Abstract
This manuscript analyzes the valorization of agricultural wastes as heavy metal adsorbents and their eco-friendly final disposal as mineral admixtures to manufacture cement-based construction materials. The adsorption properties of nutshell, pistachio shell, and agave fiber for the removal of Pb2+ and Ni2+ from aqueous solution were analyzed under different operating scenarios (i.e., pH, temperature, waste dosage, CaCO3 concentration). Taguchi L9 experimental design was applied to identify the best removal conditions for both heavy metal ions. The maximum experimental adsorption capacities of Ni2+ were 14.8, 17.8, and 16.4 mg/g for pistachio shell, nutshell, and agave fiber, respectively, whereas the maximum Pb2+ adsorption capacities of these residues were 6.25, 16.25, and 103.5 mg/g. Adsorption studies showed that CaCO3 concentration of aqueous solutions affected the removal of these heavy metal ions. However, these agricultural residues showed a competitive removal performance under different water hardness conditions. On the other hand, the setting and compressive strength of cement paste samples containing these residues loaded with heavy metals (i.e., as mineral admixtures) were also investigated. The results indicated that the setting process of the cement paste samples was accelerated when the agave fiber was used in its natural form (without heavy metals), as well as when the pistachio shell and nutshell were saturated with heavy metals. These findings were associated with the agricultural residue structure, which allowed better adherence to the cement particles by hydrating the elongated form of the agave fiber and the globular form of the nutshell and pistachio shell. Finally, the compressive strength of cement-based specimens was evaluated at 7, 14, and 28 days after their preparation. The results showed that the cement paste samples incorporating nutshell had a higher compressive strength than those of the pistachio shell and the agave fiber because of the high lignin content in the nutshell. This circular economy approach for the valorization and final disposal of agricultural wastes is attractive to face their waste management problem, and the secondary pollution problem generated by the spent adsorbents used in water treatment.
Introduction
Various activities of society generate solid, liquid, or gaseous wastes that can have negative effects on the environment and human health. In this direction, any material obtained from extraction, transformation, production, consumption, use, or treatment processes, whose quality does not allow its reuse in the same operation that generates it or its recycling in other process, can be considered a residue. The valorization and reuse of residues and subproducts obtained from different processes contribute to solving the problem of waste disposal and management including the minimization of their associated environmental impacts (Silva Santos Moreira et al., 2018). Previous studies have shown that agricultural biomass residues (which are composed mainly of lignin) are promising for various applications based on the circular economy principles. One alternative includes their utilization to the replacement of cement to obtain construction materials with certain physical, chemical, and mechanical properties (Bajwa et al., 2019).
On the other hand, it is important to indicate that the pollution caused by several heavy metals represents one of the main environmental issues to be solved today due to their toxicological effects (Mitra et al., 2022). Various technologies have been used for the removal of heavy metals from water, including ion exchange, coagulation, chemical precipitation, membrane filtration, electrochemical processes, ultrafiltration, and adsorption (Bolisetty et al., 2019). However, the adsorption is the most used and effective technique for the treatment of wastewater polluted by organic and inorganic toxic compounds including metallic cations (Moreno Virgen et al., 2018). The adsorption-based wastewater treatment is a low-cost purification technology where the adsorbent should be applied under suitable operating conditions to maximize the separation performance (Chakraborty et al., 2022; Kumar et al., 2019, Elwakeel et al., 2020). The adsorbents that have been applied in wastewater treatment for heavy metal depollution include polymers, nanoparticles, carbon-based materials, zeolites, and clays (Nilamsari et al., 2022; Elgarahy et al., 2023; Hsing-Jung and Atsushi 2023; Singh et al., 2024). These materials are generally subjected to specific activation processes to improve their adsorption properties in the aqueous solution, thereby increasing the water treatment cost and its carbon footprint. Several studies have focused on the use of lignocellulosic materials for the removal of heavy metals from water (Mendoza Castillo et al., 2015; Krishnani, 2016; Jiang et al., 2020; Kaya et al., 2020; Krishnani, 2016; Thakur et al., 2020, Elwakeel et al., 2023). This approach has been introduced due to the competitive adsorption properties and low cost of lignocellulosic materials compared to commercial adsorbents (e.g., activated carbon), whose large-scale application and regeneration can be expensive for vulnerable sectors (Malik et al., 2017). Literature review indicates that the most commonly used agricultural residues for the removal of heavy metals, in their natural or carbonized forms, include wheat straw, bamboo, avocado seed, mango and plum, banana peel, coconut shell, orange, peanut and rice, guava seeds, corn cob, cane bagasse, corn residues, sawdust, peach stone, agave fiber (AF), nutshell (NS), and pistachio shell (PS; Nayak et al., 2017; Kharrazi et al., 2021; Sharma et al., 2022).
