Abstract
Background:
Generalized anxiety disorder (GAD) is commonly co-morbid with major depressive disorder (MDD) and is associated with greater functional impairment and poorer treatment outcomes than MDD alone. However, studies on treatment with drugs for depression in patients with MDD and co-morbid GAD are limited.
Aims:
To examine the effectiveness of vortioxetine treatment in patients with MDD and co-morbid GAD in a subgroup analysis of the real-world RELIEVE study.
Methods:
The analysis included outpatients diagnosed with MDD and co-morbid GAD who initiated vortioxetine treatment at their physician’s discretion in the 24-week, observational RELIEVE study. Primary outcome was patient functioning (Sheehan Disability Scale (SDS)) after 12 and 24 weeks of vortioxetine treatment; secondary outcomes included depression severity (9-item Patient Health Questionnaire (PHQ-9)), cognitive symptoms (5-item Perceived Deficits Questionnaire – Depression (PDQ-D-5)) and cognitive performance (Digit Symbol Substitution Test (DSST)).
Results:
Overall, 180 patients with MDD and co-morbid GAD were included in the analysis. Following vortioxetine initiation, clinically significant improvements in patient functioning (SDS total score) were observed at week 12 (least-squares (LS) mean reduction from baseline, 7.5 points), sustained through week 24 (9.2 points) (both p < 0.0001). LS mean PHQ-9, PDQ-D-5 and DSST scores improved by 7.9, 4.8 and 7.4 points at week 24, respectively (all p < 0.0001 vs baseline). Adverse events were reported by 33.9% of patients (most commonly nausea, 13.3%).
Conclusions:
In routine clinical practice, vortioxetine was associated with clinically meaningful, sustained improvements in functioning, and depressive and cognitive symptoms, in patients with MDD and co-morbid GAD.
Clinical Trials Registry Name and Identifier:
Real-life Effectiveness of Vortioxetine in Depression (RELIEVE) (NCT03555136) https://clinicaltrials.gov/ct2/show/NCT03555136
Keywords
Introduction
Major depressive disorder (MDD) is characterized by a heterogeneous range of emotional, cognitive and physical impairments (Fried and Nesse, 2014; Goodwin et al., 2017; IsHak et al., 2016), and is a leading cause of disability worldwide (GBD 2017 Disease and Injury Incidence and Prevalence Collaborators, 2018). In 2020, the substantial global burden of MDD (and anxiety disorders) was increased further, associated with raised rates of SARS-CoV-2 infection and impeded human mobility during the COVID-19 pandemic (COVID-19 Mental Health Collaborators, 2021). Co-morbidity of MDD and generalized anxiety disorder (GAD) is common (McGrath et al., 2020; Saha et al., 2021; Zhou et al., 2017), and there is a strong genetic overlap between the two conditions (Morneau-Vaillancourt et al., 2020). Indeed, a meta-analysis of 171 studies found that individuals with depressive disorders had an 11.7 times elevated risk of developing GAD compared with those without depressive disorders (Saha et al., 2021). An international World Health Organization World Mental Health survey, that assessed 145,990 adult respondents according to Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, diagnostic criteria, found that those with MDD had an 11.4% risk of developing GAD within 10 years, and those with GAD had a 37.7% risk of subsequently developing MDD (McGrath et al., 2020).
The coexistence of MDD and GAD increases the duration of long-term disease beyond those of the individual disorders (Kessler et al., 2008; Meier et al., 2015), and raises the risk of relapse of depression (Buckman et al., 2018). In addition, MDD and co-morbid GAD are linked to a lower health-related quality of life (HRQoL), increased functional and cognitive impairment, and greater limitations in daily activities, than observed with either disorder alone (Armbrecht et al., 2021; Zhou et al., 2017). The combination of MDD and GAD is also associated with a poorer prognosis than the individual conditions, being more difficult to treat and having worse treatment outcomes, such as a longer time to remission and a lower chance of remission overall (Fava et al., 2008; Penninx et al., 2011). However, despite the need for direct evidence of treatment effects in patients with co-morbidity (Hirschfeld, 2001), clinical data on drugs for depression in MDD and co-morbid GAD – particularly data on functional outcomes – are very limited. A post-hoc analysis of data from a placebo-controlled double-blind clinical trial has demonstrated the efficacy of extended-release venlafaxine on depressive and anxiety symptoms in a subset of 92 patients with MDD and co-morbid GAD (Silverstone and Salinas, 2001). Another (pilot) study of 34 patients with MDD and co-morbid GAD showed numerical improvement in the symptoms of depression and anxiety with the multi-modal drug, extended-release quetiapine, versus placebo (Li et al., 2016). In both cases, the findings require confirmation in larger studies.
Vortioxetine is a multi-modal drug for depression with a unique combination of mechanisms of action (Gonda et al., 2019). It directly and indirectly influences multiple neurotransmitter systems relevant to the neurobiology of MDD, acting as both a serotonin transporter inhibitor and a modulator of several serotonin receptor subtypes (Gonda et al., 2019; Sanchez et al., 2015). In randomized controlled trials, vortioxetine (5–20 mg administered orally once daily) has demonstrated broad efficacy against the symptoms of MDD (Gonda et al., 2019; Thase et al., 2016), and vortioxetine-treated patients have shown significant improvements in cognition and overall functioning (Florea et al., 2017; McIntyre et al., 2016). Vortioxetine has also demonstrated anxiolytic effects (vs placebo) in patients with MDD and high levels of anxiety symptoms (Baldwin et al., 2016), in patients with MDD who showed inadequate response to first-line serotonin reuptake inhibitor (SERT), or serotonin and norepinephrine reuptake inhibitor (SERT and NET) treatment (demonstrating superiority to agomelatine) (Montgomery et al., 2014), and in the long-term maintenance treatment of patients with MDD (Vieta et al., 2017). Vortioxetine was also significantly more efficacious than placebo in the treatment of GAD in a meta-analysis of four short-term studies (Pae et al., 2015), and was effective in treating symptoms of depression and anxiety in a study of patients with MDD and co-morbid social anxiety disorder (SAD) (Liebowitz et al., 2017). Moreover, in the recent 8-week, open-label, RECONNECT study, patients with severe MDD and severe co-morbid GAD who were treated with vortioxetine showed clinically meaningful and statistically significant improvements in depression and anxiety symptoms, alongside improvement in overall functioning and HRQoL (Christensen et al., 2022).
