Abstract
Background:
Natural treatments may be used as an alternative or adjunct treatment for childhood neuropsychiatric disorders. Knowledge of benefits and harms is needed to inform use guidelines. We aimed to systematically identify how and which adverse events are monitored, assessed, and reported in pediatric trials that tested nutraceutical and phytoceutical treatments.
Methods:
We searched MEDLINE, Embase, PsycINFO, ProQuest Dissertations and Theses Global, Cochrane Library, and Google Scholar from 2012 to 2024. Eligible studies included nutraceutical and phytoceutical trials, experimental or quasi-experimental in design, involving children or adolescents (age 4–19 years) with neuropsychiatric conditions.
Results:
Ninety-eight trials were included with 75 reported as completed (77%). The most common natural treatment tested was polyunsaturated fatty acids (36%, 35/98). Most trials focused on treating attention-deficit/hyperactivity disorder (59%, 58/98) or autism spectrum disorder (21%, 21/98). Investigators from 74/98 trials (76%) reported methods that indicated adverse event monitoring. For these trials, events defined a priori for monitoring were identified in 43% (32/74), methods for collecting and recording events were described in 68% (50/74), and assessment of event severity and attribution was described in 49% (36/74) and 26% (19/74), respectively. Over 100 different adverse events were reported across completed trials. The most common events reported were gastrointestinal distress (65%, 49/75) and headache (33%, 25/75).
Conclusions:
We found variability in monitoring, assessing, and reporting adverse events in pediatric trials of natural treatments. The adverse events identified in this review reinforces that specific events should be prospectively monitored in future trials.
Introduction
Psychotherapy and pharmacotherapy are mainstay treatments for many neuropsychiatric disorders in childhood including depression, anxiety disorders, and attention-deficit/hyperactivity disorder (ADHD; Bukstein et al., 2020; Cheung et al., 2018; Wolraich et al., 2019). When these treatments are no longer effective, not well tolerated, or do not meet expectations, caregivers and clinicians may seek out other available options. The use of natural products—nutraceuticals (nutrient-based; e.g., polyunsaturated fatty acids, vitamins, minerals) and phytoceuticals (herbal- or plant-based; e.g., St John’s Wort, kava)—as an alternative or adjunct treatment may reflect a growing interest in nutritional psychiatry, which focuses on the relationship between nutrition and emotion, cognition, and behavior (Marx et al., 2017). Although this industry is growing, these products are not prescribed or regulated, which may lead to issues with improper use, undesirable drug interactions, and harmful or undesirable effects (Puri et al., 2022).
In 2022, the World Federation of Societies of Biological Psychiatry and the Canadian Network for Mood and Anxiety Disorders published a consensus-based, graded, clinical guideline for nutraceutical/phytoceutical use. The group made a “weak” recommendation for use of two natural products, broad-spectrum micronutrient, and vitamin D, with children with ADHD based on supportive but limited data from meta-analytic level results (Sarris et al., 2022). The group noted the challenge in interpreting the evidence base due to heterogeneity in product manufacturing and standardization as well as varying administration routes and doses. Authors of systematic reviews of trials of natural treatments for childhood ADHD (Abdullah et al., 2019), autism spectrum disorders (Jiang et al., 2023), and depression (Campisi et al., 2021) have also commented on this challenge stating that variability in dosage (Campisi et al., 2021; Jiang et al., 2023) and outcome measures (Abdullah et al., 2019; Campisi et al., 2021) limits cross-study comparisons and brief evaluation time-periods in trials limit the understanding of the effects of natural treatments over time (Abdullah et al., 2019; Jiang et al., 2023).
One aspect missing from the existing reviews is a focus on the type and consistency in the methods used by investigators for monitoring, assessing, and reporting harmful or undesirable effects that could occur in trials studying natural treatments. Identifying these effects in pediatric trials of nutraceutical and phytoceutical products ensures the evidence base used to inform product-related decision-making accounts for both potential benefits and harms. The objective of this systematic review was to identify how and which adverse events have been monitored, assessed, and reported in trials of nutraceuticals and phytoceuticals for childhood neuropsychiatric conditions.
Methods
The reporting of this study follows the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guideline (Page et al., 2021), and the study protocol was registered in the international systematic review registry, PROSPERO (CRD42023421182).
