Abstract
Background:
Cannabis constituents, including Δ9-tetrahydrocannabinol (THC) and cannabidiol (CBD), show distinct pharmacological profiles with therapeutic relevance for neurological and psychiatric conditions. THC exerts euphoric effects primarily via CB1 receptor activation, while CBD displays non-euphoric properties affecting various pathways.
Aims:
This study evaluated the effects of THC, CBD, and their combination on brain functional connectivity (FC) and cerebral blood flow (CBF) using multimodal neuroimaging.
Methods:
Adult male Sprague Dawley rats received intraperitoneal doses of 10 mg/kg THC, 150 mg/kg CBD, 10.8:10 mg/kg THC:CBD, or vehicle. Resting-state blood oxygenation level dependent magnetic resonance imaging and arterial spin labelling assessed FC and CBF, approximately 2 h after drug administration. Graph-theory metrics and seed-based analyses identified connectivity and perfusion alterations, while plasma analyses determined cannabinoid concentrations.
Results:
THC increased whole-brain FC and clustering coefficient, with elevated CBF in cortical and subcortical regions. CBD decreased FC metrics without affecting CBF, while THC:CBD induced moderate increases in both. Seed-based analysis revealed THC-driven increases in cortical-hippocampal and cortical-striatal connectivity, attenuated in the THC:CBD group. A multivariate combined analysis of FC and CBF revealed a divergent pattern of changes induced by each drug.
Conclusions:
In conclusion, we show that THC and CBD induce distinct neurophysiological profiles in rats, with THC increasing both connectivity and perfusion, moderated by CBD when combined. These findings corroborate existing knowledge about the effects of cannabinoids on the brain, while also supporting the potential of preclinical functional neuroimaging to delineate cannabinoid-induced endophenotypes, offering insights for therapeutic development.
Introduction
Growing evidence supports the therapeutic potential of cannabis and its constituent phytocannabinoids in treating a range of neurological and psychiatric conditions (Friedman and Devinsky, 2015; Montero-Oleas et al., 2020; Sarris et al., 2020; Serpell et al., 2014). Cannabis (Cannabis sativa (C. sativa)) contains around 100 cannabinoids, as well as various non-cannabinoid phytochemicals such as terpenes, flavonoids and phytosteroids (Baron, 2018; Fordjour et al., 2023). Among these compounds, Δ9-tetrahydrocannabinol (THC) and cannabidiol (CBD) are the most abundant and extensively studied phytocannabinoids.
THC is increasingly recognised for its therapeutic benefits. It is approved as an anti-emetic in cancer treatment, has shown anti-spastic effects beneficial for managing multiple sclerosis (MS), and provides analgesic relief for chronic pain (Argueta et al., 2020; Collin et al., 2007; Davis, 2016; Marinelli et al., 2017). Pharmacologically, THC acts as a partial agonist at both cannabinoid receptors 1 (CB1R) and 2 (CB2R; Paronis et al., 2012; Pertwee, 2008; Sim et al., 1996). While its euphoric effects are primarily mediated through CB1R activation (D’Souza et al., 2005; Sherif et al., 2016), its therapeutic effects likely involve additional actions, including interactions with CB2R, and modulation of glutamatergic and opioid signalling pathways (Baron, 2018; Leinen et al., 2023).
CBD is another prominent constituent derived from C. sativa (Grotenhermen and Muller-Vahl, 2012), widely recognised for its therapeutic potential. Unlike THC, CBD lacks any euphoric effects (Pertwee, 2008), likely due to its distinct pharmacological profile, showing minimal affinity for CB1R and CB2R. Plant-derived highly purified CBD (>98%) is approved as Epidiolex® for the treatment of seizures associated with Dravet syndrome, Lennox–Gastaut syndrome and tuberous sclerosis complex in the US and other territories, and anticonvulsive efficacy of CBD has also been demonstrated in animal models of seizure and epilepsy (Gray et al., 2020; Klein et al., 2017; Patra et al., 2019). Generally, CBD has been shown to have diverse actions; preclinically, there is potential evidence in treating neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease by reducing neuroinflammation and enhancing neuronal protection. Its anxiolytic properties suggest applications for anxiety-related disorders, while it also possesses anti-psychotic properties, offering benefits without the euphoric effects associated with THC (Bergamaschi et al., 2011; Crippa et al., 2004; McGuire et al., 2018; Moreira and Guimaraes, 2005; Zuardi et al., 2012a).
Current evidence suggests that CBD’s diverse actions result from multiple, only partially understood mechanisms, including its negative allosteric modulation of CB1Rs (Laprairie et al., 2015; Jakowiecki et al., 2021; Ibeas Bih et al., 2015) and modulation of glycine and opioid receptors (Baron, 2018). CBD also reduces neuronal hyperexcitability and inflammation through modulation of transient receptor potential vanilloid (TRPV1) channels, GPR55 antagonism and inhibition of adenosine uptake via the ENT-1 transporter (Gray et al., 2020). Additionally, its interactions with serotonin 5-HT1A receptors may contribute to its anxiolytic-like and neuroprotective effects (Baron, 2018; Leinen et al., 2023; Stella, 2023).
Nabiximols, marketed as Sativex®, is a botanically derived agent containing a wide variety of cannabinoids and non-cannabinoids with the most abundant cannabinoids being THC and CBD in a 1.08:1 ratio, that has been approved in several countries for the treatment of spasticity in patients with MS (Patti et al., 2022). Unlike plant-based cannabis extracts, which can have varying concentrations of THC and other cannabinoids, nabiximols offers a consistent formulation with a higher concentration of CBD. This balanced ratio is thought to enable higher THC doses while minimising undesirable side effects, as CBD may partially counteract THC’s euphoric and sedative properties, as well as some of its pharmacodynamic effects (Britch et al., 2017; Boggs et al., 2018; Zuardi et al., 2012b). The combination of THC and CBD has also shown to be effective in chronic pain management (Henson et al., 2022). It is also thought that nabiximols may provide therapeutic benefits for inflammation, digestive disorders and anxiety (Bilbao and Spanagel, 2022; Bridgeman and Abazia, 2017), although the formulation has not received marketing authorisation to treat these disorders. However, the precise mechanisms governing the interaction between CBD and THC remain unclear and are likely influenced by factors such as the compounds’ ratio and the timing of their administration and individual responses to both THC, CBD, metabolites and other non-phytocannabinoid species. Gaining further insight into the distinct central effects (endophenotypes) induced by CBD, THC or their combination in controlled settings will refine drug development strategies and deepen the understanding of their therapeutic potential (Bilbao and Spanagel, 2022; Turner et al., 2017).
Non-invasive brain imaging is increasingly used to capture drug effects on the brain in both clinical and preclinical settings (Khalili-Mahani et al., 2017; Reneman et al., 2021). Functional magnetic resonance imaging (fMRI) can reveal fluctuations in brain blood flow and oxygenation linked to drug-induced inhibitions and excitations within distinct neural circuits (Bifone and Gozzi, 2012; Borogovac and Asllani, 2012; Grade et al., 2015; Matthews and Jezzard, 2004; Zhang et al., 2018). Here, we used these imaging techniques – specifically, blood oxygenation level dependent (BOLD) contrast fMRI and arterial spin labeling (ASL) – to examine how two cannabinoids, CBD and THC, and their combination (nabiximols, THC:CBD in 1.08:1 ratio), alter brain connectivity and regional blood flow, respectively, in anaesthetised rats. Given the ethical and logistical challenges of drug testing in humans, preclinical pharmacological imaging provides a valuable means to identify functional connectivity (FC) signatures that can be compared to those of novel and putative therapeutic compounds in future studies.
Our approach builds on prior neuroimaging studies indicating that cannabis constituents like THC and CBD modulate brain activity (Grimm et al., 2018; Madularu et al., 2017; Mathew et al., 2002; van Hell et al., 2012; Volkow et al., 1996). We conducted whole-brain analyses along with targeted assessments of preselected regions, informed by clinical literature (Bossong et al., 2019; Volkow et al., 1996; Wall et al., 2019). By integrating multimodal analyses across scales, we aimed to establish comprehensive endophenotypes following acute administration of each intervention and model treatment-induced changes in FC.
Methods and materials
Please see Supplemental material for complete details of methods and analyses employed (Supplemental materials).
Animals
Experiments were approved by the UK Home Office under the Animal (Scientific Procedures) Act 1986 and the local KCL Animal Welfare Ethical Review Body. Forty-eight adult, male, Sprague Dawley rats (Charles River, UK; mean ± SD: 342 ± 37 g) were 10–12 weeks of age at the time of scanning. They were given free access to food and water and were otherwise kept under standard conditions for laboratory animals, including group-housing (4 per cage) under standard light (12:12, light:dark), temperature (21 ± 2°C), and humidity (55% ± 15%). Environmental enrichments were provided including nesting materials and chew sticks. Animal studies are reported in compliance with the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines (Kilkenny et al., 2010).
Drugs
Purified, botanically-derived THC and CBD were supplied by Jazz (formerly GW) Pharmaceuticals Research UK Ltd. (Cambridge, UK). All drugs were administered intraperitoneally (i.p.) at a volume of 1 mL/kg in the vehicle composed of 5%–10% ethanol, Kolliphor EL and saline (see Supplemental Methods) at following doses: THC, 10 mg/kg, CBD, 150 mg/kg, and THC:CBD mixture, 10.8:10 mg/kg. THC dose was selected on the basis of unpublished data (GW Pharmaceuticals), and additionally published data following a single acute dose (Long et al., 2010; Taffe et al., 2021); the CBD dose selected demonstrated efficacy in an epileptogenesis model (Patel et al., 2019), and the THC:CBD dose was based on efficacy in MS spasticity-relevant studies that used nabiximols (Sativex®) containing the same 1.08:1 mix of THC and CBD (Hilliard et al., 2012).
Experimental design
Rats were randomly assigned to four treatment groups with 12 animals per group. Four rats were excluded due to suboptimal image quality, with final groups and numbers as follows: (a) vehicle (n = 11), (b) THC (n = 10), (c) THC:CBD (n = 11) or CBD (n = 12). Drugs were administered to the conscious rats, 30 min prior to induction of anaesthesia. The groups were counterbalanced to account for order and time-of-day effects across conditions and drug treatments.
Rats were anaesthetised prior to scanning with isoflurane followed by medetomidine hydrochloride (‘Dormitor’, Orion Pharma) bolus (0.05 mg/kg, s.c) and infusion (0.1 mg/kg/h) (see Supplemental methods). Mild anaesthesia levels were maintained based on published protocols optimised to maintain physiological stability while retaining neural activity (Grandjean et al., 2014, 2023; Sirmpilatze et al., 2019).
The timing of BOLD and CBF scans was chosen based on an estimated Tmax of 2 h for THC and CBD (in-house GW data and (Deiana et al., 2012)). To this end, BOLD scan was started 108 ± 5.2 min after the drug dose, and CBF scan was started approximately 15 min later (See Figure 1).
