seen in 5-HT7 KO mice, suggesting that activity at the 5-HT7 receptor may be specifically associated with potential antidepressant properties. Another study using citalopram, imipramine, desipramine, and moclobemide found that while low doses of these agents had no significant effect, antidepressant properties were evident once combined with SB-269970, a selective 5-HT7 receptor antagonist, suggesting that it was enhanced 5HT7 antagonism that produced potential antidepressant-like effects (Wesolowska et al., 2007).
Two studies found mixed results, each with a 5-HT7 antagonist compound showing potential antidepressant effects in the FST, but not TST (Lax et al., 2018; Wang et al., 2019). A further three studies showed no significant antidepressant effects of their tested compounds in either the TST or FST (Delcourte et al., 2017; Maxwell et al., 2019; Volk et al., 2011).
Anxiolytic effects
In addition to modulation of depressive symptoms, there is also evidence from preclinical animal studies that 5-HT7 receptor modulation may mediate changes in measures of anxiety. A total of 23 studies measured anxiety behaviors using a variety of paradigms.
Similarly to preclinical depression studies, 11 studies found potential anxiolytic effects in animal models associated with the use of 5-HT7 antagonist agents, which also have strong to moderate affinities at other serotonergic and dopaminergic receptors (Chlon-Rzepa et al., 2013; Guilloux et al., 2013; Latacz et al., 2018; Pytka et al., 2015, 2017b, 2018; Volk et al., 2008, 2011; Zagórska et al., 2015, 2016; Zajdel et al., 2012b). Eight studies found potential anxiolytic effects when investigating selective 5-HT7 receptor antagonists (Canale et al., 2016a; Hedlund and Sutcliffe, 2007; Kucwaj-Brysz et al., 2018; Lax et al., 2018; Mnie-Filali et al., 2011; Wesolowska et al., 2006a, 2006b; Zajdel et al., 2015).
These studies used animal models of anxiety where anxiolytic effects were measured by a decrease in rodents’ motion or increase their searching behaviors in anxious environments (e.g. maintaining motionless to escape from electric shock in Four-Plate Test, increasing attempts to get food under electrical shocks in Vogel Conflict Drinking Test, or burying more marbles in their bedding in Marble Burying Test). The selective 5-HT7 antagonist, SB-269970, increased antianxiety behaviors in the Vogel Conflict Drinking Test and in the elevated plus-maze test (Wesolowska et al., 2006a). A similar finding was identified in the shock threshold test and open-field test (Wesolowska et al., 2006b). In addition, mice showed less anxious behavior in the marble burying test, which may measure obsessive-compulsive behaviors (Hedlund and Sutcliffe, 2007).
Balcer and colleagues found mixed results, with 5-HT7 gene KO mice being significantly less anxious during a novelty suppressed feeding task, but not during an elevated maze plus task (Balcer et al., 2019). Two studies found no significant anxiolytic effects associated with any compounds with 5-HT7 antagonistic properties (Cates et al., 2013; Maxwell et al., 2019). One study found that the 5-HT7 agonists, LP-211 and LP-378, improved potential anxiety-related behaviors in the light/dark test and increased exploration of black and white boxes (Adriani et al., 2012).
Other effects
In addition to antidepressant and anxiolytic effects, 5-HT7 antagonists have been investigated in animal models of mania, sleep, and cognition. One study found that hyperlocomotion in rodents induced by sleep deprivation, a behavioral model of mania, was reduced after treatment with asenapine (Delcourte et al., 2017). Rodents treated with JNJ-18038683, a selective 5-HT7 antagonist, and 5-HT7 gene KO mice both displayed increased latency to REM sleep and decreased REM duration (Bonaventure et al., 2012; Hedlund et al., 2005). Additionally, the selective 5-HT7 antagonist SB-269970 increased REM latency, decreased REM sleep duration, and reversed increases in sleep fragmentation induced by citalopram (Bonaventure et al., 2007).
