Abstract
Background:
The fear of taking away hope hinders clinicians’ willingness to share serious news with patients with advanced disease. Unrealistic illness expectations, on the other hand, can complicate decision making and end-of-life care outcomes. Exploration of the association between hope and illness expectations can support clinicians in better communication with their patients.
Aim:
The aim of this study was to explore whether realistic illness expectations are associated with reduced hope in people with advanced cancer.
Design:
This is a cross-sectional secondary analysis of baseline data from a primary palliative care cluster-randomized trial CONNECT (data collected from July 2016 to October 2020). Hope was measured by Herth Hope Index. Illness expectations were measured by assessing patients’ understanding of their treatment goals, life expectancy, and terminal illness acknowledgement. Multivariable regression was performed, adjusting for demographical and clinical confounders.
Setting/participants:
Adult patients with advanced solid cancers recruited across 17 oncology clinics.
Results:
Data from 672 patients were included in the study, with mean age of 69.3 years (±10.2), 53.6% were female. Proportion of patients indicating realistic expectations varied based on which question was asked from 10% to 46%. Median level of hope was 39 (IQR = 36–43). Multivariate non-inferiority regression did not find any significant differences in hope between patients with more and less realistic illness expectations.
Conclusions:
Our results suggest that hope can be sustained while holding both realistic and unrealistic illness expectations. Communication about serious news should focus on clarifying the expectations as well as supporting people’s hopes.
Keywords
Hope is a fundamental coping mechanism for people living with advanced disease.
Clinicians often hesitate to discuss poor prognosis with their patients as they fear of taking the hope away.
Only 10%–46% (based on the question used) of patients with advanced cancer in the study reported realistic illness expectations
There was no significant difference in hope between patients with realistic and unrealistic illness expectations.
Hope can be sustained by patients with both realistic and unrealistic illness expectations.
Communication about serious illness should focus on both improving illness expectations and supporting people’s hope.
Background
Hope is a complex phenomenon with a number of psychological theories explaining its role and structure. 1 Snyder 2 defines hope as “positive motivational state that is based on an interactively derived sense of successful (a) agency (goal-directed energy) and (b) pathways (planning to meet goals).” Hope is distinct to optimism by being an emotional state rather than a personality trait, containing more important but less likely outcomes, and by being more focused on beliefs about the self.3,4
For people living with advanced disease, hope is a core coping mechanisms.5,6 People who live with serious illness often hope for many things, such as cure, quality of life, or peaceful death, and sometimes their hopes are fluent and inconsistent with each other. 7 Previous research found hope to be positively associated with quality of life, social support, and spiritual and existential well-being as well as with reduced depression and anxiety.6,8,9 Patients, family caregivers as well as health care professionals all agree that supporting hope is crucial component of prognostic discussions. 10 However, balancing realism and hope while communicating prognosis is a challenging task for clinicians, who report the fear of taking away people’s hope as one of the reasons why they hesitate to engage in prognostic discussions with their patients. 11 During formulation of the classic SPIKES protocol for delivering bad news, the authors reported the results of a survey among 500 oncologists, showing that “being honest but not taking away hope” was the most difficult part of discussing bad news. 12 This led to the recommendation to explore people’s hopes as part of the conversation about serious news (step 2 of SPIKES).
People living with progressive, life-limiting disease face many difficult decisions regarding the goals of their care. Modern patient-centered care for people living with serious illness is based on a shared decision model: clinicians support patients to make informed decisions about the treatment options based on their values and goals.13–15 At the same time, physicians are expected to elicit patients’ values and preferences and incorporate them into decision-making process. 16 However, with one exception of a study at one small cancer center, 17 less than a half of patients with cancer report realistic illness expectations or accurate prognostic awareness18–20 that can be contributed to doctor, patient, as well as family factors. 21 Associations between prognostic awareness and variables such as depression, quality of life, or health care utilization are inconsistent. 22 Longitudinal studies describe a rather stable character of prognostic awareness over time,23–26 suggesting potential importance of other predispositional factors, such as personality traits, in the development of illness expectations. Formation of unrealistic illness expectations in people living with advanced cancer depends on their individual coping styles and defense mechanisms that they use, including various cognitive biases such as rationalization or denial.27,28
Evidence about the relationship between illness expectations and hope is scarce and requires further exploration as it could address the dilemma of choosing between honesty and empathy.8,20,29 The research question of this study was whether realistic illness expectations are associated with reduced hope in adult patients with advanced cancer.
