Abstract
Background:
Care costs rise towards the end of life. International comparison of service use, costs and care experiences can inform quality and improve access.
Aim:
The aim of this study was to compare health and social care costs, quality and their drivers in the last 3 months of life for older adults across countries. Null hypothesis: no difference between countries.
Design:
Mortality follow-back survey. Costs were calculated from carers’ reported service use and unit costs.
Setting:
Palliative care services in England (London), Ireland (Dublin) and the United States (New York, San Francisco).
Participants:
Informal carers of decedents who had received palliative care participated in the study.
Results:
A total of 767 questionnaires were returned: 245 in England, 282 in Ireland and 240 in the United States. Mean care costs per person with cancer/non-cancer were US$37,250/US$37,376 (the United States), US$29,065/US$29,411 (Ireland), US$15,347/US$16,631 (England) and differed significantly (F = 25.79/14.27, p < 0.000). Cost distributions differed and were most homogeneous in England. In all countries, hospital care accounted for > 80% of total care costs; community care 6%–16%, palliative care 1%–15%; 10% of decedents used ~30% of total care costs. Being a high-cost user was associated with older age (>80 years), facing financial difficulties and poor experiences of home care, but not with having cancer or multimorbidity. Palliative care services consistently had the highest satisfaction.
Conclusion:
Poverty and poor home care drove high costs, suggesting that improving community palliative care may improve care value, especially as palliative care expenditure was low. Major diagnostic variables were not cost drivers. Care costs in the United States were high and highly variable, suggesting that high-cost low-value care may be prevalent.
Large variation exists in the health service use near the end of life and a 10% of patients are especially high-cost users.
A high proportion of care costs in the last year of life occur in the last 3 months.
Patients nearing the end of life often prefer to be cared for at home, but habitually spend much time in hospital.
Our samples are from older patients with higher risks and more complex needs, having had contacts with specialist palliative care services in three countries where palliative care services are established and integrated into the health care system.
In the last 3 months of life, more than 80% of the total health and social costs were attributable to hospital care and the costs of palliative care were only ~10%, despite the fact that all patients had accessed specialist palliative care and reported high satisfaction with these services.
Uniquely we were able to compare actual costs, based on utilization, therefore providing a more robust comparison between countries with different reimbursement systems.
Cost distributions in England were more homogeneous, and with lower mean costs, than in both the United States and Ireland.
Financial difficulties and poor experience with home care services were associated with being a high-cost patient, but having cancer or non-cancer, or having multimorbidity was not.
Increasing community palliative care services may help older patients at the end of life avoid unwanted hospital care and increase care quality, value and access, and it should be a policy priority.
People with poverty, increased age and receiving poor home care should be a target for future interventions to improve care at the end of life.
The lower and more homogeneous costs in England require investigation, including the effects of different payment/reimbursement systems for health care.
Introduction
Health and social care costs are high in the last 3 months of life, yet patients and those close to them often report poor experiences.1,2 Much time is spent in hospital, despite most people preferring to be at home.3,4 In Manitoba, Canada, in the last 6 months of life, decedents (who represent 1% of the adult population) generated 21.3% of the total cost. 5 Aldridge and Kelley suggested that 13% of the US$1.6 trillion spent on the US health care was for individuals in their last year of life; much accounted for by a few very high-cost patients. 6 A recent analysis of Medicare costs suggested the last year of life was ~20% of spending, with costs increasing rapidly in the last days of life, especially when patients are in hospital. 7 Ensuring an appropriate balance of treatments with curative and palliative intent and avoiding inappropriate treatments or transitions is not only desirable for patients and families8,9 but also benefits health care policy and expenditure.10–12
Annual numbers of deaths will escalate in many countries,13–15 stressing future services. 16 Adults over 65 years of age represent >12% of the population (17.7% in England, 12.6% in Ireland and 13% in the United States). Most deaths occur in this age group. Variations in national policies and palliative and other care entitlements mean that international comparisons can inform improving access, care quality and cost-effectiveness.17–19 There are no direct international comparisons of cost patterns at the end of life across different health systems. Cost comparisons rarely consider care quality alongside costs to gauge value (quality/costs), despite quality being a palliative care cornerstone. To fully support informed choices, we need to understand patterns of service use and associated factors, including services outside of hospitals, which are often missed. 20
Therefore, we aimed to determine and compare across three countries, and disease groups focusing on older patients with complex needs, who might have higher chances of a better experience because of getting in touch with the specialist palliative care: (1) patterns of hospital, primary and community health service use by older adults known to palliative care services; (2) main drivers of variation in use of services and resultant costs and (3) experience of bereaved carers with services (as a measure of care quality) and its relation to care costs. We tested the a priori null hypothesis that there was no difference between countries in health and social care costs.
Method
Study design
We conducted a mortality follow-back postal survey of key informants (normally relatives and carers) of decedents identified by palliative care services in participating hospitals. Reporting follows STROBE 21 and MORECARE statements. 22 See declarations for ethical approvals.
