Abstract
Objective
Early mobility after cardiac surgery in the intensive care unit improves patient outcomes, yet implementation by clinicians remains inconsistent. To inform interventions to increase adoption, this study sought to explore clinicians’ perspectives on the definition of early mobility in the cardiac surgery intensive care unit and describe perceived barriers and facilitators to implementation.
Design
This qualitative study was guided by interpretive description.
Setting
A tertiary hospital that performs 1000 cardiac surgeries annually.
Participants
A total of 26 clinicians, including nurses, physicians, healthcare aides, respiratory therapists, and physiotherapists, participated.
Main Measures
The nine focus groups and four individual interviews were audio-recorded, transcribed verbatim, and analyzed using thematic analysis.
Results
Three themes and 13 categories were created from the data: (1) Incongruent operationalization and conceptualization (across micro, meso, and macro levels), (2) uncertainty, and (3) inconsistency in mobilization practices. Themes were rooted in ambiguous definitions of “early” and “mobility,” discrepancy between conceptualization and implementation of timing, varied interpretations of success, and perceived safety concerns. In addition, 4 barriers and 4 facilitators were identified and categorized into patient-care and clinician-related.
Conclusion
Developing early mobility protocols and education informed by clinicians may enhance its implementation in the cardiac surgery intensive care unit. Incorporating the perspectives of clinicians into early mobility protocol development is essential to support behaviour change and provide this beneficial care.
Keywords
Introduction
Early mobility is any active movement immediately after a specific event,1–3 such as cardiac surgery. Active movements include sitting, standing, getting out of bed to a chair, ambulation, or cycling.1,2 Early mobility in the intensive care unit after cardiac surgery is recommended, safe, and beneficial;4–9 however, it is inconsistently integrated into care.2,10 This may, in part, be related to the lack of engagement with cardiac surgery intensive care unit clinicians. Research teams thus far appear to be dominated by academic physicians, physiotherapists, and nurses.11–14 None of these teams has explicitly described if their work included or was being informed by practicing cardiac surgery intensive care unit clinicians. Practical expertise of cardiac surgery intensive care unit clinicians is needed to support the successful development and integration of early mobility interventions into clinical practice.10,15
Clinician feedback is key because integrating early mobility into clinical practice requires a behaviour change. 10 Behaviour change is complex but can be planned through intervention mapping, a systematic method of developing, implementing, and evaluating interventions targeted at changing behaviours.16,17 Intervention mapping originated in health education 16 and has been successfully applied in healthcare. 17 For this reason, we are using intervention mapping to develop, implement, and evaluate an intervention to integrate early mobility into cardiac surgery intensive care unit clinical care. The first step in intervention mapping involves exploring the behavioural and environmental aspects of the behaviour targeted for change through qualitative and quantitative means.16,17 We previously completed a quantitative investigation of early mobility practice in our cardiac surgery intensive care unit. 2 We found that sitting in a chair was observed 10 times more frequently than any other mode of mobility. 2 Mobility was supported more frequently by family, either through direct assistance with mobility, alongside the healthcare team, or by being present with the patient while they sat in a chair. 2 Nurses, physiotherapists, and physicians were the healthcare providers observed supporting mobility. 2 These valuable insights into mobility practices would benefit from being supplemented by qualitative data. Understanding why mobility occurs as observed is as important as understanding the current state of mobility.10,15 Also, understanding the healthcare team's perceived barriers and facilitators to early mobility is important, as behaviours are difficult to change if barriers are not removed, but change can be supported through leveraging facilitators.16,17 Therefore, this study aimed to gain insight into the perspectives of cardiac surgery intensive care unit clinicians on early mobility through qualitative methods. The research questions were: What are the perspectives of healthcare providers on early mobility in the cardiac surgery intensive care unit? How do they think about early mobility? How do they operationalize early mobility? What additional barriers and facilitators to early mobility do they perceive?
