Abstract
Background:
Circadian rest-activity rhythm disorders, affecting up to 60% of patients with advanced cancer, are associated with poor clinical outcomes. As disease severity correlates with circadian disruption, a multi-modal approach may be necessary to improve circadian rhythmicity in this population.
Aim:
To assess the feasibility, primarily in relation to recruitment, retention, usability and acceptability, of a multi-modal, non-pharmacological intervention in patients with advanced cancer and a circadian rest-activity rhythm disorder.
Design:
A prospective, single arm, outpatient feasibility study took place over 8-weeks incorporating a clinical review, daily bright light therapy, online cognitive behavioural therapy for insomnia course, and personalised physical activity plan.
Settings/participants:
Adult (⩾18 years old) outpatients from an acute hospital and hospice service with locally advanced or metastatic cancer and a circadian rest-activity rhythm disorder.
Results:
Participants (n = 20) completed questionnaires (80%) and accelerometry (75%) and engaged with the CBT-I course (100%), bright light therapy (80%) and physical activity plan (100%). Participants reported acceptability and usability of the intervention, with no intervention-related adverse events noted. Preliminary improvements were seen in scores for fatigue (p = 0.003) and insomnia (p = 0.021), and symptom-related distress scores (all p < 0.01). Scores also improved for social (p = 0.001), cognitive (p = 0.006), emotional (p = 0.012), and physical (p = 0.046) functioning.
Conclusions:
The study demonstrated acceptability, usability and preliminary efficacy for the intervention, and warrants further investigation with a definitive trial to assess the impact on clinical outcomes of interest.
Keywords
Circadian rest-activity rhythm disorders are common in patients with cancer, particularly in advanced disease.
Non-pharmacological interventions have demonstrated small-to-medium size effects at improving circadian rhythmicity in patients with cancer.
It is unknown if a multi-modal, non-pharmacological approach is feasible in patients with advanced cancer and whether it can improve circadian rhythmicity and/or clinical outcomes.
A multi-modal intervention consisting of a clinical review, personalised activity plan, bright light therapy and a cognitive behavioural therapy for insomnia course was feasible in patients with advanced cancer.
The multi-modal intervention supports preliminary efficacy with improved patient reported measures (symptom burden, symptom-related distress and quality of life) and to a lesser degree, objective circadian rhythmicity.
The study supports theoretical frameworks that suggest structured activity can reduce symptoms such as fatigue and insomnia, and psychological burden.
The study provides encouraging evidence that multi-modal, digital and behaviourally oriented interventions can be incorporated into routine care for patients with advanced cancer and supports policy development into embedding non-pharmacological interventions into management pathways.
With acceptability, usability and impact of digital therapeutics, such as CBT-I, increased funding and infrastructure support is justified.
Introduction
Circadian rest-activity rhythms, describing daily patterns in rest and physical activity, are disrupted in up to 60% of patients with advanced cancer and have implications for symptom burden, quality of life and prognosis. 1 Patients may demonstrate nighttime restlessness alongside daytime inactivity, or a lack of similarity in rest and activity levels between days. 2 Interventions targeting circadian rest-activity rhythm disorders aim to improve the regularity of rest-activity rhythms between days, increase daytime physical activity and/or reduce nighttime physical activity. Similar to insomnia, although pharmacological options exist, there is little evidence to support their efficacy and tolerability in the management of sleep disturbance and circadian disruption.3,4 Hence, effective non-pharmacological options should be considered first-line. As circadian rest-activity rhythm disruption progresses with advanced disease, individual non-pharmacological interventions lead to small-to-medium size effects, and combined treatments can act synergistically, it was postulated that a multi-modal approach may be required to overcome abnormal rhythmicity in those with advanced cancer.5–8 Reversible factors, such as anaemia, medication and lifestyle factors (i.e. alcohol and caffeine intake), should also be considered.
The presence, and absence, of light is the strongest factor in aligning internal circadian rhythms with the environment. 7 Bright light therapy is a simple, easy to use, and inexpensive intervention that is not associated with tolerance. 7 In patients with cancer, bright light therapy combined with sleep hygiene advice can significantly improve insomnia symptoms, sleep quality and time spent asleep whilst in bed.7,9 Bright light therapy can also stabilise rest-activity rhythms and consolidate sleep.9,10
Sleep disorders are common in patients with cancer and impact on the restfulness of sleep and the ability to be physically active the following day. 11 Cognitive behavioural therapy for insomnia (CBT-I) utilises five approaches: (i) limiting exposure to stimuli before bed (‘stimulus control’); (ii) restricting time in bed not sleeping (‘sleep restriction’); (iii) sleep hygiene advice; (iv) addressing maladaptive sleep beliefs (‘cognitive restructuring’) and (v) relaxation training.12,13 CBT-I has long-lasting effects, minimal side effects, is as effective as sedative medications in the management of insomnia across cancer groups and is preferred by patients.12,14,15
Finally, up to 70% of patients with cancer adhere to international guidance on physical activity.16,17 Effective interventions to improve physical activity in cancer populations need to be personalised, address psychosocial barriers and be mindful of the limitations in this patient group.18,19 Regular physical activity has also been shown to improve sleep parameters, sleep quality, and circadian rhythmicity.20,21
Aims
This study aimed to assess the feasibility of a novel multi-modal, non-pharmacological intervention to improve circadian rest-activity rhythms in patients with advanced cancer. The study also aimed to preliminarily assess the impact of the intervention on patient-reported symptoms and quality of life and objectively measured circadian parameters.
