Abstract
Background
The effect of the anticoagulant, dabigatran, and its antagonist, idarucizumab, on coagulation remains poorly quantified. There are few pharmacokinetic-pharmacodynamic data available to determine dabigatran dose in humans or animals undergoing cardiopulmonary bypass.
Methods
Five sheep were given intravenous dabigatran 4 mg/kg. Blood samples were collected for thromboelastometric reaction time (R-time) and drug assay at 5, 15, 30, 60, 120, 240, 480 min, and 24 h. Plasma dabigatran concentrations and R-times were analyzed using an integrated pharmacokinetic-pharmacodynamic model using non-linear mixed effects. The impact of idarucizumab 15 mg/kg administered 120 min after dabigatran 4 mg/kg and its effect on R-time was observed.
Results
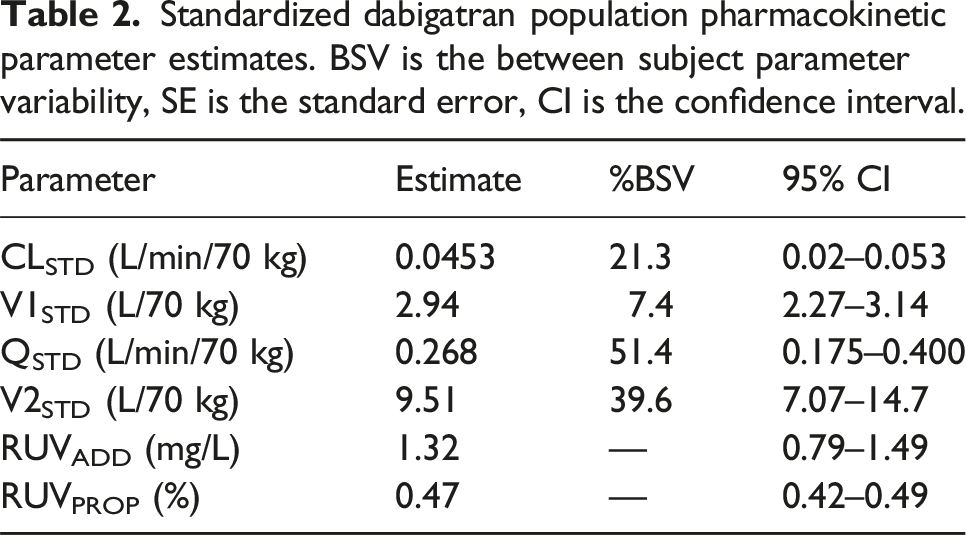
A 2-compartment model described dabigatran pharmacokinetics with a clearance (CL 0.0453 L/min/70 kg), intercompartment clearance (Q 0.268 L/min/70 kg), central volume of distribution (V1 2.94 L/70 kg), peripheral volume of distribution (V2 9.51 L/70 kg). The effect compartment model estimates for a sigmoid EMAX model using Reaction time had an effect site concentration (Ce50 64.2 mg/L) eliciting half of the maximal effect (EMAX 180 min). The plasma-effect compartment equilibration half time (T1/2keo) was 1.04 min. Idarucizumab 15 mg/kg reduced R-time by approximately 5 min.
Conclusions
Dabigatran reversibly binds to the active site on the thrombin molecule, preventing activation of coagulation factors. The pharmacologic target concentration strategy uses pharmacokinetic-pharmacodynamic information to inform dose. A loading dose of dabigatran 0.25 mg/kg followed by a maintenance infusion of dabigatran 0.0175 mg/kg/min for 30 min and a subsequent infusion dabigatran 0.0075 mg/kg/min achieves a steady state target concentration of 5 mg/L in a sheep model.