Methods
Approvals were obtained from both the University of Manitoba Research Ethics Board (HE2024-0036) and the St Boniface Hospital Research Review Committee (RRC/2024/2159) prior to the commencement of the study. All participants reviewed and signed a consent form prior to participating in a focus group. Participants were made aware that consent was an ongoing process, and they could withdraw consent until data analysis began, at which point it was no longer possible to remove their data from the study. No person withdrew their consent.
Our study was guided by interpretive description because the objective was grounded in the cardiac surgery intensive care unit, the area of clinical practice of the first author (EKP). 18 Unlike other qualitative methodologies that require the researcher not to use their clinical expertise in data analysis, a strength of interpretive description is that it values this expertise as a means to explore clinical research questions inductively. 18 Moving from the specific to the general, data are first described. Interpretation occurs through understanding qualitative data through a socially constructed reality that includes clinical expertise. 18 The reporting of this work follows the Standards for Reporting Qualitative Research. 19 See the Supplemental Materials for the checklist. See the Supplemental Materials for details on the guiding research paradigm and researcher characteristics and reflexivity.
Context and sampling strategy
The cardiac surgery intensive care unit at St Boniface Hospital, a tertiary hospital in Winnipeg, Manitoba, Canada and the setting of this study, has been described in detail elsewhere. 2 Briefly, St Boniface Hospital is the only hospital in the province of Manitoba where cardiac surgeries are performed. Around 1000 are performed annually, with the majority being coronary artery bypass grafting, valve surgery, or a combination of the two. 2 Thoracic aortic and short-term and durable ventricular assist device implantation surgeries are also performed. 2 There are 13 beds in this cardiac surgery intensive care unit. Staff who support mobility include nurses, healthcare aides, physiotherapists, and physiotherapy assistants. 2
The sample size was driven by information power. 20 The cross-case analysis and lack of guiding theory were balanced by the narrow aim and dense sample specificity of the study, leading to high information power and a moderate required sample size. 20 We aimed to recruit 30 clinicians from the cardiac surgery intensive care unit, including physicians, nurses, physiotherapists, healthcare aides, respiratory therapists, and perfusionists, through a combination of purposive and convenience sampling. 21 We targeted the specific clinician groups to attempt to gain representation from each through purposive sampling by sharing the recruitment materials with each group. 21 Within each clinician group, we included participants who volunteered to participate in the study through convenience sampling. 21 The inclusion criteria were clinicians currently working in the cardiac surgery intensive care unit with at least two years of experience.
The sample size was confirmed with data adequacy, as constant comparative analysis allowed for the recognition of alignment of data with the emerging themes and categories. 21 Passive recruitment occurred through sharing study information on notices in the unit and through an email from the cardiac surgery intensive care unit manager. Interested clinicians contacted EKP directly to determine a mutually agreeable time to participate. Recruitment occurred from June to July 2024. After signing the consent form, participants received an honorarium ($25 gift card).
Data collection and analysis
Demographic information was collected, including age, self-identified sex, years of experience in the cardiac surgery intensive care unit, and profession. Nine audio-recorded facilitated focus groups of 2–4 clinicians and four individual interviews were conducted to explore experiences with early mobility, including barriers and facilitators. Individual interviews were conducted with participants who could not attend a focus group session but wished to participate in the study. The focus groups and interviews lasted 30–90 min and were conducted in person at a mutually convenient time and place. Focus groups and interviews were conducted using a semi-structured guide (Supplemental Table 1). The facilitators/ interviewers (EKP and AA) documented perceptions of the focus group and interview processes, focusing on theoretical insights and methodological issues, in reflective journals used as another data source.