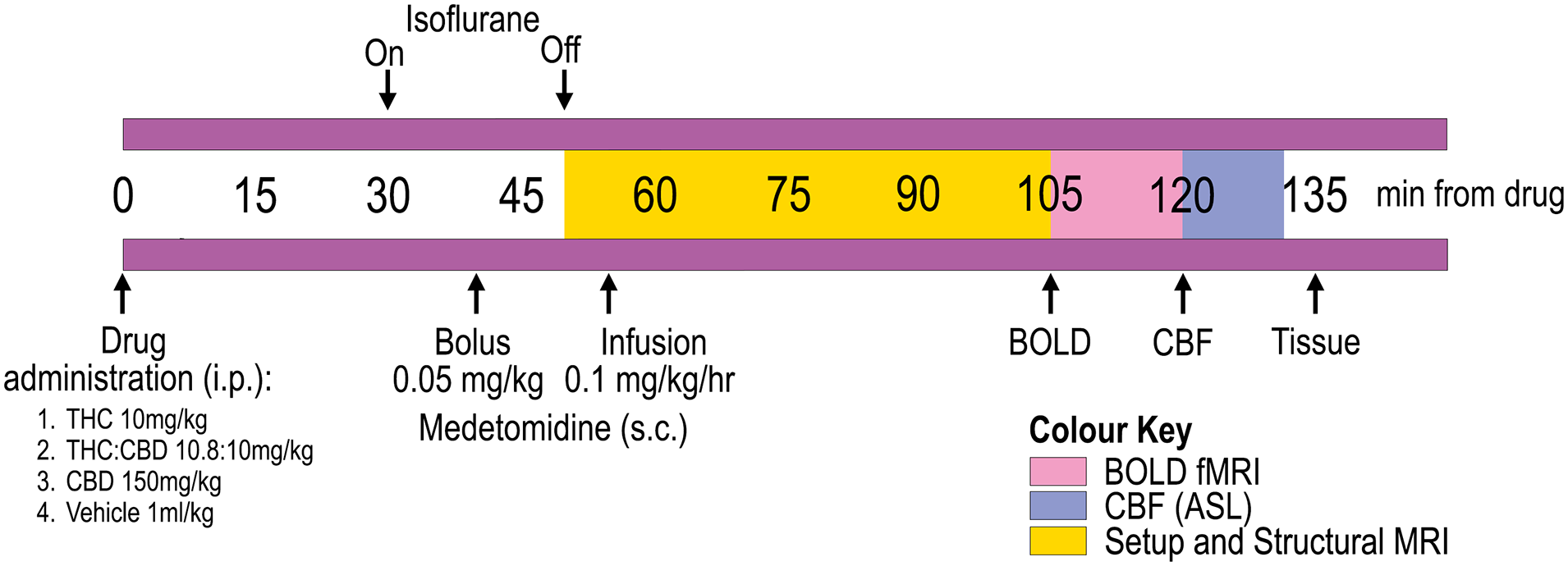
Experimental timeline: Drug or vehicle was administered to awake animals, which were then anaesthetised with isoflurane 30 min later. Approximately 10 min after anaesthesia induction, a medetomidine bolus was administered, followed by a gradual reduction of isoflurane over the next 10 min. Fifteen minutes after the bolus (i.e. 5 min after cessation of isoflurane), a continuous medetomidine infusion was initiated. MRI began with animal setup and structural scans, followed by BOLD fMRI acquisition approximately 105 min after drug administration and quantitative CBF measurement around 120 min post-administration.
Imaging acquisition
Data acquisition was performed on a 9.4 T Bruker small animal scanner, using an 86 mm diameter transmit volume coil and a rat brain 2 × 2 surface array receiver coil. T2-weighted Rapid Acquisition with Relaxation Enhancement (RARE) images were acquired as structural images to facilitate image registration. Two types of functional scans were acquired: resting state fMRI (rs fMRI) to measure BOLD contrast fluctuations, and Continuous arterial spin labelling (CASL) MRI to quantify CBF.
Rs fMRI data were acquired with a gradient-echo echo-planar imaging (EPI) using the following parameters: TE = 15 ms, TR = 1500 ms, 600 repetitions, field-of-view (FOV) = 24 × 19.2 mm, matrix = 60 × 48, partial-FT acceleration factor = 1.2 in phase-encoding direction, slice thickness = 0.6 mm, 36 contiguous slices, bandwidth = 300 kHz, scan time 15 min.
CASL data were acquired using a spin-echo EPI sequence with parameters: TE = 14.1 ms, TR = 4000 ms, 60 control/label image pairs, labelling time = 3000 ms, post-labelling delay = 300 ms, FOV = 25 mm × 25 mm, matrix = 100 × 100, partial-FT acceleration factor = 1.69 in phase-encoding direction, slice thickness = 1 mm, slice gap = 0.2 mm, 18 slices, bandwidth = 375 kHz, scan time 8 min.
Data pre-processing and outcomes measured
Cerebral blood flow (CBF)
The processing workflow for generating subject CBF maps encompassed standard steps for perfusion imaging (Alsop et al., 2010). CBF maps were registered to the atlas in a multi-stage ants Registration (Avants et al., 2011) call by combining a rigid transform to the respective RARE structural image, which in turn was normalised to the associated template image (Valdés-Hernández et al., 2011) via non-linear warping. The maps were analysed using voxel-based statistical parametric map comparison and a region of interest (ROI) analysis approach (Hawkins et al., 2018). A custom Matlab® application was used to calculate the mean CBF values (in mL/100 g/min) in 44 ROIs according to an in-house atlas (Supplemental Figure 1A) and within a whole brain mask. Further details of processing are given in Supplemental Methods.
Resting state BOLD fMRI imaging
The pre-processing workflow contained standard steps for resting state pre-processing pipelines (Mandino et al., 2019): each BOLD volume was subject to slice-timing and motion correction. The time-mean of the corrected image was non-linearly registered to a template (Valdés-Hernández et al., 2011) using antsRegistration before spatial smoothing with a 1 mm full-width at half maximum (FWHM) gaussian kernel and temporal filtering by simultaneously band-pass filtering between 0.01 and 0.1 Hz and regressing out nuisance variables, comprised of the six motion correction parameters, mean CSF signal and mean vascular signal.
The mean BOLD time-course from all voxels within a given atlas region was extracted from the pre-processed image, variance normalised and Fisher-transformed to Z-scores. A total of 16 regions from the atlas in Supplemental Figure 1B were selected for seed-based analysis, which were compared with statistical parametric mapping. Additionally, the mean time course from all 152 atlas regions (Supplemental Figure 1B) was subject to pairwise correlations to generate a weighted 152 × 152 graph of functionally connected brain regions per subject. These were analysed with methods including network-based statistics and calculation of graph theory metrics to describe the network structure.
Graph theory metrics were derived using MATLAB 2020a, and the Brain Connectivity Toolbox (Rubinov and Sporns, 2010). To minimise the influence of arbitrary threshold boundaries and spontaneous correlations, graphs were subject to proportional thresholding at a range of graph densities from 5% to 50% in 5% increments. Overall FC, global efficiency (GE) and average clustering coefficient (CC) were calculated for each subject at each graph density. The group mean values for each metric were integrated as a function of graph density, and the area under the curve (AUC) was calculated using trapezoidal numerical integration (Ginestet et al., 2011). Additional graphs were generated from bilateral regions as defined by the functional atlas (44 nodes, Supplemental Figure 1B; MacNicol, 2021). These regions were identified a priori for the default mode network (DMN), the salience network (SN) and the basal ganglia network (BGN), based on previous clinical literature that suggest changes in network connectivity following administration of cannabis (Batalla et al., 2020; Gunasekera et al., 2022; Volkow et al., 1996; Wall et al., 2019). The ROIs for DMN were dorsal and ventral hippocampus, and cortices (prefrontal, auditory, cingulate, parietal, retrosplenial and visual; Lu et al., 2012). The ROIs for SN were insula and cingulate (Tsai et al., 2020). The ROIs for BGN were striatum, motor cortex and cingulate cortex (Sierakowiak et al., 2015). Due to the drastically reduced number of edges per network graph compared to the whole brain graph, thresholding was not applied before calculating the graph theory metrics.
Tissue sampling
After scanning, the rats were deeply anaesthetised with 5% isoflurane in an oxygen/air mixture (1:9), then decapitated. Trunk blood and hemisected brains were collected. Blood samples were collected in heparin-lithium syringes (Protech Medical Ltd., UK), aliquoted into centrifuge tubes and centrifuged for 2 min at 10,000 rpm to separate the plasma, which was then processed along with the brain samples for bioanalysis.
Bioanalysis
The determination of CBD, THC and the metabolites: 7-COOH CBD, 7-OH CBD, 6-OH CBD, 11-COOH THC and 11-OH THC in rat plasma and brain samples was performed with liquid chromatography with tandem mass spectrometry (LC-MS/MS), using qualified methods. In summary, brain homogenate samples were prepared by homogenising brain tissue with 75% acetonitrile (aq) in the ratio of 1:3 w/v. Plasma and brain homogenate samples were then extracted using an SLE+ extraction plate prior to analysing the sample extracts on a Sciex QTrap API5500 (Applied Biosystems) mass spectrometer. Chromatographic separation of analytes was performed on an Agilent 1290 UHPLC system, using an ACE Ultracore SuperPhenylHexyl column (2.5 µm, 2.1 mm × 100 mm). Quantification of analytes, using peak-area ratios with deuterated internal standards to correct for extraction recovery and instrument response, was performed using AB Analyst™ software. Bioanalysis was performed at Pharmaron UK Ltd.
Statistical analysis
Sample size selection was based upon estimated Cohen’s d effect size of >1, for which a minimum of 10 rats per group were required for 90% power with 5% risk of false positives (using http://powerandsamplesize.com/Calculators/Compare-2-Means/2-Sample-Equality). All group comparisons are between each treated group and vehicle unless otherwise stated.
Univariate statistical analysis (i.e. analysis that does not require mass testing, such as comparison of correlation matrices or voxelwise analysis) was performed using GraphPad Prism v9.0.1. Data were tested for normality using d’Agostino-Pearson tests, and those following a normal distribution are presented as mean ± standard error (SEM), with p-values <0.05 considered statistically significant. For the analysis of network graph-derived metrics (Figure 4) and regional CBF in ROIs (Table 1, Figure 7), values that were more than 2 SD from the mean were excluded as outliers; if half of the ROIs for one subject were outliers, that subject was excluded. In this way, one rat was excluded as an outlier from both measures in the CBD group, and one from graph theory metrics in the vehicle group. Final group numbers for analysis were therefore vehicle (n = 10 graph theory, n = 11 CBF), THC (n = 10), THC:CBD (n = 11) and CBD (n = 11).
Cerebral blood flow values (mL/100 g/min) for all groups (mean, SD, n).
