Abstract
A type B dissection involves the aorta distal to the subclavian artery, and accounts for 25–40% of aortic dissections. Approximately 75% of these are uncomplicated with no malperfusion or ischemia. Multiple consensus statements recommend thoracic endovascular aortic repair (TEVAR) as the treatment of choice for acute complicated type B aortic dissections, while uncomplicated type B dissections are traditionally treated with medical management alone, including strict blood pressure control, as open repairs have a prohibitively high morbidity of up to 31%. However, with medical treatment alone, the morbidity, including aneurysm degeneration of the affected segment, is 30%, and mortality is 10% over 5 years. For both chronic and acute uncomplicated type B aortic dissections, emerging evidence supports the use of both best medical therapy and TEVAR. This paper reviews the current diagnosis and treatment of uncomplicated type B aortic dissections.
Keywords
Introduction
An aortic dissection occurs when an intimal tear leads to blood flow in a false lumen between the intimal and medial layers of the aortic wall. A type B dissection involves the aorta distal to the subclavian artery and accounts for 25–40% of aortic dissections. 1 Traditionally, the acute phase of a dissection has been defined as being within 14 days of symptom onset. This was based on an early autopsy series of both type A and B aortic dissections demonstrating that up to 74% of the associated mortality occurred within the first 2 weeks. 2 A more recent study of over 1000 type A and B dissection patients from Tokyo showed that there was an 11.7% mortality rate, and that 93% of deaths from dissection occurred within the first 24 hours, making control of the dissection during that period crucial. 3
Historically, the incidence of acute aortic dissections was 3 in 100,000 people per year. 4 However, a more recent review of over 30,000 patients in Sweden demonstrated that the incidence was much higher than previously thought, at 15 in 100,000 people per year. 5 The majority of type B dissections are associated with hypertension. 6 The goal of treatment is to decrease the pressure on the aortic wall, or impulse force, and decrease propagation of the false lumen. If the false lumen continues to propagate, it can lead to a complicated aortic dissection with visceral ischemia, paraplegia, and/or renal failure. Approximately 25% of type B dissections are complicated (with patients having rupture of the aorta or branch vessels, malperfusion of the viscera or extremities, aneurysmal degeneration, rapid aortic expansion/dilatation, uncontrolled hypertension on adequate medical therapy, persistent pain attributed to the dissection) on presentation and warrant urgent intervention. 7 The standard of care for uncomplicated type B dissections has been medical management with strict blood pressure control, as historically open repairs had a prohibitively high morbidity of up to 31%.8–10 However, with medical treatment alone, the morbidity, including aneurysm degeneration of the affected segment, is 30%, and mortality is 10% over 5 years.11,12 Multiple consensus statements recommend thoracic endovascular aortic repair (TEVAR) as the treatment of choice for acute complicated type B aortic dissection.3,12–16 Thus, as minimally invasive techniques have improved and mortality from aortic procedures has declined, there is new interest in the benefits of surgical therapy for the treatment of uncomplicated type B aortic dissection.
History
The first paper to introduce medical therapy for the treatment of aortic dissections was in 1965 by Wheat and colleagues. 17 The paper included two patients with type A dissections and four patients with type B dissections and showed a 100% survival rate with aggressive management of their hypertension. A follow-up paper by Daily and colleagues at Stanford evaluated 35 patients with aortic dissection, of which 30 were acute dissections. 18 In 23 patients the ascending aorta was found to be involved, while in the remaining 12 the dissection was limited to the descending aorta. The in-hospital mortality for dissection of the ascending aorta treated medically was 67% versus 28% for those treated surgically. On the other hand, the mortality for dissection limited to the descending aorta was not different for medical and surgical treatment at 20% and 28%, respectively. This led to the Stanford classifications for aortic dissections where Stanford A includes any dissection involving the ascending aorta and Stanford B includes dissections involving only the descending aorta. Daily proposed that type A dissections should be managed surgically and type B dissections should be managed medically. 18 The results of the Daily paper led to a paradigm shift in the treatment of descending aortic dissections that is the basis of the treatment algorithm we use today.
