Abstract
The increasing CO2 emission leads to significant ecological changes, and the control of CO2 emissions has been a major concern worldwide. Solid adsorbents are a highly promising carbon capture technology; the regeneration energy, visually representing the operating cost, is a key parameter to judge the merit of different solid adsorbents. In this paper, a uniform energy consumption calculation method was proposed to compare the characteristics of CO2 adsorbents in temperature swing adsorption process. The results showed that, for chemisorbents with strong interactions with CO2 (e.g. alkali and alkaline earth metal-based adsorbents), due to the high adsorption heat and the high regeneration temperature required, their energy consumption in CO2 capturing was quite high. It could be even higher than that of 30% monoethanolamine solution. Adsorption heat plays an important role in the cyclic regeneration of adsorbents; a very low adsorption heat (<25 kJ/mol) will make it difficult to form the difference in adsorption capacities between low and high temperatures. Among all the adsorbents, metal–organic frameworks, zeolites, and amine-functionalized adsorbents perform best, all of which possess moderate adsorption heats and large adsorption capacities at relatively low regeneration temperature.
Introduction
Background
Carbon dioxide (CO2) is the main anthropogenic greenhouse gas (Zhang et al., 2024); increasing carbon dioxide in the atmosphere is responsible for climate change (Song et al., 2025; Xue et al., 2022). In 2023, about 37.4 Gt CO2 was emitted to the atmosphere (CO2 emissions in 2023, 2023). The current concentration of CO2 sits at ∼420 ppm, which is excessively high as compared with what it was in the middle of the last century, and has become a huge environmental concern (Singh et al., 2020).
Of all CO2 sources, the electricity sector accounts for 36% of the total emission, greater than any other sector. To break down, coal remains the largest single source for electricity generation worldwide, and by far the largest source of CO2 emission in the electricity sector: it contributes just over one-third of electricity supply but is responsible for nearly three-quarters of CO2 emissions in the electricity sector (Birol, 2023; Dale, 2022). Therefore, decarbonization of power systems, especially coal-fired power plants, is of great importance for carbon reduction. And the current carbon capture scale is only about 40 Mt CO2, while the International Energy Agency (IEA) predicts that the carbon capture scale needs to reach 7602 Mt by 2050 (Birol, 2023). There is a great demand on the development of CO2 capture, utilization, and storage (CCUS) technologies.
At present, there are three ways to capture CO2, i.e. pre-combustion capture, post-combustion capture, and oxy-fuel combustion process (Soo et al., 2024). Among them, post-combustion capture technology can directly capture CO2 in flue gas by separating CO2 from N2 or other gas species. This is the most mature technology and can be used to retrofit the already-exist power stations (Hasan et al., 2024). It has demonstrated the most promising results in typical power plant configurations (Akeeb et al., 2022). Of China's current installed thermal power capacity, approximately 900 million kW will still be in service in 2050 (Cai et al., 2021). The incorporation of CCUS in coal-fired power plants is expected to capture 90% of the carbon emitted, enabling a relatively low-carbon power generation. The deployment of CCUS technology can help to fully utilize the existing coal-fired power units, avoid wasting resources due to the early retirement of some coal assets, and stabilize the electricity supply under extreme weather conditions. Therefore, post-combustion capture, which can be well adapted to existing power plants, becomes a practical decarbonization technical route for the future.
Application scenario
Before choosing a suitable carbon capture technology, what needs to be clarified is the application scenario, such as operating temperature, pressure, gas composition, and other conditions. Different operation conditions determine different capture technologies. Coal-fired power plant flue gas has typically the characteristics of moderate temperature (50–60 °C), huge flow rate (about 500 m3/s for a 600 MW unit), low CO2 content (10–15 vol.%), and humidified (6–10 vol.% moisture) (Ho et al., 2008; Sayari et al., 2011; Siegelman et al., 2021). As benchmark technology for carbon capture, 30% monoethanolamine (MEA) solution faces the problems of releasing toxic substances (Poste et al., 2014), equipment corrosion (Gunasekaran et al., 2017), amine degradation, and the most fatal drawback of high energy consumption (over 4 GJ/t CO2), which means that this technology will consume approximately 25–40% of the fuel energy and the additional cost will be approximately 70% (Haszeldine, 2009). Solid adsorbents can be a good solution to the above problems. First, there would be no problem of releasing contaminants and of corroding to equipment (Nie et al., 2018). Second, their relatively low specific heat capacity may reduce the regeneration energy consumption. This study will focus on the adsorbent energy consumption, by comparing different solid adsorbents with 30% MEA solution to determine the applicability of solid adsorbents.