Methods
Design
The STROBE statement 30 was followed in reporting the study (full checklist is available as Supplemental 1). We performed a cross-sectional secondary analysis of baseline data from a cluster-randomized trial of primary palliative care intervention CONNECT. The CONNECT intervention was led by oncology clinic nurses who received specialized training to provide primary palliative care with the aim to support quality of life and mood in patients and their caregivers. The effect on the intervention arm was compared to patients receiving the usual care. The trial was described in full detail previously. 31
Setting and sampling
The CONNECT trial was conducted in 17 oncology clinics across the University of Pittsburgh Medical Center Hillman Cancer network in Western Pennsylvania between July 2016 and October 2020. The unit of randomization was the oncology practice, using the practice size (number of patients with metastatic solid tumors seen in 2015) as the key for stratification.
Recruitment
Designated staff at each site reviewed upcoming appointments lists in order to identify potentially eligible patients, who were subsequently provided a one-page study overview and asked by their clinician if they would be willing to receive more information about the study. A trained researcher would contact those who wanted more information. Across the 17 sites, 1188 patients were invited to the study, 672 were enrolled (57% participation rate). There was no statistically significant difference between participants and those who did not enroll in the study regarding their age, race/ethnicity, or gender.
Ethics
All the enrolled patients provided written informed consent. Full details of the CONNECT The study was approved by the University of Pittsburgh Institutional Review Board (IRB: STUDY19090204) and is registered at ClinicalTrials.gov with identifier NCT02712229. No prior analysis focused on the association between illness expectations and hope.
Participants
Eligible participants were adult patients with metastatic solid tumors for whom the oncologists responded negatively to the surprise question (“Would you be surprised if this patient died in the next 12 months?”) 32 and had Eastern Cooperative Oncology Group Performance Status Scale scores of 0–2, indicating good performance status when patients might not be able to carry out work activities (score 2), but are fully ambulatory and capable of all selfcare, being up and about more than 50% of waking hours. 33
Measures
All enrolled patients completed a set of baseline questionnaires, including the Herth Hope Index (HHI), a validated tool to assess hope that is widely used in patients with advanced cancer. 34 The HHI consists of 12 questions, each answered on a 4-point Likert scale. Total scores range from 12 to 48, with higher score indicating higher level of hope. In consultation with the author of HHI, a 6-point difference has been identified as the minimally important difference that would have clinical relevance. 35
Illness expectations were assessed at baseline using three questions adopted from previously published studies of prognostic awareness in patients with cancer.29,36,37 Life expectancy was assessed by a question used previously in the VOICE trial 38 : “When you think about how having cancer might affect your life expectancy, do you think in terms of: months / years / do not know,” with “months” being considered the realistic response. Goals of treatment was assessed by asking “the goals of my therapy are to get rid of all my cancer (Yes/No), used previously by Temel et al. 39 Realistic expectations regarding the goals of treatment were assessed as answering “no.” Terminal illness acknowledgement was assessed using the question “How would you describe your current health status: relatively healthy but terminally ill/seriously and terminally ill/relatively healthy/seriously but not terminally ill,” developed originally by Prigerson et al. 40 and adapted in Coping with Cancer project, 36 with the first two options considered to be realistic.
Patients provided their data on age, gender, race, degree of religious importance, education level, cancer type, and marital status. Patients also completed the Hospital Anxiety and Depression Scale (HADS) 41 to evaluate their baseline depression and anxiety (seven items for anxiety and seven items for depression, score 0–3, with total score of 7 or less indicating no depression/anxiety, 8–10 indicating borderline score, and 11 and above indicating abnormality) and The Edmonton Symptom Assessment System (ESAS 42 ) to assess their symptom burden (10 items, score 0–100, higher score indicating higher symptom burden).
