Abstract
Background:
About 25% of palliative medication incidents involve continuous subcutaneous infusions. Complex structural and human factor issues make these risk-prone interventions. Detailed analysis of how this safety-critical care can be improved has not been undertaken. Understanding context, contributory factors and events leading to incidents is essential.
Aims:
(1) Understand continuous subcutaneous infusion safety incidents and their impact on patients and families; (2) Identify targets for system improvements by learning from recurrent events and contributory factors.
Design:
Following systematic identification and stratification by degree of harm, a mixed methods analysis of palliative medication incidents involving continuous subcutaneous infusions comprising quantitative descriptive analysis using the
Setting/participants:
Palliative medication incidents (n = 7506) reported to the National Reporting and Learning System, England and Wales (2016–2021).
Results:
About 1317/7506 incidents involved continuous subcutaneous infusions with 943 (72%) detailing harms. Primary incidents (most proximal to patient outcomes) leading to inappropriate medication use (including not using medication when it was needed) were underpinned by breakdowns in three major medication processes: monitoring and supply (405, 31%), administration (383, 29%) and prescribing (268, 20%). Recurring contributory factors included discontinuity of care within and between settings, inadequate time, inadequate staffing and unfamiliarity with protocols. Psychological harms for patients and families were identified.
Conclusions:
System infrastructure is needed to enable timely supply of medication and equipment, effective coordinated use of continuous subcutaneous infusions, communication and continuity of care. Training is needed to improve incident descriptions so these pinpoint precise targets for safer care.
Keywords
The third World Health Organization Global Patient Safety Challenge ‘Medication without harm’, emphasises the need for improved infrastructure through better reporting practices and cross-organisational learning from adverse events and near misses.
Medication is implicated in one-fifth of serious palliative care patient safety incidents, with approximately 25% of these incidents involving continuous subcutaneous infusions.
Inadequate analysis of continuous subcutaneous infusions as safety-critical, risk-prone interventions dependent on complex structural and human factor issues is a lost opportunity for learning.
Continuous subcutaneous infusion incidents occur across all settings including the home, hospices and hospitals and particularly after the transfer of patients between settings with harm present in nearly three-quarters of reports.
Multiple points of system failure were identified in continuous subcutaneous infusion incident reports including monitoring and supply (405, 31%), administration (383, 29%) and prescribing (268, 20%); recurring contributory factors included discontinuity of care within and between care settings, inadequate time, inadequate staffing and unfamiliarity with protocols.
Narrative descriptions of psychological and social harm, alongside physical harm risk, are not being adequately recognised or responded to through existing approaches to measure harm in palliative care, hindering learning in practice
The structural changes needed to minimise harm and maximise safety in palliative care are likely to be replicated in other parts of the world where patient safety reporting practices are less well established, for example, shifting from focussing on lack of experience and competency at an individual practitioner-level to addressing deficits in working environments and infrastructures for care provision.
When patients move between care locations, more attention should be given to the timeliness and effective transfer of medication management (e.g. if someone is discharged from hospital to a care home that rarely uses continuous subcutaneous infusions for palliative care, this needs to be preceded by refreshing staff skills and ensuring they can access further community support if needed).
Professional training and further research are needed to increase quality of reporting of psychological and social harms (including for families and other stakeholders involved) to facilitate organisational learning and pinpoint precise targets for further improvement.
Background
Unsafe healthcare contributes significantly to global morbidity and mortality.1,2 The third World Health Organization Global Patient Safety Challenge ‘Medication without harm’, aimed to halve severe, avoidable harms to patients due to medicines by 2022, emphasising the need for improved infrastructure through better reporting practices and cross-organisational learning from adverse events and near misses.
Medication is implicated in one-fifth of all serious patient safety incidents reported from palliative care, with patients at home disproportionately affected.3,4 Patients receiving palliative care often require considerable input at home5 –7 but incidents also occur in hospitals, hospices and during transfers between settings.3,8 Multiple professional roles in all settings3,8,9 make coordination essential for safe care. Prescribing is widely researched but is not necessarily the most incident-prone medication process.10 –16
Continuous subcutaneous infusions are commonly used in palliative care when the oral route is either ineffective (e.g. ongoing vomiting or other causes of poor absorption) or not possible (e.g. patient unable to swallow) and/or when subcutaneous delivery is least burdensome and most practical (i.e. when intravenous access is either difficult or not provided for, as is common in community settings). Approximately 25% of reported palliative medication incidents involve a continuous subcutaneous infusion.3,17 Medications including strong opioids and sedatives are routinely prescribed and administered by infusion when managing symptoms with oral medication ceases to be viable or effective (see Supplemental Files: Use and definition of continuous subcutaneous infusions).5,18 Infusion devices (syringe drivers/pumps) are used globally to optimise end-of-life care. 1 These battery-powered devices are usually replenished once every 24 h unless symptom control is inadequate, necessitating earlier changes.
The use of continuous subcutaneous infusions involves multistep, complex, risk-prone processes dependent on structural and human factors.4,19 For example, assessment of the patient’s fluctuating needs, agreeing (reviewing/revising) treatment plans, prescribing, dispensing, sourcing equipment, adequate staffing and skillsets, administration and disposal.4,19,20 Steps involved in monitoring patients’ needs and titrating medications are of particular concern18,21,22 but there also remain considerable gaps in evidence regarding how and where unsafe care can occur.15,8,19,21,22,23,24,25,26,27,28,29,30
Methods
The aims of this research were to:
RQ1: To understand continuous subcutaneous infusion safety incidents and their impact on patients and families.
RQ2: To identify targets for system improvements by learning from recurrent events and contributory factors.

Design
Incident reporting systems of healthcare-associated harms and near misses are intended to aid learning and inform strategies to reduce future harm. Utilising these data can enhance understanding and learning from reported continuous subcutaneous infusion incidents in palliative care.