Introduction
Dabigatran is an anticoagulant drug that reversibly binds to the active site of the thrombin molecule; it is classified as a direct thrombin inhibitor. It also reduces thrombin-mediated inhibition of fibrinolysis. The USA Food and Drug Administration (FDA) has approved oral dabigatran for the management of adult humans with thromboembolic disease and stroke prevention in those with atrial fibrillation. It is also available for the treatment of children ages 3 months to 12 years with venous thromboembolism directly after receiving injectable anticoagulants for at least 5 days. 1 Dabigatran has a volume of distribution of 50–70 L in humans with an elimination half-life of 12–17 h. When administered orally the relative bioavailability (F) is only 3%–7%. Adult human apparent clearance estimates (CL/F ∼ 4 L/min)2,3 are influenced by renal function, 4 although hepatic glucuronide metabolism to acyl metabolites also contributes. 2 Dabigatran anticoagulant effect can be reversed using the monoclonal antibody, idarucizumab. Intravenous idarucizumab immediately decreases unbound dabigatran concentration. 5 A linear correlation was observed between unbound dabigatran and diluted thrombin time and ecarin clotting time (ECT). 5 Dabigatran is contraindicated in patients with active pathological bleeding and its major adverse effect is gastrointestinal bleeding. 6
Other direct thrombin inhibitors (e.g., bivalirudin, hirudin, argatropan) have been successfully used as an alternative to heparin anticoagulation for patients undergoing cardiopulmonary bypass.7–9 Dabigatran also has promise as a direct thrombin inhibitor for cardiopulmonary bypass,10,11 in part because the effects of dabigatran can be rapidly and completely reversed with the monoclonal antibody, idarucizumab. 12 Preliminary results suggest that effects of dabigatran concentration on diluted thrombin time (dTT), ecarin clotting time (ECT) and activated partial thromboplastin time (aPTT) are largely comparable between adults and children. 13
Unfractionated heparin is the most commonly used anticoagulant in both cardiopulmonary and extracorporeal circuits. Concerns about heparin induced thrombocytopaenia has spurred examination of direct thrombin inhibitors. 14 However, bivalirudin and argatropan have no reversal agent available. Dabigatran is a potential alternative because effect is reversable. Investigation using animal models is a first step used to guide investigation in humans. We investigated the dabigatran pharmacokinetic-pharmacodynamic relationship in sheep using a delayed effect compartment model.15,16 The effect measure of reaction time (R-time) was used as a convenient point-of-care whole blood test reflecting inhibition of the tissue factor pathway. Our intent was to use the target concentration strategy17,18 to determine a dabigatran dose that could be used for anticoagulant investigation of sheep undergoing cardiopulmonary bypass. This strategy involves identification of a target dabigatran concentration for cardiopulmonary bypass use in sheep. A dose to achieve and maintain this target concentration can then be investigated using compartment models describing drug disposition in a cardiopulmonary bypass circuit.
Methods
Animals and materials
Demographic data and dabigatran injectate volume of sheep who underwent PKPD analysis.

Dabigatran 4 mg/kg IV was injected over 1 min immediately after the baseline (time 0) draw and flushed with 3 mL of normal saline. Blood samples for dabigatran assay and R time were taken at 5, 15, 30, 60, 90, and 120 min. Idarucizumab 15 mg/kg IV was given over 30 s immediately after the 120 min of dabigatran injection time, and additional blood samples were collected at the subsequent times of 5, 15, 30, 60, 120, 240, 480 min, and 24 h. All experimental protocols were approved by the Association for Assessment and Accreditation of Laboratory Animal Care-accredited University of Rochester Committee on Animal Resources.
Dabigatran (4 mg/kg) was injected through the venous cannula manually over 60 s. Samples (2 mL) were collected into citrate tubes from the arterial cannula at baseline (prior to injection) and at 5, 15, 30, 60, 90 and 120 min while the sheep were under anesthesia. Idarucizumab 15 mg/kg was injected through the venous cannula manually over 30 s, and blood samples were collected at 5, 15, 30, 60 min while the sheep were under anesthesia. Then sheep were recovered from anesthesia and moved back to their cages with food and water ad libitum. The remaining late blood samples at 120, 240, 480 min and 24 h after idarucizumab administration were collected. The total amount of collected blood was less than 7.5% (4.5 mL/kg body weight). At the conclusion of the study period sheep had intravenous and arterial cannulas removed and hemostasis from cannulation sites achieved with pressure. Sheep were monitored for 24 h for adverse reactions. Markers of hepatic and renal injury including alanine aminotransferase (ALT), aspartate aminotransferase (AST), and creatinine concentration were measured at baseline and at 24 h.
Reaction time (R-time) was measured using a Thromboelastograph Analyzer 5000 (Haemoscope Corp.). Plasma dabigatran concentrations were measured using liquid chromatography/mass spectrometry (LC-MS/MS) with a Dionex Ultimate 3000 UHPLC coupled to a Q Exactive Plus mass spectrometer (Thermo Scientific).
Dabigatran solutions
An intravenous formulation was required for study. One milligram of Dabigatran (Clearsynth, Ontario, Canada) was dissolved in 60 µL of 0.075 M HCl, and then the solution was added into 0.2 mL of 20% N,N-dimethylacetamide. The dabigatran injectate concentration was 3846 ug/mL. Dabigatran volume injected intravenously over 1 min is shown in Table 1.
Reaction time (R), and dabigatran concentration were measured in all blood samples. One milliliter of blood was centrifuged at 370 g for 15 min to obtain plasma dabigatran concentrations measured using liquid chromatography/mass spectrometry (LC-MS/MS) with a Dionex Ultimate 3000 UHPLC coupled to a Q Exactive Plus mass spectrometer (Thermo Scientific, San Jose, CA).
