Abstract
Introduction
Although morphological and anatomical studies indicate that venous wall weakening and subendothelial fibrosis characterize varicose veins (VV), the pathogenesis of VV remains poorly understood. The aim of this study is to obtain protein expression profiles in patients with VV and thereby get a step closer to understanding the pathogenesis of VV.
Methods
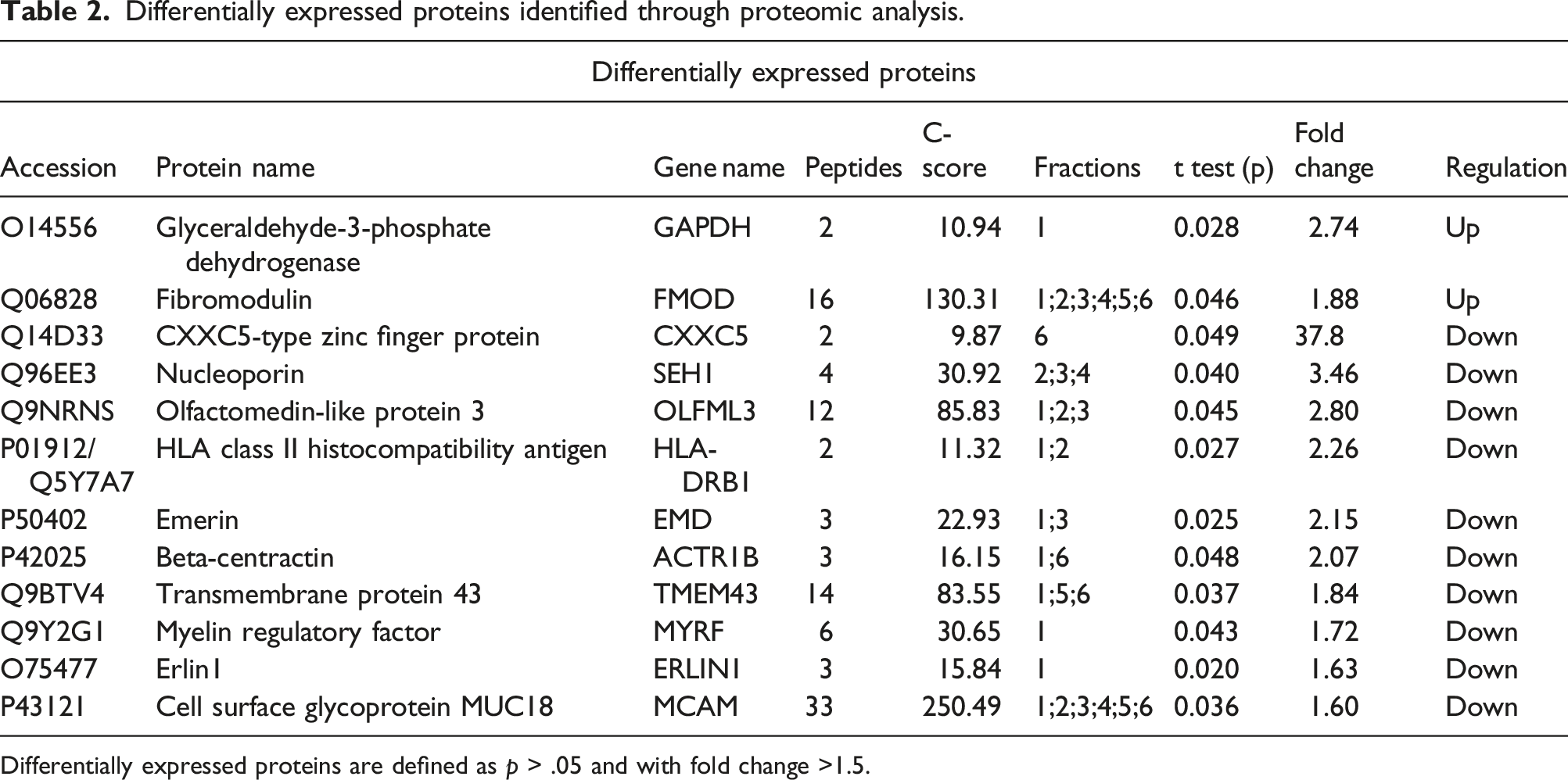
Specimens were obtained from total of 10 patients, that is, from 5 patients undergoing VV surgical stripping and from 5 non-VV patients undergoing bypass surgery. Specimens were collected from the same layers of venous wall. Proteins were extracted from each specimen and analyzed by ion mobility spectrometry (IMS-MS). In total, 1387 were identified and 486 proteins were identified in all samples. From these, 15 proteins were differentially expressed between VV and non-VV samples (p < .05) and 12 of these showed a fold change >1.5.