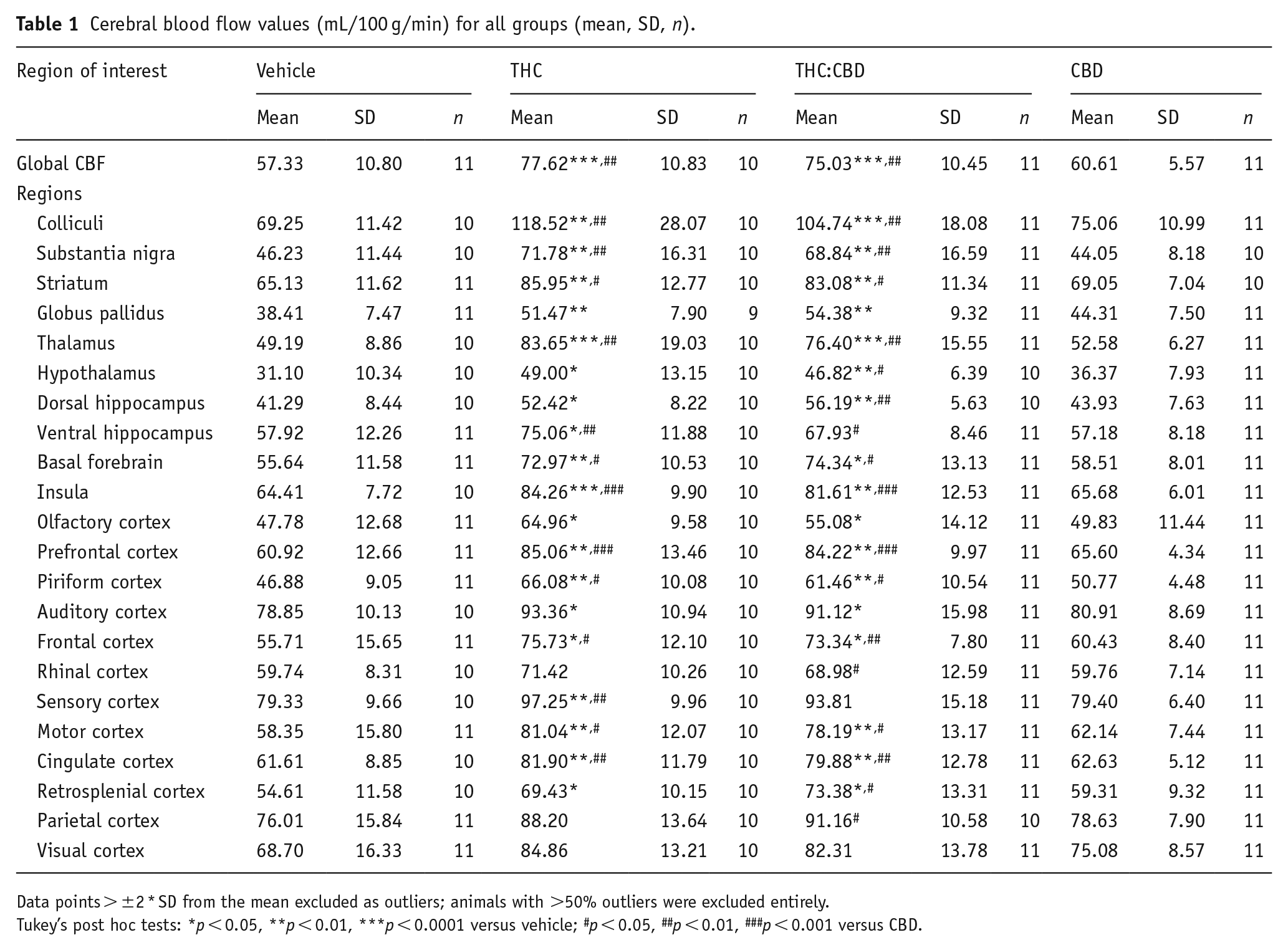
Data points > ±2 * SD from the mean excluded as outliers; animals with >50% outliers were excluded entirely.
Tukey’s post hoc tests: *p < 0.05, **p < 0.01, ***p < 0.0001 versus vehicle; #p < 0.05, ##p < 0.01, ###p < 0.001 versus CBD.
Regional CBF, and graph theory metrics (FC, GE and CC) for a priori networks were compared between drug and vehicle by two-way repeated measures analysis of variance (ANOVA). CBF data were analysed using both voxelwise and ROI-based analysis approaches (Figure 7). The voxelwise analysis informs localised changes that may be muted by averaging across parcellations, while ROI approach minimises noise and provides more statistical power.
For the whole brain graph-derived metrics (Figure 3), the difference in AUC was tested with a non-parametric permutation test, whereby the group labels were interchanged 5000 times to generate a null distribution of possible differences in AUC, against which the observed difference was compared. p-values were calculated as the count where the absolute value of the random distribution is equal to or greater to the absolute value of the observed difference, divided by the number of permutations.
Voxelwise between-group difference maps are displayed as coloured overlays on a greyscale rat brain template. A ‘dual coding’ approach is used, in which effect size (e.g. magnitude of group differences) is represented by colour – warm colours indicating increases relative to vehicle, cold colours indicating decreases – while statistical significance is conveyed by transparency. Voxels surpassing p < 0.2 are shown, with clusters at p < 0.001 outlined in black. This method allows the full extent of observed effects to be visualised, rather than obscured, while still highlighting the most statistically robust regions, in line with recommended best practices (Allen et al., 2012; Taylor et al., 2023).
Finally, a multivariate partial least squares analysis was conducted using in-house Python code to identify compound features that can discriminate between the groups across the CBF and FC (Figure 8, Table 2). In brief, CBF (44 regions and whole brain) and nodal strength derived from the same CBF parcellation were arranged as features for each subject (matrix Y). These were predicted by a matrix (X) of dummy variables encoding group identity for the four drugs. Three latent variables (LVs) were identified and tested for statistical significance by permuting the group identity matrix 10,000 times and developing a null hypothesis of the variance explained by each LV. The variable loadings of significant LVs (p < 0.05) were subject to bootstrapping with replacement. The resulting standard errors were used to normalise the loadings, creating bootstrap ratios that can be interpreted as pseudo Z-scores. The difference in scores for each LV was tested between drugs with non-parametric permutation testing and corrected for multiple comparisons using the Holm method (Holm, 1979).
PLS latent variable difference in mean group scores (Z).
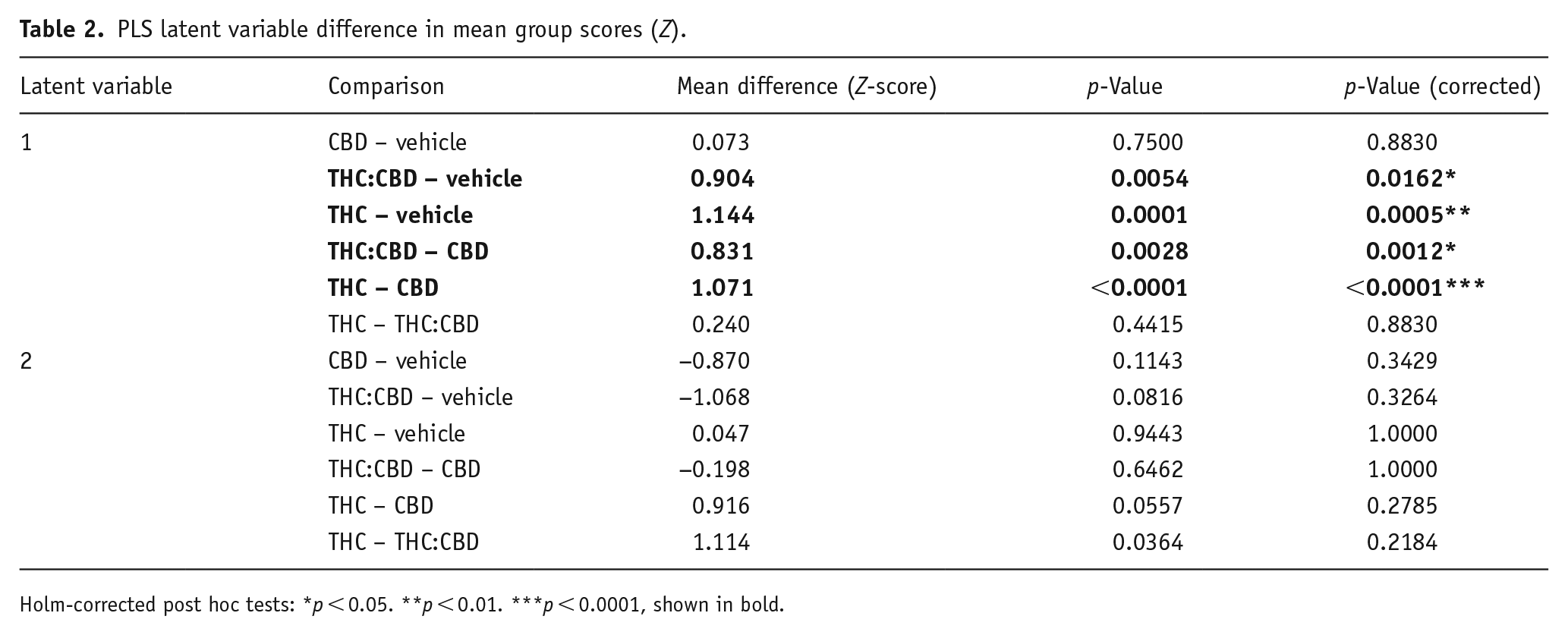
Holm-corrected post hoc tests: *p < 0.05. **p < 0.01. ***p < 0.0001, shown in bold.
Results
Cannabinoid induced brain FC endophenotypes
FC, as defined by correlation coefficients between regional time courses, describes the synchrony (or lack thereof) in activity fluctuations across many regions. Using ROIs from across the brain provides a global description of the intervention. Correlation coefficients from 152 brain-wide grey matter ROIs (76 per hemisphere; Supplemental Figure, S1A) were compared between the groups, and the resulting connectograms (Figure 2) represent the difference in region-to-region FC between drug treated groups and the vehicle treated group. THC was associated with many regions showing both higher covariance (stronger connections) and reduced covariance (weaker connections) relative to vehicle (Figure 2, left panel), which overall were significant (p < 0.01) according to the network-based statistics (NBS) analysis. Comparatively, CBD was associated with fewer connections that passed the statistical threshold (Figure 2, right panel), and there were no significant connections that surpassed correction. In between the two, THC:CBD showed more connections that differed from vehicle than CBD alone, but less than THC (Figure 2, middle panel); like CBD, THC:CBD had no significant connections that survived correction.
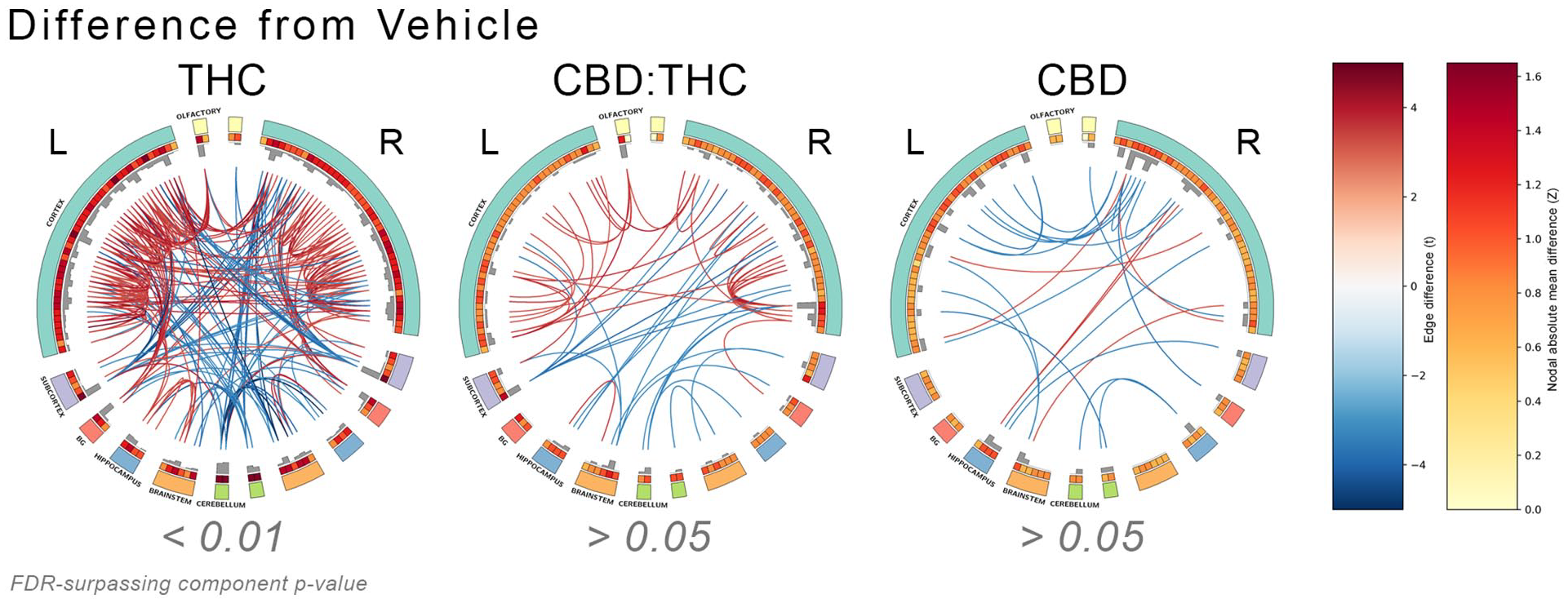
Connectograms of drug(s) versus vehicle. Connectograms depicting differences from the mean vehicle (n = 11) region–region functional connectivity strength for groups treated with THC (left; n = 10), CBD:THC (middle; n = 11) or CBD (right; n = 12). The lines represent difference in regional functional connectivity between pairs of ROIs, which are displayed around the circle; only connections which differed between the drug and the vehicle in a two-sample t-test by |t| > 3.2 are shown. Warm colours represent connections that are higher in drug compared to vehicle and cold colours denote the connections that are lower. The colours in the outer ring are arbitrary and differentiate anatomical parcellations, colours in the middle ring represent the mean difference for that node (darker colours have higher Z-values), and the height of grey histograms in the inner ring represents the number of connections that surpass the statistical threshold.