Settings
Three countries included are in the top 10 of the Economist Intelligence Unit’s Quality of Death Index; rankings (scores) are the following: England – 1 (93.9), Ireland – 4 (85.8) and the United States – 9 (80.8), 23 with differences in health care systems (England: National Health Service; Ireland: National Health Insurance; the United States: Private Health System; palliative care covered by most insurance agencies and Medicare and Medicaid.) 24 and roles of philanthropy supporting hospice and palliative care, 19 and participating palliative care services in London (England), Dublin (Ireland) and New York, San Francisco (USA) were the established hospital palliative care consulting teams in all countries, a hospital-based community outreach team in London and an inpatient palliative care ward in New York. Details of the participating services are found elsewhere. 25
Inclusion criteria
We identified patients aged ⩾ 65 years who had accessed (⩾ 1 contact) a participating palliative care team and died 4–10 months prior to the survey date. Their next of kin (referred to hereafter as ‘carer’), as recorded in clinical records, was sent study information and a postal questionnaire from their clinical service (following data-protection regulations), with a pre-paid envelope addressed to the research team. All data were analysed anonymously.
Questionnaire and data collection
Consenting carers returned a self-report questionnaire,26,27 pre-piloted in all countries. The Client Service Receipt Inventory (CSRI)28–30 collected health and social care use in the last 3 months of life. This captured the detail of service utilization. Carers rated the quality of services in different places (e.g. hospital, home) using Likert-type scales. Health-related quality of life of patients 3 months and 1 week before death was asked using standard five items in EQ-5D-3L.31,32 Carers reported demographic data, including socio-economic status, clinical and living arrangements. This was supplemented by patient record data on age, diagnosis and co-morbidities.
Analysis
We analysed demographic and clinical data for each country and compared age and place of death with population-based national statistics. We could not compare the characteristics of responders and non-responders due to ethical and data-protection constraints. We converted five EQ-5D dimensions (mobility, self-care, usual activities, pain/discomfort and anxiety/depression) into an index score using English population values, 33 which ranged from −0.594 to 1.
To determine the care costs for each patient, we multiplied the quantity of specific services used according to the CSRI with corresponding country-specific unit costs.34–36 Where published unit costs were unavailable, in Ireland and the United States, we used the World Health Organization (WHO)-CHOosing Interventions that are Cost-Effective (CHOICE) method, 37 which estimates unit cost values for primary and secondary health care services. All costs were translated into USD (US$) for comparison, using the purchasing power parity (PPP) index. To calculate aggregated care costs, we grouped services into the following:
Hospital = services provided in hospitals (intensive care, inpatients and outpatients), emergency room visits, ambulance services and day case treatments;
Community = services received either in care homes or at home;
Palliative care = services from a dedicated palliative care unit (e.g. within specific units in hospitals or inpatient hospices), from specialist palliative care teams, dedicated or specialist nurses (e.g. Marie Curie, Macmillan or Irish Cancer Society/Irish Hospice Foundation nurses; see notes under e-Table 4 for roles of these nurses).
We tested our null hypothesis using one-way analysis of variance (ANOVA) tests. We analysed the costs for people with cancer separately from those with non-cancer because of their different access levels to palliative care, which also varies internationally and may have influenced costs (the Bonferroni correction for multiple comparisons). Missing values in service use volume were imputed with medians. Sensitivity analyses applied English unit costs to service use in all countries, in case differences in aggregate costs were due to differences in country unit costs, rather than service use.
We plotted the cost distributions for each country. We identified individuals in the top 10% care costs, using Aldridge and Kelley’s 6 approach and conducted logistic regression analysis to identify factors associated with being a high-cost patient. Regression analyses explored the relationship between care costs and carers’ experience with care, with and without adjustment for co-morbidities. We included explanatory variables based on theories and literature. Sensitivity analysis also explored factors associated with total costs using regression analysis. We used complete cases only. We imputed missing values, assuming missing at random, as a sensitivity analysis to see if this influenced the results.
Our sample size is based on being able to detect a difference in the mean care costs between countries, with 80% of power and α = 0.05 (0.025 with Bonferroni correction for two pairs of comparison), which would require 229 individuals in each group. To allow regression analysis, anticipating 25 explanatory variables: 50 + 25 × 8 = 250. 38 Therefore, we aimed to recruit > 750 people and to allow country comparison and subgroup analysis > 230 per country. Allowing for 30% response, as is usual in such surveys, 39 we approached ~2000 individuals across the three countries.
Results
Sample characteristics (deceased patients and their bereaved carers)
We received 767 completed surveys: 245 (32.4%) of 756 delivered surveys in London, 282/580 (48.6%) in Dublin, 131/548 (23.9%) in New York and 109/342 (31.9%) in San Francisco.
Patients’ mean age at death was 80 years; 52.8% were women, most were married/had a partner (51%) or widowed (33%) (Table 1). Sample decedents were slightly younger than in national populations; the proportion was slightly higher for hospital deaths and slightly lower for home deaths (e-Tables 1 and 2). In the United States, decedents were ethnically more diverse (58% White) than in England and Ireland (88%, 99% White). Overall, 49% died from cancer; higher in England (60%) than in Ireland (48%) and lowest in the United States (39%); 50% of patients had ⩾ 2, and 6% ⩾ 6, co-morbidities; data not available in Ireland; 4% had been finding it difficult or very difficult financially and 10% reported being just about alright. As expected in this population, EQ-5D index scores were low and fell towards death with no significant difference between countries. Most responding bereaved carers were spouses/partners (36.3%) or daughters/sons (47.7%); 70% were women and average age was 60 years. Missing values were infrequent and scattered with no particular pattern, 2%–5% of variables.
