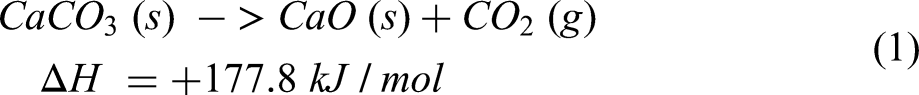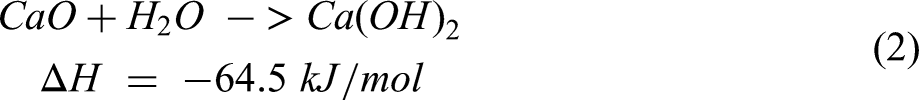Abstract
Quicklime is a noninterchangeable multi-purpose industrial product produced by heating and calcining limestone in high-temperature kilns. Quicklime slaking reactivity (reaction rate and heat development when mixed with water) is a key quality parameter of quicklime, and it is influenced by characteristics of the limestone used as well as by the calcination conditions. In this paper results are presented from a systematic experimental study focused on how the slaking reactivity of quicklime calcined in CO2 atmosphere depends on calcination parameters (time and temperature). Limestone samples were calcined at a range of temperatures (1000–1200°C) and times (10–60 min.). CO2 atmosphere was chosen to simulate the conditions of electrified industrial quicklime production, which is of relevance for emerging technologies aiming at efficient CO2 separation and sequestration. The calcination parameters and specific surface area of the quicklimes produced were correlated with slaking reactivity. The results show that the calcination temperature has greater effect on the slaking reactivity than calcination time within the conditions tested. It was also found that the slaking reactivity was not easily explained by the measured specific surface area. The findings show that, from a reactivity point of view, there is potential for an electric calciner to, combined with carbon capture and storage (CCS), provide a solution for zero-emission production of quicklime of controlled medium to high reactivity.
Introduction
Limestone and quicklime are among the oldest construction materials. There is evidence for them having been used by the Egyptians for plastering in the construction of the pyramids around 2500 BC. The Romans later developed the structural properties and uses of quicklime by producing slaked lime and mixing it with volcanic ash, which was used as a mortar to build pavements and slabs.1,2 In addition to being used for construction purposes, quicklime also has metallurgical, chemical, and agricultural applications. In steel production quicklime is used as a flux and slag-forming agent. 3 Industrial production of steel highly contributes to CO2 emissions, and therefore there are many on-going studies aiming for zero-carbon steel production,4,5 in which CO2 neutral quicklime needs to be considered. Another application of quicklime is in Kraft pulping plants, 6 where calcium oxide reacts with waste sodium carbonate solution to regenerate sodium hydroxide. Quicklime is also used in sludge treatment to remove numerous pollutants from biological waste. 3 One of the oldest applications of quicklime is in agriculture, 1 and it has also been demonstrated that quicklime can improve the aggregate stability of agricultural soils. 7
Quicklime is produced in industrial kilns by calcination of limestone at temperatures of 1100–1450°C, 8 according to endothermic reaction (1).
The most common conventional lime-kiln technologies are rotary and shaft kilns. Limestone is fed into the pre-heating zone, where it is dried and heated by gases leaving the burning zone. In the burning zone the limestone is calcined through direct contact with the flame. After calcination the produced quicklime passes into the cooling zone where it is typically cooled by air.9,10 A consequence of conventional quicklime production is CO2 emissions; around 30% of this originates from the fuel burning, and 70% originates from limestone dissociation.9,11 During calcination, every 100 grams of CaCO3 dissociates into 56 grams of CaO and 44 grams of CO2. In 2023, approximately 768 Mt limestone was used to produce 430 Mt of quicklime corresponding to approximately 473 Mt CO2 emitted.9,12 This means that around 331 Mt of CO2 was emitted from quicklime dissociation, and around 142 Mt of CO2 was emitted from the burning of fuel for heating. Since it is difficult to replace limestone with another Ca-rich source when producing quicklime, the focus of CO2-minimizing efforts is on process modifications such as introducing renewable fuels and using oxyfuel combustion. Electrification for heating,13–19 or the emerging technology of calcination with the use of concentrated solar power, 20 –22 in combination with carbon capture, utilization and storage (CCUS) tocould potentially reduce CO2 emissions to zero.
The overview of difference between conventional and electrified quicklime production is presented in Figure 1. In conventional quicklime production, the flue gas containing CO2 is released into the atmosphere and the CO2 concentration in the flue gas varies, depending on kiln type and the type of fuel used. A potential route (marked by dashed arrows) includes a CO2 separation unit, which separates most of CO2 for utilization and storage. The remaining constituents of flue gas are released into the atmosphere. The implementation of an electric calciner allows for efficient capture of CO2 concentrated process gas, for utilization and storage.14,23
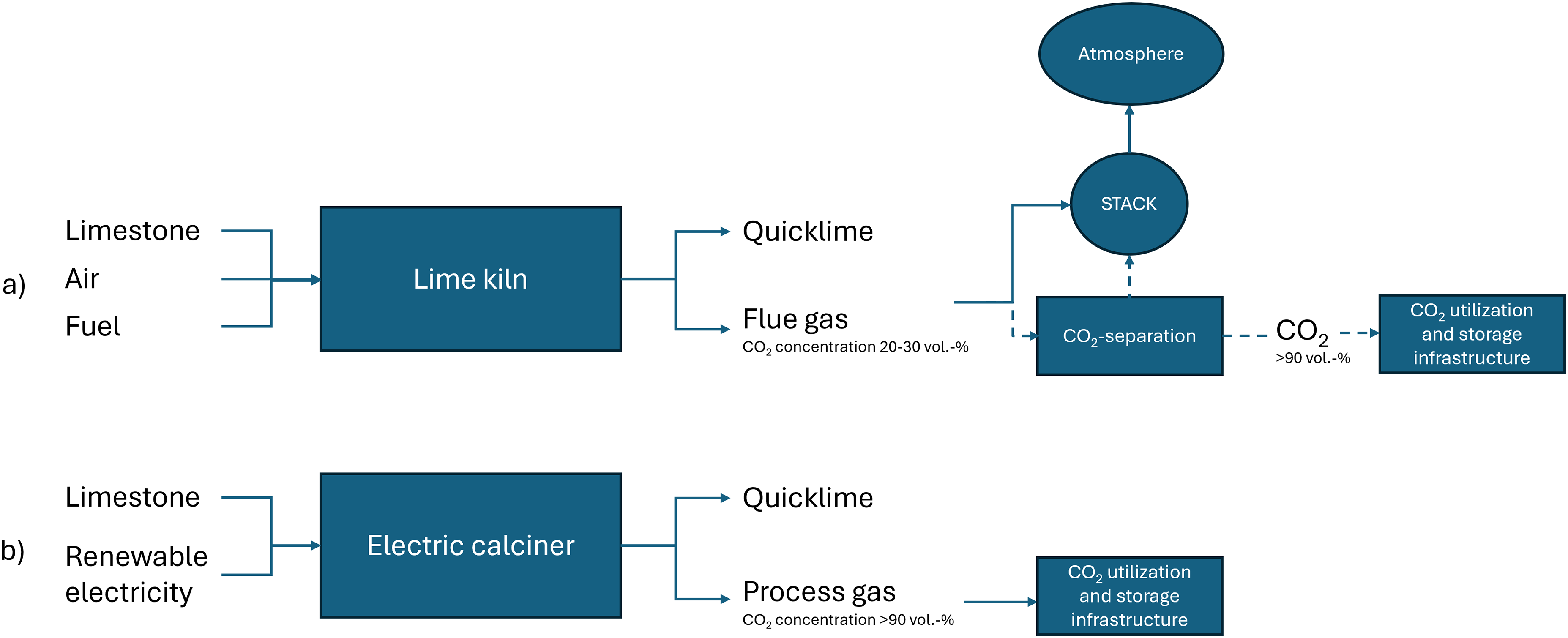
An overview of the a) conventional with potential CO2 separation unit, marked by dashed arrows and b) future electrified quicklime production with CO2 capture and storage infrastructure.