Search strategy
One reviewer generated search results from six bibliographic databases: MEDLINE, PsycINFO, Embase, ProQuest Dissertations and Theses Global, Google Scholar, and the Cochrane Library. Filters were used to restrict search results to English-language records from January 2012 to March 2024. Our search strategy included broad terms such as “pediatric” and “mental disorders” as we were unsure if all trials would be indexed using nutraceutical or phytoceutical terms, including specific compounds. From the broad search results, two reviewers searched specifically for nutraceutical and phytoceutical trials. See Supplemental File 1 for the MEDLINE search strategy. To ensure details were comprehensive for each included trial, we conducted a manual search for information sources that involved reviewing results-based papers for trial registrations and vice versa.
Selection criteria
We included protocols (i.e., online trial registrations and published protocols) and results-based papers for clinical trials of nutraceutical or phytoceutical efficacy or effectiveness meeting the following criteria: (1) available in English language, (2) experimental (randomized controlled trial) or quasi-experimental (nonrandomized trial) design, and (3) participants were children aged 5–19 years with one or more of the following diagnoses: anxiety disorder, obsessive-compulsive disorder, posttraumatic stress disorder, mood disorder, behavioral disorder, adjustment disorder, eating disorder or related body dysmorphic disorder, personality disorder, schizophrenia or psychosis-related disorder, high-functioning autism spectrum disorder, tic disorder or syndrome, ADHD, or substance use disorder. To reflect real-world product use, we included trials that evaluated a nutraceutical or phytoceutical as a unadjunct to psychotherapy and/or pharmacotherapy, provided the evaluation of the natural treatment was the trial’s primary focus. Trials with a pediatric population that was outside of our preferred range by 1 year (e.g., aged 4–9 years) were included; we reviewed results-based papers to ensure that the mean participant age was within the preferred age range. Conference abstracts needed to be linked to a protocol, dissertation, or results-based paper to be included in the review as stand-alone abstracts did not contain the details on adverse event monitoring, assessment, and reporting that we were interested in reviewing.
Article screening
We imported retrieved citations into Covidence (https://www.covidence.org/) for screening. Two reviewers used the selection criteria to independently screen the titles and abstracts of records. Full-text articles identified as potentially relevant were also screened in duplicate. Disagreements were resolved by consensus between the reviewers in consultation with two other authors. Reasons that articles were excluded after full-text review were documented.
Data extraction
We linked different types of information sources (e.g., registration, published protocol, and results-based paper) belonging to the same trial and extracted data across linked sources. One reviewer extracted data with accuracy and a second reviewer checked for completeness. We extracted information on trial characteristics, including design features, participant age parameters, and clinical diagnoses, as well as details on the experimental and comparison interventions. We also extracted information on what was considered to be an adverse event during the trial, adverse event-related hypotheses and/or objectives, and methods reported for adverse event data collection and recording (e.g., how an event was identified and by whom, types of participant questioning methods; Allen et al., 2018). From results-based papers, we extracted data on which events were reported, the described approach to statistical analyses, and author comments on potential sources of bias related to trial methods (Ioannidis, 2009; Ioannidis et al., 2019; Junqueira et al., 2022).
Assessment of adverse event monitoring and reporting practices
We assessed and extracted information related to adverse event monitoring, assessment, and reporting using a guide that was based on the Focused, Extensive, Applied, Transparent principles (Frampton et al., 2022) and contained items based on Good Clinical Practice guidelines and the Consolidated Standards of Reporting Trials (CONSORT) Harms Extension (Junqueira et al., 2022; U.S. Department of Health and Human Services et al., 2018). In this review, we defined an adverse event as any reported unfavorable outcome (i.e., harmful or undesirable effects) for a child not necessarily caused by the intervention they received (Agency for Healthcare Research and Quality, 2019; World Health Organization, 2010). Using this term allowed us to include data from trials that did not involve assessing event causality (Chou et al., 2010). Trial investigators could use other terminology commensurate with the methods used in the trial including adverse effect or side effect. We recorded monitoring and reporting practices (who reported the adverse event) as being conducted (yes/no) or partially conducted/unclear (in cases where some practices were used but not all or lacked clarity in description). Assessment was conducted by two reviewers with disagreements resolved through discussion.