Statistical analysis
The sample size of 672 participants was calculated for adequate power regarding the primary aim of the CONNECT trial to detect an 11-point difference in FACIT-Pal quality of life measure, with effect size of 0.45 and 83% power, taking into account cluster randomization effects and estimating 40% attrition due to death or loss to follow-up. 31 For this secondary data analysis, all data available from the sample were used to be analyzed as described below.
In the analysis, we analyzed frequency and percentages for each of the illness expectations questions separately. Continuous variables are presented as mean ± standard deviation as well as median and inter-quartile range in descriptive measures; categorical variables are presented as frequencies and percentages. We calculated a composite “full awareness” score combining the realistic answers to all three questions in one variable, indicating the level of realistic expectations on scale from 0 (no realistic responses) to 3 (all questions answered realistically), which was also used in previous research. 26 To assess for difference in baseline characteristics by level of illness awareness, we conducted ANOVA for continuous variables and chi-squared tests for categorical variables. Comparisons of the Herth Hope Index scores between groups are performed using t-tests for two-group comparisons (not realistic vs realistic for each of the questions individually) and ANOVA for multiple-group comparisons (0, 1, 2, and 3 on the full-awareness score).
We computed multivariable linear regression models to test for differences in Herth Hope Index by levels of illness expectations while adjusting for covariates identified as relevant in previous research.8,35,43 (age, religious importance, education, marital status, income, length of time since cancer diagnosis, baseline ESAS score, baseline HADS-Depression score, baseline HADS-Anxiety score). In these models, we present statistical significance tests for differences between groups as well as for a noninferiority test with a 6-point margin of noninferiority. Only cases with complete data were used for calculations. All statistical analyzes were performed using SAS version 9.4 (SAS Institute, Cary, NC, USA).
Results
Baseline demographic and clinical characteristics of the sample are shown in Table 1. The mean age was 69.3 ± 10.2 years. Most participants were Caucasians (94%) and there were slightly more women in the sample (53.6%). Most frequent diagnoses were lung cancer (36.2%), gastrointestinal cancer (19.5 %), and breast/gynecological cancer (16.8%). Using the composite “full awareness” measure, patients with more realistic expectations were more likely to have higher depression (p = 0.042) and anxiety (p < 0.001) scores (more detailed results for the correlates of HADS scores are in the Supplemental 2). Patients with higher education were more likely to have higher awareness score (p < 0.001). Higher symptom burden was also associated with higher awareness score (p = 0.019).