The quality of quicklime is of special importance to quicklime producers and their customers, and is assessed by standardized methods.24,25 The major quality parameters for these assessments are available CaO, residual CO2, chemical composition, and slaking reactivity. The quality requirements vary depending on the application. For example, in Precipitated Calcium Carbonate (PCC) production, quicklime with high available CaO content and low Mg, Si, Al, Mn and Fe content is desirable. Impurities affect the brightness of the product, which is an important parameter as PCC is used as a filler and pigment in paints and paper coatings.
2
High-calcium quicklime is used in the purification of drinking water, where there are strict requirements on e.g., trace elements such as As, Cd, Hg, Ni, Pb, Sb and Se, as high concentrations of these can be detrimental to human health.
26
In steelmaking, quicklime is used to form slag; here, a high-calcium, low-impurity quicklime is very important. The required concentrations are CaO >95 wt.-%, SiO2 < 1 wt.-%, S < 0.03 wt.-% and MgO <1.5 wt.-%.2,27 It is important that quicklime dissolution in the slag occurs rapidly.2,27,28 It has been reported that the increase in solution rate increases with quicklime's slaking reactivity.2,29,30 The slaking reactivity is determined by measuring the temperature increase, according to e.g., standard ASTM C-110
1
or standard SS-EN 459-2: 2010,
24
when mixing quicklime with water according to the exothermic reaction (2).
Depending on the application, quicklime with higher or lower reactivity is preferred. High-reactivity quicklime is generally soft-burned, i.e., produced at a low enough temperature that the CaO crystallites do not sinter, and has applications in e.g., the steel industry. Low-reactivity quicklime is generally hard-burned, i.e., it is produced at high temperatures that result in product densification, and is used in e.g., the production of aerated concrete and sand bricks.
Several previous studies have investigated the influence of various factors on the reactivity of quicklime, and strong connections between the microstructure of the limestone and reactivity of quicklime have been found.31–35 The presence of impurities, either from the limestone36–38 or from the fuel ash13,39 have been found to influence sintering and consequently quicklime's reactivity. Also impurities from the quarry adhered to the limestone, together with high temperature in a kiln, have been found to result in new phases in the quicklime, decreasing its quality. 40
Regarding the conditions and parameters during the calcination, several publications have addressed how the calcination temperature,33–35,41–45 calcination time31,37 both calcination temperature and time32,46 or calcination temperature and atmosphere 47 affect the reactivity of quicklime. Most of the literature reports on reactivity measurements on limestone calcined in undefined atmosphere,31,32,37,41–46 in air,33–35 and only one study on calcination in 100% CO2 has been found. 47 Alaabed et al. 32 studied six different limestones which were calcined at a range of temperatures (800–1100°C) and times (0.25–2 h). The quicklimes had their highest slaking reactivity at different calcination conditions and the slaking reactivity of the resulting quicklimes was found to be mostly attributed to the microstructural features of the limestones. Akande et al. 46 studied the influence of different calcination temperatures (800 and 1000°C) and times (30 and 60 min) on the reactivity of quicklime. They concluded that temperature had the greatest effect on quicklime's reactivity, and that the combination of temperature and time had the second-greatest effect. The quicklime with the highest reactivity was produced through calcination at 1000°C in 30 min. 46
Moropoulou et al. 41 performed a slaking reactivity study on two types of limestone, calcined at a range of temperatures (900–1200°C) for 24 h, and found a correlation between specific surface area (SSA) and reactivity, suggesting that with an increase in SSA quicklime is more reactive. According to the authors, the SSA might be a reliable factor for estimating the reactivity of quicklime. Commandré et al. 47 reported a correlation between SSA and the reactivity of quicklime and compared quicklime's slaking reactivity after calcination of limestone particles (315-400 µm) under 100% CO2 and 100% N2. It was noted that the quicklime produced under the CO2 atmosphere had a lower SSA than that produced under N2, and that the slaking reaction was faster for quicklime produced under the N2 atmosphere, as the increase in CO2 concentration enhanced sintering. However, although correlations between the SSA and slaking reactivity of quicklime have been found, predicting reactivity based on that parameter alone has so far not been proven feasible.44,47
The literature is not conclusive on either the combined effect of time and temperature on the reactivity of quicklime, or on the relation between SSA and reactivity. Furthermore, most of the studies did not focus on the calcination atmosphere affecting the reactivity. Emerging technologies with oxyfuel combustion or fuel-free heating result in calcination taking place in a CO2 atmosphere. Concentrated CO2 enables efficient carbon capture,14,23 but it also alters the reaction environment within a kiln. Therefore, the objective of the present study was to systematically evaluate calcination temperature and time as factors influencing the slaking reactivity of quicklime produced in CO2 atmosphere in order to give first indications if a high reactive quicklime can be produced under these conditions. The study was performed using a well-characterized medium to high purity limestone granules in the size of 5-10 mm. Slaking reactivity was measured according to a standardized method to generate comparable data to that available in the literature, and in addition, the SSAs of the quicklime samples were measured and correlated with the calcination parameters and slaking reactivities of the quicklime samples. The results were discussed from the perspective of product quality in industrial production of quicklime in electric calciners.
Experimental
The material used for the experiments consisted of sedimentary limestone samples from the Jutjärn quarry in Sweden, owned and operated by SMA Mineral AB. This limestone has high to medium purity48–50 based on chemical composition. Jutjärn limestone belongs to the Boda limestone formations (Upper Ordovician in age), and consists of massive wackestone, rich in void-filling stromatactis with small bioclasts, and irregularly distributed grainstone lenses with calcareous algae and trilobites. Jutjärn limestone is fine to medium grained. A detailed geological description of the limestone can be found elsewhere.51,52 Elemental composition based on XRF analysis is presented in oxide forms in Table 1. Loss on ignition (LOI) analysis was performed at 980°C.
Bulk chemical composition and LOI for the jutjärn limestone. Composition measured by XRF and expressed as oxides.
