Abstract
Background:
The Integrated Palliative Care Outcome Scale (IPOS) validly and reliably measures symptoms and concerns of those receiving palliative care.
Aim:
To determine the equivalence of the paper version with an electronic version of the IPOS (eIPOS).
Design:
Multicentre randomised crossover trial (NCT03879668) with a within-subject comparison of the two modes (washout period 30 min).
Setting/Participants:
Convenience sample of specialist inpatient and palliative home care patients aged over 18 years with cancer and non-cancer conditions was recruited. Scores were compared using intraclass correlation coefficients (ICC), Bland-Altman plots and via a mixed-effects analysis of variance.
Results:
Fifty patients were randomised to complete paper-electronic (n = 24) and electronic-paper (n = 26) IPOS with median age 69 years (range 24–95), 56% male, 16% non-cancer. The ICCs showed very high concordance for the total score (ICC 0.99, 95% CI 0.98–1.00), lowest ICCs being observed for symptoms ‘Appetite loss’ and ‘Drowsiness’ (ICC 0.95, 95% CI 0.92–0.97). Nine of seventeen items had ICCs above 0.98, as did all subscales. No statistically significant mode, order, age, and interaction effects were observed for IPOS total score and subscales, except for ‘Communication’ (Fmode = 5.9, p = 0.019). Fifty-eight percent preferred the electronic version. In the group 75+ years, 53% preferred the paper version. Only three entries in the free-text main problems differed between the versions.
Conclusion:
The very high equivalence in scores and free text between the IPOS and the eIPOS demonstrates that eIPOS is feasible and reliable in an older palliative population.
Keywords
The implementation of electronic self-reported versions may offer several advantages to palliative and hospice care, especially when fully integrated within an electronic patient health record or to help patients with sensory impairment.
The typically older palliative population might pose a barrier to the electronic implementation of patient-centred outcome measures.
Psychometric properties cannot be assumed stable across administration modes, necessitating a careful electronic adaptation of paper versions.
This randomised crossover trial in palliative patients showed the equivalence of scores between the self-completed paper and electronic version of the Integrated Palliative care Outcome Scale with near perfect agreement reached for 17 out of 21 items.
No version took longer to complete. Overall, 58% preferred the electronic version. Only in the group of 75 years or older, slightly more than half preferred the paper version for self-completion.
The high agreement and good acceptability of the electronic version of the Integrated Palliative care Outcome Scale was achieved via careful early involvement of patients and staff within a co-design framework.
Implementation of electronic assessment of patient-centred outcomes in palliative care is feasible once setting-specific barriers and facilitators are acknowledged and addressed in close collaboration with all stakeholders.
Introduction
Outcome measures are instruments that allow the assessment of change in a patient’s health status over time. Due to their patient-centredness, they play an increasingly important role in palliative care. Outcome measures are central for identifying patients’ needs, aiming at enhancing quality of life and the relief of suffering, and support evaluating the standard of care received.1–3 In palliative care, both patient and staff-completed versions of outcome measures exist to enable outcome measurement at the end of life.4–6
One such outcome measure for palliative care is the Integrated Palliative care Outcome Scale (IPOS) that has been studied extensively over the past years and has seen many developments and adaptations to different settings, conditions and populations.6,7 Next to the Edmonton Symptom Assessment Scale,8,9 it is one of the most widely used measures in the field.4,5,10,11 The IPOS assesses how much a patient is affected by symptoms, emotional concerns as well as communication and practical issues. The validity of the measure (in terms of structural validity, content, construct and criterion validity), its reliability and sensitivity to change have been demonstrated in several international studies for the inpatient, hospice, and specialist home care setting.6,7,12,13
The implementation of electronic self-reported outcomes may offer several advantages to the palliative care and the specialist home care setting in particular, such as low administration cost, scalability, adaptability on devices for those with sensory impairments, immediate automated analysis, and the possibility for full integration into an electronic patient record system. 14 The implementation in routine care could lead to shorter reaction times of staff to emerging symptoms/problems or crises, and ultimately to improved patient outcomes. 3 A few such systems for electronic capture with full integration of automated symptom monitoring have been established in oncology in recent years. 15 Only a handful of systems have been implemented in palliative care.3,16–20
Despite these advantages and their usefulness, electronic outcome measures are not commonly implemented in the palliative care setting, with only 25% of studies in palliative care and advanced oncology reporting using electronic versions. 5 Fearing the loss of personal contact while judging face-to-face interactions as more suitable in routine palliative care are named as the most important barriers. 21 A further barrier to electronic assessment in the home care setting may be that patients are typically older, and thus belong to a population with associated lower levels of computer and internet usage.22–24 Moreover, psychometric properties of questionnaires cannot be assumed stable across administration modes, 25 and it is thus recommended to empirically evaluate score equivalence and accordance of modes.
We therefore conducted a randomised crossover trial to test the score equivalence of paper and electronic versions of the IPOS for individual items, subscale scores and the total score. We hypothesised equivalence between the two administration modes.
Methods
Study design
This study was a multicentre, randomised, single-blinded, two-arm crossover trial of 5 months’ duration and is part of the project Palli-MONITOR, a multicentre, sequential mixed-methods, two-phase development and feasibility study (NCT03879668; https://clinicaltrials.gov/show/NCT03879668). 26 The study is reported in accordance with the CONSORT crossover guideline. 27 No changes to the original study protocol were made. 26
The crossover design is the preferred design for establishing measurement equivalence between different modes of administration as per guidelines.25,28,29 Patients were randomly assigned to two groups defined by the order of mode of administration (paper version of the IPOS first, group ‘P-E’ vs. electronic completion of the eIPOS first, group ‘E-P’).
