Abstract
The discharge of organic pollutants has become a significant environmental concern, posing serious threats to aquatic ecosystems. In this study, an iron-based metal–organic framework (Fe-MOF) was synthesized using ferric nitrate and 1,2,4-benzenetricarboxylic acid (BTC) through a solvothermal method and evaluated for its dual functionality in dye removal and antibacterial activity. Unlike the commonly used 1,3,5-BTC, the 1,2,4-BTC linker positions two carboxylic groups adjacent to each other, which enables intramolecular hydrogen bonding and alters the coordination geometry, thereby enhancing framework stability and providing interaction sites for pollutant binding. The morphology and surface characterizations of the Fe-MOF were confirmed by FTIR, XRD, SEM, TEM, EDX, XPS, vibrating sample magnetometer, Raman spectroscopy, and zeta-potential analysis, confirming its successful formation and surface charge. The Brunauer-Emmett-Teller analysis showed that the surface area of Fe-based MOFs was 157 m2/g, confirming the presence of mesopores and facilitating dye diffusion to the active sites. The batch adsorption experiments showed high efficiency in removing tropaeolin OO and sunset yellow dyes with maximum removal efficiencies of 89% and 96%, respectively. According to isothermal modeling, kinetic studies, and thermodynamic analysis, the adsorption process favored multilayer adsorption behavior and spontaneous dye removal. Statistical analysis, including regression modeling and one-way analysis of variance, revealed the significant influence of operational parameters, such as pH, temperature, and adsorbent dose. Fe-MOF presented antibacterial activity, achieving a 96.47% reduction in colony-forming units. Regeneration studies confirmed the reusability of the Fe-MOF, preserving over 92% of its efficiency after two cycles.
Keywords
Introduction
Water is a fundamental resource for the survival of all living organisms, and the availability of clean water is vital for maintaining healthy ecosystems. Unfortunately, industrial activities have contaminated water bodies with synthetic dyes, particularly in the textile and dyeing sectors (Singh et al., 2024). These dyes are hazardous due to their stability, persistence, and potential carcinogenic effects on human health and ecological integrity (Bhatnagar and Sillanpää, 2010; Pattnaik et al., 2018). The toxic degradation products of these dyes can severely degrade water quality (Gosavi and Sharma, 2014). This situation necessitates effective wastewater treatment solutions to mitigate the adverse impacts of dye pollution (Zhao et al., 2020).
Among various treatment methods, adsorption has emerged as one of the most efficient techniques for removing hazardous substances from wastewater due to its simplicity and cost-effectiveness (Ghaedi et al., 2015; Motshekga et al., 2024). Adsorption processes exploit the attractive forces between adsorbates (in this case, dyes) and adsorbents (materials designed to capture these contaminants) to achieve removal (Ihsanullah, 2022). Metal–organic frameworks (MOFs) have garnered considerable attention as a promising class of porous crystalline materials composed of metal ions coordinated with organic ligands (Khan et al., 2020; Yu et al., 2021). Their exceptional adsorption capabilities, high surface area, and structural tunability make them attractive candidates for water treatment applications (Goswami et al., 2023; Jiang et al., 2019).
Iron-based MOFs (Fe-MOFs) are particularly appealing due to iron's low cost, natural abundance, and environmentally friendly profile (Du et al., 2022; Soni et al., 2020). These materials exhibit significant potential for various environmental remediation applications, especially for removing toxic organic dyes from industrial effluents (Oladoye et al., 2021). The unique properties of MOFs, including their high porosity and large surface area, facilitate the efficient capture and sequestration of harmful contaminants (Liu et al., 2023; Wang et al., 2021).
Selecting organic linkers is crucial in determining the structural characteristics and performance efficiency of MOFs. One widely utilized organic linker in synthesizing Fe-MOFs is 1,3,5-benzenetricarboxylic acid (BTC) (trimesic acid), known for its ability to form stable frameworks (Ghosh, 2019). This compound features three carboxyl groups capable of coordinating with metal ions, resulting in robust frameworks that can effectively entrap dye molecules within their porous structures (Adegoke et al., 2020; Sağlam et al., 2023).
Recent investigations have increasingly focused on the efficacy of Fe-MOFs in removing synthetic dyes from wastewater. Fe-BTC frameworks have demonstrated high adsorption behavior for various types of dyes and heavy metals, primarily attributed to their porous structure and larger surface area, as well as electrostatic attraction, hydrogen bonding, and π–π interactions (Han et al., 2019b). The BTC used in these studies is in the form of 1,3,5-benzene tricarboxylic acid, which is a symmetrical linker. However, in this study, we are using 1,2,4-BTC, in which two carboxylic groups are adjacent to each other (1,2) and the third is at the 4-position. The asymmetric arrangement changes the coordination geometry and produces chemically diverse binding environments within the MOF. The two adjacent carboxylic groups enable intramolecular hydrogen bonding, which stabilizes the linker conformations and creates localized hydrogen-bond donor/acceptor sites that enhance interactions with dye molecules (Kamal et al., 2023).
Such structural features are expected to enhance multifunctional adsorption mechanisms, like hydrogen bonding, electrostatic attraction, and π–π interactions, while potentially broadening the effective pH range and improving adsorption performance for numerous types of dyes.
For instance, tropaeolin OO (TOO), a commonly used azo dye in the textile industry, is particularly problematic due to its stability in water and the toxic nature of its degradation products (Tran et al., 2022). Similarly, sunset yellow (SY), a reactive dye, presents additional challenges due to its high solubility and resistance to conventional treatment methodologies (Encyclopædia Britannica, 2024). Both anionic dyes are persistent environmental pollutants, underscoring the necessity for their effective removal to safeguard water quality (Li et al., 2021; Wyszogrodzka et al., 2016).
Gracía et al. (2014) have demonstrated the high adsorption capacity of Fe-BTC, approximately 435 mg/g, for the azo dye Orange II, where the Langmuir isotherm, characterized by a combination of physisorption/chemisorption mechanisms, describes the adsorption behavior. Delpiano et al. (2021) investigated the removal of Alizarin red S and malachite green, achieving adsorption capacities of 80 and 177 mg/g, respectively, as determined by pseudo-second-order kinetics. According to Karami et al. (2021), Fe-BTC exhibited a significant uptake of 100.3 mg/g of methyl orange dye under both batch and fixed-bed conditions. Other MOF-based systems, such as Fe3O4@ZTB-1 composites, demonstrated a high adsorption capacity for Congo red (458 mg/g) through electrostatic attraction and hydrogen bonding interactions (Han et al., 2019b).
In addition to their adsorptive properties, Fe-MOFs also exhibit antibacterial activity, making them valuable for addressing microbial contamination in water. The ability of these materials to inhibit bacterial growth in nutrient media enhances their applicability in wastewater treatment (Zhang et al., 2022; Zhao et al., 2021). Studies have shown that Fe-MOFs can effectively reduce microbial loads by disrupting bacterial membranes and inhibiting cellular functions (Nemiwal et al., 2021). This multifaceted functionality addresses dye pollution and contributes to improved water quality by reducing microbial contamination (Wen et al., 2021).
This research aims to provide crucial insights into the development of advanced adsorbents for industrial wastewater treatment by systematically comparing the efficiency of the Fe-MOF based on 1,2,4-BTC as a precursor in removing distinct dyes, TOO and SY. Overall, integrating Fe-MOFs into wastewater treatment strategies holds great promise for addressing the challenges posed by synthetic dye pollution and microbial contamination, ultimately contributing to cleaner and safer water resources for all. The investigation will focus on key factors, including adsorption capacities, the influence of pH on dye removal, and the regeneration potential of the synthesized Fe-MOFs.
Materials and methods
Materials
1,2,4-Benzenetricarboxylic anhydride (98%), TOO dye, and SY dye were purchased from Sigma-Aldrich, and the structures of TOO and SY dyes are shown in Figure 1. Ferric nitrate nonahydrate (Fe(NO3)3·9H2O, 98%) was obtained from SRL India. N,N'-Dimethylformamide (DMF, 99.8%), sodium hydroxide (NaOH), hydrochloric acid (HCl), barium chloride (BaCl2), sodium carbonate (Na2CO3), sodium chloride (NaCl), sodium sulfate (Na2SO4), and sodium bicarbonate (NaHCO3) were obtained from PioChem. Paraffinic oil was obtained from Alexandria Mineral Oils Company. Deionized water (water for injection) was sourced from Otsuka and used throughout the experiments. Nutrient agar media was sourced from HI Media.
Structure of tropaeolin OO (TOO) and sunset yellow (SY) dyes.
Synthesis of 1,2,4-BTC
1,2,4-BTC was synthesized via hydrolysis of its anhydride derivative. Initially, 1,2,4-benzenetricarboxylic anhydride was treated with an aqueous solution of sodium hydroxide. This step facilitated nucleophilic attack by hydroxide ions, leading to the opening of the anhydride ring and formation of a tricarboxylate intermediate. Subsequent hydrolysis in aqueous media resulted in the formation of the corresponding tricarboxylic acid salt. The reaction mixture was then acidified using a suitable acid (e.g. HCl) to precipitate the free 1,2,4-BTC, which was recovered by filtration and drying (see Scheme 1).

