Abstract
Background
Lymphedema imposes a significant economic and social burden in modern societies. Controversies about its risk factors, diagnosis, and treatment permeate the literature. The goal of this study was to assess experts’ opinions on the available literature on lymphedema while following the Delphi methodology.
Methods
In December of 2019, the American Venous Forum created a working group tasked to develop a consensus statement regarding current practices for the diagnosis and treatment of lymphedema. A panel of experts was identified by the working group. The working group then compiled a list of clinical questions, risk factors, diagnosis and evaluation, and treatment of lymphedema. Fifteen questions that met the criteria for consensus were included in the list. Using a modified Delphi methodology, six questions that received between 60% and 80% of the votes were included in the list for the second round of analysis. Consensus was reached whenever >70% agreement was achieved.
Results
The panel of experts reached consensus that cancer, infection, chronic venous disease, and surgery are risk factors for secondary lymphedema. Consensus was also reached that clinical examination is adequate for diagnosing lymphedema and that all patients with chronic venous insufficiency (C3–C6) should be treated as lymphedema patients. No consensus was reached regarding routine clinical practice use of radionuclide lymphoscintigraphy as a mandatory diagnostic tool. However, the panel came to consensus regarding the importance of quantifying edema in all patients (93.6% in favor). In terms of treatment, consensus was reached favoring the regular use of compression garments to reduce lymphedema progression (89.4% in favor, 10.6% against; mean score of 79), but the use of Velcro devices as the first line of compression therapy did not reach consensus (59.6% in favor vs 40.4% against; total score of 15). There was agreement that sequential pneumatic compression should be considered as adjuvant therapy in the maintenance phase of treatment (91.5% in favor vs. 8.5% against; mean score of 85), but less so in its initial phases (61.7% in favor vs. 38.3% against; mean score of 27). Most of the panel agreed that manual lymphatic drainage should be a mandatory treatment modality (70.2% in favor), but the panel was split in half regarding the proposal that reductive surgery should be considered for patients with failed conservative treatment.
Conclusion
This consensus process demonstrated that lymphedema experts agree on the majority of the statements related to risk factors for lymphedema, and the diagnostic workup for lymphedema patients. Less agreement was demonstrated on statements related to treatment of lymphedema. This consensus suggests that variability in lymphedema care is high even among the experts. Developers of future practice guidelines for lymphedema should consider this information, especially in cases of low-level evidence that supports practice patterns with which the majority of experts disagree.
Introduction
Lymphedema is a common but often unrecognized clinicopathologic condition. Given the prevalence, lymphedema imposes a significant economic and social burden in modern societies. The pathophysiology of lymphedema is complex and not completely understood. Disruption or overload of the lymphatic system is deemed to be the initial trigger to cause secondary lymphedema in addition to aplasia or hypoplasia of lymphatic vessels in primary lymphedema. It can be a primary manifestation of a disease or secondary to other conditions, such as irradiation, surgical dissection, trauma, cancer, injury, or an infectious process. Regardless of the etiology, controversies about its risk factors, diagnosis, and treatment permeate the literature.
A consensus issued by the International Society of Lymphology (ISL) has been published. 1 The document primarily discusses the diagnosis and treatment of lymphedema. However, expert opinion about the available data is limited and no agreement or disagreement exist. Professional societies of specialists who diagnose and treat lymphedema on a regular basis collaborate infrequently. The only dedicated, internationally based, ongoing registry project, the International Lymphatic Disease and Lymphedema Patient Registry and Biorepository, which aims to collect and analyze data to improve outcomes and to further the field, is not expected to be completed until the end of 2025.
In the interim, significant debate and variability regarding the best diagnostic modality and treatment strategy endures. This ongoing debate motivated the American Venous Forum (AVF) to create a working group to address a set of questions related to risk factors, diagnosis and evaluation, and treatment of lymphedema. The goal of this study is to provide experts’ opinions on the key issues related to the management of lymphedema patients. Realizing the evidence base in this field of medicine is weak, the intent was to identify those elements of lymphedema upon which expert agree or disagree, and what evidence they use to justify their position, and guide their practice. The goal is to identify needs that may exist to support future development of lymphedema-related evidence-based practice guidelines.
Methods
In December of 2019, the AVF created a working group tasked to develop a consensus statement regarding current practices in the diagnosis and treatment of lymphedema. The working group served as the steering and writing committee for this project. This steering committee defined criteria for the selection of experts who would participate in the consensus process (consensus panel). These criteria included publications and presentations on lymphedema, participation with a specialty society, and significant representation of lymphedema patients in the expert’s clinical practice. In addition, the group wanted to ensure representation of a variety of practice settings (academic, private, hospital-based) and to include an international panel of experts.
The leadership of the American Venous Forum, American Venous and Lymphatic Society (AVLS) and Society for Vascular Medicine (SVM) were asked to nominate their representatives based on these criteria. The steering committee also defined the total number and composition of the writing committee, and the number of experts in the consensus panel (40 experts). The two leaders of the project (FL, AG) organized and oversaw the entire process, participated in the writing group, but were not included in the voting process.
The extended expert group (last name, first name in alphabetical order).

The entire process from initiation to writing and submission of the article was funded solely by the American Venous Forum. Panelist participation was voluntary.