Methods
Research questions and outcome measures
Addressing feasibility of the multimodal non-pharmacological intervention (cognitive behavioural therapy for insomnia, bright light therapy and a personalised activity plan) in people with advanced cancer and a circadian rest-activity rhythm disorder:
Is it feasible to recruit and retain people to the intervention? (Target: 30 people recruited in a 12-month period, with ⩾70% retention. Recruitment and retention rates assessed at 12-months).
Is it feasible to collect subjective (MSAS-SF, EORTC-QLQ-C30, bPSQI, ESS) and objective (wrist and thigh accelerometry) data at multiple time points? (Target: ⩾70% of assessments complete. Research assessment completion rates (baseline, end of week 4 and end of week 8) considered at 12 months.
Do people with advanced cancer and a circadian rest-activity rhythm disorder engage with the individual interventions? (Target: 100% completion of CBT-I course, ⩾ 80% reporting daily engagement with bright light therapy and the physical activity plan. Assessed weekly considering subjective participant reports for bright light therapy and physical activity, objectively monitoring Polar® watch physical activity data and objective monitoring of CBT-I course completion on the THIS WAY UP® platform).
Are the study procedures and individual interventions acceptable and usable? (Target: ⩾ 80% acceptability and ⩾ 80% usability. Assessed via participant-completed questionnaire at the end of the 8-week study, considering a seven-point Likert scale for usability and acceptability, along with positive and negative comments of study procedures and individual interventions, and any suggested changes to the study.
Is the multimodal intervention safe? (Target: no significant adverse events related to the intervention. Participants assessed weekly for the development of adverse events or new symptoms in accordance with the Common Terminology Criteria for Adverse Events (CTCAE).
Addressing preliminary efficacy:
Does the intervention demonstrate preliminary efficacy in symptom burden (particularly sleep and fatigue), quality of life and functioning scores, and objective assessments of sleep, daytime activity and circadian rhythmicity?
Design and setting
This single-arm, outpatient, feasibility study was conducted over a 12-month period recruiting patients from an acute hospital and hospice service in the Republic of Ireland.
Population and recruitment
Patients with locally advanced or metastatic cancer and a circadian rest-activity rhythm disorder were identified in a related observational study. 1 Circadian rest-activity rhythm disorders (Table 1) were diagnosed, according to recently developed guidelines, following a 72-h mid-week period of physical activity monitoring using wrist MicroMotionlogger® (Ambulatory Monitoring Inc, US) and thigh ActivPal™ (PAL technologies, UK) accelerometers. 2 Participants also completed a study-specific sleep and activity diary. Accelerometry data was analysed using dedicated software (Action-W 2.7 3401, Action-4 Version 1.16 and PALanalysis v8.11.8.75). This assessment was repeated during the final week of the study and considered daytime physical activity (steps, sit to stand, sitting, standing and active time), nighttime physical activity (sleep efficiency, sleep onset latency, wake after sleep onset), proportional day to nighttime physical activity (dichotomy index, I < O), and regularity in rest-activity levels between days (24-h autocorrelation coefficient, r24).
Diagnostic criteria for circadian rest-activity rhythm disorders (CARDs) in patients with cancer.

Eligible patients (Table 2) were invited to take part in this follow-on study, provided with a patient information sheet and the opportunity to ask questions, prior to completing written consent.
Inclusion and exclusion criteria.

Due to being a known modifier of circadian rhythms.
Due to potential adverse events when engaging in increased physical activity.
Due to the potential for bright light therapy to precipitate mania.
Due to the rare occurrence of increased suicidality with bright light therapy.
Due to the reduce effect of bright light therapy.
Sample size
We aimed to recruit 30 patients. A median sample size of 30 has been demonstrated in previous palliative care feasibility studies. 22 A formal sample size calculation was not performed.
Intervention
The study involved four interventions:
(i) A clinical review at baseline and week two considered uncontrolled symptoms, blood abnormalities, lifestyle factors and medication, which may contribute to circadian disruption. Clinical teams were contacted and a management plan put in place where relevant.
(ii) A CBT-I self-guided online programme (THIS WAY UP®, St Vincent’s Hospital, Sydney). The 4-lesson programme, with a mandatory 5-day waiting period between lessons, combined psychoeducation with evidence-based skills and strategies. Prior to each lesson, participants completed the Kessler Psychological Distress Scale (K-10 23 ), which assesses psychological distress over the past 2 weeks, and the Insomnia Severity Index (ISI 24 ), which assesses sleep quality, distress from sleep loss and functional impairment over the past 2 weeks.
(iii) Daily morning bright light therapy using the Vitamin L® light box (Lumie, UK) which provides 10,000 lx at 20 cm and 2500 lx at 50 cm. Participants were advised to place the light box within arm’s reach and in a direction whereby light reached the face and eyes. Participants were advised to use the light box on waking for at least 30 min.
(iv) A personalised activity plan and a Polar Unite® watch (Polar, UK). The watch alerts the wearer to prolonged sedentary activity and provides real-time feedback on steps, daily activity, heart rate, and sleep. At the initial meeting, interests and barriers to physical activity, and short- and long-term goals were established. Goals were co-created with the participant and reviewed weekly aiming to gradually increase physical activity levels and reduce sedentary behaviour.