Synthesis of 1,2,4-benzenetricarboxylic acid from its anhydride.
Development of Fe-MOF
Fe-MOFs were synthesized by grinding Fe(NO3)3·9H2O (0.594 g, 1.7 mmol) and 1,2,4-BTC (0.166 g, 1 mmol) for 10 min, then dissolving the mixture in 20 mL of DMF at room temperature with continuous stirring. The mixture was subjected to 30 min of sonication to enhance dispersion and facilitate interaction between the components. The resulting solution was transferred to a stainless-steel-lined Teflon autoclave and heated to 150 °C for 16 h in an oil bath under static conditions to induce crystallization, as shown in Scheme 2. Once the reaction was completed, the solid product was isolated by centrifuging at 4500 r/min and washed thoroughly with DMF, followed by distilled water to remove unreacted species and solvents. The grown powders were dried in an oven at around 100 to 120 °C. The powder was known as Fe-MOF and then stored in a vacuum desiccator to avoid moisture absorption. The yield was 0.37 g, equivalent to a 55.2% yield based on the mass of the final product.

Synthesis of Fe-MOF from 1,2,4-benzene tricarboxylic acid. Fe-MOF: iron-based metal–organic framework.
Reaction mechanism
The Fe-MOF was synthesized using a hydrothermal process involving the removal of protons from the organic ligand (a Lewis acid) as the temperature increased. This deprotonation occurs because the rising temperature causes the equilibrium to shift, promoting proton extraction from the linker and leading to precipitation, as per Le Chatelier's principle (the equilibrium shifts to counteract the effect of the temperature increase). The possible structure of Fe-MOF consists of trimers of Fe(III) octahedral units, where three iron atoms are connected by a central oxygen atom (3-oxo group). Each iron atom in the trimer is bonded to two water molecules, six carboxylate groups (from the organic linker), and hydroxyl anion groups, as shown in Scheme 3. In the synthesis process, ferric nitrate salt is the iron source, while 1,2,4-BTC acts as the ligand or proton donor. As the reaction proceeds, Fe3⁺ ions form complexes with water, producing a structure called [Fe(H2O)2(OH)]2⁺. In this complex, Fe3⁺ ions strongly bond with water molecules, forming hydroxide ions. This occurs due to the partial dissociation of water molecules, resulting in hydroxide ions that stabilize the iron within the complex.

Schematic showing the possible structure formation of Fe-MOF. Fe-MOF: iron-based metal–organic framework.
In equation (3), the gradual absorption of protons leads to a slow increase in pH, which is favorable for the formation of [Fe3(3-oxo)(H2O)2(OH)]2+ units, hydroxide ions, and oxide, alongside the deprotonation of H3BTC. Consequently, the synthesis of Fe-MOFs through the hydrothermal method using the specified precursors results in a high yield and a large surface area due to the two-step purification procedure (Nivetha et al., 2020).
Characterization of Fe-MOF
The fabricated Fe-MOF was analyzed by powder X-ray diffraction (XRD) to assess the crystallinity of its components.
The morphological changes of the MOFs were also clearly visible using a scanning electron microscope (SEM, JEOL JSM-6360 LA, Japan), equipped with an EDX accessory.
Transmission electron microscopy (TEM) images were obtained using a JEOL JEM-2100 microscope (JEOL Ltd, Tokyo, Japan) operated at 200 kV at the Central Metallurgical Research and Development Institute (CMRDI), Cairo, Egypt.
Furthermore, X-ray photoelectron spectroscopy (XPS, Thermo Scientific ESCALAB 250Xi VG) was utilized to determine the elemental composition of the adsorbent.
The components’ chemical structure was assessed using Fourier transform-infrared spectroscopy (FTIR, a Bruker Tensor 27 spectrometer (Bruker Optics, Ettlingen, Germany) in the range of 4000–400 cm−1 at the Central Laboratory, Faculty of Science, Alexandria University, Egypt.
Raman spectra were collected using a confocal micro-Raman spectrometer (alpha300 RA/S, WITec GmbH, Ulm, Germany) equipped with a 532 nm laser, a Zeiss EC Epiplan-Neofluar 100×/0.9 objective, and a 600 g/mm grating (BLZ 500 nm) at the Egyptian Petroleum Research Institute (EPRI).
Zeta-potential measurements were conducted using a Nano Zetasizer ZS (Malvern Instruments Ltd, Worcestershire, UK) at the CMRDI, Cairo, Egypt.
An area was determined through Brunauer-Emmett-Teller (BET) analysis. The samples were initially out-gassed under vacuum (10−4 Torr) at 150 °C (BELSORP max II equipment, Japan) (Petroleum Research Institution).
Magnetic properties were investigated using a vibrating sample magnetometer (VSM, Lake Shore 7400) from the CMRDI, Helwan, Cairo, Egypt.
For antibacterial activity studies, plate count experiments were conducted in a laminar airflow biological safety cabinet (JSR), followed by incubation in an ICT40 ChemTech incubator at 37 °C. Media sterilization and preparation processes were operated at 121 °C and 1.5 bar using an autoclave (SjENG) of Korean origin. Colony counting was performed using an automatic colony counter (WIGGENS) to ensure precise quantification of bacterial colonies.
Batch adsorption experiment
TOO and SY were absorbed by Fe-MOFs, and their adsorption was examined in terms of pH, adsorbent mass, contact time, initial dye concentration, and temperature. By dissolving 1 g of the appropriate dyes in 1 L of distilled water, a stock solution containing 1000 mg/L of both dyes was obtained. The prepared stock solution was diluted to provide various experimental concentrations. The experiments were conducted in 250 mL conical flasks containing 50 mL of dye solution and 300 r/min of agitation at 25 °C. Each test was repeated three times to ensure repeatability. The pH of the solutions was adjusted by adding 0.1 N H2SO4 and 0.1 N NaOH to 50 mL of the dye's initial concentration (5:50 ppm). About 0.2 g of Fe-MOF was added to the dye solution and shaken for 1 h. The mass of adsorbent varied between 0.05 and 0.30 g. The initial TOO and SY concentrations changed between 5 and 50 ppm to study the isothermal models. The temperature of the dye solutions was examined at 20, 25, 30, 35, and 40 °C to investigate the thermodynamic parameters. The contact time was conducted within the 30–180 min range to evaluate the kinetic models. The dye concentrations were determined using a ultraviolet–visible (UV–Vis) spectrophotometer at a wavelength λmax of 390 nm for TOO and 417 nm for SY. The amount of dye adsorption (q) and its removal efficiency (%Re) were assessed using the standard removal equations presented in Table 1.
Standard removal equations.