After the Daily paper, there were multiple large database studies showing the successful results of medical management of type B aortic dissections. A Yale study that included 100 consecutive patients with type B dissections from 1988 to 1998 demonstrated a 90% survival with initial medical management, with almost 60% of patients never needing surgery. 19 A Stanford study of 189 type B dissection patients treated from 1963 to 1999 showed decreased early mortality with medical compared to surgical therapy. The study showed that even with a decrease in surgical mortality over time with improvement in surgical techniques, medical therapy had a 15% mortality compared to a 27% mortality with surgical management. 10 Further insight into the management of aortic disease came with the development of the International Registry of Acute Aortic Dissection (IRAD) in 1996. It is a registry that includes data from 30 referral centers in 11 countries and at this time includes over 3800 acute aortic dissection cases. For inclusion into the registry, questionnaires are completed by the referring centers regarding 290 variables that deal with demographics, history, physical findings and outcomes. 6 The studies coming out of IRAD guide much of our current management of aortic dissections.
Diagnosis of type B acute aortic dissection
In a 2015 study from the IRAD that included 1476 patients with type B dissections, presenting symptoms included severe pain (94%), chest pain (71%), back pain (70%), hypertension (66%), and syncope (3%). 20 Laboratory tests in patients admitted with chest pain and the suspicion of aortic dissection should include complete blood count (to evaluate for blood loss/anemia), troponin (to rule out myocardial infarction), creatine kinase (to evaluate for reperfusion injury, rhabdomyolysis), creatinine (to evaluate for developing renal failure), AST/ALT (to evaluate for liver ischemia/disease), and lactate (to evaluate for bowel ischemia). 21
Historically, chest X-rays were used for diagnosis of aortic dissection, with findings including an abnormal aortic contour, mediastinal widening and/or separation of the aortic wall, and intimal calcification from the outer margin of the aorta in up to 85%. 22 However, the sensitivity of a spiral computed tomography (CT) angiogram of the chest, abdomen and pelvis for detection of aortic dissection is 97%, and the specificity is 100%, and it is now the gold standard for diagnosis of aortic dissection. 23 The key finding on CT is the intimal flap separating two lumens. Furthermore, it can delineate the extent of the dissection, the involvement of aortic branch vessels and suitability for medical management versus endovascular or open repair. 21 In patients with a contrast allergy or renal insufficiency, a magnetic resonance angiogram (MRA) or transesophageal echocardiogram (TEE) can be performed. In a systematic review and meta-analysis of 16 studies including 1139 patients, CT, MRA and TEE were found to have similar pooled sensitivity (98–100%) and specificity (95–98%) for diagnosis of type A and B aortic dissections combined. 24 For diagnosis of type B aortic dissections, a 2002 IRAD study showed sensitivities of 100% for MRA, 93% for CT, 89% for aortography and 80% for TEE. 25 Despite the lower sensitivity of TEE, it is the second line imaging modality after CT as it is faster and easier to obtain in emergency situations than an MRI. 25
Acute versus chronic aortic dissection
The distinction between acute and chronic aortic dissection is important because when the dissection becomes chronic, although aortic remodeling continues to occur, the morbidity, mortality and need for intervention decline significantly. 26 The definition of an acute aortic dissection is evolving. Historically, acute dissection was defined as being <14 days from symptom onset and chronic dissection was defined as >14 days. 27 Most studies evaluating treatments for acute dissections are based on this definition. A recent European expert multidisciplinary panel suggested that ‘acute’ should refer to <2 weeks, ‘subacute’ from 2 to 6 weeks, and ‘chronic’ >6 weeks from symptom onset. 7 IRAD investigators recently published a new classification where a dissection is not considered chronic until >30 days after symptom onset, based on an analysis of survival curves demonstrating that survival decreases significantly up to 30 days after presentation. 28
Treatment of acute aortic dissection
Open aortic repair
Based on data from the papers by Wheat in 1965 and Daily in 1970, there was a paradigm shift away from surgical repair and toward medical management with aggressive blood pressure control for type B aortic aneurysms.17,18 Subsequent large studies out of Yale, Stanford and IRAD have shown decreased morbidity and mortality with medical therapy compared to open surgical repair.6,10,13,19 Based on this, open surgery for acute uncomplicated type B aortic dissection has been nearly abandoned.