Results
Interestingly, among the differentially expressed proteins, only two proteins were significantly increased in the VV tissue, that is, GAPDH (p = .028, fold change 2.74), where several proteins involved in maintaining the homeostasis in the extracellular matrix, that is, the CXXC zinc finger protein 5 (CXXC5) and nucleoporin (SEH1) were prominently downregulated (p = .049, fold change 37.8, and p = .040, fold change 3.46). The downregulation in protein expression of CXXC5 and SEH1 as well as upregulation of GAPDH were validated by Western blotting.
Conclusion
The identified differentially expressed proteins suggest an altered profile of the connective tissue proteins as well as an increased proteolytic enzyme activity which both may be central in the pathophysiology of varicose veins.
Introduction
Chronic venous insufficiency (CVI) is one of the most common disorders of the venous system of the lower limbs and roughly 1%–2% of national healthcare resources in many countries are allocated to treatment of CVI.1,2 Most common causes of CVI are valvular incompetence and valve reflux of the deep veins, the superficial veins, or the perforating veins. These conditions are strongly associated with vein wall dilation, increase in hydrostatic pressure, and altered microcirculation. 3 If left untreated, CVI can lead to pain, edema, hyperpigmentation, lipodermatosclerosis, venous eczema, and leg ulcers among others. Varicose veins (VV) are a common manifestation of CVI in the lower extremities. 4 It affects approximately 15% and 25% of men and women, respectively. 5 Risk factors associated with varicose include family history, older age, female gender, obesity, pregnancy, and prolonged standing. 4
Various therapy approaches are available for treating VV, including endothermal ablation (using endovenous laser or radiofrequency ablation), ultrasound guided foam sclerotherapy, and conventional surgery (proximal ligation, stripping, and phlebectomies). Additionally, new methods are being adopted, particularly mechanochemical ablation and the use of cyanoacrylate glue.6,7 Despite the unquestionable clinical significance of the condition and the various treatment options, the exact etiology and pathophysiology of VV formation remain unclear. 8 One of the recent theories proposes molecular changes leading to the histologic changes in the vessel wall and extracellular matrix (ECM) composition, thus venous wall weakening and subendothelial fibrosis. 9 However, the field still awaits deep biological profiling to gain insight into the pathology of VV.
Proteomics is the study of the proteome and is able to achieve a map of the proteins expressed in tissues, cells, or fluids, including plasma. 10 A proteome refers to the total set of proteins expressed at a given time in each cell and includes the variety of modified proteins resulting from gene splicing and post-translational modifications.10–12 Proteins are involved in virtually every cellular function including regulatory mechanisms and those proteins modified in diseases may directly or indirectly affect the pathogenesis. 13 Such a proteomic analysis could provide a method for quantitative and qualitative mapping of the entire proteome, as well as assist to elucidate the molecular mechanisms involved in biological processes in VV. Thus, in this study, we have analyzed the proteomic profiles, that is, protein expression levels, in varicose great saphenous vein (VSV) specimens surgically removed during stripping at the groin level compared with the expressed proteins in segmental parts of normal great saphenous vein (NSV) specimens adjacent to varicose veins as control.
By identifying alterations in proteomic profiles of VSV from VV patients and NSV veins from non-VV patients, we aimed to get direct insight into the disease process, and hence by the identification of protein modifications enhance our understanding of the pathogenesis of VV.
Material and methods
Sample collection
This study is approved by the Central Denmark Region Committees on Health Research Ethics as well as the Danish Data Agency1–10 and was conducted in accordance with the Declaration of Helsinki. Varicose great saphenous vein (VSV) specimens (n = 5) were surgically removed during stripping at the groin level. Segmental parts of normal great saphenous vein (NSV) specimens adjacent to varicose veins as control were obtained from 5 patients, who underwent vascular bypass surgery. The segment was collected from each patient from greater saphenous vein 1 cm distally from saphenofemoral junction.
Clinical characteristic.

GSV, great saphenous vein; ns, not significant; NSV, normal great saphenous vein; VSV, varicose great saphenous vein.
All vein samples were immediately rinsed from blood and fat tissue, snap frozen in liquid nitrogen, and stored at −80°C for future analysis.