Cycling process
Before selecting a suitable sorbent, it is also necessary to determine the cycling process. The cyclic process for solid adsorbents is shown in Figure 1(a), CO2 is separated from N2 in adsorption unit, then N2 passes through while CO2 remains. By changing the operation conditions, as shown in Figure 1(b), a difference in the adsorption capacities is formed and the desorption occurs to release CO2 and regenerate the adsorbent. Common CO2 capture cycle processes include pressure swing adsorption (PSA) (Karimi et al., 2024), temperature swing adsorption (TSA) (Derevschikov et al., 2023), vacuum swing adsorption (VSA) (Chatziasteriou et al., 2025), electrical swing adsorption (ESA) (Araoz et al., 2024; Jacobs et al., 2024), and their combinations (Chen et al., 2024; Gholami et al., 2024). As shown in Figure 1(b), both PSA and VSA take the advantage of the difference in adsorption capacities under different CO2 partial pressure to achieve cyclic CO2 adsorption. It is worth noting that Figure 1(b) does not represent the actual cyclic adsorption capacity of the cycle process, as the actual desorption should occur in a rather pure CO2 environment to ensure the purity of captured CO2 (Choi et al., 2016; Mcdonald et al., 2015). For flue gas with CO2 partial pressure of 0.15 bar, it may require quite high or low pressure (for PSA:

Solid adsorbent recycling process: (a) adsorption/desorption process and (b) cyclic adsorption capacity for different cyclic processes.
Ideal properties of adsorbents
Some researchers have summarized the ideal properties of superior CO2 adsorbents as shown in Figure 2 (Patel et al., 2017; Singh et al., 2020): high adsorption capacity (≥2 mmol/g), high selectivity for CO2 (

Principal criteria for an ideal sorbent for CO2 capture (Singh et al., 2020).
The calculation method of regeneration energy
Therefore, comparison of individual factors without considering the application scenario could hardly reveal the actual capability of solid adsorbents. From this point of view, it is particularly important to establish an intuitive index capable of evaluating the performance of each adsorbent in practical scenarios. Regeneration energy 
Regeneration energy consists of two parts: i.e. sensible heat and reaction heat. Sensible heat, corresponding to the first term on the right side of the equation, is the energy required to increase to the desorption temperature. Reaction heat is the second term on the right side, represents heat required to desorb CO2 from the sorbent. The data used for all calculations are given in Tables 1 and 2.
Regeneration energy consumption and corresponding parameters for different solid adsorbents.
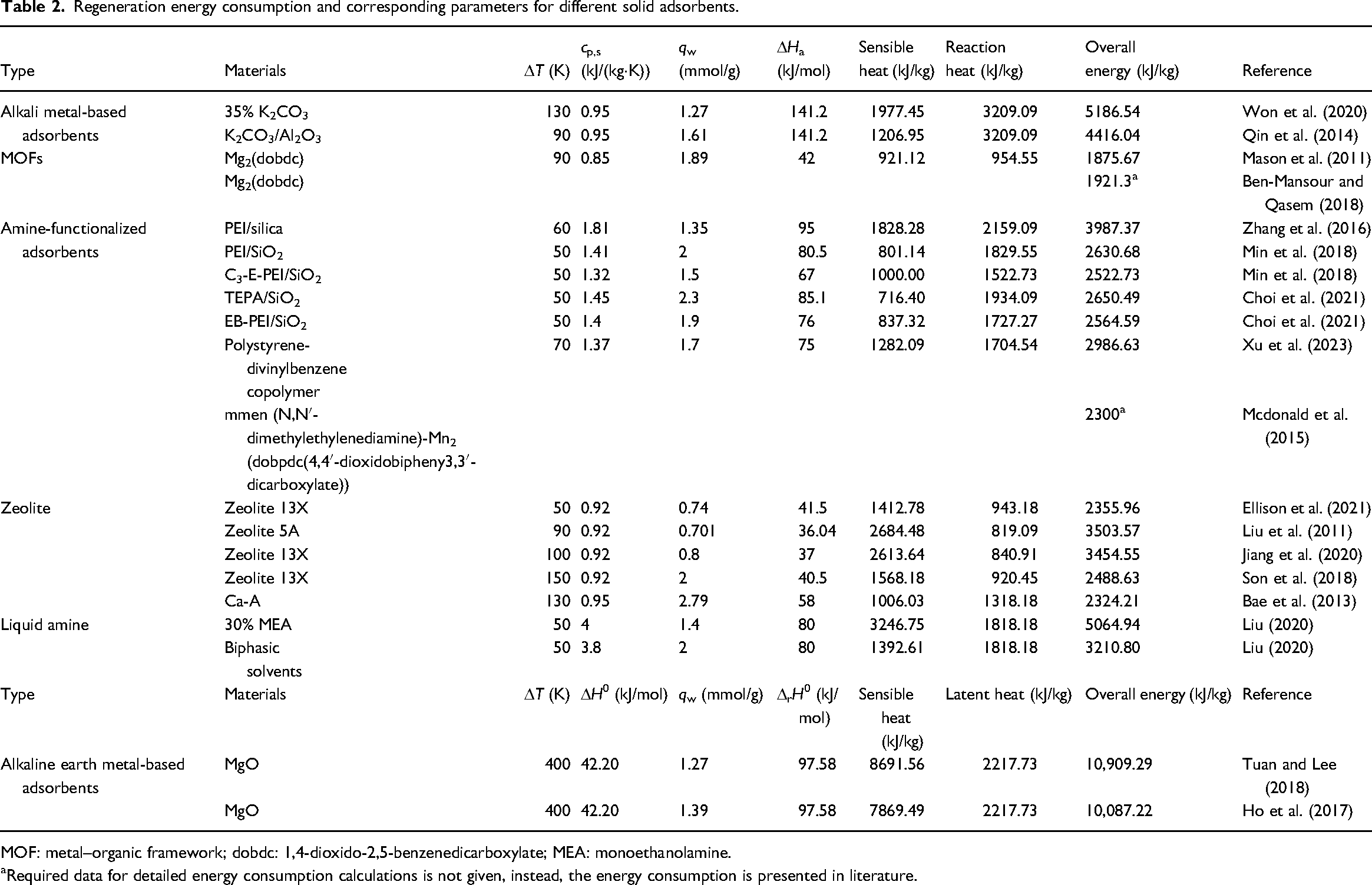
MOF: metal–organic framework; dobdc: 1,4-dioxido-2,5-benzenedicarboxylate; MEA: monoethanolamine.
Required data for detailed energy consumption calculations is not given, instead, the energy consumption is presented in literature.
To ensure the comparability of regeneration energy, the following regulations and assumptions are made: (1) Fixed flue gas outlet condition: no additional cooling or pressurization of flue gas is included, the temperature is 50–60 °C and the CO2 partial pressure is 0.15 bar. The flow rate of flue gas is too huge to be pretreated; large pressurized devices inevitably lead to very high construction and operation costs. (2) Pure CO2 gas in desorption: desorption under pure N2 conditions do enhance the cyclic adsorption capacity and regeneration rate, but is