Trial participants and settings
Participants either received specialised (five services) or general palliative home care (two services) or specialist palliative inpatient care at the university hospital’s inpatient unit. Eight study sites in Bavaria, Germany, participated from May to September 2019 representing both an urban and rural palliative population. Eligibility criteria were aged 18 years with advanced and incurable malignant or non-malignant disease, the capacity to give written informed consent and being sufficiently fluent in German to complete both questionnaire versions. Exclusion criteria were cognitive impairment or being in a poor general condition or actively dying, as judged by the patient’s clinical team. All patients who agreed to participate gave written informed consent. The study was approved by the Medical Institutional Ethics Committee of the University Hospital Munich (REC ref no: 18-871). No further ethical issues emerged during the study.
Interventions and procedures
Patients were screened consecutively for inclusion in the study. Eligible patients, who agreed to share contact details, were contacted by the study team, and informed about the study. If they were interested to participate, a member of the study team contacted them in person to give more information and take written informed consent. Patients were assigned a seven-digit identification number. Allocation to administration order was done by the principal investigator, based on a computer-generated 1:1 schedule (enuvo). 30 The patient then completed both paper IPOS and eIPOS in randomised order, with a 30-min washout period between administrations. The washout period was optimised to the palliative care setting, striking a balance between fluctuating symptom burden, and mitigating recall bias and carry-over effects. Both modes were completed in one visit lasting 45–60 min.
Neither the clinical staff nor the participants were blinded to the result of the randomisation. The statistician conducting the analysis was blinded to group allocation. Assignment to trial arm was concealed on a paper until written informed consent was obtained from participants.
The IPOS is a short 17-item outcome instrument to assess palliative-care related symptoms and concerns in generalist and specialist PC settings.6,7 The items cover physical symptoms (e.g. pain, shortness of breath, fatigue etc.), emotional concerns (patient and family anxiety, depression, feeling at peace), and communication & practical problems (sharing feelings, information needs, practical problems). Patients can designate their three main symptoms and problems as well as name and rate additional symptoms not included in the symptom list. All closed items are scored on a 5-point Likert scale ranging from 0 ‘not at all’ to 5 ‘overwhelmingly’. A sum score of all items and three subscale scores can be calculated.6,12 The paper IPOS can be obtained from www.pos-pal.org.
We used the paper-based, setting-specific 3-day or 7-day recall version of IPOS validated for the German context. 6 Its electronic adaptation was developed to resemble the paper version as closely as possible based on results from an interview study with patients testing a pilot version. 26 For free-text items, patients could enter symptoms and problems in short-answer boxes. Rated items on the IPOS could be answered by selecting the appropriate box on the five-point Likert scale. The eIPOS was provided on all operating systems (Apple, Windows, Android) and devices (e.g. laptop, computer, tablet, smartphone). Navigation buttons at the end of each screen allowed navigation through the questionnaire. Participants could progress to the next item without answers being mandated in eIPOS.
Data collection
For concordance, all answers to open-text and closed items on the IPOS were recorded for both the paper and eIPOS version. The time to complete was taken after each administration. Preference was asked using a closed question. Socio-demographic data included age, gender, nationality, main diagnosis, main care provider, device and operating system used and general use of electronic devices (daily, several times per week, once per week, less than once a week).
Statistical analysis
Sample size
With a power of 80%, a target intraclass correlation coefficient (ICC) of 0.9 and a significance level of α = 0.05, the calculated sample size is 47. Considering possible dropouts, it was planned to include 50 participants.
Data analysis
Data management and analyses were conducted with SPSS 27 31 and R 4.0. 32 Data are described via means and standard deviations (SD) for continuous and absolute frequencies and percentages for categorical variables. All data is presented for the whole sample and separately per trial arm. The distribution of scores for quantitative items is compared graphically and via the percentage of floor or ceiling effects (>15% of participants scoring the lowest or highest response option) 33 between the modes. A mixed-effects 2 × 2 analysis of variance model was used to assess mode effect (within-subject factor), order effect (between-subject factor) and the mode × order interaction effect. A significant order and interaction would indicate carry-over effects. Additionally, age was fitted as a covariate to evaluate any statistically significant mode × age effect. Four univariate mixed-effects models were run with IPOS Total Sum score, IPOS subscale scores and time to complete as dependent variables.
Following guidelines and other research,25,34–36 the concordance of the IPOS and eIPOS was evaluated using intraclass correlation coefficients (two-way mixed effects model for absolute agreement) with 95% confidence intervals, ranging from 0 to 1, for all individual IPOS items and the subscales and total score. An ICC of >0.90 was considered indicating excellent agreement.37,38 Prevalence- and bias-adjusted kappa coefficients were additionally used to take possible bias between modes and distributional floor and ceiling effects into account. 39 To assess the magnitude of possible systematic error, we also present the mean difference of scale scores between modes. Data were evaluated graphically by Bland-Altman plots. 40 The score difference (paper minus electronic) was plotted against the average paper and electronic score for each individual, including 95% limits of agreement calculated by 1.96 × SDdifference. Any systematic bias is thus separated from random measurement error.
A statistical significance level of 5% was used for all analyses. Missing items were imputed with the scale’s median. A sensitivity analysis with the imputed data did not produce different results due to the very low rate of missing data.
Results
Participants
A total of 66 eligible patients were invited to participate. Of these, 50 accepted the invitation and were randomised to either ‘paper-electronic (P-E)’ or ‘electronic-paper (E-P)’ order. Of those participating in the trial, almost all patients completed all items in both versions. Only one score on the item Poor mobility was missing for the paper version, and one score for the item Sharing feelings was missing for eIPOS. The trial flow is shown in Figure 1.