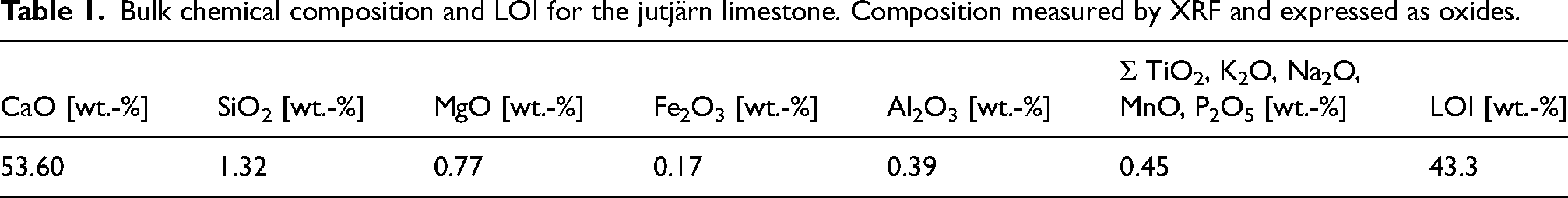
The limestone was sieved and a 5–10 mm fraction was collected and dried in a drying cabinet at 105°C for 4 h. This size fraction was chosen as it is a typical rotary kiln feed material fraction. Representative samples weighing 500 g ± 1 g were placed in crucibles and calcined in a muffle furnace (Carbolite CWF 12/13, Figure 2) in a 100% CO2 atmosphere with 0.5 l/min gas flow and 10°C/min heating rate. The furnace temperature was controlled by a thermocouple by the furnace wall (TC1). Temperature uniformity of the furnace was ±5°C according to specifications. The temperature increase in the limestone bed was monitored (TC2) in pre-tests to ensure complete calcination at the lower temperatures and times, and during these tests the final temperatures were also within the ±5°C, as specified. The CO2 gas flow was added to ensure the furnace volume was free from air during the entire experiment. Since the calcination reactions release CO2, a comparably small gas flow was used, which also ensured efficient heating of the purge gas.
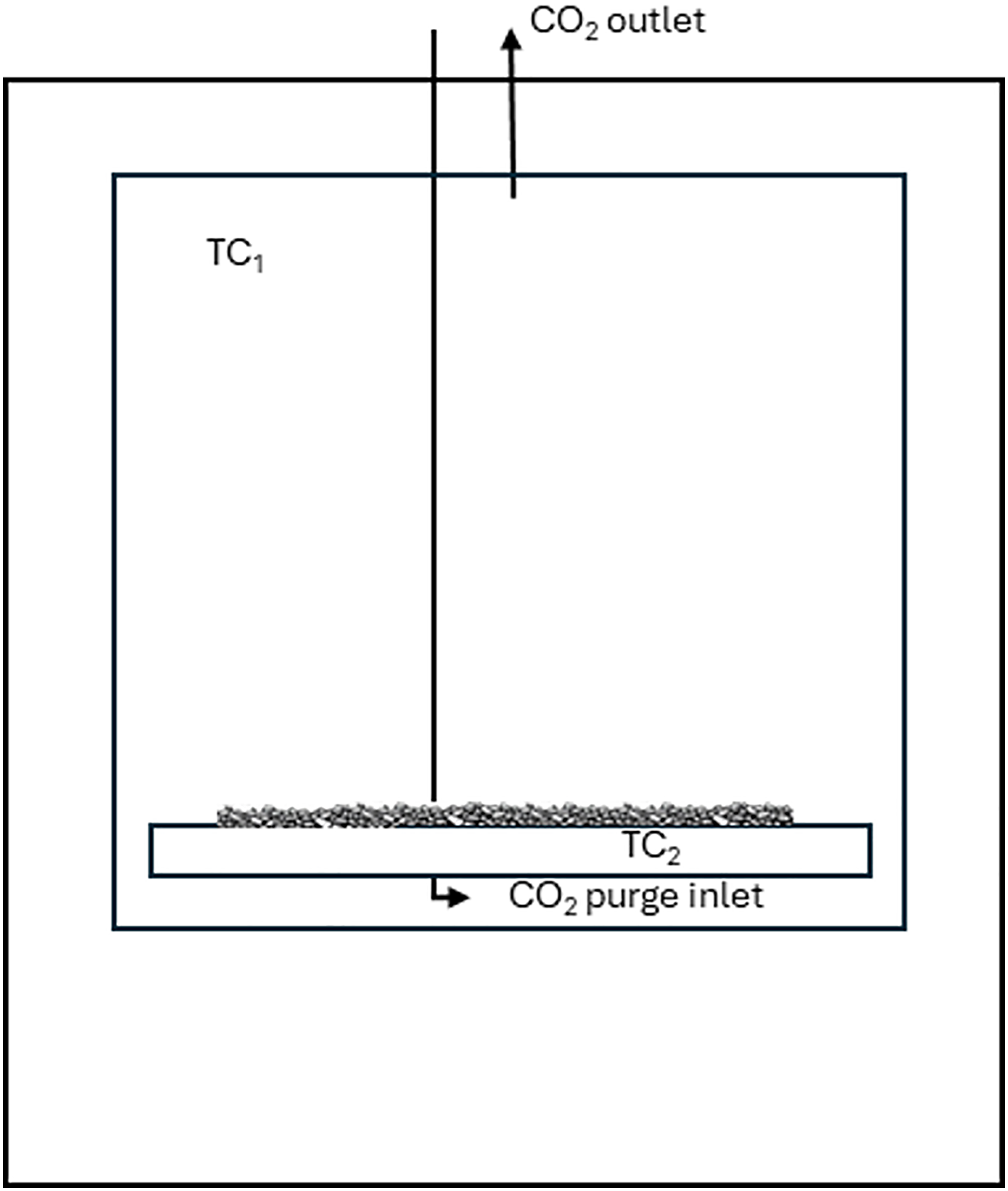
Calcination furnace.
The temperature of thermal decomposition of CaCO3 in 100% CO2 atmosphere has been reported to be 895-898°C.1,53 Pre-tests with weighing before and after were conducted to ensure full calcination at the low temperatures and times. On this basis, temperatures ranging from 1000°C to 1200°C were selected, and times were varied between 10 and 60 min.
The 100% CO2 gas atmosphere can influence the initial part of the calcination when the reactions occur close to the surface. Here, the CO2 pushes the equilibrium towards higher temperatures compared to if it would take place in another gas (e.g., air or N2). However, as soon as the calcination progresses it will move into the particle and CO2 will form causing local overpressure, and CO2 will continuously diffuse outwards creating a local environment totally dominated by CO2 regardless of gas composition on the outside of the particles. Therefore, it is likely that the decomposition temperatures of large calcite particles are governed by the thermodynamic equilibrium in 100% CO2 regardless of purge gas composition. The gas atmosphere probably has a greater influence after the actual calcination when no more CO2 is released from the stone; CO2 is known to promote sintering of quicklime, and the CO2 purge ensured 100% CO2 in the furnace also during this period. After the specified calcination times, the CO2 purge was replaced with 0.5 l/min of N2 during the cooling (natural cooling, overnight) to avoid recarbonization of the material.