To complement and extend existing clinical evidence, the RELIEVE study assessed the real-world effectiveness of a heterogeneous population of patients with MDD who were initiating treatment with vortioxetine (Mattingly et al., 2022). Integral to its real-world design, RELIEVE had no exclusion criteria for concomitant anxiety disorders, and thus included a significant proportion of patients with diagnoses of both MDD and GAD. Findings in the overall study population of patients with MDD in RELIEVE revealed significant and sustained improvements in patient functioning, as well as in measures of depressive and cognitive symptoms, sexual function and HRQoL, across a period of 6 months of vortioxetine treatment (Mattingly et al., 2022). In this subgroup analysis of data from the RELIEVE study, the effectiveness and safety of vortioxetine in patients with MDD and co-morbid GAD was assessed through measures of self-reported patient functioning, depression, cognition, sexual function, HRQoL and adverse events (AEs).
Methods
Study design and participants
RELIEVE was a 24-week, observational, multinational (Canada, France, Italy, USA), prospective cohort study in outpatients with MDD initiating treatment with vortioxetine in routine clinical practice (NCT03555136). The study was conducted between November 2017 and January 2021.
Full details of the study design have been reported previously (Mattingly et al., 2022). In brief, the study enrolled outpatients aged ⩾18 years who were diagnosed with MDD (according to local criteria), experiencing a major depressive episode (MDE), and initiating treatment with vortioxetine at their primary care or psychiatric outpatient practice according to the locally approved label. The present post-hoc subgroup analysis included eligible patients who were also recorded as having a diagnosis of GAD (according to local criteria) at baseline. Patients with schizophrenia, bipolar disorder, substance abuse or any neurodegenerative disease significantly impacting on cognitive functioning, and those considered at significant risk of suicide, were excluded. As this was a real-world study, there were no exclusion criteria for concomitant medications during the study period, including other pharmacotherapy for MDD and/or other psychoactive medications.
Data were collected at routine clinic visits at study entry (baseline) and after 12 and 24 weeks (±4 weeks) of vortioxetine treatment. A critical management plan (implemented due to the COVID-19 pandemic) permitted patients to have remote follow-up visits and complete patient-reported outcome assessments at home, if needed. Adherence to medication was monitored as per routine clinical practice, and the patient could be withdrawn from the study for compliance reasons at the physician’s decision.
The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. Local ethics committee approval for the study was obtained at all participating sites and all patients provided written informed consent for participation.
Study assessments
The primary objective was assessment of functioning as measured by the Sheehan Disability Scale (SDS), which has been widely used to assess function in patients with MDD (Sheehan et al., 1996; Sheehan and Sheehan, 2008). This well-validated, brief and simple self-report measure is easily administered in routine care and assesses the degree of functional impairment over the previous 7 days across three domains: work/school, family life/home responsibilities and social/leisure activities. Patients rate the level of impairment for each domain using a visual analogue scale ranging from 0 (not at all) to 10 (very severe). Scores from the individual domains are combined to generate the SDS total score, ranging from 0 (unimpaired) to 30 (highly impaired). An improvement of more than 4 points in SDS total score, and 1–2 points on a subdomain score, is considered meaningful for patients (Sheehan and Sheehan, 2008). Work productivity measures of absenteeism and presenteeism were derived from SDS scores on lost and underproductive days, respectively, for the working population (patients in full/part-time work or school). The number of days taken as sick leave during the past 12 weeks (prior to baseline, week 12 and week 24) was also assessed in the working population.
Secondary objectives were assessments of depressive symptoms (9-item Patient Health Questionnaire (PHQ-9), scored 0–9 with higher score indicating more severe symptoms; and the Clinical Global Impression – Severity (CGI-S) scale, scored 1–7 with higher score indicating greater disease severity); cognitive symptoms (5-item Perceived Deficits Questionnaire – Depression (PDQ-D-5), scored 0–20 with higher score indicating more severe symptoms); cognitive performance (Digit Symbol Substitution Test (DSST), scored 0–133, with higher score indicating better performance); sexual functioning (Arizona Sexual Experiences Scale (ASEX), scored 5–30, with higher score indicating greater dysfunction); and HRQoL (EuroQol 5-Dimension 5-Level Questionnaire Utility Index (EQ-5D-5L), scored 0–1 with higher score indicating better health).
Safety was assessed through the spontaneous reporting of AEs by the patient or the investigator, summarized by lowest level Medical Dictionary for Regulatory Activities (MedDRA, Version 23.1) preferred terms.
Statistical analysis
Full statistical details for the RELIEVE study have been published previously (Mattingly et al., 2022). For this subgroup analysis, the effectiveness of vortioxetine was assessed in the full analysis set of the co-morbid GAD subgroup (FAS-GAD), which comprised patients who met the study eligibility criteria, initiated vortioxetine ⩽7 days before the study baseline visit and had at least one post-baseline assessment. The primary study endpoint was mean change from baseline in SDS total score at 12 and 24 weeks. Secondary endpoints included mean change from baseline at weeks 12 and 24 for SDS domain scores and PHQ-9, CGI-S, PDQ-D-5, DSST, EQ-5D-5L and ASEX scores. The proportion of patients with sexual dysfunction (defined as ASEX total score ⩾19, any individual ASEX item score ⩾5 or a score ⩾4 for any three ASEX items) was also calculated at baseline, week 12 and week 24. Safety (AEs) was assessed in all patients with co-morbid GAD who received at least one dose of vortioxetine (safety population).
Least-squares (LS) mean changes in assessment scores from baseline at weeks 12 and 24 were estimated by linear mixed models for repeated measures, adjusted for clinically relevant baseline variables: age, sex, educational level, duration of current depressive episode, presence of somatic and psychiatric co-morbidities, and depression severity as measured by PHQ-9 score as a continuous variable. Except for SDS total score (where an SDS work/school domain score was imputed based on an average of the other two domains for those not at work/school), missing data were not imputed. Safety variables were summarized over the entire study period, with percentages calculated using the safety population as denominator.