Characteristics of the deceased patients and respondents (bereaved carer) (unit: %, mean (SD)).
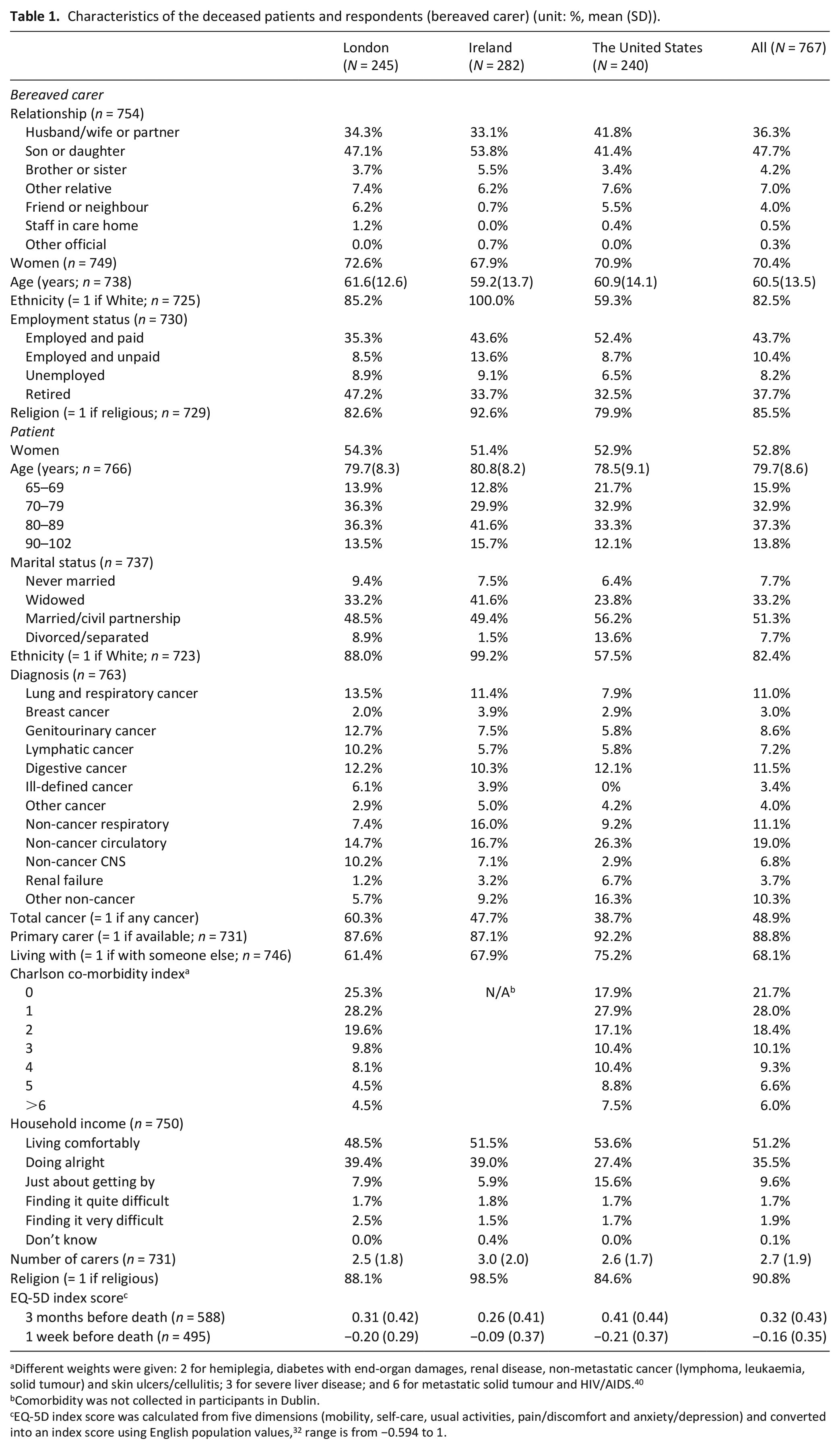
Different weights were given: 2 for hemiplegia, diabetes with end-organ damages, renal disease, non-metastatic cancer (lymphoma, leukaemia, solid tumour) and skin ulcers/cellulitis; 3 for severe liver disease; and 6 for metastatic solid tumour and HIV/AIDS. 40
Comorbidity was not collected in participants in Dublin.
EQ-5D index score was calculated from five dimensions (mobility, self-care, usual activities, pain/discomfort and anxiety/depression) and converted into an index score using English population values, 32 range is from −0.594 to 1.
Patterns of hospital, primary and community health service in the last 3 months of life
Almost all patients used hospital services, with 70%–85% hospitalized at least once. Over half of the non-cancer patients in the United States had an intensive care unit (ICU) stay, as did over one-third of cancer patients in Ireland and the United States. A general practitioner/family doctor (GP/FD) was the main health care professional in community settings, especially in England and Ireland. Patients in the United States (36% cancer, 39% non-cancer) were far more likely to stay overnight in a palliative care unit/inpatient hospice than patients in other countries; only 14%–15% of cancer and 2%–3% of non-cancer patients had hospice/palliative care unit stays in England or Ireland. Cancer patients received more palliative care than non-cancer patients in England and especially in Ireland (e.g. palliative care team was seen by 47% of cancer and 21% of non-cancer patients in England; 37% of cancer, 6% of non-cancer patients in Ireland) (e-Table 4).