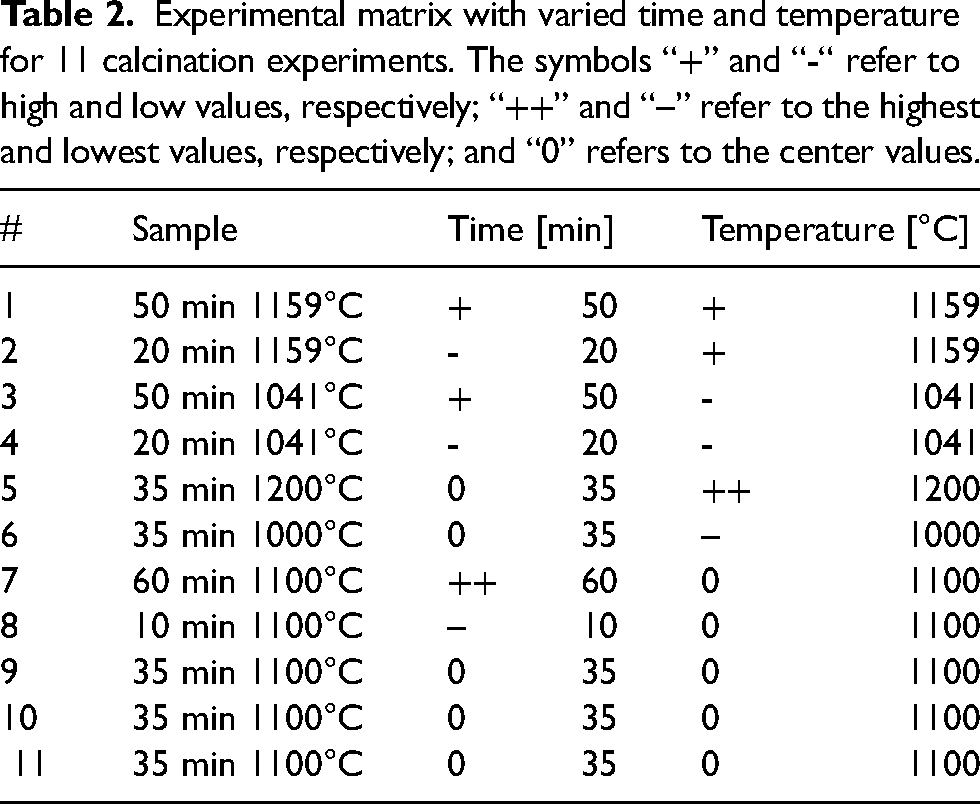
Calcination experiments were performed according to a factorial design, presented in Table 2; the experiment run order was randomized. The center values were marked by “0”. The high values were marked with “+”, and low values with (-), while the highest values with “++” and the lowest values with “–“. The three repeated center points (Experiments 9, 10 and 11) had the same temperatures and times and were values halfway between the highest and lowest ones and were included to assess reproducibility. Experiments 5 and 6 had the same times (halfway values) but different temperatures (highest and lowest), and Experiments 7 and 8 had the same temperatures (halfway values) but different times (highest and lowest). 54
Experimental matrix with varied time and temperature for 11 calcination experiments. The symbols “+” and “-“ refer to high and low values, respectively; “++” and “--” refer to the highest and lowest values, respectively; and “0” refers to the center values.
The quicklime produced was used for the slaking reactivity tests. After cooling and before the reactivity measurements, the samples were stored in a container flushed with nitrogen to avoid air-slaking. It has been found by Potgieter et al. 55 that quicklime storage conditions are very important and can influence the slaking rate; there is a possibility of air-slaking and carbonation, and exposing quicklime to ambient atmospheric conditions can lead to a decrease in quicklime's reactivity.
The data analysis was done by multiple linear regression (MLR) in MODDE 12 software (Sartorius, Germany) with time and temperature of calcination as input parameters, and reactivity parameters or the nitrogen-adsorption method of Brunauer-Emmett-Teller (BET) area as output.
The slaking reactivity tests were, with the exception of the sample fraction, performed as specified in the standard SIS-EN 459-2: 2010 24 using 150 g of quicklime and 600 g of 20°C water. The materials were mixed in an isothermal Dewar vessel with automatic stirring at 300 rpm; the temperature was measured at 0.5-s intervals for 10 min. The maximum temperature (Tmax), time to reach 60°C (t60), time to reach 80% of full slaking (tu) and temperature increase during the initial 30 s (ΔT30) were used as parameters for classifying slaking reactivity, consistent with the aforementioned standard. 24 These parameters are commonly used by industry in evaluating slaking reactivity. Temperatures were measured and plotted as a function of time to obtain a wet slaking curve, from which the parameters were extracted. The starting temperatures varied (18.57-21.09 °C) and the temperature data was shifted up or down to normalize the starting temperature to 20°C.
Tu (temperature at 80% of full slaking) is calculated using Equation (3), with the maximum temperature of the reaction (Tmax) and initial temperature for each experiment (T0); tu (time to reach 80% of full slaking) is later read from the slaking curve:
It has been found by Potgieter et al. 38 that the presence of chlorine ions in water can increase the slaking rate, and that the presence of sulphate and carbonate ions can decrease the rate. In another study, a series of slaking reactivity experiments was performed, and no difference was observed between local tap water and distilled water. 56 The tap water in Umeå is classified as mild, 26 with 19 mg/l Ca and 1.67 mg/l chlorine. On that basis, the decision was made to use tap water for the experiments, which makes the results relevant from an industrial, cost-effective point of view.
The material characteristics of the quicklime in terms of average pore diameter, pore volume and SSA were evaluated using the BET method and a Micromeritics TriStar 3000 gas adsorption analyzer. The quicklime samples were crushed into 3–4 mm particles in order to fit into the BET sample inlet tube. The weight of the samples varied between 0.06 and 0.15 g. Before analysis, the quicklime samples were kept under vacuum for 48 h before degassing at 150°C for 9 h in a continuous nitrogen flow using a Micromeritics Smart Prep degassing unit.
Results and discussion
Slaking reactivity
The outcome of the slaking reactivity measurements was a series of wet slaking curves, presented in Figure 3. The curves show the temperature increase as a function of time from the initiation of the slaking tests.
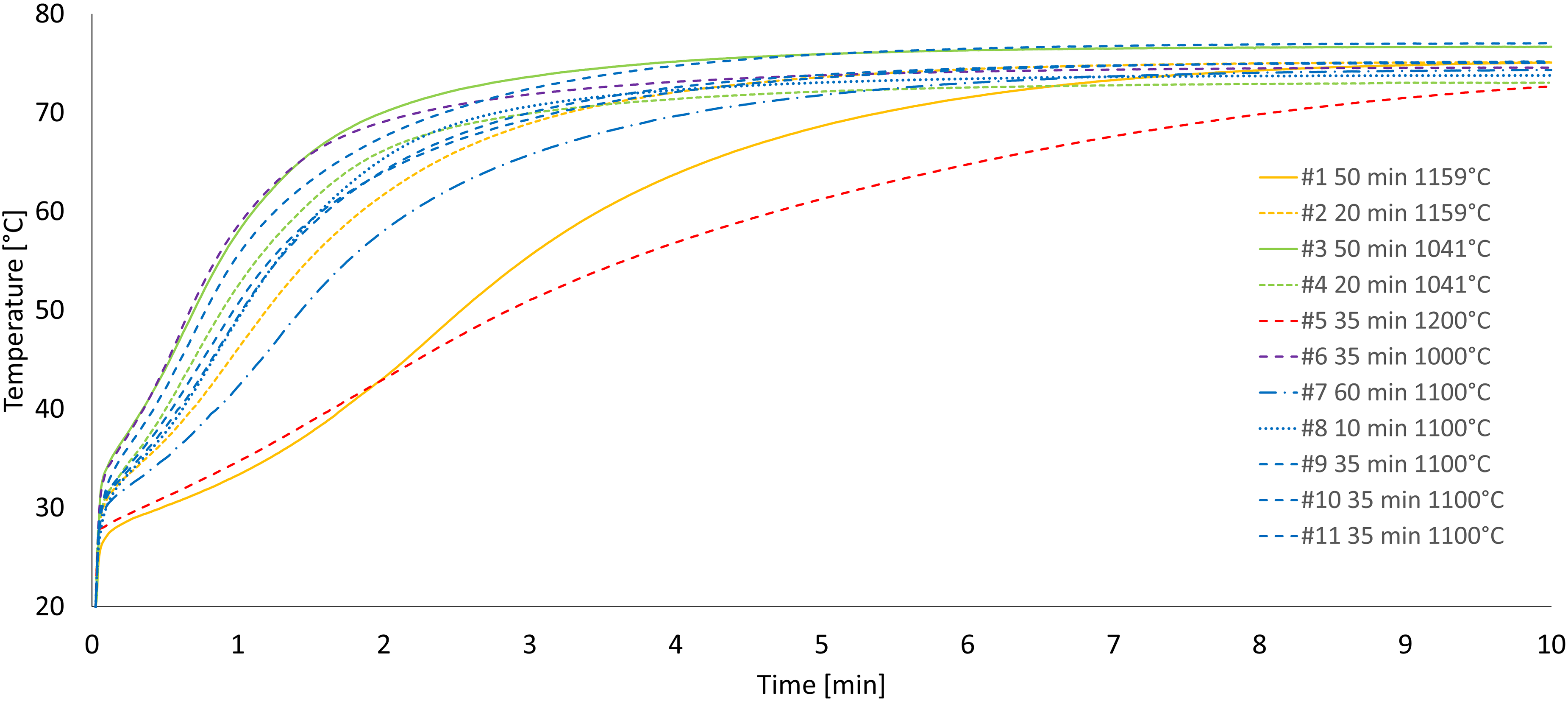
The slaking reactivity curves of the 11 experiments.