Effect of coexisting ions
To investigate the effect of coexisting ions on the adsorption efficiency of the synthesized Fe-MOFs for dye removal, a series of experiments was conducted using 50 mL of 50 ppm dye solutions. Specifically, 0.05 g of various salts, including sodium chloride (NaCl), sodium sulfate (Na2SO4), sodium carbonate (Na2CO3), and sodium bicarbonate (NaHCO3), were added to the dye solutions in conical flasks, followed by the addition of 0.2 g of the Fe-MOFs. The experiments were performed using deionized water to eliminate the presence of interfering ions. After adding the salt, the mixtures were stirred at 300 r/min for 1 h to ensure adequate interaction between the coexisting ions and the dye molecules. Following the stirring period, the mixtures were centrifuged to separate the Fe-MOFs from the solution. UV–Vis absorbance measurements were performed before and after the addition of salts to assess their impact on dye adsorption.
Reusability and regeneration of Fe-MOF
The reusability of the synthesized Fe-MOF for the adsorption of TOO and SY dyes was evaluated through regeneration experiments over two cycles. After each adsorption step, the spent MOF was washed with a 0.1 M NaOH solution to promote desorption of the anionic dyes. The basic environment deprotonates the MOF surface, weakening the electrostatic interactions and facilitating dye release. Following the NaOH treatment, the material was rinsed with deionized water to remove residual base, then centrifuged and dried at 100–120 °C for reuse. The regeneration process demonstrated the MOF's potential for two adsorption cycles with maintained efficiency.
Antibacterial activity
The antibacterial activity of the synthesized Fe-MOF was evaluated using the pour plate method with nutrient agar media. The agar was prepared by dissolving 28 g of the medium in 1000 mL of purified water, heating it to boil to ensure complete dissolution, and then sterilizing it in an autoclave at 120 ± 1 °C and 1.5 bar pressure for 15 min. After sterilization, the medium was poured into sterile Petri dishes under laminar airflow conditions. A wastewater sample was collected from an industrial source (Elvan Knitting and Dyeing) to test the antibacterial efficacy of the Fe-MOF. Five plates were prepared: one inoculated with 1 mL of untreated wastewater, and the others with 1 mL of wastewater treated with 0.05, 0.1, and 0.2 g of the synthesized Fe-MOF. A control plate containing only sterile nutrient agar was included to verify the absence of contamination. The experiment was repeated using different masses of Fe-MOFs to investigate the effect of MOF mass on the inhibition ratio. All plates were incubated at 37 °C for 72 h. After incubation, the bacterial colony count on the plate treated with the MOF showed a significant reduction, with colonies decreasing by approximately 96%, indicating the strong antibacterial activity of the MOF (Han et al., 2022).
Statistical analysis
The data analysis function in Microsoft Excel was used to perform the statistical analysis. The influence of specific parameters, including pH, adsorbent dosage, initial dye concentration, contact time, and temperature, on the efficacy of TOO and SY removal was assessed using regression analysis and one-way analysis of variance (ANOVA). The correlation between variables was modeled using regression analysis, and the statistical significance of each factor was evaluated using ANOVA, where p-values less than 0.05 were regarded as statistically significant results.
Results and discussion
Analysis of structure and morphology
XRD analysis
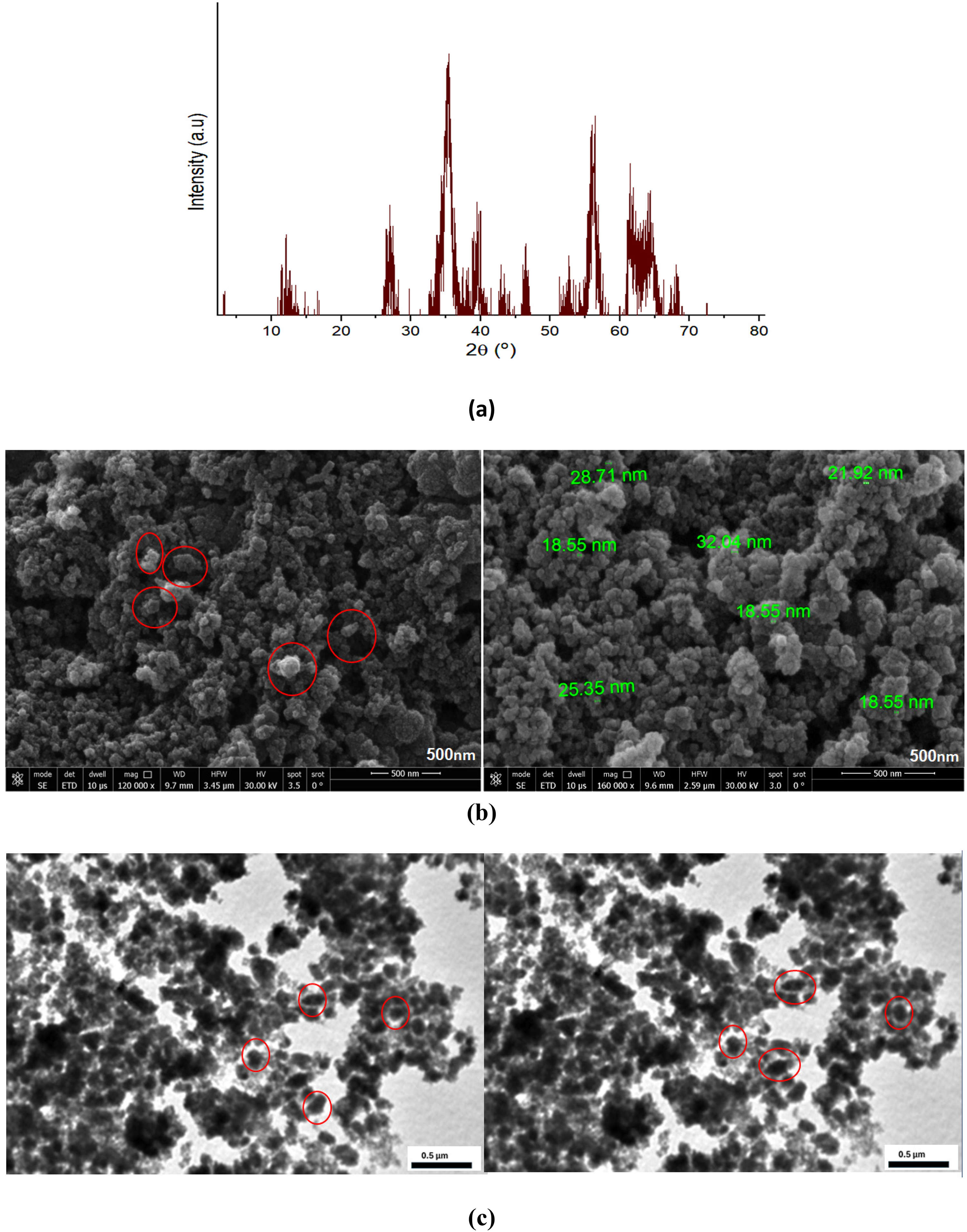
As shown in Figure 2(a), the XRD pattern of the synthesized Fe-MOFs displays a broad but distinguishable set of diffraction peaks, indicating that the material exhibits a degree of crystallinity. The observed diffraction peaks at 2θ = 12°, 28°, and 35° are in good agreement with the literature report for Fe-BTC MOF (Delpiano et al., 2021). While the peaks lack sharp definition, their presence confirms the formation of an ordered framework structure. This level of crystallinity is typical of MOFs synthesized under mild conditions. It is considered beneficial for adsorption applications, as it supports structural stability while maintaining accessible active sites for effective dye removal (Kalita et al., 2020).
Fe-MOF characterization: (a) XRD, (b) SEM, (c) TEM, (d) EDX and XPS, (e) FTIR, (f) Raman spectra, and (g) zeta-potential, (h) Adsorption-desorption isotherm, and (i) VSM. Fe-MOF: iron-based metal–organic framework.
SEM analysis
As presented in Figure 2(b), the SEM analysis of the synthesized Fe-MOF revealed well-defined, polyhedral-shaped particles with a relatively uniform size distribution. The average adsorbent size was measured in the 20–200 nm range, which is typically expected for Fe-MOFs (Bhomick et al., 2025). Additionally, a degree of particle clustering is observed, which may be attributed to the high surface energy of nano-sized particles rather than severe agglomeration.
TEM analysis
TEM analysis (Figure 2(c)) further confirms the nanoscale size, polyhedral shape, and good dispersion of Fe-MOF particles, with no major aggregation visible, indicating a stable synthesis process. These additional insights from TEM underscore the material's suitability for adsorption applications. All of these showed that the produced Fe-MOF has crystallinity and uniformly distributed nanoparticles, in agreement with the SEM observations (Sheikhhosseini and Yahyazadehfar, 2022).
EDX analysis
The EDX analysis is used to determine the elemental composition of the adsorbent. As reported in Figure 2(d), the Fe-MOF composition, where the characteristic peaks corresponding to Fe:O:C are 30.83%:53.09%:16.08%, respectively, revealed the successful synthesis of the Fe-MOFs (Singh et al., 2024).
XPS analysis
To confirm the data obtained from the EDX analysis, the XPS was performed as shown in Figure 2(d). The Fe 2p peaks at 716.85 eV with a full width at half maximum (FWHM) of 11.17 eV indicate the presence of iron, accounting for 30.22% of the sample. The O 1s peak at 534.12 eV, with a FWHM of 5.78 eV, corresponds to oxygen species, primarily from the carboxylate groups and hydroxyls, contributing to 53.57% of the sample's composition. The C 1s peak at 288.17 eV with a FWHM of 5.73 eV represents carbon, mainly from the organic ligands, making up 16.21% of the sample. Overall, the XPS data confirm a high oxygen content and significant contributions from iron and carbon.
FTIR spectra analysis
To confirm the successful conversion of the anhydride to the acid, Figure 2(e) shows the FTIR spectrum of the product, showing the disappearance of anhydride-specific peaks at around 1730 and 1850 cm−1. In contrast, a broad O–H stretching band emerged near 3100–3300 cm−1. According to Figure 2(e), the FTIR spectrum shows significant changes where the C=O stretching vibration shifts to 1600 cm−1, indicating the formation of metal–carboxylate bonds. The O–H stretching band is greatly diminished or disappears, reflecting the deprotonation of the carboxylic groups. A new strong band appears at 580 cm−1 corresponding to the Fe–O stretching vibrations. These specific changes in the FTIR spectrum confirm the successful coordination of BTC with iron to form the Fe-MOF (Zhao et al., 2017).
Raman spectra analysis
The Raman spectrum of the synthesized Fe-MOF is shown in Figure 2(f). Several distinct vibrational bands were observed, confirming the coordination between the BTC ligand and iron centers. Notable peaks include: a broad band at 1560 cm−1, attributed to the asymmetric stretching vibrations of the carboxylate groups (–COO−) coordinated to Fe3⁺ ions. A band at 1382 cm−1, assigned to the symmetric stretching of carboxylate groups, further indicates strong metal–ligand interactions. The presence of peaks at 650 and 513 cm−1 is characteristic of Fe–O vibrations, confirming the successful coordination of Fe centers with the organic linker.
Zeta-potential analysis
Zeta-potential measurements were conducted to evaluate the surface charge and colloidal stability of the Fe-MOF particles in aqueous suspension. The recorded zeta-potential was +24.9 mV, as shown in the accompanying distribution plot (Figure 2(g)). A positive zeta-potential of this magnitude indicates: moderate electrostatic stability, reducing the likelihood of particle aggregation in dispersion. A positively charged surface, which may enhance interactions with anionic dye molecules such as TOO and SY, potentially improving adsorption efficiency through electrostatic attraction.
BET analysis
Figure 2(h) illustrates the N2 adsorption–desorption isotherm of the Fe-MOF adsorbent. According to the figure, the profile exhibits a type IV with a distinct hysteresis loop, confirming the mesoporous nature of the material. The BET data obtained revealed that the Fe-MOF has a specific surface area of 157 m2/g, a total pore volume of 0.1895 cm3/g, and a mean pore diameter of 4.83 nm. These results suggest that the adsorbent contains mesopores, which facilitate the diffusion of dye molecules to the active sites.
VSM analysis
Figure 2(i) shows the VSM analysis curve of the Fe-MOF adsorbent. The VSM test studies the magnetic properties of the Fe-MOF adsorbent, revealing a typical S-shaped profile with negligible hysteresis, which indicates a superparamagnetic nature. The adsorbent showed a saturation magnetization (Ms) value of 5.5 emu/g, a very low coercivity (coercive field [HC] = 9.14 G), and a retentivity (Mr) of 0.127 emu/g. These data show that the Fe-MOF can be easily magnetized under an external field and loses its magnetization when the field is removed. This property is desirable for the recovery of the adsorbent. This magnetic behavior, along with the adsorption and antibacterial performance of the Fe-MOF, enhances its potential as a multifunctional and reusable material for wastewater management.
Factors affecting adsorption
pH of the solution
The pH of the solution is a critical factor in adsorption processes as it affects both the adsorbent's surface charge and the adsorbate's ionization degree. In this study, the optimum removal efficiency (%Re) was obtained at pH 3 for both TOO and SY dyes, as presented in Figure 3(a). At lower pH values, the surface of Fe-MOF is expected to become positively charged due to the protonation of surface functional groups, as observed in similar Fe-MOFs (Chong et al., 2021). This enhances electrostatic attraction between the positively charged Fe-MOF surface and the negatively charged sulfonate groups of the anionic dyes. As both TOO and SY are anionic in nature, the low pH facilitates stronger interactions, leading to increased adsorption efficiency. Still, at higher pH values, the surface charge of the Fe-MOF may become neutral or negative, reducing the electrostatic attraction and thus decreasing the adsorption efficiency (Sun et al., 2022).

