Abstract
Objective
This scoping review aimed to examine current literature on the use of point-of-care ultrasound (POCUS) in paediatric patients supported by extracorporeal membrane oxygenation (ECMO). It assessed existing evidence, summarised key findings, explored theoretical limitations, and identified knowledge gaps.
Methods
A comprehensive search of Medline, CINAHL, Embase, and the Cochrane Library was conducted using a strategy developed with an academic librarian. Studies involving patients aged 0–18 years receiving any form of extracorporeal life support were included; in-utero interventions and non POCUS scans were excluded. Data on participants, context, concepts, study design, and outcomes were extracted and presented in tabular and narrative form.
Conclusion
This review highlights the increasing use of POCUS in paediatric ECMO care, despite a limited and heterogeneous evidence base. Physiological differences in ECMO patients challenge the direct application of findings from non-ECMO studies. The absence of standardised protocols and governance frameworks limits consistency and reliability. Developing unified guidelines and strengthening paediatric-specific research are essential to support safe and effective integration of POCUS into clinical practice.
Introduction
Point-of-care ultrasound (POCUS) has emerged as a valuable clinical assessment tool in paediatric intensive care (PIC) as benefits are increasingly recognised in the literature. Ultrasound performed by bedside clinicians has long been used to reduce the complication rate from central venous access but is now routinely used in practice to assess lung, brain, heart and abdominal pathologies. As practitioners become increasingly comfortable and familiar with its use, the range of applications is growing. POCUS negates the requirement for further time-consuming modalities such as chest x-ray, computed tomography (CT) scan or formal echocardiograms, enabling clinicians to make faster decisions and track patient progression.1,2 It is not intended to replace comprehensive diagnostic imaging performed by specialists but rather to answer specific, binary questions. Paediatric extracorporeal membrane oxygenation (ECMO) patients have a significant risk of life-threatening pathology due to their underlying disease and due to iatrogenic complications of ECMO. They require continuous bedside assessment to ensure its efficacy and safety and frequently require imaging. The exposure to ionising radiation of radiographs and computer tomography can be significant and intra-hospital transfers carry inherent risks, including patient instability, logistical challenges, and increased demands on personnel and financial resources.3,4
Common POCUS applications in ECMO patients include lung assessment, cardiac assessment, transcranial doppler (TCD), and percutaneous cannulation. Interpreting the findings of these specific modalities often adds complexity in ECMO-supported patients due to the interaction between ECMO and the patient’s physiology. The objective of this study is to conduct a comprehensive review of the current literature regarding the application of POCUS in patients supported with ECMO and to gain an understandinghow the physiological changes caused by ECMO affect the interpretation of POCUS findings. It aims to identify, characterise and critically appraise published studies, summarise key findings around the common POCUS applications listed above, identify theoretical and practical limitations, and highlight gaps in the literature. Given the exploratory nature of the research question, a scoping review methodology was employed.
Methods
This scoping review was conducted in accordance with the Joanna Briggs Institute methodology 5 and the six-stage framework outlined by Arksey and O’Malley. 6 The study protocol was registered on Open Science Framework (OSF) on 27 November 2023 (https://doi.org/10.17605/OSF.IO/27ZAW).
Search strategy
A comprehensive search strategy was developed in conjunction with a librarian and was applied across MEDLINE, CINAHL, Embase, and the Cochrane Library (Supplemental Table 1). The keywords and index terms, were customised for each database. Grey literature and snowballed documents were also reviewed. Only English language publications were considered.
Study selection
All identified citations were collated and uploaded into RefWorks. All titles and abstracts were screened by two independent reviewers to assess their eligibility against the inclusion criteria. The full-text of potentially relevant articles were assessed for inclusion. Borderline articles were discussed between the reviewers, this resolved all differences of opinion. The selection process was reported in full using a Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) flow diagram (Figure 1).