Results and Comments
The Delphi consensus flowchart is depicted in Figure 1. Twenty-five statements were identified by the steering committee. At the completion of the first round, the entire panel voted whether to include each item in the final list. Four items that met the criteria for dropping were eliminated from the list. Twenty-one statements that met the criteria for consensus were included in the second round. This list was further refined based on the second round votes, and the resulting 19 statements were submitted for a third round. Seventeen of the 19 items met the criteria for including in the consensus document (more than 50% of the panelists voted) during the third round, with consensus on 12 statements (63%), and the Delphi process was stopped, as it met the pre-defined criteria (third round with a consensus of for more than 60% of the statements). The writing group reviewed and analyzed the findings from rounds 1 through 3, reviewed and verified supporting references, and wrote this consensus statement. Delphi consensus flowchart.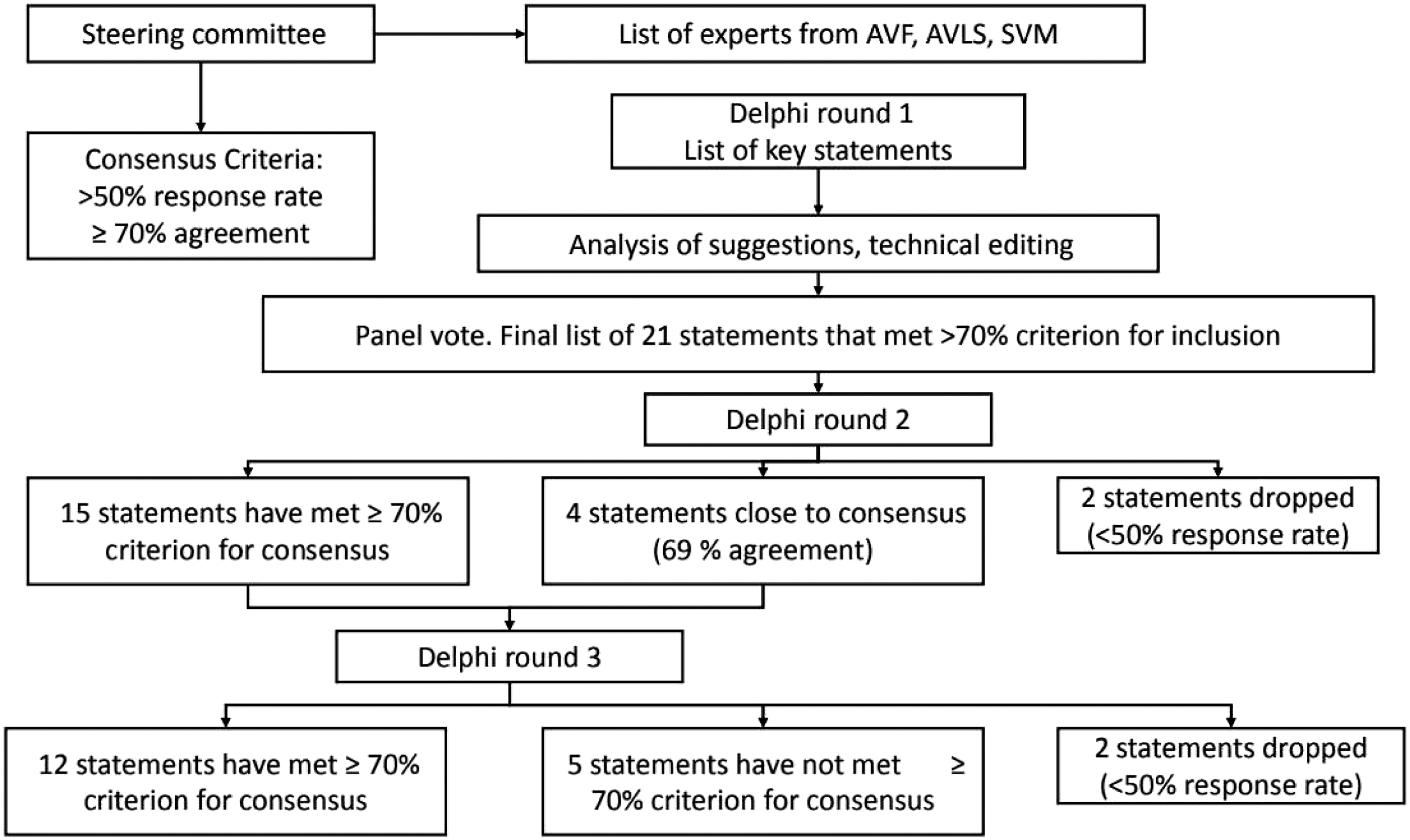
Statements Addressing Common Risk Factors for Lymphedema
The panel’s agreement rates on infection as a risk factor of lymphedema.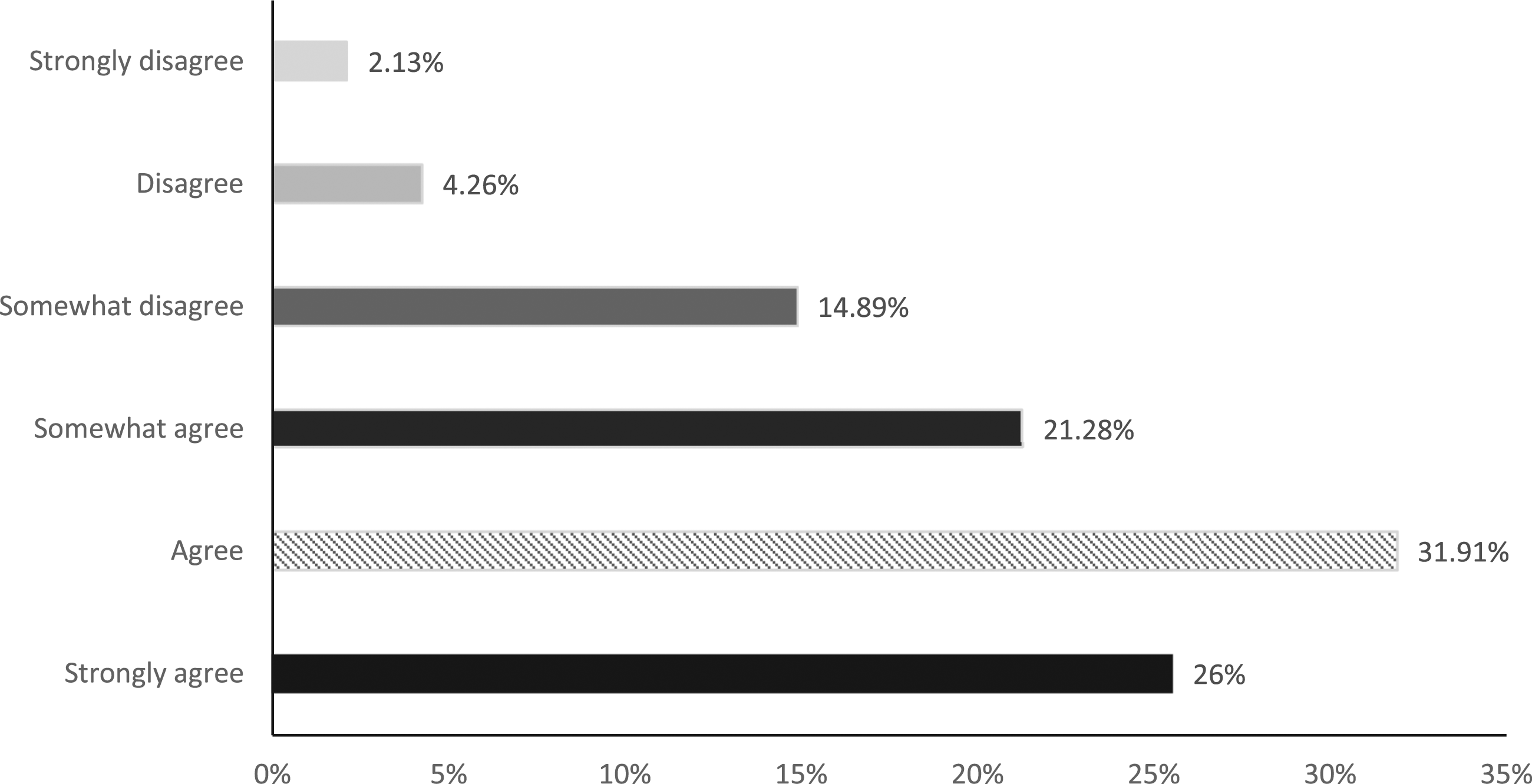
The panel’s support for a cancer-related therapy (CRT) and lymphedema association is drawn from the extensive breast cancer literature. 5 However, there is equally impressive and extensive literature regarding the broad diagnosis of cancer and the risk/prevalence of lymphedema. 6 Edema has been estimated to occur in 19% of people with advanced cancer. 7 There is little if any evidence to contradict this decision.