Since detailed and quantitative comparisons between the curves is difficult based only on Figure 3, further evaluation of slaking reactivity was based on the four parameters described in the experimental section: Tmax, ΔT30, tu and t60. These reactivity parameters are summarized in Table 3. Interpretation of these parameters varies in the literature. For example, t60 [min] is used to classify the reactivity of quicklime as: high < 3 min, medium 3–6 min, low > 6 min. 34 It can also reflect the degree of burning (hard-burning generally decreases slaking reactivity): soft < 3 min, medium 3–9 min, hard > 9 min.1,2 The mean value of calcination weight loss based on the weight before and after calcination, which is a similar measurement to LOI, was 42.2 ± 0.6 wt.-%.
The parameters for the slaking reactivity of quicklime are calcined under different conditions.
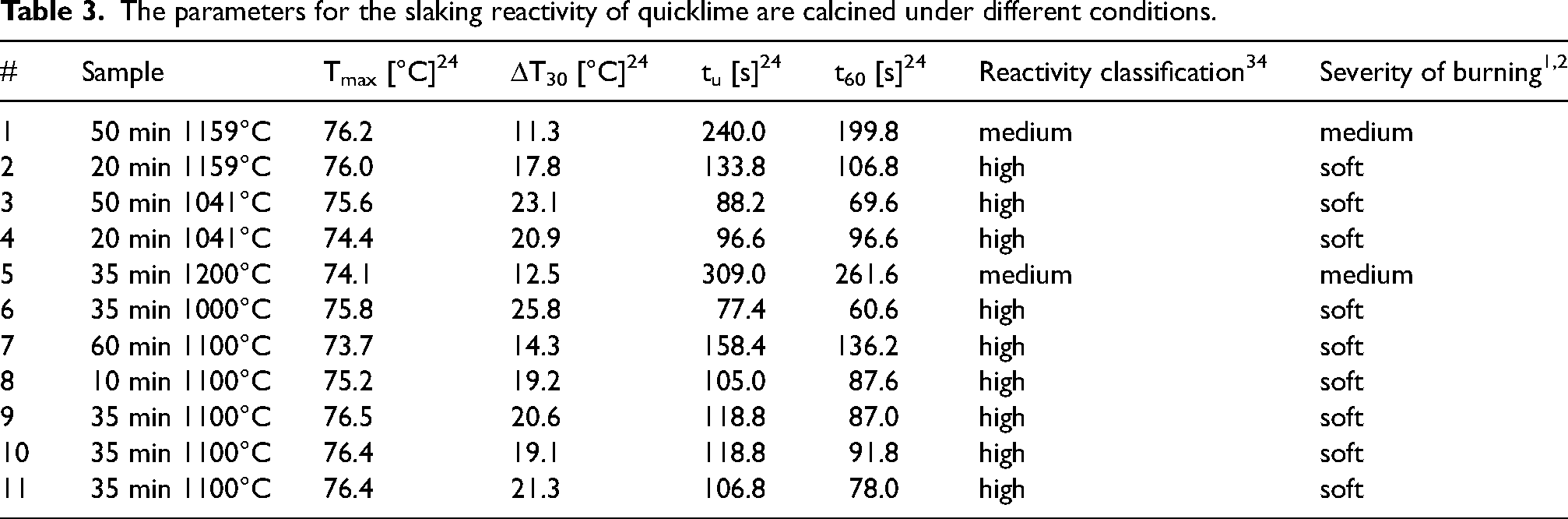
Based on the results presented in Table 3 all samples, apart from samples 1 and 5, were soft-burned and had a high reactivity. The results show that it is possible to calcine limestone under CO2, as expected in e.g., electrified processes, and that a product with controlled medium to high reactivity can be obtained.