The National Reporting and Learning System in England and Wales is a world leader in patient safety incident reporting. NHS England encourages reporting of all incidents and near misses without a specific threshold. No demographic data are included. We used this centralised database to identify patient safety incidents from January 2016 to December 2021. It contains structured information such as incident type, harm severity, outcome and location8,24,25 and unstructured free-text prompts to describe what happened, and why and how reoccurrence could be prevented. A systematic approach was developed and applied to identify and analyse incidents. 17
A cross-sectional quantitative descriptive analysis was undertaken using the
Study dataset eligibility
Ethical approval was granted by Cardiff University School of Medicine Research Ethics Committee (Ref 19/28). Secure access to the most recently available anonymised palliative medication incidents (1st January 2016–31st December 2021) in the National Reporting and Learning System was granted by National Health Service Improvement. As individual people/parties are not identifiable, data were exempt from United Kingdom research regulations regarding informed consent.
Using previous approaches to studying incident datasets,3,8,26,29,30,32,33 we identified a sample of 7506 palliative medication incidents stratified by degree of reported harm, including those reported as no harm or harm unclear, as well as those reported as resulting in low/medium/severe harm and death (see Supplemental Files: Sample Stratification). A palliative medication was defined as a medication used for symptom control, that is with palliative intent. We used the Palliative Care Formulary alongside the standard British National Formulary.17,34,35 Incidents specific to continuous subcutaneous infusions were systematically identified using the search terms: syringe, pump, driver or continuous subcutaneous infusion; and McKinley or Graseby (the two commonest UK brands). A total of 1692 potentially relevant reports were manually screened using pre-defined exclusion criteria (Table 1) by AB; SY and BB double screened 20% and 5% respectively. Uncertainty regarding eligibility resulted in all three discussing to reach consensus.
Criteria used to identify reports containing continuous subcutaneous infusion palliative medication incidents.

Classification of reports: Coding
Each incident report was coded by AB using the
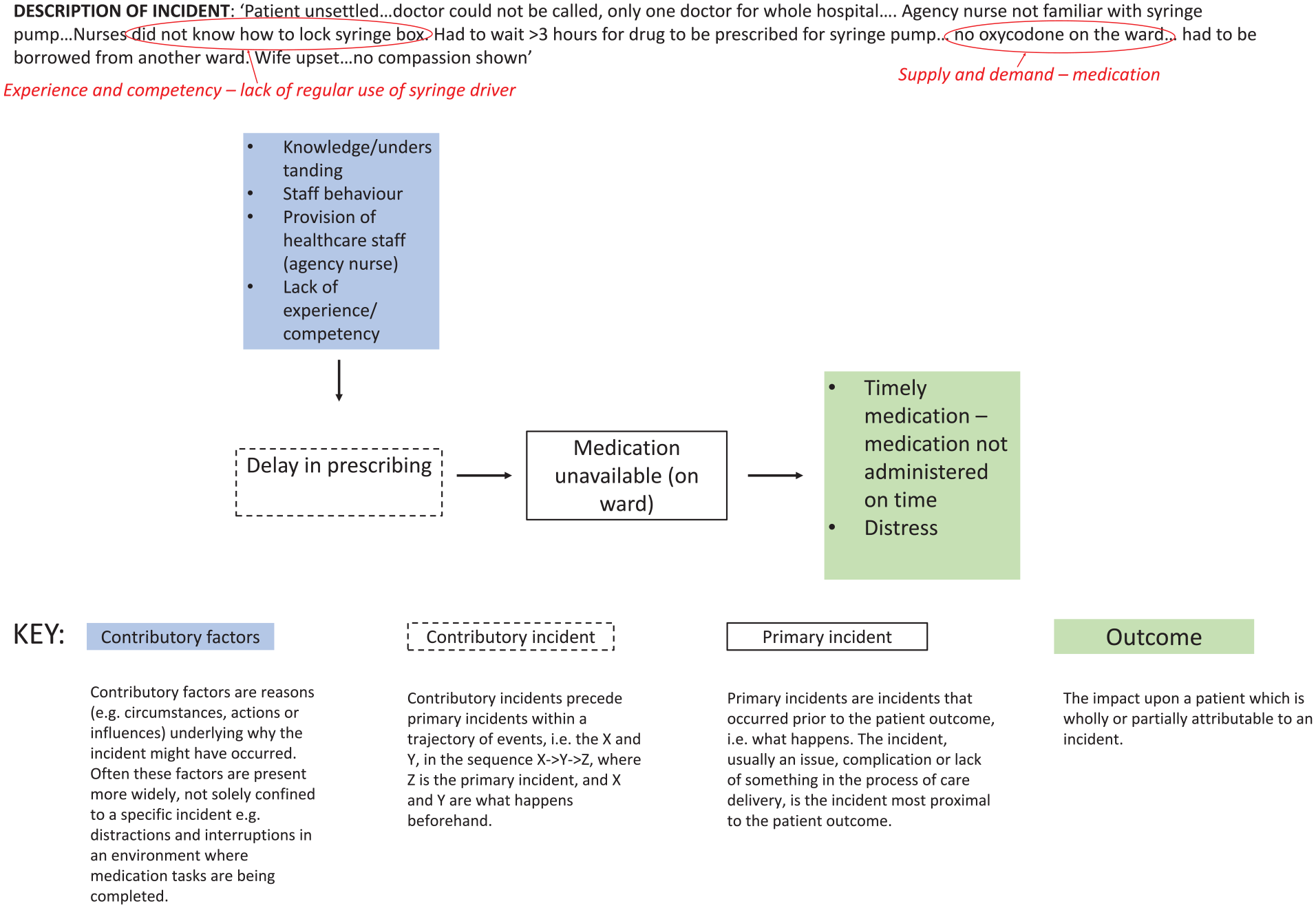
Example continuous subcutaneous infusion incident using the recursive model of incident analysis 27 and qualitative narrative analysis (plus coding/theme identification via qualitative narrative synthesis process highlighted in red.
Harm severity
Where reported harm severity clearly did not align with the free-text incident description, it was adjusted using World Health Organization International Classification of Patient Safety definitions (Table 1). 3 AB, SY, BB and ACS discussed this when information was ambiguous in individual reports. Regardless of our classification (unclear, no/low/medium/severe harm or death) all incidents were included in our full analysis to explore the learning potential (e.g. themes in contributory factors).