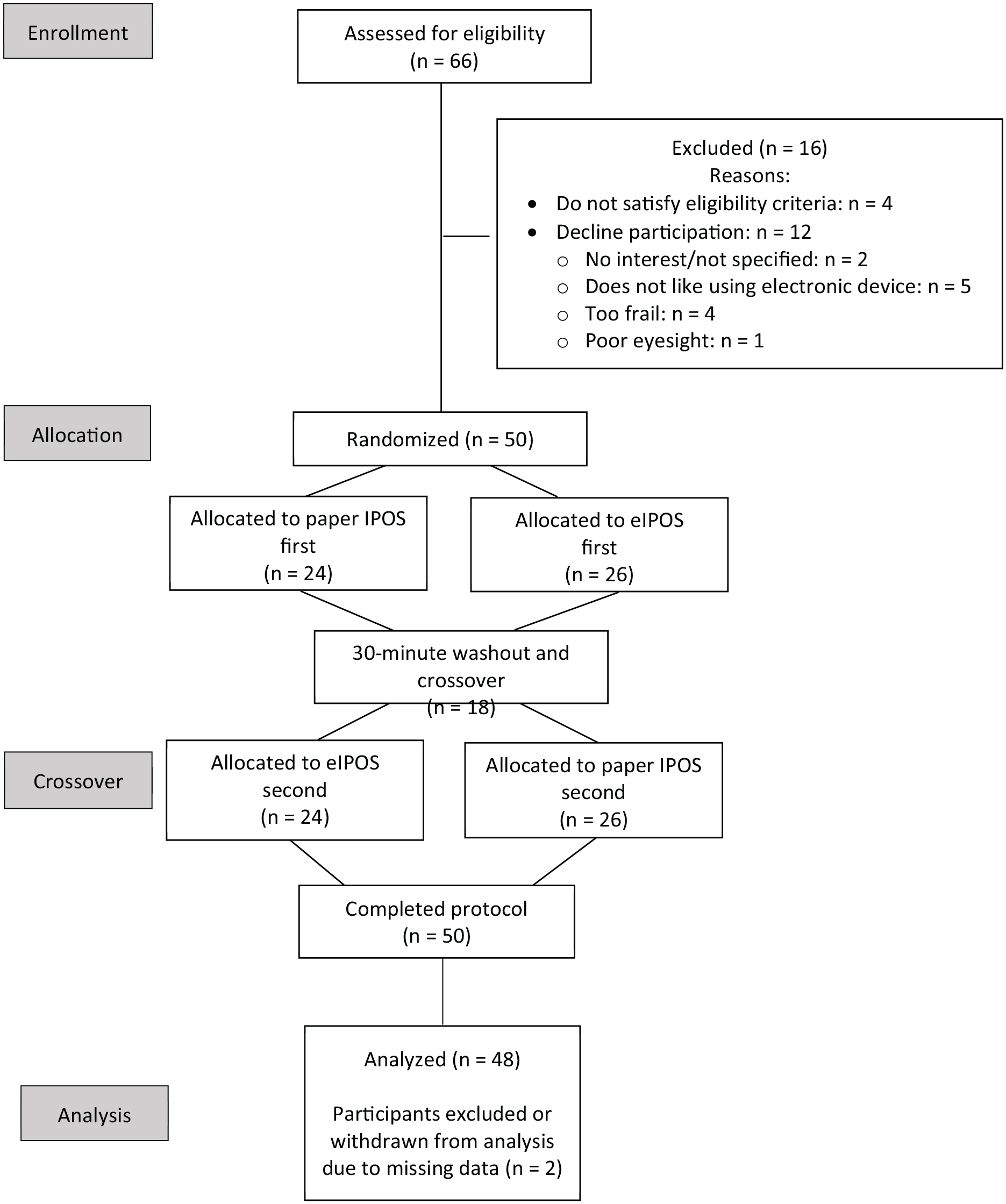
Flow diagram for crossover trial of paper versus electronic version of IPOS in palliative care.
There were no significant differences between those allocated to the two orders. The mean age of participants was 67.9 years (SD: 13.6), 56% were male. Demographic details are given in Table 1. Participants accessed the eIPOS most commonly on a tablet (68%), followed by laptop (16%) and PC or mobile phone (8%, respectively). Slightly over half of the sample used their device daily, 6% only used it less than once per week.
Demographic and clinical characteristics of study participants (n = 50).