The experiments were performed in well-controlled laboratory-scale conditions. The reactivity of industrially produced quicklime may be lower due to e.g., locally higher temperatures in the kiln; in the case for Long Rotary Kiln (LRK) the material temperature of the product can reach up to 1500°C 8 and in a shaft kiln, it can reach more than 1600°C.9,10,57 The consequence of a high product temperature is calcium oxide sintering and reduced quicklime reactivity. 47 High reactivity products are produced at lower temperatures, e.g., 1000-1200°C. 18
The slaking reactivity results were further analyzed using MLR, compiled below in a summary-of-fit plot and a response-contour plot. The summary-of-fit plot is presented in Figure 4 with the output parameters. N denotes the number of experiments. The R2 value describes how good the fit of the data is and should be close to 1 for a good model fit. The Q2 value indicates the predictive ability of the model and should be higher than 0.5. Model validity indicates whether the right type of model was chosen, and the value should be above 0.25. Reproducibility is a performance indicator that numerically summarizes the error value in a model, and should be above 0.5. 54
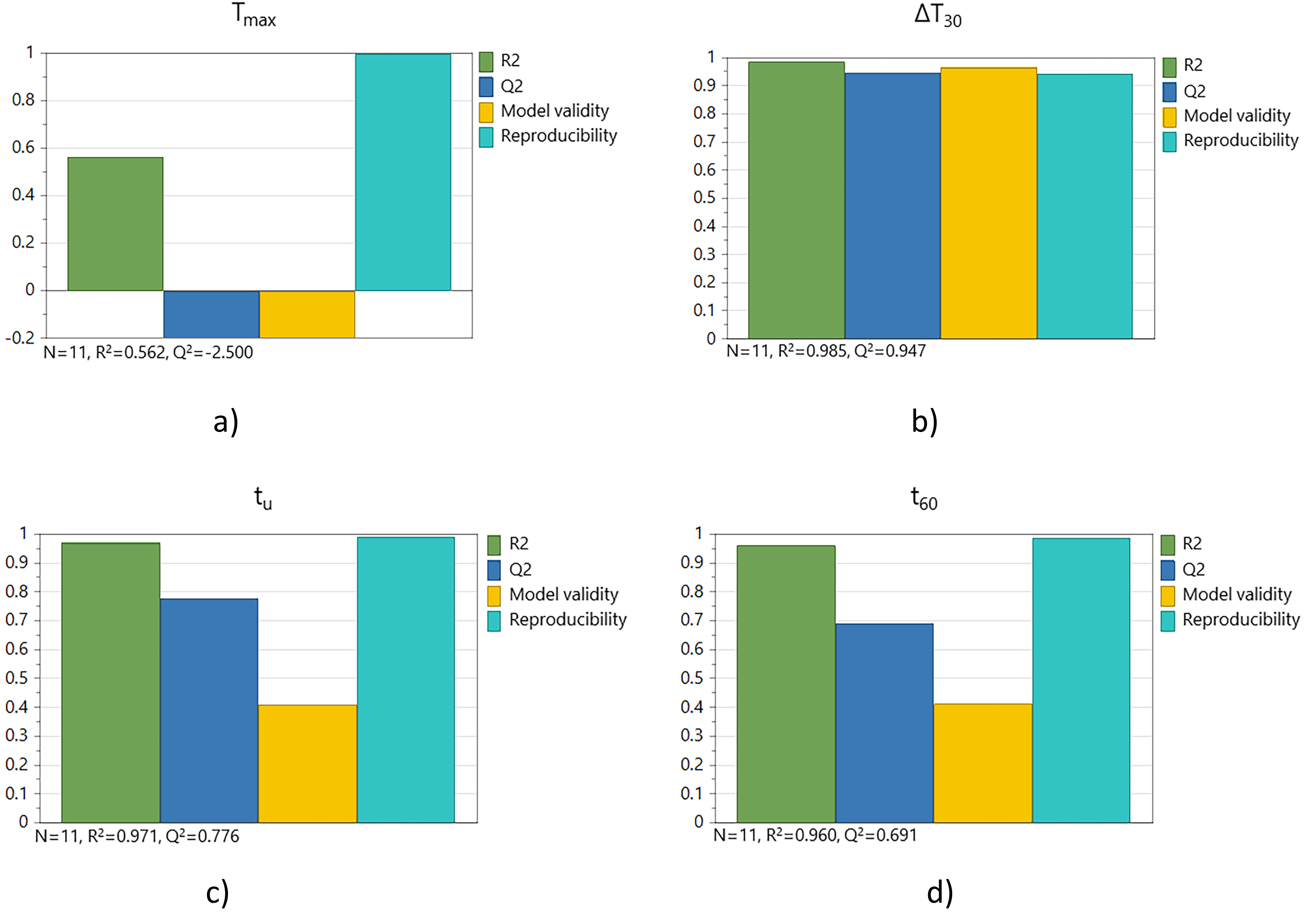
The summary-of-fit for the slaking reactivities: a) Tmax, b) ΔT30, c) tu and d) t60.
The results presented in Figure 4 a) show that Tmax appears to be an inadequate parameter for correlating slaking reactivity with the calcination parameters because the Q2 value is negative, meaning that within the ranges investigated the Tmax value was not well explained by the experimental variables. Tmax corresponds to the total heat produced by the exothermal slaking reaction, and thus depends strongly on the concentration of calcium oxide in the quicklime sample which did not vary much in the present study since all samples originate from the same limestone material. The corresponding model for ΔT30 shown in Figure 4 b) had a better fit based on the values of R2, Q2, reproducibility and model validity, which were all close to 1. The models for tu and t60 presented in Figure 4 c) and Figure 4 d), respectively, fulfilled all the numerical requirements of the criteria, indicating the data fit is adequate and useful. Based on Table 3 it had already been concluded that the samples in this study had medium to high reactivity, and that is probably why the ΔT30 parameter has the highest validity.
The coefficients of the different prediction models are presented in Table 4, and any point in the experimental domain is described by an equation: f(T[°C], t[min.]) = a + bT + ct + dt2 + eT2 + fTt.The corresponding variable contour plots for each of the three useful parameters, corresponding to Figure 4 b) – d) and to coefficient models are presented in Figure 5.
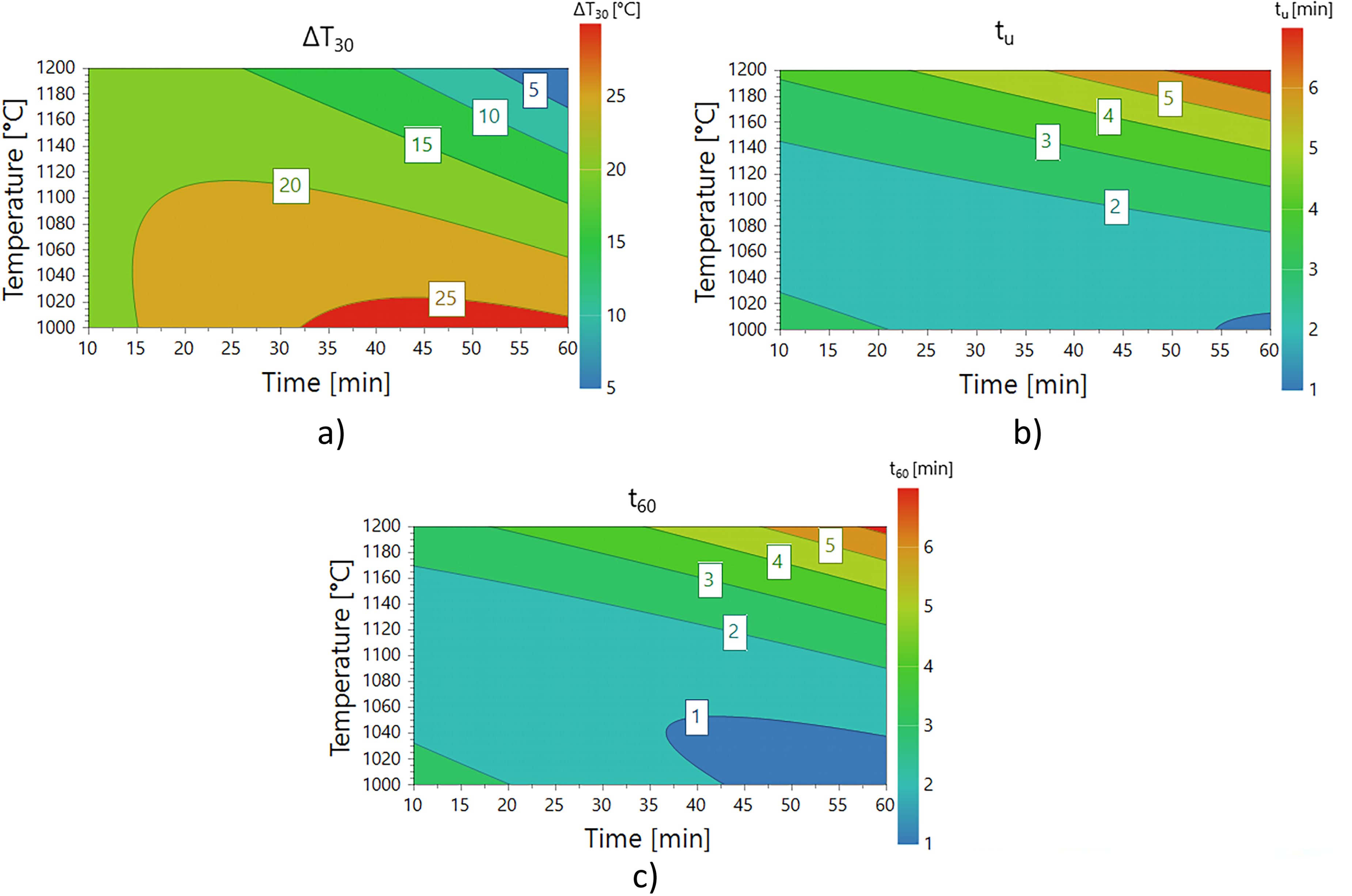
The contour plots for the fitted model of calcination parameters with a) ΔT30, b) tu and c) t60.