significantly different from the actual CO2 capture process. Desorption under other gas conditions would inevitably end up with CO2 gas mixture, after which a secondary separation process is subsequently required with additional cost and system complexity (Jahandar Lashaki et al., 2019). (3) Neglected heat recovery or heat transfer efficiency: the application of heat recovery do affect the overall energy consumption, but the performance of heat recovery devices is beyond of the scope of this study, which concentrates on the capability of adsorbent itself. (4) Neglected impact of other gas species (e.g. H2O and O2) on CO2 adsorption: the impact of H2O on the solid adsorbent has be detailed by Kolle et al. (2021) and Ray et al., (2021). In fact, for the subsequently mentioned adsorbents, most of them are strongly influenced by H2O, e.g. the competition between water and CO2 adsorption occurs on the surface of adsorbents that adsorb CO2 with metal cations (Kizzie et al., 2011; Mason et al., 2015), like zeolites and most metal–organic frameworks (MOFs). However, many scholars added hydrophobic material like polydimethylsiloxane into hydrophilic adsorbents with chemical vapor deposition method and found that the CO2 adsorption capacity of these adsorbents remained unchanged under moisture conditions (Jin et al., 2020; Zhou et al., 2022). Therefore, the regeneration energy under dry conditions still makes sense. In addition, the impact of H2O and other gas species is complicated, and the impact of water on the regeneration energy has been only studied for amine-functionalized adsorbents; there is a lack of support data for other sorbents. Even within the limited studies, the conclusions remain controversial. Some believe the H2O regeneration heat is constant and neglects the integrated role of water and CO2 (Choi et al., 2021), while the others believe the involvement of H2O in adsorption reaction with influences on regeneration heat (Siegelman et al., 2019). For the above reasons, the impact of H2O on regeneration energy is not considered in this study.
Purpose and content of the review
The review focuses on the regeneration energy of different adsorbents in the context of post-combustion CO2 capture in coal-fired power plants by TSA process. The regeneration energy of the liquid absorbers including 30% MEA and novel biphasic solvent is first calculated as a baseline, then the energy consumption of different solid adsorbents, including alkali metal-based adsorbents, alkaline earth metal-based adsorbents, MOFs, amine-functionalized adsorbents, zeolites, and porous carbon is calculated and compared with each other.
The cyclic regeneration energy of solid adsorbents has attracted more and more attention, but there is no comparison among the solid adsorbents yet. By comparing the regeneration energy of different solid adsorbents in TSA process, suitable adsorbents used for post-combustion carbon capture are found. And through the analysis of parameters affecting regeneration energy, such as cyclic regeneration capacity, specific heat capacity, and adsorption heat, the improvement direction of solid adsorbents in terms of reducing regeneration energy is determined. In addition, the lower limit of adsorption heat is given for the first time. Physical adsorbents like porous carbon with low adsorption heat (<25 kJ/mol) may not be suitable for post-combustion carbon capture in TSA process because they struggle to achieve adsorption capacity difference through temperature changes.
