Abstract
Introduction
Acute kidney injury (AKI) is commonly observed after Type A aortic dissection (TAAD) repair and is associated with increased morbidity and mortality. The long-term impact of postoperative AKI, including the influence of severity and operative factors, remains incompletely defined.
Methods
We conducted a systematic review and meta-analysis of studies reporting long-term survival after surgical TAAD repair stratified by postoperative AKI status, published through February 2025. Kaplan-Meier curves were digitized to reconstruct individual patient data.
Results
Nine studies with 6,608 patients were analyzed. Postoperative AKI occurred in 35.6% (95% CI 26.4–45.9%) and was associated with three-fold higher long-term mortality (HR 3.00, 95% CI 2.59–3.49, p < 0.001). Survival for AKI versus non-AKI patients was 75% (95% CI 73–77) versus 93% (95% CI 92–94) at 1 year and 52% (95% CI 47–58) versus 68% (95% CI 59–77) at 10 years, with a 2-years reduction in expected survival (95% CI -2.35 to −1.64, p < 0.001). Mortality was time-dependent, with early 3-months risk markedly elevated (HR 4.72, 95% CI 3.82–5.83) and sustained beyond 3 months (HR 1.74, 95% CI 1.41–2.15). Risk increased with AKI severity: stage 1 HR 1.79 (95% CI 1.30–2.48), stage 2 HR 2.88 (95% CI 2.02–4.10), stage 3 HR 5.06 (95% CI 3.75–6.81). Multiple sensitivity analyses confirm robustness.
Conclusion
Postoperative AKI affects one-third of TAAD patients and triples long-term mortality, especially early after surgery. These findings highlight the need for renal-protective strategies, careful operative planning, and structured follow-up to improve outcomes.
Keywords
Introduction
In the setting of surgical repair of Type A aortic dissection (TAAD), AKI is particularly prevalent, reflecting the complexity of the procedure, prolonged cardiopulmonary bypass, and frequent visceral and renal malperfusion.1–3 Reported rates of postoperative AKI after TAAD surgery range widely, from approximately 20% to over 60%, reflecting differences in patient populations, operative strategies, and definitions of AKI across studies.1–3 Despite the high incidence and clinical significance of AKI, the long-term prognostic impact of postoperative AKI in TAAD patients remains incompletely defined. Existing studies are limited by small sample sizes, heterogeneity in AKI definitions and management, and inconsistent follow-up. Furthermore, the influence of AKI severity, as well as patient- and procedure-related factors, on long-term outcomes has not been systematically quantified.
Given the burden of morbidity, mortality, and healthcare resources associated with AKI, a better understanding of its long-term impact could inform clinical decision-making, perioperative preventive strategies, and policy initiatives aimed at improving outcomes. To address these gaps, we conducted a systematic review and meta-analysis of published studies reporting long-term survival after TAAD repair stratified by postoperative AKI status.
Methods
This systematic review and meta-analysis was performed in adherence to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Supplemental Table 1). 4 The study protocol was prospectively registered (CRD420250654959). Ethical approval was not required for this study-level meta-analysis.
Search strategy
A comprehensive search of electronic databases (MEDLINE, Embase, Web of Science) was performed to identify studies published up to February 1st, 2025. The search strategy was developed using the PECOS framework to address the research question: In patients with Type A aortic dissection undergoing surgical repair, does postoperative acute kidney injury (AKI) affect long-term overall survival compared with no AKI? The complete search strategy is provided in Supplemental Table 2. In addition, reference lists of eligible studies and prior meta-analyses were screened to identify additional relevant publications.
Two investigators (X.J., M.P.S.) independently screened titles, abstracts, and full texts using prespecified inclusion and exclusion criteria. Studies were eligible if they enrolled patients with Type A aortic dissection treated surgically, reported Kaplan–Meier curves for overall survival stratified by postoperative AKI status, and provided sufficient data for extraction. Reviews, case reports, single-arm studies, and studies including Type B aortic dissection patients were excluded. When overlapping populations were identified, the most recent or most comprehensive report was included.
Data extraction, quality assessment and statistical analysis
Patient characteristics and Kaplan-Meier curves were extracted independently by two reviewers (X.J, M.P.S). The methodological quality and risk of bias were also assessed independently by the same authors (X.J, M.P.S) using the Cochrane Risk of Bias tool.
Kaplan-Meier curves were digitized using Web Plot Digitizer software. Reconstructed time-to-event data and individual patient data (IPD) were derived from the digitized Kaplan-Meier plots, along with the corresponding total number of patients, total events, and the number of patients at risk at various time intervals for each study arm. 5 Hazard ratios (HR) with 95% confidence intervals (CI) were calculated using a Cox frailty model with robust standard errors. Heterogeneity across studies was assessed by testing for an interaction between the trial and treatment effect, incorporating a γ frailty term to account for between-study variability, with studies modeled as random effects (Supplemental Table 3). The significance of the variance parameter was evaluated using the likelihood ratio test. Proportional hazards were checked using the Grambsch-Therneau test and Schoenfeld residual diagnostic plots. As a further analysis, conventional “two-step” meta-analysis using the DerSimonian Laird random-effects model were conducted forest plots were used to display the pooled estimates. Between-study heterogeneity was assessed through Cochran Q statistic and I2. Publication bias was assessed via funnel plots and Egger’s test for each outcome of interest. The restricted mean survival time (RMST), interpretable as a quantification of loss of life expectancy, was calculated and the difference between both study arms was compared. Jackknife resampling analyses were performed excluding each study in turn. Pre-specified random-effects meta-regression analyses were also conducted to examine the impact of moderator variables on the outcomes. Significance testing was performed at the two-tailed 5% significance level. All analyses were completed with R Statistical Software (version 4.4.0, Foundation for Statistical Computing, Vienna, Austria).
