Abstract
Objective
To develop and test a novel programme to support post-stroke fatigue management for testing in a future clinical trial.
Design
Staged approach to development and preliminary testing. Phases 1 and 2: co-design groups held to agree programme content and resources. Phase 3: training for facilitators developed and delivered. Phase 4: feasibility of programme delivery tested with questionnaires completed at beginning and end, and feedback interviews conducted.
Setting
Community, online.
Participants
Stroke survivors with post-stroke fatigue, carers and healthcare professionals.
Intervention
Post-stroke fatigue management programme supported by ‘buddies’ with lived or professional experience of managing fatigue.
Main measures
In phase 4, we examined feasibility of programme delivery (recruitment, retention, engagement and acceptability) and preliminary signals of efficacy.
Results
In phases 1 and 2 we recruited 23 participants (16 stroke survivors, 2 carers and 5 healthcare professionals) and designed resources. In phase 3, 10 people supported training development: 7 received training to be buddies. In phase 4 we recruited 15 people with fatigue; 13 completed the programme. Of these, 13 completed baseline and 12 follow-up questionnaires. All buddies (7/7) and 12/13 participants were interviewed. Mean number of sessions delivered was 9 (SD 3.24; range 3–13) and ranged from 10 to 60 minutes (mean 28 minutes; SD 10.04). Overall, mean fatigue severity reduced, activity increased, and mood measures improved. Participants and buddies were positive about the programme and believed it was worthwhile.
Conclusions
The programme was acceptable and feasible, with preliminary evidence of efficacy, but some issues need to be addressed in future trial design.
Introduction
There are 1.3 million stroke survivors in the United Kingdom, 1 and it is estimated that post-stroke fatigue affects 50% of stroke survivors.2,3 A recent international consensus group 4 has defined post-stroke fatigue as ‘A feeling of lack of energy, weariness, or exhaustion that can be overwhelming, and which involves physical, emotional, cognitive, perceptual aspects and is not relieved by rest and affects a person's daily life’. Post-stroke fatigue is linked to poor outcomes such as disability, 5 increased mortality, 6 places significant burden on caregivers 7 and profoundly affects everyday life. 2 It is therefore a national 8 and international stroke research priority. 9
Current clinical practice guidelines rely on low levels of evidence, such as expert consensus, to generate recommendations for management of fatigue. Although systematic reviews2,3,10 have demonstrated that some interventions show promise, the overall evidence base is weak. In addition, researchers have often tested a single approach, such as exercise, when research11,12 indicates that a much more comprehensive, multi-faceted approach is required.
Researchers have also previously failed to actively engage those with lived experience of post-stroke fatigue who already use a variety of successful strategies day-to-day to cope with manging their fatigue.12,13 Our previous research has shown that those with post-stroke fatigue indicated they would value contact with others with lived experience of managing fatigue and felt this would be important in helping them to cope. 12 Moreover, the concept of providing peer support in stroke rehabilitation is gaining interest and several reviews 14 and studies15–17 have reported this could be important.
The overall aim of this research is to design and test a clinical and cost-effective post-stroke fatigue management programme. However, we first needed to develop and refine the intervention and test this. Therefore, the aim of the Nottingham Fatigue after Stroke study (NotFAST3) was to co-design and test the delivery of the materials required to support a post-stroke fatigue self-management intervention in a future definitive trial.
Methods
Ethical approval was granted by the Nottingham University Research Ethics Committee for phases 1, 2 and 3 (June 2023; REF 294-0523) and for phase 4 (January 2024; REF 77-1123). All participants in each phase were sent a Participant Information Sheet (tailored to the appropriate phase) electronically and given at least 24 hours to decide whether to participate. All who participated completed a consent form for the relevant phase.
The study comprised four separate phases. We used the TIDieR checklist 18 to support reporting.
The aim of phases 1 and 2, was to recruit people to participate in co-design groups to agree the content of the self-management intervention (phase 1) and to develop supporting resources (phase 2). Our recruitment inclusion criteria for phases 1 and 2 were (i) participants with lived experience of post-stroke fatigue and (ii) people with expertise in managing post-stroke fatigue, such as partners of stroke survivors who provided informal support and (iii) professionals with expertise in post-stroke fatigue. We wanted to include people with communication difficulties, from different socio-economic and ethnic backgrounds. We recruited participants via social media and aimed to recruit 20 participants to discuss content, presentation, duration, timing and staffing of the programme.