Demographic and clinical characteristics of patient participants (N = 672), overall and by illness expectations.
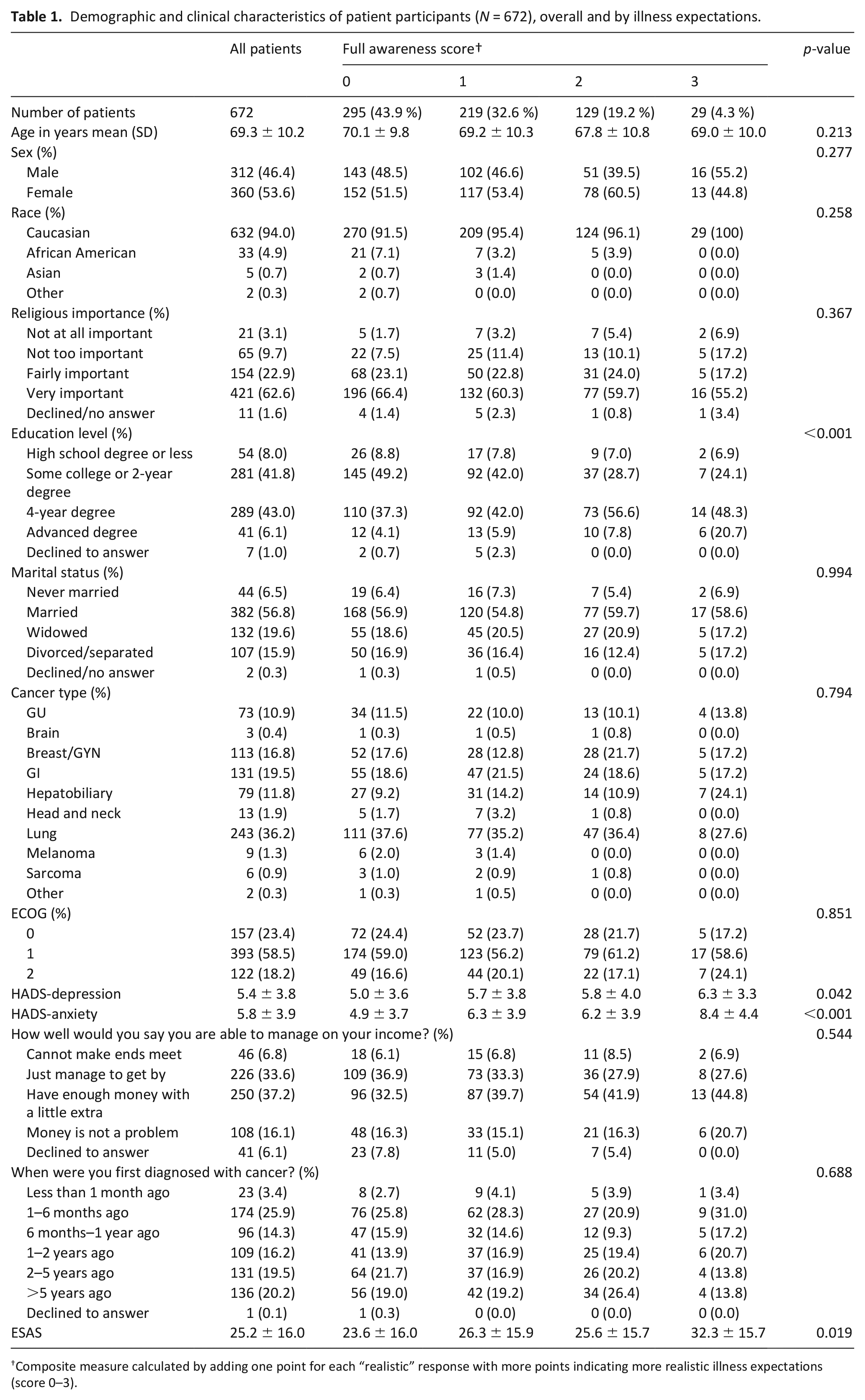
Composite measure calculated by adding one point for each “realistic” response with more points indicating more realistic illness expectations (score 0–3).
The proportion of patients indicating realistic expectations varied based on which question was asked, ranging from 10% to 46% (Figure 1, bold font corresponding to realistic expectations). All three questions were answered realistically only by 29 (4.3%) participants.
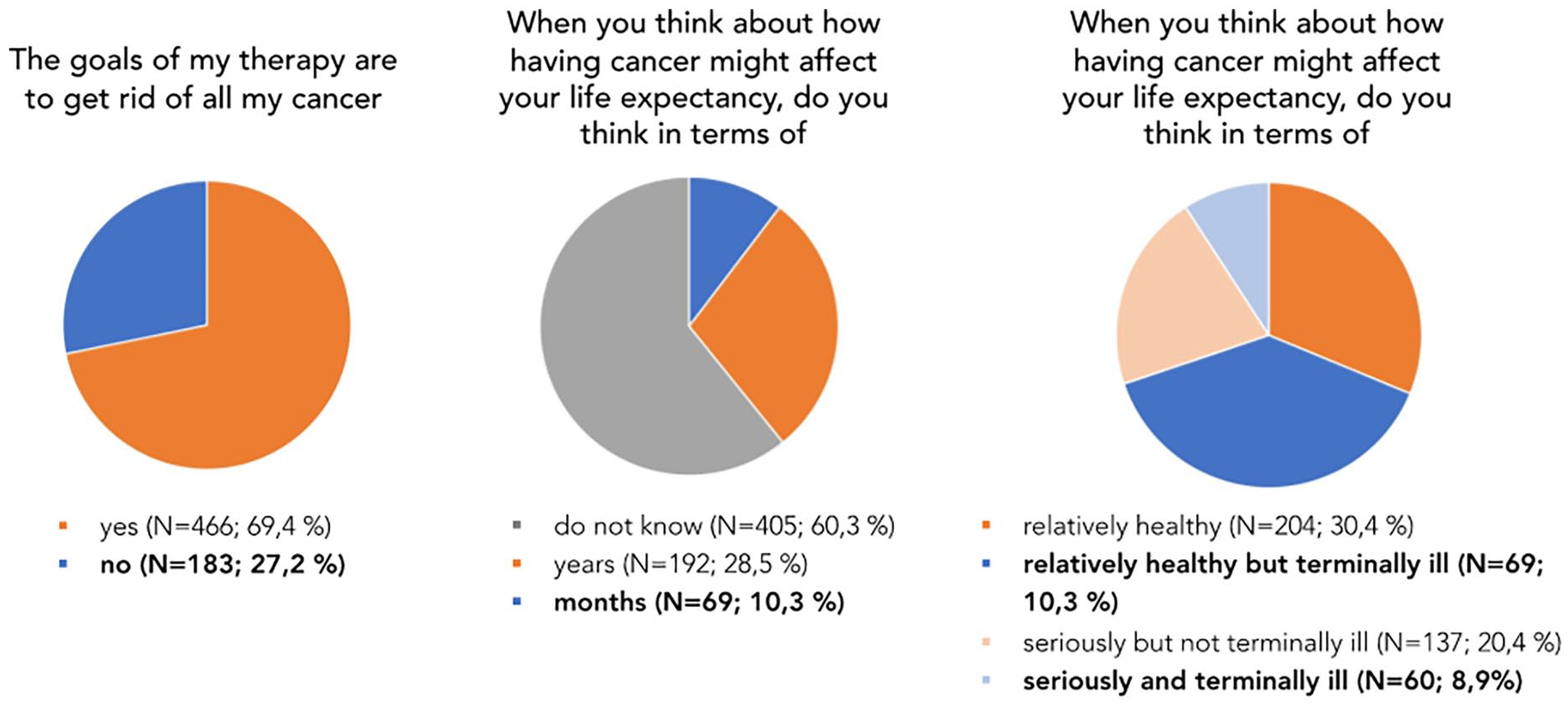
Realistic vs unrealistic illness expectations, asked by three different questions.
Overall median score for hope measured by HHI in the sample was 39 (IQR = 36–43), indicating a high level of hope. Patients with realistic illness expectations generally tended to have lower hope. However, the observed differences were very small (HHI median difference of 1, 1, and 2) and all within the 6-point margin of clinical relevance (Table 2). In a multivariate non-inferiority analysis adjusted for age, religious importance, education, marital status, income, time since cancer diagnosis, ESAS, HADS-D, HADS-A, and clinic site, the differences in hope were no longer statistically significant and all comfortably met a noninferiority conclusion at the 6-point margin (Table 3).
Distribution of Herth Hope Index (HHI) scores for patients with realistic versus not realistic illness expectations (N = 672).