Analyses of the overall RELIEVE study population were performed using R version 3.6.1 (R Core Team, 2016); the co-morbid GAD subgroup analyses were performed using SAS 9.4. Significance was set at p < 0.05.
Results
Study population
Of the 737 patients in the RELIEVE study, 413 (56.0%) had symptoms of anxiety co-morbid with MDD at baseline, including 203 patients (27.5%) reporting anxiety symptoms of ‘unspecified type’. A total of 180 of the 737 patients (24.4%) had a diagnosis of GAD and formed the FAS-GAD and safety population for the present analysis.
Baseline demographics of patients with MDD and co-morbid GAD, and the overall study population, are shown in Table 1. In total, 85% of patients with MDD and co-morbid GAD were white/Caucasian, 68.3% were female, and the mean age at baseline was 47.8 years. Most patients (66.7%) reported at least one co-morbid condition at baseline. The most common co-morbidities (aside from GAD) were sleep disorders (30.6%) and cardiovascular disease (20.0%), with sleep disorders, panic disorder, chronic pain and social phobia present more frequently in the co-morbid GAD subgroup than in the overall study population (Table 1).
Patient demographic and general clinical characteristics at baseline (FAS) in the RELIEVE study overall population and subpopulation with co-morbid GAD.
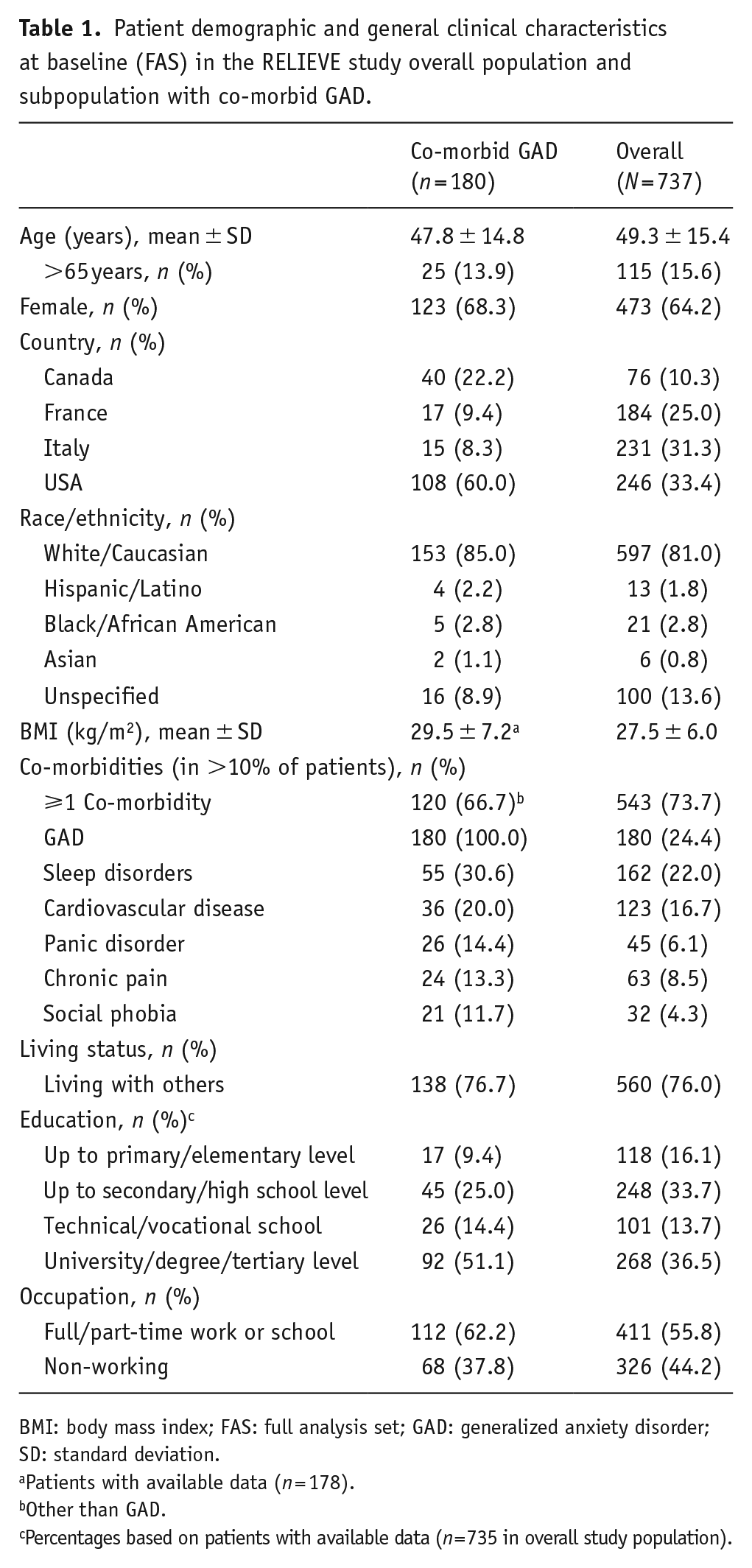
BMI: body mass index; FAS: full analysis set; GAD: generalized anxiety disorder; SD: standard deviation.
Patients with available data (n = 178).
Other than GAD.
Percentages based on patients with available data (n = 735 in overall study population).
Patient disease characteristics at baseline, including concomitant medications, are shown in Table 2. The majority of patients (62.2%) had ⩾2 previous MDEs, and the mean duration of MDD was 13.4 years. Vortioxetine (5–20 mg/day) was initiated as first-line treatment for the current MDE in 27.8% of patients, as second-line treatment in 34.4% of patients, and as third-line treatment or beyond in 37.8% of patients. At baseline, patients displayed impaired functioning across all SDS domains, moderate-to-severe depressive and cognitive symptoms, impaired sexual function and impaired HRQoL. In general, levels of impairment in the co-morbid GAD subgroup at baseline were slightly greater than in the overall RELIEVE study population. In addition, the mean duration of the current MDE was longer in the co-morbid GAD subgroup (101 weeks) than in the overall study population (47 weeks).