Data analysis
We used frequencies and proportions to summarize findings across the included trials. We grouped adverse event data using terms from the Medical Dictionary for Regulatory Activities (International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use, n.d.) to understand monitoring/reporting foci: nervous system, respiratory, cardiovascular, musculoskeletal, psychiatric, sleep-related, metabolism and nutrition, skin and tissue, gastrointestinal, pain or discomfort, infections and infestations, investigations (e.g., laboratory results), injury or poisoning or procedural complications, and other (e.g., hospitalization, procedures). Events considered to be serious by investigative teams (e.g., suicide attempt) were also identified.
Results
The search strategy yielded 16,713 unique records (Figure 1). Ninety-eight trials met the inclusion criteria: 23 (23%) were protocols (no results yet available) and 75 (77%) were completed (results-based publication).
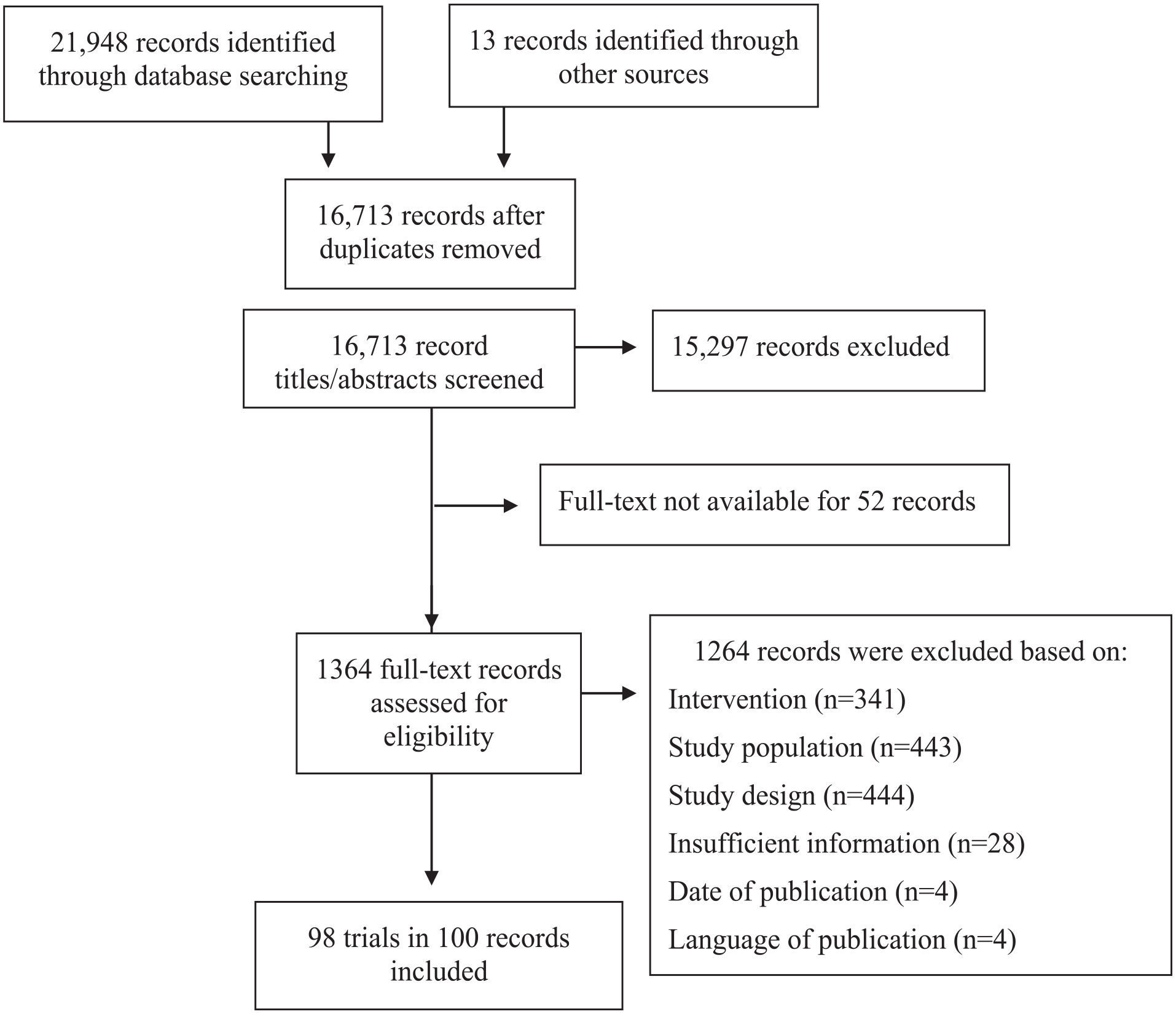
Literature search flow diagram.