Sample preparation (protein extraction)
Samples (about 160 mg) were grinded in liquid nitrogen to a fine powder using a mortar and pestle. Tissue powder was transferred to microcentrifuge tubes and resuspended in lysis buffer containing 7 M urea, 2 M thiourea, 4% w/v CHAPS, and 100 mM DTT.
Filter-aided protein sample preparation (FASP) for mass spectrometry analysis
Samples were concentrated on Amicon Ultra-0.5 mL 30 kDa centrifugal filter unit and were denatured in 8 M Urea, 100 mM DTT solution with continuous rotation at 800 r/min in the temperature-controlled shaker for 3 h at 37°C.
Trypsin digestion was done according to a modified FASP protocol as described by Wisniewski et al. 14 Briefly, phage particles were washed with buffer containing 8 M urea. The proteins were alkylated using iodoacetamide. Buffer was exchanged by washing two times with 50 mM NH4HCO3 and proteins digested overnight with TPCK Trypsin 20233 (Thermo Scientific, USA). After overnight digestion, peptides were recovered by centrifugation and then two additional washes using 50% CH3CN were combined, acidified, lyophilized, redissolved in 0.1% formic acid, and then analyzed by mass spectrometry.
Liquid chromatography and mass spectrometry
Peptides were separated off-line on a 200 × 2.1 mm, 5 µm SCX column (300 µm, that is, 15 cm, packed with POROS 10S) using a gradient of 0–60% B over 20 min with a flow rate of 300 µL/min. Solvent A was 5 mM NaH2PO4, pH 3.0 in 5% acetonitrile. Solvent B was solvent A with the addition of 1 M NaCl. The separation was monitored at 214 nm and 2 min, six fractions were collected. The fractions were dried using a vacuum centrifuge and resuspended in 30 µL of 0.1% formic acid (FA).
Each of the SCX fractions was analyzed by nano-LC-MSE analysis. Peptides were loaded on reversed-phase trap column PST C18, 100 Å, 5 µm, 180 µm × 20 mm (Waters Corporation, UK) with a flowrate of 15 µl/min using loading buffer of 0.1% FA and subsequently separated on HSS-T3 C18 1.8 μm, 75 μm × 250 mm analytical column (Waters Corporation, UK) in 120 min linear gradient (A: 0.1% formic acid, B: 100% CH3CN and 0.1% FA) at a flow rate of 300 nL per min. The analytical column temperature was kept at 40°C.
The nano-LC was coupled online through a nanoESI 7 cm length, 10 mm tip emitter (New Objective, USA) with HDMS Synapt G2 mass spectrometer (Waters Corporation, UK). Data were acquired using Masslynx version 4.1 software (Waters Corporation, UK) in positive ion mode. LC-MS data were collected using data independent acquisition (DIA) mode MSE in combination with online ion mobility separation.
The trap collision energy of mass spectrometer was ramped from 18 to 40 eV for high-energy scans in MSE mode. The trap and transfer collision energy for high-energy scans in HDMS mode was ramped from 4 to 5 eV and from 27 to 50 eV. For both analyses, the mass range was set to 50–2000 Da with a scan time set to 0.9 s. A reference compound [Glu1]-Fibrinopeptide B (Waters Corporation, UK) was infused continuously (500 fmol/µl at flow rate 500 nL per min) and scanned every 30 s for online mass spectrometer calibration purpose. The samples were run in triplicate.
Data processing, searching, and analysis
Raw data files were processed and searched using Progenesis QI for proteomics (Nonlinear Dynamics, Waters). The following parameters were used to generate peak lists: (i) minimum intensity for precursors was set to 100 counts, (ii) minimum intensity for fragment ions was set to 30 counts, and (iii) intensity was set to 500 counts. Processed data was analyzed using trypsin as the cleavage protease, one missed cleavage was allowed, and fixed modification was set to carbamidomethylation of cysteines, variable modification was set to oxidation of methionine. Minimum identification criteria included 2 fragment ions per peptide, 5 fragment ions per protein, and minimum of 2 peptides per protein. The false discovery rate (FDR) for peptide and protein identification was determined based on the search of a reversed database, which was generated automatically, when global false discovery rate was set to 4%. UniprotKB/SwissProt human database (2013-10) was used.