Results
Study selection
Our initial search yielded 533 unique citations (Figure 1), of which 43 publications were potentially relevant (Figure 1(A)). A total of 9 retrospective, observational studies were included, encompassing over 6,608 patients undergoing surgical repair for TAAD (Table 1).1,3,6–12 Study periods ranged from 1990 to 2022, and the cohorts represented populations from North America, Europe, and Asia. Two studies utilized prospective registry data (IRAD and the Nordic Consortium), while the remainder were single- or multicenter retrospective analyses. Supplemental Figure 1 presents the qualitative assessment of the included studies using the ROBINS-E tool, highlighting several sources of concern. Overall, the risk of bias was judged to be moderate. Study selection and incidence of postoperative acute kidney injury (AKI). Legend: (a) PRISMA flow diagram illustrating the identification, screening, eligibility assessment, and inclusion of studies in the systematic review and meta-analysis. (b) Forest plot showing the pooled incidence of postoperative AKI using a random-effects meta-analysis of proportions. The pooled estimate (diamond) and 95% confidence intervals (CI) are displayed. Characteristics of the studies evaluating outcomes of acute kidney injury following type A aortic dissection. Legend: Study design, time period, total sample size, number of patients in each group.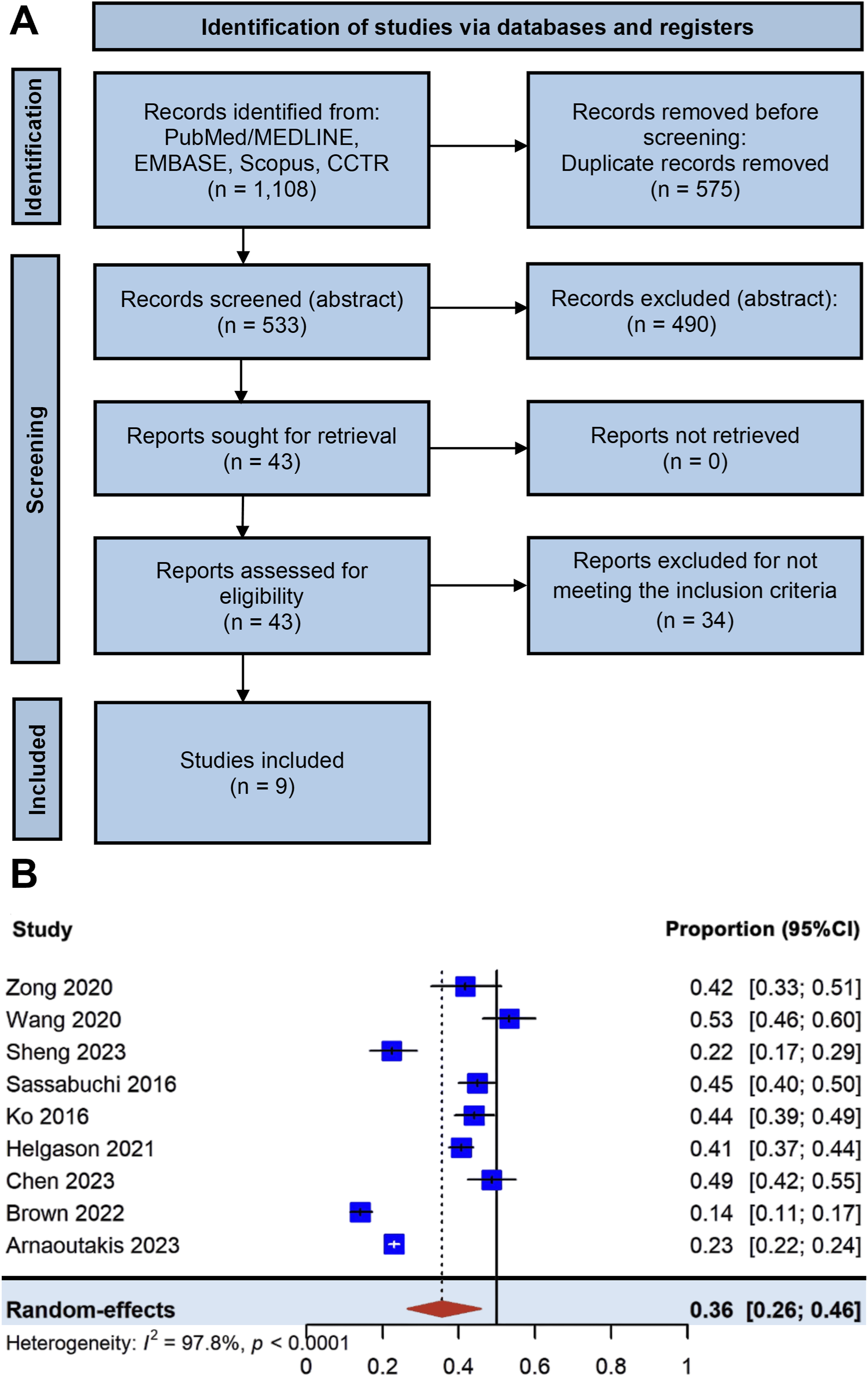
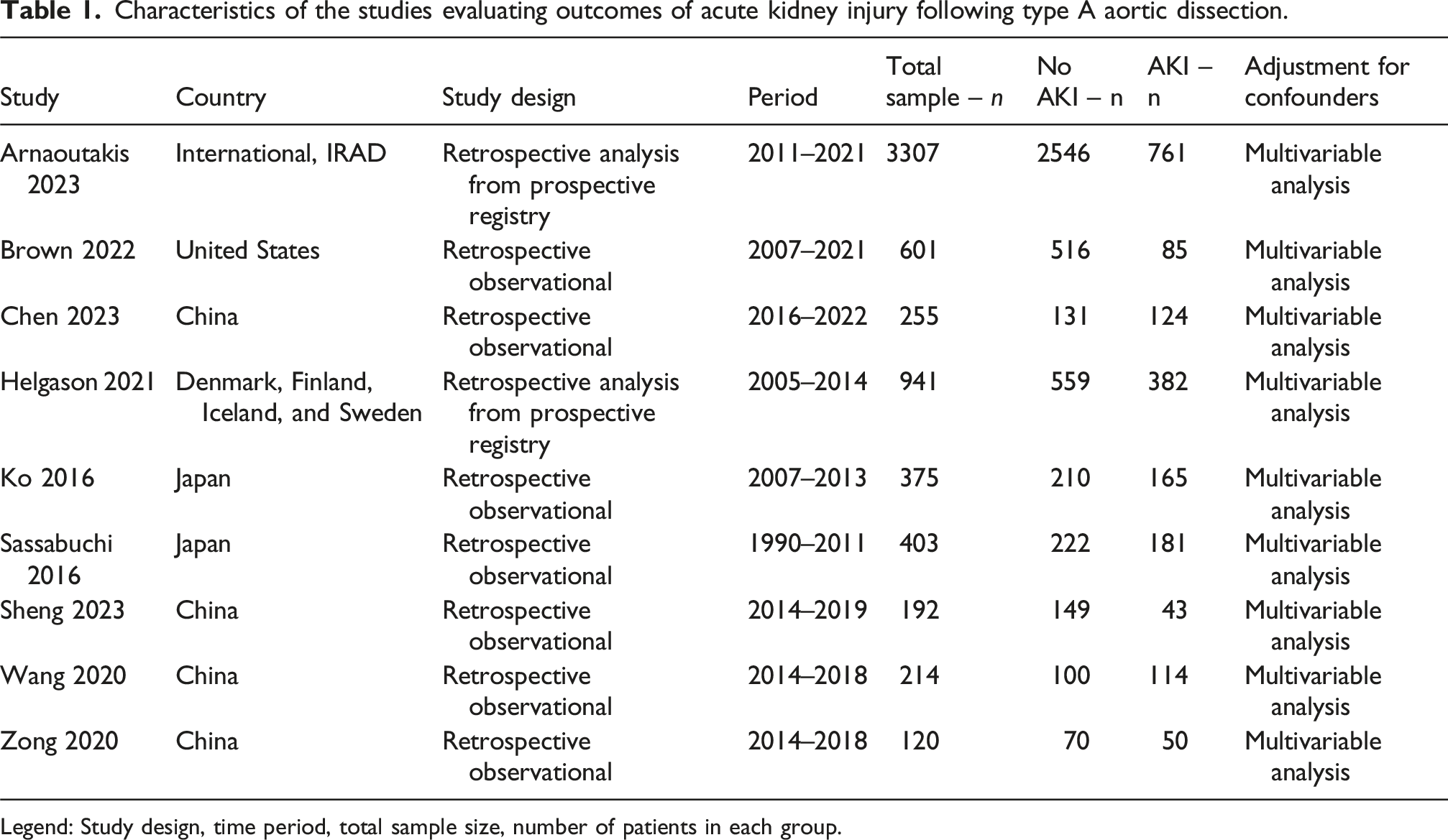
Patient characteristics of the included studies.
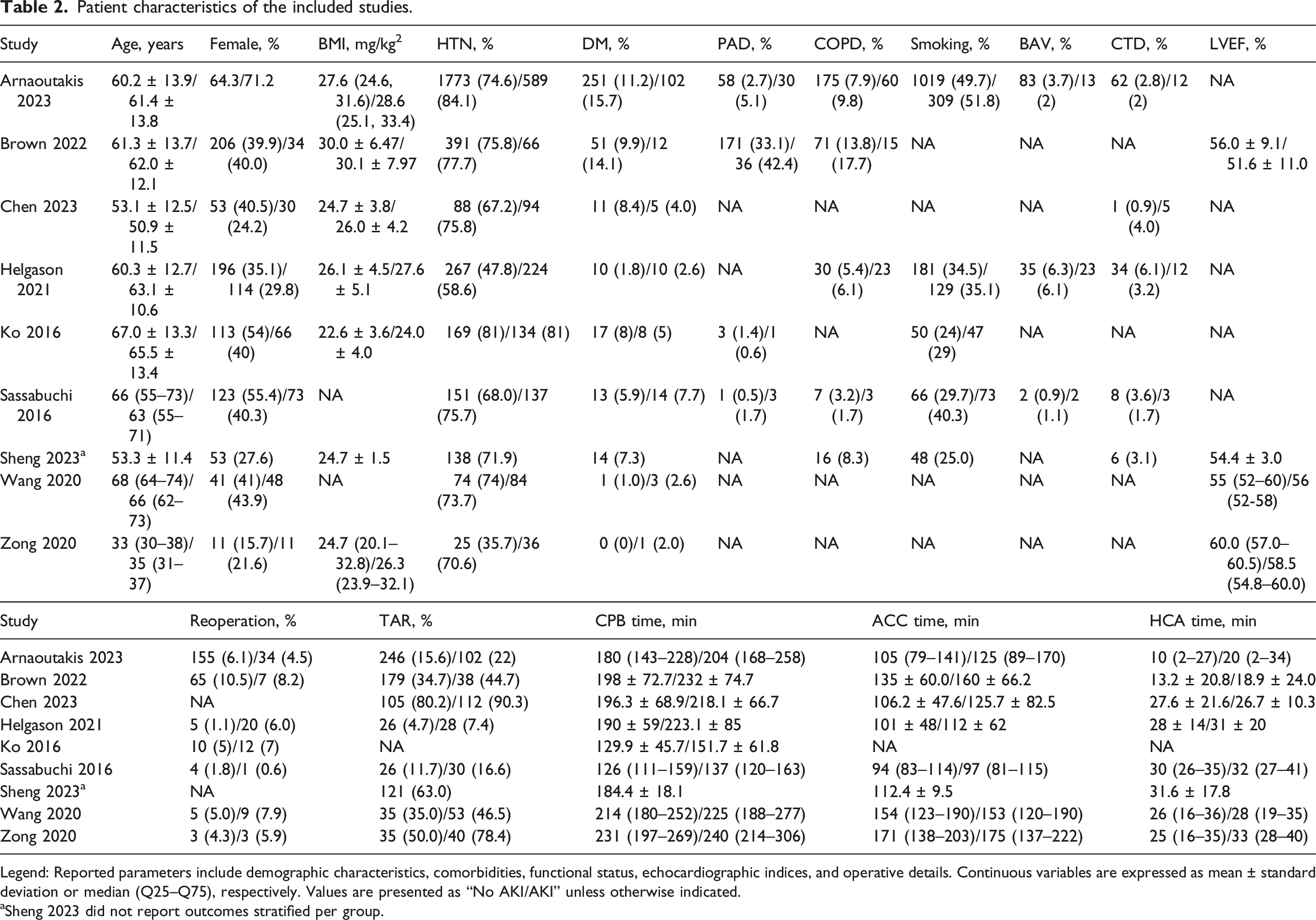