We built on our previous findings 12 where people wanted a ‘pick and mix’ programme (term suggested by participants) where they could try out several fatigue management strategies and use some in combination. Using this research as a foundation, we held co-design groups to refine the components of the programme, and the resources required (web-based information resources and a workbook).
Using co-design methods, we sought to develop a fatigue management programme that could be tailored to the unique needs of stroke survivors with fatigue. Our co-design approach was based on an Experience-Based Co-Design (EBCD) methodology and participatory methods utilising the findings and narratives of people living with fatigue to trigger ideas and solutions and inform individual and joint priority setting. 19 Key issues, informed by recent, wide-ranging reviews of the relevant literature3,10 were summarised and discussed. This included the fact that evidence was not robust for any strategies but that there were some potentially promising results around pacing (i.e. spreading out activities during a day and having rest periods throughout), keeping a diary to understand fatigue, and taking exercise. The Health E-Learning and Media (HELM team at the University of Nottingham who have specific expertise in health informatics) guided the development of the web-based information resources. The co-design groups were held online (using MS Teams) and, a researcher was available to trouble shoot technical problems.
In phase 3 the aim was to build on the previous co-design phases of this study and previous studies11,12,13 and develop online training for peer supporters. The training, designed to be delivered online (using MS Teams), was developed by gathering examples about similar training, accessing literature (e.g. the SUPERB materials 17 ) and with direct input from our Patient and Public Involvement (PPI) group, our co-design groups and from participants in previous studies. Our inclusion criteria for phase 3 were the same as for phases 1 and 2. Peer supporters were recruited by contacting stroke survivors and carers who had been involved in previous studies 11 and who had consented to be contacted in the future about further studies. We also recruited via stroke groups.
A series of online meetings were held (via MS Teams) that included discussions to explore and decide on the content of the training. We aimed to have input from 10 participants, to have a spread of responses, and drew up suggestions of priority areas, for example, establishing boundaries and safeguarding. Following the suggestion of PPI group members and participants, peer supporters were called ‘buddies’.
In our final phase, phase 4, we conducted a feasibility study to test the delivery of the programme designed in phases 1, 2 and 3. Specifically, our aim was to determine: recruitment and refusal rates; retention and engagement; acceptability of the intervention (including the resources and use of buddies); and any preliminary efficacy signal on the Fatigue Severity Scale (as advised by the international consensus paper 4 ).
Our phase 4 recruitment target was 20 participants with post-stroke fatigue. Given little consensus in the literature on the optimal sample size for a feasibility study, we considered (a) the numbers of people with fatigue and (b) our recent studies on post-stroke fatigue which recruited to target, 11 and that this sample size would be appropriate for a feasibility study. 20 This number was also because we were not randomising nor testing trial parameters or outcome measures because we already know these from our previous research.
Participants were recruited from the community via stroke clubs and via social media. Participants were eligible to participate if they were aged 18 years or over, had current fatigue, and had had their stroke more than 6 months prior, and not more than 5 years before. This was chosen because, before 6 months, people could still be actively involved in rehabilitation and after 5 years other factors might come into play. We actively recruited participants with aphasia and provided materials and support to facilitate their participation (based on experiences from previous research 12 ).
All those who expressed an interest were asked to confirm eligibility criteria. Those who consented were sent electronic links to the digital resources and the workbook was posted to them. The topics agreed through co-design stages and covered in the resources are listed in Table 1. Participants were asked to meet with their ‘buddy’, who had received the training developed in phase 3, via video-call twice a week with each meeting lasting 15 to 20 minutes. At each meeting, the ‘buddy’ provided peer support and encouraged the participant to make use of the resources and to try different strategies to self-manage their fatigue.
Topics covered in the feasibility study participant interviews.

At the end of each session, the ‘buddy’ was asked to complete an electronic form recording the length of the meeting, a tick box of strategies discussed and any key issues that arose (e.g. safeguarding concerns or technical problems). The forms were designed to be completed in under 5 minutes and were screened by the study co-ordinator regularly. Any issues of concern raised by the buddy would trigger a contact with the study co-ordinator.