Characteristics of included trials
Eight-one percent (79/98) of the trials involved testing the effects of a nutraceutical product (Supplementary File 2; Anand and Sachdeva, 2016; Assareh et al., 2017; Barragan et al., 2017; Batebi et al., 2021; Bent et al., 2014; Bloch, 2022; Bos et al., 2015; Carucci et al., 2022; Chang et al., 2019; Cornu et al., 2018; Crippa et al., 2019; Dashti et al., 2014; Davies, 2023; Dean et al., 2014; Doaei et al., 2021; Dubnov-Raz et al., 2014; Elhossiny et al., 2023; Feng, 2024; Föcker et al., 2018; Fristad et al., 2015, 2019; Gabbay et al., 2012, 2018; Ghajar et al., 2018; Ghanizadeh et al., 2013; Gröbner et al., 2022; Gyrgo, 2014; Haberling et al., 2019; Hariri et al., 2012; Hemamy et al., 2020, 2021; Johnstone et al., 2022; Kean et al., 2017; Keller et al., 2022; Khalaj et al., 2018; Kumperscak et al., 2020; Kvernmo, 2024; Lawrence et al., 2023; Lawrence, 2023; Li et al., 2020; Liu et al., 2019; Llevadot, 2019; Manor et al., 2012; Mehrazad-Saber et al., 2018; Milte et al., 2012; Mirhosseini et al., 2023; Mohammadpour et al., 2018; Mohammadzadeh et al., 2019; Moradi et al., 2020; Mostajeran et al., 2020; Motaharifard et al., 2019; Naeini et al., 2019; Ni, 2023; Nikoo et al., 2015; Noorazar et al., 2020, 2021; Palmer et al., 2024; Panahande et al., 2017; Perera et al., 2012; Pongpitakdamrong et al., 2022; Pusponegoro et al., 2015; Rahmani et al., 2022; Raine et al., 2019; Rheims et al., 2024; Rhoads, 2024; Rizzo et al., 2022; Rodríguez et al., 2019; Rucklidge et al., 2018; Salehi et al., 2016; Sangouni et al., 2022; Scahill, 2021; Sepehrmanesh et al., 2021; Straussberg, 2014; Tohidi et al., 2021; Trebatická et al., 2020; Tzang et al., 2016; Widenhorn-Müller et al., 2014; Wink et al., 2016; Wozniak et al., 2015; Wu et al., 2021; Zhou et al., 2021).
Nineteen percent (19/98) involved testing the effects of a phytoceutical product (Supplementary File 3; Baziar et al., 2019; Blasco-Fontecilla et al., 2022; Ford, 2023; Golsorkhi et al., 2022; Guo et al., 2022; Hannant et al., 2021; Hasanzadeh et al., 2012; Hendouei et al., 2020; Hosseini et al., 2019; Huang et al., 2020; Kean et al., 2015; Khaksarian et al., 2021; Lee et al., 2019; Momtazmanesh et al., 2020; Murdoch Children’s Research Institute, 2024; Shakibaei et al., 2015; Soltanifar et al., 2023; Verlaet et al., 2017; Wang et al., 2012).
Across all trials, products were most commonly tested for ADHD (59%, 58/98) and ASD (21%, 21/98), while the most commonly tested product was polyunsaturated fatty acids (36%, 35/98). In 30% (29/98) of the trials, a nutraceutical (69%, 20/29) or phytoceutical (31%, 9/29) product was investigated as an adjunct to a pharmaceutical treatment. The majority of trials were reported as funded by the investigators’ university (38%, 37/98) or a government-funded agency (19%, 19/98). The most common terminology used by investigators was adverse event (45%, 44/98) followed by side effect (33%, 32/98) and adverse effect (14%, 14/98).
Adverse event monitoring, assessment, and reporting practices
In 21/98 (22%) trials, investigators reported an objective or hypothesis specific to monitoring for adverse events. In 74/98 (76%) trials, the methods described indicated that adverse event monitoring took place. Of the 29 trials that involved assessing natural treatments as adjuncts to pharmaceutical products, adverse event monitoring was indicated as a component in 26 (90%); this was a greater proportion than trials that evaluated a nutraceutical or phytoceutical alone (68%, 47/69), and these differences were statistically significant (p = 0.04).