Western blotting
For one-dimensional Western blotting (WB), protein concentrations of selected samples were determined using a noninterfering assay (NI Protein Assay, Geno Technology Inc., St Louis, Mo). Identical amounts of protein from each sample (5 or 10 µg total protein) were added to each lane of a 10%–20% and 4%–20% tris-glycine gel (Invitrogen, Carlsbad, Calif., USA). After electrophoresis, the proteins were transferred to nitrocellulose membranes. For immunodetection of the proteins, the nitrocellulose sheets were incubated overnight at 4°C in phosphate-buffered saline (PBS) (2.7 mM KCl, 1.8 mM KH2PO4, 10.1 mM Na2HPO4, 140 mM NaCl, pH 7.3) containing 0.05% Tween-20 and 5% skimmed milk. The membranes were washed five times in PBS with 0.05% Tween-20. Rabbit polyclonal antibody to nucleoporin (SEH1) 0.05 mg/mL (Abcam, Cambridge, UK) was diluted to 1:50 rabbit polyclonal antibody to CXXC zinc finger protein 5 (CXXC5) 1 µg/mL (Santa Cruz Biotechnology, Inc, Santa Cruz, Calif) was diluted 1:1000, and rabbit polyclonal antibody to glyceraldehyde-3-phosphate dehydrogenase (GAPDH) 0.2 mg/mL (Abcam, Cambridge, UK) was diluted 1:2500 The blots were incubated with antibodies for 1 h at room temperature. After five washes in PBS with Tween-20, the blots were incubated for another hour with 1:1000 diluted peroxidase-conjugated swine antirabbit IgG (Dako, Glostrup, Denmark). Finally, the membranes were washed five times in PBS with Tween-20 and developed with the enhanced chemiluminescence technique (Amersham Biosciences Inc.). Blots were quantified using Imaging Densitometer GS710 and Quantity One software (Bio-Rad, Switzerland).
Results
The study included 5 VV patients with one VSV sample each and 5 non-VV patients with a NSV sample each. The characteristics of the patients are given in Table 1. There were no significant differences between the two groups with respect to gender, age, severity of varicosity, smoking status, BMI-index, or ingestion of non-steroidal anti-inflammatory drugs.