Qualitative narrative analysis
To identify a purposive sample for more in-depth qualitative analysis of the most salient semantic relationships between primary incident type, contributory incident type and contributory factors, a cross-tabulation (using a median frequency of 4 as a cut off) was created between:
(1) the 14 most common primary incidents and 11 recurring contributory factors;
(2) 5 contributory incidents and 9 recurring contributory factors.
This created a subsample of 129 incidents, of which 111 reports contained adequate free-text descriptions to enable a detailed thematic analysis of incident narratives (see Figure 1 for an example of this process).
AB re-read incident descriptions to iteratively identify and describe recurring patterns and themes. Further thematic analysis was carried out by SY. 36 Themes and their implications for practice were refined through critical reflection meetings of AB, SY, BB and ACS, plus weekly wider research team discussion meetings.36,37
Results
1317 reports were included in the quantitative analysis and 111 in the thematic analysis (see Supplemental Files: PRISMA diagram summarising incident handling and selection of continuous subcutaneous infusion incidents for quantitative and qualitative analysis). Double/triple screening for inclusion produced consensus without discussion in 150/158 (95%); 8 reports were discussed to reach consensus on harm severity. Abridged individual unattributed examples are provided to contextualise and illustrate the analysis, whilst retaining anonymity.
Quantitative results
Harm severity and outcomes
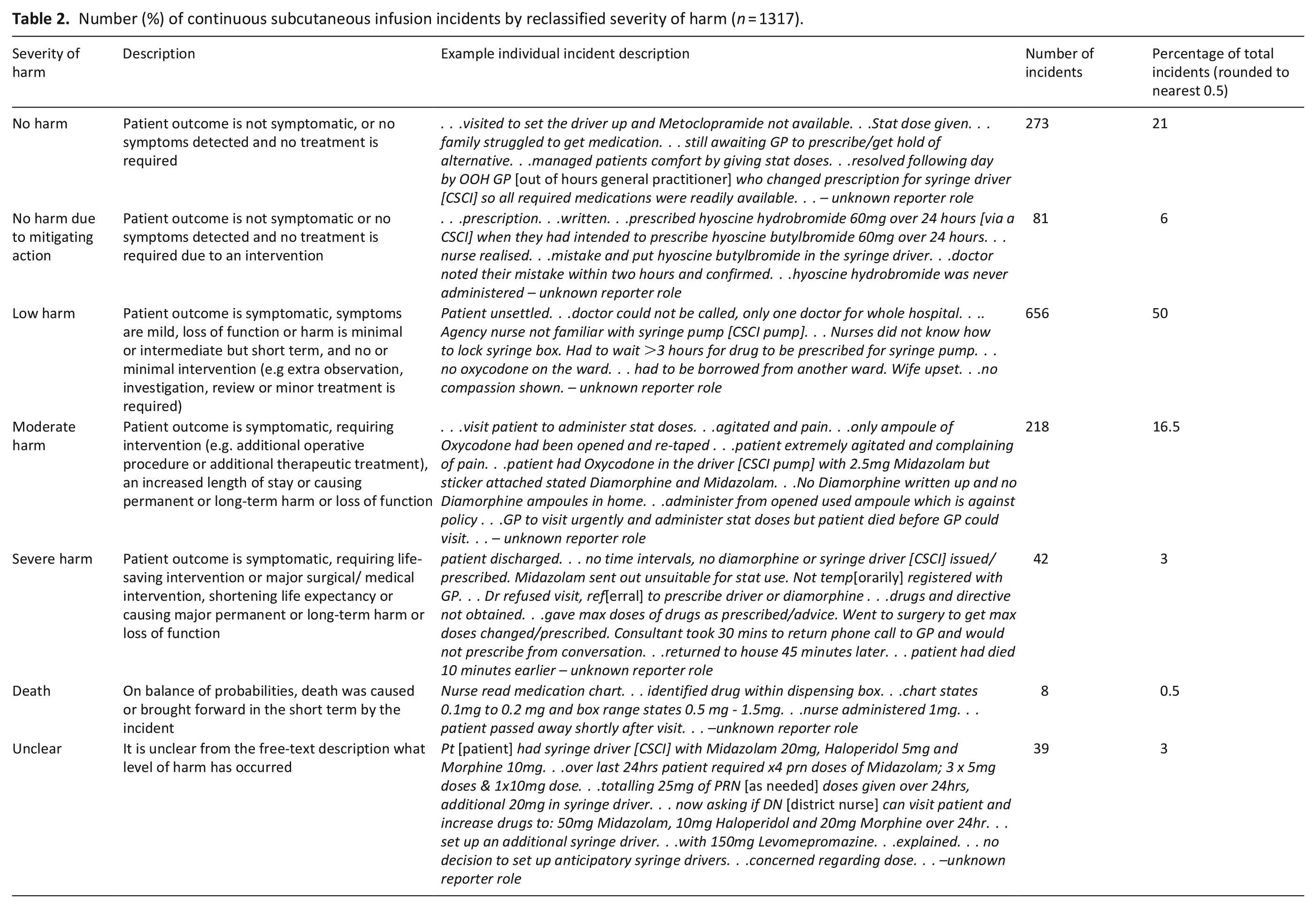
About 924/1317 (70%) of included reports were characterised by the initial reporters of incidents as resulting in harm, the other 30% as no harm. Where harm was recognised by reporters it was most commonly characterised as low. The code ‘unclear’ harm was not used by initial incident reporters and thus 39 incidents were changed to an unclear harm outcome by the research team when in our analysis (Table 2), 118 incidents (9%) were reclassified to align with World Health Organization International Classification for Patient Safety definitions. Overall reclassification led to 77 reports being upgraded, and 41 reports downgraded. Following reclassification, 924/1317 (70%) reports were now characterised as resulting in harm (including death); the other 393 (30%) were now characterised as no or unclear harm (including no harm due to mitigating action).
Number (%) of continuous subcutaneous infusion incidents by reclassified severity of harm (n = 1317).