The regeneration energy consumption analysis of liquid amine
Ammonia solution is the most mature technology used for post-combustion carbon capture (Dutcher et al., 2015), which is the benchmark and has a large-scale commercial demonstration. The parameters of 30% MEA solution are shown in Table 1. Since the evaporation temperature of water is 100 °C, and the latent heat of water evaporation is large at 2260 kJ/kg. To avoid the evaporation,
To reduce the regeneration energy, the improvement in liquid absorbents includes the development of biphasic solvents, non-aqueous amine solvents, ionic liquids, etc. (Gao et al., 2020). Biphasic solvent could be divided into rich-liquid and poor-liquid layers after absorbing CO2, and regenerating only the rich-liquid can effectively reduce the mass of absorbent regenerated, thus reducing the heat-up energy (Wang et al., 2018). Many researchers have already measured the regeneration energy of biphasic solvents with the distribution between 2 and 2.2 MJ/kg CO2 (Liu et al., 2019; Lv et al., 2019; Shihan et al., 2018; Zhang et al., 2018), but all with energy recovery. Liu (2020) found that as the overall CO2 adsorption capacity increases, the mass ratio of the rich-liquid phase gradually increases. When the adsorption capacity is 2 mmol/g, the mass ratio of rich-liquid is between 62% and 67%. The detailed parameters are shown in Table 1; the total energy consumption is calculated to be 3210.79 kJ/kg. Nearly 1800 kJ/kg is reduced compared with that of the conventional 30% MEA solution.
In addition, in the process of absorbent heat-up, although the temperature does not reach the boiling point of water, with the increase in saturation vapor pressure, there will still be part of the water vaporization. This part of water is difficult to calculate because it changes with experimental conditions, such as adsorption time and temperature rise rate. The energy used for water evaporation is large, leading to the development of non-aqueous amine solvents (Yang et al., 2019).
The regeneration energy consumption analysis of solid adsorbents.
Alkali metal-based adsorbents
The combining force between CO2 and alkali metal-based adsorbents is tight. The adsorption properties of Na-based and K-based adsorbents are close, and due to the stronger basicity, K2CO3 is regarded as a more effective adsorbent compared to Na2CO3 and Li2CO3 (Lee et al., 2006; Zhao et al., 2024). The adsorption reaction of these adsorbents requires water and can be expressed as (Wang et al., 2025):

K2CO3 often needs to be loaded on other materials to enhance its effectiveness (Angkanawisan et al., 2022; Zheng et al., 2024), like K2CO3//activated carbon (Liu et al., 2025), K2CO3/Al2O3 (Gao et al., 2024), etc. The CO2 adsorption capacity increases with increasing K2CO3 mass ratio. However, if the mass ratio is over 50%, the adsorption capacity would no longer increase but remains a plateau (Wang et al., 2019). The presence of support will result in a drop of the adsorption capacity per unit mass because most support materials cannot adsorb CO2. For example, the theoretical CO2 adsorption capacity of pure K2CO3 is 7.25 mmol/g, the maximum adsorption capacity of 50% load K2CO3-based adsorbent could hardly exceed 3.6 mmol/g. Considering the difficulty of reaching 100% utilization, the actual adsorption capacity may be even lower (Guo et al., 2021). The theoretical CO2 adsorption capacity is helpful in confirming the upper limit of the adsorbent. Xie et al. (2019) also pointed out that the optimal mass ratio of K2CO3 is 30–40%. The adsorption usually occurs at the temperature of 50–80 °C, while the desorption occurs at 120–200 °C (Wu et al., 2023; Zheng et al., 2025).
Won et al. (2020) determined a cyclic adsorption capacity of 1.27 mmol/g for a solid sorbent containing 35 wt.% K2CO3, under the adsorption temperature of 70 °C and the desorption temperature of 200 °C. The corresponding calculation parameters for this adsorbent are shown in Table 2. Its regeneration energy is 5186.54 kJ/kg, which is close to that of 30% MEA. Although the adsorption heat and the regeneration temperature of K2CO3-based adsorbent are much higher, its lower specific heat capacity leads to an overall medium regeneration energy. Qin et al. (2014) used 35% K2CO3/Al2O3 under the adsorption/desorption temperature at 60 °C and 150 °C respectively, and found the cyclic adsorption capacity of 1.61 mmol/g with decent cycle stability. The overall regeneration energy for this sorbent was 4416.04 kJ/kg.
