Abstract
This work presents the results of the investigations of the adsorption process of nitrate, nitrite, and ammonium ions on carbon adsorbent with the application of the solid phase extraction method to isolate and enrich the investigated ions from aqueous solutions of the concentration below 1 mg/L. For the determination of the concentration of the analyzed ions, we applied the ion chromatography method. The sorption capacity for the tested adsorbents as well as the efficiency of the enrichment of the investigated ions were determined.
Introduction
Nitrogen compounds are necessary for the proper functioning of the environment and living creatures, because nitrogen is an element of biogeny. However, the presence of nitrogen ions in excess in different forms results in the negative effects on the ecosystem, and consequently it can damage human health and even kill. This component is present in the continuous circulation cycle. In water, it creates inorganic compounds, mainly nitrate ions, nitrite, and ammonium. In these forms, it occurs in drinking water as well as in the surface or underground water (Bielicka-Giełdoń et al., 2014). Too high levels of nitrogen compounds are dangerous for people; therefore, it was necessary to create specific standards and conditions, which should be proper for the water for human consumption (Stepnowski et al., 2010). The maximum permitted amount of nitrogen compounds can reach the following values: 0.50 mg/L NO2− ions and NH4+ ions, and 50 mg/L for NO3− ions. These recommendations have been introduced in the European Union and in Poland. The recommended standards of the amount of nitrite in water intended for human consumption were applied primarily to prevent methemoglobinemia (cyanotic). These compounds are mainly harmful for infants, but in some rare cases also for adults (Fan and Stienberg, 1996).
The purpose of this work was to investigate the possibility of applying carbon adsorbents for the separation and enrichment of nitrite, nitrate, and ammonium ions from aqueous solutions. The solid phase extraction (SPE) was applied as the method of enrichment of the determined ions.
The adsorption processes of nitrate ions were the object of research of a few authors (Balci, 2004; Hanafi and Azeema, 2016; Jankowska et al., 1991; Kalantary et al., 2016; Mizuta et al., 2004; Öztürk and Bekta, 2004; Zhang et al., 2014). These studies were carried out for the removal of nitrate ions from water. The authors of these works use carbon adsorbent with a very large surface area up to 2800 m2/g (Hanafi and Azeema, 2016), or synthetic activated carbon with magnetic nanoparticles (Kalantary et al., 2016). The aim of our work was to research on the possibility of the use of carbon adsorbents to isolate and enrich nitrate, nitrite, and ammonium ions from the aqueous solutions to determine their concentration in low concentrations below the level of determination of the instrumental analysis methods. In our investigations on the nitrate ions determination, at first we used the solid phase extraction method (SPE) and next for its quantitative determination we applied the ion chromatography technique with conductometric detection.
Adsorption ions on activated carbon
Activated carbon had already been known and used before our era, and from 1929, for the first time, it was used for the purification of drinking water. Carbon adsorbent possesses high sorption capacity, its surface area reaches the value of up to 2500–3000 m2/g (Dąbek, 2007). The adsorption of ions does not depend only on the specific surface area of the adsorbent but also on the presence of the surface groups. The oxidation of active carbons can lead to the increase amount of the oxygen functional groups on the surface of the adsorbent, and the result of this is an increase in polarity and hydrophilicity of the surface of the carbon adsorbent. The applied oxidizers as well as the conditions applicable during the modification of carbon adsorbent influence the content and nature of the emerging oxygen functional groups (Figueiredo et al., 1999; Jankowska et al., 1991). The surface of carbon adsorbents, due to the chemical properties, is divided in two kinds of functional groups:
acidic functional groups of, among them are the carbonyl, lactone, phenolic, carboxylic, anhydrous, alkaline groups, such as chromanone and others (Figueiredo et al., 1999; Jankowska et al., 1991).
The carbon materials that have been chemically oxidized in the gas phase within the limits of the temperature of 300℃ to 500℃, or oxidized in the liquid phase can exchange cations (Jankowska et al., 1991).