Different operating parameters for dye removal using Fe-MOF. Fe-MOF: iron-based metal–organic framework.
Contact time
Figure 3(b) illustrates the effect of time on the removal of TOO and SY dyes. As time increases, the %Re increases to equilibrium, where the active sites of the Fe-MOF become saturated with the dyes. Upon comparing the two dyes, the SY dye was found to be the faster. It yielded higher removal than the TOO, which could be attributed to the greater number of functional groups contained in this dye (Figure 1). The ideal contact time ensures maximum adsorption efficiency without unnecessarily prolonging the process (Ahmad et al., 2021).
Effect of mass
Figure 3(c) and (d) shows that as the adsorbent's mass increases, the number of available active sites for dye molecules also increases. However, the ratio of dyes to the number of sites decreases, causing a decrease in the adsorption capacity (qe).
Regarding %Re, increasing m generally enhances the removal efficiency due to the greater availability of active sites, as shown in Figure 3(c) and (d). However, further increases in m may not significantly improve removal efficiency beyond a certain threshold because the adsorption sites become saturated, or excess adsorbent may aggregate, thereby reducing the effective surface area available for adsorption. For optimal adsorption, the mass of the adsorbent should be carefully controlled to maximize both the amount of dye adsorbed and the removal efficiency. This balance ensures that the adsorbent is used efficiently, providing the maximum removal capacity without waste (Singh et al., 2023).
Temperature
Temperature plays a critical role in the adsorption of TOO and SY dyes onto Fe-MOFs. In endothermic processes, higher temperatures increase molecular mobility, according to collision theory, which leads to more frequent and energetic collisions, enhancing adsorption. TOO reaches its maximum adsorption at 40 °C, where the increased temperature promotes the necessary molecular movement and diffusion into the MOF's porous structure. In contrast, SY achieves optimal adsorption at 30 °C (Figure 3(e)), where stronger interactions with the MOF occur at a lower temperature. Beyond the optimal temperature, increased thermal energy in exothermic adsorption processes disrupts molecular interactions, reducing adsorption efficiency. Therefore, optimizing temperature conditions is essential for maximizing dye removal, as adsorption decreases when temperatures exceed the optimal range for each dye (Zhang et al., 2023).
Dye concentration
The impact of initial dye concentration on the adsorption performance of Fe-MOFs was evaluated for two anionic dyes: TOO and SY (Figure 3(f) and (g)). The study was conducted by varying the concentration of each dye while maintaining constant conditions, including pH, temperature, contact time, and the mass of the MOF used as the adsorbent. For the TOO dye, Fe-MOF exhibited efficient removal of TOO at lower concentrations, with a maximum efficiency of approximately 84.8% at a concentration of 10 mg/L. As the initial dye concentration increased, the percentage of recovery (%Re) gradually declined. For instance, at 30 mg/L, the %Re dropped to approximately 51%, indicating that the available active sites on the Fe-MOF surface became saturated with the dye molecules, thereby decreasing the adsorption capacity at higher concentrations.
A similar trend was observed for SY, but with a comparatively higher removal efficiency. At low concentrations (e.g. 10 mg/L), the Fe-MOFs achieved a removal efficiency of approximately 93.7%. As the concentration of SY increased, the adsorption efficiency decreased, although it remained higher than TOO for equivalent concentrations. At 30 mg/L, the removal efficiency of SY was about 70%.
As a result of the factors mentioned above, the two dyes reveal that SY consistently demonstrates higher removal efficiency than TOO across all concentrations tested. This difference can be attributed to the stronger affinity of the Fe-MOFs toward SY, possibly due to differences in molecular properties such as size, molecular active sites, charge distribution, and binding interactions with the framework. As the concentration of the dyes increases, competition for the limited adsorption sites intensifies, resulting in a gradual decrease in the percentage recovery (%Re) for both dyes (Chen et al., 2023).
Effect of coexisting ions
The presence of coexisting ions such as NaCl, Na2CO3, NaHCO3, and Na2SO4 influenced the adsorption efficiencies of TOO and SY dyes, as shown in Figure 3(h). The addition of NaCl had a negligible effect on dye adsorption, suggesting that Cl− ions did not significantly interfere with the interaction between the dyes and the Fe-MOF. In contrast, the presence of Na2CO3 led to a decrease in dye removal efficiency, with TOO's removal percentage dropping from 84% to 62%, and SY's from 91% to 74%. This decline is likely due to competitive adsorption between carbonate ions and dye molecules for the active sites on the MOF surface. A similar trend was observed upon the introduction of NaHCO3, where bicarbonate ions likely competed with the anionic dyes, further hindering the adsorption process. The addition of Na2SO4 also resulted in reduced dye removal efficiencies, with TOO's adsorption decreasing to 70% and SY's to 81%, suggesting that SO42− ions likewise competed with the dyes for adsorption on the MOF. Additionally, these changes were not due to an increase in dye concentration (which would violate the Beer–Lambert law), but rather to less dye being adsorbed, thereby remaining in solution and leading to higher measured absorbance values comparable to those of Fe-MOF without ions.
To counteract this competitive effect, BaCl2 was introduced to the system, precipitating carbonate and sulfate ions as BaCO3 and BaSO4, respectively, and effectively removing them from the solution (Figure 3(i)). After this treatment, the dye removal efficiencies were restored: in the presence of carbonate ions, TOO's removal increased to 81%, and SY's to 85%; with sulfate ions, TOO's removal rose to 83%, and SY's to 88%. These results highlight the importance of managing interfering coexisting ions in dye removal processes and demonstrate that selective precipitation is an effective strategy for enhancing MOF performance in complex aqueous environments (Kumar et al., 2024).