Health and social care costs in the last 3 months of life
Cost distributions were positively skewed; median costs were lower than mean costs (Figure 1). In England costs were more homogeneous, many patients had low per-user cost. In the United States and Ireland, more patients, especially with non-cancer, had higher cost. Mean per-patient costs were lowest in England; the United States had more than double English care costs. Results were similar in the sensitivity analyses using the same unit costs for all countries. Hospital costs were 79%–88% of total care cost (Table 2, e-Table 5).
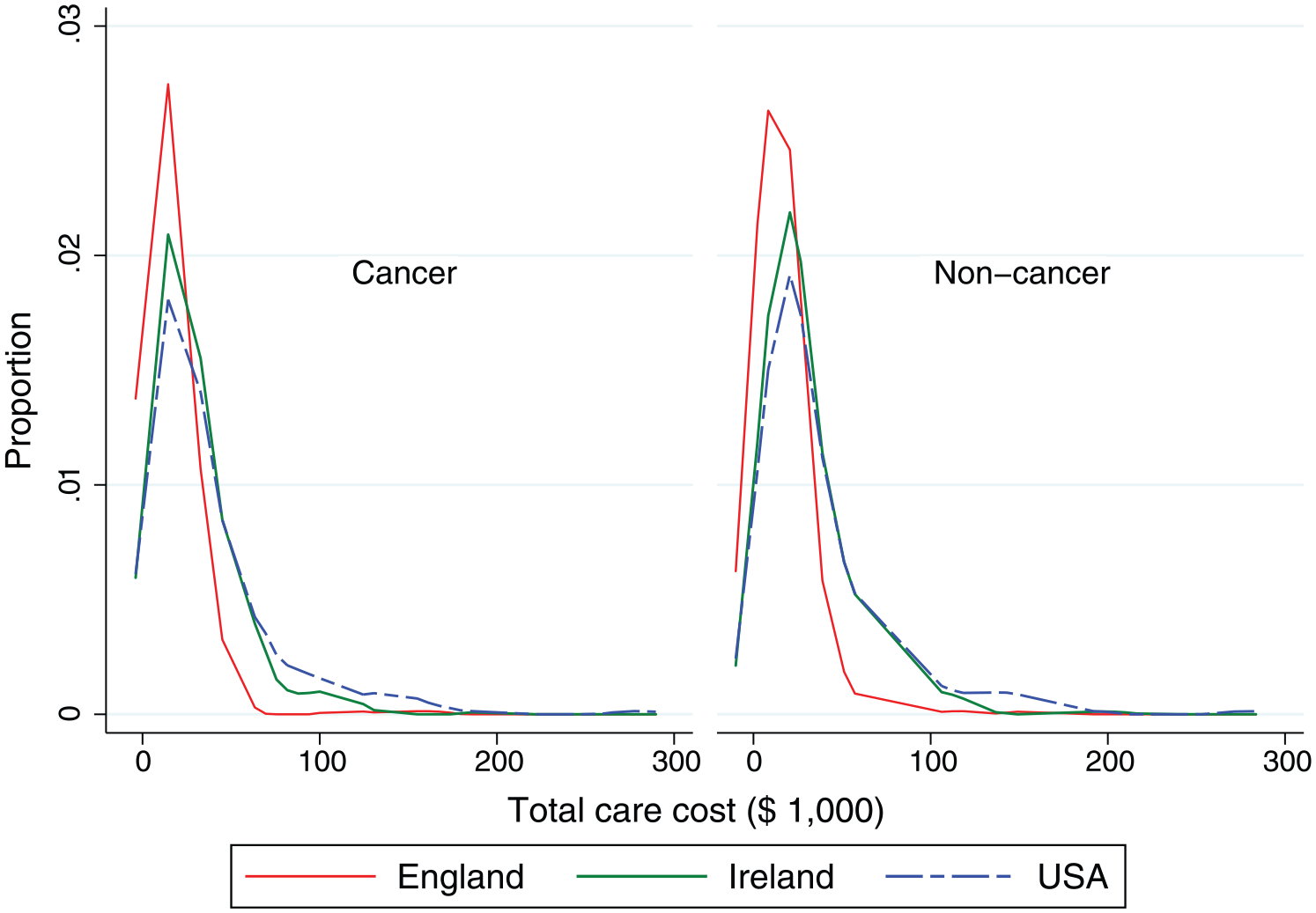
Distribution of hospital care costs in the last 3 months of life for older patients who had access to specialist palliative care in England, Ireland and the United States.
Hospital and total health and social care costs in the three countries.