From a Western medical context, CRT remains the most prevalent cause of lymphedema. The main practical consideration here is related to patient education. When addressing the individual who will be treated for cancer, the subject of lymphedema risk should be addressed and, where appropriate, surveillance and risk-reduction measures should be undertaken.
The association of lower extremity infection with lymphedema is universally acknowledged. Globally, lymphedema frequently occurs due to non–surgically-related lymphatic filariasis, which is estimated to affect more than 40 million individuals worldwide, according to the World Health Organization. 8 In this context, it would be incorrect to say that CRT is the most common factor for developing lymphedema. The theory that an initial episode of cellulitis could damage lymphatics initiating lymphedema though appealing is difficult to establish. Damstra et al. 9 performed lymphoscintigraphy on patients presenting with an initial case of erysipelas. They found evidence of significant subclinical lymphedema in the uninfected limb at baseline, supporting the theory that lymphedema contributes to the initiation of cellulitis infection cycles as opposed to the converse.
Infection is a common cause of severe illness and hospitalization in lymphedema patients. A recent meta-analysis of papers reporting on non-purulent cellulitis found that lymphedema was strongly associated with cellulitis having an odds ratio of 6.8. 10 Lymphedema directly results in impaired local immune responses which affects bacterial and fungal clearance and results in cellulitis. A recent 3-year review of 440 lymphedema patients followed closely by a comprehensive team of therapists and physicians found that approximately one in three (35.7%) of the population sustained one or more episodes of cellulitis, but patients with stage 3 lymphedema had roughly twice the rate of soft tissue infection as patients with stage II, 61.7% vs 31.8%, respectively (p < 0.001). 11 The implication is that more severe lymphedema causes more cellulitis. However, it could also be that more infections cause worsening lymphedema and swelling. This notion that recurrent episodes of cellulitis damage the cutaneous lymphatics resulting in progression of skin injury and worsening clinical stage of lymphedema is widely accepted though again difficult to prove.
The strength of the evidence supporting the association between CVI and consequent lymphedema was deemed strong enough to yield near unanimous agreement from the panelists. Only two of 47 respondents disagreed with the assertion that CVI and lymphedema are interrelated. Panelists also lamented the fact that this association remains unrecognized by many in the medical community. Several panelists intimated that only advanced CVI was a lymphedema risk factor.
Evidence supporting the association of CVI with lymphedema is based on both expert opinion12-16 and recent observational data.10-11, 17-18 A recent study identified CVI, not CRT, as the predominant cause of lower extremity lymphedema in 440 patients who presented for lymphatic physiotherapy to a university medical center’s cancer-based physical therapy department. 11 The venous and lymphatic systems are intimately related and mutually interdependent systems. Although a comprehensive overview of this unique veno-lymphatic axis was eloquently outlined by Mortimer 15 in 2000, subsequent recognition of this unique vascular association did not initially become more widely accepted. A decade later, several articles referenced the association between initial venous and subsequent lymphatic disease.12-14, 16 A dysfunctional venous system ultimately leads to a dysfunctional lymphatic system. Specifically, CVI initially increases venous filtration that triggers an initial increase in lymphatic transport. Eventually, the augmented filtration overloads lymphatic capability, and venous lymphedema or “phlebolymphedema” results. Untreated venous hypertension can permanently damage the lymphatic architecture, yielding a low flow obstructive lymphedema with worsening swelling and stereotypical skin changes. In 2014, Partsch and Lee 16 emphasized that “CVI (CEAP C3 to C6) is always a chronic venous-lymphatic insufficiency.” Recent observational studies have not only confirmed that CVI is a common risk factor for lymphedema but have also refuted the long-held doctrine that CRT is the most common cause of secondary lymphedema.10-11, 17-18
The panelists’ support surgery as a risk factor for lymphedema and this belief is specifically addressed within the cancer literature where surgery is strongly identified as an independent risk factor for lymphedema development. The extent of surgical intervention also bears a role in determining the likelihood of lymphedema sequelae.5-6 The difficulty here is determining the individual role of surgical trauma in lymphedema pathogenesis, with the proviso that the majority of these patients also undergo radiotherapy, which has a strong, independent lymphedema impact. In breast cancer, at least, there is evidence that surgery alone is not a strong risk predictor in the absence of other treatment factors that predispose to lymphedema. 19 Non–cancer-related surgical causation is commonly cited as a potential cause for lymphedema, but the literature support is not as robust as that for cancer-related mechanisms.