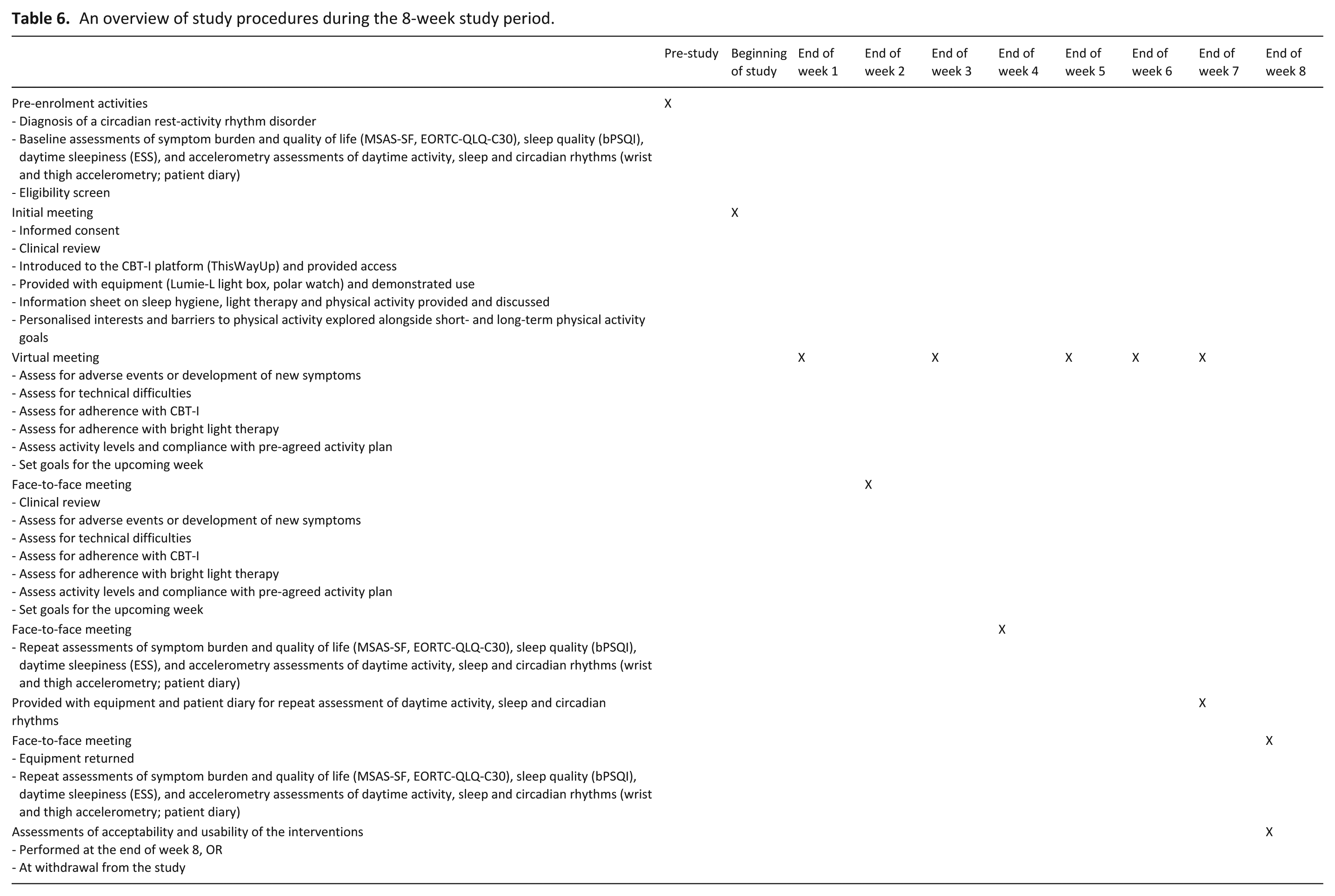
Study procedures
An overview of study procedures is detailed in Appendix. Eligible participants met with the research team, provided informed consent and received relevant equipment and information sheets detailing the intervention. Four face-to-face meetings occurred (baseline, and ends of weeks 2, 4 and 8) with virtual weekly follow up in between. Participants completed questionnaires on symptoms (Memorial Symptom Assessment Scale – Short Form, MSAS-SF 25 ), quality of life (European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire, EORTC CQC-C30 26 ), sleep quality (brief Pittsburgh Sleep Quality Index, bPSQI 27 ) and daytime sleepiness (Epworth Sleepiness Scale, ESS 28 ) at face-to-face meetings in healthcare settings or the participant’s home at baseline, and the end of weeks 4 and 8. Weekly contact (via telephone, text or email) assessed: (1) Adverse events or development of new symptoms; (2) Technical difficulties; (3) Adherence with interventions CBT-I; 4) Goal setting for the upcoming week. Assessments of sleep, daytime activity and circadian rhythmicity were repeated during the final week of the study.
Data analysis
IBM®SPSS® statistic version 29.0.1.1 (244) software was used for the analysis which included descriptive statistics for participant characteristics, recruitment rates, retention rates, engagement with interventions, completion of research reviews and assessments, adverse events and clinical outcomes. Content analysis was used for participant reported interests and barriers to physical activity. One-way repeated measures ANOVA and the Friedman test was used to consider the significance of change in clinical outcomes over time, with a significance threshold of p < 0.05.