Divergent network properties of CBD, THC and THC:CBD
Graph theory was used to measure global network metrics using all 152 pairwise ROI correlations (Figure 3(b)). THC, relative to vehicle, resulted in an overall significant increase in FC and CC, with no changes in GE. CBD, relative to vehicle, resulted in an overall significant reduction in FC, CC and GE across multiple graph densities. While THC:CBD significantly increased FC compared to vehicle over multiple sparsities, the increase was smaller than that observed with THC alone (AUCTHC:CBD:17.87; AUCTHC: 20.44). Moreover, there was no significant difference in CC or GE between rats treated with THC:CBD and rats treated with vehicle.
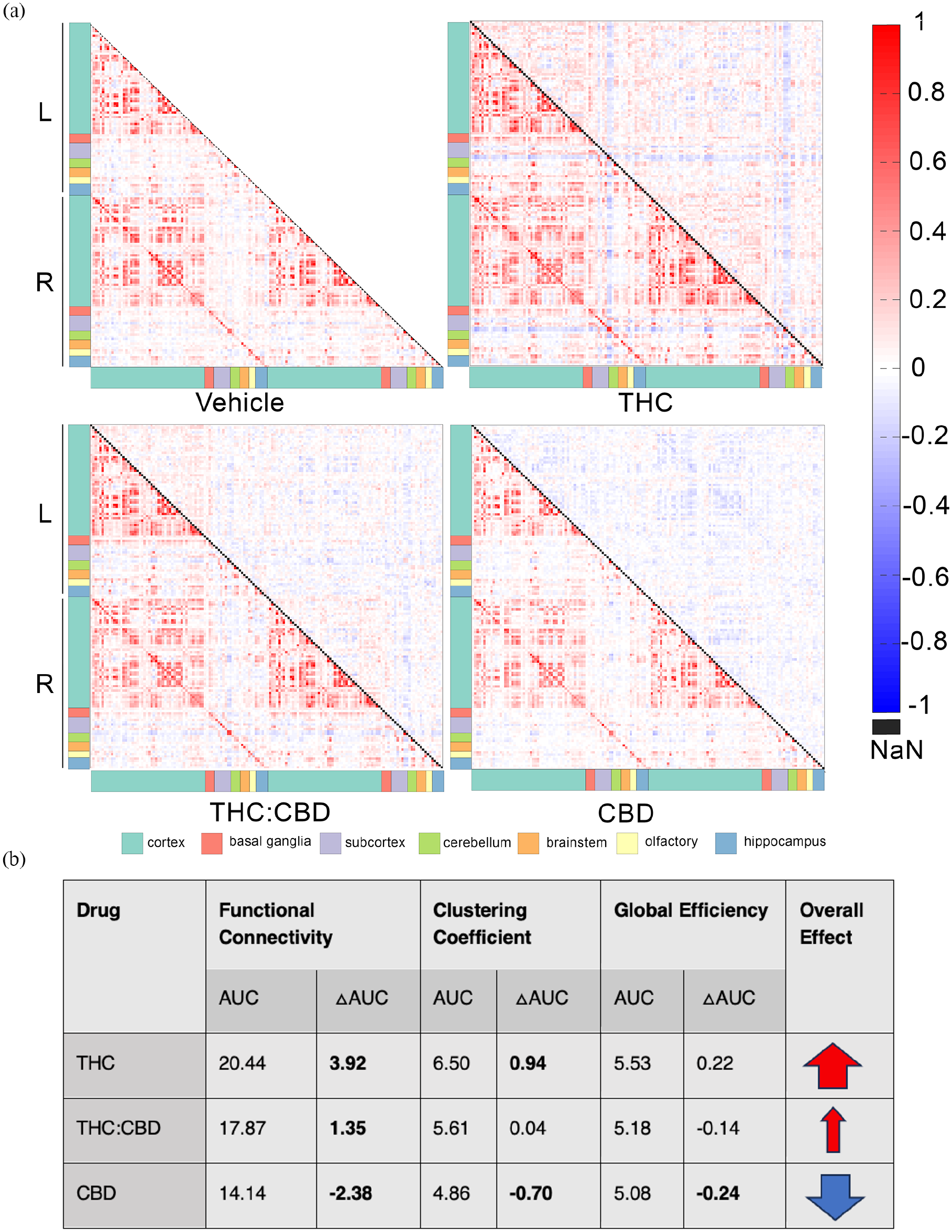
Graph theory analysis (a) Functional connectivity between 152 regions. Heat maps depict pairwise correlations between BOLD time courses from 152 ROIs (lower triangle) in rats treated with vehicle (n = 11), THC (n = 10), THC:CBD (n = 11) or CBD (n = 12) and the difference in correlation between drug and vehicle (upper triangle). The colours on the sides of the heat maps denote the following regions: red – cortex, purple – basal ganglia, yellow – subcortex, orange – cerebellum, pink – brainstem, green – olfactory bulb, light blue – hippocampus. (b) Summary of the effect of CBD, THC and THC:CBD, relative to vehicle, on global graph theory metrics measured as the AUC fitted from calculating the metrics across a range of graph sparsities. Metrics that surpassed p < 0.05 are highlighted in bold. Size of the arrow denotes the strength of changes, and red represents increase, blue represents decrease.
Furthermore, graph theory metrics were analysed within three a priori networks (Figure 4): the DMN, the SN and the BGN. Each metric underwent a two-way repeated measures ANOVA with network as a within-subject factor and drug as a between-subject factor. Significant effects of both drug and network were found for each metric, with FC and GE showing significant drug-network interaction effects. Tukey’s multiple comparison tests indicated significant increases in FC across all three networks due to THC. Additionally, THC increased GE in the BGN and SN, but not in the DMN, and enhanced the CC in the SN. No significant differences were observed between the THC:CBD combination and either THC or CBD alone in any metric. Lastly, a significant difference was found between CBD and THC: compared to THC, CBD resulted in lower FC and GE in all networks, and a lower CC in the DMN.
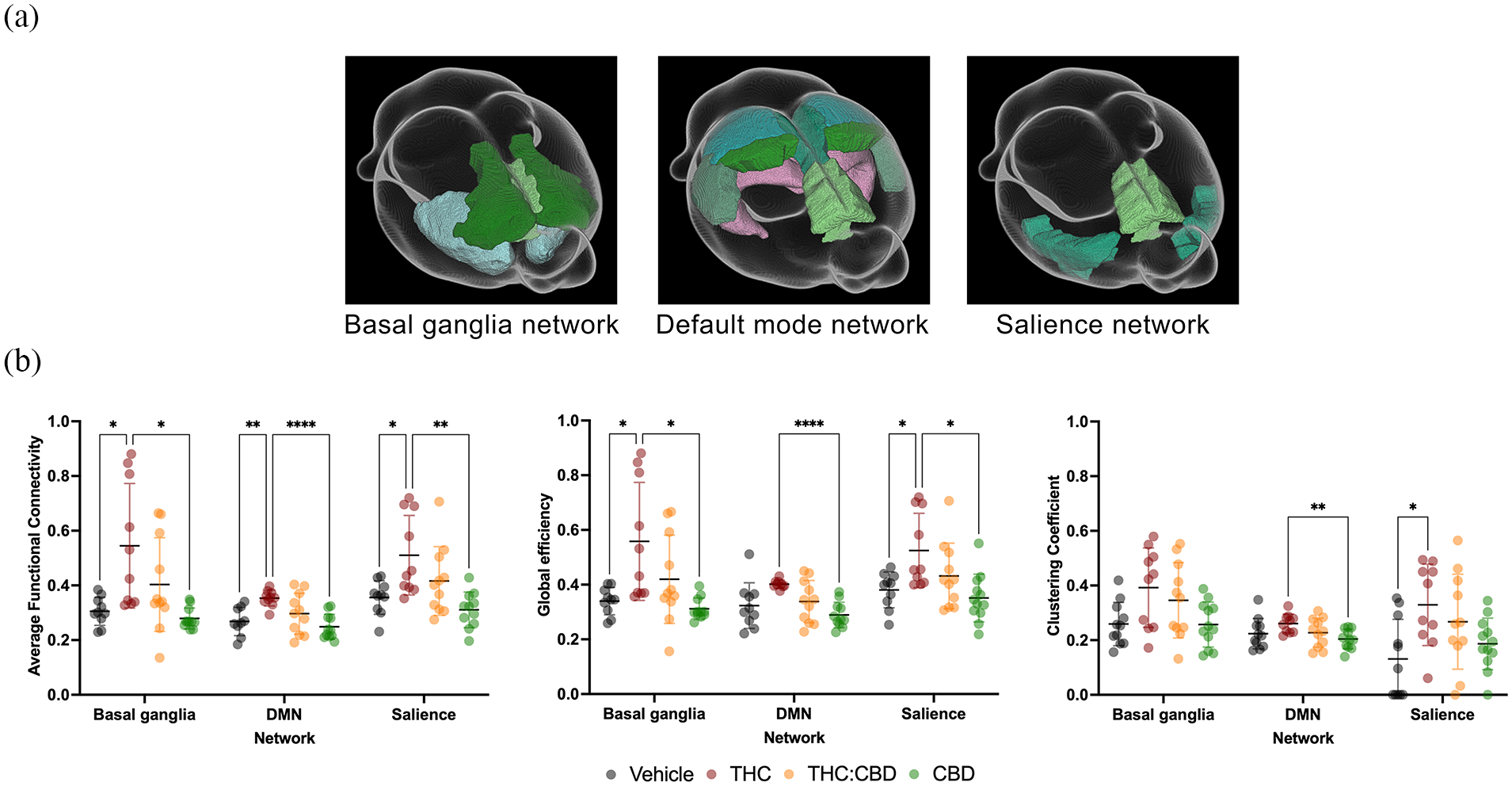
Graph theory metrics derived from a priori networks. (a) The atlas delineation of the nodes, derived from the atlas in S1, within a 3D glass brain to illustrate the spatial distribution of the three networks chosen for network-specific analyses. (b) Global graph theoretical metrics (average functional connectivity, global efficiency and clustering coefficient) derived from three a priori networks of interest. Each metric was subject to a two-way repeated measures ANOVA (network: within-subject, drug: between-subject). Drug (functional connectivity: F(3, 38) = 9.035, p < 0.0001; global efficiency: F(3, 39) = 6.79, p = 0.0009; clustering coefficient: F(3, 40) = 4.76, p = 0.0063) and network (functional connectivity: F(1.44, 54.01) = 23.24, p < 0.0001; global efficiency: F(1.501, 56.3) = 17.08, p < 0.0001, clustering coefficient: F(1.777, 70.20) = 14.61, p < 0.0001) had significant effects on each metric, while only clustering coefficient did not have a significant interaction effect (functional connectivity: F(6, 75) = 2.656, p = 0.0217; global efficiency: F(6, 75) = 2.439, p = 0.033; clustering coefficient: F(6, 79) = 2.156, p = 0.056). Tukey’s multiple comparison’s test was to compare between the groups, with p < 0.05, p < 0.01 and p < 0.0001 represented by *, ** and ****, respectively.