Several studies have also reported that compounds with 5-HT7 antagonist properties were associated potential pro-cognitive effects, such as reversing natural forgetting impairment in rats (Canale et al., 2017; Pytka et al., 2017a) and reversing drug-induced memory impairments (Canale et al., 2016b; Jankowska et al., 2020; Zajdel et al., 2015).
Human study results
Study characteristics
Twelve RCTs were included in the systematic review. Most of the studies (11) were between 6 and 8 weeks long (mean study duration 7.4 weeks); however, one study lasted for 32 weeks. Eight studies explored effects in participants with MDD, two in participants with bipolar disorder, and two in participants with generalized anxiety disorder (GAD). Only one study used a highly selective 5-HT7 antagonist (JNJ-18038683), while the others used medications that had higher affinity to other receptors, in addition to 5-HT7 receptor activity. All clinical trial details and findings are summarized in Table 2.
Description of human studies.
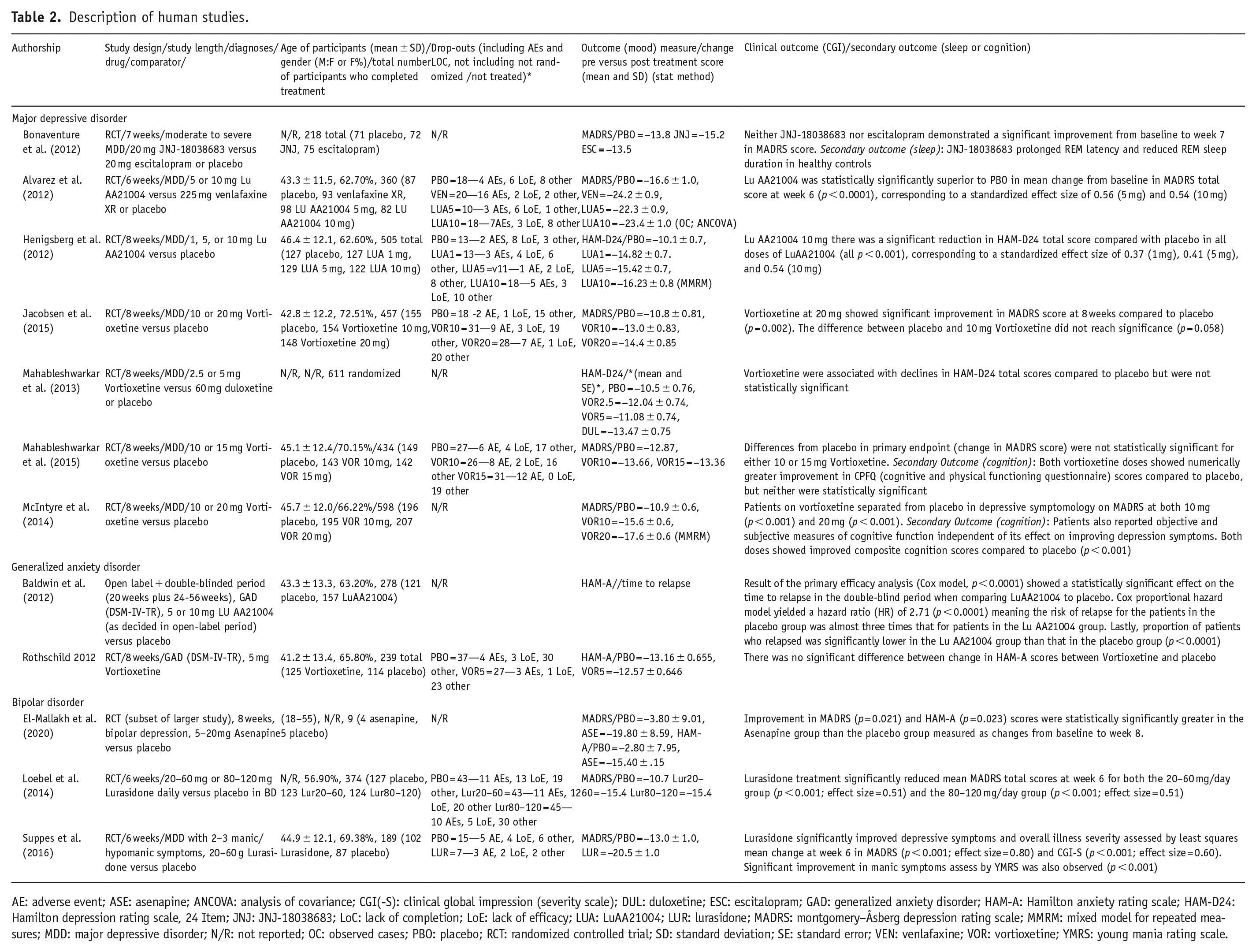
AE: adverse event; ASE: asenapine; ANCOVA: analysis of covariance; CGI(-S): clinical global impression (severity scale); DUL: duloxetine; ESC: escitalopram; GAD: generalized anxiety disorder; HAM-A: Hamilton anxiety rating scale; HAM-D24: Hamilton depression rating scale, 24 Item; JNJ: JNJ-18038683; LoC: lack of completion; LoE: lack of efficacy; LUA: LuAA21004; LUR: lurasidone; MADRS: montgomery–Åsberg depression rating scale; MMRM: mixed model for repeated measures; MDD: major depressive disorder; N/R: not reported; OC: observed cases; PBO: placebo; RCT: randomized controlled trial; SD: standard deviation; SE: standard error; VEN: venlafaxine; VOR: vortioxetine; YMRS: young mania rating scale.
Major depressive disorder