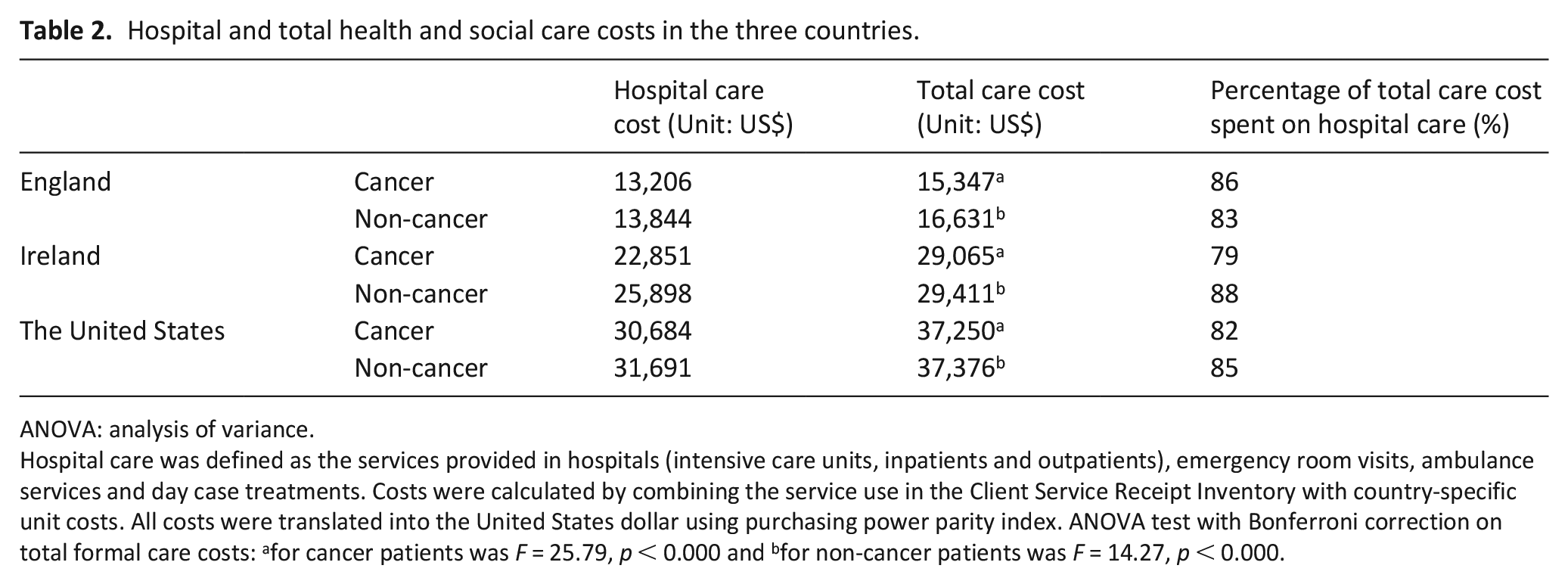
ANOVA: analysis of variance.
Hospital care was defined as the services provided in hospitals (intensive care units, inpatients and outpatients), emergency room visits, ambulance services and day case treatments. Costs were calculated by combining the service use in the Client Service Receipt Inventory with country-specific unit costs. All costs were translated into the United States dollar using purchasing power parity index. ANOVA test with Bonferroni correction on total formal care costs: afor cancer patients was F = 25.79, p < 0.000 and bfor non-cancer patients was F = 14.27, p < 0.000.
Of total care costs, community care costs were 9(16)%, 5(8)% and 9(11)% in England, Ireland and the United States in cancer(non-cancer), respectively. In cancer patients, palliative care comprised 5% (US$775), 13% (US$4444) and 8% (US$3058) of costs in England, Ireland and the United States, respectively (e-Table 5, e-Figure 1).
Main drivers of variation in use of services and resultant costs
Across the three countries, the mean total costs were US$76,919 per patient in the highest 10% and US$19,865 in the rest (Table 3): a small number – 25, 28 and 25 patients – used 29%, 30% and 34% of the total costs in England, Ireland and the United States, respectively. These highest cost patients (10%) spent 3.9-fold the total costs and 4.3-fold the hospital costs of the remaining 90%.