Medical and endovascular therapy
Based on the data out of Yale, Stanford, IRAD and multiple smaller studies, there are numerous consensus statements recommending only best medical therapy (BMT) for acute uncomplicated type B aortic dissection.3,12–16 The aim of medical therapy in acute aortic dissections is to reduce the blood pressure to 100–120 mmHg systolic and reduce the heart rate to less than 60 beats per minute. The preferred medication is selective beta blockade, which lowers blood pressure by decreasing the force of left ventricular ejection. In refractory cases, diuretics, angiotensin converting enzyme (ACE) inhibitors, calcium channel antagonists, and alpha blockade may be required.12,14
Although the short-term outcomes with medical therapy have been shown to be superior to open surgery, the long-term complications are not trivial. The 1, 5 and 10-year mortality rates for patients treated by BMT alone are 10%, 20% and 30%, respectively. The 1-year false lumen thrombosis with medical therapy alone is 35%. Additionally, at 4 years there is a 25–50% rate of aneurysmal degeneration of the aorta, and by 6 years up to 59% of patients have undergone a surgical intervention.7,29,30 As surgical therapy outcomes have improved, these long-term complications have led to more interest in endovascular management of uncomplicated type B aortic dissections.
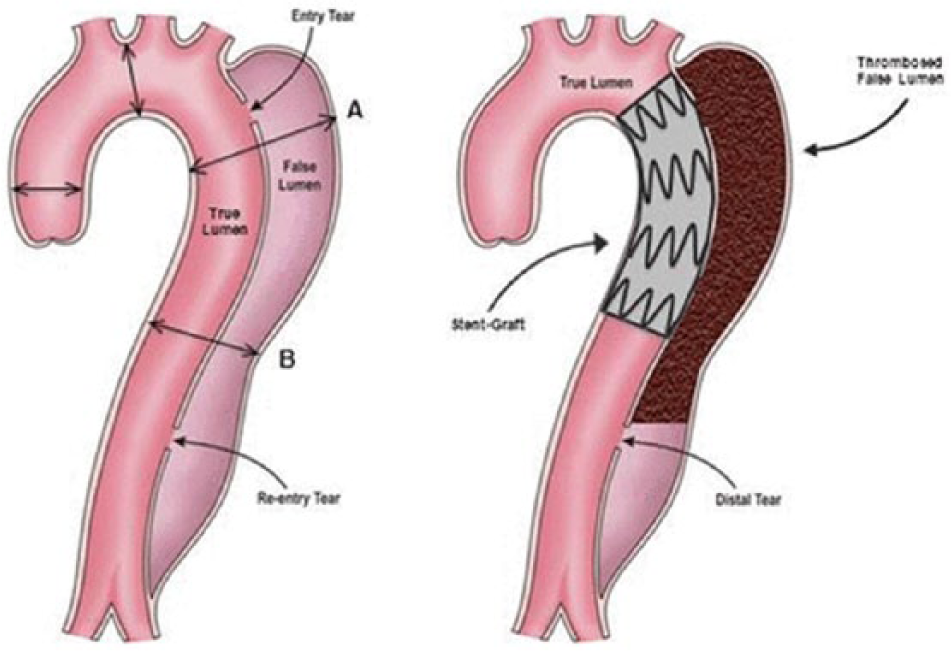
The rationale for endovascular therapy for the management of type B dissections is to close the primary entry tear, thereby leading to decreased pressure in the false lumen and ultimately false lumen thrombosis (Figure 1). 31 The first published use of an endovascular aortic stent graft for management of type B dissections was in 1999. 32 In 15 patients with complicated type B dissections and four patients with type A dissections, the 13-month mortality was 16% and morbidity was 21%. Complete thrombosis of the false lumen was shown in 79% of patients. These results were verified in subsequent larger, physician-sponsored investigational device exemption (IDE) clinical trials evaluating the use of TEVAR in patients with complicated type B aortic dissections. 33 A 2010 meta-analysis comparing open and endovascular repair for acute complicated type B dissection in 76 observational studies showed decreased mortality, paraplegia and vascular complications for TEVAR compared to open repair. 34 Additionally, in a 2013 study from the IRAD investigators of 1129 patients with both complicated and uncomplicated type B aortic dissection, TEVAR was associated with a lower 5-year mortality than medical therapy. 35 However, there were limited data evaluating TEVAR to BMT for uncomplicated aortic dissections.