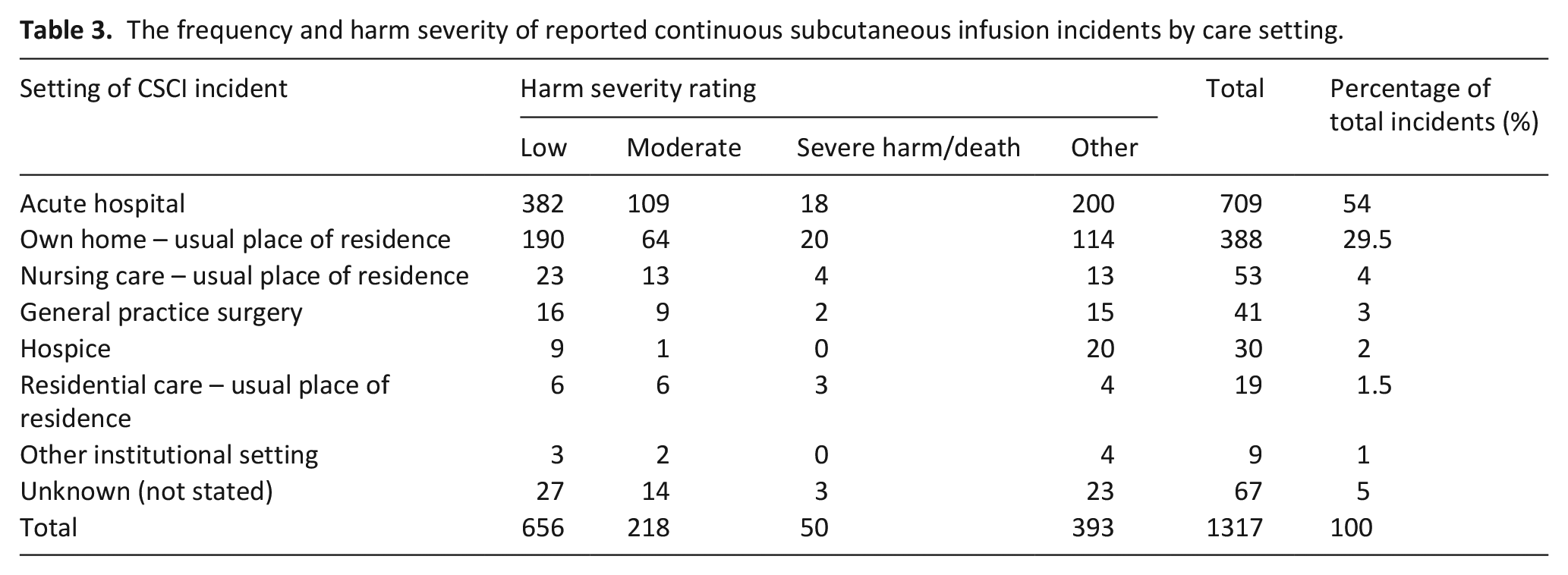
Most incidents occurred in acute hospitals or in patients’ homes (Table 3). An outcome was described in 1092 (83%) incidents, most frequently this was stated as the patient experiencing pain (195 incidents, 15%). Incidents resulting in severe harm or death in the community (the most frequent location of these levels of harm) were reviewed for common themes. This identified that most of these incidents occurred out of hours. Frequently the clinicians involved were the out of hours GP and the district nursing service, and communication between these services was impaired in many of these incidents. Discharging from the hospital frequently resulted in uncontrolled symptoms due to the lack of medications and failure to hand over the patient to the district nursing service in a timely manner.
The frequency and harm severity of reported continuous subcutaneous infusion incidents by care setting.
Process breakdowns
Three medication processes were identified as those most prone to breakdowns and resulting in a wrong dose being administered, that is, a different medication and/or giving too much or too little medication (Table 4). These medication processes were:
• administration (383 incidents of wrong doses administered in 115 cases);
• monitoring and supply (405 incidents with wrong doses subsequently administered in 117 cases) and;
• prescribing (268 incidents with wrong doses subsequently administered in 5 cases).
Number of incidents by main medication process involved.
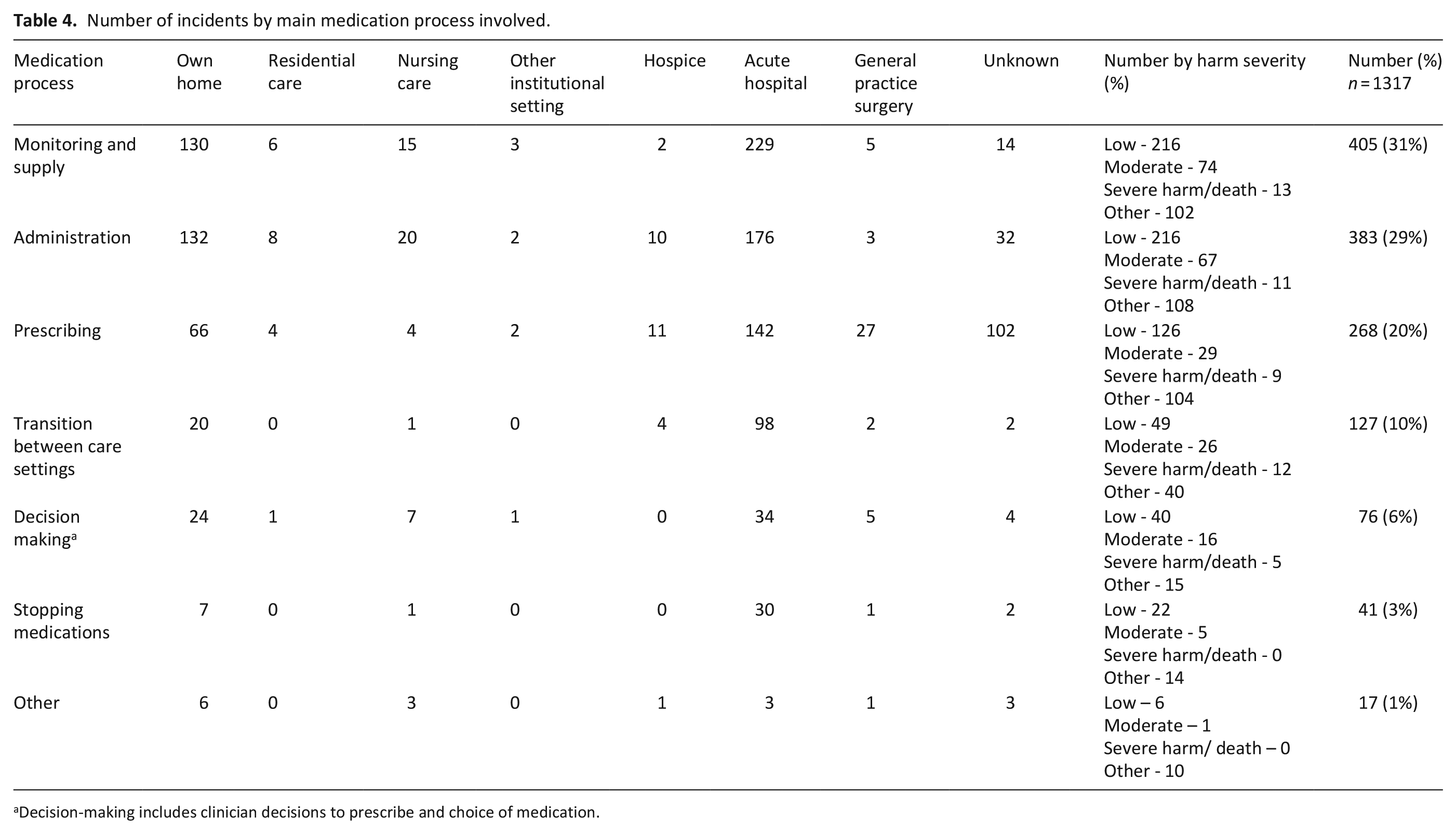
Decision-making includes clinician decisions to prescribe and choice of medication.