Statements Addressing Diagnosis and Evaluation
There was universal opinion by the panel experts that, in almost all cases, physical examination supplemented by venous ultrasound are sufficient to make a lymphedema diagnosis. However, no high-quality studies have been performed to determine the accuracy of clinical examination nor the inter- and intra-observer reproducibility of a clinical diagnosis. In addition, no cost analysis of clinical examination in comparison to combinations of examination and imaging have been performed from which strong recommendation can be made and be applied to most patients in most circumstances without reservation. The most accepted clinical staging system, developed by the International Society of Lymphography (ISL), uses only physical examination to classify patients, 1 although several national lymphatic societies have proposed using combinations of history, clinical examination, and imaging findings. It is well recognized that the diagnosis of Clinical Stage 2–3 lymphedema can be confidently made with clinical examination alone based on very characteristic findings. Most patients with Clinical stage 0–1 can also be diagnosed on clinical grounds based on the combination of identified risk factors, specific physical findings, and exclusion of alternate causes of limb enlargement. 20
In reviewing the panelist’s comments, one theme emerged. In order to evaluate the effectiveness of treatment, one has to be able to quantify the amount of swelling at intervals during follow up. Since swelling is the presenting symptom in lymphedema, it is important to have an objective measure of initial swelling in the pretreatment phase, and progressively evaluate whether the treatment reduces swelling. Thus, swelling needs to be quantified. Some concerns were raised however, as to the practicality and standardization of quantifying swelling. Some of these concerns may be addressed by application of the AFTD-pitting test. This test includes 4-factors:
Patients want to have confidence in their diagnosis; anecdotally, when patients are presented with the opportunity for imaging to increase the confidence of the diagnosis, some are interested. In addition, some of the experts consider additional assessment such as limb volume, physiologic changes, or lymphatic imaging necessary to make a confident diagnosis of Stage 0 and 1 lymphedema. It was noted that an imaging or physiological test is required for diagnosis when invasive treatment for lymphedema was planned or if a patient were part of a research trial.
Ultrasound will be done in nearly all patients to evaluate the venous system. One expert pointed out that there are ultrasound findings that can support a lymphedema diagnosis in Stages 0 and 1. 22 The panelist expressed that this information is also valuable to help educate patients, encourage the use of compression, and to guide and assess treatment responses. However, the experience and initiative to evaluate patients with ultrasound for lower stage lymphedema is limited to centers of excellence. Lymphoscintigraphy was the most commonly cited confirmatory test used in the lower stages of lymphedema. This is a modestly expensive procedure that involves several hours of a patient’s time and includes intradermal injection of radiotracer into the dorsum of the foot. Indocyanine green near-infrared fluorescence lymphography requires equipment that is currently only found in select lymphatic surgery centers. 23 However, it is increasingly being used by lymphatic surgeons to refine assessment of the clinical stage, observe lymphatic function real-time, and localize lymphatics relative to ultrasound-identified veins prior to lymphovenous anastomosis. 24
Reliable evidence linking CVI and lymphedema or phlebolymphedema is based on updated veno-lymphatic physiology, expert opinion, and radiographic and histologic data. From a practical standpoint, awareness that CVI is a risk factor for secondary lymphedema explains many of the clinical consequences of chronic venous hypertension including swelling, advanced trophic skin changes, and susceptibility to infection as well as loco-regional malignancy. Abundant corroborative radiographic studies have documented the association of CVI with lymphatic dysfunction via abnormal fluorescence microlymphography,25-26 lymphoscintigraphy,27-28 indirect lymphography, 29 magnetic resonance imaging, 30 and near-infrared fluorescence lymphatic imaging. 31 Histological studies from patients with CVI and stasis dermatitis demonstrate structural changes within the dermal lymphatic vessels including luminal obliteration, loss of the open intercellular junctions, and destruction of the anchoring filaments. 32 A histological study in lipodermatosclerosis documented destruction of lymphatics within the ulcer bed and a marked reduction in the number of peri-ulcerative lymphatics. 33
Treatment
The panel’s agreement rates on surgery as an appropriate strategy for high-risk patients, or stages 0–1 lymphedema.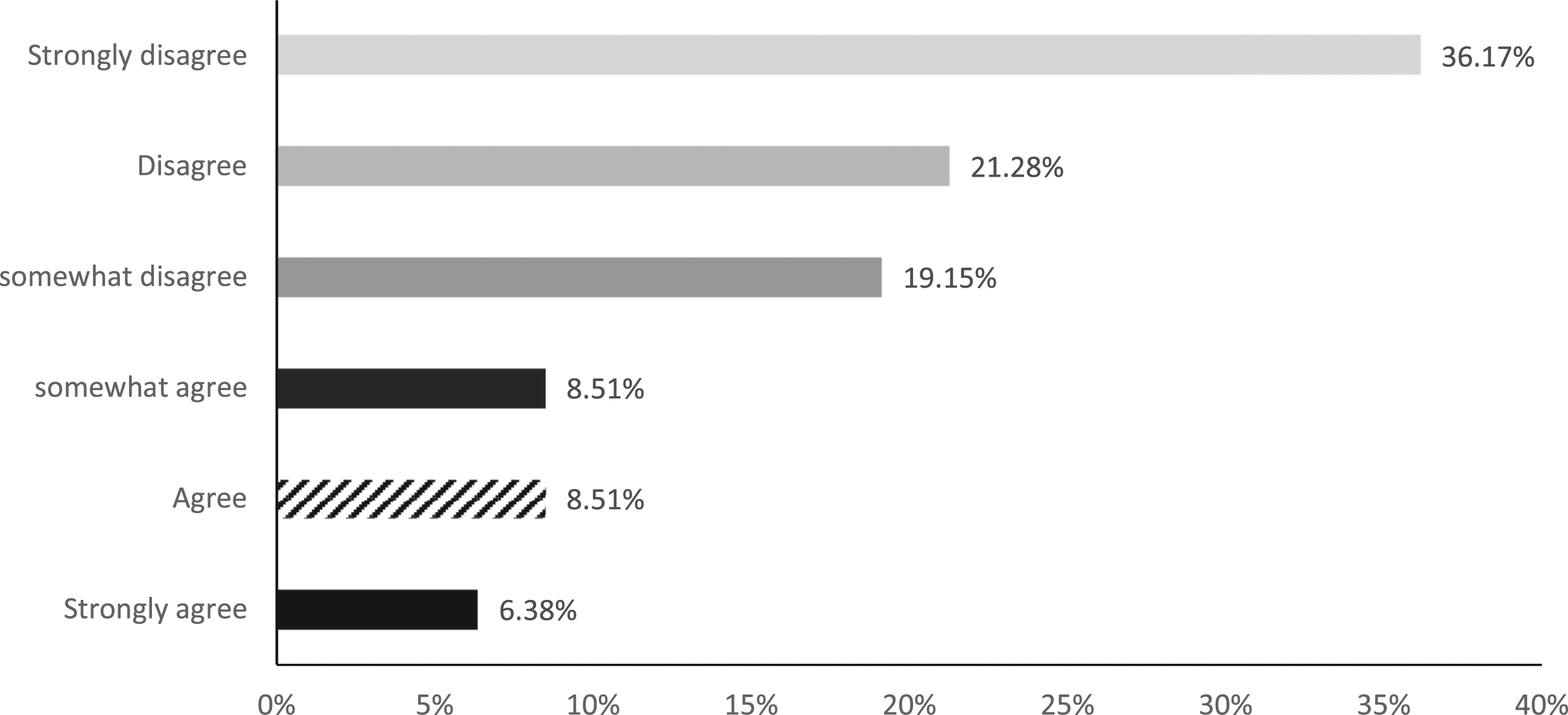
In a large cohort study, it was demonstrated that patients who adhere to wearing the compression sleeve have the lowest risk for regaining edema. 34 A systematic review by Gebruers and colleagues 35 also demonstrated that regular use of compression garments did not reduce swelling but prevented worsening. In studies evaluating the effects of compression sleeves with or without the addition of exercise in the intensive phase of therapy, the effect on edema volume reduction was limited to 50 mL.36-38 These results are to be expected since a compression sleeve is not a treatment modality to reduce volume but to maintain the leanest volume. In the treatment of severe lymphedema, a compression sleeve should be provided in the maintenance phase to limit the risk of volume increase. Therefore, a sleeve should not be used in the intensive phase unless the sleeve is provided very early after onset of lymphedema, as was the case in the study of Stout-Gergich. 36