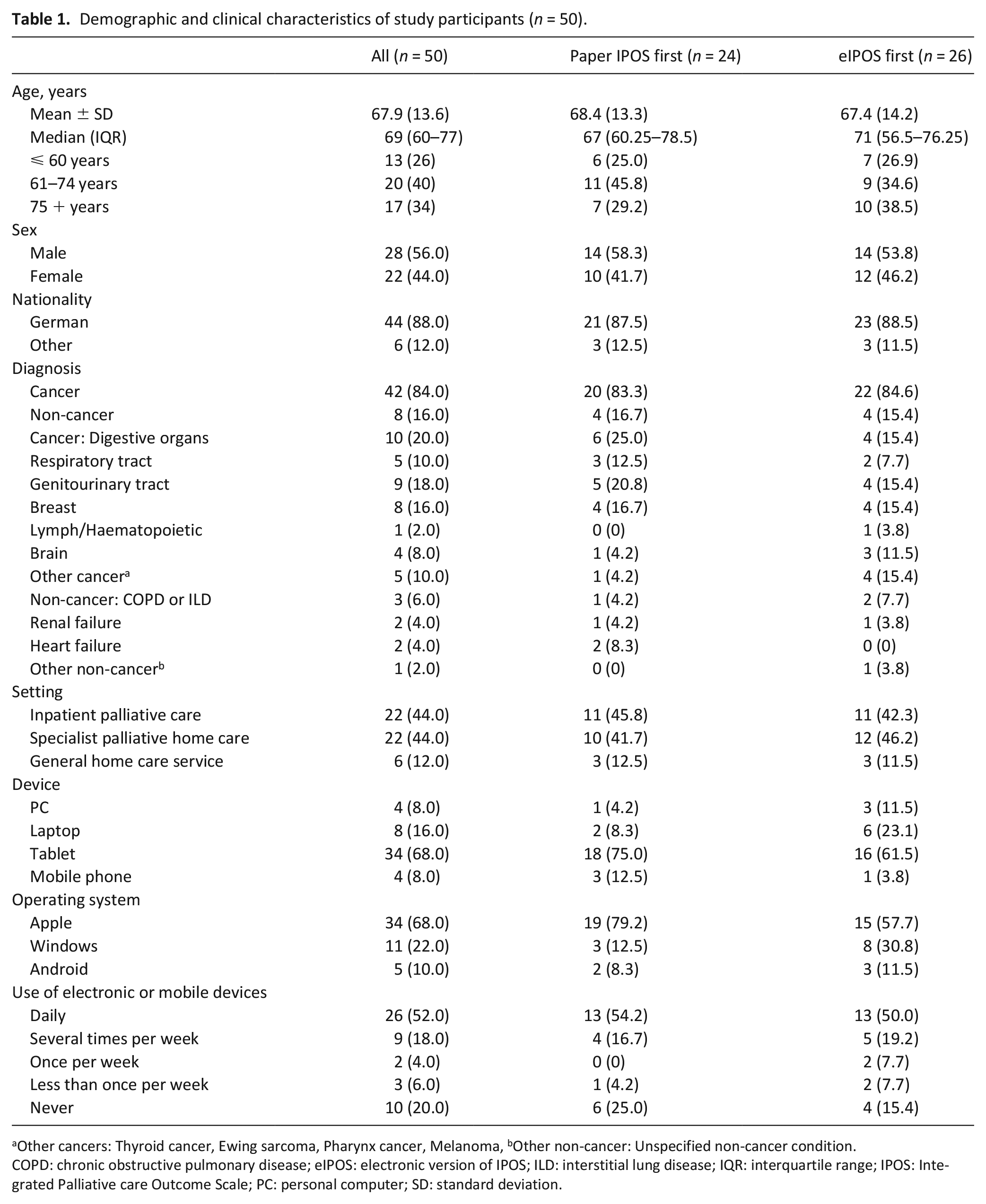
Other cancers: Thyroid cancer, Ewing sarcoma, Pharynx cancer, Melanoma, bOther non-cancer: Unspecified non-cancer condition.
COPD: chronic obstructive pulmonary disease; eIPOS: electronic version of IPOS; ILD: interstitial lung disease; IQR: interquartile range; IPOS: Integrated Palliative care Outcome Scale; PC: personal computer; SD: standard deviation.
Descriptive statistics
Descriptive statistics for the distribution of scores on the IPOS and the eIPOS are provided in Table 2. The mean and median scores between the two modes only differed in the first decimal. Small consistent differences existed for the proportion of floor and ceiling effects with the eIPOS showing a slightly smaller proportion of floor effects in four symptom items, two of the four emotional subscale items, and all Communication & practical problems subscale items. The score distribution is presented graphically in Supplemental Figures 1 and 2 for the IPOS and the eIPOS. Fifty-eight percent of patients preferred the electronic version, 40% the paper version.
Sample statistics for the paper and electronic IPOS versions (n = 50).
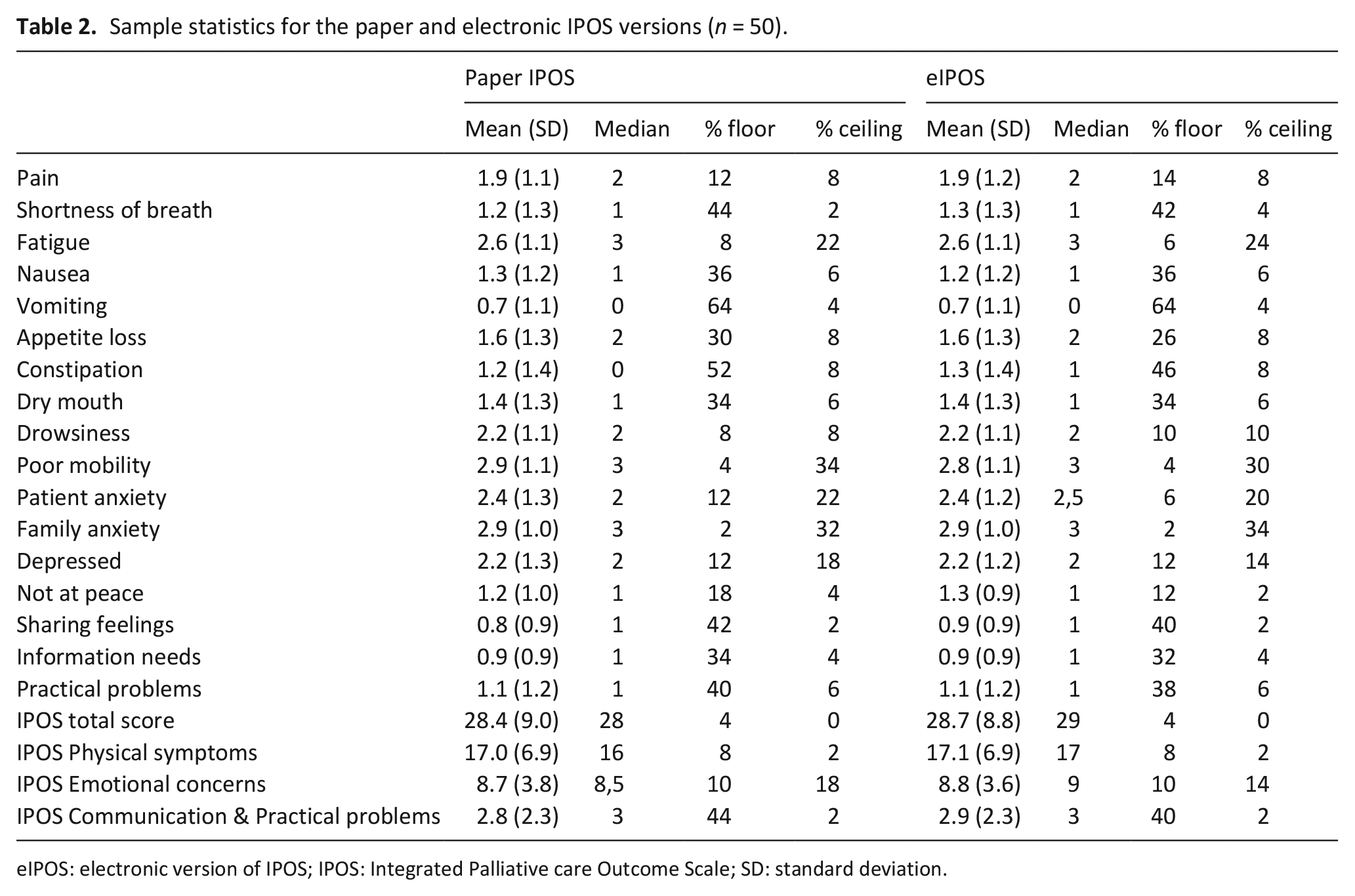