Commonly more than one medication process was involved as illustrated in this example:
. . .syringe driver had already been commenced with Morphine sulphate and cyclizine. . .diluent used was normal saline which was indicated on prescription sheet. . .cyclizine not compatible with normal saline. Patient’s leg became red and syringe driver needed to be re-sited
Medication processes were broken down into harm severity. Monitoring and supply incidents commonly resulted in low harm in 216 incidents (53%), administration resulted in low harm in 197 incidents (51%) and prescribing incidents resulted in low harm in 125 (47%) of incidents (Table 4). In all three of the major medication processes (monitoring and supply, administration and prescribing), the harm level was nil or it was difficult to ascertain the level of harm. This category of other was 102 (25%) of incidents in monitoring and supply, 108 (28%) in administration and 104 (39%) in prescribing process incidents.
Identifying a single point of failure was often challenging due to insufficient detail in reports, although interacting contributory factors and multiple weak points increasing risk were clear. Nevertheless, the wrong dose administered (248 incidents, 19%); medication timeliness (233 incidents, 18%); prescribing error (114 incidents, 9%); and drug omission (107 incidents, 8%) were identified most frequently as the primary incident.
Two fifths of reports (540 incidents, 41%) contained one or more contributory factor (i.e. at least one perceived reason why the incident occurred ) and a similar proportion (537 incidents, 41%) had contributory incidents (i.e. events leading up to the incident that resulted in the harmful outcome). Most commonly, an inadequate skill set or knowledge were cited by reporters, including limited prior experience of continuous subcutaneous infusions (84 incidents), and inadequate education or up-to-date training (69 incidents). Other contributory factors included lack of equipment (56 incidents) or continuity of care (56 incidents). Often when a drug omission (not administered to the patient in error) was reported as the primary incident, no contributory factors were mentioned (67/107 incidents, 63%).
Qualitative narrative analysis: Themes
Four major themes were identified in free-text narratives: continuity of care; communication and collaboration; supply and demand; and experience and competency. Intersections between themes were common. For example, incidents were often caused or exacerbated by poor communication; and a lack of awareness of receiving teams’ processes and procedures for using continuous subcutaneous infusions was notable in incidents involving patient transfers between teams and settings.
Continuity of care: Coordination and transitions
Incidents arising in transfers between settings featured in 127 (10%) reports, with inadequate exchange of patient information frequently leading to reported harm. About 84% (26/31) of discharge-related incidents reported harm, frequently coinciding with other incidents, such as delayed prescribing. Valid community medication authorisation and administration records, equipment and diluents (e.g. water for injection) were frequently missing:
. . .end-of-life anticipatory medications. . .would be coming home with patient. . .found to be in a great deal of pain. . .no anticipatory medications in place. . .did have analgesic patch in situ but this had not been handed over by hospital staff and was not effective
Several incidents reported no medication or incorrect medications being supplied by hospitals on discharge:
Patient discharged to care home with anticipatory medication and drug chart, but drug chart not signed by doctor. . . Hyoscine prescribed but the patient had been discharged with Glycopyrronium. . .delay in district nurses being able to set up the syringe driver
Hospital admissions and discharges also often lacked coordination with community-based services:
. . .neither DNs [district nurses] or SPCT [specialist palliative care team] had received direct verbal or written referral. . .found distressed patient and family. . .patient symptomatic. . .unable to swallow. . .no anticipatory medication sent home. . .
In one case an error in transfer-related communication included a 10-fold increase in medication:
. . .Syringe and prescription both stated 187.5mg of levomepromazine, this has been administered for previous 4 days. . .site in the patient left leg appeared very red. . .checked through patient notes. . .handwritten GP referral to AMU [acute medical unit] stated 187.5mg levomepromazine. . .this dose seemed too high. . .Community Nursing Team who had been doing the syringe driver in the community. . .confirmed that the dose they were administering had been 18.75mg. . .
Mismatches in perceptions of respective responsibilities resulted in patients not receiving planned and timely care. Cultural working practices varied, and consequently insufficient communication was reported:
. . .advised ward doctors to start syringe driver with midazolam and oxycodone spoke to the doctor left written instructions in the medical notes and asked nursing staff if there was a driver available. . .as they would be able to obtain one from our office until 5pm. . .colleague reviewed the patient the next day and found that the driver had not been started. . .This resulted in the patient being agitated and in pain. . .
Variations occurred both within (as shown above) and between healthcare teams/settings:
. . .Staff commenced syringe driver at 16:20 with Oxycodone 20mg at 0.67 mls/hour to run over 24 hours. . .he already had a syringe driver in situ from previous facility which staff had not been made aware of.