When patients are provided elastic compression garments, care should be taken to ensure they are well fitted, providing the highest tolerated compression. In addition to size, length, and compression gradient; the manufacturing process also differs between garments and should be understood when making clinical recommendations.
“Elastic compression garments” can be manufactured with a variety of materials; but manufacture technique can be separated into circular knit and flat knit garments. Circular knit garments are seamless and generally not as dense or stiff as flat knit garments. A circular knitting machine creates a seamless tube of fabric. Yarn tension is varied to create the desired shape and Lycra is incorporated into the weave to create the desired compression. Compression is highest at the ankle and lower at the top of the garment. This largely automated manufacturing process allows for a more affordable mass-produced product. Although circular knit garments vary widely between products, they tend to be lighter and more compliant than the flat knit alternative. These garments are typically the first line of compression garments used by patients with relatively mild swelling and normal shaped legs. Garments which have more compliance are said to have low “containment.” Thus, a garment with low containment stretches during the day allowing some degree of expansion and swelling. Circular knit garments are thought to provide relatively less containment and therefore are usually ideal only in patients with mild/moderate lymphedema.
Flat knit machines create a sheet of fabric that must be cut and sewn by a seamstress or by a specialized automated sewing machine. This process allows for customization to accommodate any shaped leg. This process inherently creates seams in the garment as the edges are joined. The flat knit fabric tends to be thicker and stiffer. By sewing the garment according to a pattern, the density of the knit is held constant, and this garment is thought to provide enhanced “containment” even in the face of severe lymphedema. A product with high containment would be stiff and would fully resist expansion. This stiffer compression allows the garment to cross skin folds without cutting into the adjacent skin. Typical indications for flat knit garments include significant differences in leg circumference as well as deep skin folds and edema of the toes/forefoot. Unfortunately, these garments tend to be significantly more expensive which is often a barrier to acquisition.
The prevailing opinion that flat-knit garments provide intrinsically better containment than other garments was challenged by a study published by the International Compression Club (ICC) which concluded that the dynamic stiffness of circular and flat knit fabrics are not different. 42 Although flat knit may be a heavier fabric, the study concluded that once a piece of fabric has a stretch to it (which both flat and circular knit have), then the stiffness is not very high. We believe that the above analysis provides a good working understanding of the circular and flat knit garments. It is also true that the stocking manufacturers are constantly innovating which has resulted in more customizable circular garments and flat knit fabrics which are lighter and more complaint. Practical considerations regarding compression include issues related to patient compliance, cost, and reimbursement.
Velcro circumferential wraps are inelastic devices that are an alternative to compression garments. However, in early or uncomplicated lymphedema stage I, Velcro devices may be overtreating and elastic compression stockings (ECS) may just be as effective. Velcro may be a better option for lymphedema stage II and III. They have advantages in very large legs, can accommodate a variety of leg shapes and are adjustable. However, they require education to ensure proper donning and when and how to adjust the wrap. Some patients require assistance with application, and the devices may become uncomfortable in warmer humid weather. Patient compliance and preference are significant factors in the successful use of Velcro devices.
There was general consensus that SPC be recommended as an adjuvant of a multidisciplinary therapeutic treatment program that includes manual decongestive therapy, compression, and skin care for the more advanced stages of lymphedema. Some panelists felt SPC should be used in all stages of lymphedema, whereas others felt there is limited data to demonstrate its benefit for the lower classes of lymphedema when compared with inelastic compression of various forms. It was remarked that SCP may be the only decongestive therapy available to patients in certain regions with limited access to manual decongestive care. Several panelists stated that the data demonstrate that APCDs (advanced pneumatic compression devices with calibrated gradient pressure) were superior to simpler devices (without calibrated gradient pressure). Other than a small prospective randomized study which shows the effectiveness of an APCD over a standard device in breast cancer related lymphedema, 39 there is no GRADE A or Level 1 evidence supporting any treatment method, including pneumatic compression, for reduction and/or maintenance of swelling from lymphedema.
A study of 100 consecutive lymphedema patients (78% with a secondary etiology) who used an advanced sequential (intermittent) pneumatic compression (SPC) device in addition to compression demonstrated that SPC was well tolerated and associated with a significant reduction in limb girth, improved quality of life (QOL), and a reduction in cellulitis over a 12-month period compared to the prior year. 40 Two commercial claims-based studies demonstrated significant reduction in healthcare costs associated with the use of an advanced SPC device; one was in a mixed population of cancer and non-cancer associated lymphedema of both the upper and lower extremities, 41 and the other was in a population of phlebolymphedema patients, almost all of whom had either C5 or C6 CVI. 42 In both cases, the cost reduction was primarily driven by the reduced requirement for inpatient and outpatient treatment for cellulitis as well as reduced use of physical and occupation therapy.