Dabigatran pharmacokinetic-pharmacodynamics
Population parameter estimates were obtained using nonlinear mixed effects models (NONMEM 7.5, ICON Development Solutions, MD, USA). These models account for population parameter variability (between subjects) and residual variability (random effects) as well as parameter differences predicted by covariate (fixed) effects. Population parameter variability was described using exponential models, which is equivalent to assuming a log-normal distribution and avoids biologically inappropriate parameter values of zero or less. Residual unidentified variability (RUV) was modeled using both proportional (RUVPROP) and additive residual (RUVADD) errors. The ADVAN6 subroutine was used to solve differential equations. NM-TRAN code is available in supplementary material (Supplementary NM-TRAN Code). A sequential PPPD method was used for final pharmacodynamic parameter estimates. 19 Convergence criterion was three significant digits.
Pharmacokinetics
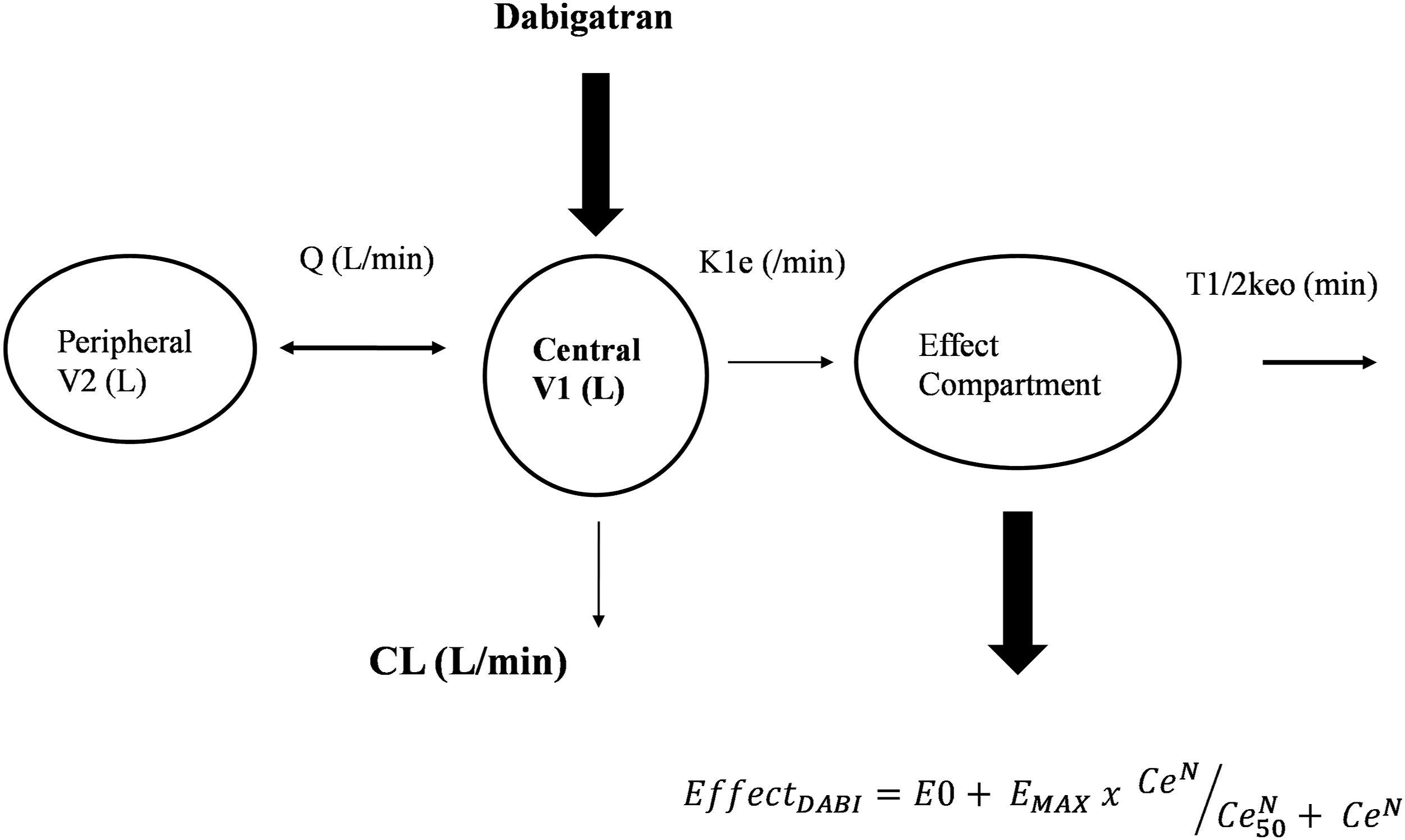
A two-compartment (central and peripheral) pharmacokinetic model was used to fit data. The model was parameterized in terms of clearance (CL), between compartment clearance (Q), central volume (V1) and peripheral volume of distribution (V2). An additional effect compartment was linked to the central compartment by a rate constant (keo). That constant was expressed as a half-time (T1/2keo = Ln(2)/keo), demonstrated in Figure 1. The pharmacokinetic parameter values were standardized for a body weight of 70 kg using allometric models.