The audio recordings were transcribed verbatim to produce 13 transcripts (9 focus group transcripts and 4 individual interview transcripts). Transcripts and reflective journals were analysed using thematic analysis. 22 Specifically, EKP, AA, and JLH (the coders) first read the transcripts several times to gain familiarity. 22 The coders met in person and analysed the first transcript together. Each coder independently read and analysed two transcripts and then met to compare and contrast their coding. 18 Coding continued with each coder reading and analysing two additional transcripts. Coders independently analysed the same transcripts (i.e., EKP and JLH analysed transcript #3 and #8, and JLH and AA analysed transcript #5) to support the analysis of transcripts from diverse perspectives. 22
Initial codes were generated by grouping responses together based on similarities. 22 These initial codes were reviewed, and themes were created by conceptualizing the relationships among the codes. 22 The themes and codes were shared for review and revision by the broader research team, including clinical partners (CS and GB). 22 The themes and codes were then named and defined in a coding scheme. 22 Memoing was used to create an audit trail and allow for transparency on coding decisions. 18
Barriers and facilitators to early mobility beyond the themes created above were also inductively analysed. This analysis focused on identifying barriers and facilitators in a qualitative manner to ensure the breadth of these was made explicit. This differs from other approaches that are deductive, as these approaches may limit what barriers and facilitators are identified through the use of pre-existing frameworks. The interview guide included questions about what barriers and facilitators to early mobility participants perceive. We then reviewed the transcripts again, specifically looking for other barriers and facilitators mentioned throughout the discussions or those that were identified through the coder's clinical and professional backgrounds, particularly in relation to early mobility and supporting behaviour change. We inductively analysed all identified barriers and facilitators in the same manner that codes were created, moving from initial codes through revisions, then naming them, and finally creating a coding scheme. 22 The barriers and facilitators were categorized as patient-specific, clinician-specific, and unit-specific. This categorization occurred to support organizing the barriers and facilitators in a manner that supports their use in the development of future interventions. This analysis did not explore relationships among barriers, facilitators, and the themes created from the discussions. Rather, we only sought to identify the breadth of barriers and facilitators perceived by participants. The goal of identifying barriers and facilitators was to explicitly share these so that they can be considered in the development of future interventions. Memoing was again used to create an audit trail and allow for transparency on decisions. 18
Credibility
As appropriate for the methodology of interpretive description, credibility was evaluated rather than trustworthiness through the evaluation criteria described by Thorne. 18 These criteria include integrity, representative credibility, interpretive authority, and transparent analytic logic. 18 The integrity of this study is rooted in the epistemological approach. 18 The findings have representative credibility because they relate to early mobility in the cardiac surgery intensive care unit based on data sourced from healthcare providers in the cardiac surgery intensive care unit. Multiple team members independently analysed and compared coding to address interpretive authority. 18 Finally, creating an audit trail and facilitating reflection on coding decisions via memoing ensured transparent analytic logic. 18
Results
A total of 26 healthcare providers participated in focus groups (n = 22) or individual interviews (n = 4). All who volunteered to participate were eligible. See Supplemental Table 2 for the participant demographics. We were able to achieve representation from most identified clinician groups through purposive convenience sampling. Seventeen participants were nurses, three were healthcare aides, two were physicians, two were respiratory therapists, and two were physiotherapists. Nineteen of the participants identified themselves as female (not shown in Supplemental Table 2 to support anonymity). The median age of the participants was 40 years (interquartile range [IQR] 34 to 51). The median years of experience in the cardiac surgery intensive care unit was 8 (IQR 3 to 13).
Three themes and 13 categories were created from the data. Incongruent Operationalization and Conceptualization of Early Mobility was the first theme, supported by categories grouped at the micro (clinician), meso (unit), and macro (hospital) levels. These incongruencies lead to the next theme, Uncertainty. This theme was supported by the categories Getting Hurt and Following the Protocol. The final theme of Inconsistent Mobilization Practices is supported by the categories It Depends… and Rationale. These themes and categories are described below and visualized in Figure 1.