FTIR analysis of adsorbed dyes
FTIR spectroscopy was employed to investigate the interaction mechanisms between Fe-MOF and the azo dyes TOO and SY. The spectra for Fe-MOF, the pure dyes, and dye-loaded Fe-MOF are presented in Figures 4 and 5. For the pristine Fe-MOF, characteristic absorption bands are observed at approximately 1600 and 580 cm−1, corresponding to the asymmetric stretching of carboxylate groups and Fe–O vibrations, respectively. These bands are retained in the dye-loaded spectra, indicating the structural stability of the Fe-MOF framework upon dye adsorption. In the spectrum of pure TOO dye, notable bands are seen at 1640 cm−1 (C=N stretching), 1443 cm−1 (aromatic ring vibrations), and 1120 cm−1 (S=O stretching), along with a broad peak at ∼3300 cm−1 indicative of the O–H or N–H stretching. After adsorption onto Fe-MOF, new bands appear at 2100 and 1390 cm−1, indicating shifts in the original dye peaks. These changes indicate successful adsorption and possible interactions, such as hydrogen bonding or electrostatic attractions, between the dye molecules and functional groups on the MOF surface. In the case of SY dye, characteristic bands at 1340 and 920 cm−1 are attributed to the azo (–N=N–) stretching and sulfonate (SO3−) vibrations, respectively. After adsorption, the dye-loaded Fe-MOF spectrum exhibits shifts in these peaks, along with the emergence of new signals at 1570 and 1415 cm−1. These alterations confirm the incorporation of SY dye within the MOF and indicate interactions. Overall, the FTIR analysis confirms the effective adsorption of both TOO and SY dyes onto Fe-MOF, with spectral shifts and the appearance of new bands providing evidence of specific interactions between the dye functional groups and the MOF surface.