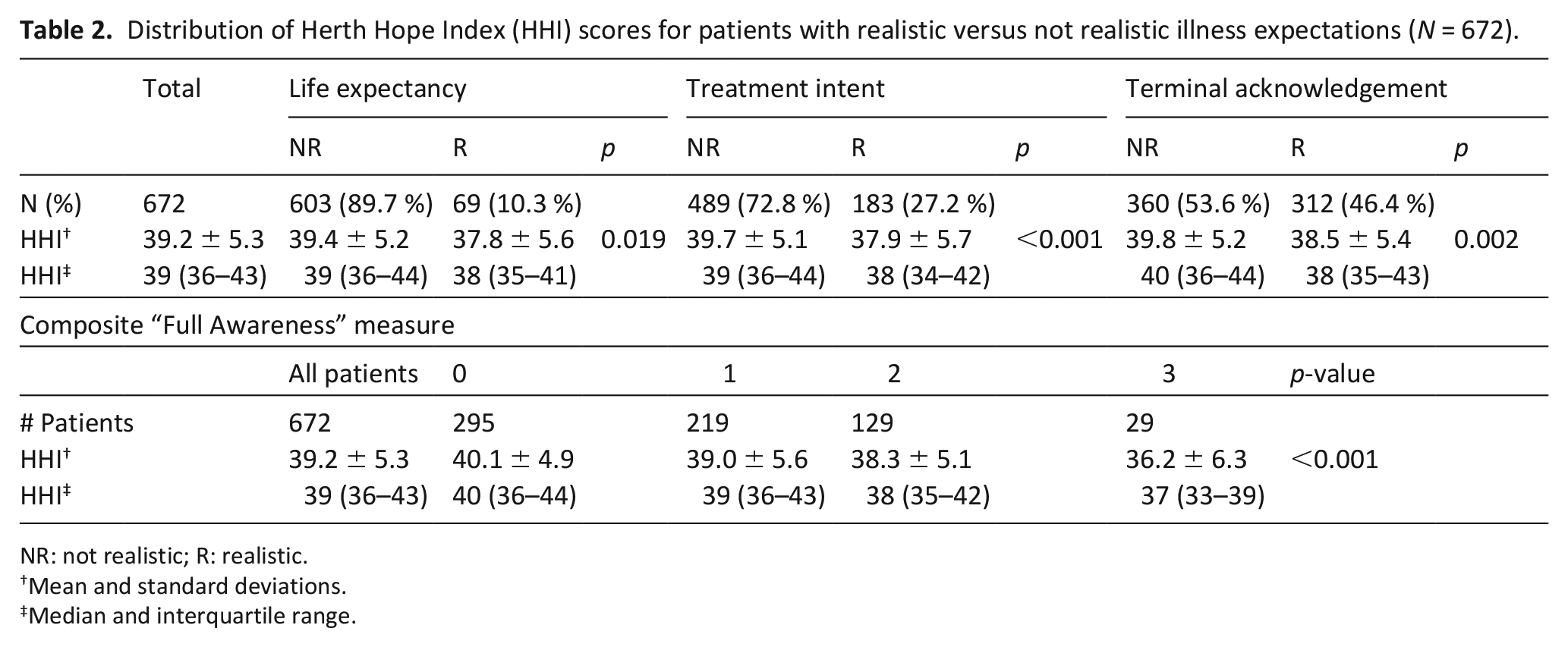
NR: not realistic; R: realistic.
Mean and standard deviations.
Median and interquartile range.
Adjusted† difference in Herth Hope Index (HHI) score by level of illness expectations.
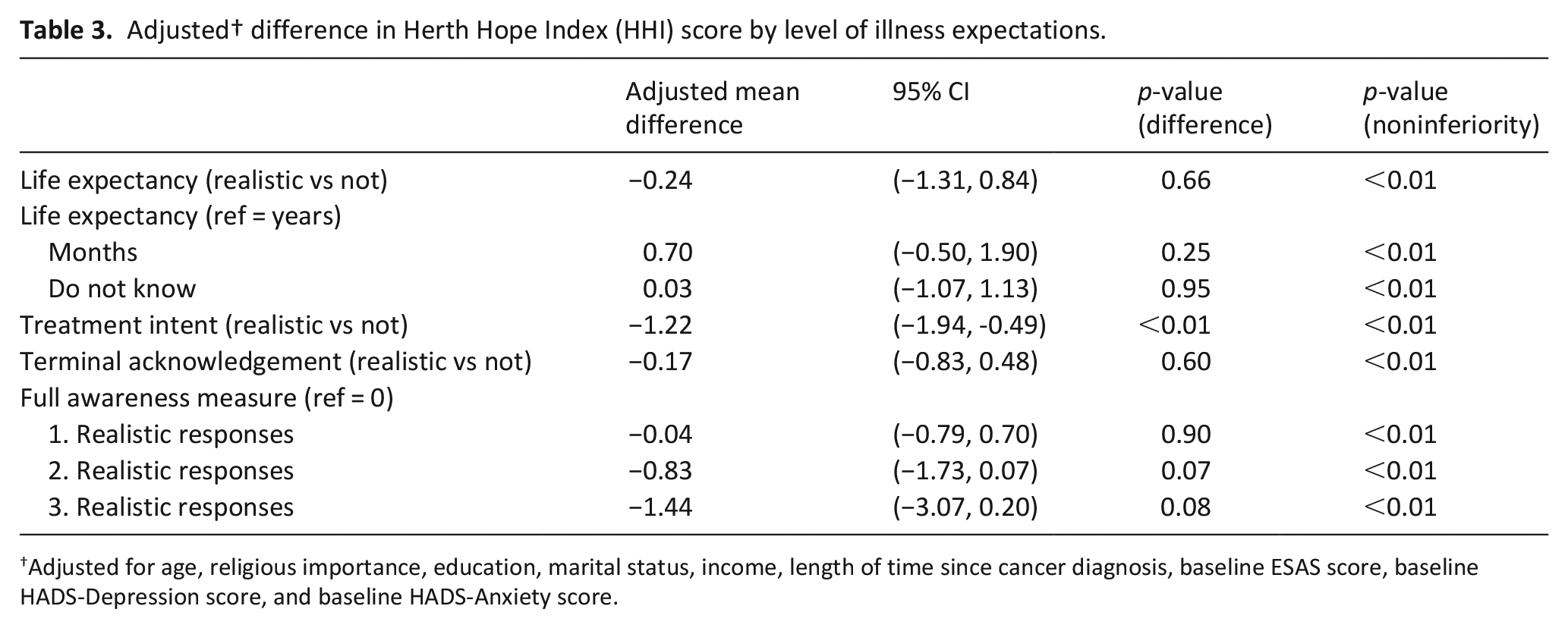
Adjusted for age, religious importance, education, marital status, income, length of time since cancer diagnosis, baseline ESAS score, baseline HADS-Depression score, and baseline HADS-Anxiety score.
Discussion
Main findings of the study
Our study explored the association between illness expectations and hope among people living with advanced cancer, with results indicating that people can have similar levels of hope while holding both realistic and unrealistic illness expectations. Participants with higher education and higher symptom burden reported more accurate illness expectations. We also found that patients’ responses about their illness expectations varied significantly based on the question used, however no clinically important differences were found for the level of hope regardless of which question was asked.
What this study adds?
Our findings of only weak associations between realistic expectations and hope support previous qualitative studies, which found that prognostic disclosure might not lead to the loss of hope. Kogan et al. 44 reported on people’s ability to redefine hope based on the disclosure of terminal prognosis when patients highlighted the value of setting up goals that are achievable under their circumstances. Another qualitative study based on 76 interviews with palliative care patients showed that the intensity of hope is more linked to the importance of the object it is attached to, rather than to the likelihood that it would become reality. 45 Smith et al gave printed very realistic (weeks to a few months) prognostic information to 27 patients, which they found helpful, but most remained overly optimistic. 46 Even with the dismal prognosis given explicitly, hope was maintained and even increased in some patients. 47
Our findings differ from those of a recent study from Singapore, where Finkelstein et al. 48 described the importance of several cognitive biases, such as optimism bias or illusion of superiority, on developing inaccurate survival expectations. Finkelstein et al. also suggested that these biases can be amplified by higher level of hope as each one-point increase in hope measured by the HHI was associated with 6% greater odds of patient believing their illness is curable and a 4-month larger expected survival. There are three potential explanations for the difference in our results. First, although all patients in Finkelstein et al. study had life expectancy of less than 12 months (indicated by their treating oncologists), 41% stated that the goal of their treatment was to completely cure the disease. In our study, we found similar level of hope (39 vs 39.7) but even more patients in our sample had unrealistic expectations about their treatment intent (69% vs 41%). The second possible explanation is that we adjusted our analysis for more covariates, including ESAS score, depression, and anxiety. The last important difference in our methodological approach was that we factored in the 6-point clinically important difference for HHI score to test the clinical relevance of the results.