Alkaline earth metal-based adsorbents
Alkaline earth metal-based adsorbents mainly include CaO and MgO; the reaction is:
Different from alkali metal-based adsorbents, this type of adsorbent does not need support and the reaction can occur in the absence of moisture (Alhalawani et al., 2025). CaO is commonly used in chemical chain combustion, which adsorbs CO2 between 600 °C and 700 °C and desorbs CO2 at about 900 °C (Afandi et al., 2024; Zhao et al., 2021b). Due to the high adsorption temperature, CaO is not suitable for post-combustion CO2 capture, so it will not be discussed any more.
MgO can adsorb CO2 at a relative low temperature (<100 °C) (Chai et al., 2024; Ruhaimi et al., 2021), but due to the tight combination, the desorption always occurs over 450 °C (Xu et al., 2024), which is quite high. Overall, alkaline earth metal-based adsorbents are more suitable for CO2 adsorption at higher temperatures (over 200 °C), as they can reduce energy consumption by lowering the temperature difference during adsorption and desorption processes. Furthermore, the cycle stability of MgO is bad because easy-sintering characteristics lead to a dramatical decrease of surface area; the adsorption capacity reduces by 10–50% in 10 cycles (Gao et al., 2018; Ho et al., 2017).
For substances with well-defined thermochemical properties, i.e. clear correlation of enthalpy and entropy with temperature before and after adsorption (Mcbride et al., 2002). Overall regeneration energy consists of two parts: one is heating sorbent after CO2 adsorption to desorption temperature, which equals to enthalpy difference of the sorbent between the adsorption and desorption temperatures. Another is heat required to desorb CO2 at desorption temperature, which can be calculated by subtracting the enthalpy of all products from the enthalpy of all reactants with the stoichiometric coefficient of CO2 as 1. Hence, equation (1) can be then rewritten as:


Equation (4) uses state function obtained from the thermodynamic properties handbook (Mcbride et al., 2002); the reliability of the calculated regeneration energy is greatly improved. The only variable in equation (4) is the cyclic adsorption capacity
Metal–organic framework
MOF, a relatively new type of material, is a porous crystalline material consisting of metal ions and organic ligands (Ge et al., 2024; Li et al., 2024). This combination of metal nodes and organic linkers produces a diverse structural library, and different MOFs differ a lot in their properties (Hui et al., 2025; Rohde et al., 2024). MOFs may have significant difference in hydrothermal stability, and similarly, may have significant difference in CO2 adsorption properties (Sher et al., 2024; Soleimani et al., 2024; Wan et al., 2023).
Mason et al. (2011) selected two typical MOF materials, i.e. MOF-177 and Mg2(dobdc) (dobdc4− = 1,4-dioxido-2,5-benzenedicarboxylate). The adsorption isotherms of the two materials at different temperatures are shown in Figure 3(a) and (b). The two vertical black dotted lines represent the CO2 partial pressure of 0.15 and 1 bar, respectively, corresponding to the partial pressure of CO2 in flue gas and the pressure of pure CO2 during desorption. To attain cyclic adsorption, the adsorption capacity under

(a) Adsorption isotherm of MOF-177 and (b) adsorption isotherm of Mg2(dobdc). MOF: metal–organic framework; dobdc: 1,4-dioxido-2,5-benzenedicarboxylate.
For Mg2(dobdc), the adsorption capacity is high at 0.15 bar and the adsorption capacity increases slowly with the further increasing pressure. The adsorption capacity at 60 °C and 0.15 bar is 3.85 mmol/g. At the pressure of 1 bar, when

(a) Variety curves of cycle adsorption capacity and energy consumption of Mg2(dobdc) with Δ
The reason for the two adsorbent materials showing huge differences in the adsorption properties is not only due to their own adsorption capacity, but also due to the difference in their adsorption heat. For MOF-177 and Mg2(dobdc), Δ
When the adsorption condition is fixed as
Amine-functionalized adsorbents
Amine-functionalized solid adsorbents have the same functional groups with amine solution, both uses amino groups to adsorb CO2 (Cui et al., 2025; Singh et al., 2025). Amine-functionalized adsorbents have several advantages over amine solution, for example, low corrosion, low volatility, low toxicity, and low specific heat capacity (ca. 1.5 vs 4.1 kJ/kg·K) (Du et al., 2024; Zhao et al., 2021a). Similar to K2CO3, amines also need to be loaded on other objects; common support materials include silica (Kikkawa et al., 2024), alumina (Wang et al., 2025), activated carbon (Hamdy et al., 2021), MOFs (Si et al., 2025), zeolites (Liu et al., 2020), resins (Tao and Dong, 2025), and porous polymers (Wang et al., 2024; Zhao et al., 2025). Most of the support materials are often mesoporous, which can ensure sufficient surface area and facilitate the migration of CO2 into the adsorbent at the same time (Yan et al., 2022).