Outcome measures
Participants were asked to complete a questionnaire electronically at baseline and on completion of the programme. The questionnaire included measures to assess current activity and fatigue levels. The length of the questionnaire and the time it would take to complete were key considerations in selecting the tools. Our PPI group assessed the final version to ensure it was not overly burdensome. Overall, the questionnaire was estimated to take around 20 to 30 minutes to complete and participants were encouraged to rest as needed when completing it.
Fatigue Severity Scale
21
(as recommended by international consensus
4
). Nottingham Extended ADL Scale
22
to determine activity. EQ5D-5L
23
measures five dimensions of health (mobility, self-care, usual activities, pain/discomfort and anxiety/depression). Patient Health Questionnaire 8
24
to measure depression. GAD-7
25
(General Anxiety Disorder-7) to measure anxiety. Fatigue and Energy Scales
26
to determine fatigue severity and energy levels.
At baseline, we also collected basic demographic data including date of birth, sex, date of stroke, and which side of the body was affected by stroke.
Interviews
On completion of the programme, all participants and ‘buddies’ were invited to participate in a one-to-one interview via video-call. The semi-structured interview included questions to explore perceptions of what worked well or less well in terms of the usability and usefulness of the resources and peer support. Questions about acceptability and useability were posed in the interviews rather than in the follow-up questionnaire to enable participants to share their experiences in their own words. We had considered using a user questionnaire to do this, but our PPI felt this data would be better collected in interviews.
The interview guide covered similar questions for participants (see Table 1) and buddies (see Table 2). Participants with aphasia were given extra time, breaks and accessible materials to facilitate their participation.
Topics covered in the buddy facilitator interviews.
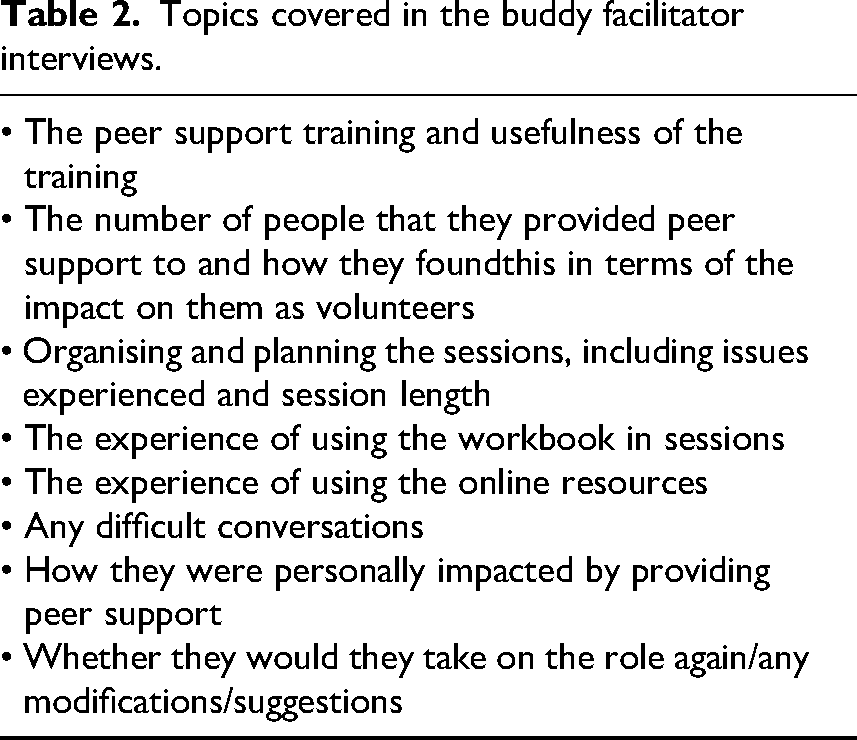
Interviews were conducted at a time agreed in advance with the interviewee. The interviews were audio-recorded, and the transcripts downloaded. These were then checked for accuracy and anonymised by a member of the research team.
Data analysis
Descriptive statistics were used to analyse the quantitative measures to test whether the study was feasible and identify any signals of efficacy in the data. The interview data were analysed using framework analysis. 27
To determine whether we could progress to a definitive trial, our a priori progression criteria were:
Recruitment of ≥50% of eligible participants; Retention: ≥ 80% of participants complete all questionnaires; Engagement: ≥ 80% of participants access ≥3 of the sessions available and Acceptability: ≥ 80% of participants regard/score the intervention as useful, relevant, and appropriate.