Legend: Reported parameters include demographic characteristics, comorbidities, functional status, echocardiographic indices, and operative details. Continuous variables are expressed as mean ± standard deviation or median (Q25–Q75), respectively. Values are presented as “No AKI/AKI” unless otherwise indicated.
aSheng 2023 did not report outcomes stratified per group.
Operative characteristics were markedly different in patients who developed AKI. The overall frequency of reoperation was similar between groups (OR 1.25, 95% CI 0.65–2.39, p = 0.50), with study-level rates ranging from 0.6% to 10.5%. In contrast, total arch replacement (TAR) was significantly more frequent among AKI patients (OR 1.61, 95% CI 1.35-1.93, p < 0.001), with individual study rates ranging from 4.7% to 90.3%. Furthermore, on average, cardiopulmonary bypass (CPB) time was 20.9 min longer than in non-AKI patients (95% CI 14.1–27.7, range +9 to +34 min), aortic cross-clamp (ACC) time was 11.5 min longer (95% CI 4.2–18.8, range −1 to +25 min), and circulatory arrest time was 4.3 min longer (95% CI 1.4–7.1, range +2 to +10 min). There was significant heterogeneity across studies ranging from substantial to extremely high for almost all operative characteristics (I2 = 73–93%). For total arch replacement frequency, heterogeneity was minimal and not significant (I2 = 5%).
Overall outcomes
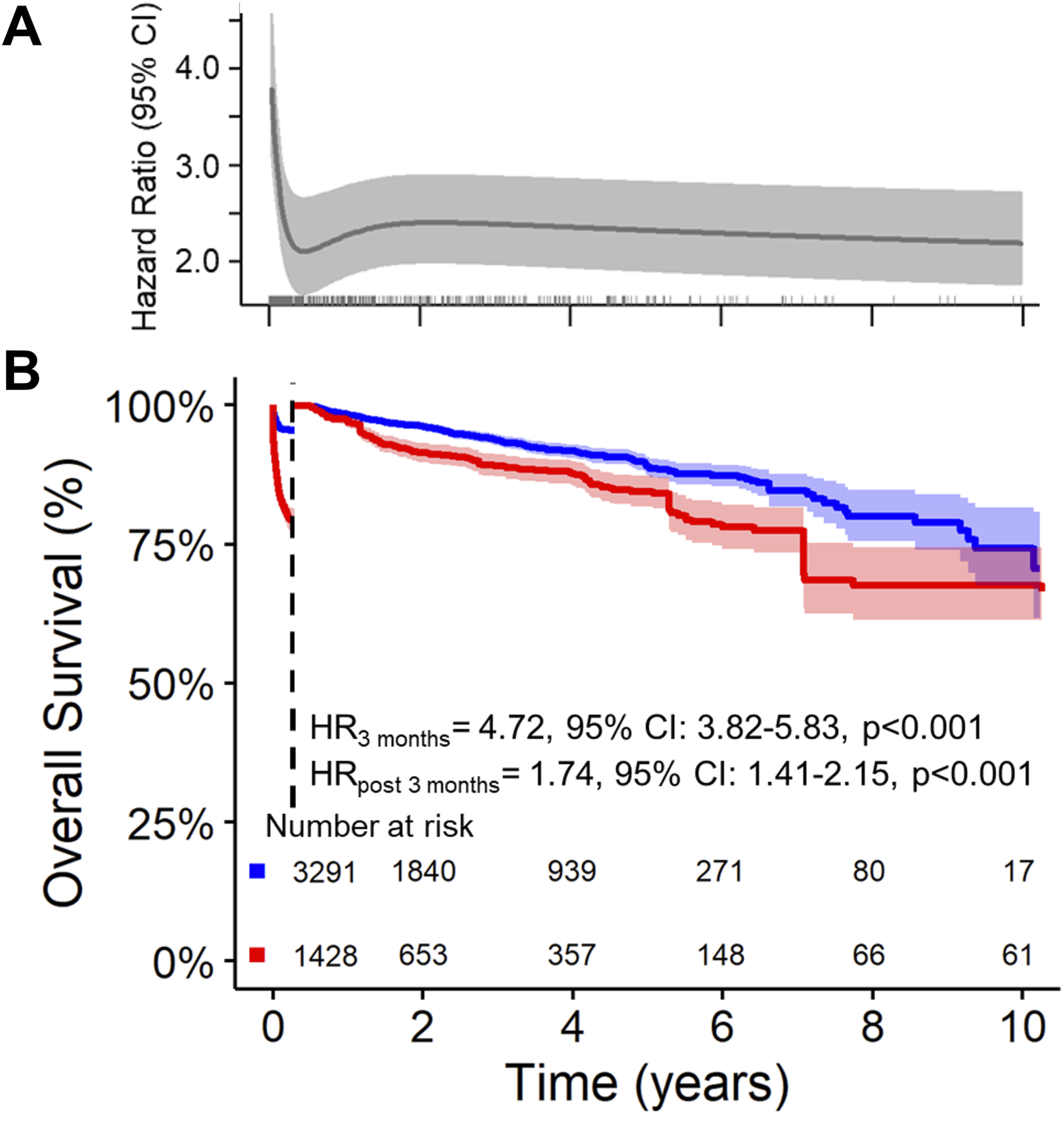
Nine studies were included, comprising 1,428 patients with AKI and 3,291 without, with a median follow-up of 2 years (IQR 0.8–4.2) and up to 10 years of total follow-up. Patients who developed AKI had a three-fold higher hazard of death compared with