Communication and collaboration
Collaboration between healthcare professionals, family carers and other staff members was reported as preventing or mitigating adverse patient outcomes. Collective decisions to commence infusions were reported as important for preventing harm and distress. In some cases, a breakdown in the multiple steps of initiating a syringe driver subsequently caused further incidents. For example, the absence of a continuous subcutaneous infusion contributed to a family carer using medications with unintended consequences:
. . .Syringe driver medication not written on correct prescription sheet. . .daughter instructed verbally and written on discharge letter to give 1/4 a tablet 6.25mg every 2 hours prn [as needed] nausea and vomiting. . .proceeded to give a total of 25mg of levomepromazine in 4 x 6.25mg doses as prescribed. . .Patient heavily sedated by drug and barely rousable
Supply and demand
Inadequate staff numbers or skill mix, and lack of medication or equipment, led to delays in starting and replenishing and/or incorrect setting up of infusions and subsequently led to inadequate symptom control in hospital and in the community:
. . .imminently dying admitted from A&E. . .required continuous subcutaneous infusion to improve comfort. . .included midazolam, glycopyrronium. . .neither drugs kept as stock. . . delays in obtaining supply which delayed administration and control of symptoms. . .additionally CDU [clinical decision unit] does not stock basic equipment needed for syringe driver medication. . .many of the staff unfamiliar and not skilled/experienced in the use of subcutaneous infusions. . .
The decision was made to commence a syringe driver. Patient also taking oral dexamethasone so needing to convert this to a subcutaneous route. . .Those pharmacies that I called were also unable to order in as item saying out of stock.
Many reports identified that the reporter knew equipment was needed, but they were unsure of procedures to access these or experienced barriers to following established protocols. Equipment malfunctions (operator or technical failures) were recurring. Ongoing shortages of syringe drivers in hospital settings were frequently highlighted:
No syringe drivers available in the Trust. . . she required numerous separate subcutaneous injections, into cachexic tissue, which was very painful. . .died 24 hours after.
Several incidents could not be mitigated until equipment became available from another ward or until another patient had died.
There were cases when supply was sufficient, but equipment was not utilised correctly, set at incorrect rate or ran at a different rate than expected:
When changing patient’s syringe driver I noticed. . .end of the giving set still had the sheaf on it. . .no medication was being administered. . .patient had been complaining of a lot of pain.
. . .I checked the syringe driver at start of my shift and initially had no concerns. . .as the night went on, I noticed the infusion was running quicker than it should be and consequently realised that the patient had been overdosed. . .the syringe driver had ran for 17.5 hours instead of 24 hours.
Medication and equipment supply issues were more pronounced out of hours, when fewer staff were working with limited access to specialist advice in both hospital and community settings:
Contacted 111 [UK urgent care phone number] just before 9am on Saturday still no response or returned call by 3.30pm, patient had no meds for syringe driver at home so eventually FP10 [prescription document] completed from inpatient unit so that community nurses could refill the driver. . .On Sunday. . .patient in pain dropped oramorph bottle and it smashed. . .he had no extra doses at home unable to get hold of doctor.
Experience and competency: Systematic gaps in infrastructure
Poor decision making (76/1317 incidents, 6%) around dose ranges and titrating medication occurred, as did incidents because prescribers were unwilling or uncertain in prescribing infusions for patients with whom they were unfamiliar:
Locum GP asked to prescribe medication for syringe driver. . .As locum did not know patient this request was declined and advised the family to call out of hours GP if syringe driver was required. . .
Reporters commonly attributed these incidents to lack of experience/incompetency without considering contextual/infrastructure factors, suggesting there was a problematic tendency to view incidents as individual-level human errors rather than seek to identify and address structural deficiencies. For example, lone working, practical barriers to following protocols, reliance on agency staff and lack of timely access to guidelines and specialist advice may all have contributed to incidents but were rarely considered:
. . .On reviewing the prescription charts it appears the drop in conscious level is more likely to be related to the addition of midazolam. . .for which the antidote is flumazenil - this drug was not considered for use. . .200mcg dose of naloxone caused the patient to have a pain crisis. . .I asked [name] why flumazenil was not considered - he is unfamiliar with its use and therefore did not consider it.
Other examples attributed to ‘simple’ staff error included administration errors:
. . .new syringe driver prescribed containing 45mg morphine over 24 hours (ten times the dose he should have had). . .also 90mg cyclizine prescribed. . .which is incorrect dosing. . .
duplicate prescriptions:
patient has two prescriptions. . .patient had syringe driver changed containing Hyoscine Butylbromide 60mg. Correct prescription as per Palliative care advice should have contained Hyoscine Butylbromide 60mg, Midazolam 10mg, Morphine Sulphate 10mg to volume of 24mls over 24-hours. . .
and dose miscalculations:
instead of using 2 [x] 30mg ampules[sic], used 2 [x] 10mg ampules. . .patient received 18.3mg instead of 55mg via syringe driver over 24hr period.
as well as knowledge deficiencies such as failure to appropriately discontinue alternative opioids once an infusion commenced:
Twilight service commenced a syringe driver to deliver pain relief overnight and dosage to be reviewed next day. . .nurse informed patient’s wife to still give oral medication including the slow release Oxycodone. . .wife also gave drug next morning and patient became semiconscious
Similar sounding medication names, were commonly confused at the prescribing, dispensing and administration level:
. . .patient had been discharged from our hospital with Hyoscine hydrobromide although Hyoscine Butylbromide was prescribed.
Inadequate knowledge and understanding of prescribing appropriate medications and doses, and using infusion equipment, underpinned over half the common incident/contributory factor reporting patterns; the lack of identifiable protocols, equipment or prescribing guidance compounded these issues. Of the 44 incidents that reflected this theme, 30 (68%) involved nursing staff not being trained in using, or even if they had received training, not being confident to use syringe drivers.
Lack of peer support and blame were notable in many report narratives:
4am nurse contacted OOHs GP for advice on what dose to commence syringe driver on. . . . . .verbal advice by GP to start with Diamorphine 40mg and Midazolam 40mg. . .night staff commenced. . .stat doses over past 24 hours indicated a total of midazolam 12.5mg and Diamorphine 7.5mg. . .