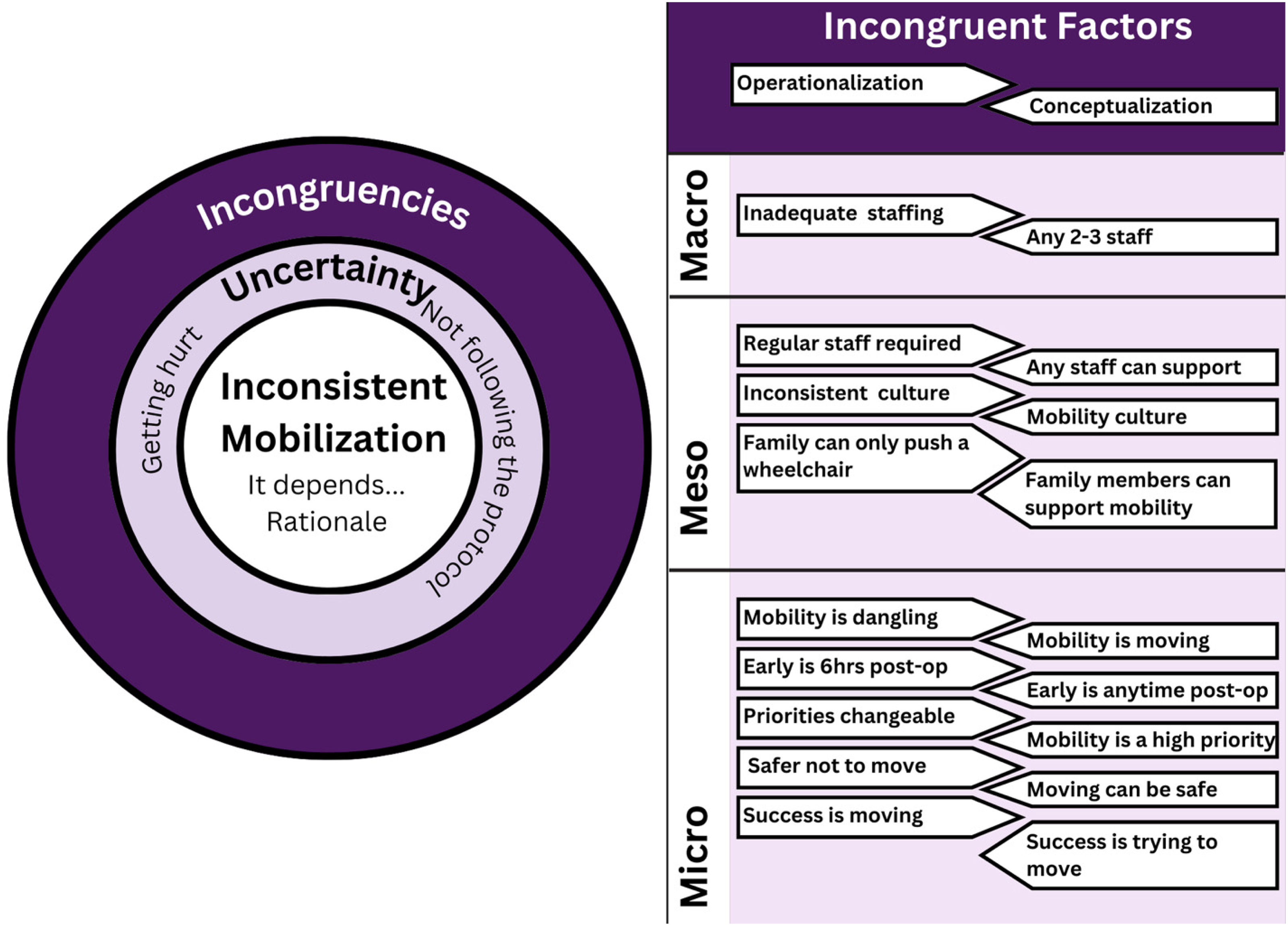
A visual overview of the themes and categories leading to inconsistent mobility.
Four barriers and four facilitators to early mobility were identified. These are organized as patient care-related (n = 2 barriers, n = 1 facilitator) and clinician-specific (n = 2 barriers, n = 3 facilitators). The barriers and facilitators generated are shared in Supplemental Table 6.