non-AKI patients (HR 3.00, 95% CI 2.59–3.49, p < 0.001). Estimated survival in the AKI group was 75% (95% CI 73–77) at 1 year, 66% (95% CI 64–69) at 5 years, and 52% (95% CI 47–58) at 10 years, compared to 93% (95% CI 92–94), 85% (95% CI 83–86), and 68% (95% CI 59–77) in non-AKI patients (Figure 2(A)). RMST analysis indicated that patients with AKI experienced a clinically significant reduction in expected survival of approximately 2.0 years compared with non-AKI patients (RMST difference −2.0 years, 95% CI −2.35 to −1.64, p < 0.001). The two-stage meta-analysis confirmed these findings, with pooled hazard ratio for mortality associated with postoperative AKI of 3.12 (95% CI 2.34–4.15, p < 0.001) (Figure 2(B)). Heterogeneity was substantial (I2 = 69.5%), reflecting variability in study-level effect sizes. In the main analysis, the relative hazard of death associated with AKI was time-dependent, with the proportional hazards assumption violated (p < 0.001, Figure 3(A)). Kaplan-Meier analysis showed markedly increased mortality in AKI patients. Within the first 3 months, the hazard of death was over four-fold higher than in non-AKI patients (HR 4.72, 95% CI 3.82–5.83, p < 0.001), and beyond 3 months, risk remained elevated (HR 1.74, 95% CI 1.41–2.15, p < 0.001), reflecting both early and sustained adverse effects on survival (Figure 3(B)). Long-term outcomes following type A aortic dissection repair according to postoperative acute kidney injury (AKI) status. Legend: (a) Pooled Kaplan-Meier survival curves for patients with and without postoperative AKI, reconstructed from included studies. (b) Forest plot