Balancing hope and honesty are a challenging task for clinicians who might be afraid of taking hopes away by sharing serious news about patients’ prognostic status. Our study suggests that the association between patients’ hope and their illness expectation is more complex, and that hope can be sustained while holding both realistic and non-realistic expectations about the future illness trajectory. Workshops for people living with metastatic breast cancer that concentrate on enhancing hope appear to be successful in raising hope scores and allowing participants to integrate more hope into their daily lives. 49 Rosenberg et al. also suggest to think about patients’ hope as a fluid and complex phenomenon, often consisting of diverse hopes. 7 Data from bereaved parents showed that hope can shift its focus from cure to life outside of illness (have as normal life as possible) to comfort and prevention of suffering, 50 which might be especially challenging for clinicians to address. 51 Communication about serious news should focus on clarifying the expectations as well as supporting people’s hopes.
Study limitations
This study has several limitations. We used several different questions to assess patients’ illness expectations, and our analysis showed how results differ based on the chosen assessment method (realistic expectations rates varied from 10% to 46%). This suggests that illness expectation is a complex phenomenon and different operationalizations of this concept hinders the comparability of the published evidence. 37 Complex and robust methods of assessment, such as recently published Prognostic Awareness Impact Scale, 52 are needed for future research in this field. Our sample also consisted predominantly of White respondents (94%), representing the population of suburban and rural Western Pennsylvania, which might limit the generalizability of the results to populations with more racial/ethnic diversity. Another limitation of the study is that the patients reported lower scores of depressions than patients with advanced cancer in other studies (mean HADS score for depression in our sample was 5.4 ± 3.8, which is below even the borderline level), which might limit the generalizability of the results to more depressed populations. Also, 86% of our sample reported religion being fairly or very important for them, potentially limiting the representativeness for other regions or countries with less religious communities. Future studies could test the importance of other confounding variables such as type of health services, personality, family status, or social support, including the role of primary informal caregivers, as these factors might impact people’s hope. To further expand our knowledge in this area, future research should also explore whether the objective of hope among patients with advance disease (e.g. life extension, peaceful death, or symptom control) is associated with their illness expectations. Another important area to explore is what are the effective ways to clarify people’s expectations and how unrealistic expectations may influence the patient-clinician relationship.
Supplemental Material
sj-pdf-1-pmj-10.1177_02692163231214422 – Supplemental material for Hope and illness expectations: A cross-sectional study in patients with advanced cancer
Supplemental material, sj-pdf-1-pmj-10.1177_02692163231214422 for Hope and illness expectations: A cross-sectional study in patients with advanced cancer by Martin Loučka, Andrew D Althouse, Robert M Arnold, Thomas J Smith, Kenneth J Smith, Douglas B White, Margaret Q Rosenzweig and Yael Schenker in Palliative Medicine
Supplemental Material
sj-pdf-2-pmj-10.1177_02692163231214422 – Supplemental material for Hope and illness expectations: A cross-sectional study in patients with advanced cancer
Supplemental material, sj-pdf-2-pmj-10.1177_02692163231214422 for Hope and illness expectations: A cross-sectional study in patients with advanced cancer by Martin Loučka, Andrew D Althouse, Robert M Arnold, Thomas J Smith, Kenneth J Smith, Douglas B White, Margaret Q Rosenzweig and Yael Schenker in Palliative Medicine
Footnotes
Author contributions
ML, YS, ADA, and RMA contributed to the conceptualization and writing of the manuscript. ADA provided the statistical computations. YS provided the access to the original data. TJS, KS, DBW, and MQR contributed to conducting the study and editing the manuscript.
Data availability statement
The data that support the findings of this study are available from Dr Yael Schenker (
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the NIH grants number R01CA197103 and K24AG070285. ML’s work on this paper was supported by Czech Science Foundation grant number 23-07747S. The CONNECT trial also used resources provided through the Cancer Center Support Grant P30CA047904 from the National Cancer Institute.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