By means of proteomic analysis, we were able to obtain and compare the protein profiles of the VSV and NSV specimens from the patients in the two groups. In total, the analysis identified 1387 proteins over the 10 specimens. Of these, 486 proteins were identified in both types of the venous samples and the expression level of each of the proteins were compared between the groups. Interestingly, 220 proteins demonstrated differences in protein expression (>1.5 fold), but only 12 of these proteins reached significance (p < .05) Among these differentially expressed proteins, 10 proteins were identified as significantly decreased in the VV patients, including CXXC5-type zinc finger protein (CXXC5), nucleoporin (SEH1), emerin, and beta-centractin (p = .049–.025, fold change 37,8-2,15), and only two proteins had increased expression, that is, glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and fibromodulin (p = .028, fold change = 2.74; p = .046, fold change = 1.88), Figure 1. Interestingly, the CXXC5 protein, a well-known receptor for vascular endothelial growth factor (VEGF), is the most prominent with a 38-fold less expression in the VSV tissue, Table 2. As validation, protein expression of both SEH1, CXXC5 and GAPDH was investigated by Western blotting showing a decrease of SEH1 and CXXC5 proteins and an increase in GAPDH (X fold) corresponding to the differential expression was found by the MS-based proteomic analysis, Figure 2. Functional interaction network of identified proteins. The protein expression of the 486 proteins identified in all samples were compared between samples from the VSV and NSV. The blue color indicates downregulation of a protein in the VV group compared to the non-VV group, and the red color indicates upregulation. Differentially expressed proteins identified through proteomic analysis. Differentially expressed proteins are defined as p > .05 and with fold change >1.5. Western blotting of SEH1, CXXC5, and GAPDH proteins. The protein expression of SEH1, CXXC5, and GAPDH proteins were investigated in 6 specimens. Up/downregulation correlates with the differential protein expression identified in the MS-analysis.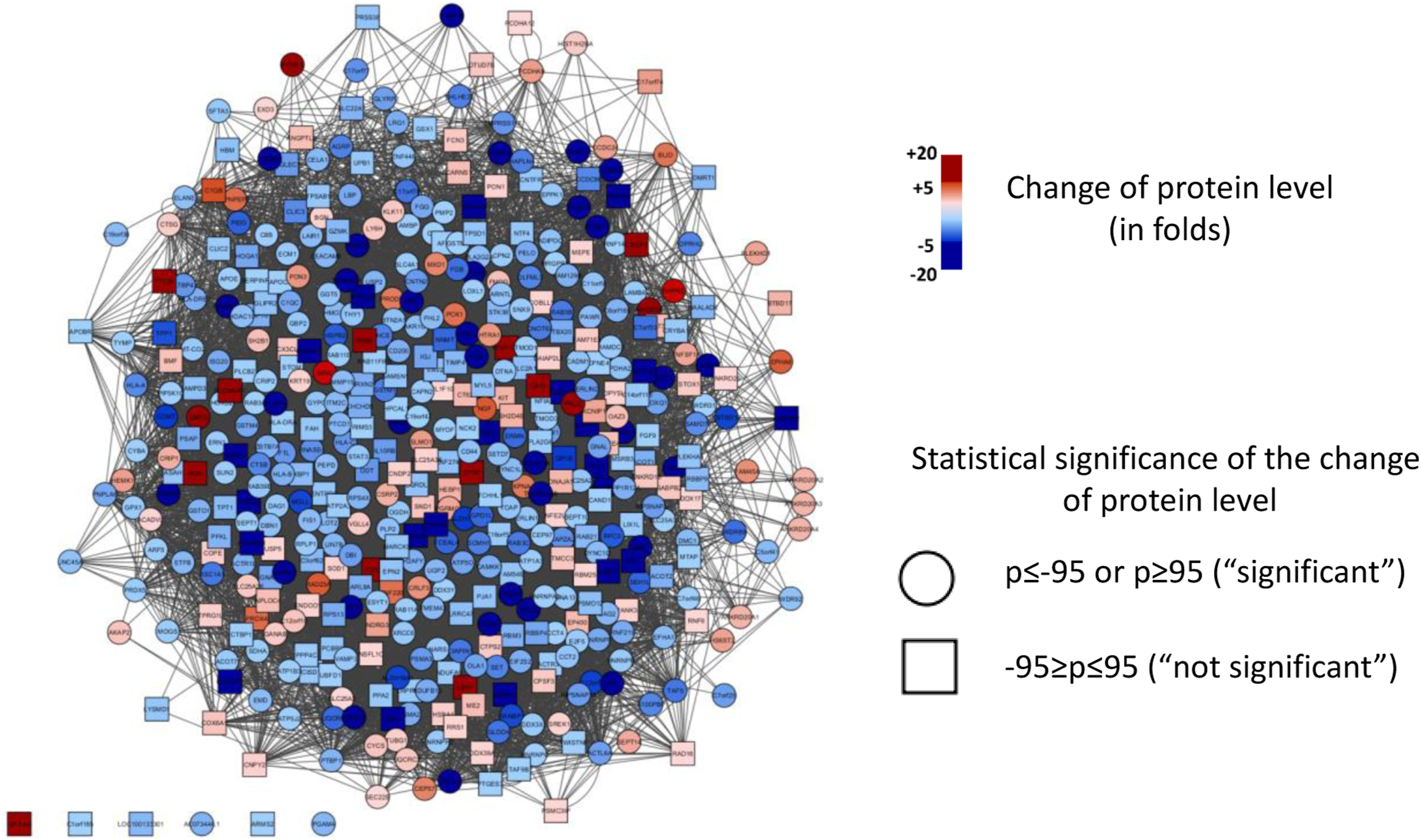