showing hazard ratios (HR) for long-term mortality associated with postoperative AKI from both random-effects and fixed-effects meta-analyses, including individual study estimates with 95% confidence intervals (CI). Time-dependent mortality risk associated with postoperative acute kidney injury (AKI). Legend: (a) Time-varying hazard ratio (HR) for long-term mortality, illustrating how the relative risk associated with AKI changes over the follow-up period. (b) Landmark analysis at 3 months comparing early (≤3 months) and late (>3 months) mortality risk in patients with versus without AKI.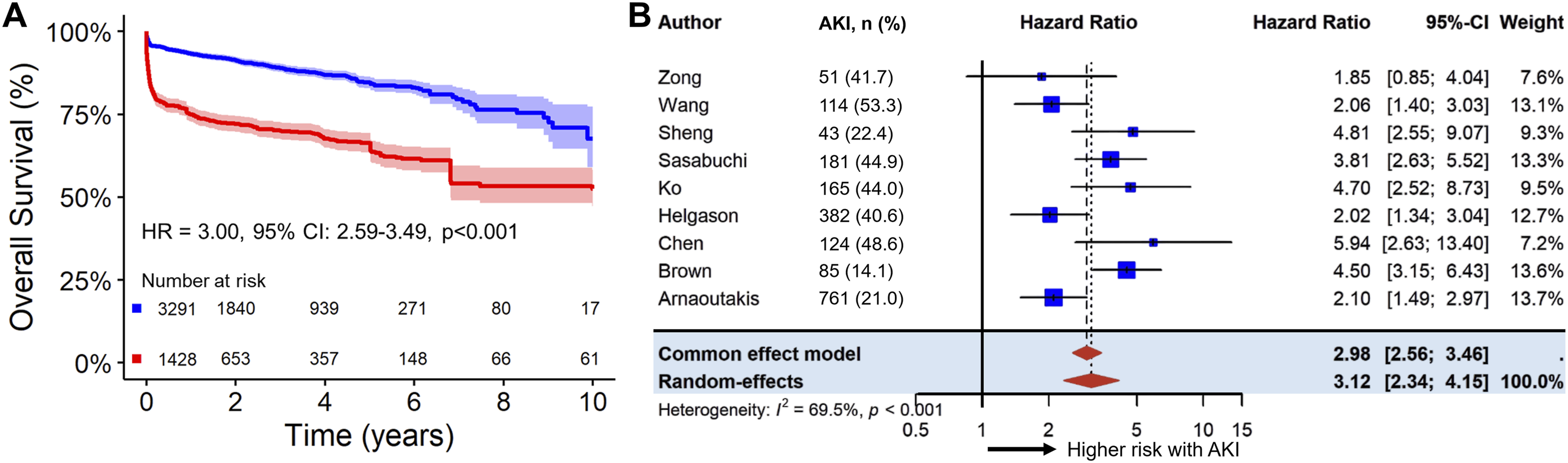
Overall outcomes according to AKI severity
Among 1,674 patients across 3 studies, 950 did not develop AKI, while 342, 164, and 218 patients developed AKI stages 1, 2, and 3, respectively. Median follow-up was 3.1 years (interquartile range, 1.3–5.4 years), with follow-up extending up to 10 years. As expected, survival declined progressively with increasing AKI severity (Figure 4(A), log-rank p < 0.001). Non-AKI patients had highest survival at 1, 5, and 10 years: 97% (95% CI 96–98), 88% (95% CI 85–91), and 67% (95% CI 56–79). In contrast, estimated survival probabilities for AKI stage 1 were 92% (95% CI 89–95), 82% (95% CI 77–87), and 67% (95% CI 59–77), for stage 2, 82% (95% CI 76–88), 68% (95% CI 60–77), and 63% (95% CI 54–74), and for stage 3, 69% (95% CI 63–75), 61% (95% CI 54–69), and 48% (95% CI 35–65) at 1,5 and 10 years respectively. Compared with non-AKI patients, the hazard ratios were 1.79 (95% CI 1.30–2.48, p < 0.001) for stage 1, 2.88 (95% CI 2.02–4.10, p < 0.001) for