Additional learning derived from researcher-led qualitative narrative analysis
Commonly, psychological and social harms to patients and their families were identified or implied in free-text reports but these were difficult to quantify due to insufficient description. In addition to family involvement in some of the communication examples above, one incident described the involvement of a daughter in a subcutaneous medication error:
According to [patient] the OOH GP asked her [daughter] to pass him Midazolam 2.5mg which he administered s/c. . .then the daughter realised it was actually Haloperidol. . .daughter felt she was to blame.
While numerically rare, the identification of incidents where family carers’ actions and opinions diverged significantly from the healthcare professionals involved is concerning. Three instances were reported where a continuous subcutaneous infusion was found ‘switched off’ by family carers, and professionals were unable to appropriately use a continuous subcutaneous infusion for another patient because of family disagreement:
. . .syringe driver commenced previous day for symptom management, but district nurses found it discontinued. . .wife had stated she had stopped it through the night as she felt drugs were killing him. . .she did not want her husband to receive any further stat doses to relieve symptoms. . .
Despite this patient’s desire to remain at home, he was instead transferred to hospital as the professionals felt it would not be possible to negotiate symptom control at home.
Patients’ and families’ perspectives in these reports were reframed in what the healthcare professional reporter deemed appropriate. Professionals also appeared to default to the use of additional authority to impose their views when faced with challenges from family carers for example, safeguarding referrals, seeking input from the primary responsible clinician.
Discussion
Harm was present in nearly three-quarters of continuous subcutaneous infusion incident reports, often resulting in uncontrolled symptoms and significant distress for patients and family carers. We found reports solely indicating physical harm when it was beyond reasonable doubt that broader psychological and social harm occurred. This suggests that these harms, though prevalent, are frequently overlooked in incident reporting, highlighting a need for systematic recognition of the impact of care deficiencies. More focus also needs to be given to assuring continuity and quality of palliative care at interfaces between different care providers, particularly during hospital admission and discharge processes. 38
Inadequate continuity when patients were transferred between settings and providers frequently resulted in safety incidents.38,39 Effective communication between multiple clinical teams as well as between professionals, patients and families is vital for safe use of palliative medications to achieve symptom control.6,9,16,18,39 Mechanisms embedded within cross-organisational working patterns are needed to provide time-sensitive training for professionals irregularly involved with continuous subcutaneous infusions, accompanied by the support of professionals who regularly use these regardless of patient location. Other issues are more complex, requiring the weighing of different risks. For example, the use of a single system-wide continuous subcutaneous infusion model reduces risks associated with confusion of pumps with different functions, whereas using different models in healthcare systems mitigates supply chain issues and product recalls.
While existing literature acknowledges individual-level issues, this research emphasises that multiple points of system failure are contributing to incidents with continuous subcutaneous infusions. It underscores the importance of ensuring timely access to medications when needed while addressing infrastructure and communication deficits to improve patient safety effectively.23,40 A focus in reports on lack of experience and competency at an individual practitioner-level also demonstrates the need to consider how working environments and infrastructure deficits place undue reliance on human mitigation in care provision.
Tools like the Systems Engineering Initiative for Patient Safety (SEIPS) could aid healthcare professionals in considering broader contributory factors and implementing systemic changes.41,42 SEIPS is a well-established healthcare improvement and patient safety framework that integrates evaluation of the structural, organisational and human factors that typically interact in care outcomes 43 .
Strengths and limitations
This study’s strengths lie in identifying what is overlooked in incident reporting, such as psychological and social harms, and highlighting the need for systemic solutions. We also identified that the existing taxonomy 27 for grading harm is structured to be systematically biased to focus on physical functional outcomes, and on the patient in isolation. This is at odds with the goals of palliative care, and with what is known about the lasting psychosocial impact of, for example, disturbed dying with respect to complex bereavement for families.9,43,44 Undertaking a narrative analysis of the incidents was important for maximising system learning, and would be further enhanced with better reporting of psychological and social harms.
Limitations include the self-selecting nature of incident reporting. Incident databases are inherently dependent on reporting culture and training for reporters; not all incidents are reported, nor all contributing factors and outcomes detailed. 24 Despite these limitations, our analysis highlights what is perceived important by reporters and, through considering gaps in free-text reporting, key factors that may be overlooked. Our study design initially sought to identify isolated incidents in the multi-step processes of continuous subcutaneous infusion use. In practice, we identified that reporters commonly fail to provide information that exactly pinpoints system breakdowns in process steps, and, when they do, incidents can be seen to involve several inter-related steps in medication processes.
Recommendations for policy, practice and future research
Examples of recommendations from our results synthesis are provided in Table 5.
Recommendations.