Incongruent operationalization and conceptualization
How clinicians described the operationalization (“doing”) of early mobility did not align with their conceptualization (“thinking”) of early mobility. This was noted through the incongruences in definitions provided compared to how these were described when discussing a successful mobilization or barriers and facilitators to early mobility. This theme is broken down into the micro (clinician), meso (unit), and macro (hospital) levels. This theme is summarized below with details and participant quotations available in Supplemental Table 3.
There are five incongruencies at the micro level: mobility definition, timing, priority, safety, and defining success. For example, the operationalization of mobility as sitting at the side of the bed is incongruent with how mobility is conceptualized, as a wide range of movements. As well, six hours post-operative was a consistent time for operationalization of “early”, however, this time does not align with the conceptualization of early. At the meso level, three incongruences exist: who can mobilize, culture, and family support. For example, when operationalizing early mobility, clinicians considered the presence of cardiac surgery intensive care unit-specific staff as a requirement; yet, this was not the case when early mobility was conceptualized. At the macro level, the incongruency relates to staffing. Early mobility is not operationalized even when there are two clinicians (one-eighth to one-seventh of the clinicians on each shift), which is the conceptualized requirement for early mobility.
Uncertainty
Clinicians are uncertain how to mobilize patients due to the incongruencies between the conceptualization and operationalization of early mobility. This theme comprised two categories: getting hurt and not following the protocol. This theme is summarized below with details and participant quotations available in Supplemental Table 4.
Clinicians described immobility as the preference to reduce the risk of getting hurt. Not engaging the family more actively than pushing a wheelchair was shared as a way to keep them and the patient safe. Clinicians also described being uncertain of whether to mobilize a patient before the six-hour mark when the patient wished to move.
Inconsistent mobilization practices
Uncertainty ultimately leads to the theme of Inconsistent Mobilization Practices, which comprises two categories: it depends… and rationales. The categories are summarized below with details and participant quotations available in Supplemental Table 5. In it depends…, clinicians argued with themselves about engaging in early mobility as their incongruent conceptualizations, operationalizations, and uncertainties came together. They either talked themselves into mobilizing or out of mobilizing based on where the internal argument ended. Mobility depends on the arguments clinicians use to justify their early mobility practices. The uncertainty can also lead to a lack of action by clinicians as they make rationales for not mobilizing their patients. In this way, clinicians provide a rationale for their inconsistent mobility practices.
Barriers and facilitators to early mobility
Four barriers to early mobility were identified, along with four facilitators. These are in addition to the themes and categories already described. The barriers and facilitators are grouped as patient care-related and clinician-related to support their future use in changing behaviours and supporting early mobility practices. See Supplemental Table 6 for an overview of the barriers and facilitators with details and participant quotations.
Two barriers were grouped as patient care-related: the number and location of intravenous lines and continuous renal replacement therapy. The facilitator grouped as patient care-related were the adjunct patient care elements of massage, pain control, deep breathing and coughing. These were thought to facilitate early mobility by helping patients move better. The participants believed that massage would relax tense muscles and improve blood flow which would help mobility. Participants were clear that they did not think deep breathing and coughing was early mobility but that it was important for patients to be able to breathe to be able to move.
Two barriers were grouped as clinician-related: lack of knowledge of the bed bike and prioritizing discharge education over mobilizing patients. A discussion about prioritization is shared in Supplemental Table 6 and highlights how patients who require the most support for mobility are prioritized behind patients who are leaving the cardiac surgery intensive care unit due to the focus on patient flow out of the intensive care unit. This leads to patients not being mobilized and remaining in the intensive care unit for longer periods of time. Three facilitators were grouped as clinician-related: experience as a healthcare provider, familiarity with the team, and conceptualization of mobility as key to recovery and discharge. Experience was not tied to years working in the intensive care unit but rather working in healthcare in general. Participants tied experience to recognizing the deconditioning that can happen to patients and to how quickly a healthcare provider will begin to mobilize a patient. Reframing early mobility from a safety issue that requires mitigation to conceptualizing it as key in recovery and discharge supports early mobility practices. Participants believe this facilitates prioritizing early mobility. This reframing of early mobility was shared by one participant as impactful and something that will change their healthcare practices.