7
The PRISMA-ScR checklist has been provided as Supplemental Table 3. PRISMA flow diagram.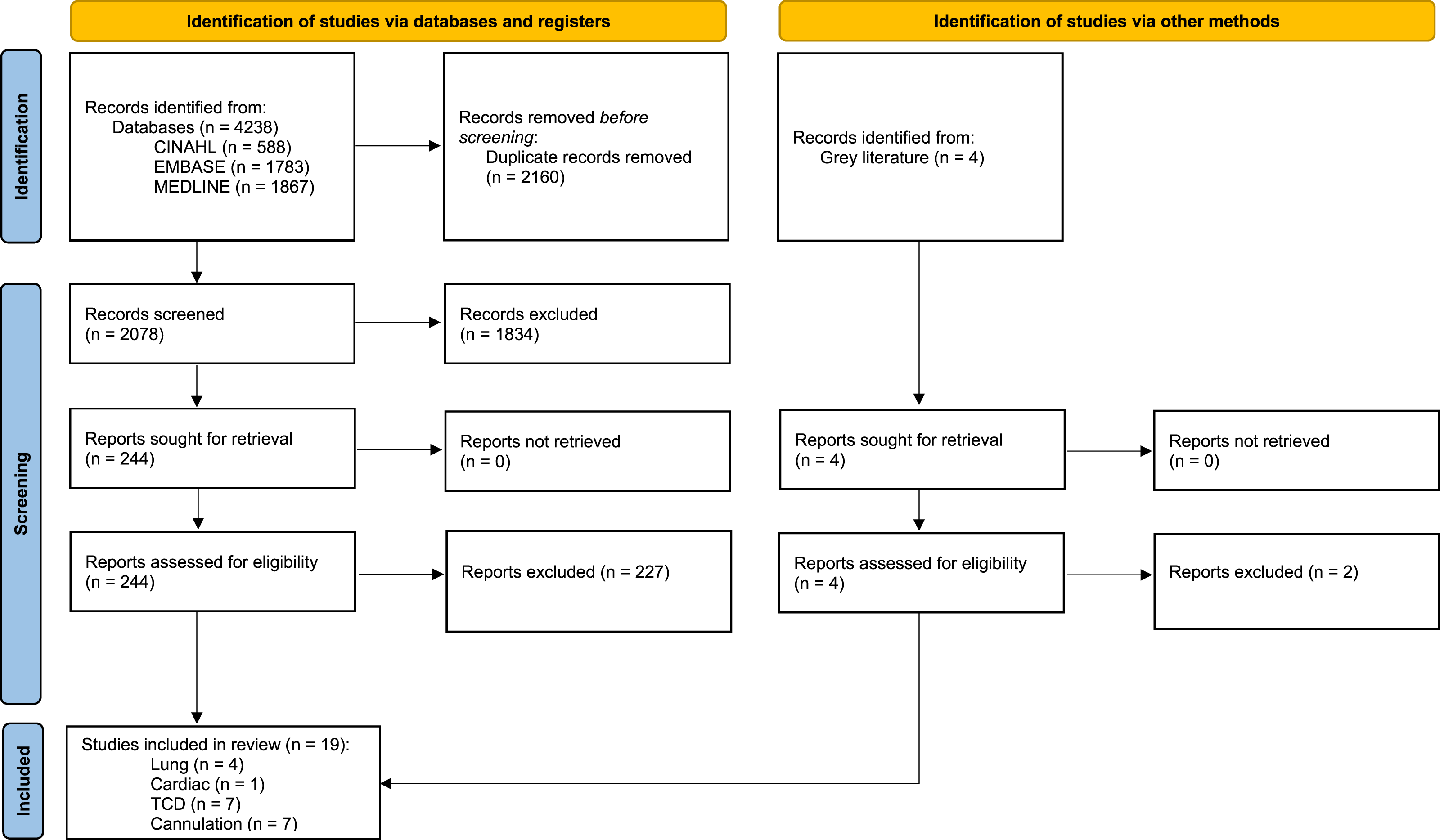
Inclusion criteria (Population, concept, and context)
Preliminary database searching revealed limited evidence for POCUS on ECMO prior to 1990. Studies published from 1990 (when ECMO and POCUS technologies became widely adopted due to advancements in technology) until the search (25 January 2024) were considered. Studies were included if they focused on children from birth up to 18 years of age. Studies that included a mixture of paediatric and adult patients were included if the findings were relevant to the paediatric population. Prenatal studies were excluded. We included only studies describing the use of POCUS performed by the treating clinician (e.g. intensivists, advanced practitioners, physiotherapists), explicitly excluding those conducted by clinicians with specific training in imaging (radiologists, sonographers, or cardiologists).