Herein, it is also important to highlight that the implementation of adsorption for the removal of heavy metals from water generates a residue that corresponds to the saturated (exhausted) adsorbent used as separation medium. This saturated adsorbent usually can be regenerated for its recycling in water depollution. At the end of the adsorbent lifetime where its adsorption properties cannot be regenerated, the residual adsorbent should be properly disposed, or reused in another process as a raw material, with the aim of immobilizing the loaded pollutants and minimizing the environmental impact caused by its final disposal. In this sense, cement is one of the most adaptable binder materials for the immobilization of heavy metals and has been widely used for the management and reduction of leaching rates of metal-saturated wastes (Vyšvařil and Bayer, 2016; Niu et al., 2018; Piekkari et al., 2020).
Portland cement is effective to carry out the solidification process of wastes and can be utilized for the eco-friendly final disposal of materials containing toxic heavy metals, mainly because the high pH of the cement keeps metals in their less soluble form (Pantazopoulou et al., 2020; Sim et al., 2023). Therefore, previous studies have analyzed the preparation of cement products via the incorporation of different hazardous residues. For example, lignocellulosic wastes have been used to replace cement and to minimize the consumption of this construction material, whose production process is highly polluting (Teixeira et al., 2020). It is convenient to mention that lignocellulosic residues, in addition to their excellent properties for the removal of pollutants, have gained remarkable importance in recent years because of their high specific resistance, coupled with their high availability in nature and low cost (Barra et al., 2015; Santos et al., 2015). However, the use of mineral admixtures in cement pastes may change the final product properties, including the tensile and compressive strength (Inozemtcev et al., 2018), setting times (Khan et al., 2014; Burris and Kurtis, 2018; Altynbekova et al., 2023), and hydration (Kaminskas et al., 2015; Deboucha et al., 2017). The partial replacement of binders and mineral admixtures in Portland cement using nontraditional construction materials (e.g., granulated slag, fly ash, cane bagasse, LCD glass residues, and tire rubber particles) modifies the physical and chemical properties of the cement microstructure (Ramezanianpour, 2014; Soltan et al., 2017; Miller and Myers, 2020). Another important factor is the amount of water added during the mixing process because it has a significant impact on its workability. Most of the water is consumed in chemical reactions with the cement (i.e., the process of cement hydration), while the rest remains in the liquid state inside the microstructure, which is directly responsible for the porosity of the hardened cement paste, thus affecting its mechanical properties, transport, and durability (Scrivener et al., 2019). The temperature is also an important factor during the development of the strength of cement samples because it has shown that a temperature increase improves the hydration rate and thus, a greater compressive strength can be obtained at early ages under these conditions (Xu et al., 2018; Huang et al., 2022). Consequently, the recycling of heavy metal-loaded lignocellulosic wastes to obtain cement products implies a detailed analysis of the best preparation conditions and the corresponding property characterization of the final product to establish the potential benefits and drawbacks.