Sequential pneumatic compression, often in conjunction with exercise, meticulous skin hygiene, static compression, and complete decongestive therapy (CDT), has shown benefit in treatment of lymphedema in numerous studies. However, most studies to date fail to distinguish the effectiveness of pneumatic compression based on ISL stage of lymphedema. A prospective study of 196 lower extremity lymphedema patients treated with APCDs demonstrated consistent reductions in limb volume (with 35% of patients enjoying >10% reduction) as well as a clinical improvement in skin fibrosis and physical function. 10 Others have shown significant improvement in quality of life (QOL) with use of APCDs. Blumberg et al. 40 studied 100 consecutive patients with lower extremity lymphedema treated with APCDs and demonstrated significant decrease in limb girth at ankle and calf at 12.7 months as well as improvement in QOL. The higher patient QOL may be related to a significant decrease in complications related to lymphedema in patients treated with SPC. Cellulitis is a recurrent complication resulting in regular hospital encounters (emergency room, clinic visits, or hospital admissions) and has been shown to be positively impacted by use of SPC. A retrospective analysis of a deidentified private insurance database and multivariate regression analysis comparing outcomes for the 12 months before and after APCD purchase identified 718 patients (374 in the cancer cohort, 344 in the non-cancer cohort). The study found an association between significant reductions in cellulitis (cancer vs non-cancer cohorts), outpatient care, and costs of APCD acquisition within a 1-year time frame in patients with both cancer-related and non–cancer-related lymphedema. 41
While the opinion of the panel was largely in agreement with this use of SPC in early stage lymphedema, the reason for lack of consensus appears to be a reluctance for some to endorse such a recommendation without high-quality evidence showing clear benefit, especially in light of their cost. In addition, some noted that early stages of lymphedema are poorly defined in this statement and that some differences may exist in benefit derived depending on the etiology of lymphedema (primary vs secondary). Specifically, the presence of phlebolymphedema (associated CVI) may be relevant.
Evidence supporting the use of SPC for early stage lymphedema comes from studies using indocyanine green (ICG) as a lymphatic contrast agent. In one small study, Rasmussen et al. 31 imaged 12 subjects with active leg ulcers (CEAP C6) using ICG to image lymphatics in patients before and after SPC. Baseline imaging showed impaired lymphatic function and bilateral dermal backflow in all subjects with CVI, even those without ulcer formation in the contralateral limb (C0 and C4 disease). APCD therapy promoted proximal movement of ICG away from the active wound in 9 of 12 subjects. Importantly, subjects with the longest duration of active ulcers had few visible lymphatic vessels, and proximal movement of ICG was not detected after APCD therapy. This finding supports intervention with APCDs at early stages (ISL 0–2) prior to lymphatic destruction and fibrosis commonly seen in later stages of lymphedema.
Phlebolymphedema may indeed be an under-recognized etiology for lymphedema that may benefit from earlier intervention with compression of all forms, including SPC. Few studies focus specifically on treatment of phlebolymphedema especially early stage disease. Studies have demonstrated that SPC results in more rapid healing of chronic venous stasis ulcerations compared to traditional compression bandages.43-44
MLD is a major component of Complete Decongestive Therapy (CDT) or conservative care (CC), which also includes bandaging, compression garments, exercise, and self-care. This light massage-like technique is done in a specific sequence to clear proximal congestion and redirect fluid to lymphatic beds/pathways with capacity to absorb the extra volume. 45
The evidence for proving that MLD alone is an effective treatment is poor. Because MLD is used as one component of decongestive lymphedema therapy, it is difficult to delineate the effect of each component.
Utilization of MLD on midline lymphedema (e.g. breast lymphedema) appears logical as there are few other treatments. While the theory behind using MLD to “clear the way ahead” to normal lymph drainage basins is sound. MLD technique can vary substantially between therapists, even between those taught through the same four training schools (Vodder, Foeldi, Casley-Smith, and Leduc).
One of the few RCTs demonstrating a positive effect of MLD was published by Williams and colleagues. 46 Another study on 42 patients randomized to 2 weeks of MLD plus hosiery or to hosiery alone showed that improvements seen in both groups were attributable to the use of compression sleeves and that MLD provided no extra benefit at any point during the trial. 47
A Cochrane review found little quality evidence to demonstrate the effectiveness of MLD. 48 The recent introduction of indocyanine green (ICG) lymphography-guided MLD may clarify what is most effective. 49
Mueller and associates identified eight studies in their systematic review to determine the impact of MLD on QOL. The majority of the RCTs were conducted with breast cancer patients only, but two included patients with phlebolymphedema. 50 Only one of the eight studies described a statistically improved QOL. Moreover, although pre- and post-treatment intervention changes in volume were observed, only one study of five showed a significant reduction in the absolute lymphedema volume compared to control.
A Cochrane systematic review reported six trials of MLD for breast cancer patients. 48 Trial numbers were small, ranging from 24 to 45 participants, and not adequately blinded. In two trials comparing MLD plus compression bandages versus compression bandages alone, the authors noted that the response was dependent on the objective outcome measure. There was a significant percent of volume reduction (the proportion of fluid reduced relative to the baseline excess volume, calculated as volume reduction divided by baseline lymphedema volume multiplied by 100) of 30%–38.6% for compression bandaging alone, and an additional 7.11% reduction for MLD (MLD 7.11%, 95% CI 1.75%–12.47%; two RCTs; 83 participants).
Volume reduction, defined as the amount of fluid reduction from before to after treatment, was not significant (p = 0.06). The lymphedema volume (the amount of excess fluid remaining in the arm after treatment) was also not significant (p = NS). Most importantly, subgroup analyses demonstrated that participants with mild‐to‐moderate breast cancer-related lymphedema responded better to MLD than those with moderate‐to‐severe disease. Brandao et al. carried out a systematic review, which specifically addressed CDT treatment of lower extremity lymphedema in a review of 2119 studies. In their systematic review, only five clinical trials met the eligibility criteria. 51 The largest RCT (n = 272) in this systematic review, conducted by Casley–Smith in 1996, showed that lymphedema volume was reduced in both groups (CDT alone versus CDT plus benzopyrones, either oral or topical). The CDT plus benzopyrones groups, however, demonstrated greater volume reduction and better maintenance of results. 52 Brandao and colleagues observed a discrepancy in many of the RCTs between the perception of clinical improvement by patients and researchers. 51 They suggested this difference is related to the outcome measures valued by the two groups. Researchers focus on volume reduction of lymphedema in contrast to patients, who focus on functionality, mobility, and complication rates.