We used a feasibility traffic light system 28 : Red (stop) where A is not met; Amber (modify) where A is met AND either B OR C OR D reach at least 70%: and Green (proceed) where all criteria are met.
Results
Phases 1 and 2
A series of co-design groups were convened (August–October 2023) and 23 participants were recruited; 16 stroke survivors with post-stroke fatigue, 2 informal caregivers, and 5 healthcare professionals with expertise in fatigue management, 2 of whom also lived with fatigue as a part of a long-term health condition.
The co-design groups designed two web-based digital resources and a workbook. The resources agreed by the groups included information on how fatigue may impact a person, what it can feel like, and potential strategies for self-management. The resources included ‘real world’ anecdotes and analogies that others had experienced. All resources were designed to be informative and visual. We embedded self-management principles into the design and delivery of the intervention such as exploring problem solving and setting goals. We wanted to build on confidence (individual self-efficacy) as well as confidence in other forms of support (collective efficacy). The content and priorities were informed and enriched by the shared lived experiences of the group. Several participants volunteered to contribute to the writing and production of resources on ‘Understanding fatigue after stroke’ and ‘Managing fatigue after stroke’ by providing personal tips and recording short audio-visual messages about their experiences of fatigue. They also provided personal quotations to illustrate the workbook content. The web-based resources included an audio option so that people could choose to watch, listen or both as they accessed them. See Table 3 for topics which the co-design groups agreed should be covered.
Topics agreed for inclusion in the online and workbook resources.

Phase 3
It was agreed that the training should be delivered in under 90 minutes, with breaks, by two facilitators (healthcare professional and stroke survivor with post-stroke fatigue). The areas agreed to be included were: (i) Introduction to peer support, stressing active listening, supporting not advising. (ii) Managing safeguarding, boundaries and confidentiality. (iii) Handling difficult conversations and what to do if someone is distressed. (iv) Understanding biases and prejudices. (v) Responsibilities of a buddy, such as time commitment, being mindful of safety. (vi) Potential benefits of being a buddy. (vii) Wider team support available for buddies.
Following this, the training of buddies occurred in February and March 2024. Ten people supported the development of the training and seven people (four women and three men) received the online training. Of these, four had lived experience of managing post-stroke fatigue, while three had experience of supporting people with it either as professionals or carers.
Phase 4
The feasibility study occurred between 1 April and 30 June 2024. Although our aim was to recruit 20 people, the time taken to recruit, train and allocate buddies, manage technical set-up issues and deliver the programme meant we had a shorter recruitment window and could only recruit 15 people in the time allocated.
We received 34 expressions of interest and sent out Participant Information Sheets. Of these, seven did not meet eligibility criteria, and eight did not respond again despite follow up. Of the remaining 19, 15 completed baselines, 15 started the programme, 13 completed it and all 13 were invited to complete follow up questionnaires and interviews. All seven people who received the training in phase 3 acted as buddies. We were not able to collect any data on reasons why people did not complete follow ups.
Participants included 7 men and 8 women with a mean age of 59 (SD 9.59; range 39–81). We had three people with aphasia. Participants were from a wide geographical location; the Midlands (4), Southeast England (4), Wales (4) and East of England (4).
We planned to hold the sessions over 6 weeks but had to be flexible as the study was over the summer holidays. The mean number of sessions delivered was 9 (SD 3.24; range 1–13). A total 107 sessions were delivered. Sessions ranged in length from 10 to 60 minutes with a mean of 28 minutes (SD 10.04). We had 14 reports of technical problems. No safeguarding issues were raised.
In terms of the content of sessions, participants met with their buddy to discuss the resources available, progress at trying out suggestions in the workbook and in the online training, and any more general issues around fatigue and its management. The most popular issue discussed was rest and relaxation (67 sessions) followed by sleep (55), what fatigue feels like (54), keeping a diary (53), exercise (51), identifying triggers (50), family and friends (42), pacing (32) and nutrition (28).
The results of the baseline and final questionnaires are reported in Table 4. Data were collected at baseline (
Baseline and final questionnaire results.