eIPOS: electronic version of IPOS; IPOS: Integrated Palliative care Outcome Scale; SD: standard deviation.
Mode equivalence
The mixed-effects analysis of variance with mode and order as the two main effects showed mean differences of −0.1 for the IPOS subscale scores and −0.3 for the IPOS total score when comparing the IPOS to the eIPOS. Table 3 shows that all mode and order effects as well as interaction effects were statistically non-significant, except for a statistically significant mode effect for the IPOS Communication & Practical problems subscale (F (2,48) = 5.9, p < 0.019). None of the mode × age interaction effects were statistically significant. Table 4 presents results for the outcome mean time to complete for both modes. There was no statistically significant order, mode, or interaction effect for time to complete. However, the paper and electronic versions differed significantly between age groups with patients aged 60 years or younger requiring the shortest time to complete both modes.
Results of mixed-effects 2 × 2 analysis of variance (Mode: Paper or electronic, Order: P-E vs. E-P, Interaction, Covariate: age) in n = 50 palliative patients.
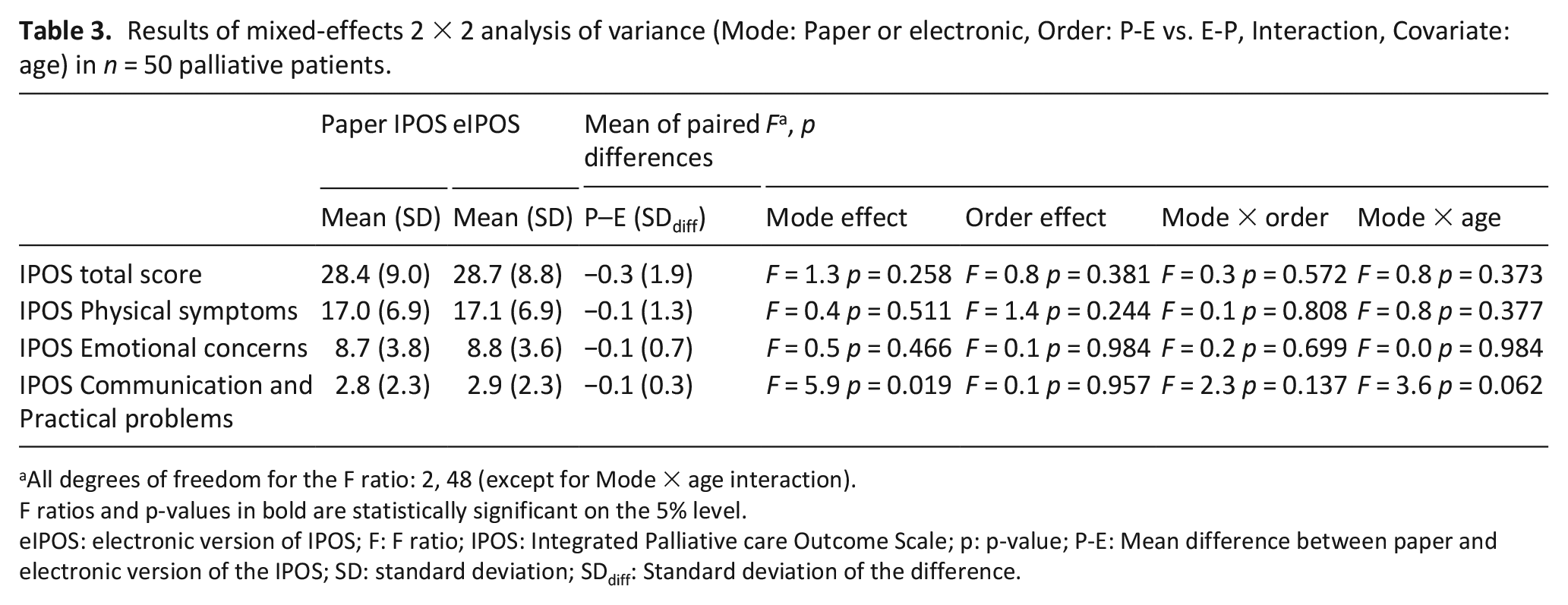
All degrees of freedom for the F ratio: 2, 48 (except for Mode × age interaction).