Data extraction
Data from the included studies were extracted by two independent reviewers using a standardised data extraction tool (Supplemental Table 2). The data extracted included details about each study’s participants, concept, context, methods, and key findings relevant to the review question. In cases where clarification or additional data were required, the authors of the original studies were contacted. Findings were categorised and discussed by common POCUS applications with altered interpretation in this population: lung assessment, cardiac assessment, transcranial Doppler (TCD), and percutaneous cannulation. Given the nature of a scoping review, the quality of included studies was not formally assessed.
Results & discussion
Summary of studies.

Cardiac
Only one study met the inclusion criteria. 8 The majority of studies were excluded because the examinations were conducted by a cardiologist or a sonographer. In the only included study, POCUS was used in the context of extracorporeal cardio-pulmonary resuscitation (E-CPR). The primary aim was to investigate reversible cardiac causes, such as tamponade, and to assess for cardiac standstill. The subcostal view was the only window required during resuscitation. All three patients in the study exhibited cardiac standstill during resuscitation, but subsequently regained cardiac contractility with repeat scanning while on ECMO. The authors concluded that POCUS during cardiac arrest should not be used to predict cardiac recovery.
This review did not demonstrate sufficient evidence supporting the use of cardiac POCUS in paediatric ECMO patients. Although previous authors have emphasised the importance of cardiac ultrasound in intensive care and its applications in ECMO patients,9,10 most studies utilised echocardiography rather than POCUS. Physiological changes due to ECMO need to be considered when assessing the traditional components of cardiac POCUS (function, form, filling and fluid). In VA ECMO, the heart is unloaded and the assessment of ventricular function and filling loses sensitivity.11–13 Additionally, the assessment of pulmonary pressures and RV function may be further affected by tricuspid regurgitation caused by the ECMO drainage cannula.12,14 POCUS remains an essential tool for promptly diagnosing pericardial effusion and life-threatening pericardial tamponade.9,15 In clinical practice, cardiac POCUS practitioners are acquiring more advanced skills. This could be further applied to ECMO patients, for example determining patency and shunting through the ductus arteriosus and interatrial septum, crucial in the initial phase of patients with pulmonary hypertension supported by ECMO.16,17
Lung
Four studies met the inclusion criteria, with patients on either VV or VA ECMO.18–21 One study included both adults and children. 20 All paediatric patients had abnormal scans, except in the neonatal study, where persistent pulmonary hypertension (PPHN) was the primary indication for ECMO. 19
Diagnostic adjuvant
The neonatal study found that POCUS was superior to chest X-rays at distinguishing between parenchymal and non-parenchymal causes of PPHN. The authors suggested that this could be used to identify which patients would benefit from a lung biopsy whilst on ECMO. 19
Prognostication and monitoring disease progression