Patient MDD-related clinical characteristics at baseline (FAS) in the overall RELIEVE study population and subpopulation with co-morbid GAD.
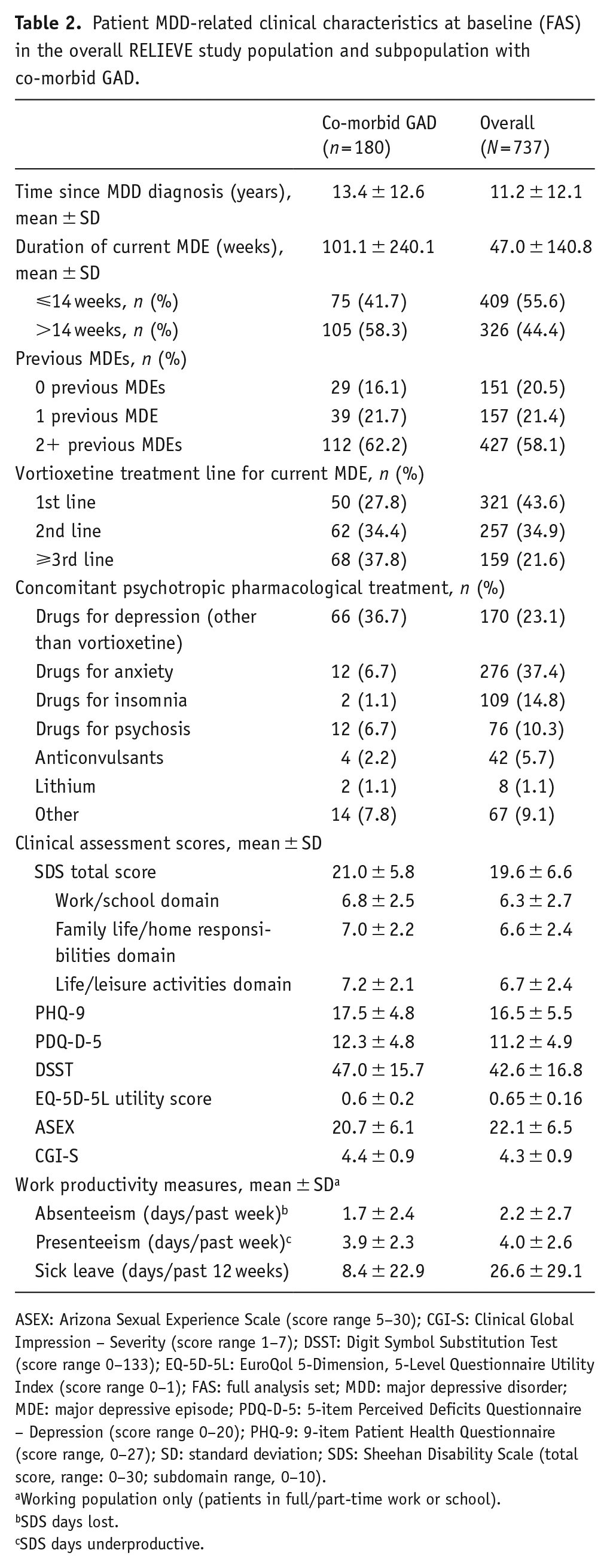
ASEX: Arizona Sexual Experience Scale (score range 5–30); CGI-S: Clinical Global Impression – Severity (score range 1–7); DSST: Digit Symbol Substitution Test (score range 0–133); EQ-5D-5L: EuroQol 5-Dimension, 5-Level Questionnaire Utility Index (score range 0–1); FAS: full analysis set; MDD: major depressive disorder; MDE: major depressive episode; PDQ-D-5: 5-item Perceived Deficits Questionnaire – Depression (score range 0–20); PHQ-9: 9-item Patient Health Questionnaire (score range, 0–27); SD: standard deviation; SDS: Sheehan Disability Scale (total score, range: 0–30; subdomain range, 0–10).
Working population only (patients in full/part-time work or school).
SDS days lost.
SDS days underproductive.
Effectiveness
Clinically meaningful improvements from baseline in patient functioning (LS mean SDS scores) were sustained across 24 weeks of vortioxetine treatment in patients with MDD and co-morbid GAD (Figure 1(a)). The change from baseline in LS mean (standard error) SDS total score was −7.5 (0.6) points at week 12 and −9.2 (0.7) points after 24 weeks of vortioxetine treatment (both p < 0.0001 vs baseline). An improvement of ⩾4 points in SDS total score is considered meaningful for patients (Sheehan and Sheehan, 2008). Statistically significant reductions from baseline were also observed for all three SDS subdomain scores (work/school, family and social) at both time points (p < 0.001 for all changes at weeks 12 and 24) (Figure 1(a)).