Health and social care costs in the last 3 months of life among older patients.
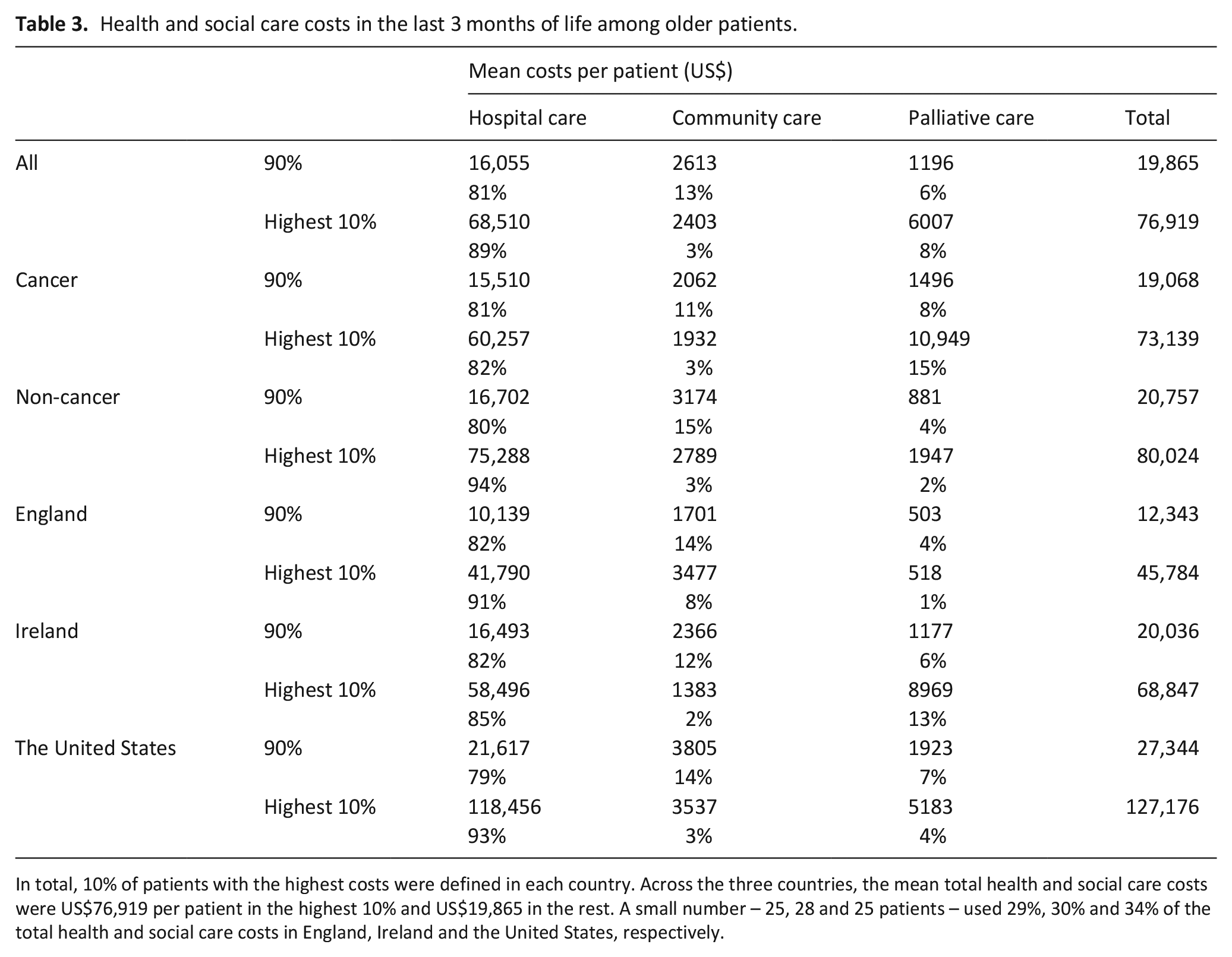
In total, 10% of patients with the highest costs were defined in each country. Across the three countries, the mean total health and social care costs were US$76,919 per patient in the highest 10% and US$19,865 in the rest. A small number – 25, 28 and 25 patients – used 29%, 30% and 34% of the total health and social care costs in England, Ireland and the United States, respectively.
Being aged > 80 years (β = 0.07, 95% CI 0.03– 0.10) and experiencing financial difficulty (β = 1.92, 95% CI 0.42–3.41) were significantly associated with being in the highest 10% in all countries. When we included the number of co-morbidities in the model, age and financial difficulties remained associated with being a high-cost user. The number of co-morbidities was not associated with care costs (e-Table 6).
Bereaved carer’s experience of services and relationship to care cost
Bereaved carers rated the services provided to patients as ‘good’ or ‘better’, except at care homes in the United States which were rated lower, as ‘fair’ to ‘good’. Consistently, bereaved carers rated care in hospices/palliative care units as the highest (Figure 2, e-Table 7).
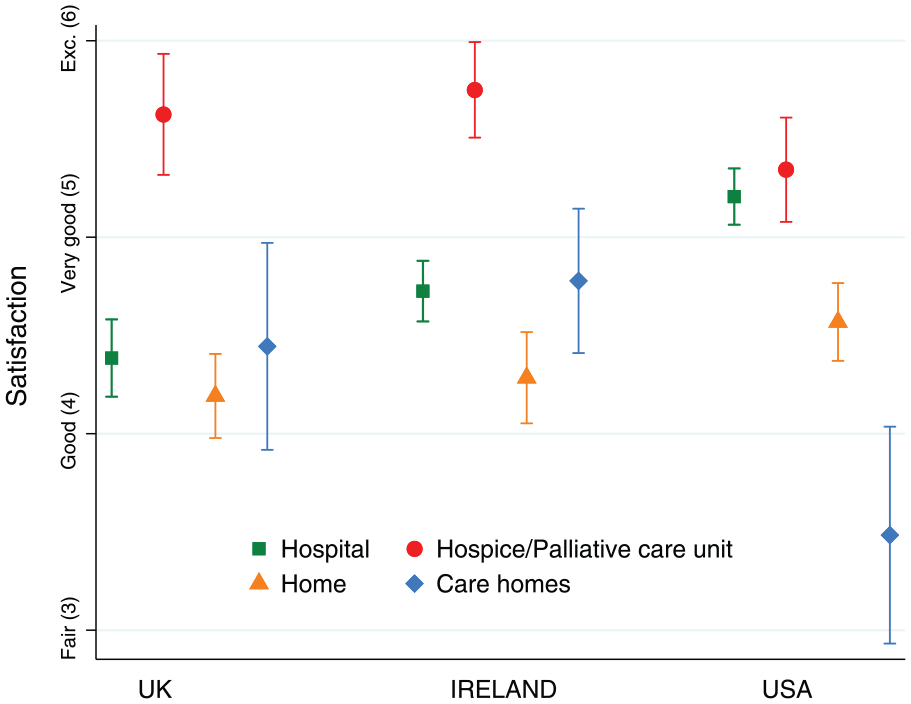
Carer’s ratings of the quality of care received at home, in hospitals, care homes and hospices/palliative care units separately for England, Ireland and the Unites States.