All four studies utilised a system to observe lung aeration and monitor disease progression. Of the four articles reviewed, three employed the adult lung ultrasound scoring system (from 0 to 36).18,20,21 The neonatal study instead classified patients into diagnoses based on ultrasound images. 19 The lung scores were correlated with dynamic lung compliance. The studies that employed a scoring system compared their lung ultrasound score with different measurements of illness severity, including oxygenation index (OI) and lung dynamic compliance (Cdyn).18,20,21 All articles employing a scoring system concluded that these scores could aid in predicting outcomes. Two of the studies (including both children and older adults, primary lung injury and cardiac dysfunction, VV and VA ECMO) found that patients experienced worse outcomes if lung ultrasound scores remained elevated at 72 h of ECMO.20,21 However, in the study looking specifically at paediatric acute respiratory distress syndrome (pARDS), no significant changes were observed in the first 48 h of ECMO. 18 The study investigating patients with cardiogenic shock found that as left ventricular dysfunction persisted in non-survivors, lung scores worsened with prolonged ECMO duration. 20
Evidence outside of ECMO, demonstrates that using a lung scoring system reduces inter-operator variability, enhancing the ability of clinicians to monitor disease progression and to prognosticate.22–26 The underlying disease and cohort characteristics differed significantly between the studies suggesting that this finding may be applicable across multiple pathologies. Observing and quantifying the progression of B-lines (B-lines are generated by increased interstitial fluid and therefore can be used as a marker and differentiator for any condition in which the lung parenchyma have pathophysiological increases of fluid, such as cardiogenic pulmonary oedema or ARDS) and subpleural consolidation may allow clinicians to improve management decisions.27,28 When performed serially, POCUS provides equivalent bedside information to a CT scan.29,30
POCUS can go beyond answering binary questions assisting clinicians in formulating a diagnosis.24,31,32 The review found no evidence for POCUS assessment of pleural effusions but clinically POCUS is frequently employed to evaluate pleural effusions. While this is a valuable tool, there are pitfalls; mathematical formulas for estimating pleural effusion volumes have proven unreliable. 33 When used, these formulas should focus on ensuring a systematic approach to reporting effusion size.28,34 Adult studies have highlighted the role of POCUS in targeted recruitment manoeuvres and in personalising and optimising PEEP levels to enhance safety during these interventions.35,36 POCUS has been shown to facilitate the assessment of proning therapy by evaluating anterior-posterior changes. 37 These applications could be particularly valuable in ECMO patients who are often at increased risk of complications from these manoeuvres.
Transcranial doppler
Transcranial doppler assesses cerebral blood flow in the major intracranial arteries. Cerebral blood flow velocities (CBFV) are measured in terms of peak systolic velocity (PSV/PS), end diastolic velocity (EDV/VD) and mean flow velocities (MFV/VM). These measurements can be used to determine cerebrovascular resistance via calculating indices such as the pulsatility index-PI (PSV-EDV/MFV), and resistive index-RI (PSV-EDV/PSV). Higher values can be indicative of raised intracranial pressure (ICP). 38
Seven studies met the inclusion criteria. Six of these studies39–44 aimed to assess how CBFV were affected by ECMO with only one of these studies including patients on VV ECMO. 40 A further article surveyed the use of TCD across Argentinian PICUs. 45 The methods used in the included articles are heterogenous, reporting different insonation locations (middle cerebral arteries, pericallosal arteries, internal carotid arteries) and different measurements. The first type of measurements describes CBFV in terms of systolic, diastolic, or mean velocities. The second type of measurements (PI and RI) are markers of cerebrovascular resistance. Both types are affected in VA ECMO, through variations in systemic blood flow and pulsatility.