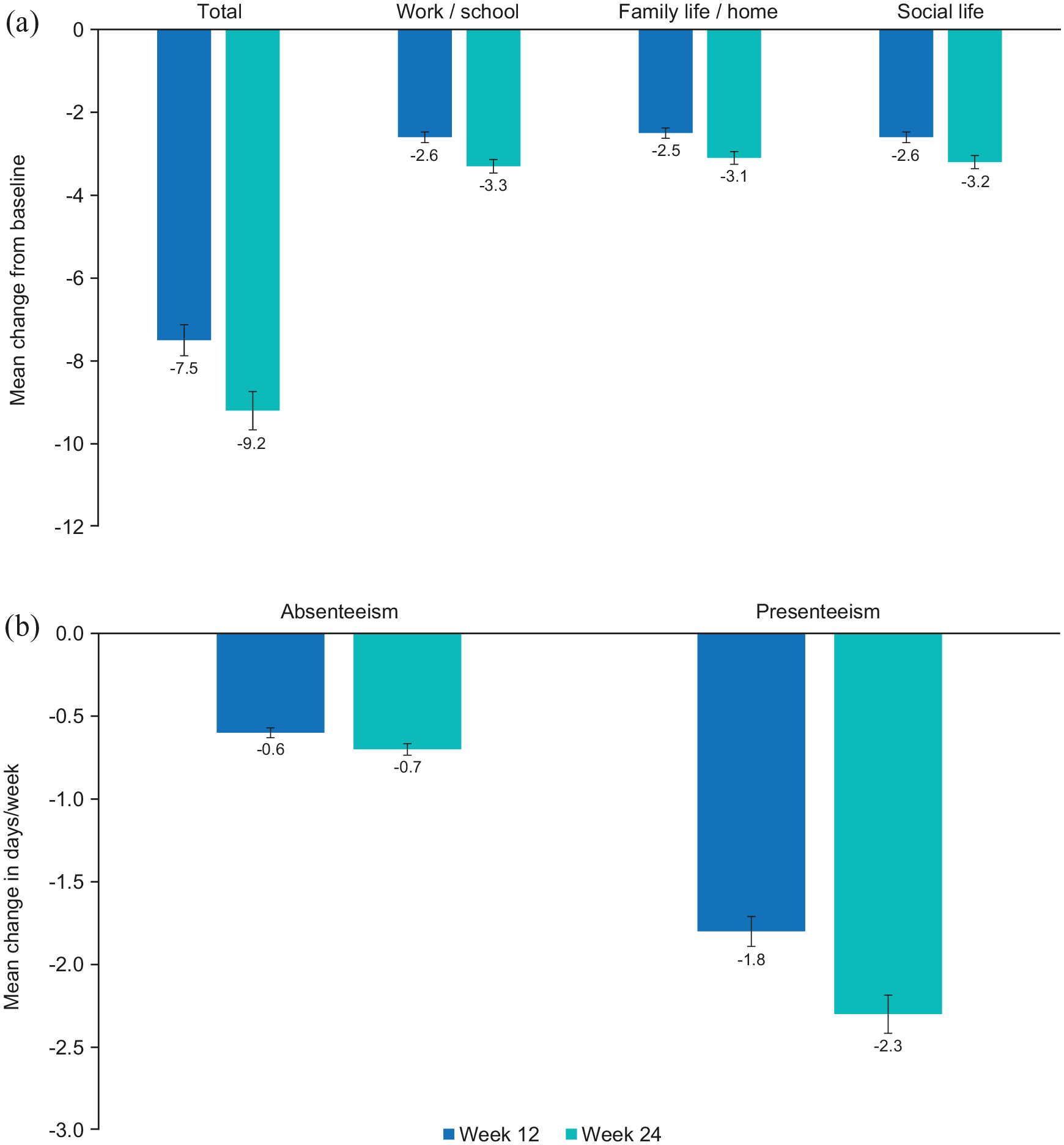
Functional activity–adjusted LS mean change (SE) from baseline to week 12 and week 24 in (a) SDS total and subscores (FAS-GAD) and (b) work productivity (working populationa of FAS-GAD).
In working patients, improvements from baseline were seen in all measures of work productivity. At weeks 12 and 24, respectively, there were mean reductions in absenteeism (work days lost) of 0.6 and 0.7 days/week, and in presenteeism (underproductive work days) of 1.8 and 2.3 days/week (Figure 1(b)). The number of days of sick leave (taken during the preceding 12 weeks) increased at week 12 (by 0.6 days), but had reduced by 1.0 day at week 24.
Clinically relevant improvements in patient- and clinician-rated measures of depression severity, cognition, sexual function and HRQoL were also observed in patients with MDD and GAD, and sustained across the 24 weeks of vortioxetine treatment (Figures 2 and 3). LS mean changes in score from baseline were statistically significant at both week 