To further investigate pertubated pathways between VV and non-VV tissue, we utilized the DAVID bioinformatic resource system and found several pathways being affected. To broaden our search, we included all proteins with fold change >1.5. Interestingly, among others, we identified a profound disturbance in response to inflammation, blood vessels development, and oxidative phosphorylation all with known implication in VV, Figure 3. Protein-protein interaction. Bioinformatic analyses were performed with DAVID with input of those proteins with fold change >1.5 (220 proteins) between VV and non-VV. Protein-protein interaction showed profound disturbance in several mechanisms. (a) Inflammatory response cluster (n = 15); (b) Blood vessel development (n = 15); (c) Oxidative phosphorylation (n = 10). Protein–Protein interactions were obtained from the STRING database.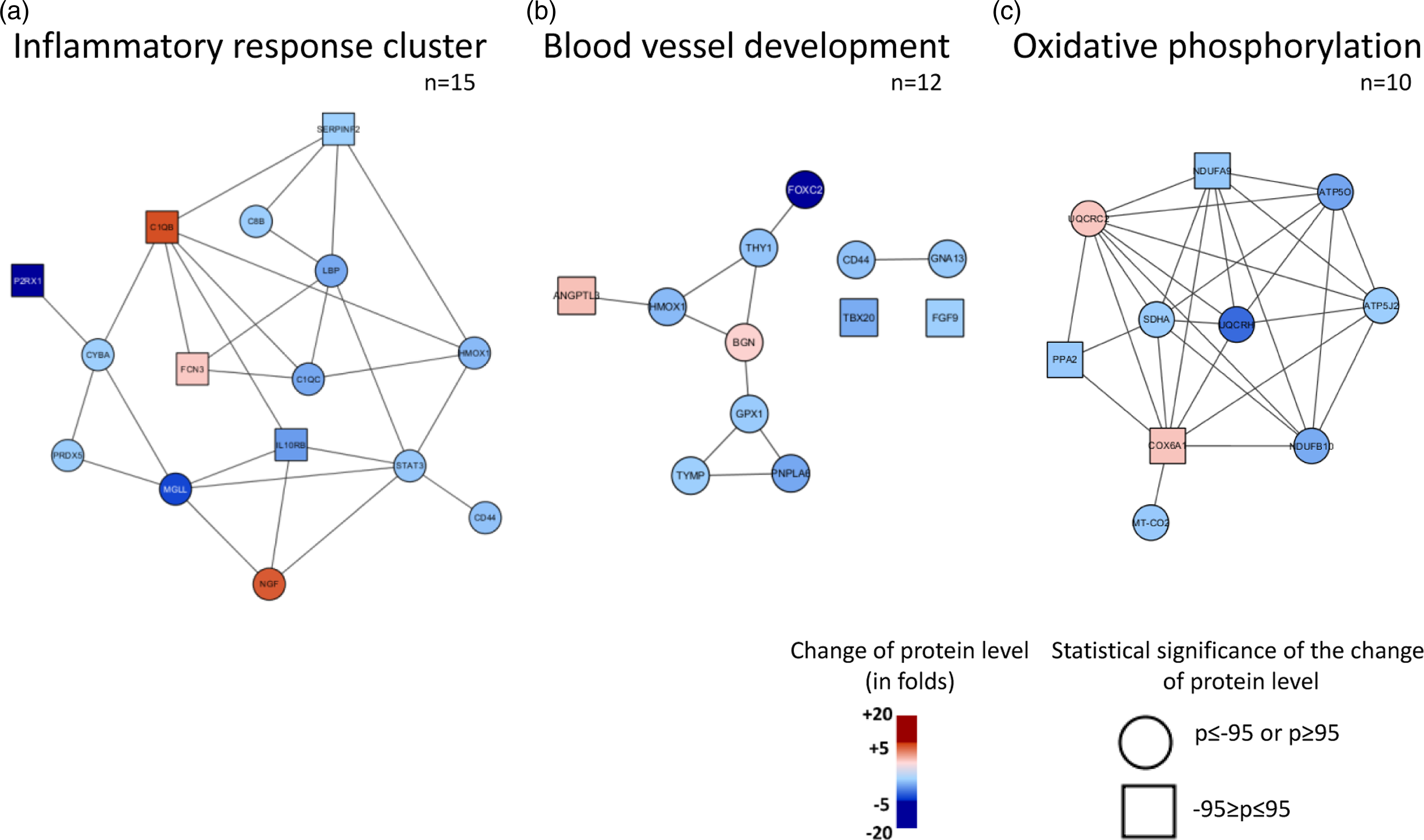
Discussion
Varicose veins of the lower limbs are one of the most common diseases treated by vascular surgery. The dilated and tortuous great saphenous veins are not only cosmetics problem, but also seriously affects patients’ quality of life. Treatment of VV will often be interventional therapies, which can achieve immediate results. Therefore, research on the treatment of VV of the great saphenous vein of the lower limbs has mainly focused on clinical treatment with relatively fewer basic research studies. In the present study, we collected the remaining normal veins from peripheral vascular bypass grafting surgery and varicose veins after great saphenous vein stripping surgery and analyzed them using an MS-based proteomic approach, and based on the results, we hypothesized that patients with VVs display a specific protein expression profile different from non-VV patients. As such, we found that particular differences in protein expression of proteins involved in the ECM, that is, fibromodulin, olfactomedin-like 3 protein (OLFM13), emerin, transmembrane protein 43 (TMEM43), erlin1, fibromodulin (FMOD), SEH1, and CXXC5, were all identified as differentially expressed presumably due to that they are either component of the primary structural behind biomechanical properties of the vascular wall or has a role in the mechanism of ECM homeostasis. As such, we also identified GAPDH, and when considered, this metabolic protein’s non-glycolytic function also seems to play a role in EMC remodeling.