F ratios and p-values in bold are statistically significant on the 5% level.
eIPOS: electronic version of IPOS; F: F ratio; IPOS: Integrated Palliative care Outcome Scale; p: p-value; P-E: Mean difference between paper and electronic version of the IPOS; SD: standard deviation; SDdiff: Standard deviation of the difference.
Results of mixed-effects 2 × 2 analysis of variance for outcome mean time to complete in n = 50 palliative patients.
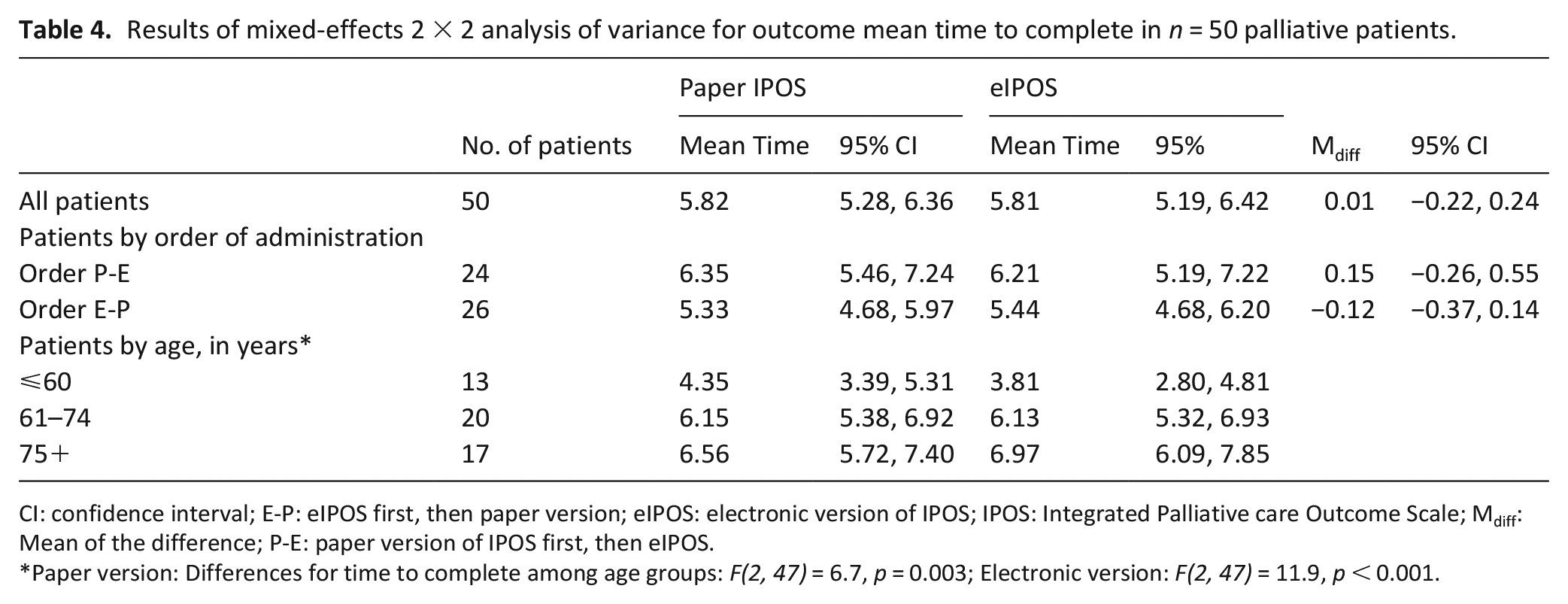
CI: confidence interval; E-P: eIPOS first, then paper version; eIPOS: electronic version of IPOS; IPOS: Integrated Palliative care Outcome Scale; Mdiff: Mean of the difference; P-E: paper version of IPOS first, then eIPOS.
Paper version: Differences for time to complete among age groups: F(2, 47) = 6.7, p = 0.003; Electronic version: F(2, 47) = 11.9, p < 0.001.
All ICCs for the comparison of IPOS and eIPOS were ⩾0.95 (see Table 5). Appetite loss and Drowsiness presented the lowest ICCs with 0.95 (95% CI: 0.92; 0.97). Constipation and Feeling not at peace (spiritual concerns) had ICCs of 0.96 (95% CI: 0.94; 0.98). Four items (Family anxiety, Feeling depressed, Fatigue, Nausea) had ICCs of 0.97 (95% CI: 0.95; 0.98). All other items showed ICCs of 0.98 or above. The lowest kappa score was found for Drowsiness (κ = 0.78), followed by Appetite loss and the IPOS Emotional concerns subscale with κ = 0.82, respectively. Nine items, two subscales and the IPOS total score showed κ above 0.92.
Spearman correlations, prevalence and bias adjusted kappa coefficients (PABAK), mean differences (with 95% confidence interval), intraclass correlation coefficients for the agreement between paper IPOS and eIPOS (n = 50).