12 and week 24 (all p < 0.0001 vs baseline, except ASEX (p < 0.05 at week 12, p < 0.01 at week 24)) (Figures 2 and 3). The proportion of patients who met the definition for sexual dysfunction decreased over the 24 weeks of vortioxetine treatment, from 73% at baseline to 62% at weeks 12 and 24. Notably, improvements in cognition to week 24 were observed on both the self-report PDQ-D-5 measure of cognitive symptoms (−3.3 points at week 12; −4.8 points at week 24) and the objective DSST measure of cognitive performance (4.8 points at week 12; 7.4 points at week 24); all p < 0.0001 versus baseline (Figure 2).
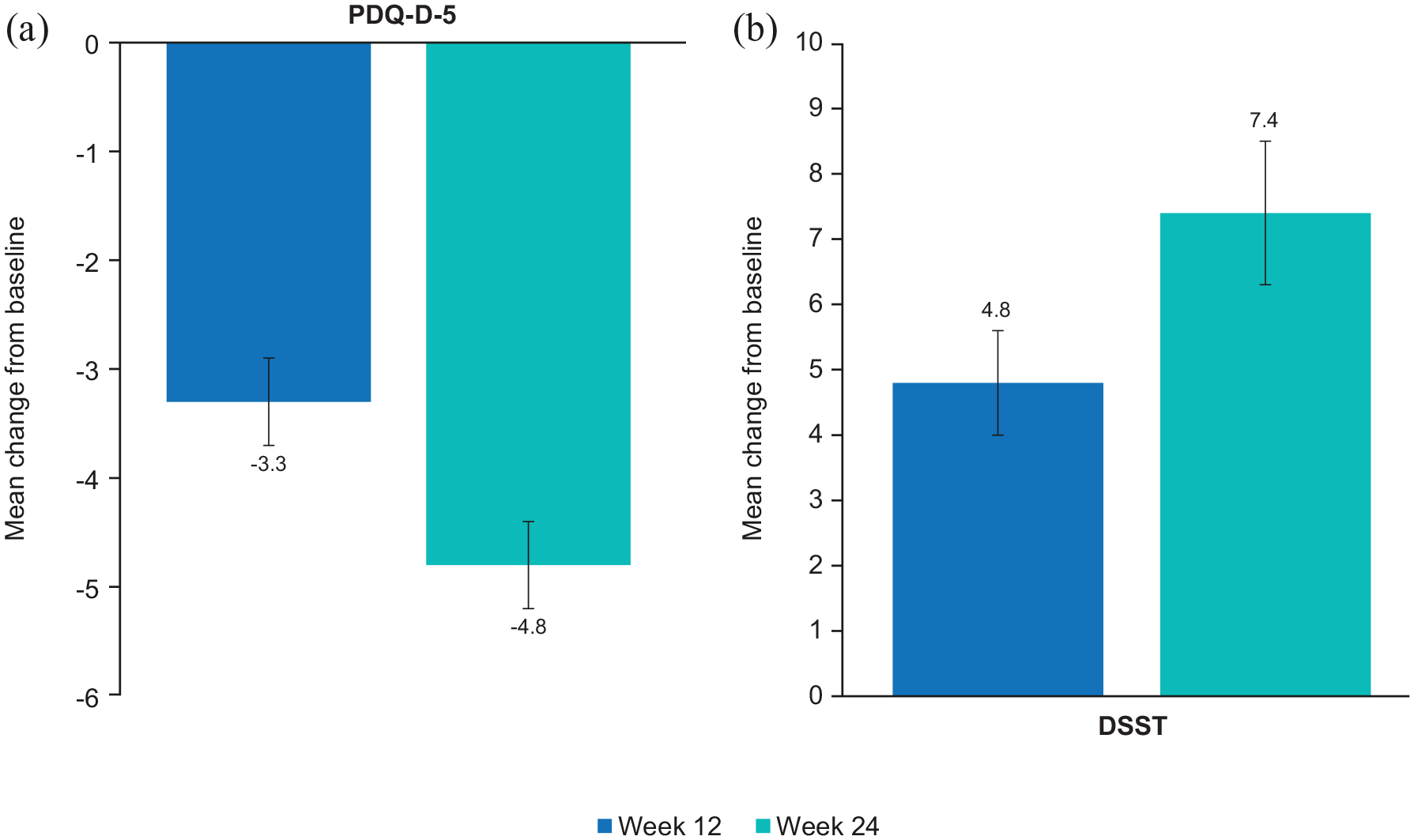
Cognitive function: adjusted LS mean change (SE) from baseline in (a) cognitive symptoms (PDQ-D-5) and (b) cognitive performance (DSST) (FAS-GAD).
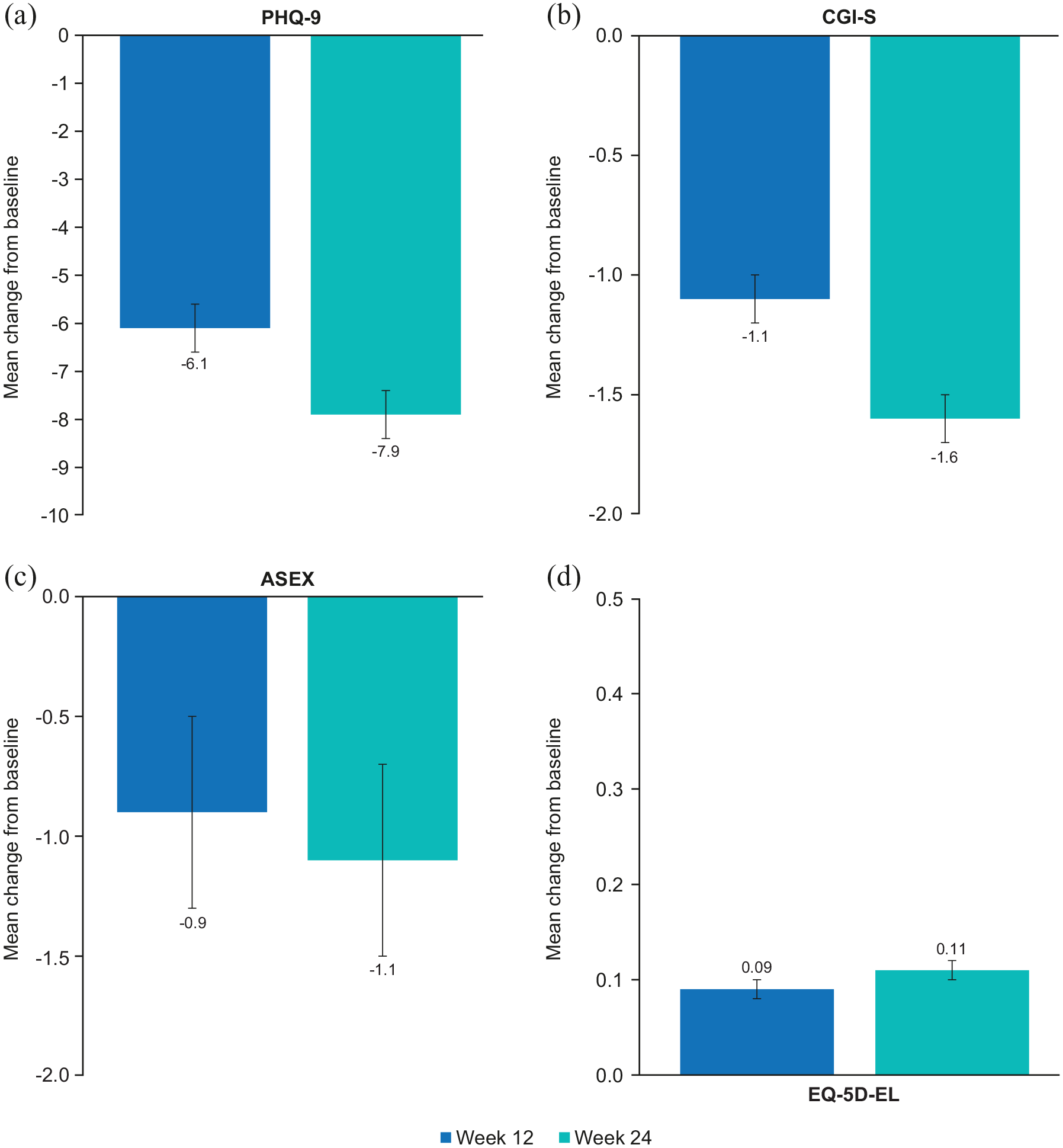
Disease severity, sexual function and quality of life: adjusted LS mean change (SE) from baseline to weeks 12 and 24 in (a) PHQ-9, (b) CGI-S, (c) ASEX and (d) EQ-5D-EL (FAS-GAD).