Impact of CBD, THC and THC:CBD on seed correlations of the resting-state BOLD-derived FC
Each subject’s reference mean BOLD time course was extracted from each seed (we chose 8 cortical and 6 subcortical ROIs) and regressed as a covariate of interest against all brain voxels generating subject-level correlation maps. The spatial distribution and magnitude of these maps were subsequently analysed for between-group differences. For clarity, only maps of three seeds, representing areas rich in cannabinoid receptors (cingulate cortex, hippocampus and dorsal striatum) are shown in Figure 5; whereas all 14 seed correlation results are shown in the Supplemental Materials (Figure S2).
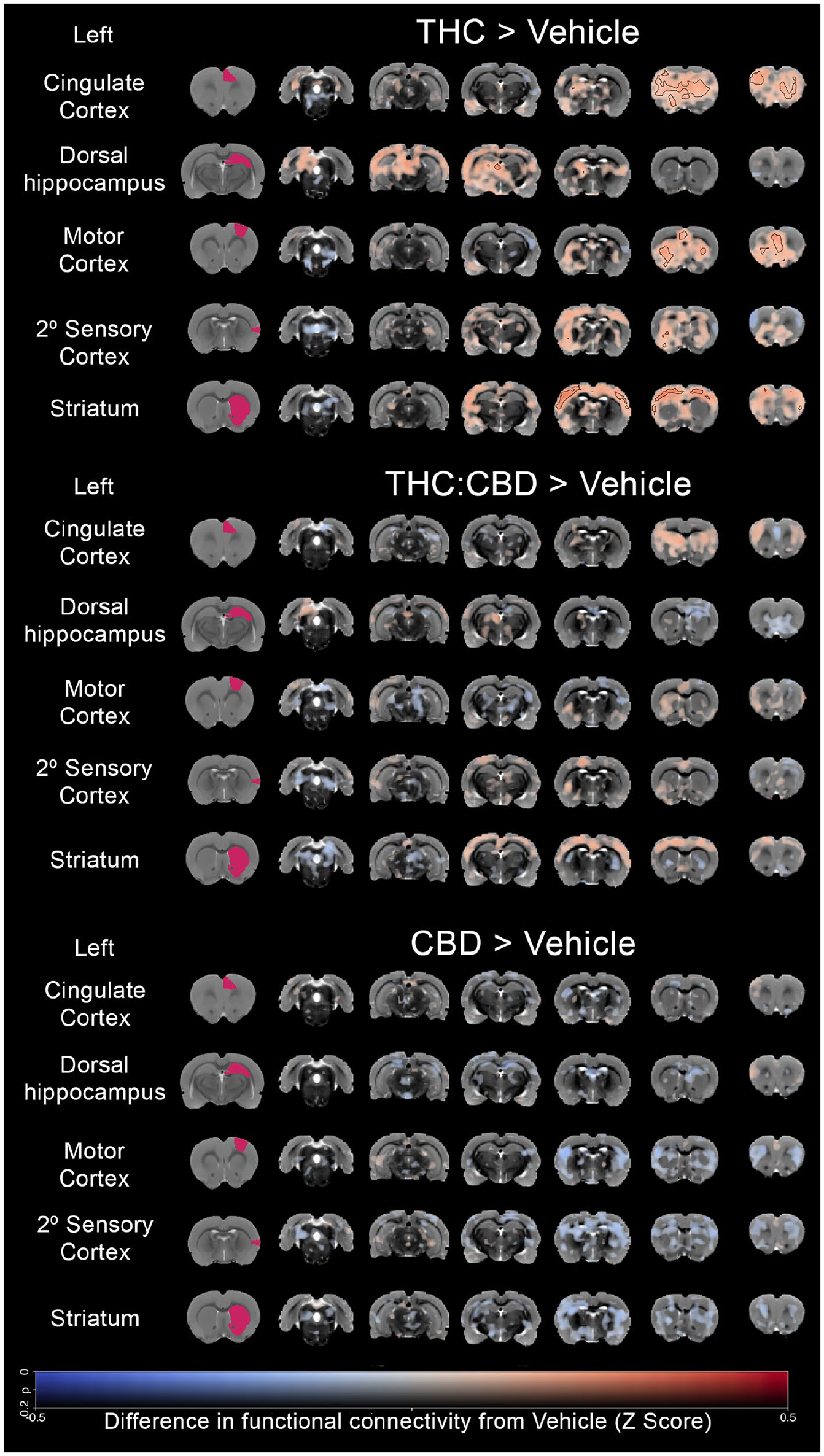
Seed analysis. Seed-based analysis maps derived from regressing the mean BOLD signal time course from a given seed region with each brain voxel, showing difference from vehicle (n = 11) Fisher-transformed correlation coefficients (Z-scores) in rats treated with THC (n = 10), THC:CBD (n = 11) or CBD (n = 12) with representative seeds located in left-sided regions of interest. The colour of the overlay represents the magnitude of the group differences (effect size), with red hues indicating areas where the correlation with the seed time course is higher in drug compared to vehicle and blue hues showing areas where correlation coefficient is lower. The overlay transparency represents the statistical significance from a two-tailed, two-sample t-test, given as p-values. p-values > 0.2 are fully transparent, while values closer to 0 are more opaque. Contours highlight voxel clusters that are above the statistical threshold (p < 0.001 uncorrected).
THC, relative to vehicle, resulted in a statistically significant increase in covariance from cortical ROIs to many regions across the brain (Figure 5; left column). Specifically, the cingulate seed had significant clusters in the prefrontal cortex and striatum (Figure 5; upper left column). When subcortical ROIs were used as seeds, THC administration relative to vehicle, resulted in statistically significant increases in connectivity between the striatum and the sensorimotor cortex (Figure 5; lower left column) and between the hippocampus and the thalami (Figure 5; centre left column).
On the contrary, CBD resulted in a general decrease in covariance relative to vehicle, but very few regions surpassed statistical significance (Figure 5; left column). The largest changes were seen when the striatum was the seed (Figure 5; lower left column), which occurred in the sensorimotor cortices, although they were not statistically significant.
The direction of changes relative to vehicle was mixed when rats are administered with THC:CBD, although these changes were almost entirely not significant (Figure 5; middle column). The only significant change occurred using the striatal seed (Figure 5; lower middle column): there are very small significant clusters, indicating increased covariance with the sensorimotor and cingulate cortex regions. The same region appears to show decreased covariance with the thalamus, compared to vehicle, but these non-significant results should be treated with caution.
The absolute strength of FC for each of the ROIs, normalised to vehicle, is shown for the three drug groups in Figure 6. The difference from vehicle illustrates the divergent effect of the three drugs in the brain, with overall strong increases in FC after THC, that are weaker after THC:CBD, and show predominant decreases with some increases in rats treated with CBD.
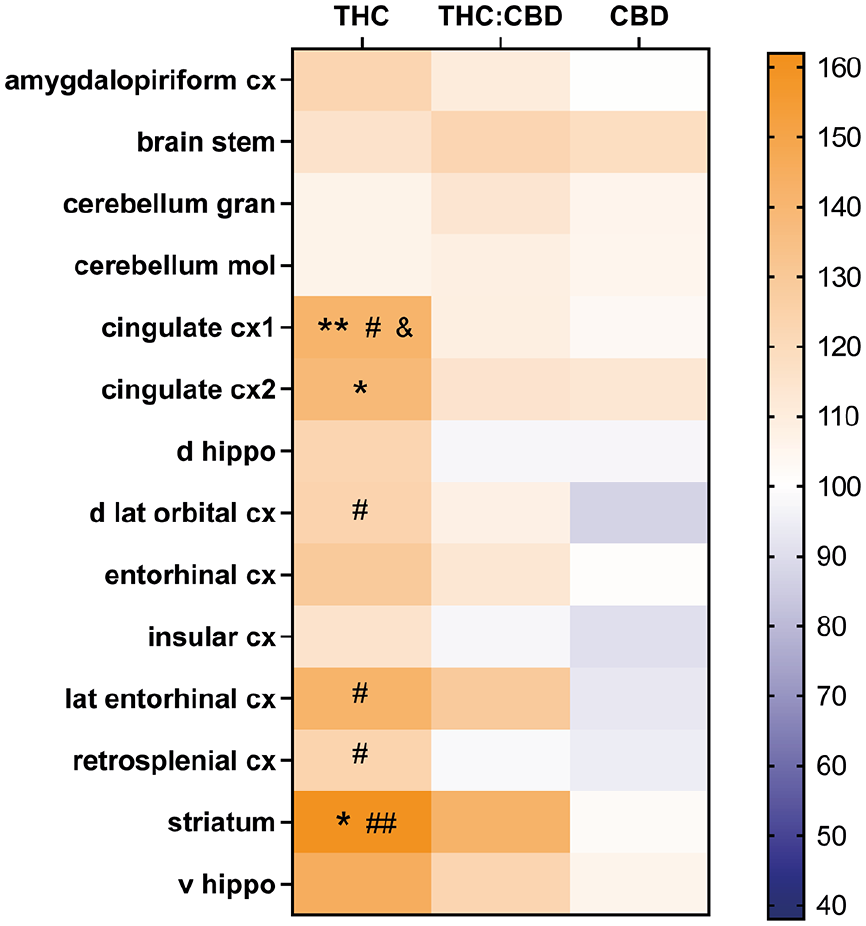
Mean FC in seeds functional connectivity strength of a priori selected ROIs expressed as mean % difference from vehicle, with orange colours indicating increases (>100) and blue decreases (<100). Mixed effects ANOVA was used for analysis of all four treatment groups with drug (Veh, THC, THC:CBD, CBD) as between-subject and ROI as within-subject factors. There were significant effects of drug treatment (F(3, 40) = 2.893, p = 0.047), ROI (F(95.906, 236.2) = 80.47, p < 0.0001) as well as drug x ROI interaction (F(45, 600) = 1.659, p = 0.0054). Tukey’s multiple comparison’s test compared the groups, where *p < 0.05, **p < 0.01 versus vehicle; #p < 0.05, ##p < 0.01 versus CBD; and &p < 0.05 versus THC:CBD.