The CO2 adsorption reactions are exothermic, thus the adsorption decreases as the temperature increases (Chen et al., 2018). However, for amine-functionalized solid adsorbents, the adsorption increases first and then followed by a fall with increasing temperature (Lin et al., 2023; Zhu et al., 2025), which is due to the competition between the migration resistance of CO2 and the exothermic adsorption reaction (Hu et al., 2025; Wang et al., 2025). Similarly, there is often an optimal amine loading (Yang et al., 2022).
As for the effect of amine type on CO2 adsorption, 1° amine often corresponds to the fastest reaction rate and the highest adsorption capacity if compared with 2° and 3° amine, and also it has the highest alkalinity and the highest adsorption heat. Moreover, 1° amine is unstable and has poor cycling stability (Didas et al., 2015). As the molecular weight of the amine increases or experiences alkylation, some 1° amine would be converted to 2° amine and 3° amine; the reaction heat, adsorption capacity, and adsorption rate would all tend to decrease (Min et al., 2018). In addition, 1° amine is more difficult to regenerate because it is more strongly bound to CO2, leading to a lower cyclic adsorption capacity. As shown in Figure 5, Choi et al. (2021) found that TEPA (tetraethylenepentamine)/SiO2 has an adsorption capacity as high as 5.36 mmol/g (60 °C, 0.15 bar), but only 43% of the CO2 can be desorbed. Meanwhile, the adsorption capacity of alkylated PEI/SiO2 (EB-PEI (1,2-epoxybutane-polyethyleneimine)/SiO2) is only 1.75 mmol/g, but 90% of CO2 can be desorbed. Although the adsorption capacity of these two adsorbents differs a lot, their cyclic adsorption capacity is very similar. This indicates that the mere pursuit of high adsorption capacity may not be very meaningful.

(a) Amine state distributions. CO2 and H2O adsorption–desorption profiles of (b) TEPA/SiO2, (c) EB-PEI/SiO2 under TSA process with adsorption (15% CO2, 10% H2O, 2% Ar in N2 balance) at 60 °C and regeneration (100% CO2) at 110 °C. Arrows represent the working capacities of CO2 (red) and H2O (blue). (d) The adsorption heat of CO2 and H2O (Choi et al., 2021). TSA: temperature swing adsorption.
The regeneration energy of amine-functionalized solid adsorbents is shown in Table 2. Zhang et al. (2016) experimentally measured the parameters of PEI/silica required for regeneration energy calculation, including cyclic adsorption capacity, specific heat capacity, adsorption and desorption temperatures, and adsorption heat. Its energy consumption was 3987.37 kJ/kg, lower than that of 30% MEA, but still energy intensive. Subsequent studies (Choi et al., 2021; Mcdonald et al., 2015; Min et al., 2018) showed that amine-functionalized solid adsorbents had lower adsorption heat, higher cyclic adsorption capacities, and hence lower regeneration energy.
Zeolites
Zeolites are microporous crystalline materials and mainly consist of two parts: metal ions and silicate/aluminate skeleton (Wang et al., 2024; Zhang et al., 2025). Since the aluminum–oxygen tetrahedra in zeolites are negatively charged, metal cations are needed to maintain electrical neutrality. Zeolites with higher CO2 adsorption have a lower silica–alumina ratio and are able to carry more metal cations. Unfortunately, metal cations are not only attracted to CO2 via a large quadrupole moment, but also attracted to water with high polarity (Yang et al., 2024), which leads to a significant decrease in the adsorption capacity of CO2 under moisture conditions (Xu et al., 2021; Yang et al., 2024). In addition, for molecules with different sizes, molecular sieves like zeolites with different pore sizes can achieve selective adsorption (Bai et al., 2022).
Typically, zeolites are not considered as an ideal CO2 adsorbent because it is well-known that zeolites are sensitive to moisture. While no matter in flue gas carbon capture or in direct air capture, there is much moisture. But in recent years, many scholars focus on the improvement of water-resistance properties of zeolites, as shown in Figure 6. Datta Shuvo et al. (2015) developed a new zeolite, i.e. SGU-29, whose adsorption capacity for CO2 may not be affected by water (Figure 6(a)). According to progressive change in the Fourier transform infrared spectra, the authors found that the adsorption peak of CO2 and H2O did not affect each other and inferred that CO2 and H2O have independent adsorption sites (Figure 6(b)). Zhou et al. (2021) found that FeO4 tetrahedra inside the 12-MR of Fe-MOR (mordenite monolith) zeolite could reduce the pore diameter to 0.33–0.34 nm (Figure 6(c)), which was slightly bigger than the kinetic diameter of CO2; CO2 can be selectively adsorbed through sieving effect. At the same time, the high Si/Al ratio enhanced the water-resistance properties.