20
This standardization allows comparison of sheep parameter estimates with those reported for human adults
21
: A diagram representing the pharmacokinetic effect compartment model. Drug is delivered into a central compartment (V1) that distributes to a peripheral compartment (V2) linked by an intercompartment clearance (Q). A rate constant (k1e) links the central compartment to an effect compartment. The rate constant k1e is the same as keo when the system is at equilibrium.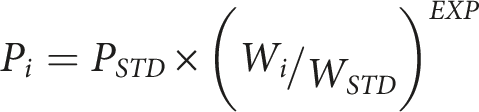
Pharmacodynamics
A sigmoidal EMAX model was used to describe thromboelastogram reaction time (R). The reaction time (R-time) represents the time until initial fibrin formation after mechanical stress and reflects the ability to generate thrombin. Population parameter estimates were estimated using an effect compartment model, a model valid for situations where there is an apparent temporal displacement between plasma concentration (Cp) and response e.g., neuromuscular blocking drugs.
15
A rate constant (keo, T1/2keo = Ln(2)/keo) links plasma concentration with effect site concentration (Ce).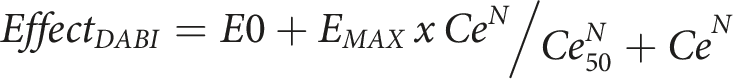
The parameter E0 is the baseline measure (e.g., ACT 100, R 0.4 min), EMAX is the maximum drug effect, Ce50 is the effect site concentration eliciting half of EMAX and N is the Hill coefficient describing the steepness of the concentration–response curve 15