Ethical issues and approvals
Ethical approval was granted by the joint St James’ Hospital – Tallaght University Hospital Joint Research Ethics Committee (ID 1926). The study was registered at ClinicalTrials.gov (NCT06329479).
Results
Primary outcomes
Recruitment and retention (target 30 patients with ⩾70% retention)
The related observational study identified 39 patients with a circadian rest-activity rhythm disorder diagnosis. Twenty participants (51%; Table 3) were recruited to this feasibility study (Figure 1) and 15 (80%) completed the 8-week study.
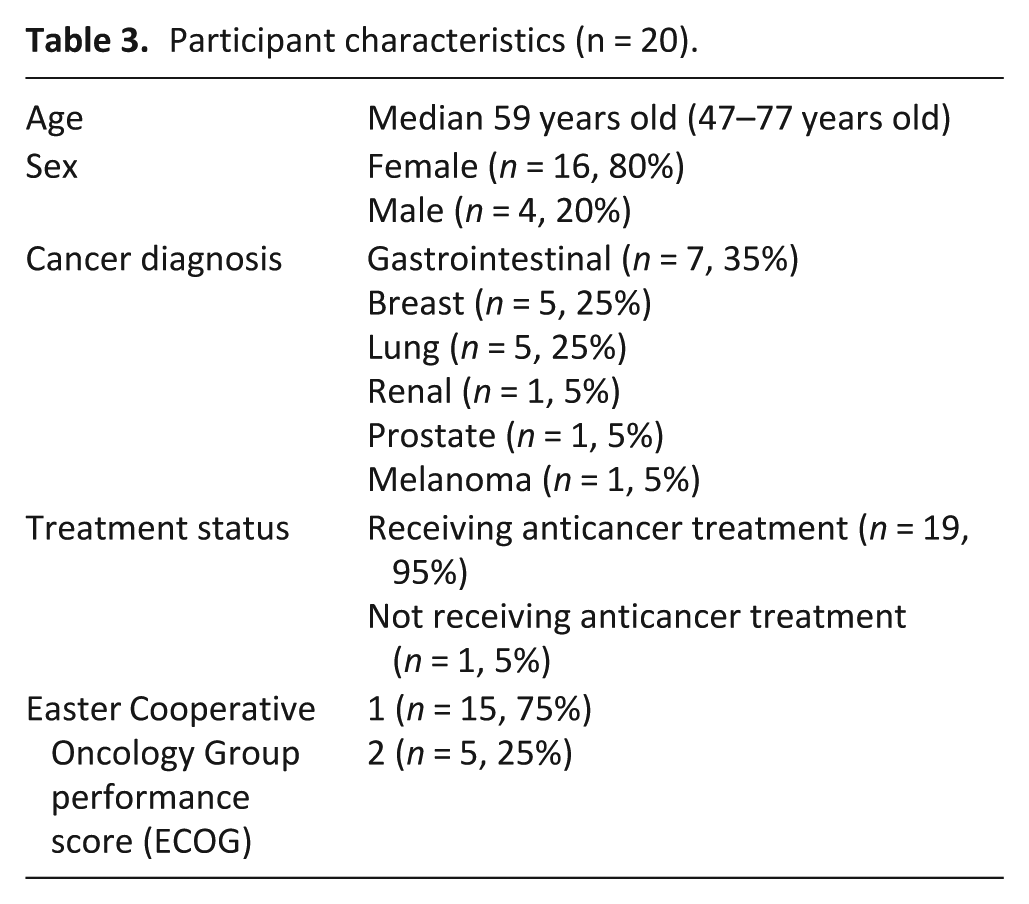
Participant characteristics (n = 20).