Six studies investigated the effect of vortioxetine (LU AA21004), a medication with 5-HT1A, 5-HT1B, 5-HT3, and 5-HT7 receptors and 5-HT transporter affinities, on MDD symptoms. All studies administering vortioxetine at 20 mg found significant improvements in mood as measured by Montgomery–Åsberg Depression Rating Scale (MADRS) (Montgomery and Asberg, 1979) between baseline and week 8 (Jacobsen et al., 2015; McIntyre et al., 2014). Three studies found significant differences in mood at a 10-mg dose (Alvarez et al., 2012; Henigsberg et al., 2012; McIntyre et al., 2014); however, two only found a trend toward significance at this dosage (Jacobsen et al., 2015; Mahableshwarkar et al., 2015). Lower doses of vortioxetine (5 mg or less) found no significant changes associated with treatment (Alvarez et al., 2012; Henigsberg et al., 2012; Mahableshwarkar et al., 2013). It is important to emphasize that vortioxetine is not a 5-HT7-specific agent, and vortioxetine also blocks 5-HT1D and 5HT-3 receptors and stimulates 5-HT1A and 5HT1B receptors.
Only one study examined change in depressive symptoms after 7 weeks of 20-mg JNJ-18038683, a highly selective 5-HT7 receptor antagonist, compared to placebo and escitalopram 20 mg (Bonaventure et al., 2012). Although JNJ-18038683 did not significantly decrease depression symptoms compared to placebo, escitalopram (20 mg) also did not significantly change mood scores in this study.
Anxiety disorders
Two studies examined the effects of medications with 5-HT7 antagonistic properties in participants with GAD. Rothschild and colleagues measured anxiety symptoms using the Hamilton Anxiety Scale (HAM-A) (Hamilton, 1959) before and after 8 weeks of treatment with 5 mg vortioxetine, but found no differences compared to placebo (Rothschild et al., 2012). Baldwin and colleagues investigated the effect of either 5 or 10 mg of vortioxetine on time to relapse in participants with GAD. At the end of a 20-week open-label treatment period, participants who responded were then randomized to 24–56 weeks of a double-blind treatment of vortioxetine (n = 229) or placebo (n = 230). The study found a statistically significant effect of vortioxetine relative to the placebo in time to relapse (Baldwin et al., 2012).