Table 1 presents the monitoring, assessment, and reporting practices described for the 74 trials. Of these trials, 43% of trials (32/74) identified defined events a priori for prospective monitoring. A variety of methods for collecting and recording events were described for 50/74 (68%) trials. In 51% of trials (38/74), investigators described using structured tools to elicit and record information on adverse event occurrence. Publicly available measures reported as used across multiple trials were the Extrapyramidal Symptom Rating Scale (Chouinard and Margolese, 2005; n = 3) and Safety Monitoring Uniform Report Form (Greenhill et al., 2004; n = 2), while measures reported in single trials included the Treatment Emergent Symptom Scale (Guy, 1976), Adverse Events Profile (Gilliam et al., 2004; Helmstaedter et al., 2024), National Initiative for Children’s Healthcare Quality (NICHQ) Vanderbilt Assessment Follow-up (American Academy of Pediatrics and NICHQ, 2002), the Suicidal Ideation Questionnaire Junior (Reynolds, 1987), Antidepressant Side Effect Checklist (Uher et al., 2009), and Barkley Side Effect Rating Scale (Barkley, 1981). An overview of these measures is presented in Supplemental File 4. In 28 trials, investigators described the use of a study-developed interview, measure, and/or form, and in 9 trials, investigators cited other author groups’ publications rather than a direct source for a measure; the measures used in these 37 trials were not publicly available for review. The use of spontaneous report by caregivers (reporting not prompted by investigator questioning or timed data collection) was relied on in 8 trials in addition to solicited data collection methods.
Adverse event monitoring and reporting practices described in 74 trials.

>100%; multiple counts as more than one could be reported.
Calculated based on 45 trials during which an adverse event was reported during monitoring.
Corresponding trial numbers are found in Supplemental Files 2 and 3.
The individuals responsible for identifying events varied across trials, with most individuals unidentified (53% of trials; 39/74). The blinding of event assessors was reported for almost all the trials (93%, 69/74). An assessment of event severity was reported as a component in 49% (36/74) of trials, while assessing event attribution was reported less frequently (26%, 19/74; Table 1).
Among completed trials that involved monitoring for adverse events, events were reported as having occurred in 75% (45/60); adverse events were also reported in two completed trials for which monitoring was not described as part of the trial design. Reporting on the number of participants that contributed to the analysis, methods used to compare events between intervention groups, and reporting data separately for each intervention group varied (Table 1). Table 2 details over 100 types of adverse events that were prospectively surveilled across the 74 trials that involved monitoring. The most common events that were specifically monitored for included gastrointestinal distress (36% of trials; 27/74), headache (18% of trials; 13/74), and decreased or loss of appetite (14% of trials; 10/74), which were reported to have occurred in 65% (49/75), 33% (25/75), and 23% (17/75) of completed trials, respectively. Nine different types of serious adverse events were prospectively monitored in three trials and reported as having occurred in one trial.
Adverse events that were monitored and/or reported in trials.

>100%; multiple counts as more than event could be monitored or reported.
Considered in the trial to be a serious adverse event.
For example: “others,” “data not shown,” “side effects were uncommon and mild.”
Discussion
The study of adverse events alongside the potential benefits of nutraceutical and phytoceutical products is important to generate a comprehensive evidence base for clinical decision-making. Using guidelines from Good Clinical Practice (U.S. Department of Health and Human Services et al., 2018) and reporting recommendations from the CONSORT Harms Extension (Junqueira et al., 2022), the results from this systematic review highlight: (1) differences across pediatric natural treatment trials in the amount of detail reported for adverse event monitoring; (2) in a modest proportion of trials, the type of adverse events identified for prospective monitoring; and (3) that event monitoring differed between trials that involved studying natural treatments as an adjunct to pharmaceutical treatment versus a stand-alone treatment.