Discussion
This interpretive description study explored perspectives of early mobility and barriers and facilitators to early mobility using focus groups and individual interviews. An incongruent operationalization (“doing” of early mobility) and conceptualization (“thinking” of early mobility) of micro, meso, and macro-level elements were found to lead to uncertainty, which culminates in inconsistent mobilization practices. Four additional barriers and four additional facilitators of early mobility were identified. These are important additional considerations when seeking to change clinician behaviours related to early mobility. They are groups as patient care-related and clinician-related to support their use.
These findings present a novel understanding of clinicians’ perspectives of early mobility and present unique barriers and facilitators to early mobility. These findings may help integrate early mobility into cardiac surgery intensive care unit clinical care by advancing our collective insight into the underlying reasons for non-uptake. The perspectives, barriers, and facilitators are multifaceted and diverse, suggesting that interventions to support the integration of early mobility into clinical practice also need to be multifaceted and diverse. This aligns with the complexity of behaviour change and the process of intervention mapping.16,17 Interventions can be co-developed to target specific incongruencies or barriers and, together, they may lead to behaviour change. A combination of education, mobility champions, and protocols may be required to support overcoming identified incongruencies and barriers and leveraging facilitators. Together, these components would provide clarity and consistent conceptualizations of early mobility and would provide guidance to support aligned and consistent operationalization of early mobility.
Providing education may support consistent conceptualizations of early mobility. For example, education about the risks of bedrest could be shared to overcome the safety concerns and the preference to keep patients immobile to keep them safe. Studies such as the revisited Dallas Bed Rest Study could be shared to demonstrate the impacts of prolonged bed rest on healthy individuals. 23 Discussions about the effects of bed rest on the population of patients undergoing cardiac surgery could occur to generate an understanding that bed rest must be perceived as unsafe and incongruent with healing and maintaining a good quality of life after cardiac surgery. Education for cardiac surgery intensive care unit clinicians about early mobility, the benefits, and the outcomes of implemented early mobility programs has been shown to build a culture of mobility and sustain successful implementation.10,24 This education must occur for new staff and existing staff.10,24 Educating patients and families preoperatively about the importance of early mobility adds to the culture of mobility.10,24 One team took this further and provided families with three tangible ways to actively participate in early mobility with their loved one while in the cardiac surgery intensive care unit. 24 Families were involved in reinforcing the importance of early mobility, motivating patients through coaching or cheerleading, and providing support and encouragement for mobility. 24 Education could also support the prioritization of mobility. Sharing that in one study, the first mobilization took a median of 10 min and an average of 20 min 25 may help cardiac surgery intensive care unit clinicians plan for early mobility throughout their shifts. Early mobility must be reframed to be equally regarded as important as titrating medications, diagnostic tests, and other “medical” care to support its prioritization by cardiac surgery intensive care unit clinicians.