Differential effects of THC, CBD and THC:CBD on regional CBF
CBF data were analysed using both voxelwise and ROI analysis approaches (Figure 7). CBF maps represent the mean regional CBF values from drug treated and vehicle treated groups (Figure 7(a)). Voxelwise analyses suggested that THC, relative to vehicle, resulted in significant increases in CBF in large clusters in the cortex, such as the insular and cingulate cortices, and in sub-cortical areas specifically the striatum, thalami, colliculi and brainstem (Figure 7(b)). These regions were similarly affected by THC:CBD, while there were no statistically significant differences between CBD and vehicle.
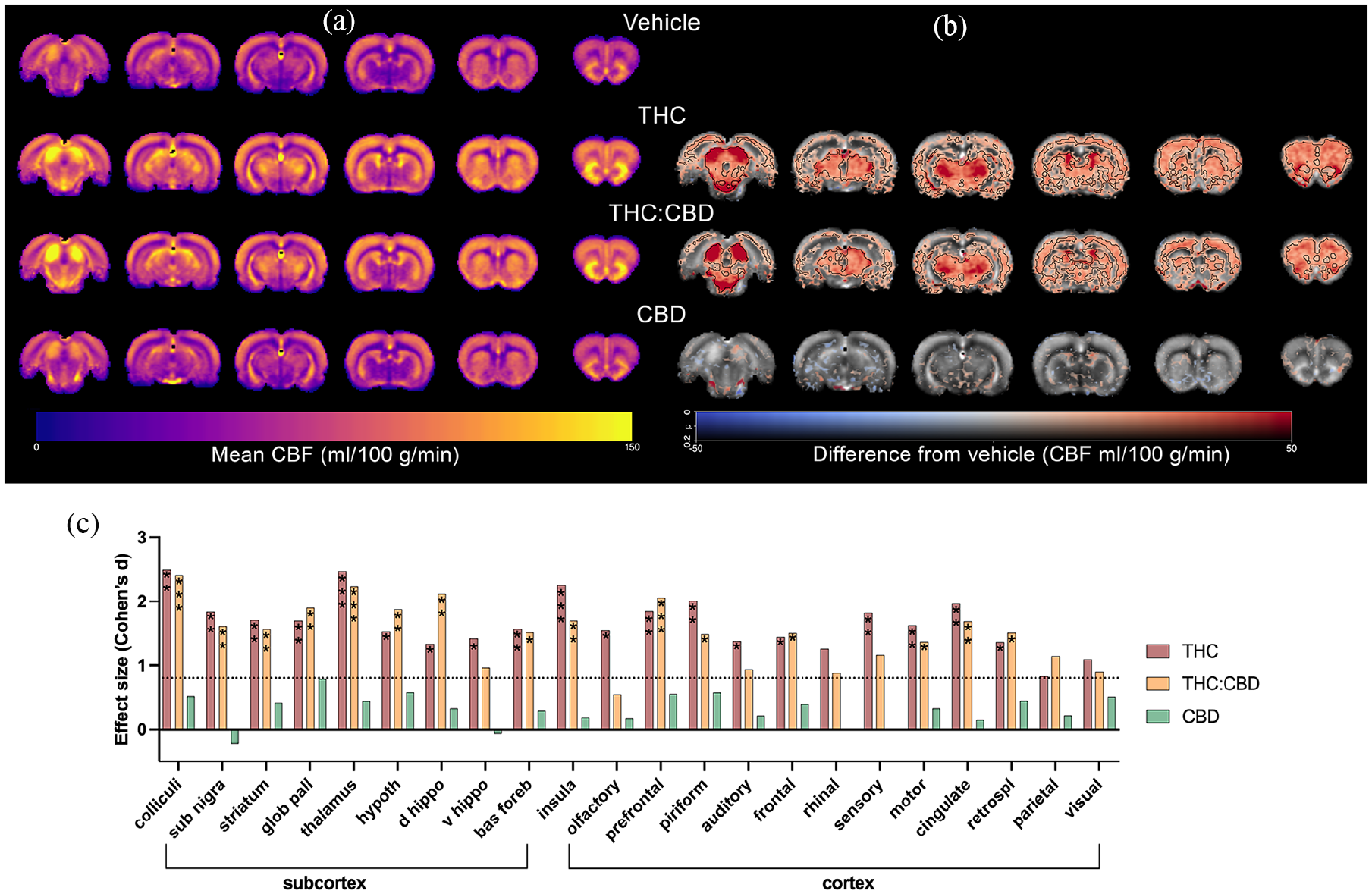
Cerebral blood flow (a) Voxelwise maps of mean CBF per group: vehicle (n = 11), THC (n = 10), THC:CBD (n = 11), CBD (n = 11). (b) Difference in mean CBF from vehicle for each of the groups. (c) Mean CBF across all voxels within selected ROIs, defined by an in-house atlas, was analysed using a two-way repeated measures ANOVA (factors: region as within-subject, drug as between-subject). Significant main effects were observed for drug (F(3, 39) = 12.77, p < 0.0001) and region (F(7.953, 304.1) = 144.6, p < 0.0001), with a significant interaction (F(63, 803) = 3.8, p < 0.001). Post hoc comparisons were performed using Tukey’s multiple comparison test. Bars in the figure represent effect size differences from the vehicle, with the dotted line at 0.8 indicating a large effect size. Stars indicate significance levels from post hoc comparisons to vehicle (*p < 0.05, **p < 0.01, ***p < 0.001).
Similarly, regional analyses (Table 1 and Figure 7(c)) showed both THC and THC:CBD resulted in significant increases in CBF relative to vehicle in specific regions of interest, namely, inferior colliculi, striatum, thalamus, insula, PFC and cingulate cortex. CBD, relative to vehicle, had no effect on regional CBF.
Differentiating vehicle, THC, CBD and THC:CBD with partial least squares analysis
Multivariate partial least squares analysis found two significant LVs (Figure 8). The first, LV1, explained 60% of the variance (p = 0.0006) while the second explained 27% of the variance in the data (p = 0.0208). The profiles of the LVs are shown in Figure 8(a). LV 1 had many features that surpassed a pseudo z-score threshold, |bootstrap ratio| > 1.96, and the majority of these were CBF features. LV 2, on the other hand, had fewer features surpass the threshold but the most were FC measures. The LV scores (Figure 8(b)) indicate how strongly each subject displays this LV profile. LV 1 could distinguish between all pairwise comparisons of drugs except CBD and vehicle, and THC and THC:CBD drugs (Table 2), and all comparisons surpassed correction for multiple tests. LV 2 could distinguish between some pairs of drugs, but no comparison was robust to multiple comparisons correction. Figure 8(c) displays a simplistic demonstration of how these LVs can be used as a two-dimensional discriminatory tool to differentiate and identify drug groups.
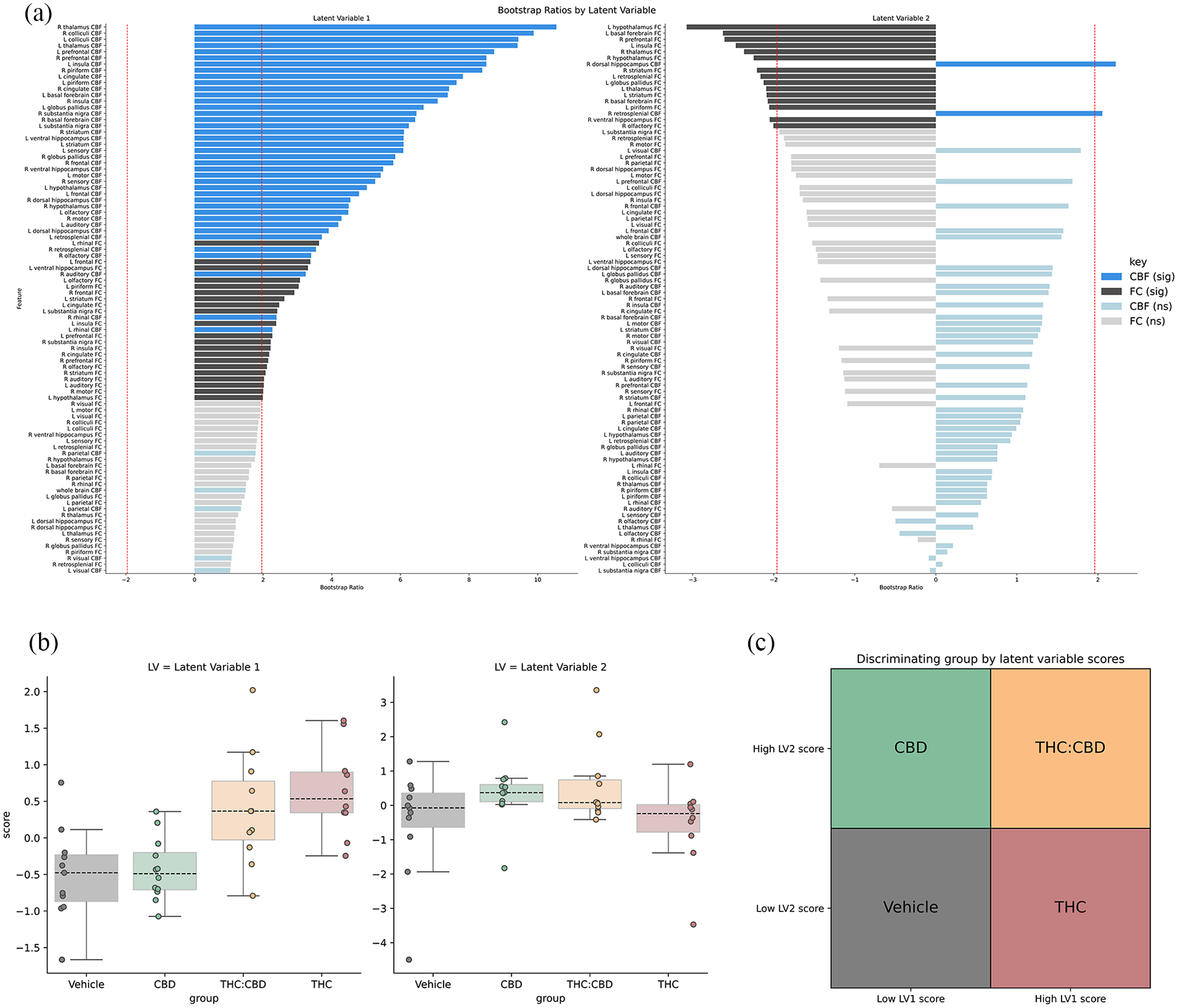
Partial least squares of CBF and FC (a) Features of each latent variable, organised by the absolute strength of the bootstrap ratio. A red dotted line indicates the significance threshold of |bootstrap ratio| > 1.96. Bars that surpass this line are coloured in dark hues, and bars that don’t are in lighter hues. Blue bars indicate CBF-derived features and grey bars indicate FC-derived features. (b) Latent variable scores per subject with respect to group: vehicle (n = 11), THC (n = 10), THC:CBD (n = 11), CBD (n = 11). (c) A simplified decision matrix indicating the discrimination of drug by LV score.
Total plasma and brain concentration of CBD, THC and metabolites
Total plasma and total brain concentrations of THC, CBD and metabolites following the administration THC, CBD and THC:CBD at approximately 4 h post-dose are shown in Supplemental Table 1. In THC treated rats, brain levels of THC and 11-OH THC metabolite were at higher levels than in the plasma showing a preferential partitioning into the brain compartment. The 11-COOH metabolite of CBD showed similar levels in the brain compared to plasma. Profiles and levels are consistent with unpublished in-house data.