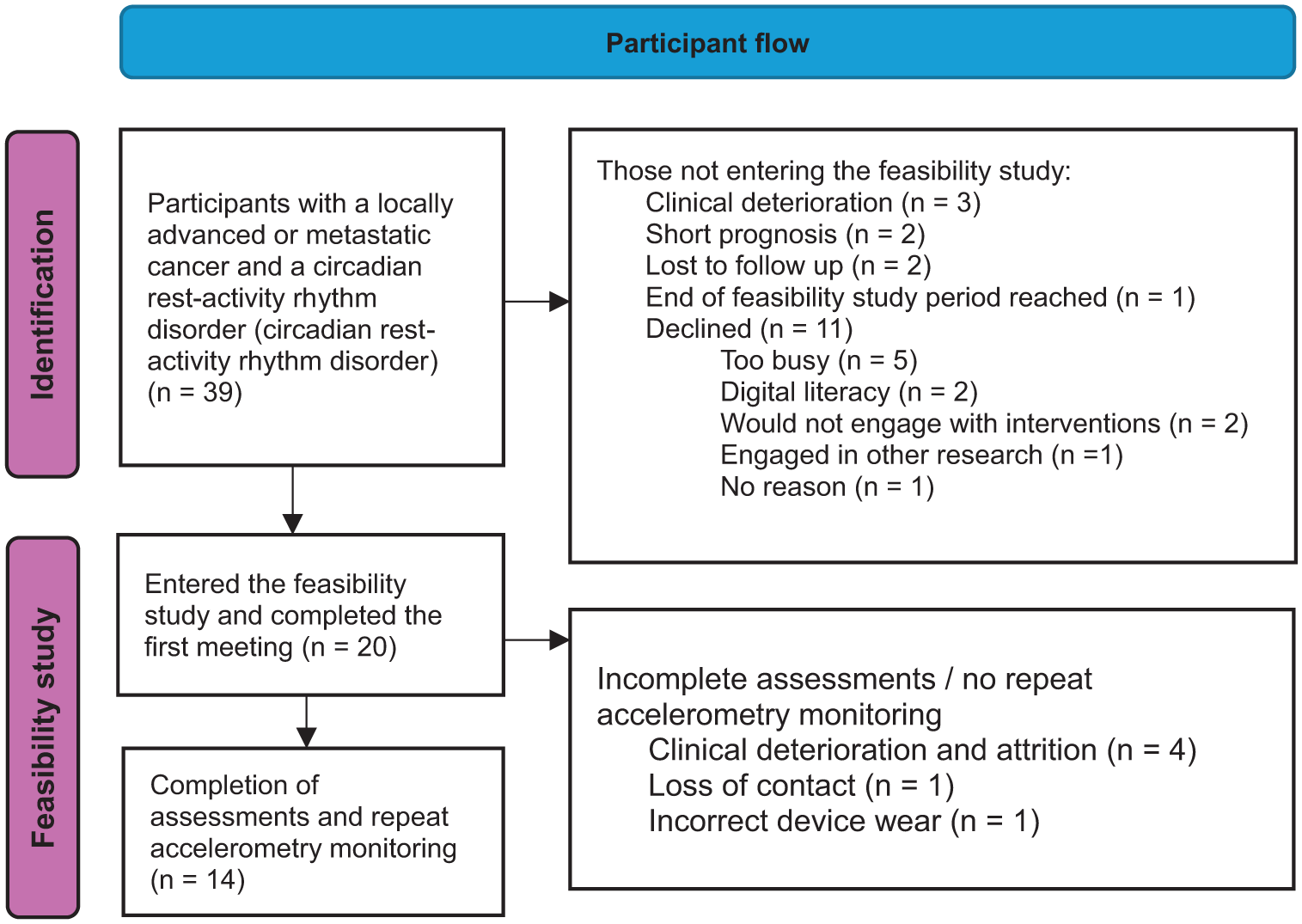
Flow chart of potential participants identified in a related observational study.
Completion of research reviews and assessments (target ⩾70% of assessments complete)
All participants (n = 20) completed questionnaires at baseline, whilst 18 (90%) completed questionnaires at the end of week four, and 15 (75%) completed the questionnaires at the end of week eight. Fourteen participants had comparative wrist and thigh accelerometry data; one participant removed the wrist accelerometer overnight during the final week of assessments, meaning there was no comparative accelerometry data.
Weekly bi-directional contact was possible for six participants (40%). The remaining participants missed one–three contacts. In this instance, goals were re-iterated, and potential improvements suggested, via email or text.
Engagement with interventions (target: 100% completion of CBT-I course, ⩾80% engaging daily with bright light therapy and the physical activity plan)
Fifteen participants (100%) completed the CBT-I course, which was completed at variable time points (week four (n = 5), week five (n = 3), week six (n = 2)), week seven (n = 3), and week eight (n = 2). Twelve participants (80%) reported engaging daily with bright light therapy; two participants (13%) reported using bright light therapy intermittently and one participant (7%) did not use bright light therapy. Fifteen participants (100%) reported engaging with the activity plan and the majority increased their physical activity levels according to watch data. Participants struggled intermittently with activity due to anticancer therapy side effects (n = 2), hospitalisation (n = 1), lack of motivation (n = 1), and the weather (n = 1).
Participant acceptability, ease of use and general feedback (target: ⩾80% acceptability and ⩾80% usability)
Participants reported that the clinical reviews and weekly contact were easy and acceptable. Participants reported that the CBT-I course was easy to use (n = 14, 93%) and acceptable (n = 15, 100%). Challenges with the CBT-I course focussed on difficulty logging in to the website. Feedback was generally positive in terms of its educational nature, particularly in relation to sleep hygiene, setting a routine and learning new techniques. Participants liked the visual displays and found the concise summaries useful. Two participants felt the course did not relate to their situation. One participant felt the course to be repetitive, one felt the associated questionnaires were burdensome, and another felt the summary sheet alone would have been sufficient. The Australian origin of the course was highlighted as a negative for two participants.
All participants (n = 15, 100%) reported bright light therapy was easy to use and acceptable. Participants found the light box to be relaxing, energising and that it improved mood, motivation and alertness. Two participants reported buying their own devices after the study. One participant felt that the light was too bright. During sunny mornings, one participant opted to be outdoors as opposed to use the light box.
Participants reported that the physical activity plan (and Polar watch) was easy to undertake (n = 14, 93%) and acceptable (n = 15, 100%). Participants noted that the watch was motivating, and helped with goal setting. Three participants had difficulty engaging with the physical activity plan due to fluctuating health.
Participant’s suggestions for a future study included a tailored CBT-I course, incorporating cognitive activity, a daily diary or tick box exercise, and reduced researcher involvement over time with increasing responsibility on the participant.