More than education is required to overcome the incongruencies and barriers identified. Engaging an interdisciplinary team to champion early mobility within a cardiac surgery intensive care unit has been found to support behaviour change and improve mobility practices in other studies.10,24 These champions support the development and integration of early mobility intervention elements in the cardiac surgery intensive care unit and can help communicate outcomes to their fellow healthcare providers.10,24 Communicating outcomes, such as the number of patients eligible for early mobility who were mobilized according to the intervention, is important to successful behaviour change as it supports culture change. 10 When a behaviour is expected and this expectation is communicated and reinforced, it becomes part of the cultural routine of the cardiac surgery intensive care unit. 24
Operationalization and barriers identified could be removed through the creation of early mobility protocols that provide specific definitions and criteria to support clarity. For example, protocols that clearly define early mobility and outline a time to begin mobility that focuses on patient factors rather than time from extubation could help overcome the incongruencies of mobility definition, time, and defining success. One quality improvement project focused on increasing early mobility in the cardiac surgery intensive care unit shared a clear goal of early mobility: to perform safe, progressive mobility based on a patient assessment and to ambulate patients at least three times each day. 24 This definition includes a mode of mobility but also centres the cardiac surgery intensive care unit team around a shared goal to support operationalization. Teams can identify when mobility is successful by measuring when they have achieved the goal of early mobility. Adding patient-specific inclusion and exclusion criteria for when to start mobility is common 26 and allows cardiac surgery intensive care unit staff to use their critical thinking and judgment in applying an early mobility protocol, tailoring it to the unique patient context. This would also overcome many of the patient-specific barriers identified in this study. Concerns related to the safety of early mobility could be addressed through protocol elements such as monitoring suggestions, possible adverse events and potential solutions, and criteria for ceasing mobilization would help explicitly guide staff and remove interpersonal differences. One study found that these elements were only included in just over half of the early mobility protocols studied. 26
This work has several strengths and limitations. One strength is the inclusion of a variety of cardiac surgery intensive care unit clinicians. This reflects the population of clinicians who support early mobility in this cardiac surgery intensive care unit. 2 Another strength is the diversity of the research team. As described above, the team has diverse research, lived, and clinical expertise which supports a deep, rich, and multi-faceted analysis. Finally, a limitation is that this study was conducted at one site within one intensive care unit. This limits the transferability and generalization of the findings. Other sites would need to confirm and likely revise the findings before applying them to their institutions.
In conclusion, the incongruence between the operationalization and conceptualization of early mobility leads to uncertainty and inconsistent mobility practices. The successful integration of early mobility into cardiac surgery intensive care unit clinical care may require protocols and education to address incongruencies and barriers. In this way, incorporating the perspectives of clinicians into early mobility protocol development is essential to support behaviour change and provide this beneficial care.
Supplemental Material
sj-docx-1-cre-10.1177_02692155251405747 - Supplemental material for Perspectives of early mobility in the cardiac surgery intensive care unit: an interpretive description study
Supplemental material, sj-docx-1-cre-10.1177_02692155251405747 for Perspectives of early mobility in the cardiac surgery intensive care unit: an interpretive description study by Emily K. Phillips, Amy Abegglen, Jacqueline L. Hay, April Gregora, Kathy Smith, Carly Shaski, Gavin Bozek, Sarah Gilchrist, Andrew Fagan, Anna M. Chudyk, Maureen C. Ashe, Annette S. H. Schultz, Sheila O'Keefe-McCarthy, Rakesh C. Arora and Todd A. Duhamel in Clinical Rehabilitation
Footnotes
ORCID iDs
Ethical statement
Approvals were obtained from both the University of Manitoba Research Ethics Board (HE2024-0036) and the St Boniface Hospital Research Review Committee (RRC/2024/2159) prior to the commencement of the study. All participants reviewed and signed a consent form prior to participating in a focus group. Participants were made aware that consent was an ongoing process, and they could withdraw consent until data analysis began, at which point it was no longer possible to remove their data from the study. No person withdrew their consent.
Authors’ contributions
EKP conceptualized the study, collected, analyzed, and interpreted data and drafted the manuscript. AA collected, analyzed, and interpreted data and drafted a section of the manuscript. JLH analyzed and interpreted data, supported manuscript revision, and created the figure. AG, KS, CS, GB, SG, AF, AMC, MCA, ASHS, SOM, RCA, and TAD supported data analysis and revisions of the manuscript and figure. All authors read and approved the final manuscript.
Funding
Emily Phillips's doctoral studies are supported by a Canadian Institutes of Health Research Vanier Canada Graduate Scholarship, grant number 186981. Jacqueline Hay's is supported by a GROWW (Guiding interdisciplinary Research On Women's and girls’ health and Wellbeing). Maureen Ashe is supported by the Canada Research Chairs program. Anna Chudyk is supported by Canadian Institutes of Health Research Patient-Oriented Research Awards - Transition to Leadership Stream - Phase 2 award (reference number 188352). This work was supported by a Canadian Institutes of Health Research grant SCT-191275.
Conflicts of interest
The authors report no conflicts of interest.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