Mechanisms that can be distinguished in the adsorption process of electrolytes on the carbon adsorbent occur as: equivalent exchange of ions, the electrochemical exchange which occurs between ions of electrical double layer of the adsorbent and ions in a solution of electrolyte, molecular mechanism of adsorption of the whole molecules of the substance on the surface of the carbon material, adsorptive mechanism of quasi-molecular, in case when strongly polarizing ions are present in the electrolyte (Jankowska et al., 1991).
The solid phase extraction process is a dynamic process of adsorption and it can be illustrated graphically with curves, which, in this case, are called breakthrough curves of the exchange or 
Experiment
In these researches, we used commercial carbon AG-5 (GRYFSKAND) and carbon oxidized with 30% hydrogen peroxide solution. The surface area (SBET) of the commercial carbon AG–5 equaled 750 ÷ 850 m2/g (
The concentration of the investigated ions was determined by ion chromatography. Quantitative analysis was carried out based on calibration curves for nitrate and nitrite ion for which we received the coefficient of determination (R2) close to unity (equal 0.99). The method of the calibration curve was also used for the determination of concentrations of ammonium cation. Calibration curve for this ion also had the coefficient of determination (R2) close to unity (equal 0.98).
Adsorption of nitrate and nitrite ions was performed on the SPE column filled with an initial carbon AG-5 or the oxidized carbon AG-5. The sorption process went on as follows: a portion 100 or 500 mg carbon were deposited in SPE column, next they were washed with three portions of 2 ml of methanol and 2 ml of deionized water, and then by the SPE column the aqueous solutions of the investigated ions in quantities from 20 ml up to 700 ml was passed, at a flow rate of 1 to 3 ml/min. The individual parts of the investigated ions passed by SPE column were analyzed on the ion chromatograph and received the dependency graph of the quotient c/c0 from the volume of the applied solutions (where c is the concentration of NO3−, NO2−, or NH4+ ions in the flowing portions of the filtrate, c0 is the concentration of the analyzed ions, Figures 1(a) and 2(a)). The process is graphically presented on the curve in Figure 1(a) for NO2− ions adsorbed on carbon adsorbent AG-5.
(a) Breakthrough curve in the process of the SPE (c/c0 = f(V)) for nitrite ions on adsorbent AG-5 (mcar = 0.502 g, concentration of ion cNO2- = 0.135 mg/L). The ratio c/c0 for each 10 ml of the filtrate is designated. (b) The desorption curve (c = f (V)) of nitrite ion from adsorbent AG-5 after analysis each 2 ml of the eluent passed through SPE column. (a) Breakthrough curve in the process of the SPE (c/c0 = f(V)) for ammonium ion (NH4+) on the oxidized carbon AG-5. (mcar = 0.104 g, concentration of ions cNH4+− = 0.661 mg/L). The ratio c/c0 for each 5 ml of the filtrate is designated. (b) The desorption curve (c = f(V)) of ammonium ion from oxidized carbon adsorbent AG-5 after analysis each 2 ml of the eluent passed through SPE column.

The second part of the research referred to the process of adsorption of ammonium ions from aqueous solutions on the initial carbon AG-5 and on the oxidized adsorbent AG-5. The process was carried out in the same conditions as for the anions of nitrates and nitrites, by passing the solution of ammonium nitrate through SPE column packed with test adsorbents.
After the adsorption step, desorption process was conducted (for ions NO3−, NO2−, and NH4+) in the following ways: by SPE column small portions (2 ml) of the eluent were passed, the same which were used as the mobile phase of ion chromatography. Every portion of eluents passed through the column was analyzed chromatographically and desorption curves were drawn up (elution curves), i.e. the graph of the concentration of the designated ions from the volume of the applied eluent phase (c = f(V)) (Figures 1(b) and 2(b)).