FTIR spectrum of TOO dye adsorption by MOF. TOO: tropaeolin OO; Fe-MOF: iron-based metal–organic framework.

FTIR spectrum of SY dye adsorption by MOF. SY: sunset yellow; Fe-MOF: iron-based metal–organic framework.
XPS analysis of adsorbed dyes onto Fe-MOF
Figure 6 presents the XPS spectra of Fe-MOF after the adsorption of TOO and SY. For TOO, the XPS analysis reveals significant peaks for S 2p (10.83%), O 1s (51.58%), Na 1s (17.38%), and C 1s (19.75%), indicating that the dye primarily interacts with the MOF through its sulfur and oxygen groups, with minimal involvement of the iron sites (Fe 2p at 0.46%). In contrast, the XPS spectrum of SY shows a higher oxygen content (O 1s at 56.96%) and moderate carbon content (C 1s at 32.13%), along with a notable contribution from iron (Fe 2p at 6.22%), suggesting that the interaction with the MOF involves both the organic functional groups and the iron sites of the Fe-MOF. The minimal sulfur content (S 2p at 0.33%) indicates that sulfur groups play a less significant role in SY adsorption. The third XPS spectrum shows Fe-MOF, without any adsorbed dyes, providing a comparison of the MOF's surface composition before dye adsorption.
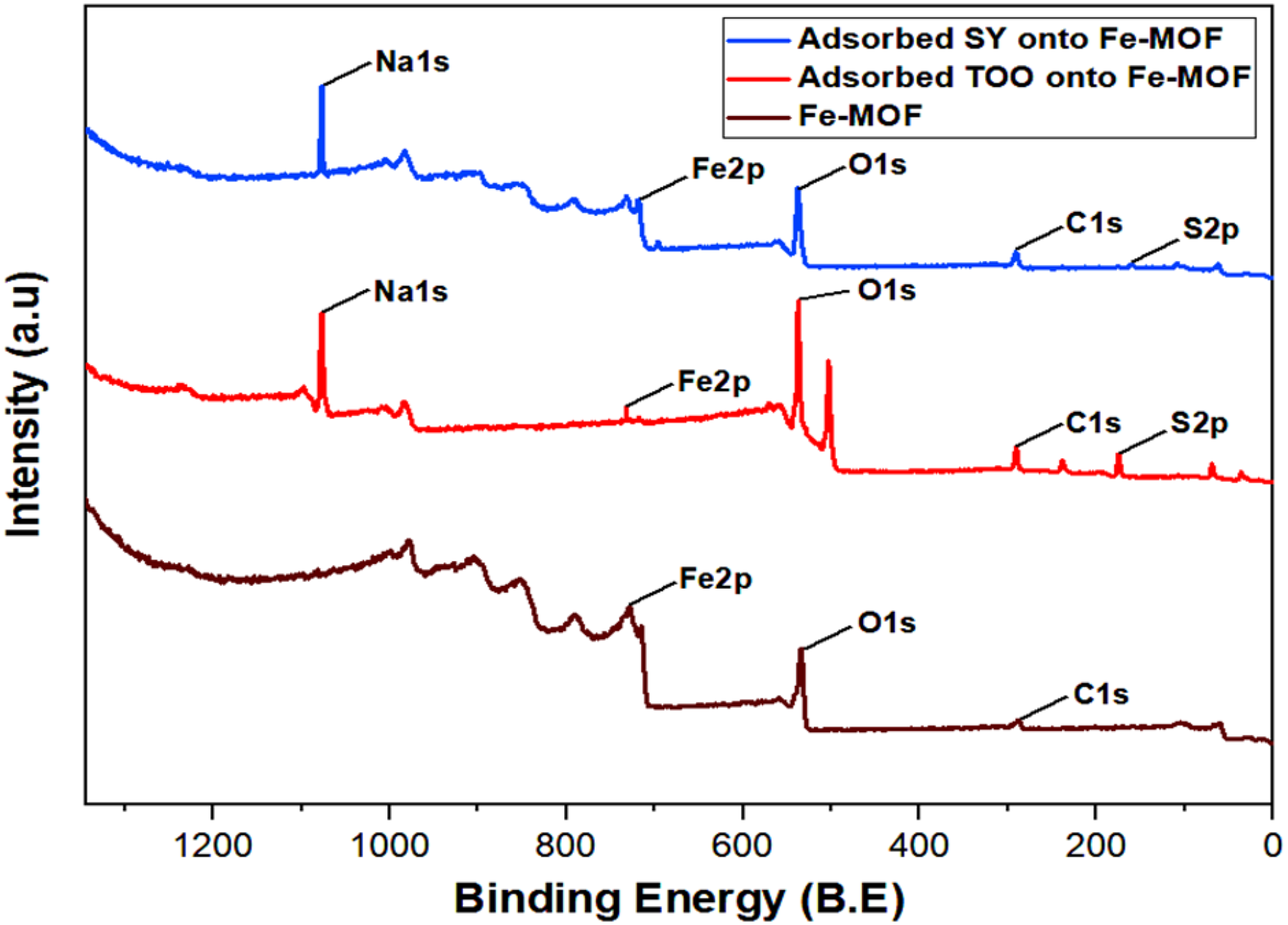
XPS after dye adsorption of TOO and SY dyes by Fe-MOF. TOO: tropaeolin OO; SY: sunset yellow; Fe-MOF: iron-based metal–organic framework.
Adsorption kinetics and isotherm studies
The adsorption isotherms and kinetics are beneficial in describing the adsorption mechanism (Wang and Guo, 2023). Several isotherm and kinetic models have been studied using the linear and nonlinear equations presented in Table 2, and their plots are shown in Figures 7, 8, and 9. As presented in Table 3, according to the correlation value (R2), the SY adsorption aligns better with the Langmuir model, indicating monolayer adsorption. In contrast, the TOO dye aligns with the Temkin model, underscoring the importance of adsorbate–adsorbent interactions.

Linear plots of different adsorption isotherm models, (a) Langmuir, (b) Freundlich, and (c) Temkin Models.