In the CBD treated rats, brain levels of CBD and 6/7-OH metabolite were at similar or higher levels than the plasma showing ready partitioning into the brain compartment. The 7-COOH metabolite of CBD showed lower levels in the brain compared to plasma indicating it was not readily partitioning into brain compartment. Profiles and levels are consistent with unpublished in-house data.
In the THC:CBD treated group, the THC levels were higher in brain and plasma compared to THC alone dose group by around fourfold. It is noted that despite the higher levels of THC in the combination group, the brain:plasma ratio remained similar (around 4) to that observed for the THC only dose group. For the THC-related metabolites measured following administration of THC:CBD, 11-OH THC levels appeared to be similar to those measured from the THC alone group, whereas 11-COOH THC levels appeared to be lower. For CBD and metabolites, both brain and plasma levels observed in the combination group were dose proportional when compared to the CBD alone group.
Discussion
A combination of fMRI, to assess the brain FC, and ASL, to measure regional CBF, was used in rats to identify endophenotypic signatures of the acute pharmacological effects of THC, CBD, and their combination, THC:CBD. We began by applying global measures of whole-brain connectivity using network-based statistics and graph theory. This was followed by characterisation of connectivity within specific subnetworks and, finally, in several a priori selected regions of interest. CBF data were initially analysed independently to quantify blood flow in brain regions. Subsequently, we explored the covariance between BOLD fMRI and CBF using both linear regression and partial least squares analyses.
The difference in FC from vehicle, depicted as connectograms (Figure 2), illustrates the pronounced distinction between cannabinoid compounds. THC elicited the most changes, with approximately 1% of connections showing statistically significant alterations, involving both increases and decreases relative to the vehicle. Increases were primarily in the cortical areas, while decreases were observed in subcortical and posterior regions, including the cerebellum. The THC:CBD group had fewer connections surpassing the statistical threshold, and there were even fewer in the CBD group. Neither CBD nor THC:CBD showed overall network-level significant differences to vehicle when correcting for multiple comparisons using NBS.
Whole-brain changes were further probed using graph theoretical metrics (Figure 3(b)). To this end, THC significantly increased two out of three global measures: FC and CC, whereas GE remained unchanged. Increased FC suggests that the regions are more synchronised that is, correlated with respect to temporal activity, whereas increased CC reflects a higher level of segregation, which impacts on the efficiency of information processing (Sporns, 2018). Increases in FC and CC – both might in fact reflect a level of disorganisation (Hillary et al., 2015) which, coupled with increased perfusion (see below, Figure 7 and Table 1) could indicate higher overall load on the brain, consistent with the euphoric action of THC (Burggren et al., 2019).
In contrast, THC:CBD increased FC only, with GE and CC unchanged (Figure 3(b)). This finding corroborates the suggested tempering effect of CBD on THC, perhaps lowering its euphoric properties as previously suggested (Boggs et al., 2018; Britch et al., 2017; Gunasekera et al., 2022; Stella, 2023; Zuardi et al., 2012b). Interestingly, CBD alone significantly decreased all three global metrics (FC, GE and CC) – an effect that could be related to its proposed anxiolytic properties (Schouten et al., 2023).
Graph theory metrics were next calculated within smaller networks: the Default Mode (DMN), Salience and BGNs, which were selected a priori based on the proposed effects of cannabinoids on the brain (Batalla et al., 2020; Gunasekera et al., 2022; Figure 4). In agreement with the whole brain results, THC in comparison to vehicle significantly increased most metrics in all three networks, with the exception of CC in the BGN and DMN, and GE in the DMN. Conversely, neither THC:CBD nor CBD showed significant effects compared to the vehicle, although there were some significant differences between the CBD and the THC group, demonstrating the divergent effects of CBD and THC where CBD commonly shows the lowest mean value while THC often has the highest. Moreover, while no significant differences were found between THC and THC:CBD, the study was likely underpowered to show these, as the data demonstrate a trend suggesting that CBD attenuates the effects of THC.
Voxelwise ‘seed-based’ analyses (Figures 5, 6 and Supplemental Figure 2) evaluated correlations between the whole brain all-voxel signal versus time courses of signal from the specific a priori selected ‘seed’ regions. Cannabinoid receptor-rich areas (Batalla et al., 2020; Gunasekera et al., 2022; Stella, 2023) – the dorsal hippocampus, cingulate cortex, striatum and sensorimotor cortices) – showed divergence between the three compounds. THC caused the most pronounced changes throughout the brain, with significant increases in inter-cortical and cortical-hippocampal connectivity, and some notable decreases in seed-to-brain covariance in the posterior regions, notably the brain stem, and the cerebellum. The THC:CBD combination produced a similar, but less pronounced and more diffuse pattern, lacking the posterior decreases seen with THC alone. CBD alone showed no significant changes and only a few small magnitude, but widespread, regional covariance alterations.
Divergence between these compounds is further highlighted in the averaged regional FC differences between the drugs and the vehicle (Figure 6): connectivity was primarily increased with THC, negatively modulated by THC:CBD, and predominantly decreased with only a weak increase in some ROIs in the CBD group. This underscores that THC significantly altered overall brain connectivity and efficiency, and this was modulated by the presence of CBD. The effects of CBD alone were more subtle, pointing toward generally weakening connections across the brain.
Preclinical literature reveals a paucity of fMRI studies with cannabinoids that use clinically relevant methodologies similar to ours. Broadly agreeing with our results are two studies in rats that employed BOLD-sensitive fMRI and showed both BOLD signal increases and decreases with THC (Madularu et al., 2017), whereas CB1R agonist HU210 showed mostly increases (Shah et al., 2004). However, direct comparisons with these studies are limited by methodological considerations regarding the timing of drug administration and the dose(s) used, and importantly not measuring FC or blood flow as we have done. Moreover, their approach is limited to studies where a drug is administered acutely in the scanner, which affects clinical translatability.
The existing clinical (human) literature on imaging and cannabinoids is highly heterogeneous with respect to methods, subjects and paradigms (Cupo et al., 2021; Gunasekera et al., 2022). Most human studies focus on stimulus- or task-related fMRI, unlike our focus on a stimulus-free paradigm. While these studies report mixed effects of THC and CBD on task-related activations, they are not easily generalisable to resting-state fMRI studies performed in anesthetised animals. Moreover, many are conducted in (chronic) cannabis users, who may also use other substances, complicating the interpretation of fMRI results due to observed baseline perfusion alterations as a result of chronic substance use (Filbey et al., 2018), and therefore potential receptor changes as well as test substance heterogeneity.
There are variable results amongst clinical studies that used acute THC or cannabis in the naïve or washed out subjects to measure resting-state BOLD signal fluctuations. Van Hell et al. (2012) and Klumpers et al. (2012) reported resting signal increases, aligning with our results of increased FC. Conversely, Ramaekers et al. (2016) and Mason et al. (2019) both found decreased connectivity between nucleus accumbens and the cortical regions (but did not examine any other areas), Bossong et al. (2019) detected decreases in one out of four regions examined, whereas Grimm et al. (2018) reported no acute effect of THC, possibly due to too low plasma exposure (ca. 0.4 ng/mL compared to typical values of 15–30 ng/mL (Lawn et al., 2023)). Wall et al. (2019, 2022), like us, demonstrated that the effects of THC-rich cannabis are attenuated in a CBD-rich THC + CBD combination; however, they observed THC-induced decreases in several networks, and not increases. Comparisons with and between such studies are difficult as the majority are conducted with highly selective a priori hypotheses and restricted to very few regions or networks. Where whole-brain dynamic FC was assessed (Zaytseva et al., 2019), both increases and decreases were seen in the brain connectome after cannabis in occasional users, with a particular brain state of hyperconnectivity being associated with an intoxication element of the drug, echoing our results of increased connectivity. Although we did not measure chronic effects of the drugs, it is interesting to note that in chronic cannabis users, similar hyperconnectivity of the brain was reported (Ramaekers et al., 2022; Vergara et al., 2018).
In addition to rs-fMRI, we measured cerebral perfusion using ASL, potentially enhancing the translational relevance of our findings (Jonckers et al., 2015; Khalili-Mahani et al., 2017). ASL provides a non-invasive, quantitative assessment of brain perfusion, specifically regional CBF (Wang et al., 2011). Given the direct relationship between CBF and neural firing, CBF is often used as a proxy for changes in neural activity (Attwell and Iadecola, 2002; Hosford and Gourine, 2019; Paulson et al., 2010). Our results show that both THC and the THC:CBD combination increased CBF in cortical and subcortical areas across most regions analysed, while CBD alone did not affect CBF (Figure 7). In contrast to rs-fMRI findings, where CBD modulated THC’s effects, the addition of CBD did not alter THC’s impact on CBF. Additionally, since the dose of CBD in the ‘CBD alone’ condition was higher than in the THC:CBD mixture (150 vs 10 mg/kg, respectively), it remains unclear what effect, if any, CBD at 10 mg/kg would have on CBF. These CBF data support that THC markedly influences brain perfusion, while CBD alone does not impact brain perfusion, and it does not appear to modify THC’s perfusion effects when combined.
The clinical literature generally aligns with findings on cannabinoid effects on cerebral perfusion. THC is notably associated with increased CBF, especially in the frontal and cortical regions (Batalla et al., 2014; O’Leary et al., 2002; Ogunbiyi et al., 2020; Richter et al., 2018). Similarly, animal studies have observed increases in cerebral blood volume, a metric related to CBF, following the administration of CB1R agonists (Chin et al., 2008; Yao et al., 2009). However, no quantitative ASL studies have measured CBF after cannabinoid administration in rodents to date. Interestingly, several studies using an older technique, in vivo 14C-iodoantipyrine autoradiography, reported decreases in CBF following THC (Bloom et al., 1997; Goldman et al., 1975), or anandamide, a CB1/2R agonist (Stein et al., 1998). The discrepancies between MR-based findings and these early autoradiography results remain unclear, likely due to methodological differences.
The effects of CBD on CBF in humans appear smaller and more varied, likely due to its proposed polypharmacological properties. Some studies report CBD-induced increases or decreases in CBF within hippocampal regions, while others show a normalisation of elevated hippocampal CBF in patients at risk of psychosis (Bloomfield et al., 2020; Crippa et al., 2004; Davies et al., 2023), underscoring the subtler impact of CBD compared to THC. CBD’s effects are often most pronounced when a pre-existing deficit is present, as seen in patient populations or animal disease models (Batalla et al., 2020; Davies et al., 2023; Gunasekera et al., 2022). This suggests that CBD’s effects may be challenging to detect in healthy individuals, whether human or animal.
When considering the effect of cannabinoids on brain perfusion, it is important to note that THC can directly affect the vasculature through vasodilation via activation of CB1 receptors on endothelium (Ho and Kelly, 2017; Morse et al., 2023; Richter et al., 2018). CBD does not cause vasodilation or increased brain perfusion (Crippa et al., 2004). Therefore, it is possible that at least some of the effects of THC and THC:CBD on cerebral perfusion may be driven by a direct effect of THC on the vasculature. Disentangling the relative contributions of cannabinoid-induced vascular versus neural-driven CBF changes is challenging (Batalla et al., 2014). While THC and THC:CBD similarly impact CBF, their global and regional effects on fMRI patterns differ. CBD administration decreases regional brain connectivity without significantly affecting CBF. Thus, it is unlikely that the observed global connectivity changes are primarily dictated by CBF impacts, though interactions between drug-induced CBF changes and brain connectivity cannot be entirely ruled out.