Until now, only few proteomic analyses of NSV and VSV have been performed.5,15 Noteworthy, these analyses also identified a highly disturbed ECM. However, none of the significant differentially expressed proteins identified in our study were identified as significantly differential expressed in the previous studies. Even so, much knowledge is present combining ECM remodeling and several vascular conditions, including venous hypertension and VV. 15 Interestingly, using our quantitative proteomic approach, most proteins were identified with low expression in the VSV compared to the NSV samples which may allude that more degradation or lack of transcription/translation of proteins is present in VSV. Several of the identified proteins are affiliated with the ECM which may correlate with the changed structure in the VSV tissue. OLFML3 is known to exhibit proangiogenic properties. Similarly, to other matricellular proteins, OLFML3 is highly expressed during development, while its expression in adults is limited largely to tissues undergoing normal or pathological remodeling. 16 Interestingly and a putative future consideration for treatment, a recent study has demonstrated blockade of OLFML3 by anti- OLFML3 antibodies is highly effective in reducing tumor vascularization, pericyte coverage, and tumor growth as well as in vitro studies have shown OLFML3 targeting is sufficient to inhibit endothelial cell migration and sprouting. 17 These results must surely be investigated further, and even though our results show a downregulation of OLFML3 in the VSV tissue, this may pose an interesting novel putative insight into the pathogenesis of VV. Furthermore, it is well known that blood vessels are subjected to mechanical loads of pressure and flow, inducing smooth muscle circumferential and endothelial shear stresses. The perception and response of vascular tissue and living cells to these stresses and the microenvironment they are exposed to are critical to their function and survival. These mechanical stimuli not only cause morphological changes in cells and vessel walls but also can interfere with biochemical homeostasis, leading to vascular remodeling and dysfunction. The stretch caused by the distention and relaxation of the vascular wall is an important factor in the regulation of vascular modeling and remodeling.18,19 There is growing evidence that mechanical stretch modulates the functions (e.g., apoptosis, proliferation, and migration) of vascular smooth muscle cells (VSMCs) in the media of the vascular wall 19 and that chronically elevated stretch stimulates VSMC functions to mediate vascular remodeling during hypertension. 20 The proliferation of VSMCs in response to excessive stretch is crucial in vascular remodeling in hypertension. To elucidate the molecular mechanism, Qi et al. studied the mechanobiological roles of emerin and lamin A/C, two important components of nuclear envelope proteins localized beneath the inner nuclear membrane. The authors found that emerin and lamin A/C play significant roles in the mechanical modulation of VSMC proliferation. 18 Also, in our study, we found the vascular remodeling proteins emerin, TMEM43, and erlin1 downregulated in the VSV tissue. In summary, elevated venous pressure in the lower extremity veins during prolonged standing generates circumferential stretch of the vein wall and imposes mechanical stimulation on both ECMs and VSMCs.
In VSV, the perivascular space is surrounded by ECM proteins. This perivascular cuff is an initial response to an increased mechanical load and is accompanied by collagen deposition. However, the collagen fibers show abnormal distribution and morphology. Several studies demonstrated that there is significant hypertrophy of the media layer of the wall of VV compared with normal veins 21 with other studies showing that the medial layer consisted of increased number of collagen fibers and that elastin fibers constantly fragmented with paralleled interruption of the internal elastic lamina. The adventitial layer in VV showed decreased density and size of elastin fibers and increased degradation.22,23 Interestingly, in our study, we found an increased FMOD protein expression in VSV tissue, which further adds to our hypothesis of a remodeled ECM and suggest that it may be due to media layer hypertrophy. In tread with the deregulation of the ECM with a pattern of downregulated proteins, we demonstrated a higher expression of GAPDH in the VSV tissue. In that, hypoxia up-regulates the expression of GAPDH in a cell type-specific manner and that GAPDH is a protein with great biochemical and biophysical interests because of its functional importance in glucose metabolism, this may imply a changed metabolic turn-over in the VV patients. This is further implied by recent discoveries on GAPDH that in addition to its role in glucose metabolism has unraveled GAPDH’s non-glycolytic roles which show a more multifaced function of GAPDH both associated with DNA repair and thereby affects cell death, autophagy, and apoptosis, depending on its cellular location and post-translational modifications.24,25 Under hypoxia and cellular stress, GAPDH has showed enhanced secretion and may correlate with our finding of higher expression levels in the VSV tissue. 26