Non-linear plots of different adsorption isotherm models, (a) TOO dye, (b) SY dye.
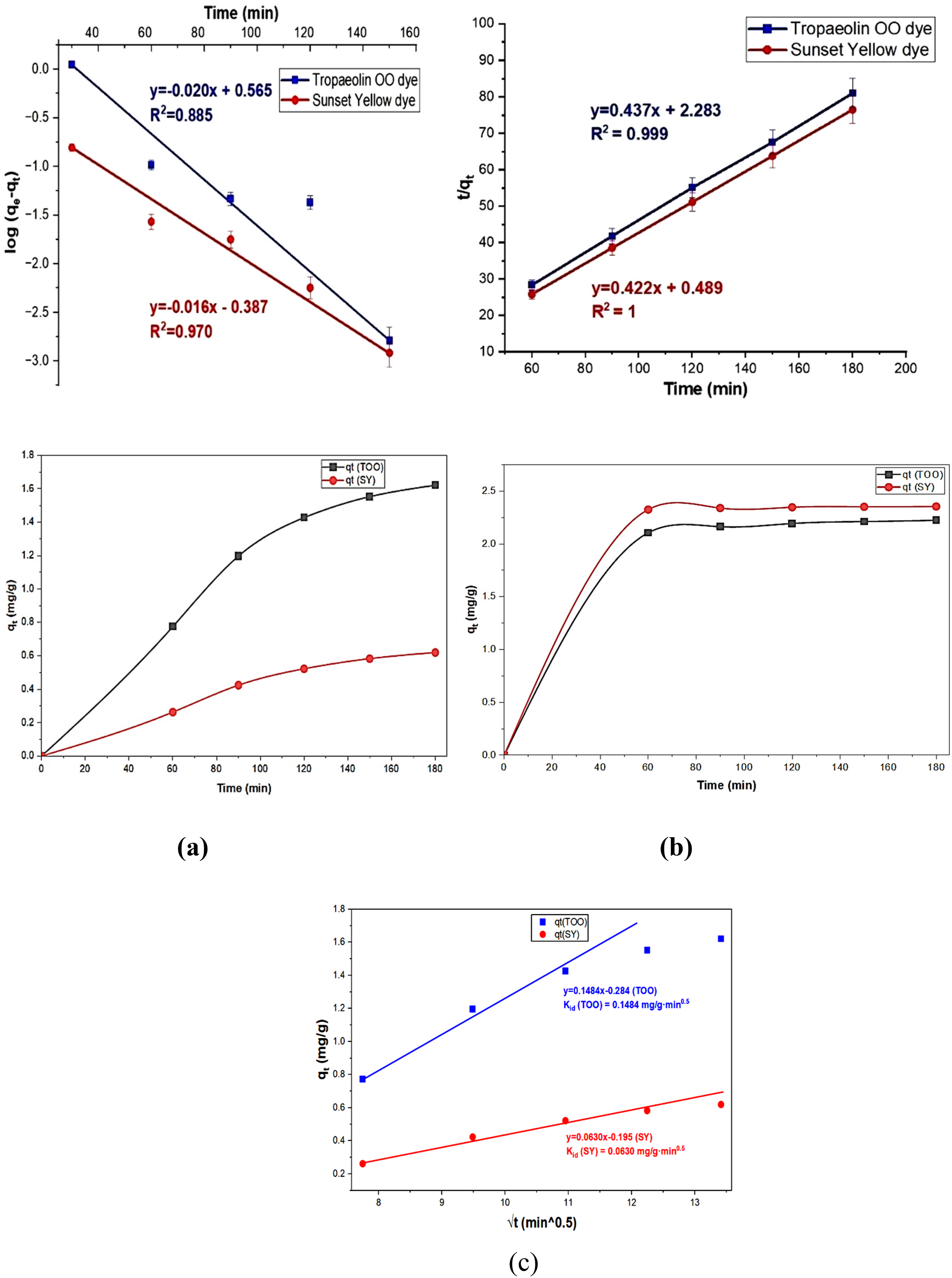
Linear and Non-linear plots of different kinetic models: (a) First-order Model, (b) Second-order Model, (c) Weber-Morrisintra-particle diffusion Model.
Linear and non-linear models of the adsorption isotherm and kinetics.

Adsorption isotherm and kinetic models for dye adsorption on Fe-MOFs.

Fe-MOF: iron-based metal–organic framework; TOO: tropaeolin OO; SY: sunset yellow.
In kinetic studies, both dyes are best described by a second-order model, indicating that chemical adsorption mechanisms predominate for both dyes, although SY exhibits a higher adsorption affinity. Furthermore, the values of qe calculated from the second-order model are nearly identical to the experimental value obtained from the experiment. The values of R2 and qe suggest that the removal mechanism follows the pseudo-second-order kinetic model.
Thermodynamic investigations
The thermodynamic parameters reflect the distinct adsorption mechanism and interactions of the dyes with Fe-MOF (Atkins et al., 2014). The changes in Gibbs free energy, enthalpy, and entropy were calculated using the equations presented in Table 4.
Thermodynamic parameters.

For the TOO dye, and according to Table 5, which illustrates the thermodynamic parameters for the dye removal, the
Thermodynamic parameters for dye removal by Fe-MOFs.

Fe-MOF: iron-based metal–organic framework; TOO: tropaeolin OO; SY: sunset yellow.
Selective adsorption study
Figure 10 presents the removal efficiency of a binary mixture of TOO and SY dyes as a function of adsorbent dose. The %Re of both dyes increased steadily with increasing Fe-MOF dosage, reaching approximately 69.3% and 75% for TOO and SY dyes, respectively. These values are lower than those obtained for each dye individually, which could be due to competition between the two dyes for the active sites of the adsorbent and possible repulsion between the dyes. However, SY dye showed a higher percentage of recovery (%Re) than TOO dye across all tested dosages and experiments, confirming its stronger affinity for the Fe-MOF. This behavior could be due to the higher number of functional groups in the SY dye compared to the TOO dye, as indicated by the structure in Figure 1, which enhances electrostatic and coordination interactions with the active sites of the Fe-MOF.

Effect of mass on % Re of a mixture of TOO and SY dyes.
Antibacterial activity
The antibacterial effectiveness of the synthesized Fe-MOFs was evaluated using nutrient agar media through a series of experiments involving five Petri dishes (Figure 11). The first dish contained wastewater without any addition to the MOF. In contrast, subsequent dishes added increasing amounts of MOF: 0.05, 0.1, and 0.2 g. A negative control plate containing only media was also prepared to assess the sterility of the agar. Initial bacterial cultures were inoculated onto the agar media, resulting in a bacterial count exceeding 1000 colony-forming units (CFU) in the wastewater sample. The bacterial counts recorded for each dish are presented in Table 6.

Anti-bacterial activity of Fe-MOF.
Inhibition ratio of Fe-MOFs and the bacterial counts recorded.

Fe-MOF: iron-based metal–organic framework; CFU: colony-forming units.
To quantify the extent of bacterial inhibition, the inhibition ratio was calculated using the following equation:
The results indicate that the Fe-MOFs exhibited a significant antibacterial effect, particularly at a concentration of 0.2 g, with an inhibition ratio of 96.47%. The reduction in bacterial count demonstrates MOF's effectiveness as an antimicrobial agent. The mechanism underlying this antibacterial activity is likely multifaceted. MOFs, especially those incorporating metals such as iron, can disrupt bacterial cell membranes and generate reactive oxygen species, inducing oxidative stress and ultimately leading to cell damage and bacterial cell death. Further research may elucidate the precise mechanisms at play, but the notable reduction in CFUs across the experiments provides clear evidence of the MOF's potential in antibacterial applications (Dhaka et al., 2019; Yan et al., 2022; Yuan and He, 2020).
Reusability and regeneration of Fe-MOFs
Figure 12 presents the two cycles of adsorption–desorption for the SY dye. The regeneration experiments showed a gradual decrease in the adsorption efficiency of the Fe-MOFs over successive cycles. The %Re of the dye was 96.79% before regeneration; however, it dropped to 93.70% and 87.10% after the first and second regeneration cycles, respectively. The regeneration efficiencies were calculated according to equation (5) and were found to be 96.80% and 92.95% for the first and second cycles, respectively, indicating a loss of performance (Asghar et al., 2019). This loss can be attributed to two factors: the degradation of active adsorption sites on the Fe-MOFs structure and the physical loss of material during the regeneration process.