To further characterise brain activity patterns and endophenotypes associated with each compound, we leveraged the within-subject design to probe FC and CBF data acquired from the same subject within the same session. Although FC and CBF are known to be associated and moderately correlated to each other, they reflect distinct physiological processes (Attwell and Iadecola, 2002; Liang et al., 2013a). CBF primarily reflects local neural activity, with highly active ‘hub’ regions – such as those in the DMN or auditory structures like the inferior colliculus – displaying greater perfusion. In contrast, FC captures temporal synchrony between regions, which may not correspond to the magnitude of activity within individual regions; a region can be highly active but weakly connected to others, or vice versa. Furthermore, physiological changes induced by factors such as illness, ageing or drugs can modulate the coupling between FC and CBF (Galiano et al., 2020; Qiu et al., 2017; Zhu et al., 2017).
We first confirmed a linear relationship between nodal FC strength and mean CBF in rats, consistent with findings from human studies (Liang et al., 2013a; Supplemental Figure 3). While a slight reduction in correlation coefficient was qualitatively observed in the THC group, differences in the relationship were not formally tested across groups due to a predicted lack of power to detect small differences. Overall, the correlation between FC and CBF across regions appeared preserved in all groups despite anaesthesia.
Instead, we utilised the within-subject design to perform a multivariate PLS analysis (McIntosh and Misic, 2013), incorporating FC and CBF data from all brain regions and subjects (Figure 8) to explore how these measures jointly vary with drug treatment. This revealed two LV1 and LV2 with distinct feature loadings: LV1 was primarily driven by CBF, and LV2 by FC. These latent patterns more effectively distinguished the treatment groups. THC showed strong positive LV1 scores, followed by THC:CBD, whereas LV2 best distinguished CBD from both THC and vehicle. Each drug group displayed a unique LV1/LV2 profile, suggesting a unique neural signature that could inform the classification of novel compounds. Notably, the strong contribution of CBF to THC’s LV1 profile is consistent with its robust regional perfusion effects (Figure 7), while CBD’s elevated LV2 scores reflect its overall decrease of FC (Figures 3 and 6).
Considering the tight coupling between cerebral metabolism and blood flow, it is also relevant to evaluate studies that have measured glucose consumption in the brain following cannabinoid administration. An animal study using 18F-fluorodeoxyglucose Positron Emission Tomography (FDG PET) with a cannabinoid agonist indicated increased metabolism (Nguyen et al., 2012), which aligns with our findings of elevated CBF. However, several earlier studies using 14C-2-deoxyglucose (2DG) autoradiography in rats showed metabolic decreases at the doses of THC we used (Freedland et al., 2002; Margulies and Hammer, 1991; Pontieri et al., 1999; Whitlow et al., 2002). These findings not only contradict our results of increased CBF but also contradict similar findings of increased brain perfusion in humans. Reasons for these discrepancies are unclear and may include factors such as anaesthesia, animal restraint (used in 2DG method) or analytical methodologies.
We also quantified CBD, THC and their metabolites in the animals in our study, and the specific concentrations are summarised in Table S1. Comparing doses of cannabinoids between animals and humans is complex due to logistical and metabolic reasons: human administrations of cannabinoids are typically via inhalation, although sometimes oral, and less frequently injection, whereas animals are usually injected. For instance, in human applications, typical plasma exposures approximately 20 minutes after vapour-based administration are around 15–30 ng/mL of THC, or 40 ng/mL of CBD (Lawn et al., 2023). In our study, median plasma concentrations in rats, measured approximately 4 h after administration of THC:CBD, ranged from 40 to 175 ng/mL for THC (depending on whether it was administered alone or combined with CBD), and approximately 100–1530 ng/mL for CBD depending on dose (Table S1). Considering the difference in timing (4 h in rats vs 20 min in humans) and an estimated conversion factor of 4–6 between human and rodent doses due to differences in metabolic rate (Nair and Jacob, 2016), these plasma concentrations appear to be comparable between both species although this would have to be verified by the appropriate drug metabolism and pharmacokinetic analysis that was outside the scope of this study.
In line with previous clinical (Lawn et al., 2023) and preclinical (Hlozek et al., 2017) studies, our study found that co-administration of CBD with THC led to a significant (approximately 4×) increase in THC concentration in both brain and plasma, compared to similar doses of THC alone. This effect might be attributed to the known impact of CBD on THC’s P450 enzymatic degradation in the liver (Smith and Gruber, 2022). Contrary to Hlozek et al. (2017), in our study, levels of CBD in the brain and plasma did not appear to be influenced by THC co-administration, when corrected for dosage difference (150 vs 10 mg/kg; Table S1). Additionally, CBD may influence THC metabolism in other ways, such as modulation of the ATP-binding cassette (ABC) transporter p-glycoprotein, which mediates THC efflux across the blood-brain barrier (Stella, 2023). If THC efflux is reduced, its effect could be extended and/or mitigated; or via CB1R – TRPV1 cross-talk (Chen et al., 2016), which could create regional interaction patterns between THC and CBD according to the distribution of both receptors (Britch et al., 2017). This interaction is complex and its unravelling is beyond the scope of this study. Nevertheless, our findings reinforce the hypothesis that the effect of co-administering CBD and THC is more substantial than the sum of their individual impacts, underscoring the need for further exploration into THC:CBD’s effects on the brain. Additionally, we observed a small amount of THC in the brains (but not plasma) of rats administered CBD alone, as also found by (Hlozek et al., 2017), which is highly likely due to the extremely low concentrations of THC being present in the CBD botanical formulation.
Our findings have implications for stratifying therapeutic indications in neurological and neuropsychiatric disorders, even though we used healthy animals rather than a disease model. Cannabinoids, especially CBD, have demonstrated clinical efficiency and received marketing authorisation for the treatment of rare epilepsy disorders including Lennox–Gastaut syndrome, Dravet syndrome, and Tuberous Sclerosis Complex (Devinsky et al., 2017, 2018; Miller et al., 2020; Thiele et al., 2018, 2021). Preclinical studies suggest increased brain connectivity in the models of epilepsy (Gill et al., 2017; Otte et al., 2012), in particular higher clustering and GE (Carboni et al., 2020; Richter et al., 2018), though weaker network connections have also been reported (Christiaen et al., 2019). Our finding that acute CBD reduces these metrics aligns with CBD’s use in epilepsy (Klein et al., 2017).
In clinical applications, THC is suggested to be effective in managing pain, particularly chronic pain (Stella, 2023; van de Donk et al., 2019). Key brain areas involved in modulating such pain include the prefrontal and cingulate cortex (Stella, 2023). In our study, THC significantly modulated FC in the cingulate cortex and, to some extent, in the orbital cortex, a part of the prefrontal cortex. Additionally, individuals with chronic pain exhibit altered activity within the SN and DMN (van Ettinger-Veenstra et al., 2019), both of which were also affected by THC in our study. However, THC also induces acute psychotic symptoms, which might exacerbate symptoms of schizophrenia, and potentially lead to addictions (Stella, 2023), all of which have been linked to aberrant processing within the SN and DMN (Bolton et al., 2020). The cingulate cortex and prefrontal cortex mediate many reward and emotional circuits in the brain – highly relevant to the experience and processing of both pain and psychosis. Therefore, the acute effect of THC we report may reveal alterations in the neural substrates of aberrant salience processing, reward mechanisms and pain pathways.
There are general methodological considerations that may have influenced the results of this study. Specifically, anaesthesia presents a confound for all rodent pharmacological fMRI studies unless conducted in awake animals. The majority of rodent fMRI studies use anaesthesia, which inevitably affects FC – it is used to reduce stress and immobilise the animals while scanning, since motion degrades image quality (Liang et al., 2012; Mandino et al., 2024). To minimise the impact of anaesthetics, we used a protocol that is associated with robust and stable BOLD responses as well as synchronised activity networks (Grandjean et al., 2014, 2020, 2023); You et al., 2021). Moreover, we included an appropriate vehicle group in the study design; therefore, it is less likely that the effects we see on FC are driven by the anaesthesia protocol used. In fact, our results are in line with another preclinical study investigating the effects of THC on BOLD response in awake rats (Madularu et al., 2017).
However, a particular concern regarding anaesthesia is the reported interaction between cannabinoids, particularly via CB1Rs, and noradrenaline signalling (Cathel et al., 2014). This interaction may involve α2-adrenergic receptors, which are also the primary site of action of medetomidine (Sinclair, 2003), the anaesthetic used in this study. We cannot dismiss a potential interaction between medetomidine and the cannabinoids administered, especially in the THC alone group (Tapley and Kellett, 2019). To conclusively address this interaction, future studies could compare different anaesthetic agents or perform the experiment in conscious animals. However, the former is complicated by the broad pharmacological profile of cannabinoids, which can interact with various systems implicated in anaesthesia – such as opioid, glutamatergic, or GABAergic pathways – potentially confounding results with alternative anaesthetics. The latter is made challenging by the well-documented influence of cannabinoids and endocannabinoids on stress-related processes (Navarrete et al., 2020), which are particularly relevant in conscious rodent fMRI, where stress effects are difficult to eliminate despite careful experimental control (Mandino et al., 2024).
Another limitation is that we only used male animals to minimise physiological variability and to keep the groups manageable as the inclusion of both sexes would have required larger groups. As thus far most cannabinoid research has been conducted in male animals, the inclusion of both sexes in future studies is recommended.
In summary, we have demonstrated that acute THC administration resulted in increases in FC and regional CBF, acute CBD administration resulted in an overall reduction in FC with negligible effect on CBF, and the combination drug THC:CBD resulted in effects similar to, but lower than THC alone. Our application of functional neuroimaging has thus identified differential pharmacodynamic signatures for THC and CBD in anaesthetised adult male rats. Further work should encompass an investigation of the effects of sub-chronic administration of phytocannabinoids on brain activity in animal models with relevance to selected disease indications to investigate changes on FC in a perturbed system, more applicable to the disease state. Overall, this neuroimaging work supports the role of preclinical fMRI to generate functional neuroimaging signatures as endophenotypes that could help inform indication selection as well as potential site(s) of action in drug discovery and development (Carmichael et al., 2018).
Supplemental Material
sj-pdf-1-jop-10.1177_02698811251360745 – Supplemental material for Acute cannabidiol (CBD), tetrahydrocannabinol (THC) and their mixture (THC:CBD) exert differential effects on brain activity and blood flow in rats: A translational neuroimaging study
Supplemental material, sj-pdf-1-jop-10.1177_02698811251360745 for Acute cannabidiol (CBD), tetrahydrocannabinol (THC) and their mixture (THC:CBD) exert differential effects on brain activity and blood flow in rats: A translational neuroimaging study by Eilidh MacNicol, Michelle Kokkinou, Maria Elisa Serrano Navacerrada, Donna-Michelle Smith, Jennifer Li, Camilla Simmons, Eugene Kim, Michel Mesquita, Loreto Rojo Gonzalez, Tierney Andrews, Sally Loomis, Royston A Gray, Volker Knappertz, Benjamin J Whalley, Andrew C McCreary, Steven CR Williams, David Virley and Diana Cash in Journal of Psychopharmacology
Footnotes
Acknowledgements
We would like to thank Jim Taylor for his help with assessing the BioAnalytical data.
Data availability statement
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: At the time of the study MK, D-MS, JL, SL, RAG, VK, BJW, ACM and DV worked for and held shares in GW/Jazz. The remaining authors have no conflicts of interest to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a grant from Jazz Pharmaceuticals to DC and SCRW.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