Typical features of a type B dissection with an entry and re-entry tear with flow in both the true and false lumen (A). With TEVAR, a stent is placed over the major proximal entry tear, reconstructing the true lumen with subsequent thrombosis of the false lumen (B). Reproduced with permission. 39 TEVAR, thoracic endovascular aortic repair.
Most studies evaluating TEVAR for uncomplicated type B aortic dissection are uncontrolled prospective or retrospective cohorts or case series. Additionally, the timing of intervention after dissection onset and complications are not uniformly reported and defined. 7 The first randomized control trial (RCT) comparing BMT versus BMT plus TEVAR for uncomplicated type B dissections was the INSTEAD trial. It enrolled 140 patients with chronic dissections where TEVAR was performed between 2 and 52 weeks after symptom onset (clustering at 10–12 weeks). It demonstrated no improvement in short-term outcomes with TEVAR. 26 However, at the 5-year follow up, the aorta-specific mortality was lower with TEVAR (6.9% vs 19.3%, p = 0.04), and progression of aortic disease was lower with TEVAR (27.0% vs 46.1%, p = 0.04). However, there is no RCT evaluating outcomes for BMT versus BMT plus TEVAR for acute uncomplicated type B dissections. There is currently an ongoing study from the Society of Vascular Surgery Patient Safety Organization (SVS PSO) TEVAR Dissection Project to evaluate effectiveness of TEVAR to treat acute and chronic descending aortic dissections. This 5-year project will follow at least 200 patients with acute dissections treated with TEVAR and 500 chronic dissection patients treated with TEVAR. 36 The results of this study will likely guide much of the future management of both acute and chronic uncomplicated aortic dissections.
Anatomic considerations for treatment
Prediction of which patients are more likely to progress to a complicated dissection or to aneurysmal degeneration could potentially differentiate which patients should be treated with open or endovascular surgical intervention from those who will do well with medical therapy alone.
Association of progression with location of initial tear
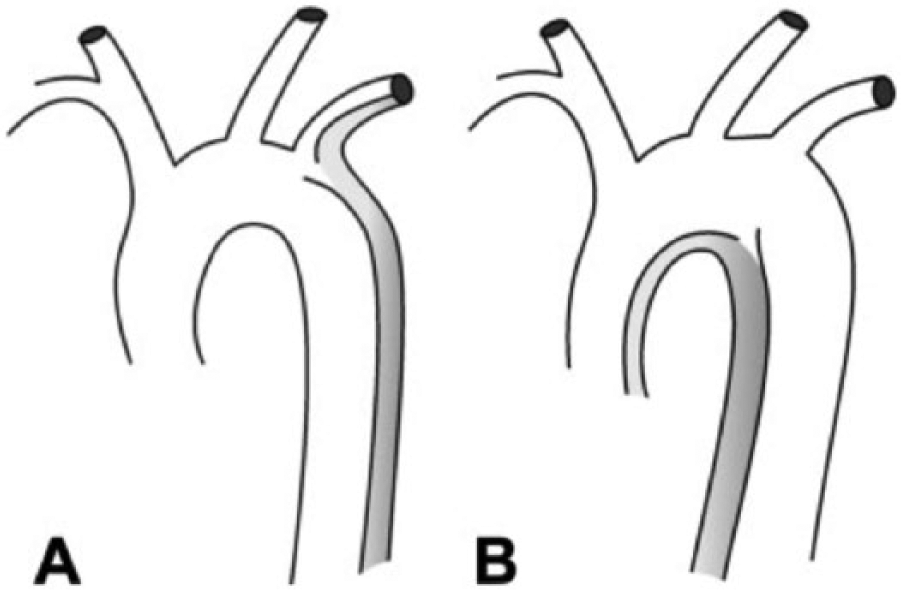
Two recent publications found that for patients with acute type B aortic dissection, those with the primary entry tear located on the concavity (undersurface) of the distal aortic arch more frequently had complicated dissections either at presentation or during initial hospitalization, had retrograde extension of the dissection process, and required intervention for management. The authors theorized that the reason for the higher incidence of retrograde extension in the concavity group is the lack of an anatomic barrier, the arch vessels, on the undersurface of the aortic arch.27,37 In these studies, a Cox regression analysis demonstrated the location of the primary tear on the concavity of the distal arch to be the sole independent predictor of developing a complicated dissection. The authors suggested a modified terminology where a primary entry tear on the convexity is referred to as B1 and a primary tear on the concavity as B2 (Figure 2). They also concluded that localization of the primary tear should be implemented in risk stratification of acute type B aortic dissection. 38
Acute type B dissection with a primary tear on the convexity of the aortic arch (left) and with a primary tear on the concavity (right). For tears on the convexity/outer circumference of the arch, the arch vessels provide an anatomic barrier limiting retrograde extension of the dissection process. The authors describing this relationship suggested a modified terminology whereby a primary entry tear on the convexity is referred to as B1 and a primary tear on the concavity as B2. Reproduced with permission.27,32