In this study, the valorization of heavy metal-loaded agricultural residues for the cement paste preparation was analyzed and discussed. First, the adsorption properties of PS, NS, and AF for the removal of lead (Pb2+) and nickel (Ni2+) from water were studied under different conditions of temperature, pH, biomass dosage, and CaCO3 concentration. For the case of Mexico, PS, NS, and AF are agricultural waste generated in large quantities and their valorization is fundamental to generate environmental and economic benefits. These residues also display favorable mechanical strength and/or morphology that can be exploited to obtain alternative materials. In the present study, these biomass wastes saturated with these heavy metals (after water purification) were used as mineral admixtures in the preparation of cement paste samples where their mechanical properties in the fresh and hardened states were evaluated and compared. The novelty of this study lies in the valorization of residual biomass to remove heavy metals from water and the safe final disposal of these residues containing toxic heavy metals for the preparation of cement-based materials. The results of this study allow to assess the potential benefit of recycling these agricultural residues to reduce the environmental impact and production costs of cement pastes, besides introducing a sustainable alternative for the final disposal of biomass wastes used in the removal of toxic pollutants from water.
Methodology
Materials
Residues of AF, PS, and NS were selected and collected to perform this study. They were used as natural adsorbents to remove heavy metal ions from water and recycled as mineral admixtures for cement preparation. These agricultural wastes were ground using an IKA MF 10 blade mill. The particle size of these materials was homogenized using ASTM (American Society for Testing and Materials) sieves, which ranged from 0.85 to 0.25 mm for studying the effect of this parameter on the physical properties of cement pastes in fresh state (evolution of setting). For the adsorption studies, PS and NS were utilized with particle sizes of 0.50–0.84 mm, whereas a particle size of 0.25–0.42 mm was selected for AF. These lignocellulosic materials were washed with deionized water to remove impurities and then dried at 70°C for 24 h. Therefore, the adsorption properties of tested biomass residues were calculated on a dry basis. Metal solutions were prepared from Ni(NO3)2·6H2O (Golden Bell) and Pb(NO3)2 (J.T. Baker).
The cement pastes were obtained with composite Portland cement classified as CPC 30 R according to the Mexican standard (NMX-C-414-ONNCCE-2004), with a 7-day compressive strength of 30 MPa. Deionized water was used to prepare all the cement pastes.
Characterization of heavy metal adsorption properties of agricultural residues
The adsorption properties of lignocellulosic residues were analyzed at batch operating configuration. Heavy metal adsorption studies were carried out using the experimental conditions defined in a Taguchi L9 array for each biomass sample, see Table 1. This experimental design was applied to determine the impact of heavy metal removal conditions on biomass adsorption properties. A brief explanation of Taguchi experimental design is provided in the Supplementary Information file. Adsorption studies were performed varying pH, temperature, and CaCO3 concentration of heavy metal solutions. Specifically, this experimental design implied the assessment of solution pH (3, 4, and 5) and temperature (15, 25, and 35°C), concentration of CaCO3 added to the heavy metal solution (5 × 10−5, 1 × 10−4, and 1.3 × 10−4 M), and biomass dosage (10, 25, and 40 mg/mL), while the response variable was Pb2+ and Ni2+ adsorption capacities of tested agricultural wastes. Note that CaCO3 was utilized to analyze the heavy metal removal performance of these residues under different water hardness conditions, which is a typical characteristic of real-life fluids like groundwater. The impact of these operating variables on the heavy metal adsorption properties of tested biomass wastes was evaluated via a statistical analysis with the signal-to-noise (S/N) approach under the premise “higher-the-better” (Durán Jiménez et al., 2014). This statistical analysis also included the identification of the best operating conditions that favored Pb2+ and Ni2+ removal. Supporting information file provides the details of the statistical analysis used to assess the results of experimental design using S/N methodology.
Experimental conditions tested to analyze the heavy metal adsorption properties of agricultural residues.