(a) Ratio of CO2 capture adsorption capacity of different adsorbents under humid versus dry conditions. (b) Schematic illustrations showing the presence of only H2O-specific and CO2-specific sites but not sharing sites in SGU-29 and H2O-specific, CO2-specific, and H2O/CO2 sharing sites in NaX (Datta Shuvo et al., 2015). (c) Schematic illustrations of the formation of specific pore size of Fe-MOR zeolite (Zhou et al., 2021).
The regeneration energy of zeolites and the related parameters (Ellison et al., 2021; Liu et al., 2011; Neilsen et al., 2022) are shown in Table 2. Most zeolites have lower adsorption heat of about 40 kJ/mol and lower cyclic adsorption capacity (<1 mmol/g) when compared with other CO2 capture technology. However, Bae et al. (2013) found zeolite Ca-A having a higher adsorption heat of 58 kJ/mol and larger cyclic adsorption capacity. It is conjectured that there might be some positive correlation between the cyclic adsorption capacity and adsorption heat, and due to the opposite effect of these two parameters on the regeneration energy, there should be an optimal adsorption heat to minimize the regeneration energy.
Porous carbon
Porous carbon is also a popular adsorbent with low regeneration heat of 20–30 kJ/mol (Sun et al., 2025) and high adsorption capacity of >4 mmol/g (273 K/298 K, 1 bar) (Ma et al., 2022; Shen and Wu, 2025). In addition, porous carbon has the advantages of high hydrothermal stability, good water resistance, and low cost. Its application in CO2 capture has been widely studied (Deng et al., 2014); it mainly focuses on three areas: (1) element doping, e.g. N, S, and O (Fu et al., 2025; Shao et al., 2025); (2) pore size adjustment: micropores of <0.7 nm facilitate CO2 adsorption, and are less sensitive to the changes in temperature and pressure (Sethia and Sayari, 2015; Yang et al., 2025); and (3) produced adsorbents with different precursors, such as various biological or solid wastes. Porous carbon is also known as a carbon-negative technology due to the use of carbon-containing materials (Bai et al., 2023).
There are several problems associated with porous carbon in CO2 adsorption. The application scenario of porous carbon is not clear. Most studies set the adsorption condition as 298 K and 1 bar, which differs a lot from industrial carbon capture conditions. However, few studies describe the industrial background. With the increase of temperature and the decrease of CO2 partial pressure, the adsorption capacity and selectivity drop dramatically (Singh et al., 2022; Wang and Xuan, 2024). It is difficult to be used in high-temperature and low-pressure environments. Although porous carbon is considered as a carbon-negative material, in fact, chemical activation like NaOH and K2CO3 (Luo et al., 2025) is often required to obtain large adsorption capacity. The recovery of these alkaline substances and the preparation cost will also be an unsolved problem. Therefore, before studying how to improve adsorption capacities, it is necessary to consider the working scenario and corresponding recycle process.
We have failed to find out the recycle adsorption parameter of post-combustion carbon capture combined with TSA process. For most porous carbon adsorbents, their adsorption capacity at 60 °C and 0.15 bar are low, and as the temperature increases, the adsorption isotherms change very little (Ji et al., 2022), so it is difficult to create a cycle adsorption capacity. Therefore, it can be concluded that, porous carbon adsorbents could hardly be used for post-combustion CO2 capture via TSA process. This is consistent with the conclusion illustrated in Figure 4(b).
Comparison of regeneration energy of different adsorbents
The regeneration energy and the calculated parameters associated with all of the solid adsorbents mentioned above are listed in Table 2. Due to limited studies on desorption in pure CO2 environment, the data is relatively scarce. Nevertheless, the table highlights its value by screening for regeneration energy consumption under realistic application scenarios. Although there might be some advanced adsorbents not being explored, or the cyclic adsorption parameters are missing so that the regeneration energy cannot be calculated, the parameters listed in Table 2 do represent the general adsorption characteristics of this class of adsorbents. Furthermore, the economic viability of solid adsorbents is closely related not only to the regeneration energy but also to material costs, as discussed in detail in Halliday and Hatton (2021). However, some of the solid adsorbents in Table 2 are commercially mature, such as zeolites, alkali metal-based adsorbents, and alkaline earth metal-based adsorbents. While some still remain at the laboratory stage, such as MOFs, amine-functionalized adsorbents, and porous carbons, the development prospects and future pricing of these materials are difficult to predict. Therefore, the review focuses on the regeneration energy of solid adsorbents, which largely reflects their economic viability.
Since the thermodynamic properties of MgO and MgCO3 at different temperatures are easy to calculate, the enthalpy change and the reaction enthalpy can be used to calculate the total energy consumption. Only the cyclic adsorption capacity is needed to be measured from the adsorption–desorption experiment. The other variables can be obtained from the thermodynamic manual (Mcbride et al., 2002), thus the total energy consumption could be calculated rather accurately. For other adsorbent materials, it is difficult to calculate the energy consumption by this method because the thermodynamic properties of the substances itself or those after CO2 adsorption are unknown.