Reusability of Fe-MOF 1st and 2nd regeneration.
FTIR analysis of regenerated Fe-MOFs
FTIR analysis of the Fe-MOFs was conducted before and after the adsorption processes, as shown in Figure 13. The results indicated that the characteristic bands of the Fe-MOFs remained consistent after dye adsorption and regeneration, suggesting that the structural integrity of the Fe-MOFs was largely preserved. This stability supports the potential for reusability. However, a gradual decline in adsorption efficiency is expected with each cycle due to inevitable material loss and degradation of active sites over repeated use.
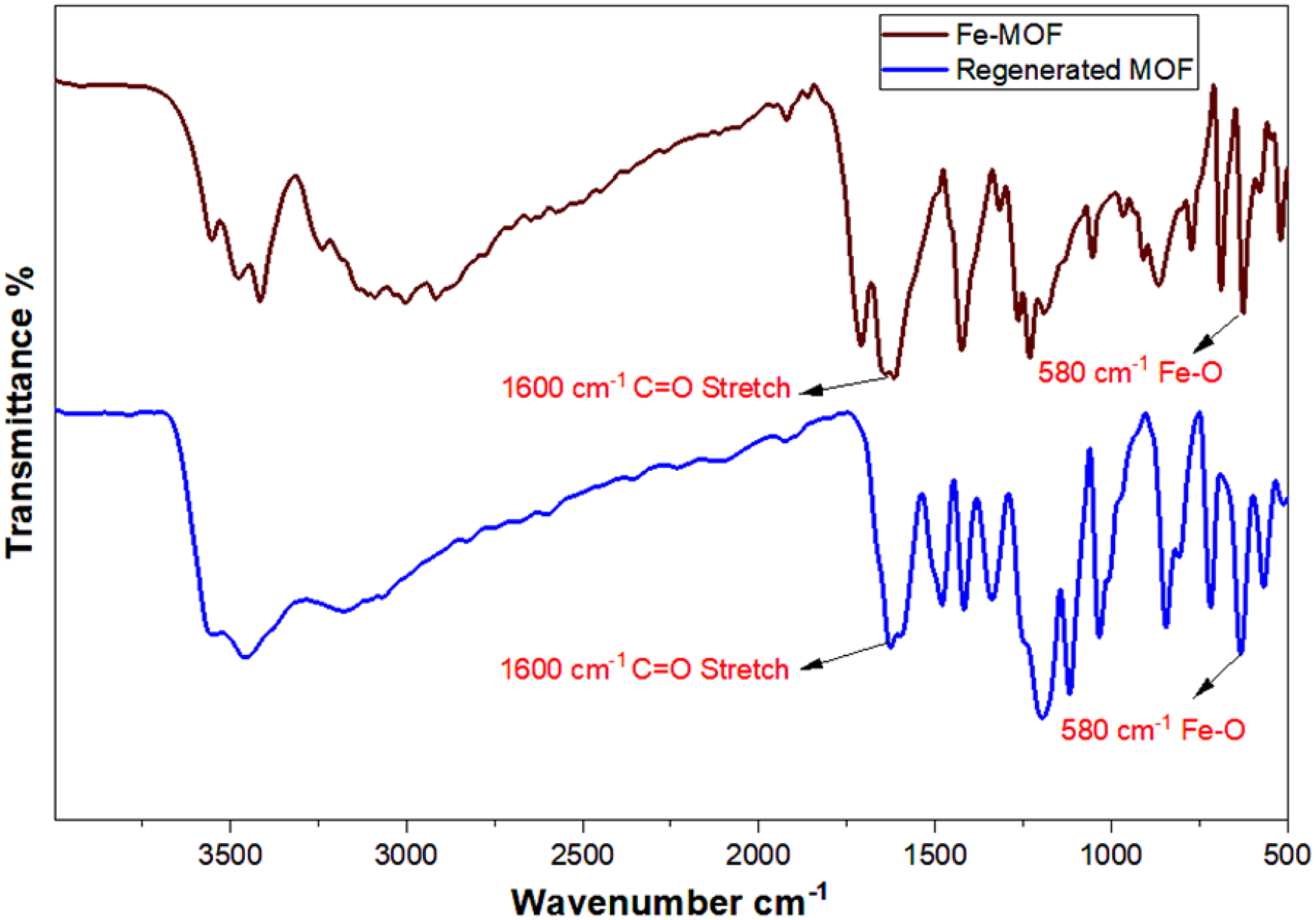
FTIR spectrum of synthesized and regenerated Fe-MOFs. Fe-MOF: iron-based metal–organic framework.
Comparison with the literature
Table 7 presents a comparison of the adsorption capacities of various Fe-MOFs and composites for different organic dyes. The reported qmax values are generally higher compared to the qmax values in our work, which could be due to several factors, including the lower initial concentration used in our study, as well as the different framework generated from the use of BTC with a different geometry.
Comparison of the adsorption capacities of Fe-MOF with other related adsorbents for organic dyes.

Fe-MOF: iron-based metal–organic framework; MIL: Materials of Institut Lavoisier.
Statistical analysis
Regression analysis
The regression analyses for the removal of TOO and SY dyes are presented in equations (6) and (7), respectively. These analyses indicate the impact of operational parameters on the percentage recovery (%Re). In both dyes, pH has a negative coefficient, indicating that an increase in pH leads to a decrease in %Re, which suggests that acidic conditions are more favorable for the adsorption in both dyes. The mass of adsorbent, on the other hand, has a strong positive impact on the %Re for both dyes, indicating that a higher adsorbent dosage significantly enhances the %Re due to an increase in the available active sites. For the contact time, the longer the interaction between the dyes and the adsorbent, the higher the %Re according to the positive coefficient obtained. Temperature has a minor negative effect on the percentage of removal (%Re), which suggests that the adsorption process may be exothermic; increasing the temperature slightly decreases the dye removal efficiency. The higher dye concentrations decrease the %Re due to the saturation of the available active sites for the TOO dye. However, for the SY dye, it shows a slight positive coefficient, indicating a minor increase in the %Re with higher concentrations, which may be due to adsorption mechanisms or surface interactions.

Analysis of variance
One-way ANOVA analysis was used to evaluate the effect of various operating parameters on the removal efficiency of TOO and SY dyes (Table 8). For both dyes, pH, mass of adsorbent, initial dye concentration, and temperature had statistically significant effects on the dye removal efficiency (p < 0.05), where the highest significant variables in both dyes were found to be the temperature and the mass of adsorbent, with F-values of 136.68 and 205.25 and 71.004 and 432.86 for TOO and SY dyes, respectively. The contact time did not show a statistically significant effect on the removal of either dye, as the p-values were greater than 0.05. These illustrations suggest that to maximize the removal of dyes, optimization of pH, adsorbent dose, dye concentrations, and temperature is crucial, whereas the contact time within the range of testing had a minimal impact
ANOVA results for TOO and SY dye removal with Fe-MOF.

ANOVA: analysis of variance; TOO: tropaeolin OO; SY: sunset yellow; Fe-MOF: iron-based metal–organic framework.
Conclusion
The present study successfully demonstrates the synthesis of Fe-MOFs using Fe(NO3)3·9H2O and 1,2,4-BTC as precursors, highlighting their effectiveness as multifunctional materials for water treatment applications. The Fe-MOF exhibited an excellent removal efficiency toward the TOO and SY dyes, along with notable antibacterial activity against microbial contaminants. A thorough characterization and statistical analyses gave valuable information on the structural, surface, and functional properties. The adsorbent also demonstrated excellent regeneration potential, with high efficacy after two cycles of regeneration. Accordingly, the dual-function ability of Fe-MOF demonstrates its sustainability and versatility in wastewater treatment.
Footnotes
Acknowledgements
The authors would like to extend their sincere appreciation to the Science, Technology & Innovation Funding Authority (STDF) with ID 46267.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
All data generated or analyzed during this study are included in this manuscript.