The impact of idarucizumab was modelled by a unit bolus input into a fourth compartment with first order elimination described by a rate constant (KIDA). Input duration (DURIDA) was estimated as a parameter. The vasopressors concentration (CIDA) was assumed to directly decrease R time and this relationship was described using a slope constant (SLOPEIDA). Observed R time was the sum of Dabigatran (EFFECTDABI) and idarucizumab effects (EFFECTIDA).
Quality of fit
Model selection required an improvement in the NONMEM objective function (OBJ) between nested models, equating to a reduction >3.84 based on a Chi square distribution (α < 0.05). A visual predictive check (VPC), was used to evaluate how well the model predicted the distribution of observed dabigatran concentrations or coagulation measures (R).
Results
Pharmacokinetics
Standardized dabigatran population pharmacokinetic parameter estimates. BSV is the between subject parameter variability, SE is the standard error, CI is the confidence interval.
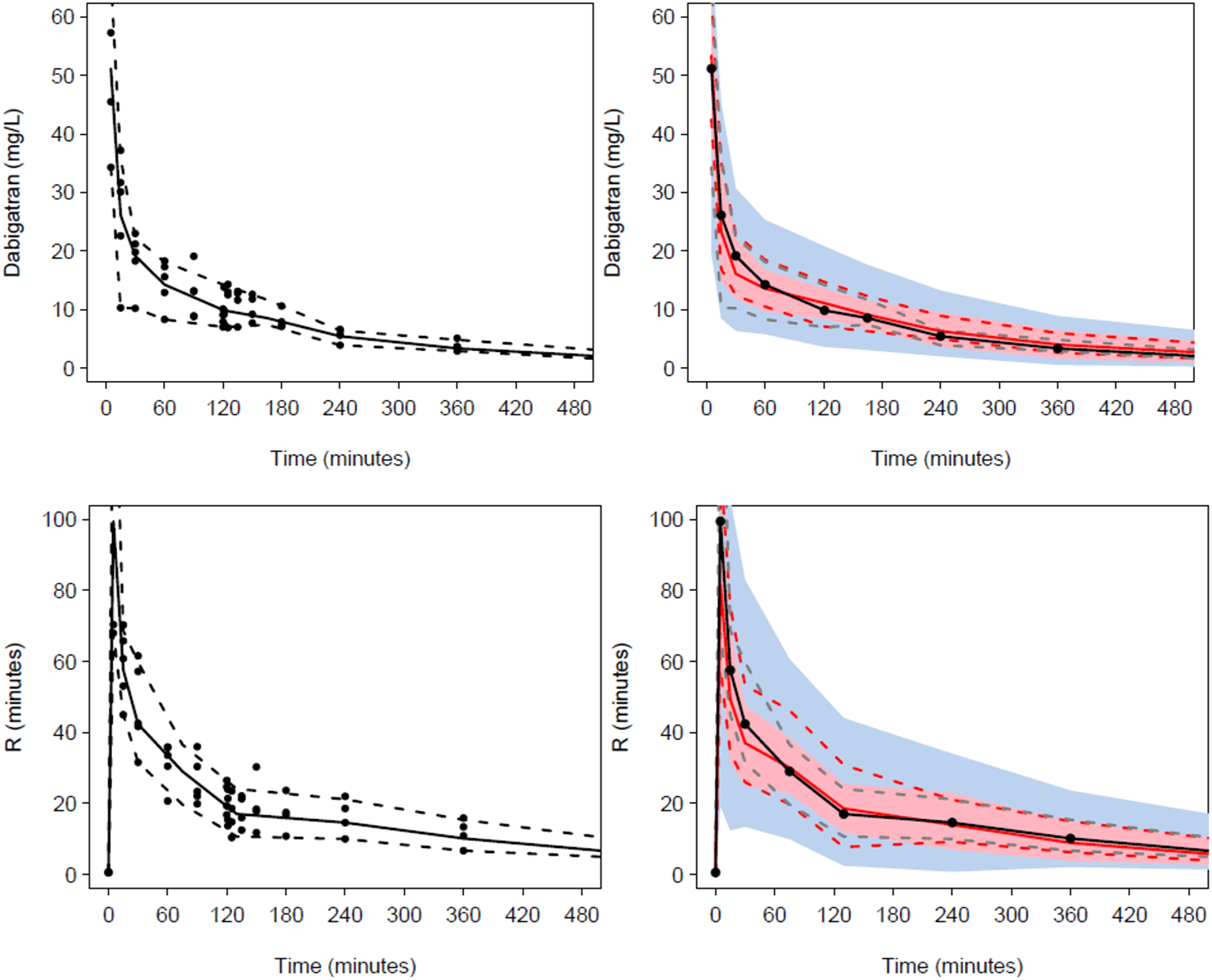
Visual predictive checks (VPC) Plots show median (solid) and 90% intervals (dashed lines). The left hand plot shows all prediction corrected observed dabigatran concentrations or effect (R-time). The right hand plot shows prediction corrected percentiles (10%, 50%, and 90%) for observations (grey dashed lines) and predictions (red dashed lines) with 95% confidence intervals for prediction percentiles (median, pink shading; 5th and 95th blue shading). The upper panel displays the VPC for the pharmacokinetic analysis. The lower panel shows the VPC for the pharmacodynamic reaction time (R) response.
Pharmacodynamics
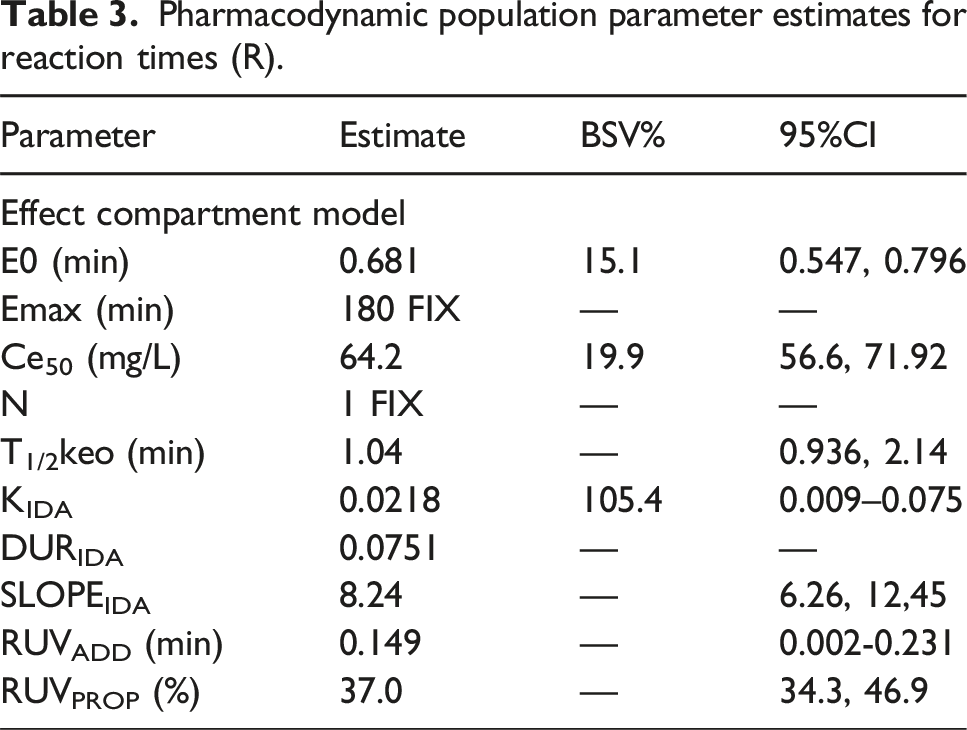
Pharmacodynamic population parameter estimates for reaction times (R).