While previous systematic reviews have involved appraising the methods used in natural treatment trials (Abdullah et al., 2019; Campisi et al., 2021; Jiang et al., 2023), these reviews have not included a focus on adverse event monitoring, assessment, or reporting. This particular focus adds important details on how trials have been conducted and reported. Across the group of trials that we reviewed, there were differences in what was described for adverse event monitoring. Based on these results, we suggest that a comprehensive approach to monitoring and reporting adverse events be used in all future natural treatment trials, a recommendation that has also made by other systematic review author groups of complementary therapy reviews (Asher et al., 2017; Fusar-Poli et al., 2019; Ng et al., 2023). This recommended comprehensive approach includes investigators: (1) using systematic assessment (e.g., structured measure) to collect adverse event data and considering the addition of nonsystematic assessment (e.g., spontaneous report) to augment their approach, (2) assessing adverse events for severity and attribution to clarify event impact and relationship to the natural treatment, and (3) using the CONSORT Harms Extension to guide reporting of methods and findings (Junqueira et al., 2022). Following these recommendations would introduce methods consistency across trials and could help to address concerns that the current evidence base on harmful or undesirable effects is variable (Puri et al., 2022).
An important contribution of this review is its finding that adverse events that were specifically monitored for in some trials were reported in a higher proportion of completed trials. For example, gastrointestinal distress and headache were prospectively monitored in 36% and 18% of trials, respectively, and reported as having occurred in 65% and 33% of completed trials. These findings highlight the concept of event expectedness and the reinforce the importance of comprehensive data collection. In terms of event expectedness, while trial investigators may not have expected specific events to occur, moving forward, the list of reported events compiled in this review, across a range of natural treatments, provides investigators with an opportunity to consider the plausibility of their relationship to the product they are testing. Those events that could be expected should be actively monitored during the trial, and which events should be reported to a regulatory body should be identified (Nebeker et al., 2004). From a data collection perspective, use of both systematic and nonsystematic approaches can help to identify rare and/or unexpected adverse events. Use of both approaches is ideal as solely relying on a nonsystematic approach may contribute to event underreporting if the event is similar to a disease-related complication or if the event is well-known (Loke et al., 2011). Investigators should also consider from whom adverse event data should be collected. For example, a child may be a more ideal informant for identifying certain somatic complaints or mood states, while a parent or clinician may be ideally suited to identify behaviors. Description of this aspect was missing for 53% of the trials included in this review, but its consideration would ensure an optimized approach to adverse event solicitation.
The findings from this review should be considered against several limitations to its scope. We were interested in contemporary studies on nutraceutical and/or phytoceutical interventions, so we limited our search to studies published or registered between 2012 and 2024. Therefore, our results may not generalize to studies published outside of this time-period. We also focused on pediatric trials in this review, so our findings do not apply to the adult population. While there may be trials testing novel nutraceutical and/or phytoceutical products that were not captured by our search, we took a broad search approach to minimize this issue. Scope is also strength of this review in terms of our inclusion of trials involving a broad range of natural treatments including those that involved their evaluation as an adjunct to prescribed pharmacotherapy. While not the focus of this review, investigators’ report of adverse events by treatment group allows other investigators to consider potential drug interactions.
Conclusions
In this review, we found variability across pediatric natural treatment trials in the amount of detail reported for adverse event monitoring and assessment methods. Because nutraceuticals and phytoceuticals are not yet prescribed or regulated, issues can arise with improper use, undesirable drug interactions, and harmful or undesirable effects. This context makes it critical for trial investigators to generate knowledge of both product harms and benefits. For investigators conducting future natural treatment trials, we recommend using systematic assessment to collect adverse event data and considering the addition of nonsystematic assessment to augment their approach, assessing adverse events for severity and attribution to clarify event impact and relationship to the natural treatment, and using the CONSORT Harms Extension to guide reporting of methods and findings (Junqueira et al., 2022).
Supplemental Material
sj-docx-1-jop-10.1177_02698811251344683 – Supplemental material for Adverse event monitoring, assessment, and reporting in nutraceutical and phytoceutical trials for pediatric neuropsychiatric conditions: A systematic review
Supplemental material, sj-docx-1-jop-10.1177_02698811251344683 for Adverse event monitoring, assessment, and reporting in nutraceutical and phytoceutical trials for pediatric neuropsychiatric conditions: A systematic review by Kalee Lodewyk, Darren B Courtney, Alexa Bagnell and Amanda S Newton in Journal of Psychopharmacology
Footnotes
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
This study is a systematic review using published, methodological data and did not require ethical approval.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