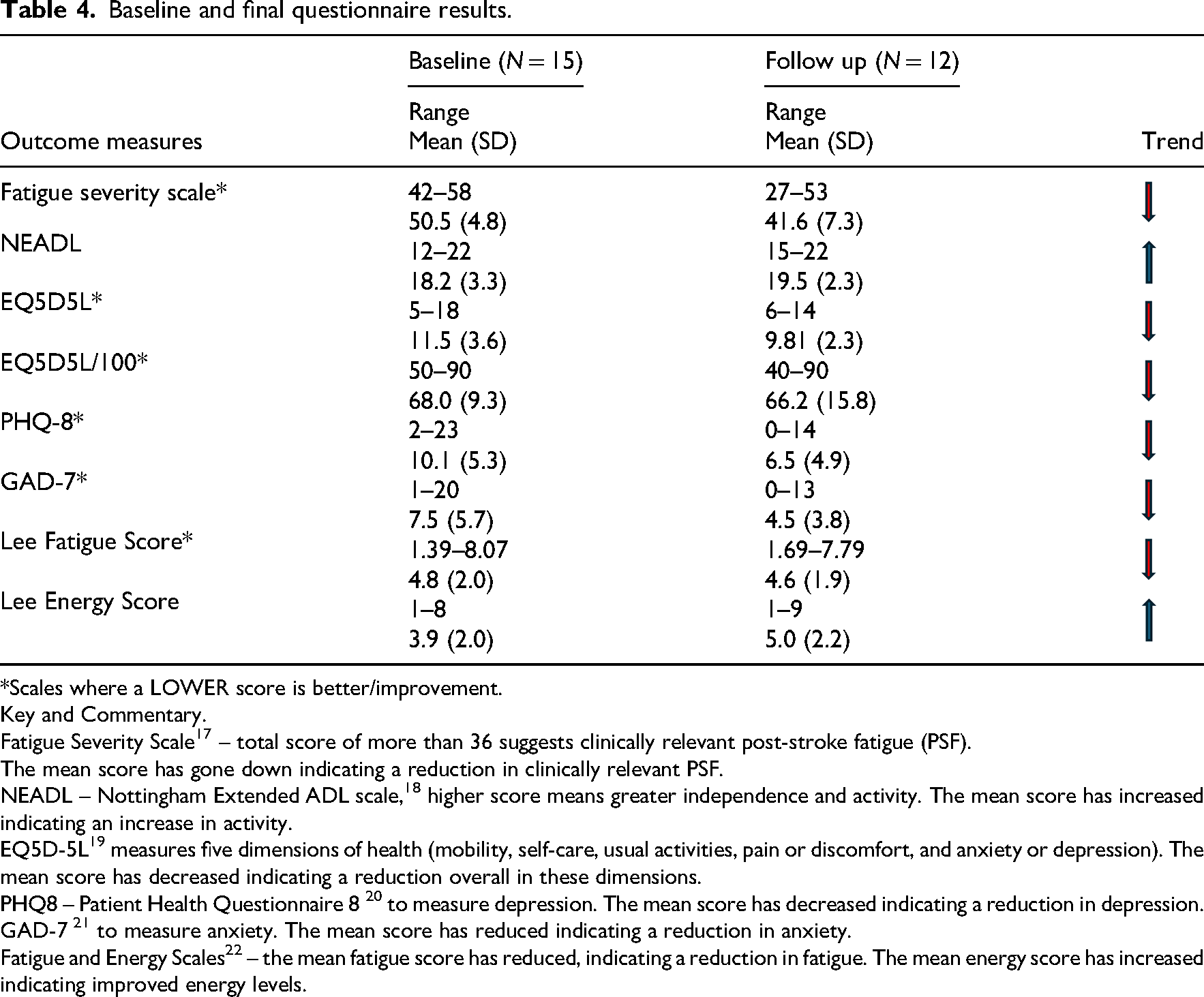
*Scales where a LOWER score is better/improvement.
Key and Commentary.
Fatigue Severity Scale 17 – total score of more than 36 suggests clinically relevant post-stroke fatigue (PSF).
The mean score has gone down indicating a reduction in clinically relevant PSF.
NEADL – Nottingham Extended ADL scale, 18 higher score means greater independence and activity. The mean score has increased indicating an increase in activity.