Safety and tolerability
Treatment with vortioxetine was well tolerated in the subgroup of patients with MDD and co-morbid GAD (Table 3). In total, 61 patients (33.9%) reported an AE over the 24 weeks of vortioxetine treatment. The most frequently reported individual AE was nausea (24 patients; 13.3%), followed by anxiety (6 patients; 3.3%) and headache (4 patients; 2.2%) (Table 3). No other AEs were reported by more than 2% of patients. AEs were most commonly gastrointestinal disorders (39 patients; 21.7%), psychiatric disorders (19 patients; 10.6%) and nervous system disorders (17 patients; 9.4%). Of the 10 serious AEs reported in nine patients, three were considered possibly/probably related to the study treatment (pancreatitis, serotonin syndrome and suicidal ideation; each in one patient). No deaths occurred during the study in the co-morbid GAD subgroup.
Overview of AEs (safety analysis set).
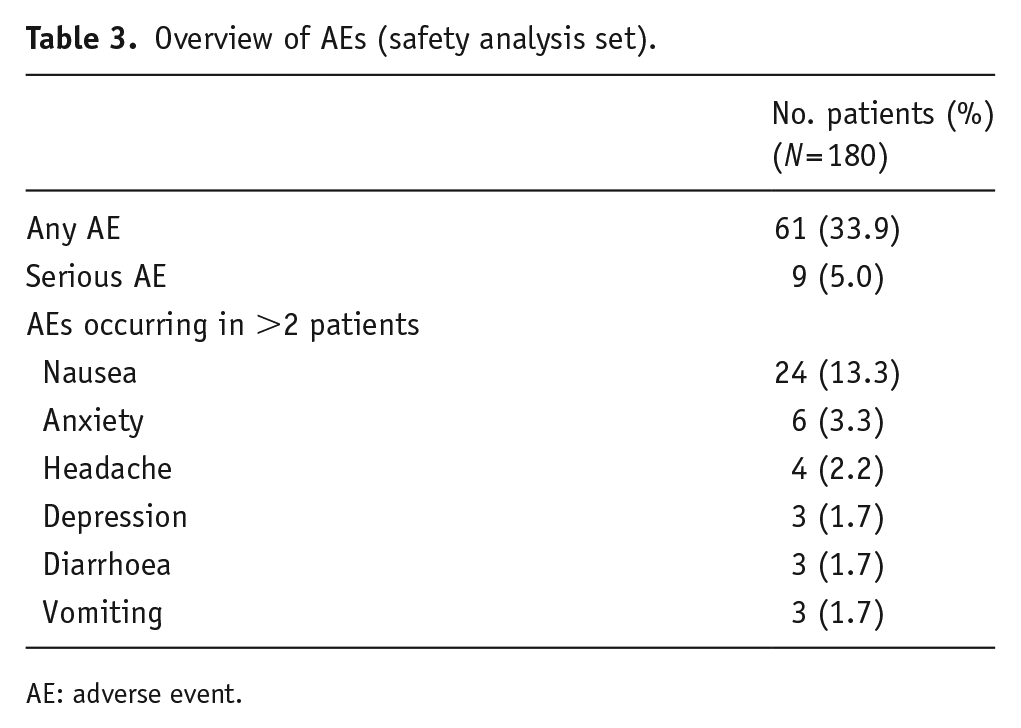
AE: adverse event.
Discussion
GAD is a common co-morbidity in patients with MDD (Saha et al., 2021), and has a negative impact on disease course and treatment outcomes (Fava et al., 2008; Meier et al., 2015; Penninx et al., 2011). However, there are limited clinical data on the treatment effect of drugs for depression on functional outcomes in patients with MDD and co-morbid GAD/anxiety. Real-world studies provide data on treatment efficacy in a heterogeneous patient population, reflective of routine clinical practice. Thus, the real-world RELIEVE study of vortioxetine enabled the analysis of patients diagnosed with MDD and co-morbid GAD, who represented approximately one-quarter of the overall MDD study population. In the current subgroup analysis of RELIEVE, vortioxetine was associated with clinically meaningful improvements from baseline in measures of functioning, as well as improved depressive and cognitive symptoms, sexual function and HRQoL, in patients with MDD and co-morbid GAD. These effects were sustained through 24 weeks of treatment, with statistically significant improvement from baseline observed at both week 12 and week 24. The profile of effectiveness was similar to that observed in the overall patient population of the RELIEVE study (Mattingly et al., 2022).
The primary outcome of this study – patient functioning – is known to be significantly impaired in patients with MDD, which has a profound impact on daily life and thus is an important treatment goal (Habert et al., 2016; IsHak et al., 2016; Lam et al., 2016; Oluboka et al., 2018; Sheehan et al., 2017). Despite there being slightly higher (worse) mean SDS scores at baseline in the co-morbid GAD subgroup compared with the overall study population of RELIEVE, functional improvements from baseline were similar, and sustained in the long term (24 weeks) (Mattingly et al., 2022). Interestingly, it also should be noted that in the co-morbid GAD subgroup, vortioxetine was most commonly administered as third-line treatment to patients (38%), in contrast to the overall study population in which vortioxetine was most often a first-line treatment (44%). This may reflect the fact that vortioxetine does not have an indication for the treatment of GAD and that patients receiving initial treatment would traditionally be prescribed a SERT, followed by a different SERT or SERT and NET, which have indications in both MDD and GAD.