Arteries examined
Three studies performed TCD on the middle cerebral arteries (MCAs).40,41,43 Two studies used the anterior fontanelle of infants to examine the pericallosal arteries.39,42 One insonated both the MCAs and internal carotid arteries via the fontanelle. 43 Another also examined flow velocity in the extracranial internal carotid artery (EICA) to calculate a Lindegaard ratio which compares velocities in the MCA to the velocities in the ipsilateral EICA to assess for vasospasm. 40
Reported measurements
The more recent studies reported systolic (PS), diastolic (VD), and mean flow (VM) velocities, along with the derived PI.40,41,43 Two older studies reported the RI.42,44 One study reported the time-averaged mean (TAM) flow velocity. 39
Alterations to cerebral blood flow velocities & neurological injury
Six studies described changes in CBFV upon initiation of ECMO.39–44 Five of the six studies reported an overall reduction in velocities with ECMO flows up to 150 ml/kg/min39–42,44. Conversely, one found a significant increase in mean velocities 1 hour after ECMO initiation but ECMO flow rates were much higher in this cohort. 43 Manipulation of the ECMO circuit by either closing a secondary cephalic drainage cannula or by opening the ECMO bridge caused a significant drop in CBFV.39,44 RI was reported to drop on higher flows. 42 All the studies show that changes in ECMO flow are associated with changes in CBFV. Non-POCUS research on ECMO and cardiopulmonary bypass patients shows that these are associated with a decrease in CBFV.46,47
Four articles reported neurological injury as an outcome.40,41,43,44 Two studies did not associate changes in TCD velocities with neurological injury.43,44 The remaining two divided their findings into cases of ischaemic stroke (AIS) and haemorrhagic stroke (HS).40,41 An elevated PI was reported in eight children suffering from cerebral ischaemia. 41 This finding is supported by another study in which one patient with global cerebral ischaemia had an elevated PI and globally reduced CBFV 6 days before the injury was clinically detected. All four patients suffering cerebral haemorrhage had significantly increased CBFV. These elevated flow velocities were detected up to 6 days before the bleeds were clinically diagnosed. These patients were hyperaemic, using the Lindegaard ratio. 40 Although these studies suggest that an elevated PI is associated with ischaemia and increased CBFV is associated with haemorrhage, a wider comprehensive systematic review of adult and paediatric patients suggested that CBFV, their clinical significance, and their association with neurological injury are heterogeneous. 48 A benefit of TCD may be in helping clinicians detect neurological injury at an earlier stage.46,49
A unique risk for ECMO patients is the potential for thrombi formation within the circuit, which in VA ECMO may extend or dislodge into cerebral arteries. Microembolic signals detected by TCD were observed in 47% of VA ECMO patients, with over 75% of these patients experiencing thromboembolic neurological events. 50 Our search did not find studies reviewing the significance of microembolic signals in ECMO patients as studies are limited to adults/non-POCUS.
The included survey found 2/20 responding centres utilised POCUS TCD for ECMO patients. 45 Further evidence reflects our finding that TCD is not widely used for neurological assessment in paediatric ECMO patients. Only 28% of European ECMO centres report regular TCD use, compared with just 8% of centres in the United States.51,52 In contrast, 93% of American paediatric neurological intensive care units routinely use TCD, with a significant proportion incorporating TCD findings to guide clinical care, albeit without established standards for its performance, interpretation, or reporting.53,54 Recent practice recommendations advocate for TCD to be performed in paediatric intensive care settings by individuals accredited by their institution. 54
Cannulation
Seven studies met the inclusion criteria. Six studied paediatric cannulation and one included both adult and paediatric populations. 55 Five studies utilised cardiac POCUS or echocardiography to guide cannulation during procedural POCUS.55,57–60 Bicaval dual-lumen cannulas were employed in six studies.55–60 One study focused on cannula positioning using POCUS but excluded this cannula type. 61 One study reported the use of multisite cannulation including femoral cannulation. 58
Vessel sizing
Only the study involving both adults and paediatrics reported the use of POCUS for vessel measurement before cannulation. 58 In cases where the arteries were deemed too small, two arteries were cannulated to allow sufficient flows.