On the other hand, the heavy metal adsorption isotherms were quantified for three biomass residues using an initial concentration range of 100–2500 mg/L for each adsorbate. These isotherms allowed to determine the maximum adsorption capacities of tested biomass wastes and to obtain the corresponding heavy metal saturated samples for the preparation of cement specimens. The equilibrium experiments were carried out in a batch adsorber system with constant agitation of 150 rpm, contact time of 24 h, and biomass dosage (i.e., adsorbent mass/heavy metal solution volume) of 10 g/L at 30°C and pH 5. CaCO3 concentrations used in these experiments were 5 × 10−5 and 1 × 10−4 M for Ni2+ and Pb2+, respectively. In all the heavy metal removal tests, the adsorption capacity of agricultural residues (q, mg/g) was calculated using equation (1):
Agricultural residues with and without loaded heavy metals were analyzed by X-ray spectroscopy (XRD) and Fourier transform infrared spectroscopy (FTIR). XRD diffraction patterns were recorded with an Empyrean Malvern-Panalytical diffractometer using an integrated PIXel 1D detector. The agricultural wastes were processed under the following conditions: 45 kV and 40 mA, Bragg-Brentano configuration, and CuKα1 radiation (λ = 1.5406 Å). HighScore Plus software was used to identify the crystallographic phases. Fourier transform infrared spectra were obtained with a Thermo Scientific Nicolet iS10 infrared spectrophotometer using KBr, and the analysis was carried out in the 4000–400 cm−1 range via 32 scans per sample. These characterization results were used to discuss the heavy metal adsorption mechanism and determine the best model to correlate the adsorption isotherms.
Preparation of cement paste samples using heavy metal-loaded agricultural wastes and characterization of their setting process and compressive strength
Cement paste samples were prepared according to ASTM C192 at different water-to-binder (W/B) ratios ranging from 0.45 to 0.55, and biomass/cement (P/C) ratios ranging from 0.1 to 0.2. Note that the binder was the sum of the cement plus the heavy metal–loaded biomass residue as a mineral admixture. These cement samples were prepared in a closed laboratory where the environmental temperature (ranging from 17 to 22°C) and relative humidity were controlled and recorded. The setting was monitored in all the cement paste samples using a Vicat apparatus, following the ASTM C 191 standard. In this test, the effects of W/B and P/C ratios and particle size of agricultural residues were studied (see Table 2). The response variables were the final setting time of cement samples and their compressive strength.
Experimental conditions used for the preparation of cement paste samples with heavy metal-loaded agricultural residues.
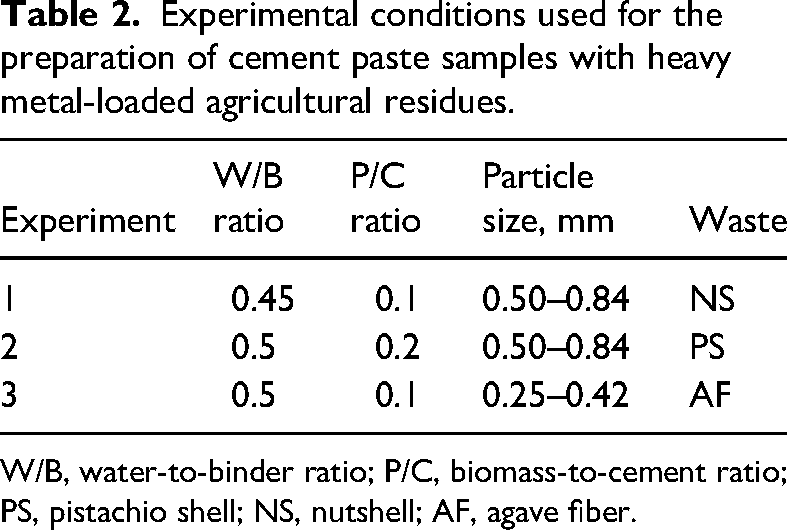
W/B, water-to-binder ratio; P/C, biomass-to-cement ratio; PS, pistachio shell; NS, nutshell; AF, agave fiber.
To evaluate the compressive strength of the cement paste samples, 50 mm × 50 mm × 50 mm cubes were prepared in accordance with the ASTM C 109 Standard Test Method for Compressive Strength of Hydraulic Cement Mortars (using 50 mm cube specimens), and the compressive strength was evaluated at ages of 7, 14, and 28 days. The particle sizes of the heavy metal–saturated biomass residues used to study the mechanical properties of cement paste samples were 0.50–0.84 mm for NS and PS and 0.25–0.42 mm for AF, respectively. The setting and compressive strength experiments were carried out by triplicate.