The effectiveness of vortioxetine in improving both cognitive symptoms (PDQ-D-5) and cognitive performance (DSST) is another interesting finding of the current analysis, as clinical data on these outcomes are limited in MDD – and even more so in patients with co-morbid GAD – despite cognitive impairment being increasingly recognized as a key therapeutic target in depression (Mattingly et al., 2016). Moreover, cognitive symptoms have been shown to significantly influence the daily functioning of patients with MDD (Chokka et al., 2019a; Hammer-Helmich et al., 2018; Haro et al., 2019; Mattingly et al., 2016; McIntyre et al., 2015; Wang et al., 2020), with evidence that cognitive impairments (e.g. selective attention and working memory) in GAD may also correlate with everyday functioning (Butters et al., 2011; Yang et al., 2015). Cognitive symptoms and performance have also been linked to the extent of functional recovery and subsequent quality of life in patients with MDD (Jaeger et al., 2006; Saragoussi et al., 2018). Before the current analysis, only the recent 8-week, open-label RECONNECT study had examined the effectiveness of vortioxetine on cognitive function in patients with MDD and co-morbid GAD (severe disease) and, consistent with our findings in the RELIEVE subgroup analysis, demonstrated effectiveness by significantly improving cognitive function (Christensen et al., 2022).
The severity of anxiety symptoms has also been associated with functional outcomes in patients with MDD (Chokka et al., 2020, 2021). Moreover, as cognitive issues such as impaired attention and concentration deficits are a common feature of anxiety (Vytal et al., 2012), due to symptom overlap the treatment/relief of anxiety is likely to contribute to cognitive improvement in MDD, and thus overall treatment benefits. Vortioxetine has previously demonstrated efficacy in treating patients with MDD and high levels of anxiety, reducing symptoms of depression and anxiety, as well as delivering long-term improvements in cognitive symptoms and patient function (including work productivity) (Baldwin et al., 2016; Chokka et al., 2019b, 2021). It has also improved symptoms of anxiety, depression and functional impairment in clinical studies of patients with GAD (Bidzan et al., 2012; Christensen et al., 2019), and in patients with MDD and co-morbid anxiety disorders (GAD and SAD) (Christensen et al., 2022; Liebowitz et al., 2017). Thus, beneficial effects on symptoms related to anxiety and to cognition are likely to contribute, in part, to improvement in functioning. The present study complements these findings, suggesting an overall favourable impact of vortioxetine on functioning, cognition and depression, alongside improvements from baseline in HRQoL and work productivity, in a population of patients with MDD and co-morbid GAD in routine clinical practice.
Vortioxetine was well tolerated in the long term (24 weeks) in patients with MDD and co-morbid GAD in this analysis, displaying a tolerability and safety profile consistent with that of the overall RELIEVE study population (Mattingly et al., 2022). The incidences of AEs and common events such as nausea appeared slightly lower in this real-world analysis than in the safety profile established for vortioxetine in randomized, controlled trials (Boulenger et al., 2014; McIntyre et al., 2014). However, as real-world assessments were made only at baseline and 12 and 24 weeks, it is possible that some AEs, including early events that resolved quickly, may not have been captured. The real-world nature of this investigation, with minimal study intervention, is, nevertheless, also a notable strength. Inclusion of a heterogeneous, co-morbid anxiety study population, comprising outpatients aged ≥18 years (including the elderly, aged >65 years), with no restrictions on concomitant medications, means that all outcomes (including tolerability and safety) are reflective of a broad patient exposure and generalizable to patients receiving treatment in routine clinical practice.
In general, the limitations of this study are those inherent to a real-world study, with no control/comparator group or intensive monitoring. The non-interventional design, with study assessments at baseline, 12 and 24 weeks, meant that the study was not able to examine onset of effectiveness or early development/resolution of AEs. In addition, MDD and GAD were not diagnosed according to standardized study criteria, but instead relied upon local criteria and physicians’ judgements, reflecting the situation in everyday clinical practice. Interestingly, patients from the North American study sites (USA and Canada) accounted for a greater proportion of patients in the co-morbid GAD subgroup (82%) than in the overall RELIEVE study population (44%). This may reflect a general difference in diagnostic practices for GAD compared with the European sites (France and Italy), and/or the potential for anxiety symptoms to be encompassed within a diagnosis of MDD in European practice – although between-country differences in the prevalence of mental disorders have been demonstrated previously (Kessler et al., 2007; Scott et al., 2018). Estimates of the prevalence of GAD in patients with MDD vary widely across studies and cohorts (Zhou et al., 2017). The overall prevalence of GAD in patients with MDD observed in the RELIEVE study (24.4%, at a mean 11.2 years since MDD diagnosis) is consistent with the findings of the international World Health Organization World Mental Health survey (McGrath et al., 2020).
Another potential limitation is that 413 patients (56%) in the overall RELIEVE study population had co-morbid anxiety, including 203 patients with anxiety symptoms of ‘unspecified type’. As it was planned that only those patients with a diagnosis of GAD were included in this analysis, the impact of vortioxetine treatment in addressing symptoms in patients with co-morbid anxiety may have been underestimated. Moreover, because Hamilton Anxiety Rating Scale scores were not recorded in this study, the severity of anxiety symptoms in these patients at baseline, and during treatment could not be followed. Finally, due to the COVID-19 pandemic, a very small number of patients did not complete their visits or completed their visits remotely. However, a sensitivity analysis (data not shown) found that this change of study conduct did not have a significant impact on the study results.
In summary, patients with MDD and co-morbid GAD who were treated with vortioxetine in this real-world study experienced clinically meaningful improvements from baseline in overall functioning over a 6-month period. In addition, benefits were observed in patients’ depressive symptoms, cognitive symptoms, cognitive performance and HRQoL compared with baseline status, and the tolerability of vortioxetine was consistent with its known profile. These findings from routine clinical practice suggest that treatment with vortioxetine may benefit patients with MDD and co-morbid GAD, and could merit further investigation in controlled clinical studies.
Footnotes
Acknowledgements
Additional statistical analysis was provided by Daniel Oudin Åstrom (H. Lundbeck A/S). Medical writing assistance was provided by Juliet George for Piper Medical Communications, funded by H. Lundbeck A/S.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SA has received a research grant from AstraZeneca and consulting/advisory board/honoraria fees from Janssen, Lundbeck, Novartis, Angelini, AstraZeneca, BMS, Krka, Lilly, Grünenthal, Tecnifar, Servier, and MDS.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: H. Lundbeck A/S funded the research reported in this article and the medical writing assistance.
MCC and MA are employees of H. Lundbeck A/S. KS was an employee of H. Lundbeck A/S at the time of the data analysis.