Bipolar disorders
Lurasidone and asenapine are both atypical antipsychotics often used to treat schizophrenia and bipolar disorders, and they both have moderate affinity to 5-HT7 receptors. Lurasidone is a dopaminergic D2 and D3 receptor, 5-HT2A, 5-HT7, and α2C-adrenergic receptor antagonist, and a partial 5-HT1A agonist (Bawa and Scarff, 2015). In one study investigating bipolar depression, participants treated with lurasidone (at both 20–60 mg and 80–120 mg doses) experienced a significant improvement in MADRS scores after 6 weeks compared to placebo (Loebel et al., 2014). One study, with a sample size of nine participants, found significant improvements in both depressive and anxiety symptoms in those with bipolar depression after 8-week treatment with asenapine (El-Mallakh et al., 2020). In participants with MDD with mixed features, who experienced least 2–3 manic or hypomanic episodes, depressive symptoms measured by MADRS and manic symptoms measured by the Young Mania Rating Scale (YMRS) improved after 6 weeks of treatment of 20–60 mg lurasidone (Suppes et al., 2016; Young et al., 1978).
Other effects
As with rodent studies, human clinical trials have observed effects of 5-HT7 receptor modulation outside of mood symptoms. McIntyre and colleagues found that both 10- and 20-mg vortioxetine significantly improved participants’ composite cognition scores compared to placebo using a predefined efficacy analysis. Additionally, they also found significant improvements in most secondary objectives and subjective patient-reported cognitive measures (McIntyre et al., 2014). Another study measuring cognitive impairment using the Cognitive and Physical Functioning Questionnaire (CPFQ) found while both 10- and 15-mg vortioxetine numerically improved scores, these improvements were not statistically significant versus placebo (Fava et al., 2009; Mahableshwarkar et al., 2015).
Only one study in the systematic review examined changes in sleep in human participants. JNJ-18038683, a specific 5HT-7 antagonist, was found to prolong REM latency and reduced REM sleep duration in healthy participants, and enhanced REM sleep suppression induced by citalopram (Bonaventure et al., 2012).
Quality assessment
Overall, the clinical trials included in this systematic review were assessed to be between strong and moderate quality using the Quality Assessment Tool for Quantitative Studies, and the scores are listed in Table 3. All studies were considered to have a strong study design and strong blinding strategies. Some studies did not describe participant withdrawals and drop out, which is vital to a well described trial. Full quality assessment results for each study can be found in Table 3.