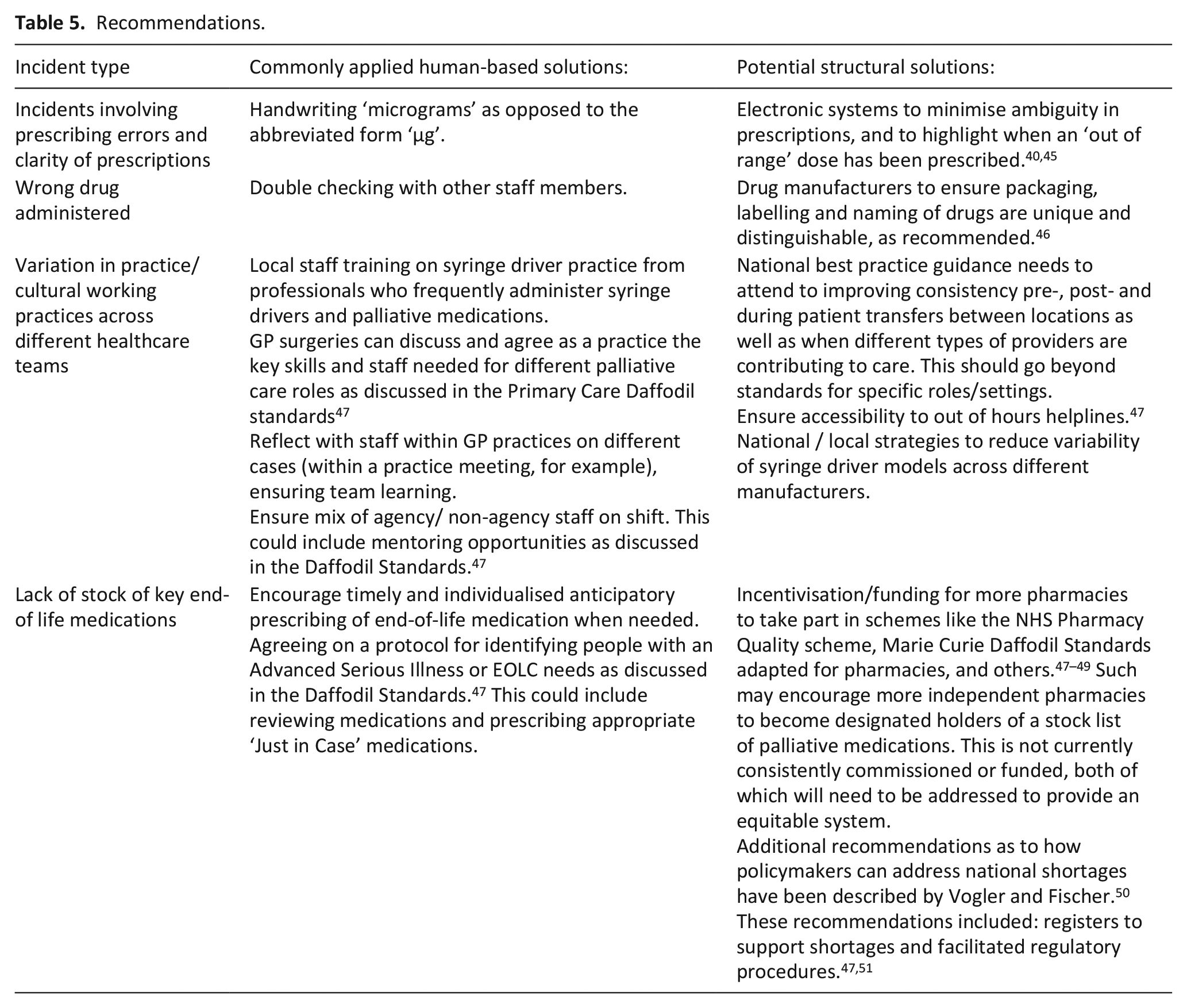
Palliative care services are structured differently in different countries but in England and Wales these services are free to access, well-established and reporting systems are relatively integrated in comparison to most international provision. 52 Consequently, shortfalls in care identified in our study are likely to also apply elsewhere. Organisations must embrace the complexity of palliative care and discuss locally their experiences and challenges of generating meaningful learning from incident reporting. 53 Many of the reported incidents were not specific to the subcutaneous route or a palliative care patient population but could equally occur with other routes and in other populations. This highlights both the importance of a whole systems analysis of incidents and the wider potential relevance of our findings.
In future incident analyses, research methods are needed to allow analysis of multiple interacting processes to understand the realities of practice and target solutions that account for these complexities. Our team are currently conducting this research. Further research is also warranted to explore how best to address medication-related psychological and social harms, including through incident reporting (e.g. new frameworks, taxonomies and alternative reporting systems) as these are not limited to the patient alone in the context of palliative care. This is recognised as being problematic worldwide. NHS England have more recently advocated reporting of psychological harms, and some countries make reporting severe psychological trauma mandatory, but neither of these actions alone will address the harms identified within this study.53 –56.
Research into changes in UK community palliative care before the COVID-19 pandemic identified changes in palliative prescribing pathways and clinical contacts; these were accelerated during the pandemic.20,38,39,56,58 Increasing use of remote patient consultations in community settings alongside the accepted (but under-researched) norms of practice with respect to prescribing of continuous subcutaneous infusions in advance of clinical need38,39,56,57,59 require monitoring for new safety risks alongside exacerbation of the risks identified in this study given that combining these practices may introduce additional systemic vulnerabilities into complex and nuanced care-critical clinical processes. 60
Conclusion
Continuous subcutaneous infusion incidents often involve multiple breakdowns across steps in key clinical processes, compromising patient safety. Narrative descriptions of psychological and social harm alongside physical harm risk are not being adequately recognised through existing approaches to measure harm in palliative care, hindering learning in practice. Structural changes are needed to minimise harm and maximise safety in palliative care.
Supplemental Material
sj-docx-1-pmj-10.1177_02692163241287639 – Supplemental material for Multiple points of system failure underpin continuous subcutaneous infusion safety incidents in palliative care: A mixed methods analysis
Supplemental material, sj-docx-1-pmj-10.1177_02692163241287639 for Multiple points of system failure underpin continuous subcutaneous infusion safety incidents in palliative care: A mixed methods analysis by Amy Brown, Sarah Yardley, Ben Bowers, Sally-Anne Francis, Lucy Bemand-Qureshi, Stuart Hellard, Antony Chuter and Andrew Carson-Stevens in Palliative Medicine
Footnotes
Acknowledgements
The views expressed are those of the authors and not necessarily those of the funders, sponsors, National Health Service, the NIHR or the Department of Health and Social Care.
Author contributions
All authors have made a substantial contribution to the research design and approved the final version of this paper. AB conducted the analysis supervised by ACS and SY. A sample of reports were double and triple coded by BB and SY. Clinical recommendations were composed by AB, SY, BB and ACS. AB drafted the article with all authors critically revising the intellectual content.
Data management and sharing
The data for this study is held by National Health Service Improvement who considers applications to use National Reporting and Learning System data on a case-by-case basis. Permission was granted to Cardiff University through a data sharing agreement for the duration of the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Central and North West London NHS Foundation Trust Starter Grant Scheme. ACS’s and SH’s contribution to the study was funded by PRIME Centre Wales. BB is supported by Wellcome Trust [225577/Z/22/Z]. Cardiff University is the sponsor of this study.
Research ethics and patient consent
Ethical approval was granted by Cardiff University School of Medicine Research Ethics Committee (Ref 19/28). A data sharing agreement exists between Cardiff University and National Health Service Improvement. Data released to the research team was anonymised based on in-house data cleaning processes led by the National Health Service Improvement team. Should identifiable content have been recognised in a report, the research team would adhere to strict Information Governance procedures which includes notifying National Health Service Improvement.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