In addition to the various ECM proteins identified, we also identified SEH1, a nucleoporin with a lower expression in the VSV tissue. A role for nucleoporins (NUPs) in vascular disorders has been reported, where they may function to drive VSMC in the setting of neointimal hyperplasia. 27 It is unclear, however, if the loss of critical interactions at the nuclear envelope or binding to chromatin or protein within the nuclear interior is the main consequence of increased nucleoplasmic localization. Given that intranuclear mobility is a normal function and characteristic of NUP, it may reflect a dysregulation of NUPs and may affect its normal function and interacting partners. These emerging data point to potential and specific functional roles for NUPs in vascular development and disease remains to be studied further. Even though, cardiovascular disease is a confounding disorder with a multigenic basis, a number of clinical studies have demonstrated consistent NUP derangement associated with disease. 28 Still, it remains uninvestigated whether SEH1 dysregulation occurs as a cause or effect of vascular pathology despite evidence for NUP malfunction as the underlying cause for cardiac disease is strongly supported by current data. 27
In the present study, we found CXXC5 to have as low as 38-fold lower expression in VSV tissue compared to the NSV tissue. This protein is a known transcription factor that plays a vital role in a variety of cellular activities, including cell development, proliferation, differentiation, and migration as well as vessel formation. 29 Among several others, CXXRC5 is a transcription factor for Flk-1/KDR, the primary receptor for vascular endothelial growth factor (VEGF) and it has been shown that high expression of CXXC5 increase the expression of VEGF. 30 With VEGF’s central role in angiogenesis and neovascularization, and its stimulatory role in endothelial survival, proliferation, and migration, inhibition of the VEGF pathway leads to decreased messenger RNA levels of endothelial-specific markers. 31 It is clear that migration of endothelial cells is essential for sprouting angiogenesis, and fusion of endothelial cells participating in sprouting angiogenesis are tightly regulated by numerous and specific signals during vessel structure formation. 32 Furthermore, Kim et al. showed that in CXXC5-/- mice vessel formation is dependent on CXXC5 protein expression. 33 Held together, this might indicate that the activation of VEGF in patients with VSV is inhibited. This led us to hypothesize that the downregulation CXXC5 may cause weakening of the venous wall and may be an additional mechanism responsible for the formation of VV.
For the present study, we included well-characterized patient samples, even though a caveat of working with patient specimens is that only end-stage disease can be assessed as well as the heterogeneous nature of patient specimens. Another factor limiting the ability of studying all aspects of the protein repertoire is the technical means currently at hand. MS in all its facets has led to a quantum leap in protein analysis and was instrumental for speeding up the process of analyzing proteins with good accuracy and reproducibility. Technical complexity of the analysis process still limit high throughput and lack of sensitivity are aspects that have been restrictive to date. As with omics studies, another obstacle is the fact that proteins do not act on their own usually but exhibit their activity together with other proteins or ligands, and for the current study, we have focused on the expression levels of the proteins. To further establish the differentially expressed proteins’ putative role in the pathogenesis of VV, it is of outermost importance to further investigate those with functional studies. Still to date, this study is a hypothesis generating study and aims to identify proteins in the patient tissue that may have a function in the pathogenesis of VV and surely future studies in larger and independent cohorts await to validate our findings.
In conclusion, this study provides novel insights into the biochemical mechanisms of VV through proteomic analysis and provides a basis for further studies. Our proteomics discovery approach suggests that extracellular matrix degradation play a pivotal role in the pathogenesis of this disease. The differentially expressed proteins identified suggest that altered connective tissue proteins and increased proteolytic enzyme activity oxidation, inflammation, and apoptosis appear to be central to the pathophysiology of VV. Abnormalities in vein wall architecture probably precede the development of valvular incompetence and overt varicosities. Confirmation of the findings and the potential clinical role of the identified proteins still awaits.
Footnotes
Acknowledgments
We would like to thank Aldona Kriksciukaitiene for her assistance and guidance in this research, for WB analysis. We would also like to thank collegues from Vascular surgery department for technical support for this study.
Contributorship
RS and JS researched literature and conceived the study. SU, RS was involved in protocol development, gaining ethical approval, patient recruitment and data analysis. SU and RS wrote the first draft of the manuscript. AK, MV and ML made proteomics expertise and supervises lab works. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Asta og Rosa Jensens Fond (j.nr. 1-16-2-2-20).
Ethical statement
Guarantor
SU.