Impact of idarucizumab on reaction time
The use of idarucizumab 15 mg/kg administered 120 min after dabigatran 4 mg/kg reduced reaction time by approximately 5 min over the 5 min after administration. This effect is demonstrated in Supplementary Figure S1 for sheep subject #4. An Emax model was used to describe the relationship between dabigatran effect site concentration and R-time. The symbols (x) are individual Bayesian predictions. The solid line demonstrates population prediction. An R time of 13 min (Normal: 4–8 min) was considered a suitable target effect. The effect site concentration correlates with a target concentration of 5 mg/L.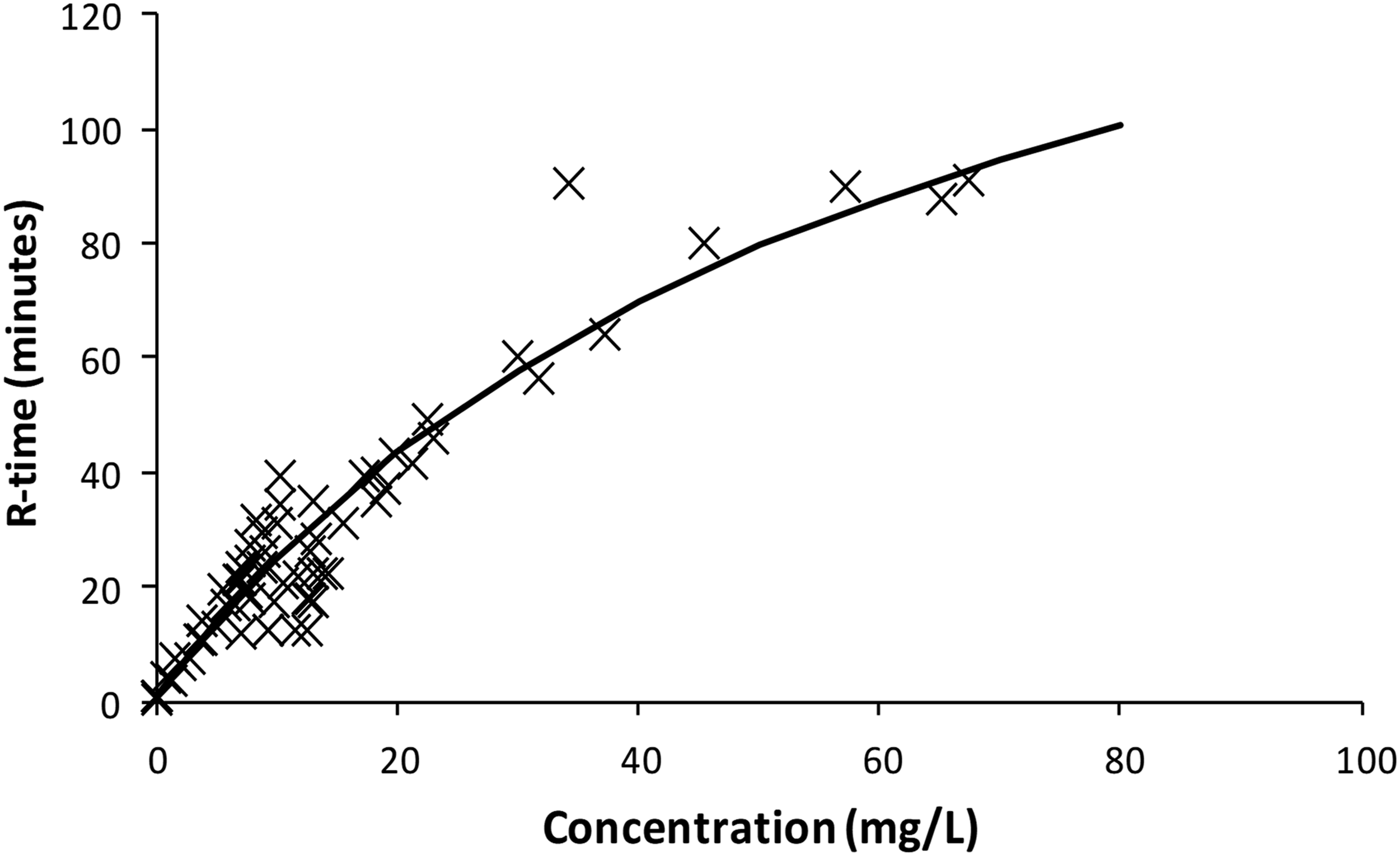
Target concentration strategy
The goal of treatment is the target effect. The target concentration strategy is useful for determining the clinical dose through an understanding of pharmacokinetics and pharmacodynamics.