The coefficient models for prediction of ΔT30, tu and t60.

In the design space, all three statistically significant reactivity parameters showed the same trend: to obtain a high-reactivity quicklime, the temperature should be low and calcination time long. The ΔT30 plot Figure 5 a) indicates high reactivity (red) for low temperature and medium to long calcination periods. For parameters tu and t60, high reactivity can be found in areas indicated with low values (blue) in Figure 5 b) and Figure 5 c). It can also be concluded that, within this design space, temperature was more influential than time on reactivity, and that, under the prevailing conditions, keeping the temperature below 1100°C would be the first measure for achieving a high-reactivity product. A similar finding has been reported, 46 where the calcination temperature had the greatest effect and a high-reactivity quicklime was produced by calcination at 1000°C for 30 min, under undefined atmosphere. From a Design of Experiments perspective, calcination time and temperature are controlled factors that influence reactivity. The results of the BET analysis: SSA, average pore diameter and pore volume are uncontrolled factors, that also influence reactivity.
BET specific surface area analysis
The SSA of a given quicklime is dependent on the calcination conditions: time, temperature and atmosphere. In this study, calcination was performed under a CO2 atmosphere to simulate an electrified calciner; CO2 is known to decrease SSA by accelerating CaO sintering.58–61
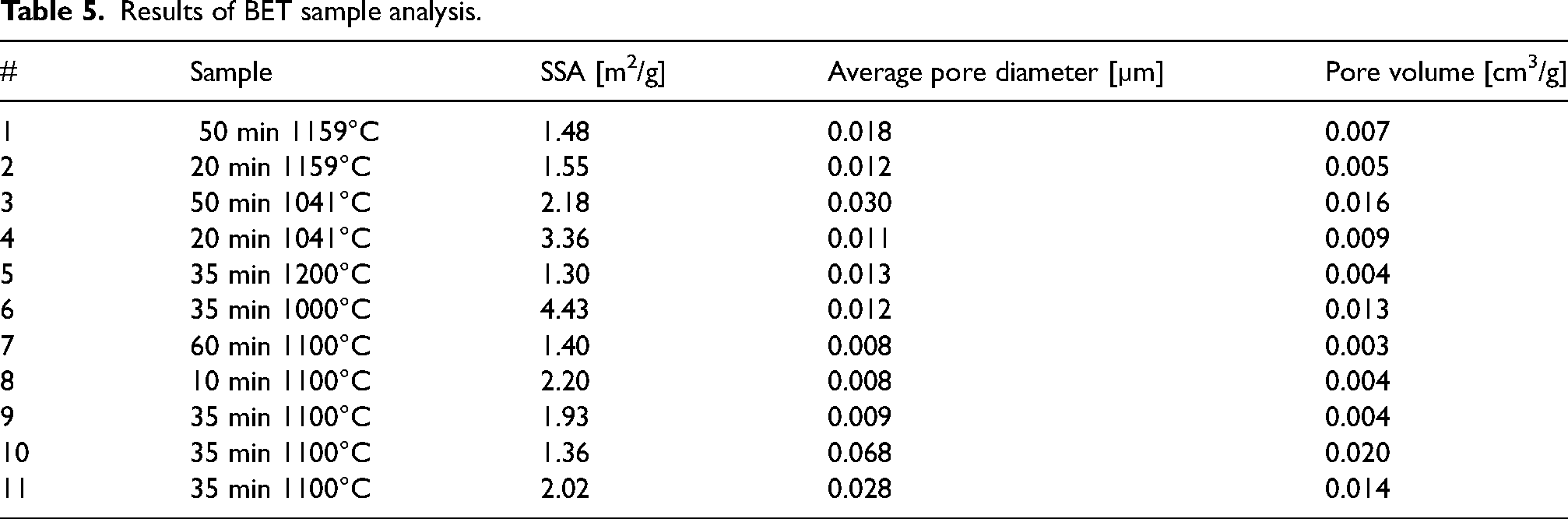
The results of the BET analysis are presented in Table 5. The highest applied experimental temperature of 1200°C resulted in the lowest SSA, of 1.30 m2/g. Conversely, the lowest applied temperature of 1000°C resulted in the highest SSA, of 4.43 m2/g. This confirms that an increase in temperature reduces the SSA of the product, possibly by sintering of CaO.35,58,60,62 This finding is consistent with the previous finding of high SSA values for lower calcination temperatures, performed under undefined atmosphere, 41 as well as performed under N2 or CO2. 47 SSA is a parameter associated with slaking reactivity, as high SSA provides a larger area for reaction. The results show a significant correlation between SSA and ΔT30 of 0.74, and moderate correlations between tu and SSA of −0.56, and t60 and SSA of −0.51.
Results of BET sample analysis.
The correlation between SSA, average pore diameter and pore volume measured using BET and the calcination variables were further analyzed using MLR. The summary-of-fit between average pore diameter and pore volume and the calcination variables resulted in a poor fit with negative Q2 and reproducibility. This means that the lack of correlation is statistically validated, and therefore, within the design space, there is no correlation between the calcination conditions and the obtained average pore diameter and pore volume.