Adverse events (target: no significant adverse events related to the intervention)
Of enrolled participants, three had progression of disease leading to a discontinuation (n = 2) or change (n = 1) of anticancer therapy. One participant received planned radiotherapy. Several participants noted recurring anticancer therapy side effects impacting on sleep and daytime activity.
Six participants developed an infection during the study (presumed viral n = 3, presumed bacterial n = 3) resulting in three antibiotic prescriptions. Four participants were hospitalised with bowel obstruction (n = 1), constipation (n = 1), diarrhoea (n = 1), and infection (n = 1). One participant had a fall not requiring a medical review. No adverse events were felt to be related to the intervention.
Secondary outcomes
Participant interests and barriers to physical activity
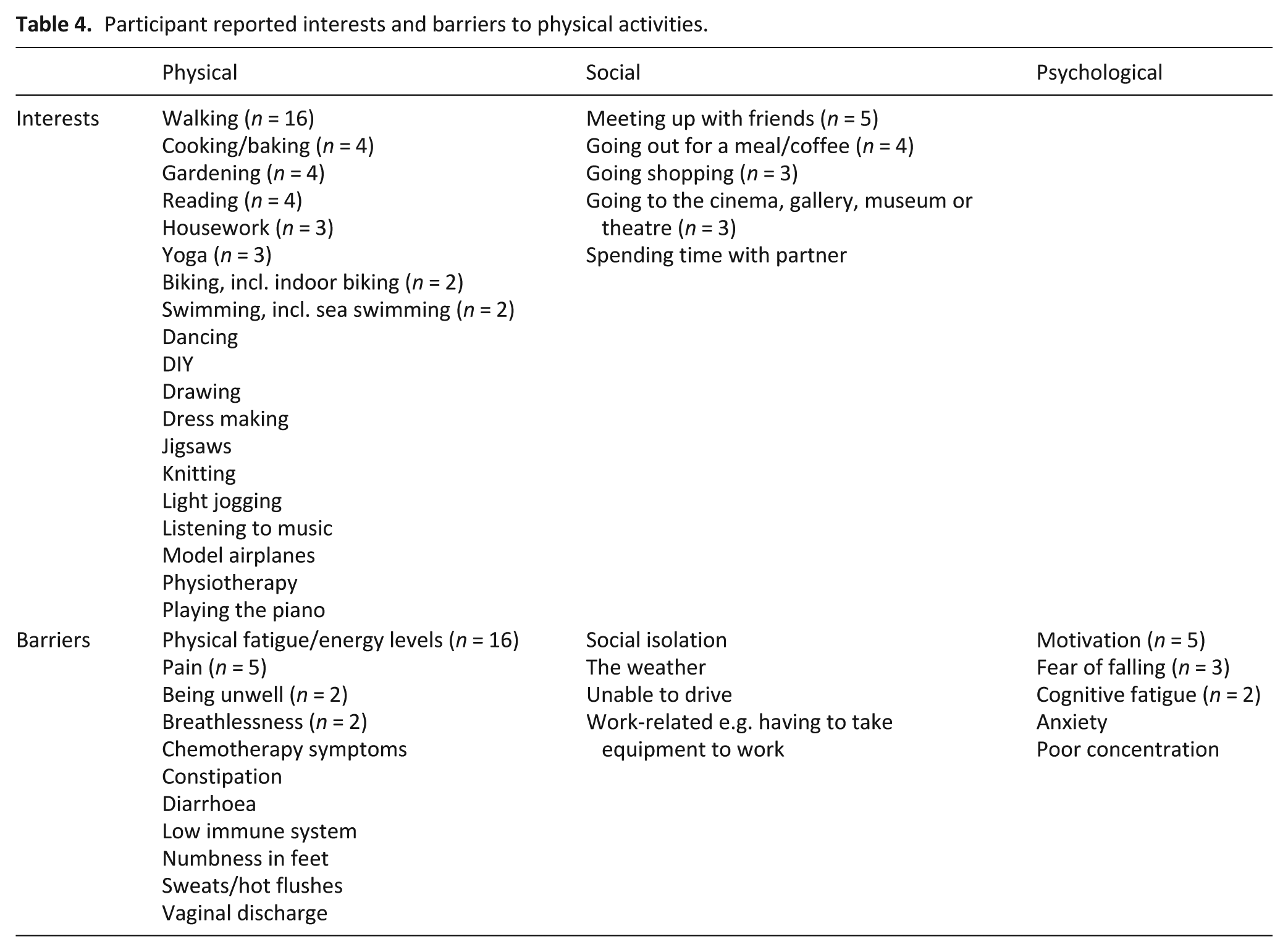
Varied interests and barriers to physical activity were noted by participants (Table 4).
Participant reported interests and barriers to physical activities.
Potentially reversible contributors
The clinical review identified the following potential contributors: uncontrolled symptoms (n = 18, 90%), medications (n = 14, 70%), or lifestyle factors (n = 8, 40%). The clinical review led to the prescription of a new medication in six participants (30%), or further assessment/investigation by the clinical team in three participants (15%).