17
The concentration-response relationship (Figure 3), determined in the pharmacodynamic analysis was used to predict a target concentration of 5 mg/L. Pharmacokinetic parameters were used to determine dose that would achieve this target concentration in sheep supported using cardiopulmonary bypass. Simulation of dabigatran concentration using a loading dose of dabigatran 0.25 mg/kg followed by a maintenance infusion of dabigatran 0.0175 mg/kg/min for 30 min and a subsequent infusion dabigatran 0.0075 mg/kg/min for 30–90 min achieved a steady state target concentration of 5 mg/L.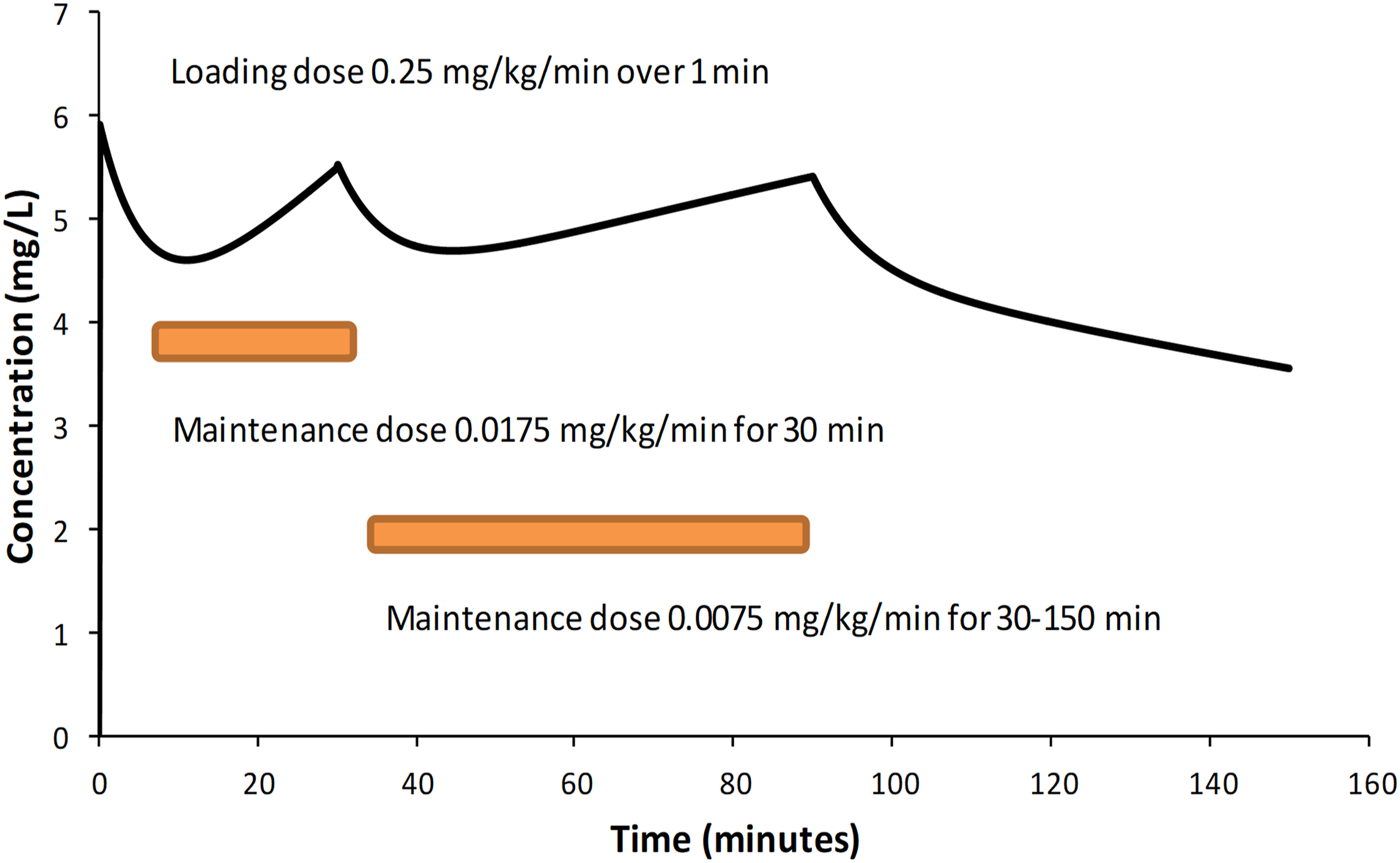
Determination of dabigatran dose
A simple pharmacokinetic model was used to determine dabigatran disposition in sheep during cardiopulmonary bypass (Supplementary Figure S2). Pharmacokinetic Parameter estimates (Table 2) were used to simulate time concentration profiles using differential equations in Berkeley Madonna™ modeling and simulation software (Robert Macey and George Oster of the University of California Berkeley, USA).
Differential equations used were:
d/dt (central) = drug rate in + Q2*C2 + Q3*C3*- (CL+Q2+Q3)*C1
d/dt (peripheral) = Q2*(C1 - C2)
d/dt (CPB) = C1*Q2-C3*Q3
where C1 is concentration in the central compartment, C2 is concentration in the peripheral compartment, and C3 that concentration in the cardiopulmonary bypass (CPB) circuit; Q2 is the intercompartment clearance and Q3 the pump flow, parameterized as intercompartment clearance Q3.