EQ5D-5L 19 measures five dimensions of health (mobility, self-care, usual activities, pain or discomfort, and anxiety or depression). The mean score has decreased indicating a reduction overall in these dimensions.
PHQ8 – Patient Health Questionnaire 8 20 to measure depression. The mean score has decreased indicating a reduction in depression.
GAD-7 21 to measure anxiety. The mean score has reduced indicating a reduction in anxiety.
Fatigue and Energy Scales 22 – the mean fatigue score has reduced, indicating a reduction in fatigue. The mean energy score has increased indicating improved energy levels.
Summary of interview findings
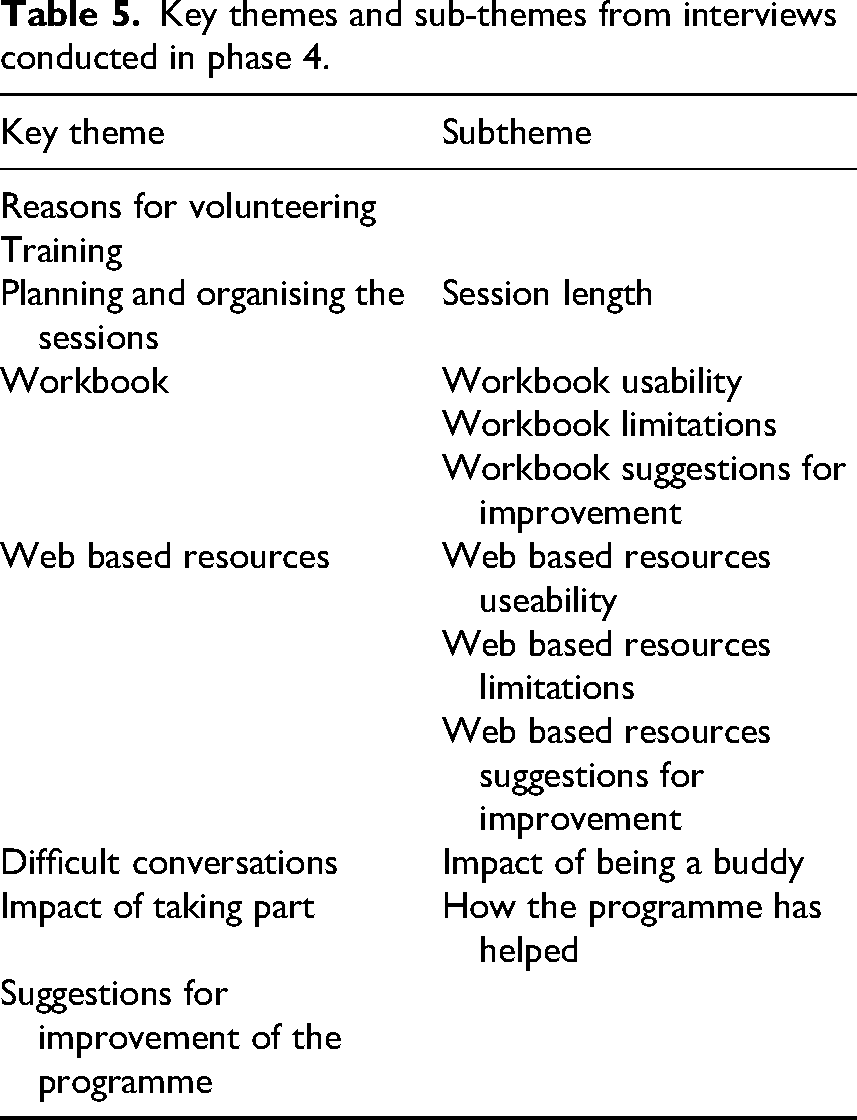
We interviewed 7/7 buddies and 12/13 participants. The key themes identified in the analysis are shown in Table 5, Supplemental materials 1 and 2 show a fuller selection of the illustrative quotations from participants and buddies in support the themes.
Key themes and sub-themes from interviews conducted in phase 4.