Another predictor of higher dissection-related morbidity and mortality is the size of the original aortic tear. A recent study by Evangelista and colleagues found that a large (⩾10 mm) entry tear in the proximal part of the dissection identified a high-risk subgroup of patients with more rapid aortic expansion and a higher incidence of dissection-related events than those with tears <10 mm. Furthermore, in a large number of patients, at least 3 years of follow up was required before dissection complications appeared. 39 It was theorized that the reason for the higher complication rate with larger tears was due to the higher false lumen pressures with larger tears. A follow-up study was performed by the same group using in vitro phantoms to evaluate the hemodynamics of the false lumen. The study found that, in the presence of multiple intimomedial tears, flow enters and leaves the false lumen through all tears simultaneously, with minimal proximal-to-distal (or vice versa) flow in the false lumen. As such, false lumen hemodynamics depend heavily on the cumulative tear size as well as the size of the primary entry tear. 40
Difference in aneurysmal degeneration of the aorta based on treatment
In a study of 1129 patients with type B dissection (both complicated and uncomplicated) from IRAD that compared endovascular therapy versus BMT alone, at 1 year, the maximum diameter of the descending aorta was the same for both groups. 26 However, at 5 years it was decreased in the endovascular group compared to the BMT group.7,31 The INSTEAD trial found no difference in the maximum diameter at up to 2 years, but did find that false lumen thrombosis was 91% for TEVAR versus 19% for BMT. 26 The INSTEAD XL trial found that, at 5 years, the maximum aortic diameter was smaller for TEVAR plus BMT (44.5 ± 11.5 mm) than for BMT alone (56.4 ± 6.8). 31
The ADSORB trial, the first RCT comparing BMT versus BMT plus TEVAR for acute uncomplicated type B aortic dissections, was aimed specifically at evaluation of the post intervention aortic morphology. The study was performed across 17 European centers and randomized 61 patients (31 BMT; 30 BMT + TEVAR). The primary study end point is a composite of incomplete or no false lumen thrombosis, aortic dilatation (⩾5 mm or maximum aortic diameter ⩾55 mm), and descending thoracic or abdominal aortic rupture through the 1-year follow-up visit.27,41 Greater freedom from the composite end point in the BMT plus TEVAR group was found compared with the BMT alone group (57% vs 3%; p < 0.001).27,42 Interestingly, this difference appears to be driven almost entirely by false lumen thrombosis, as no difference was seen in the aortic dilatation and rupture components of the composite end point. Secondary aortic remodeling end points also favored the BMT plus TEVAR group, with a larger true lumen and smaller false lumen, and a trend toward smaller overall aortic diameter when compared to BMT alone.27,42
Surveillance and follow up
Patients who undergo TEVAR for aortic dissection should undergo a CT or MRI at 6 months and then annual CT or MRI for ongoing surveillance. They should also continue on long-term anti-hypertensive medication, preferably a beta-blocker or ACE inhibitor, with a goal blood pressure of <140/90 mmHg. 21
Conclusions
Type B aortic dissections are more common than previously thought. If a dissection is complicated, there is ample evidence that emergent TEVAR is the first line choice of treatment. However, in uncomplicated type B aortic dissection, the management is less clear. For both chronic and acute uncomplicated aortic dissections, emerging evidence supports the use of both BMT and TEVAR in select patient populations. A proposed management algorithm is in Figure 3. Further randomized studies comparing medical and surgical treatment are needed for both acute and chronic dissections. Open surgery for acute type B aortic dissection should not be performed unless the patient has ruptured and has unfavorable anatomy for TEVAR.