stage 2, and 5.06 (95% CI 3.75–6.81, p < 0.001) for stage 3. Similarly to the main analysis, the relative hazard of death associated with AKI was time-dependent, with the proportional hazards assumption violated (p < 0.001, Figure 4(B)). Long-term outcomes following type A aortic dissection repair according to postoperative acute kidney injury (AKI) severity. Legend: (a) Pooled Kaplan-Meier survival curves for patients stratified by AKI stage reconstructed from included studies. (b) Time-varying hazard ratio (HR) for long-term mortality, illustrating how the relative risk associated with AKI stages changes over the follow-up period. Inset shows early follow-up (≤4 months).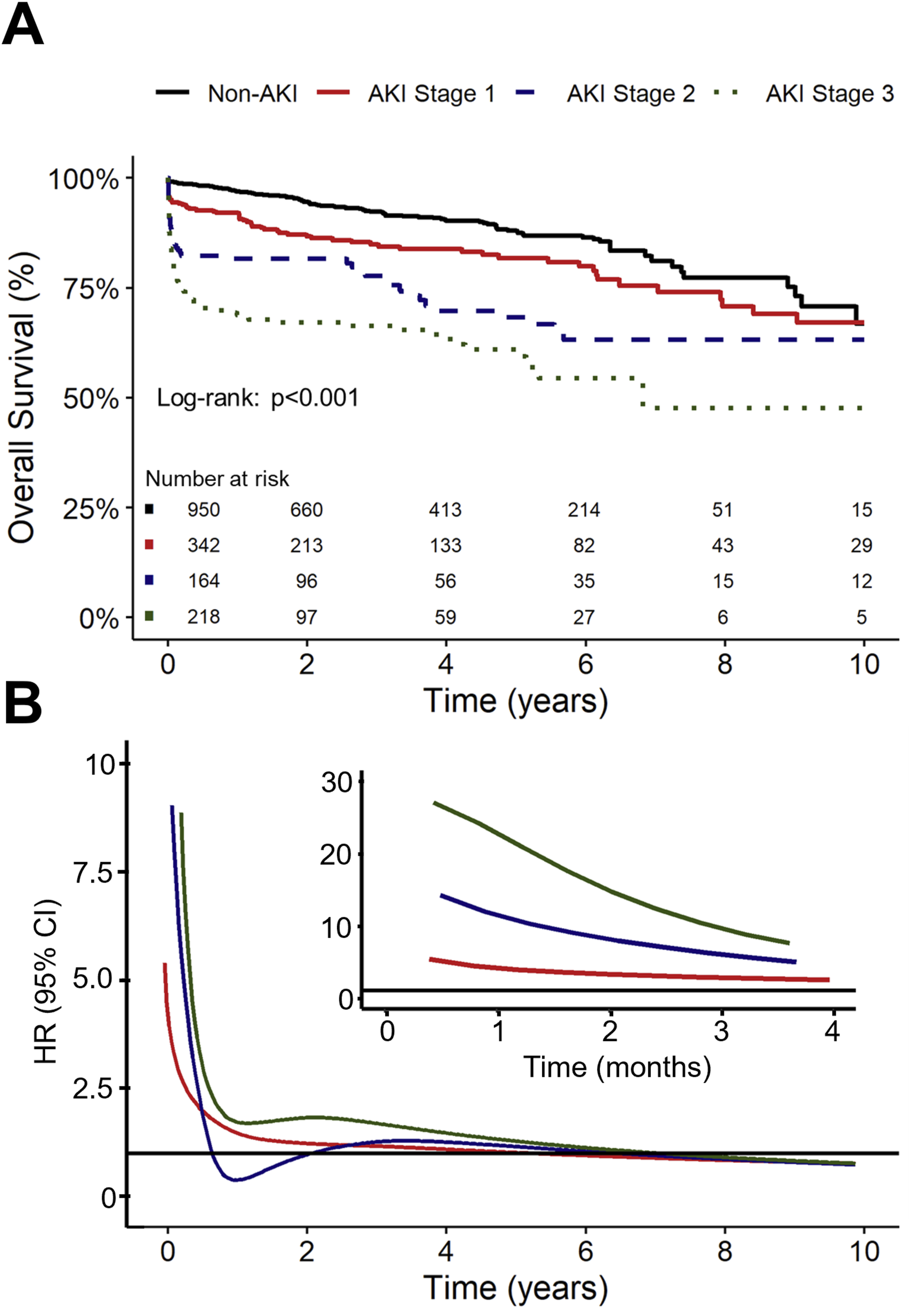
Subgroup and sensitivity analysis
Leave-one-out resampling was performed to assess the robustness of the overall survival findings. Across all, the HR remained consistently elevated, ranging from 2.78 to 3.26, with all 95% confidence intervals well above 1 and p-values <0.001. No single study disproportionately influenced the pooled estimate, confirming the stability and reliability of the association between postoperative AKI and long-term mortality. Then, pre-specified random-effects meta-regression analyses were conducted to evaluate the influence of study-level covariates on mortality. Most covariates, including age, BMI, diabetes mellitus, peripheral artery disease, COPD, smoking status, Marfan syndrome, time to intervention, and operative times (CPB, cross-clamp, and circulatory arrest) were not significantly associated with the hazard of death (all p > 0.05) (Supplemental Table 4).