Results and discussion
Adsorption properties of agricultural residues
Figure 1 shows the adsorption capacities for Ni2+ and Pb2+ using PS, NS, and AF at different removal conditions. Ni2+ adsorption properties of these residues ranged from 1.5 to 14.9 mg/g for PS, 1.2 to 17.9 mg/g for NS, and 8.6 to 16.4 mg/g for AF, while Pb2+ adsorption capacities were 2.0–6.3, 1.9–16.5, and 14.4–103.5 mg/g for the same biomass samples. Table 3 presents a comparison of the adsorption capacities reported for the removal of tested heavy metal ions using these agricultural residues. These results showed that the adsorption capacities of PS, NS, and AF reported in this manuscript agreed with most studies enlisted in Table 3 and, in some cases, a better removal performance was obtained. It is important to note that several of the heavy metal removal studies reported in Table 3 were performed using single aqueous solutions without evaluating the impact of water hardness. Therefore, despite the inclusion of CaCO3 in the heavy metal solutions used in this investigation, which may interfere with the removal of Ca2+, Ni2+, and Pb2+ ions, the adsorption properties of these agricultural residues were competitive, and they can be promising as adsorbents for the depollution of groundwater.

Adsorption capacities for the removal of nickel and lead using wastes of pistachio shell (PS), nut shell (NS), and agave fiber (AF) at different operating conditions.
Adsorption capacities of pistachio shell (PS), nutshell (NS), and agave fiber (AF) residues for the removal of Pb2+ and Ni2+ ions from aqueous solution.
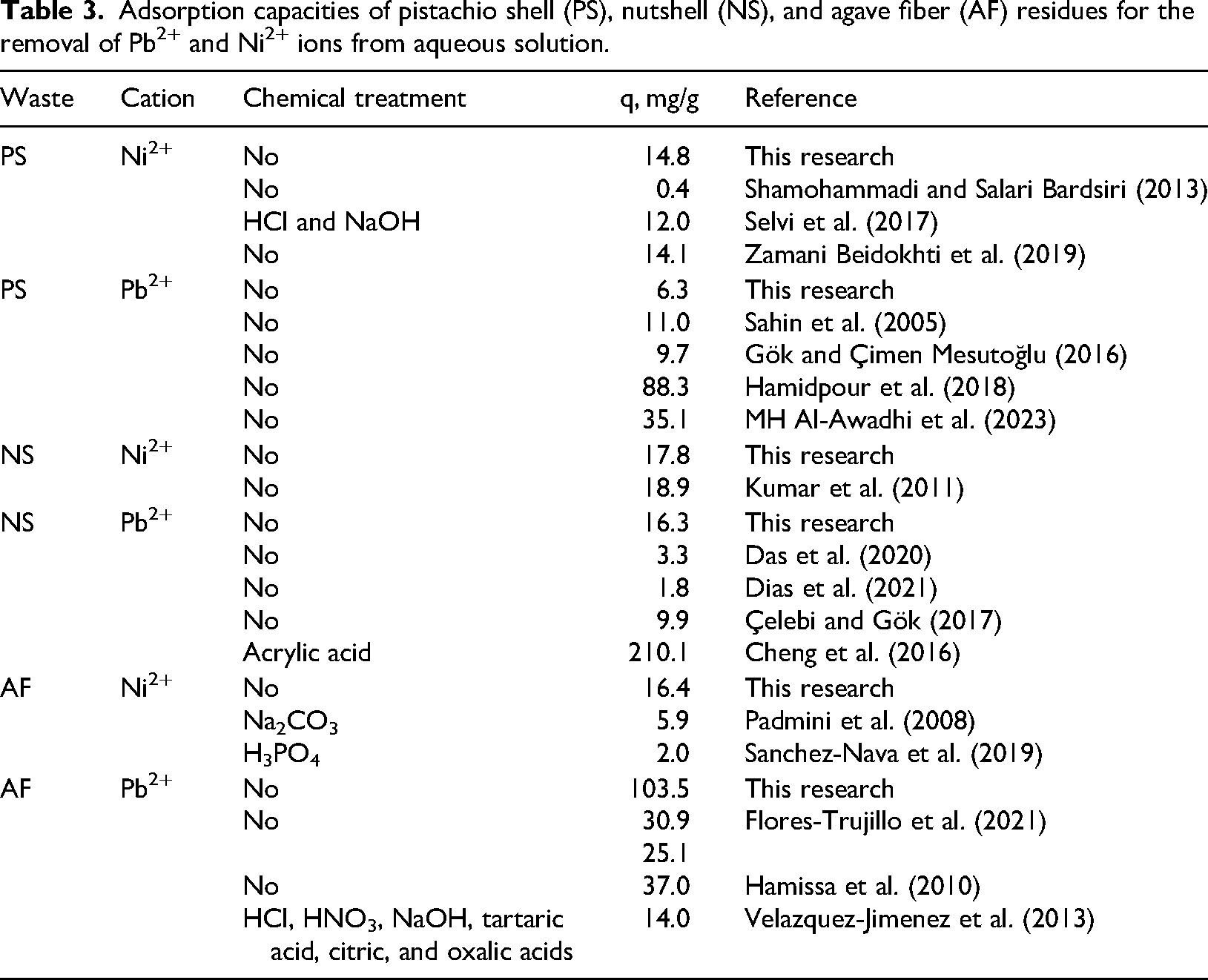
Overall, three agricultural residues had similar adsorption capacities for Ni2+, but NS achieved the best removal of this metal. AF showed a better Pb2+ removal performance and outperformed the adsorption capacities of PS and NS up to 1650 and 630%, respectively. Ni2+ adsorption capacities of tested biomass residues followed the next trend: NS > AF > PS, while Pb2+ adsorption capacities were AF > NS > PS.