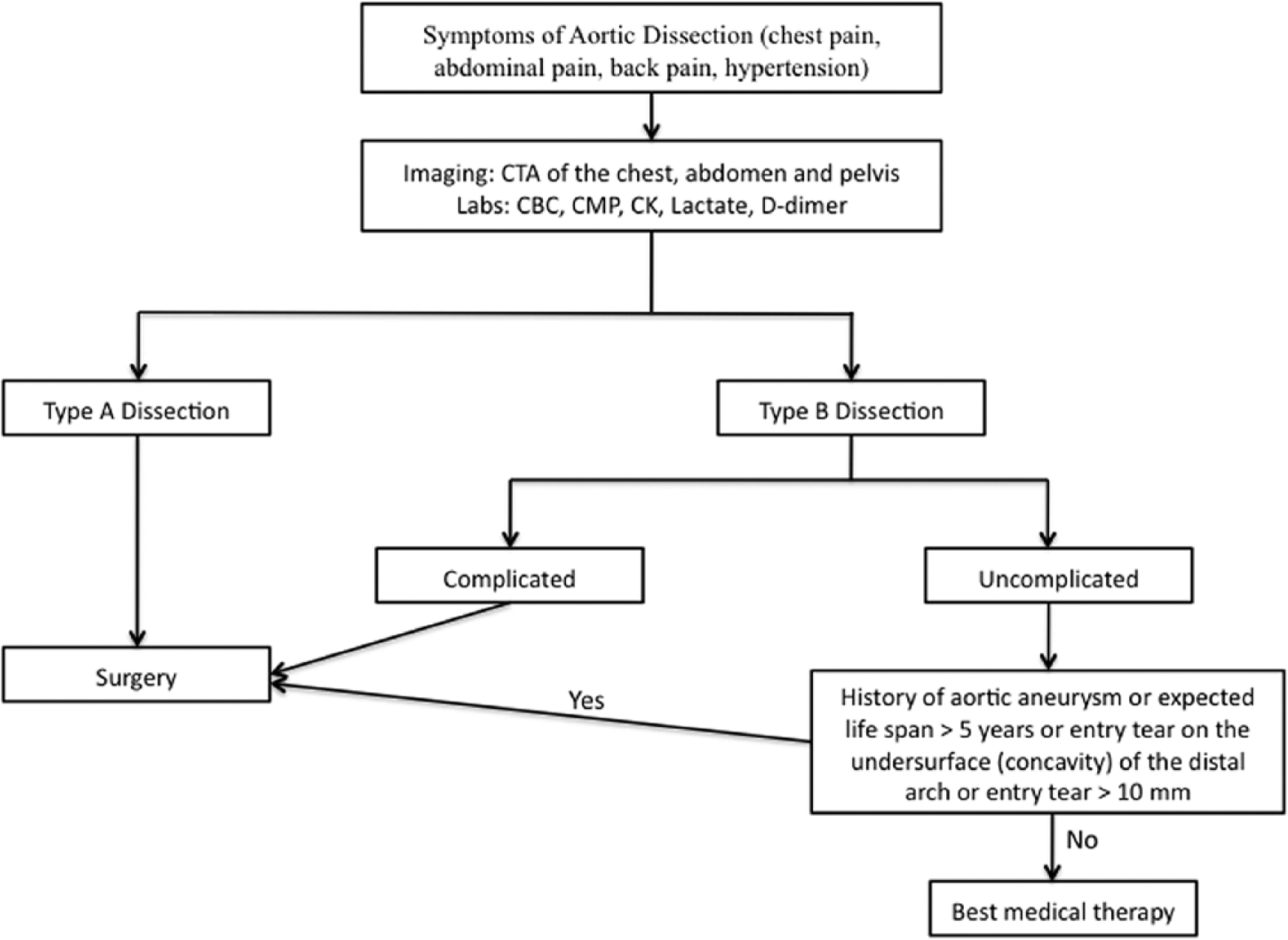
Algorithm for the treatment of acute uncomplicated type B aortic dissections. CTA, computed tomography angiogram; CMP, complete metabolic panel; CK, creatine phosphokinase.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