Overall, we found that the interview data showed clear support for the programme from both participants and buddies. The buddies volunteered because of a wish to help others and because they understood the issues facing those with post-stroke fatigue. ‘Because I know what it's like to have fatigue and well, basically nobody to talk to about it…Yeah, that was the main thing. I know how hard it is’. B4 ‘I thought it might be an interesting and potentially rewarding experience because I could share my insights of fatigue and maybe sort of share some of the coping strategies that I use and as it turned out rightly or wrongly, I got the impression that what I could offer was quite helpful’. B2 ‘It was useful having someone who's been through this process and who understood where I was coming from. I thought that worked very well’. (306) ‘But do you know the biggest tick for me? Is actually having somebody to talk to regularly about it. Who is, you know, completely giving me half an hour of their time, which I'm so grateful for…So for me, a real plus was having somebody who took an interest in me, actually asked me about things. It gave me confidence to then tackle things that I've been putting to one side… I can't tell you how grateful I am to [name] for having, you know, an hour a week to talk to somebody about the issues’. (304) ‘It was the practical ways to manage …I found extremely useful …like the fatigue diary and like trying to work out what your triggers are, the analogy of the battery I really associated with’. (308) ‘And for me personally, it's been part of like that journey of acceptance as well’. (311) ‘There were one or two little tips that I've picked up from it…I think really you know the fact of not pushing myself too much, you know to sort of do a bit and have a rest and do a bit and have a rest, which is something that I wasn't really doing’. (301) ‘I found because I'm working full time, I found that the flexibility of my buddy slash mentor was great so that we could fit in a couple of sessions a week or one session a week depending on our availability. But she was very flexible so that was extremely helpful’. (308) ‘I would say that twice a week is probably too frequent’. B2 ‘…that workbook's brilliant’. B4 ‘You can dip in and out of it, so it's kind of flexible, which I liked’. (302) ‘I could show it to my husband and my daughters for there's like, the section for the family’ [about fatigue being a symptom of stroke]. (311)
Was the trial feasible?
Using our priori progression criteria, and with regard to recruitment, we recruited ≥50% of eligible participants. However, this was with caveats that our recruitment window was small, and our data were not comprehensive. We had 34 expressions of interest and, as previously noted, of these seven were not eligible, but eight then did not respond – and we do not know if they were eligible or not. Of 19 who were eligible we recruited 15 into the study (79%). Given that we were not using NHS clinics and networks, this was potentially promising. However, we do not have details on those who decided not to participate from the very beginning. Therefore, this is Amber rating given these limitations.
We did achieve our retention target. All participants (
We also achieved green for engagement and acceptability. Participants who attended more than one meeting (13/15 = 87%) stayed in the study and completed the sessions (attending a mean of 9 sessions). The findings from interviews demonstrated a very high level on acceptability and, although suggestions were made for improvements, these were minor. Overall, in terms of our stop-go criteria, we believe we achieved a Green-albeit noting above caveats.
We also wanted to determine whether there were any efficacy signals in the data. We are mindful that the sample is small, there was no control group, and that interpretation must be very cautious. However overall mean fatigue severity reduced in the two fatigue scales, activity increased, and mood measures improved, although we are also mindful that three individuals did not return the final questionnaire.
Discussion
Given the prevalence of post-stroke fatigue 1 and the current limitations in the evidence base for management, 10 this study is timely. Although the feasibility study was small, it showed potential for testing the programme with peer support and personalised resources in a larger trial. Comments were positive from both those providing the peer support and from those receiving it. The signal on efficacy, though small, was also encouraging as the mean scores in all the relevant domains improved. Of note, the study also showed that not all strategies are valued by everyone and confirmed our previous research that not all strategies were helpful for each person 12 : a ‘pick and mix’ approach is essential.
Other researchers have also recently demonstrated positive outcomes for the use of peer supporters. Fu et al. 14 in their review of supporting meaningful outcomes in stroke rehabilitation have underlined the value of personal connections through the shared process of providing mutual encouragement and supporting social integration. Likewise, a review by Clark et al., 29 concluded that peer support facilitated sharing of experiences, vicarious learning, and was valuable in increasing motivation. The feasibility trial by Hilari et al. 17 , which was specifically designed to provide peer-support for stroke survivors with aphasia, reported similar benefits for participants to those found in our study.
Yet although the programme showed great promise and was very well received by all those involved, there were key issues that merit further attention and discussion. Technology was a crucial issue for some. For several participants, using links to online meetings was an initial challenge and caused set-up delays. Problems were overcome with support from the team, but this may mean a longer set-up is needed or that other access methods are required. We also had several people who struggled with the platform indicating that covering this prior to starting the programme would be appropriate. On a similar vein, fewer people used the online resources. On reflection, we may not have spent enough time educating and supporting participants on using these or referring to them in sessions, which may have increased their use.