Signal-to-noise ratio analysis results for Ni2+ and Pb2+ adsorption on tested biomass residues are reported in Figures 2 and 3, respectively. Overall, it was observed that the removal of both metallic cations improved as solution pH increased. It was concluded that this operating variable showed a significant statistical effect on the removal of Ni2+ and Pb2+ ions using these agricultural residues, see Tables S1 and S2 of Supporting Information. The solution pH is an operating parameter that directly affects the adsorption of ionic species. It was observed that at higher pH values, the adsorption capacity increased for both Ni2+ and Pb2+ using all agricultural residues. This trend was explained by the competition caused by H+ in the aqueous solutions for the binding sites available on the biomass surface. Consequently, the increase in solution pH decreased the competition between H+ and heavy metal cations (Kadirvelu et al., 2001), thereby favoring the presence of negatively charged surfaces of these residues that promote the electrostatic interactions (Kabwadza-Corner et al., 2015; Payne and Abdel-Fattah, 2004). The removal of these heavy metal ions increased from 174–1250% using PS, NS, and AF when the solution pH changed from 3 to 5. The maximum improvement of adsorption capacities was observed for Pb2+ – AF system.

Signal-to-noise (S/N) ratios for the nickel adsorption capacities of (a) Pistachio shell (PS), (b) Nutshell (NS), and (c) Agave fiber (AF) wastes at different removal conditions. Nomenclature: Factor A – solution pH, Factor B – solution temperature, Factor C – CaCO3 concentration, Factor D – biomass-to-volume ratio.

Signal-to-noise (S/N) ratios for the lead adsorption capacities of (a) Pistachio Shell (PS), (b) Nutshell (NS), and (c) Agave fiber (AF) at different removal conditions. Nomenclature: Factor A – solution pH, Factor B – solution temperature, Factor C – CaCO3 concentration, Factor D – biomass-to-volume ratio.