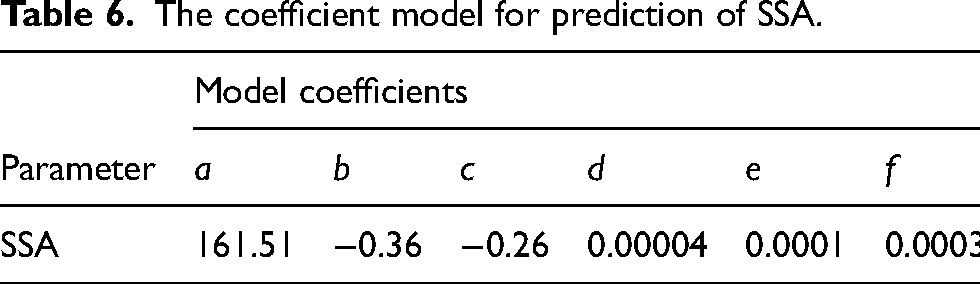
The correlations between SSA and the calcination variables are summarized in summary-of-fit, contour plots in Figure 6, and the coefficient model in Table 6. The model had high validity and predictability according to the fit parameters. As can be seen in the contour plot in Figure 6, the highest SSA values are found in the lower-left corner of the plot. The results show that, similarly to the reactivity parameters (ΔT30, tu, t60), high values were found for low calcination temperatures. However, the influence of calcination time is reversed for BET analysis as compared to the reactivity parameters; short times give high BET values whereas longer times give higher reactivities (see Figure 5). Sintering is a plausible explanation for SSA decreasing with time, 59 but the inverse correlation with reactivity was somewhat unexpected. This indicates that reactivity, although governed by surface reactions, is related to a property that is not accurately measured by the BET analysis. This is in agreement with findings by e.g., Leontakianakos et al., 44 who found that SSA alone is not a reliable factor for estimating reactivity performed under undefined atmosphere, and that the kinetic and mechanistic aspects of the slaking reaction are more important. Another study has also concluded that it is not possible to predict the reactivity of quicklime based on SSA alone, after performing the calcination in 100% CO2 or 100% N2. 47 During the BET analysis nitrogen gas penetrated the sample pores more efficiently than, and in contrast to, the water in the slaking reactions, which reacted only with the outer surface, 1 and this could be part of the explanation for the inversed correlation observed. To find information on what fundamental properties of the quicklime that is controlling the slaking reactivity further studies are needed.
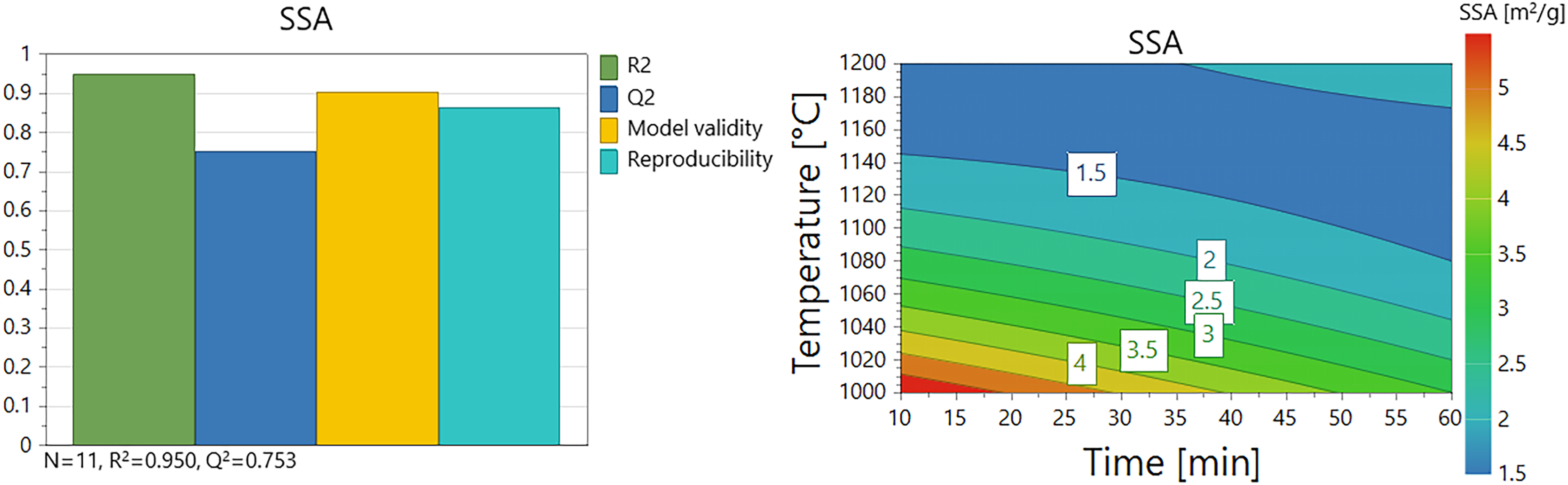
Summary-of-fit and contour plots of the fitted model of BET analysis of SSA vs. calcination parameters.
The coefficient model for prediction of SSA.
Conclusions
The slaking reactivity of quicklime is an important product quality parameter and influencing factors are of special interest for industrial applications. The reactivity of the quicklime product is known to vary depending on limestone properties, the calcination conditions and on the resulting quicklime's microstructure. In the present study, the effects of calcination time and temperature on slaking reactivity were systematically studied and evaluated. In addition, the SSA of quicklime samples was measured and correlated with the calcination parameters and resulting slaking reactivity. All calcination experiments were performed under CO2 atmosphere, which is of relevance with regards to future electrically heated zero-emission processes. Based on the analysis of the results, the following can be concluded:
Available literature has been inconclusive on the effect of calcination parameters on the slaking reactivity. The laboratory scale experiments reported in the present paper indicate that calcination temperature has greater effect on the slaking reactivity of quicklime than the time, within the range tested. For the kiln-feed limestone used in the present study, quicklime with the highest initial reactivity, ΔT30, and quicklime with the highest reactivity, t60, was produced at a low temperature and with a medium to long calcination time. The highest-reactivity quicklime at 80% of slaking, tu, was produced at a low temperature and with a long calcination time, based on statistical treatment of the experimental results and illustrated in the contour plots. Reliable predictive models for the reactivity parameters ΔT30, t60 and tu, were obtained. The reactivity parameter Tmax was found not to be a useful parameter and no reliable model was obtained. A reliable predictive model for SSA of the quicklime product was obtained. However, the inverse correlation between SSA and reactivity was unexpected and SSA analysis performed by BET cannot be used in a mechanistic explanation for the slaking reactivity. Additionally, the results show that there is no correlation between the calcination conditions and the average pore diameter and pore volume of the quicklime product.
This paper demonstrates the possibility of producing controlled, medium to high reactivity quicklime, in a CO2 atmosphere calciner, e.g., one that is electrically heated. Thus, from a reactivity point of view, there is potential for an electric calciner to, combined with carbon capture, utilization and storage (CCUS), provide a solution for zero-emission production of quicklime of controlled medium to high reactivity.
For detailed information on what fundamental properties of the quicklime that is controlling the slaking reactivity further studies are needed. Also, the results and conclusions from the present study are limited to one assortment of limestone only, meaning factors suc h as mineral composition, particle size, impurities, etc. need to be taken into consideration in future studies.
Footnotes
Acknowledgements
Nordkalk AB, SMA Mineral AB, Heidelberg Materials Cement Sverige AB, the Ellen, Walter and Lennart Hesselman Foundation, the Swedish Mineral Processing Research Association - MinFo, the Swedish Energy Agency, and Sweden's innovation agency Vinnova are acknowledged for their project support. This project has also received funding from the European Union's Horizon Europe research and innovation programme under Grant Agreement No 101138392. The authors would like to thank Piotr Jablonski from the Department of Chemistry at Umeå University for assistance, and Karin Sandström, Umeå University, for fruitful discussions.
Credit authorship contribution statement
Katarzyna Olovsson: Conceptualization, Investigation, Data curation, Formal analysis, Writing – original draft, Writing – review & editing, Matias Eriksson: Conceptualization, Investigation, Writing – review & editing, Supervision, Project administration, Funding acquisition, Erik Björnwall: Conceptualization, Investigation, Markus Broström: Conceptualization, Investigation, Writing – review & editing, Supervision, Project administration, Funding acquisition.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the European Union’s Horizon Europe research and innovation programme, (grant number 101138392).