Sorption capacity of the adsorbents
The sorption capacity of the tested adsorbents was calculated from the experimentally received data: the working sorption capacity (for c/c0 equal 10%) and the total sorption capacity (for c/c0 equal 50%). Sorption capacity (q) was calculated from the formula:

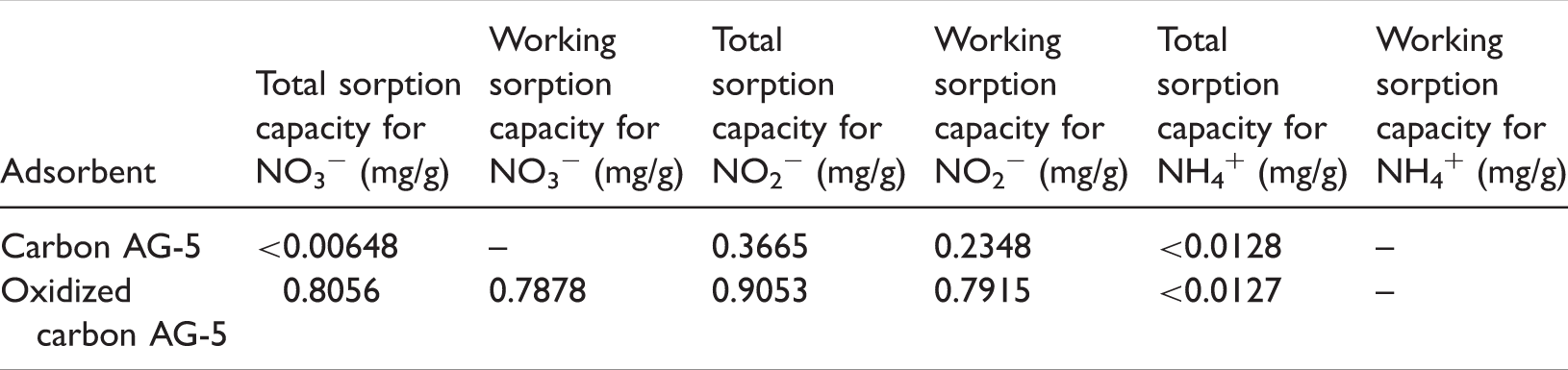
The sorption capacity of the tested adsorbents in terms of ions: NO3−, NO2−, and NH4+.
Recoveries of NO3− ion in SPE process.

Conclusions
Summing up, on the basis of the obtained results, we can conclude that the investigated adsorbent, i.e. initial carbon adsorbent AG-5 can be used for isolation and enrichment of nitrate ions from aqueous solutions with a concentration below 1 mg/L after enrichment with solid phase extraction method (SPE). In the dynamic process of adsorption, i.e. solid phase extraction test, we obtained the breakthrough curves like the theoretical curve. Oxidation of activated carbon AG-5 gives you a higher total and working capacity and sorption in relation to ion NO3−, but at the same time, it reduces the efficiency of the recovery of the nitrate ions in the SPE (see results in Tables 1 and 2). In case of adsorption of ammonium ions for tested carbon adsorbents, we obtained similar values of the total sorption capacity (for initial and oxidize carbon adsorbents) (Table 2).
From the analysis of the obtained results, it can be concluded that:
the efficiency of the nitrate ion enrichment on the SPE column does not depend on the amount of polar groups on the surface of the carbon adsorbent, because carbon AG-5 after oxidation (i.e. richer in function groups) gives lower recoveries than carbon not oxidized. what is more, the efficiency of ion enrichment is not a function of the total ion exchange capacity of carbon adsorbent, as the oxidized carbon, with a higher total capacity of ion exchange in relation to the measured ion (i.e. ions NO3−; Table 1) gives almost twice lower efficiency of enrichment nitrate ions (respectively ∼97 and ∼57% - Table 2).
Authors of the article (Zhang et al., 2014) suggested that the adsorption rate for carbon adsorbent depended on the availability of adsorption sites rather than on the ions concentration in solution, but it would have rather related to ion-exchange mechanism in double electric layer around the carbon adsorbent, and not only with the presence of the polar functional groups on its surface. Although, on the other hand, the increase in the number of ions in a double layer of carbon may be the result of the increase in the number of polar functional groups on its surface.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support received from the Polish Ministry of Higher Education and Science (UJK BS 612 490).