The use of buddies was very well received by participants and these volunteers also seemed to value the experience. However, the team had to manage the flow of participants to ensure there were enough buddies available as needed; some buddies were delayed in responding to requests to ‘check in’ which meant further delays. Our buddies were volunteers, and it may be wise to consider employing buddies in future studies so that availability can be more carefully monitored and planned. Another issue which merits attention is the fact that having oversight of what was being shared between buddies and participants was almost impossible. We covered this carefully in the training, and tried to ensure that buddies did not give advice on, for example, medication or medical issues, but the reality was that we could only identify this from the checklist or email communication with buddies. We were not party to all the conversations, and this may need further consideration. We are also aware that some buddies and participants formed friendships and were still in contact at the end of the study. This has not been a problem in this study, but further thought may be wise around this wider issue to identify further safeguards and boundaries.
Recruitment was the issue of most concern. Unfortunately, our recruitment coincided with a holiday period, and this clearly effected uptake. From the onset it seemed unlikely we could recruit and deliver the programme to twenty people in the time available. We used snowballing methods to advertise the study and therefore cannot be certain about the actual numbers approached and the reasons why people chose not to participate. However, when participants had committed to the study, attrition was low. We think that if we extend recruitment to include those people attending follow up clinics who report fatigue, many of whom attend before the 6 months exclusion criteria set in this study, recruitment could be optimised further.
It is of note that a great deal of co-ordination and preparation went on ‘behind the scenes’ to keep the study afloat. The research team worked hard to keep people engaged, to support the buddies and participants, and to sort out technical and other issues and day to day queries. The work required to deliver such studies has also been stressed by Hilari et al. 17 and this team also noted the importance of perseverance of their co-ordinator – which was also important in this fatigue study.
Overall, the design and testing of the programme was successful. Participants liked the choice of developing personal self-management options for their fatigue and valued the opportunity to try out a range of strategies, from building in rest and sleep periods, to increasing exercise and keeping a diary while being supported by ‘buddies’, Given that the post-stroke fatigue international consensus group 4 have called for more research into this area, our results are very promising. Although the feasibility trial achieved a green rating, the study showed there are still important issues to consider in designing a future definitive trial
Clinical messages
- Post-stroke fatigue has a significant impact on daily living for stroke survivors and those who provide informal support.
- People with post-stroke fatigue valued talking and receiving support from someone with lived experience or expertise of managing it.
- Not all ‘fatigue’ strategies are useful for everyone, and approaches tailored to individual needs are required.
Supplemental Material
sj-pdf-1-cre-10.1177_02692155251350084 - Supplemental material for Co-designing and testing a management programme with peer support for post-stroke fatigue: Nottingham fatigue after stroke study (NotFAST3)
Supplemental material, sj-pdf-1-cre-10.1177_02692155251350084 for Co-designing and testing a management programme with peer support for post-stroke fatigue: Nottingham fatigue after stroke study (NotFAST3) by Joanne Ablewhite, Shirley Thomas, Roshan das Nair, Fiona Jones, Nikola Sprigg, Heather Wharrad and Avril Drummond in Clinical Rehabilitation
Footnotes
Acknowledgements
We thank many participants and peer supporters who took part in the study. We particularly thank our Patient and Public Involvement (PPI) group (ET, JD, BS, RM) and the UK NIHR for funding.
Ethical considerations
Ethical approval was granted by the Nottingham University Research Ethics Committee for phases 1, 2 and 3 (June 2023; REF 294-0523) and for phase 4 (January 2024; REF 77-1123). All those who participated provided informed consent for each relevant phase and signed a consent form.
Author contributions
AD and JA designed the study with contributions from all the authors who are all grant holders. AD is the principal investigator. JA was the study co-ordinator, was a key point of contact for all the team and participants and collected the data for phase 4. AD and JA wrote the first draft of the article. All authors contributed to the interpretation of the data, commented critically on the manuscript and read and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project is funded by the National Institute for Health and Care Research (NIHR) under its Research for Patient Benefit (RfPB) Programme (Grant Reference Number NIHR204311). However, please note that the views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data is available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