Being widowed was associated with poorer reported quality of hospital care, whereas being divorced/separated was associated with poorer quality of home care. Poorer quality of home care was associated with higher hospital costs (β = −0.84, 95% CI −1.36 to −0.31); conversely, reporting better quality of home care was associated with both higher community and palliative care spending (β = 0.69, 1.34, respectively, e-Table 8). This was consistent when adjusting for co-morbidities (e-Table 9).
Discussion
Main findings
This large international study of older people known to palliative care services found that in the last 3 months of life, hospital care accounts for over 80% of total health and social care costs. Community and palliative care costs were low, ranging 6%–16% and 1%–15%, respectively. Care costs differed between countries, being the lowest and most homogeneous in England. The 10% highest cost patients accrued four times the care costs of the remaining 90%. Being aged ⩾ 80 years and having difficulty living on current income (indicating poverty) were significantly associated with higher total costs across all three countries. Clinical factors were not or only marginally influential. Care quality was highest in palliative care unit/inpatient hospices. Poor home care was associated with higher hospital costs.
Strengths and limitations
Our findings have limitations; at the University of California, San Francisco (UCSF; the United States) we recruited from a palliative care service limited to inpatients, the Irish sample included a high proportion that died in hospital, both potentially inflating hospital costs. We focussed on four major cities in these three countries, and so our results may not represent the countries as a whole. Nonetheless, these cities contain large proportions of each country’s population. Furthermore, our findings are supported by previous research revealing that hospitals are the main components of costs of care near the end of life.7,41–43 Unit costs are not fully representative due to multiple payers and providers in all countries. Intensive and critical care beds vary internationally in their supply, patterns of admission and/or discharge, as well as, interpretation and severity of patients admitted.44,45 We need more information on the health infrastructure and clinical/policy decision-making to understand how these differences affect our findings. The EQ-5D was used because it is a standard quality of life measure in health economic studies and so our values provide useful context. EQ-5D scores were low, and its limited domains may have missed symptom and patient experience differences between countries. 46
A strength is that we used published information, supplemented by the extrapolated costs using methodology adopted by the WHO, making the most of available information. Sensitivity analysis using English unit costs confirmed that the patterns were driven by differences in service use. Future research is needed to improve unit cost accuracy. By sampling from those known to palliative care services, we were able to focus on decedents who were more likely to have high levels of needs and high costs, but could not focus on very sick patients who did not access palliative care services. Mortality follow-back surveys inherently suffer from sample and recollection bias. Although our response rate is above average for this type of study,4,47 as anticipated it was below 50% in all sites. For ethical and data-protection reasons, we could not compare responders and non-responders and so relied on comparisons with national data, which suggested our sample was reasonably representative.
The sample missed many older patients without caregivers; understanding this group requires prospective study. Respondents may have inaccurately distinguished parts of hospital care (e.g. intensive care) or mistaken outreach palliative care services with other community services; sometimes palliative care might have been reported as community services and vice versa. 42 However, as community service contacts were low, major underestimation is unlikely. Linking routinely collected acute hospital and community service data could provide more information. Carers’ experiences differ from patients’ and are not patient outcomes. Nevertheless, carers’ experiences are crucial; family views influence patient preferences 48 and good experiences may reduce carers’ grief and morbidity. 4 Uniquely, we collected data firsthand, using standard procedures specifically for our objectives, rather than relying on secondary data sources as in many international comparisons. This enhances the validity of our findings.
What this study adds and implications
In this first comparison of overall care costs at the end of life between countries, overall costs for patients in England were around half of those in Ireland or the United States. These differences were significant, not supporting our a priori hypothesis. As these costs are based on utilization combined with country-specific unit costs rather than on what individuals pay, our data uniquely provide reliable comparisons of what people received, irrespective of payment methods. When individual patient costs were considered, more English patients were at the lower range and fewer at the higher range of costs, compared with Ireland and the United States. To our knowledge, this has never been reported before. Reasons for this apparent ‘standardization’ of provision and costs need investigation. It may relate to the health systems – National Health Service (England), National Health Insurance (Ireland), Private Health System (the United States). Variations in unit costs did not explain this. The cost differences result from different quantities and intensities of services used: more patients used more expensive care services in the United States and Ireland than in England. The lower quality of home care (and higher quality of hospital care) in the United States might have also driven higher hospital costs. We were surprised by the high use of ICUs, especially in this population. Between-country differences may reflect different definitions and criteria for ICU admission.44,45 We found no evidence that higher ICU use led to higher care quality, but this was not a focus of our study.
We were surprised by the low costs of palliative care, which included inpatient and a range of community and hospital services; < 4% of total cost. This was despite our study including only patients who had received some palliative care. Given a major preference for quality of life in this age group, 25 the high quality of palliative care services reported in this study and others,49–51 and growing evidence that palliative care teams lower costs and improve experiences in serious illness;52,53 these results are concerning and suggest that greater support for palliative care services is needed.