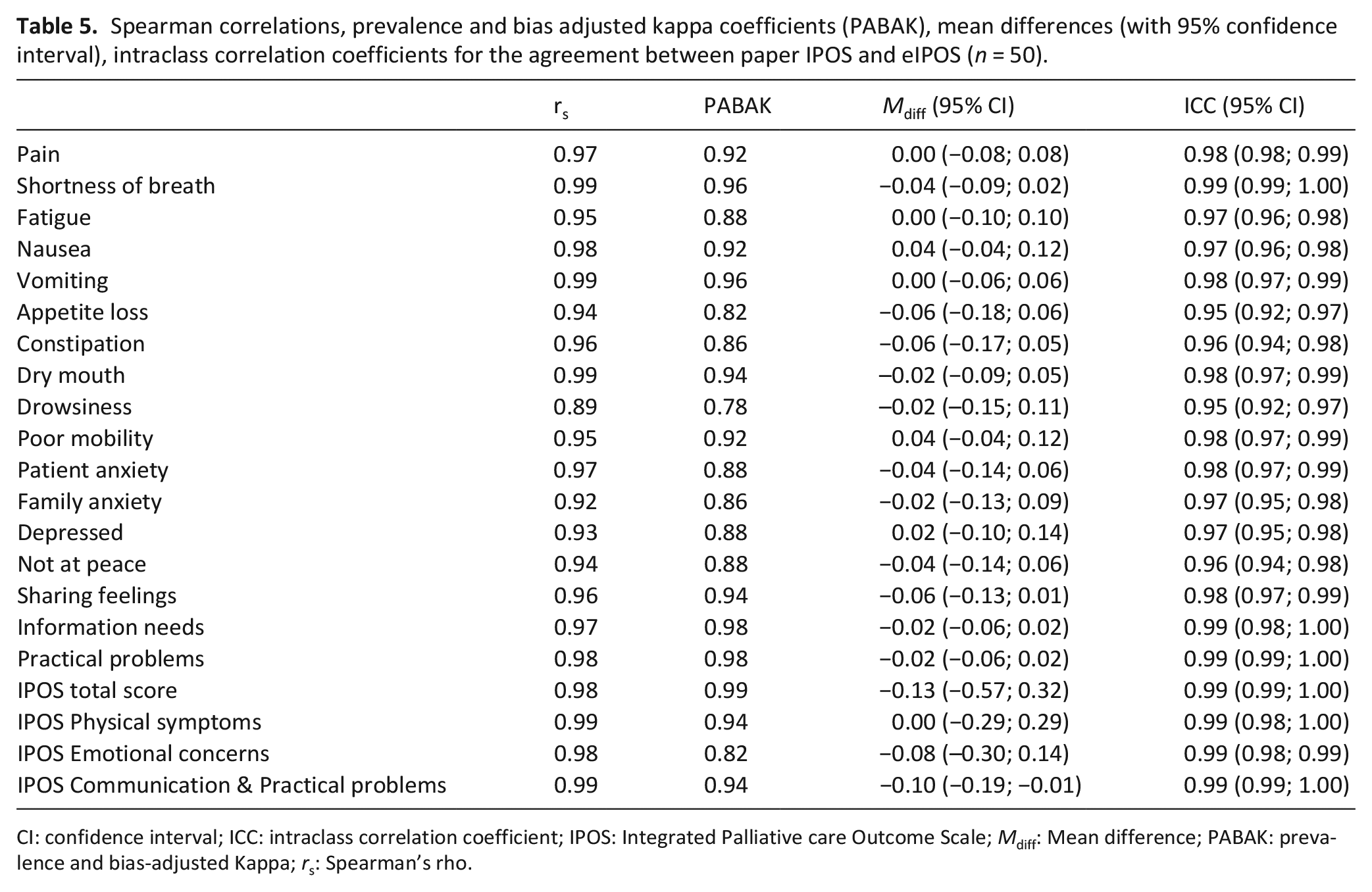
CI: confidence interval; ICC: intraclass correlation coefficient; IPOS: Integrated Palliative care Outcome Scale; Mdiff: Mean difference; PABAK: prevalence and bias-adjusted Kappa; rs: Spearman’s rho.
Bland-Altman plots for the three subscales and the IPOS total scores for the comparison of paper version and eIPOS are presented in Supplemental Appendix 2. The systematic bias was largest for the IPOS total score with mean difference −0.13 (limits of agreement: −3.14; 2.89), followed by the IPOS Communication & Practical problems subscale (Mdiff = −0.10, limits of agreement: −0.69; 0.49). The IPOS Emotional concerns subscale showed a systematic bias of −0.08 (limits of agreement: −1.61; 1.45). The IPOS Physical symptoms subscale was measured without a systematic bias between the modes (Mdiff = 0.00, limits of agreement: −1.94; 1.94).
Concordance of free-text answers
Thirteen participants overall did not volunteer any free-text main problems. Of those indicating main problems, 81% showed exact concordance of answers. Volunteered problems were mainly physical symptoms. Four persons volunteered different problems in the electronic version and three changed the order of main problems.
Twelve patients volunteered additional symptoms at the end of the IPOS symptom list. The concordance was nearly perfect, with one person scoring one symptom differently.
Discussion
Main findings/results of the study
The results of this randomised crossover trial indicate highly comparable and concordant responses between the paper version of IPOS and the eIPOS, at the total score, the subscale scores, and the individual item level. Mean summary and subscale score differences were very small (<1% of score ranges) and non-significant throughout all analyses. ICCs between paper and electronic scores were very high, significant and all exceeded the cut-off of >0.90.24,25,28,29 Appetite loss, Constipation, Drowsiness and Feeling not at peace showed the lowest ICCs in comparison to other items, the subscales, and the total score. The concordance extends to volunteered main problems and concerns as well as volunteered symptoms. The preference for the eIPOS was higher than for the paper version. A mode × age interaction effect was shown for the IPOS Communications & Practical problems subscale, and a significant age effect was shown for completion time.