For chemisorbs such as alkali metal-based adsorbents and alkaline earth metal-based adsorbents, due to the strong combination of CO2, the regeneration temperature and the adsorption heat are usually too high, so the regeneration energy consumption is large, and even bigger than that of 30% MEA solution. Therefore, these materials may be difficult to be used for post-combustion CO2 capture. Materials with very low adsorption heat are not listed in Table 2 because they cannot form the adsorption capacity difference between adsorption and desorption conditions. Comparing the composition of overall energy consumption required for adsorbents with high reaction heat, such as alkali metal-based and amine-functionalized adsorbents, using materials with lower adsorption heat may be effective. For adsorbents with high sensible heat, e.g. zeolites and MOFs, increasing their cycle adsorption capacity might be the proper solution.
Conclusion and prospective
The combination of traditional fossil fuels combined with CCUS technology is a significant way to eliminate the negative impact of fossil fuels on the environment. The main obstacle that currently limits the large-scale development of CCUS technology is the high CO2 capture cost. This study focuses on the regeneration energy accounting for the most portion of capture cost, which is important to the application of solid adsorbents’ technology. A comprehensive discussion on the regeneration energy of various solid adsorbents is present with the purpose of clearing the ambiguities associated with CO2 adsorption cycles.
The regeneration energy of 30% MEA solution and advanced biphasic solvents was first calculated for comparison. The biphasic solvents greatly reduce the sensible heat by regenerating only the rich-liquid phase.
For alkali metal-based and alkaline earth metal-based adsorbents, they possess strong interactions with CO2. On the one hand, they need high regeneration temperature, for example,
Solid amine adsorbents, usually need to be anchored to other porous media by impregnation or grafting, reduce the utilization of the adsorption sites and the adsorption capacity. Compared with amine solution, due to the lower specific heat capacity, amine-functionalized adsorbents have lower regeneration energy.
The adsorption heat of MOFs and zeolites with good CO2 adsorption properties is higher than 35 kJ/mol, which ensures both the high adsorption capacity and the adsorption capacity difference under adsorption/desorption conditions. Materials with low adsorption heat (<25 kJ/mol), like porous carbon, cannot form the difference in adsorption capacity. Therefore, these types of physical adsorbents are hard to use for post-combustion carbon capture combined with TSA process.
Higher regeneration temperature corresponds to a larger cyclic adsorption capacity; the former leads to an increase in sensible heat while the latter leads to the opposite. Thus, there is often an optimal desorption temperature for an adsorbent.
From the previous discussion, the cyclic adsorption capacity may not be positively correlated with the adsorption capacity; it could be more meaningful to improve the cyclic adsorption capacity than increasing the adsorption capacity itself. Currently, the cyclic adsorption capacity is low, usually below 2 mmol/g. A major advantage of solid adsorbents over liquid adsorbents is the lower specific heat capacity. Except for amine-functionalized adsorbents, the mass specific heat capacity of most solid adsorbents is less than 1 kJ/(kg·K). The best performing adsorbents in terms of regeneration energy tend to have moderate adsorption heat, such as MOFs, zeolites, and amine-functionalized adsorbents.
Further work is required in applying solid adsorption technology for carbon capture at coal-fired power plants. First, solid adsorbents with lower adsorption heat and larger cyclic adsorption capacity at a lower regeneration temperature worth the effort of further developing. Second, although less discussed in the review, impurities in the flue gas like O2, H2O, and low levels of nitrogen oxides and sulfur oxides may affect the CO2 adsorption properties. Developing adsorbents with high tolerance to impurities or establishing low-cost pre-desorption processes is crucial. Third, current energy consumption remains at an unacceptable level. Appropriate energy recovery processes, such as heat pump technology, can be developed to recover heat generated during the adsorption process for reuse in the desorption process. Finally, the review addresses only thermodynamic aspects; researchers should also focus on the overall duration of the adsorption/desorption process, primarily encompassing the adsorption/desorption rate of adsorbents and the heating and cooling rates of adsorption equipment.
Supplemental Material
sj-docx-1-adt-10.1177_02636174251385608 - Supplemental material for The comparison of regeneration energy of different solid adsorbents in temperature swing adsorption process: A review
Supplemental material, sj-docx-1-adt-10.1177_02636174251385608 for The comparison of regeneration energy of different solid adsorbents in temperature swing adsorption process: A review by Shun Wang, Shujuan Wang and Yuqun Zhuo in Adsorption Science & Technology
Footnotes
Ethical approval
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundamental Research Funds for the Central Universities (grant number No. 2022ZFJH04).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data and code availability
Supplemental material
Supplemental material for this article is available online.
Appendix 1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