In all countries, we identified a patient group with higher costs, with the top 10% accounting for around four times that of others; these are important for future targeted support. Low satisfaction of home care was consistently associated with higher hospital costs. Poor home support may drive admission to hospital; our findings support other studies that suggest that feeling ‘unsafe’ at home leads to emergency department (ED) attendance, 54 and that skilled and available community services are key to ensuring home care.4,55,56 Home palliative care can reduce hospital admissions, as well as provide care of better quality; strengthened home palliative care may be important to improve value (quality/costs). 4
Our finding that being over 80 years of age was associated with higher care cost was unexpected; other studies have found that care cost near death is lower for older decedents, this warrants further investigation. 3 Co-morbidity did not affect costs. This differs from studies of community-dwelling older people, where multimorbidity was associated with higher costs.57,58 A recent meta-analysis found that the economic benefits of palliative care are greatest in the face of increasing multiple morbidity. 52 Receiving some palliative care, as in our study, may have protected against multimorbidity increasing costs, adding further evidence to support palliative care referral in this population.
Conclusion
In this first major study to compare costs and experiences of people in different countries near the end of life, we found that care costs are high, hospital care comprises > 80% of care costs, and palliative and community care use is low, despite patients receiving some palliative care. These novel findings suggest that improving palliative care access and intensity is an essential priority for health care policy, especially for non-cancer patients who had least access. Cost distributions in England were more homogeneous and with lower mean costs than in the United States/Ireland, which warrants further investigation. Poverty, increased age and poor home care were drivers of high costs and should be a target for future interventions to improve care quality and value at the end of life.
Supplemental Material
Revised_PMJ-18-0419_Care_costs_and_quality_Suppl – Supplemental material for Drivers of care costs and quality in the last 3 months of life among older people receiving palliative care: A multinational mortality follow-back survey across England, Ireland and the United States
Supplemental material, Revised_PMJ-18-0419_Care_costs_and_quality_Suppl for Drivers of care costs and quality in the last 3 months of life among older people receiving palliative care: A multinational mortality follow-back survey across England, Ireland and the United States by Deokhee Yi, Bridget M Johnston, Karen Ryan, Barbara A Daveson, Diane E Meier, Melinda Smith, Regina McQuillan, Lucy Selman, Steven Z Pantilat, Charles Normand, R Sean Morrison and Irene J Higginson in Palliative Medicine
Footnotes
Acknowledgements
This study was a part of BuildCARE, an international research programme supported by Cicely Saunders International (CSI) and The Atlantic Philanthropies led by King’s College London, Cicely Saunders Institute, Department of Palliative Care, Policy and Rehabilitation, UK. Irene J Higginson is a National Institute for Health Research (NIHR) Emeritus Senior Investigator. We thank all collaborators and advisors including service users. BuildCARE members: Emma Bennett, Francesca Cooper, Barbara Daveson, Susanne de Wolf-Linder, Mendwas Dzingina, Clare Ellis-Smith, Catherine Evans, Taja Ferguson, Lesley Henson, Irene J Higginson, Bridget Johnston, Paramjote Kaler, Pauline Kane, Lara Klass, Peter Lawlor, Paul McCrone, Regina McQuillan, Diane Meier, Susan Molony, R Sean Morrison, Fliss Murtagh, Charles Normand, Caty Pannell, Steve Pantilat, Anastasia Reison, Karen Ryan, Lucy Selman, Melinda Smith, Katy Tobin, Rowena Vohora, Gao Wei and Deokhee Yi.
CLAHRC acknowledgement: A component of this study is funded by the Collaboration for Leadership in Applied Health Research and Care (CLAHRC) South London at King’s College London Hospital Foundation Trust. The views expressed are those of the authors and not necessarily those of the NHS, our funders, the National Institute for Health Research or the Department of Health and Social Care.
Author contributions
I.J.H., C.N., D.E.M. and R.S.M. were grant leads. I.J.H., R.S.M. and C.N. are co-senior authors for this paper. B.A.D., C.N., K.R., R.M., R.S.M., D.E.M., L.S. and S.Z.P. were study PIs for the sites of London, Dublin, New York and San Francisco; M.S., L.S. and B.M.J. coordinated data collection in different sites. D.Y. led the analysis and with I.J.H. drafted the manuscript, with significant contributions from C.N., B.M.J. and K.R. Also, I.J.H. and D.Y. had full access to all the data in the study and take responsibility for the integrity of the data and analysis. All authors contributed to the development of study aims, integration of the interpretation across the BuildCARE study and analysis plan and provided critical revision of the manuscript for important intellectual content.
Availability of data and materials
The anonymized datasets supporting the conclusions of this manuscript are available upon request to the corresponding authors and BuildCARE team.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
The study received ethical approval from the Research Ethics Committee in Dulwich, London in England (Ref. 12/LO/0044); the ethics committees of the Mater Misericordiae University Hospital (MMUH) and Beaumont Hospital in Ireland (Ref. 1/378/1456 and Ref. 12/07, respectively) and the Institutional Review Board of the Icahn School of Medicine at Mount Sinai (Ref. GCO#11) and the University of California, San Francisco (UCSF; Ref. 13-1099) in the United States. In addition, in England NHS research and development approval was received. If participants were agreeable to being approached, a researcher fully explained the study to them, provided an information sheet and gained written informed consent. Adults unable to give informed consent or deemed too ill to complete any part of the interview were excluded.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The authors thank Cicely Saunders International (CSI, UK charity) and The Atlantic Philanthropies for the main support for this programme. Additional support was provided by the NIHR South London CLAHRC and Higginson’s NIHR Senior Investigator award. (Further details in acknowledgements.) The sponsors did not have any role in the design, conduct, interpretation, review, approval or control of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