Results of quality assessment.
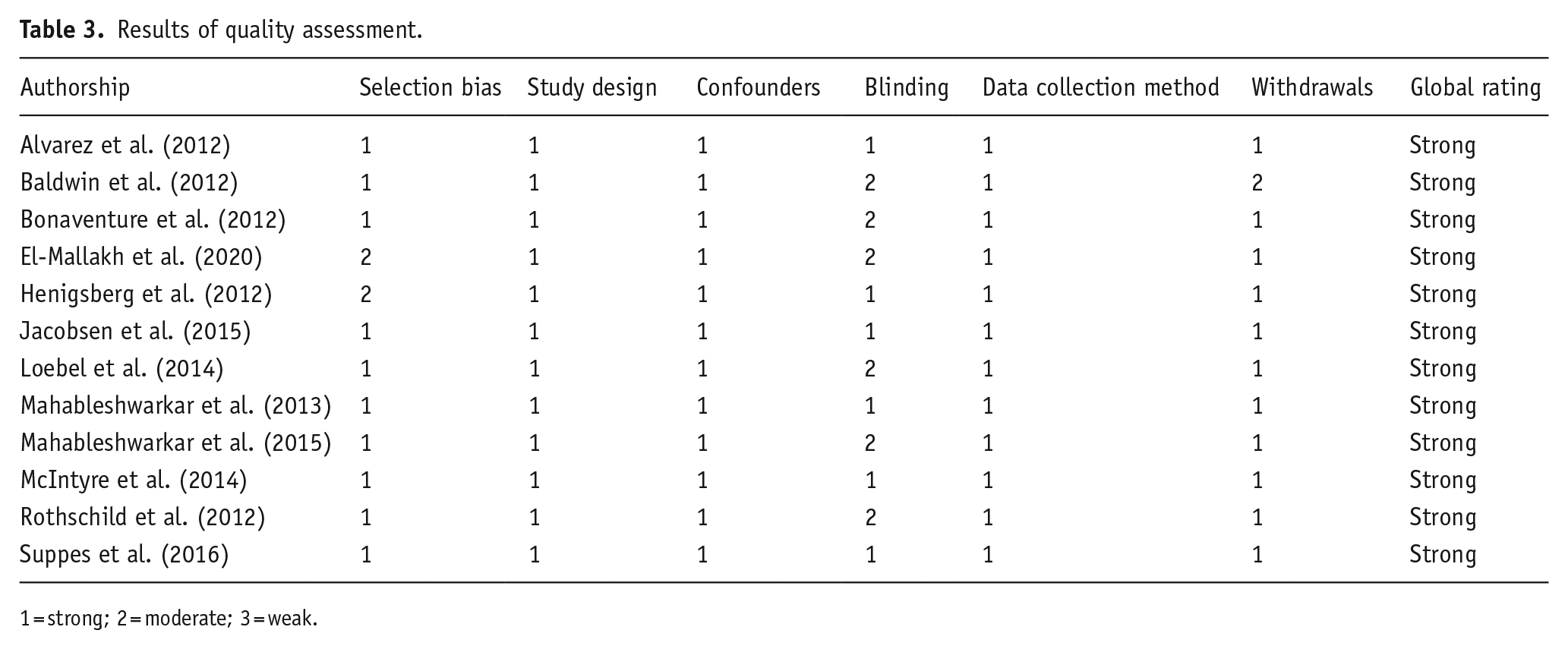
1 = strong; 2 = moderate; 3 = weak.
Discussion
Overall, the animal studies and human clinical trials included in this review provide preliminary evidence that 5-HT7 antagonists may potentially be useful for the treatment of mood and anxiety disorders, with 49 animal studies and 11 human studies reporting statistically significant changes in mood or behavior using 5-HT7-related pharmacological interventions. However, this review highlights that the evidence base is constrained by a lack of studies using specific 5-HT7 receptor agents, which do not influence dopaminergic or other 5-HT receptor subtypes. While many pharmacological interventions with activity at the 5-HT7 receptor have been found to induce significant changes in mood symptoms in clinical trials, these results are difficult to interpret due to the lack of 5-HT7 receptor specificity.
Preclinical studies report an improvement in depression-related (43 studies) and anxiety-related (21 studies) behaviors, further supporting that the notion that 5-HT7 receptor modulation may impact mood- and anxiety-related symptoms. For example, the selective 5-HT7 antagonist SB-269970 was associated with specific anxiolytic effects in the Vogel conflict drinking, elevated plus-maze, shock threshold, and open-field tests (Wesolowska et al., 2006b), without influencing gross locomotion. Selective 5-HT7 antagonists have also been shown to induce possible antidepressant-like behaviors in preclinical animal models (Bonaventure et al., 2007, 2012; Canale et al., 2015, 2016a, 2016b, 2017; Guscott et al., 2005; Hedlund et al., 2005; Kim et al., 2016; Kucwaj-Brysz et al., 2018; Lax et al., 2018; Medina et al., 2014; Partyka et al., 2019; Stroth and Svenningsson, 2015; Wesolowska et al., 2006a, 2006b, 2007; Zajdel et al., 2011, 2015) although the interpretation of these results is complicated by concerns over the validity of these tests measuring depressive symptoms and lack of validation of receptor binding profiles for many of the compounds used. The use of compounds with multiple receptor subtype affinity (such as MF-8, HBK-14, and HBK-15) was also associated with potential antidepressant-like and anxiolytic-like effects, although the lack of specificity of these compounds for the 5-HT7 receptor makes interpretation of these results difficult. For example, Wrobel and colleagues only found that MW005 may have antidepressant-related effects in animal models. However, this compound had lower 5-HT7 receptor selectivity compared to the newer compounds investigated in this study, which were not associated with antidepressant-related effects (Wróbel et al., 2019).