What this study adds
Compared to studies assessing the concordance of paper and electronic versions of outcome measures in populations of patients with advanced diseases (mostly cancer),36,41–44 our sample comprised older patients, an equal gender distribution with a more heterogenous disease variety due to its palliative sampling frame. Contrary to the age and gender bias in equivalence studies reported in general clinical populations,24,45 a significant mode × age interaction effect was only observed for items Sharing feelings with family/friends, Information needs and Practical problems. Symptoms and emotional concerns were reported in equal manner between the modes. This shows that electronic adaptations of measures are possible despite the challenging palliative setting.
With the exception of one study in cancer patients, 44 equivalence studies report a higher preference for electronic versions of PROs for 52%–67% of the sample.36,41–43 Since qualitative data is missing, this preference is not explained. Equivalence studies in general populations with non-advanced disease indicate that the preference of electronic outcome measures is strongly a function of age.24,45 In palliative care, however, advanced illness and a traditional focus on delivering interventions via expert face-to-face communication coupled with a generally older population may hinder the successful implementation of electronic versions. A successful inclusion of self and proxy-reported electronic measures within an outpatient hospice population has been shown in the past.17,46 It is also worth pointing out that electronic completion of outcome measures does not preclude face-to-face interaction and follow-up communication.
The level of concordance of the paper and electronic versions of a self-reported outcomes found in the present study was excellent. The agreement found was higher than in similar studies testing the equivalence for quality of life and/or morbidity measures and showing acceptable to good ICCs of ⩾0.7,41,43 or moderate to good agreement based on weighted kappa coefficients.36,42,44 None of these cited studies, however, reached consistently high ICCs across both the total score and almost all subscale scores as we did in our study. Systematically reviewed features leading to high agreement have been coupled to randomised designs of shorter duration,22,24,47 features clearly met in our study. High agreement may also be attributable to the deliberate early involvement of patients in the development of the electronic version via co-design. Additional research is needed to understand how visual factors contribute to high agreement between paper and electronic versions. To help older adults and/or those with peripheral neuropathy, qualitative evidence has also supported stylus or pen entry of data into electronic devices instead of the more common swipe-and-touch techniques. 48
With the demonstration of high reproducibility and concordance between the two versions, the regular use of electronic IPOS in palliative home care may help harness the power of rapid, real-time assessment and feedback to patients and clinicians. This might also enhance interdisciplinary communication and care.49,50 The setting itself need not be a barrier for the successful adoption of electronic versions.51–53 However, implementation strategies need to recognise barriers and facilitators specific to the setting and a close collaboration with care teams is paramount.49,54,55
Limitations of the study
First, including only cognitively able patients might have resulted in a sampling bias, as up to 90% of palliative patients demonstrate some form of cognitive impairment before death. 56 Proxy-rated staff versions are available, but no proxy version for informal caregivers exists yet. Both should be tested for measurement equivalence when migrating to an electronic version. Second, the timing of assessments and selecting the appropriate wash-out period is a challenge in PC due to the fast-changing symptom burden specifically in inpatient populations as evidenced by often low to moderate test-retest reliability of measures. 6 Albeit we could not detect a significant order effect, these carryover effects cannot be excluded. The lower kappa values for the emotional subscale may point towards differences in interpretation of the underlying constructs being measured by the items and should be addressed in future studies on content and cross-cultural validity. 57
Conclusion
Following the recommendations of the ISPOR guidelines, the results show that eIPOS is a valid and reliable measure in the palliative setting. Paper and electronic versions of the IPOS can be considered equivalent and interchangeable. This means a fundamental step towards a more widespread routine implementation of measures and their positive effects for the palliative home care setting. The challenge of using data from electronically implemented outcome measures effectively in routine clinical care remains, so that these measures can foster the patient-professional dialogue and help professionals deliver high-quality care.
Supplemental Material
sj-pdf-1-pmj-10.1177_02692163231157871 – Supplemental material for Measurement equivalence of the paper-based and electronic version of the Integrated Palliative care Outcome Scale (IPOS): A randomised crossover trial
Supplemental material, sj-pdf-1-pmj-10.1177_02692163231157871 for Measurement equivalence of the paper-based and electronic version of the Integrated Palliative care Outcome Scale (IPOS): A randomised crossover trial by Anna Bolzani, Sophie Kupf, Farina Hodiamont, Isabel Burner-Fritsch, Claudia Bausewein and Christina Ramsenthaler in Palliative Medicine
Footnotes
Acknowledgements
We want to thank all collaborating services (specialist and general PC home services), their staff and their patients for the collaboration and for their efforts in collecting data and supporting the study. A special thanks goes to all patients and their families for participating in our study and giving us their time.
Author contribution
CB is the chief investigator and responsible for the design of this study. AB is the principle investigator and responsible for the conduct of this study. CB, FH, CR and AB conceptualised the study. SK, IBF and AB collected the data. SK and CR analysed the data. CR drafted the manuscript, with the support of AB, IBF, and SK. All authors provided critical feedback to the manuscript, read and approved the final draft.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Federal Joint Committee German Innovation Fund (Innovationsausschuss des Gemeinsamen Bundesausschuss), grant number 01VSF17014. The funder had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Ethics and consent
This crossover trial received research ethics approval from the local research ethics committee of the LMU University Hospital Munich (REC ref no: 18-871). All participants provided informed written consent. All study procedures adhere to the World Medical Association Declaration of Helsinki.
Data management and sharing
Due to data sharing restrictions set out by the ethics committee and the local data governance department, the full datasets used and analysed are only available from the corresponding author on reasonable request. R code for analyses can be obtained via contacting the corresponding author.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