Participant reported clinical outcomes
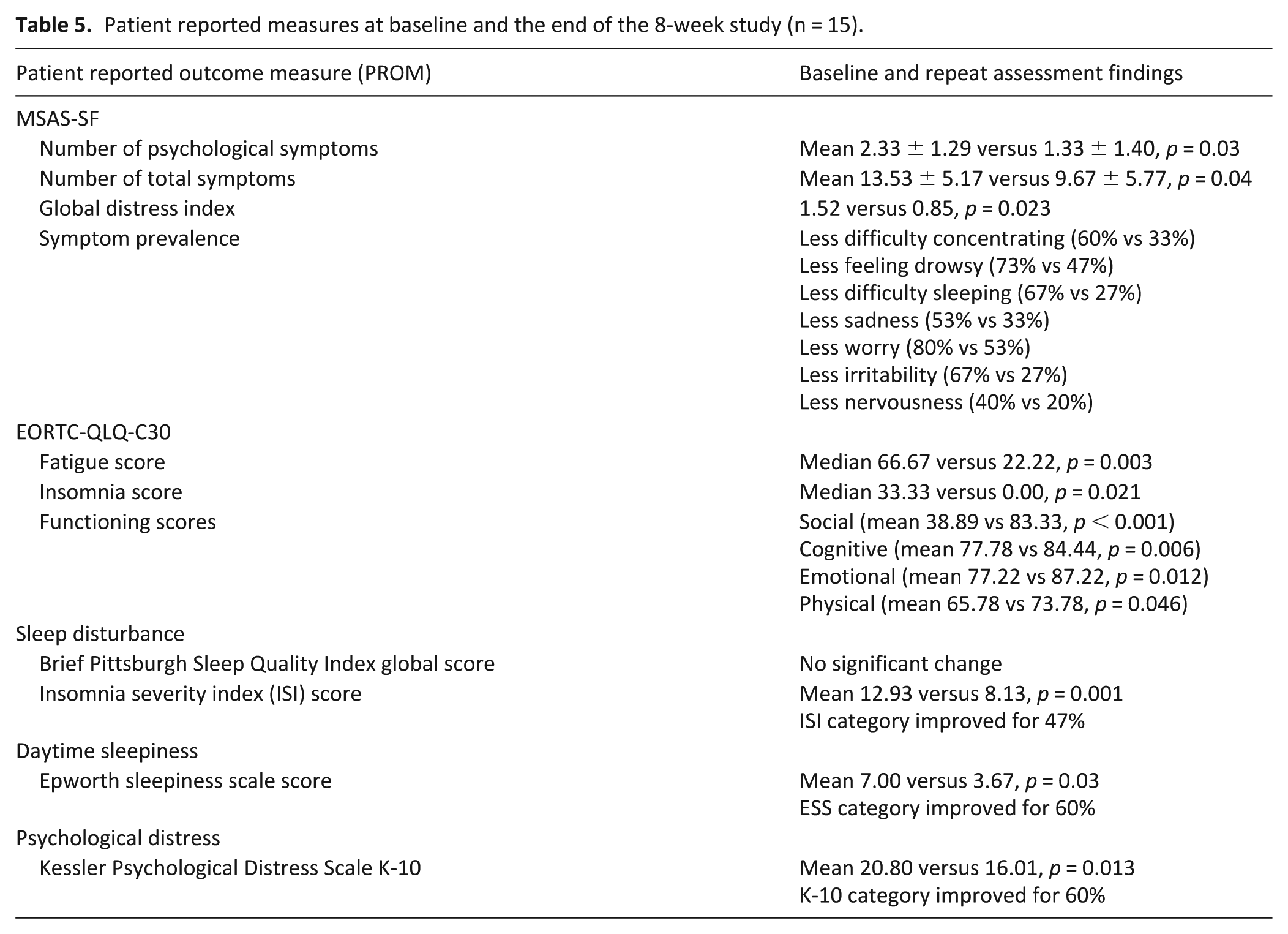
Changes between baseline and repeat patient reported outcome measures (PROMs) are demonstrated in Table 5.
Patient reported measures at baseline and the end of the 8-week study (n = 15).
Accelerometry-derived circadian parameters (n = 14)
I < O values improved for 11 participants (79%), with a reduction in mean IN bed activity for seven participants (50%) and improvement of the mean OUT of bed activity for 11 participants (79%). r24 values improved for nine participants (64%). SE values improved for seven participants (50%). At a group level, significant improvements were seen for OUT of bed activity (p = 0.045).
Discussion
Main findings
This innovative study highlights the feasibility of several aspects of this multimodal intervention in patients with advanced cancer. The findings are encouraging with reasonable recruitment (51% of eligible patients) and good retention (75% completed the study). Participants showed high engagement to the interventions (80%–100%) and completion of assessments (80%). Notably, the intervention was well-received, with participants expressing acceptance of the various components, and no trial-related adverse events were reported. Most excitingly, the study demonstrated preliminary efficacy with improvements in patient-reported outcomes.
Strengths and limitations of this study
This study offers several strengths supporting the feasibility and relevance of the intervention. The flexibly delivered study procedures tailored to participant preference (i.e. location of follow up and contact methods) demonstrates a participant-centred approach. Personalised activity plans enhanced the interventions relevance to participants, accommodating the range of participant interests in, and perceived barriers to, physical activity. The pragmatic inclusion of participants experiencing hospitalisations, which are frequent in this cohort due to fluctuating trajectories, supports the real-world applicability of the findings but may have dampened the impact of the intervention on outcomes of interest. The study is strengthened by incorporating subjective and objective measures of daytime activity, sleep and circadian rhythms, focussed on clinically relevant outcomes. The multimodal intervention is low cost, easy to upscale and suitable for delivery in outpatient settings, an increasingly valuable feature given recent drives within healthcare systems to move towards more community-based service delivery.