In contrast, hypertension was associated with a 3% increase in hazard per 1% increase in prevalence (β = 0.03, SE = 0.01, p = 0.03), while a history of cerebrovascular accident was associated with a 13% higher hazard per 1% increase in prevalence (β = 0.13, SE = 0.05, p = 0.006). Then, female sex showed a trend towards a positive association, with a non-significant 3.3% higher hazard per 1% increase in female representation (β = 0.033, SE = 0.019, p = 0.081). Residual heterogeneity remained substantial after adjustment, indicating that these factors only partially accounted for between-study variation. Lastly, no evidence of publication bias was detected, with Begg-Egger tests (p = 0.240 and p = 0.460).
Discussion
In this large meta-analysis including over 6,600 patients undergoing surgery for TAAD, postoperative AKI was common, affecting approximately one-third of patients. We observed that AKI was associated with a three-fold higher risk of death compared with those without AKI, with the excess risk being most pronounced in the first three postoperative months. Yet, the increased risk persisted up to 10 years. As expected, survival declined progressively with increasing AKI severity, with stage 3 AKI conferring a more than five-fold increase in mortality risk. RMST analysis demonstrated a clinically significant loss of 2 years of life expectancy associated with AKI over the 10-years time period. Finally, these findings were robust across sensitivity analyses, consistent between one- and two-stage meta-analytic approaches, and independent of major baseline covariates.
Our findings corroborate and extend prior single-center and registry-based studies (e.g., IRAD, NORCAAD) that have reported AKI incidence rates ranging from 23% to 55% after TAAD repair and consistently linked AKI to worse outcomes. Specifically, patients with AKI experienced higher rates of in-hospital mortality, sepsis, prolonged ventilation, and longer length of stay.3,6 We confirm that the adverse prognostic impact of AKI extends well beyond hospital discharge, with prior studies showing reduced 5- and 10-years survival after TAAAD repair, and survival decreasing progressively with increasing AKI severity.6,13 Although we observed similar trends in our cohort, it is important to emphasize that they do not necessarily imply causality, as AKI can also result from postoperative complications rather than predict them. Although the precise mechanisms linking postoperative AKI to long-term outcomes remain incompletely understood, the pathophysiology of AKI in the context of acute TAAD is likely multifactorial, including perioperative hypoperfusion, systemic inflammatory response, ischemia-reperfusion injury, and nephrotoxic exposures likely all play contributory roles.14–16 For instance, preoperative renal malperfusion is a strong predictor of postoperative AKI (3.5-fold increase in our study and others), 6 yet interestingly on the other end of the spectrum, our pooled analysis also found that approximately one-third of patients with malperfusion do not go on to develop AKI, suggesting additional patient- and procedure-specific factors modulate renal susceptibility and outcomes. While dynamic malperfusion often improves after restoration of true lumen flow following TAAD repair, patients with persistent renal artery malperfusion may be more likely to develop AKI and benefit from targeted intervention. 3
In addition, patients who developed AKI underwent more extensive aortic reconstruction, including total arch replacement, and experienced longer CPB, cross-clamp, and circulatory arrest times, all of which factors known to contribute to renal injury.15–17 In our study, meta-regression showed that CPB, cross-clamp, and circulatory arrest times did not significantly modulate the long-term mortality risk associated with AKI (Supplemental Table 4), suggesting that the adverse impact of AKI reflects more than CPB exposure alone and may indicate a frailer or more vulnerable patient phenotype. Regardless, in our cohort, most baseline characteristics were similar between AKI and non-AKI patients, suggesting that modifiable intraoperative and postoperative management factors may play a substantial role in AKI development. 18 This contrasts with some prior studies in which most risk factors for AKI at ATAAD presentation were considered nonmodifiable, such as advanced age, obesity, and local or generalized malperfusion due to the dissection itself. 6 For instance, in this context, an aortic balloon occlusion (ABO) technique during TAR with frozen elephant trunk appears promising, as it shortens circulatory arrest time, reduces transfusion requirements, and is associated with a lower incidence of postoperative AKI. 19
Then, evidence regarding sex differences in postoperative AKI after ATAAD surgery remains conflicting.20,21 Some studies report higher AKI risk in women when using creatinine-based definitions, whereas others show lower risk when using eGFR, suggesting that true differences in AKI occurrence may be minimal and heavily dependent on the definition used.20,21 However, in our meta-regression, female sex showed a nonsignificant trend toward higher long-term mortality, with a 3.3% increase in hazard per 1% rise in female representation. This indicates that while AKI rates may be similar between sexes, there could still be sex-related differences in late outcomes after acute TAAD. 22 However, it is important to recognize that findings across studies remain heterogeneous, with some reporting no significant differences in baseline characteristics between AKI and non-AKI groups. 1