In addition to 5-HT7 antagonism, 5-HT7 agonists were found to impact emotion in preclinical animal models. One study found that 5-HT7 agonist AS-19 exacerbated depressive symptoms in rodent models of progesterone withdrawal, which are believed to model hormonally induced mood disorders in women (Li et al., 2013). Conversely, Adriani and colleagues found that LP-211 and LP-378, two 5-HT7 agonists, were associated with increased disinhibition across a variety of tasks, including B/W boxes, dark/light, and novelty seeking tasks. The researchers observed pro-locomotor and pro-exploratory behaviors, with mice spending more time in the aversive light side and white boxes (Adriani et al., 2012). Further studies using 5-HT7 agonists are needed to clarify the mechanisms underlying these observations.
In human clinical trials, several studies found an improvement of symptoms in people with MDD after treatment with vortioxetine. All studies administering vortioxetine at 20 mg found significant improvements in mood (Jacobsen et al., 2015; McIntyre et al., 2014). Those using lower dosages had more mixed results, suggesting it may be only doses of vortioxetine greater than 10 mg are effective in symptom improvement. Lastly, two studies also found improvements in cognition with use of at least 10-mg vortioxetine in these participants with MDD (Mahableshwarkar et al., 2015; McIntyre et al., 2014). As only these two studies measured changes in cognition, further research in needed.
In people with GAD, 5-mg vortioxetine was associated with no differences in anxiety symptoms (Rothschild et al., 2012); however, vortioxetine at 5 and 10 mg did show a statistically significant effect of vortioxetine relative to the placebo in time to relapse in a longer 56-week study (Baldwin et al., 2012). Taken together, these studies highlight the importance of dosage and treatment duration when considering efficacy in the treatment of affective symptoms.
In the only human trial using a selective 5-HT7 antagonist, Bonaventure and colleagues found that while there was no significant decrease in depressive symptoms compared to placebo, JNJ-18038683 prolonged REM latency and reduced REM sleep duration in healthy controls. It is important to note that in this study, neither escitalopram (20 mg), a commonly used antidepressant, nor JNJ-18038683 significantly decreased depressive symptomology as measured by the MADRS compared to placebo (Bonaventure et al., 2012).
The quality of the clinical trials included in this review was considered to be strong or moderate overall. However, one study had small sample size (n = 9) due to it being a subset of a larger study that was terminated early by the sponsor for non-safety-related issues (El-Mallakh et al., 2020). Quality assessments were not carried out on preclinical rodent studies as the availability of standardized quality assessment for use in animal studies is lacking (Zeng et al., 2015).
Limitations and future directions
Despite the evidence for the potential role of the 5-HT7 receptor subtype in mood and anxiety disorders, this systematic review has several limitations.
While all the studies included in this review used pharmacological treatments with moderate to strong affinity to 5-HT7 receptor subtypes, many also have affinity to other receptor types including the SERT and 5-HT1A receptors, which are often targets for antidepressants and anxiolytics (Artigas, 2013; Nautiyal and Hen, 2017). Only 21 of the original 52 preclinical studies measuring depressive symptomology used selective 5-HT7 pharmacological interventions. While the compounds described in this systematic review were defined as selective based on a preferential binding to 5-HT7 receptors compared to others, some of these compounds did have moderate affinities to other receptors with inhibition constants less than 100 nm. For example, while PZ-766 is a potent 5-HT7 antagonist, with a Ki value of 0.3 nm, there is also indication of activity at the D2 receptor (Ki = 52 nm) (Zajdel et al., 2015). This may imply that changes in mood symptoms result in part from the modulation of SERT, 5-HT1A, or 5-HT2 receptor function. JNJ-18038683 was the only highly selective 5-HT7 antagonist used in human trials, highlighting that further research is required using more selective 5-HT7 compounds to better characterize the underlying pharmacology of the observed changes in mood and anxiety disorders.
Although antidepressant- and anxiolytic-related effects were widely examined, only one preclinical study and one clinical trial explored effects on mania-related measures using agents with wider therapeutic targets (Delcourte et al., 2017; Suppes et al., 2016). Sleep and cognition were examined as secondary outcome measures in nine animal studies and three human studies (Adriani et al., 2012; Bonaventure et al., 2012, 2007; Canale et al., 2017, 2016b; Jankowska et al., 2020; Pytka et al., 2017a; Zajdel et al., 2015). We would suggest that further research using more selective 5-HT7 compounds is necessary to fully understand the effects of 5-HT7 receptor modulation on mania, sleep, and cognition function (Gasbarri and Pompili, 2014).