These statements addressed the timing and role of surgery for advanced lymphedema. While physiologic surgical procedures are usually suggested for the early stage of lymphedema, reductive surgery is directed at late stage (II and III) lymphedema.
In 1912, Sir Havelock Charles outlined a surgical procedure for lower limb lymphedema without an actual case description. 53 This procedure was resurrected and popularized in 1950. 54 The debulking operation removed all overlying skin and soft tissue located above the deep fascia, which can be associated with morbid complications. The resultant surface was covered by a skin graft, usually harvested from the opposite thigh. Thompson 55 removed the fibrotic and fatty tissue and embedded the de-epithelialized skin flap in the nearby neurovascular bundle—known now as the “buried flap procedure.”
While principally extirpative, this procedure was also intended to be physiologic because it attempted to promote superficial lymph flow into the deep lymphatic system. Due to extensive scarring and other morbid sequela, these debulking “open” operations are now reserved for the most advanced cases. Surgical treatment for lymphedema has shifted to a less invasive approach for reducing lymphedematous tissue.
Liposuction or suction-assisted protein lipectomy (SALP), where the subcutaneous fat of the lymphedematous limb is suction-aspirated through a small metallic cannula attached to a vacuum suction machine, has become popular for reducing lymphedema. This procedure removes the solid hypertrophic component, which consists of fibrotic and hypertrophied subcutaneous adipose tissue. 56 Patients selected for this procedure usually have symptoms of discomfort (sometimes intractable pain) and/or dysfunction, where the large size and heaviness of the limb prevent movement. In addition, treatment may be indicated for recurrent episodes of cellulitis. Such patients typically present with late ISL Stage II or III non-pitting lymphedema after the lymph fluid component has been reduced by various compression methods.
Brorson and Svensson 57 combined liposuction with a compression garment in a prospective comparative study versus compression garment alone in 28 patients. They demonstrated a 104% decrease in limb volume versus the contralateral side with this combined treatment, whereas only a 47% reduction was observed in patients receiving compression garments alone. In another prospective study of 105 patients, Hoffner and associates demonstrated a mean extremity volume reduction of 117% when compared to the contralateral limb at 5 years following liposuction combined with compression. 58 Boyages and colleagues 59 performed liposuction in 21 of 55 eligible patients. Indications for treatment were 1) unilateral non-pitting ISL Stage 2, 3 LED; 2) a limb volume difference in excess of 25%; and 3) previously failed conservative therapy. Pre-surgical limb volume measurements by circumferential techniques were 44.2% for arms and 47.3% for legs. Limb volume was decreased to 3.6% for arms and 4.3% for legs 6 months after surgery. All patients reported improved function and symptoms and patient-reported outcomes were statistically significant.
Finally, Forte and colleagues carried out a systematic review of liposuction studies for lower limb lymphedema published between 2006 and 2018. From their initial literature search 129 articles were identified, but only 13 studies met their inclusion criteria. 60 Their review showed that edema was significantly decreased in all patients undergoing this treatment, but the maximum reduction in fluid was observed after 4–5 years of treatment. Another study in their SR demonstrated improvements in both QoL and function. The authors concluded that liposuction should be reserved for patients who fail conservative compression therapy with no reduction of lower limb volume and who are classified as ISL stages late II or III.
Discussion
Lymphedema is a clinicopathologic condition related to intrinsic or extrinsic impairment of lymphatic resorption and/or transportation. The Starling model of normal tissue fluid homeostasis published in 1896 posited that the preponderance of arteriolar-capillary interstitial filtrate was subsequently reabsorbed within the post capillary venules. Minimal residual interstitial fluid was processed via the lymphatic system. 61 However, Levick and Michel 62 subsequently refuted this principle as they identified that when the effect of the endothelial glycocalyx was considered, there was no steady state venous reabsorption of microvascular fluid but rather steady, dwindling filtration. Thus, the lymphatics, not the venules, are predominantly responsible for maintaining tissue fluid balance by absorbing interstitial fluid and ultimately returning it into the circulation. Based on this contemporary physiological data, experts such as the International Union of Phlebology 63 opined in 2013 that “lymphatic failure is responsible for all forms of peripheral edema.” In their 2014 article, Mortimer and Rockson 64 similarly noted that “all chronic edema indicates an inadequacy or failure of lymph drainage.” As such lymphedema has both primary and secondary etiologies. The panelists identify the commonly cited risk for lymphedema related to cancer, surgery, and infection. In addition, the experts recognize the significant contribution from CVI.
The edema associated with CVI (C3–C6) represents lymphatic failure or phlebolymphedema. Abundant corroborative radiographic studies have documented the association of CVI with lymphatic dysfunction via abnormal fluorescence microlymphography,25-26 lymphoscintigraphy,27-28 indirect lymphography, 29 magnetic resonance imaging, 30 and near-infrared fluorescence lymphatic imaging. 31 Histological studies from patients with CVI and stasis dermatitis demonstrate structural changes within the dermal lymphatic vessels including luminal obliteration, loss of the open intercellular junctions, and destruction of the anchoring filaments. 32 A histological study in lipodermatosclerosis documented destruction of lymphatics within the ulcer bed and a marked reduction in the number of peri-ulcerative lymphatics. 33
The ISL Consensus Document 65 describes three clinical stages of lymphedema: Stage 1 is early edema, which is relatively high in protein content and usually improves with leg elevation; Stage 2 represents pitting edema that fails to improve with elevation alone; Stage 3 encompasses lymphostatic elephantiasis where pitting is absent, fibroadipose deposition and trophic skin changes such as acanthosis, fat deposits, and warty overgrowths develop. In advanced stages the accumulation of protein-rich lymph combined with stasis in the interstitial space has induced an intense inflammatory response within the tissue. This cellular reaction results in the proliferation of adipose tissue and deposition of fibrous tissue. The net consequences are nearly permanent edema of the limb, which may be unresponsive to conservative measures. The ISL also recognizes a latent or subclinical condition (stage 0) where impaired lymphatic transport (as illustrated by lymphoscintigraphy) may manifest in the absence of swelling as “subtle changes in tissue/fluid composition and changes in subjective symptoms.”