From a clinical standpoint, these findings highlight several opportunities for intervention. Perioperative renal-protective strategies, including minimizing CPB and circulatory arrest times, avoiding excessive hemodilution, maintaining adequate perfusion pressures, and judicious use of blood products, may mitigate AKI risk. 2 In addition, hypothermia may be beneficial in these patients prior to lower body circulatory arrest to decrease the renal metabolic demand. In the context of general cardiac surgery, not limited to acute TAAD, recent studies have emphasized the potential of pharmacologic interventions to prevent postoperative AKI. Intravenous amino acids (AAs) reliably recruit renal functional reserve and improve kidney function, a finding confirmed by the PROTECTION trial.23,24 The recent DEFEAT-AKI Randomized Clinical Trial demonstrated that prophylactic administration of IV deferoxamine did not reduce the occurrence of AKI. 25 In contrast, the ALBICS AKI trial showed that postoperative infusion of 20% hyperoncotic albumin in high-risk cardiac surgery patients even increased the risk of AKI. 26 Additionally, in the SIRAKI02 Randomized Clinical Trial, extracorporeal blood purification (EBP) connected to cardiopulmonary bypass circuits has demonstrated a significant reduction in AKI incidence compared with standard care (28.4% vs 39.7%), further expanding the toolkit for renal protection. 27 These studies underscore that perioperative renal protection remains a critical area of research, with growing emphasis on both procedural strategies, such as minimizing CPB and circulatory arrest times, and pharmacologic approaches aimed at preserving renal function. Early identification and management of malperfusion syndromes before or during surgery complement these strategies to reduce downstream renal injury. 28 Furthermore, our findings underscore a clinical dilemma: while more extensive arch surgery may offer potential long-term benefits, less aggressive approaches, such as hemiarch replacement, 29 may be safer for patients at high risk of AKI, especially when total arch replacement is not clearly indicated. 3 Beyond the perioperative period, survivors of AKI should be considered a high-risk subgroup warranting structured follow-up for both renal and cardiovascular health, as AKI is known to accelerate chronic kidney disease (CKD) progression and may exacerbate cardiovascular risk through sustained inflammation and neurohormonal dysregulation.14,16,17,30
Limitations
This study has several limitations. First, all included studies were observational, which introduces the potential for residual confounding and selection bias despite multivariable adjustments. AKI definitions varied somewhat between cohorts, though most used KDIGO or RIFLE criteria. Then, we were unable to assess important renal-specific outcomes such as CKD progression, long-term renal replacement therapy requirement, or renal recovery, which may mediate the observed mortality differences. Finally, there was substantial heterogeneity across studies, reflecting differences in operative techniques, perioperative management, and population demographics over three decades. For instance, some of the included studies, such as the IRAD dataset, had limited follow-up beyond 5 years, 3 and others included selective surgical populations, such as patients undergoing hemiarch or TAR at higher-than-average rates, which may limit the generalizability of our findings to all patients undergoing standard TAAD repair. 8 In addition, several procedural and patient-level factors that may influence AKI risk, such as cooling strategy, extent of proximal repair, cross-clamp versus hypothermic circulatory arrest, and perfusion technique, were inconsistently reported or available in only a few studies, limiting our ability to evaluate their impact. Future studies systematically reporting these variables, along with long-term renal outcomes, would help identify modifiable risk factors and enhance the clinical utility of pooled analyses. Nonetheless, our findings were robust across sensitivity and meta-regression analyses, suggesting that the association between AKI and mortality is consistent and clinically meaningful.
Conclusions
Postoperative AKI occurs in roughly one-third of patients after Type A aortic dissection repair and is associated with a three-fold higher risk of long-term mortality, particularly within the first 3 months. Risk increases with AKI severity, and operative factors such as total arch replacement and longer CPB times contribute to its development. These findings emphasize the need for strategies to prevent AKI and improve long-term survival.
Supplemental material
Supplemental material - The impact of postoperative acute kidney injury on survival after surgery for type A aortic dissection repair: Results from over 6000 patients
Supplemental material for The impact of postoperative acute kidney injury on survival after surgery for type A aortic dissection repair: Results from over 6000 patients by Xander Jacquemyn, Ganduboina Rohit, Michel Pompeu Sá, Irsa Hasan, Takuya Ogami, Derek Serna-Gallegos, Bart Meuris, Tom Verbelen, Peter Verbrugghe, Filip Rega, Ibrahim Sultan in Perfusion
Footnotes
Author note
Presented at the 39th EACTS Annual Meeting. 8 – 11 October 2025, Copenhagen, Denmark.
Ethical considerations
Ethical approval was not required for this study as it is a study-level meta-analysis based exclusively on previously published data and did not involve individual participant data.
Consent to participate
Consequently, informed consent was not required.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: IS receives institutional funding and consults for Abbott, Artivion, Atricure, Edwards Lifesciences, Medtronic and Terumo Aortic. PV is consultant or receives honorary speaking fees from Artivion, Atricure, Edwards Life sciences, Medtronic and Corcym. None related to the manuscript.
Data Availability Statement
The data underlying this article will be shared on reasonable request to the corresponding author.
Supplemental material
Supplemental Material for this article is available online.