Finally, it is important to note that no unpublished data were identified to be included in this systematic review, which may indicate that negative findings could have been underreported.
Conclusions
This systematic review examined evidence for the 5-HT7 receptor as a therapeutic target for mood and anxiety disorders with 49 preclinical and 11 human studies demonstrating antidepressant- or anxiolytic-related effects using compounds that had at least moderate 5-HT7 receptor affinity. The 5-HT7 receptor may also be a potential target for the treatment of sleep disturbances or cognitive impairment associated with mood disorders, but further research is warranted. We would suggest that further studies investigate pharmacological agents with more distinct selectivity to the 5-HT7 receptor subtype, to ensure that effects are not related to their affinity to other receptors (such as 5-HT1A, D2, or SERT). Additionally, more research is needed to observe changes in cognition and sleep.
Supplemental Material
sj-docx-1-jop-10.1177_02698811231211228 – Supplemental material for The 5-HT7 receptor system as a treatment target for mood and anxiety disorders: A systematic review
Supplemental material, sj-docx-1-jop-10.1177_02698811231211228 for The 5-HT7 receptor system as a treatment target for mood and anxiety disorders: A systematic review by Natalie Gottlieb, Tse-Yi Li, Allan H Young and Paul RA Stokes in Journal of Psychopharmacology
Supplemental Material
sj-docx-2-jop-10.1177_02698811231211228 – Supplemental material for The 5-HT7 receptor system as a treatment target for mood and anxiety disorders: A systematic review
Supplemental material, sj-docx-2-jop-10.1177_02698811231211228 for The 5-HT7 receptor system as a treatment target for mood and anxiety disorders: A systematic review by Natalie Gottlieb, Tse-Yi Li, Allan H Young and Paul RA Stokes in Journal of Psychopharmacology
Supplemental Material
sj-xlsx-3-jop-10.1177_02698811231211228 – Supplemental material for The 5-HT7 receptor system as a treatment target for mood and anxiety disorders: A systematic review
Supplemental material, sj-xlsx-3-jop-10.1177_02698811231211228 for The 5-HT7 receptor system as a treatment target for mood and anxiety disorders: A systematic review by Natalie Gottlieb, Tse-Yi Li, Allan H Young and Paul RA Stokes in Journal of Psychopharmacology
Footnotes
Acknowledgements
We would like to thank Dr. Anthony Vernon for his help and advice on interpretation of preclinical compound binding.
Author’s Note
Natalie Gottlieb and Allan H Young are also affiliated to South London and Maudsley NHS Foundation Trust, Bethlem Royal Hospital, Monks Orchard Road, Beckenham, Kent, UK.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AY declares honoraria for speaking from Astra Zeneca, Lundbeck, Eli Lilly, Sunovion; honoraria for consulting from Allergan, Livanova and Lundbeck, Sunovion, Janssen; and research grant support from Janssen. He is an editor for Journal of Psychopharmacology and Deputy Editor, BJPsych Open. PS reports personal fees and nonfinancial support from Frontiers in Psychiatry, personal fees from Allergan and a grant from H Lundbeck outside the submitted work. All authors report support from a grant from the Medical Research Council UK and nonfinancial support from Janssen Research and Development LLC during the conduct of the study. No other declarations of interest are reported.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study represents independent research partly funded a Medical Research Council UK grant (MR/R005885/1). This work was also supported by the Ministry of Education, Taiwan as a PhD scholarship to TYL. This paper also includes independent research part funded by the National Institute for Health Research (NIHR) Maudsley Biomedical Research Centre at South London and Maudsley NHS Foundation Trust and King’s College London. The funders were not involved in any aspects of this work’s planning, execution, article preparation or in the decision to submit the article for publication. The views expressed are those of the authors and not necessarily those of the funding Trusts, the NHS, the NIHR, or the Department of Health and Social Care.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