According to our expert panel, clinical recognition of lymphedema is preferred over additional imaging. Certainly, venous duplex will be done in nearly all patients to identify or exclude treatable contributing to the lymphedema is appropriate given the non-invasive nature of ultrasound. Lymphoscintigraphy or ICG imaging while only available in specialized centers may be helpful to identify stage 0 or 1 disease or to provide confirmation to patients in whom the diagnosis is unclear. Imaging and physiologic testing is required for diagnosis when invasive treatment for is planned or when the patient is part of a research trial.
Implicit in the diagnosis of lymphedema, irrespective of the stage at presentation, is the concept that this is a progressive and incurable disease. As such, early diagnosis and intervention, analogous to treatments for cancer or other progressive disease, is paramount. Intervention at pre-fibrotic (Stages 1–2) or even subclinical (Stage 0) stages is widely considered optimal for best results. All patients with edema due to chronic venous insufficiency should be considered for treatment similar to lymphedema patients. From a practical standpoint, awareness that CVI is a risk factor for secondary lymphedema explains many of the clinical consequences of chronic venous hypertension including swelling, advanced trophic skin changes, and susceptibility to infection as well as loco-regional malignancy. Although compression and exercise-based therapy of venous and lymphatic hypertension are traditionally similar, recognition of concurrent CVI and secondary lymphedema should prompt consideration for instituting advanced compressive modalities such as complete decongestive lymphatic physiotherapy and pneumatic compression pumps.
The expert panel identified the use of compression for maintaining limb volume reduction as opposed to a primary method of decongestion. Circular knit compression, flat knit compression and Velcro devices all garner support from the panelists. Recognizing that the type of compression with need to be individualized and practical considerations include issues related to patient compliance, cost, and reimbursement. Decongestive therapy with SPC and MLD as part of an overall complex decongestive therapy program were also recognized, while surgical therapy had less support from the expert panel.
Several panelists stated that the data demonstrate that APCDs (advanced pneumatic compression devices with calibrated gradient pressure) were superior to simpler devices (without calibrated gradient pressure). The use of the trunk piece was felt by some to augment the physiologic effect. Patients usually find the use of these devices simple and comfortable although it does require at least an hour of daily commitment to use in a recumbent position. In clinical trials compliance is usually high but real-world data are lacking and some patients may discontinue using the device for various reasons. Some panelists noted that access to these devices may prove difficult because medical insurers will not cover them.
Notably, the cost of APCDs appears to be offset by savings incurred to the healthcare system. In the study by Karaca-Mandic et al., total lymphedema-related costs per patient, excluding medical equipment costs, were reduced by 37% in the cancer cohort and the corresponding decline in costs for the non-cancer cohort was 36%. 43 Others have shown similar clinical and economic benefits to APCD relative to conservative therapy alone or even simple pneumatic compression devices when treating patients with CVI and lymphedema. Specifically, a longitudinal matched case-control analysis of deidentified private insurance claims found that, compared to conservative therapy, APCD was associated with 69% lower total phlebolymphedema- and sequelae-related costs per patient and per year net of any pneumatic compression device-related costs. 42
Unfortunately, most studies, including the latter, do not distinguish among ISL stages of lymphedema as it pertains to treatment efficacy with SPC. Moreover, while numerous studies have established that SPC can improve QOL for patients with lymphedema (irrespective of etiology; primary vs secondary), inability to reliably control for patient compliance with regular use of pneumatic devices may limit the interpretation of results. Despite the need for stronger evidence supporting SPC for early stage lymphedema, the likely clinical and cost saving benefits associated with immediate intervention in this incurable disease buttress its early adoption.
Limitations. The results of this consensus should be interpreted in the context of several limitations. A systematic review of the literature was not the chosen method to gather published data on lymphedema. Therefore, the included studies level of evidence was not graded using a dedicated system (i.e. GRADE). We acknowledge the use of a 6-point Likert scale instead of more commonly used 5-point Likert scale in order to increase precision and gather the best assessment from the expert. A modified approach to present results was also implemented alongside with a careful and complete discussion of each item/question. Rather than imposing a major limitation, this stylistic strategy was intended to provide a global view from every step of the process, which pertains to a consensus document. With regards to conflict of interest (COI) reported by the authors, all compression treatments were evaluated in the questions and their positive outcome for their use based on data available. No specific pneumatic compression device was mentioned. In addition, evidence provided was based on clinical experience and judgement. The literature on all questions address was weak. When an agreement often was very strong, it was solely based on common sense and current clinical practice. Therefore, no bias based on COI was identified and no additional measures were taken. This further emphasizes the substantial need for further and more robust research in this area.
One of the aims of this project was to identify the evidence currently guiding the practices of the lymphedema experts. Included in this study panelists were aware of the most recent publications and have published on this topic themselves. Despite abundant recent literature related to lymphedema, including several randomized trials and systematic reviews, the panelists selectively referenced studies that are aligned with their clinical experience. This trend indicates a discrepancy between a formal assessment of the level of evidence of a specific publication, and the expert clinical opinion of the utility of such publication for clinical practice. It also emphasizes the need for well clinically and methodologically designed research studies addressing important aspects of lymphedema care.
Conclusion
This consensus process demonstrated that lymphedema experts agree on the majority of the statements related to risk factors and diagnostic evaluation for lymphedema patients. Less agreement was demonstrated on statements related to treatment of lymphedema. Experts justified their positions and practices by referencing to abundant literature source, most of which are low-level evidence, and systematic reviews that concluded that the evidence is of insufficient quality. This consensus suggests that variability in lymphedema care is high even among the experts. Developers of future practice should consider this information guidelines for lymphedema, especially in cases of low-level evidence that supports practice patterns with which the majority of experts disagree.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Steven M. Dean: Tactile Medical (Advisory Board), Nicos Labropoulos: Tactile Medical (Consultant), Tom Maldonado: Tactile Medical (Consultant), Thomas F. O'Donnell Jr.: Tactile Medical (Consultant), Stanley G. Rockson: Koya, Inc (Chief Medical Officer), Fibralign, Inc (Research support), Antonios P. Gasparis: Tactile Medical (Chair Advisory Board).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