Accessing vessels and positioning cannulae
All studies used POCUS to guide percutaneous cannulation. One article reported cannulation in out-of-hospital E-CPR. 61 All studies reported a POCUS-guided cannulation success rate exceeding 93%. The maximum duration for cannulation was 30 min, while the minimum time recorded was 16 min.59,61 Use of POCUS for percutaneous cannulation is standard practice for both arterial and venous cannulation, regardless of the access site.55,62–66 Most evidence in paediatric patients pertains to percutaneous cannulation via a dual-lumen cannula for VV ECMO, typically using a jugular vein approach.62,67,68,69
Four studies solely used POCUS for positioning.58–61 Three combined POCUS with fluoroscopy.55–57 One study reported that POCUS combined with fluoroscopy reduced the need for cannula repositioning. 56 In out-of-hospital E-CPR scenarios, two clinicians used POCUS to confirm cannula position. 61 Patients with congenital diaphragmatic hernia (CDH) were noted to present the greatest challenge for position confirmation using POCUS during and after cannulation. 60 Consequently, one study excluded this patient group when assessing venous cannula positioning. 56
In the adult literature, POCUS-only approaches have yielded promising outcomes, reducing the need for larger teams or patient transfers to imaging departments.64,65 However, another paper suggests that positioning of cannulae were improved when fluoroscopy is used in addition to POCUS. 70 The reviewed studies indicated that POCUS outperformed chest X-ray in verifying cannula positioning, a finding corroborated by other studies. 71 Further paediatric-specific research is needed to determine if POCUS is comparable to echocardiography. Paediatric patients frequently present with limited or challenging imaging windows, where sonographers or cardiologists trained in locating unconventional views may provide enhanced accuracy. 72 POCUS-trained specialists can develop these skills to address similar challenges. Utilising POCUS to confirm cannula positioning is critical for promptly identifying misplaced cannulas and complications such as cardiac tamponade or hepatic venous outflow obstruction.15,73,74
Limitations
Our review was limited by a paucity of evidence, and it was challenging to draw conclusions due to the heterogeneity and design of the included articles in each section, both in their results and their methodology (including the evolution of ultrasound technology over time). Most of the studies included were observational, small, single-centre studies which limits generalisability of findings, especially considering how performance of POCUS is dependent on operator experience. We chose very specific inclusion criteria, so techniques such as cranial ultrasound and measuring of the optic nerve sheath diameter whilst useful for the detection of intracranial pathology, were excluded since these measurements are not affected in the same way as TCD by the ECMO circuit. Likewise, the ability of POCUS to reduce procedural complications (arterial and venous access, chest drains) has been well studied and offers great benefit to ECMO patients, but this is not a benefit or use that is affected by ECMO. 75
Conclusion
This scoping review highlights the challenges encountered by POCUS practitioners in the care of paediatric ECMO patients. While evidence supporting the use of POCUS has grown significantly in the adult population, this review underscores the paucity of paediatric-specific data. Based on the authors’ experience, POCUS is increasingly utilised in this patient group; however, the lack of robust evidence and governance frameworks may undermine the validity and consistency of its findings. The changes in physiology of patients on ECMO means that caution must be used when extrapolating non-ECMO POCUS studies to this specific population. The development of standardised guidelines, POCUS definitions, and a stronger evidence base is essential. This would enhance the reliability of POCUS in this context, ensure its effective integration into clinical practice and produce evidence that is less heterogeneous.
Supplemental Material
Supplemental Material - A scoping review: Point of care ultrasound (POCUS) use in paediatric ECMO
Supplemental Material for A scoping review: Point of care ultrasound (POCUS) use in paediatric ECMO by Catia Pinto, Alex Williams, Jon Lillie, Benjamin Crulli in Perfusion
Footnotes
Acknowledgements
Julie Windle – Senior Library Assistant, King’s College London. Dr Nicole O’Brien – Critical Care Physician, Nationwide Children’s Hospital, Columbus, Ohio, USA. Dr Osami Honjo – Cardiovascular Surgeon, The Hospital for Sick Children, Toronto, Canada.
Authors contributions
CP and AW designed the study, undertook literature search, reviewed all the articles and wrote the initial manuscript draft. JL and BC helped design study, reviewed and contributed to the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