As most participants were female, receiving anticancer therapies and had an Eastern Cooperative Oncology Group (ECOG) score of 1, the external validity of the findings is limited and the feasibility in other patient groups remains unknown. Future research should aim to include a more diverse patient population, spanning cancer diagnoses and stages, to better understand variability in circadian rest-activity rhythm disorders and differential responses to circadian-based interventions. Although advanced disease is associated with heightened circadian disruption, it also remains unknown if circadian disruption becomes refractory to treatment (c.f. cancer-related fatigue, cancer-related anorexia), highlighting a further avenue for investigation.
Of note, factors influencing circadian rhythmicity, such as anticancer therapy delivery and clinical deteriorations, vary significantly between individuals and cancer groups. 29 Capturing patients at comparable timepoints, in relation to external factors, is difficult and more continuous accelerometry monitoring may be a more appropriate measure in a definitive study. Furthermore, seasonal variation in sunlight exposure, temperature and physical activity patterns warrant consideration to ensure balanced group allocation and accurate interpretation of outcomes in future studies.30,31
While objective engagement measures were collected for CBT-I and physical activity, bright light therapy engagement was self-reported and may be influenced by reporting bias. Additionally, participant’s pre-morbid physical activity levels and self-motivation, which undoubtably influence engagement in the intervention and retention in the study, were not assessed prior to entering the study and should be considered in a definitive study. 32 Including a follow-up assessment period post-intervention will also be important to evaluate the longevity of any treatment effects.
Although the study did not reach the recruitment target of 30, recruitment and retention rates are similar to other studies within this population. 33 Limitations stemmed from a single-researcher model, clinical deteriorations, and a fixed 12-month study period. Developing effective screening tools to identify circadian rest-activity rhythm disorders, purposive sampling patients with circadian rest-activity rhythm disorder-associated symptomatology, utilising multi-site delivery of the study and/or a longer period of recruitment may improve recruitment.
Finally, while consistent weekly contact was not possible for most participants, the study revealed valuable insight into participant preferences. Many expressed a growing desire for autonomy and self-directed participation as the study progressed, allowing the future study design to build participant independence while maintaining appropriate levels of support.
What this study adds
Research has demonstrated the positive effect of the individual interventions used within this study on sleep, daytime physical activity and circadian rhythmicity.34,35 This study highlights the feasibility of a multi-modal intervention. Although preliminary efficacy of the intervention for patient-reported outcomes and objective measures has been demonstrated, the sample size limits the ability to detect significant change, and a definitive study is required to assess this further. A recent study comparing multimodal therapy (sleep education and psychoeducation), multimodal therapy combined with aerobic exercise and aerobic exercise alone in breast cancer survivors, demonstrated superiority of multimodal and multi-modal combined groups with respect to sleep quality. 36 Future work should consider multiple arm trials, including standard care control arms, with randomisation to explore the contribution of individual interventions further. Alternative non-pharmacological interventions, such as controlled dietary habits, and pharmacological approaches for the management of circadian rest-activity rhythm disorders should also be explored.
Footnotes
Appendix
An overview of study procedures during the 8-week study period.
| Pre-study | Beginning of study | End of week 1 | End of week 2 | End of week 3 | End of week 4 | End of week 5 | End of week 6 | End of week 7 | End of week 8 | |
|---|---|---|---|---|---|---|---|---|---|---|
| Pre-enrolment activities - Diagnosis of a circadian rest-activity rhythm disorder - Baseline assessments of symptom burden and quality of life (MSAS-SF, EORTC-QLQ-C30), sleep quality (bPSQI), daytime sleepiness (ESS), and accelerometry assessments of daytime activity, sleep and circadian rhythms (wrist and thigh accelerometry; patient diary) - Eligibility screen |
X | |||||||||
| Initial meeting |
X | |||||||||
| Virtual meeting |
X | X | X | X | X | |||||
| Face-to-face meeting |
X | |||||||||
| Face-to-face meeting |
X | |||||||||
| Provided with equipment and patient diary for repeat assessment of daytime activity, sleep and circadian rhythms | X | |||||||||
| Face-to-face meeting |
X | |||||||||
| Assessments of acceptability and usability of the interventions |
X |
Acknowledgements
The authors would like to thank clinical teams at Our Lady’s Hospice & Care Services (Dublin) and St James’ Hospital (Dublin) for their contribution in identifying appropriate patients for the study.
Ethical considerations
Ethical approval was granted by the joint St James’ Hospital – Tallaght University Hospital Joint Research Ethics Committee (ID 1926).
Author contributions
CG was the lead researcher in the development of the study protocol, participant recruitment and completion of study procedures, data analysis and drafting of the manuscript under the supervision of AD. CG and AD reviewed and agreed on the final manuscript for publication.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Professor Andrew Davies’ research fund. No external funding was received.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Requests for data sharing should be made to the Academic Department of Palliative Medicine, Our Lady’s Hospice & Care Services, Dublin, Ireland (