Loading doses and maintenance infusions over 90 min that achieve steady-state concentrations of 5 mg/mL are shown in Figure 4. These infusion rates assume that the CPB circuitry is preloaded with a dabigatran to achieve the target concentration of 5 mg/L. That dose is determined by the volume of the CPB circuit (V3).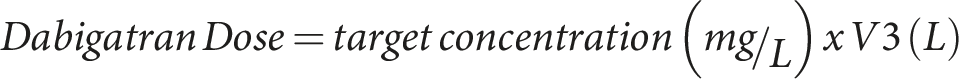
A loading dose of dabigatran 0.25 mg/kg followed by a maintenance infusion of dabigatran 0.0175 mg/kg/min for 30 min and a subsequent infusion dabigatran 0.0075 mg/kg/min achieved a steady state target concentration of 5 mg/L.
Discussion
We present an example of use of the target concentration strategy to determine dose for cardiopulmonary bypass investigation of dabigatran in sheep. This methodology is commonly used for clinical drug development and dose determination.17,18 The goal of any pharmacological treatment is the target effect. A pharmacodynamic model for dabigatran and its effect on reaction time was determined. This relationship was explored to predict the target concentration (5 mg/L) known to be associated with a specific target effect (R-time 16 min). Initial dosing that could be used to study the drug during cardiopulmonary bypass was determined using estimated pharmacokinetic parameter estimates within a model that described a simple bypass circuit used in a sheep study. Subsequent monitoring of drug effect and/or drug concentrations and Bayesian forecasting may be used to improve the dose in individual sheep undergoing cardiopulmonary bypass. 17
Monitoring of R-time is a convenient point-of-care whole blood test that is rapidly achieved. The short equilibration half-time (T1/2keo) makes this a coagulative measure useful for dabigatran dose adjustment. The need for subsequent monitoring after initiation of bypass is because the use of extracorporeal circuits are associated with additional covariates that can affect both pharmacokinetic (e.g., clearance, volume) and pharmacodynamic (EMAX, C50) parameter estimates. While it is possible to nonmaize pharmacokinetic parameters for size using allometric theory,20,22,23 pharmacokinetic maturation, particularly in children has impact.22,24–26 These considerations have been used to describe dabigatran in children suffering thromboembolic disease. 27 However, these considerations becomes more complex in situations such as extracorporeal membrane oxygenation (ECMO) where improved organ perfusion betters function. 28 In contrast, children undergoing cardiopulmonary bypass often suffer a renal insult, lessening clearance. Altered organ blood flow, temperature changes or inflammatory responses can affect clearance. Protein binding changes, haemodilution, and circuit drug adherence influence apparent volume of distribution. Haemodilution and inflammatory responses have further impact on the coagulation cascade. A lower target concentration during infancy may be required because of immaturity of the coagulation cascade. 29
We have explored the sheep as an animal model because there is an increasing focus on rapidly translating findings in the laboratory to clinical applications in cardiovascular procedures. This has resulted in a shift from small to larger animal models, such as sheep, that closely correlate with human anatomy and physiology and allow use of commercially available bypass units used in humans. This current study was performed under general anaesthesia, a clinical scenario also used in humans. It is of value that the pharmacokinetic parameter estimate for clearance was similar to that noted in adult humans.2,3 Concerns that anaesthesia drugs may interfere with dabigatran metabolism are unfounded. Dabigatran in primarily cleared by renal function. Ketamine is metabolized by CYP2B6 and CYP3A4-mediated N -demethylation at a rate approximating hepatic blood flow. 30 Midazolam is primarily metabolized by CYP3A4. 31 These hepatic clearance pathways have little or no impact on renal function, glucuronide conjugation or dabigatran clearance. 32 Isoflurane is predominantly cleared through the lungs rather than hepatic metabolism. The anaesthetic drugs are not known to have any coagulation effect. Neither renal function nor hepatic function were compromised in sheep given dabigatran.
The use of idarucizumab 15 mg/kg administered 120 min after dabigatran 4 mg/kg reduced reaction time by approximately 5 min over the 5 min after administration. An idarucizumab dose range was not explored. A larger dose may have greater effect. The reversal agent certainly had effect at this dose, reducing reaction time by 30%. The clinical impact of this effect on sheep while undergoing cardiopulmonary bypass remains untested.
Supplemental Material
Supplemental Material - Dabigatran pharmacokinetic-pharmacodynamic in sheep: Informing dose for anticoagulation during cardiopulmonary bypass
Supplemental Material for Dabigatran pharmacokinetic-pharmacodynamic in sheep: Informing dose for anticoagulation during cardiopulmonary bypass by Michael P Eaton, Sergiy M Nadtochiy, Tatsiana Stefanos and Brian J Anderson in Perfusion.
Footnotes
Acknowledgments
We thank the URMC Mass Spectrometry Resource Laboratory for measuring Dabigatran concentrations in the rabbit plasma samples.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