The removal of Ni2+ and Pb2+ cations was also affected by the solution temperature for all tested biomass residues. Ni2+ adsorption was favored and improved up to 347% with the increase in solution temperature, while Pb2+ removal showed the same trend where the adsorption capacities increased up to 880% due to the solution temperature change, see Figures 2 and 3. The highest and lowest changes in the adsorption capacities were observed for NS and AF, respectively. ANOVA of S/N ratios confirmed that solution temperature was the main operating variable that showed the highest statistical effect on the heavy metal adsorption properties of these biomass wastes, see Tables S1 and S2. ANOVA results indicated that temperature had a greater influence on Ni2+ adsorption capacity of PS and NS, and Pb2+ adsorption capacity of NS and AF, see Supporting Information. Solution temperature is a crucial factor that affects heavy metal removal due to its impact on the kinetic energy of the dissolved cations (Kumar et al., 2011). It has been often observed that the mass transfer increases at high solution temperatures because the higher kinetic energy of adsorbates facilitates their diffusion and interactions with adsorbent active sites (Uddin and Nasar, 2020; Das et al., 2020).
The presence of CaCO3 in the aqueous solution affected the removal of both heavy metal ions but in different degree. Ni2+ adsorption was more sensitive to this parameter where the adsorption capacities of biomass wastes reduced from 25–52% as CaCO3 concentration increased in the aqueous solution. The adsorption properties of AF biomass were more significantly affected by this parameter that is associated with the water hardness. Pb2+ removal using PS and NS was not significantly affected by CaCO3 concentration in contrast to AF, see Figure 3. It is convenient to recall that the impact of CaCO3 concentration on adsorption properties of these residues also appeared to be depended on the biomass dosage, pH, and temperature conditions that were applied in the removal experiments. As indicated, the addition of CaCO3 in the heavy metal solutions allowed to simulate the characteristics of groundwater. Note that the groundwater used for human consumption can exhibit significant variations in its CaCO3 content, depending on geological and environmental factors. CaCO3 is commonly found in groundwater due to the dissolution of minerals such as calcite and dolomite from the rock strata (Wen et al., 2020). Therefore, the reduction in the adsorption capacities of tested cations was associated with the competitive effective caused by calcium ions contained in the aqueous medium (Qian et al., 2024). Note that previous adsorption studies have shown that the adsorption of metallic cations is antagonistic (i.e., competitive) using several adsorbents including lignocellulosic biomass residues (Reynel Ávila et al., 2016; Liu et al., 2023). However, some studies have indicated that Pb2+ adsorption is not significantly affected by the presence of other co-ions in the aqueous solutions (Reynel Ávila et al., 2016). For example, Reynel Ávila et al. (2016) showed that Cd2+ and Ni2+ removal can be significantly affected by Pb2+ ions in binary aqueous solutions using residues of flamboyant and bush mango. In contrast, the adsorption of Ni2+ ions can be significantly affected by the competition caused by other metallic ions (Reynel Ávila et al. 2016).
ANOVA results indicated that the best conditions for the removal of Ni2+ using PS were pH 5, 15°C, 5 × 10−5 M CaCO3 concentration, and 25 g/L of biomass dosage (A3B1C1D2). For the case of NS and AF, the best Ni2+ removal conditions were pH 5, 15°C, 5 × 10−5 M CaCO3 concentration, and 10 g/L of biomass dosage (A3B1C1D1). The best operating conditions to remove Pb2+ using PS and NS were pH 5, 35°C, 1.3 × 10−4 M CaCO3 concentration, and 10 g/L of biomass dosage (A3B3C3D1); while the best Pb2+ adsorption conditions using AF were pH 5, 35°C, 5 × 10−5 M CaCO3 concentration, and 10 g/L of biomass dosage (A3B3C1D1), see Figures 2 and 3.
The adsorption isotherms of Ni2+ and Pb2+ on PS, NS, and AF are shown in Figure 4. These isotherms were obtained at 30°C and pH 5, 10 g/L biomass dosage, and CaCO3 concentration = 5 × 10−5 and 1 × 10−4 M for Ni2+ and Pb2+, respectively. The maximum experimental adsorption capacities ranged from 6.3 to 103.5 mg/g for Pb2+ and from 14.8 to 16.3 mg/g for Ni2+. These isotherms showed the next trends for the adsorption properties of tested biomass residues: Pb2+ (AF > NS > PS) and Ni2+(NS > AF > PS), thus confirming the results obtained from the experimental design given in Table 1.
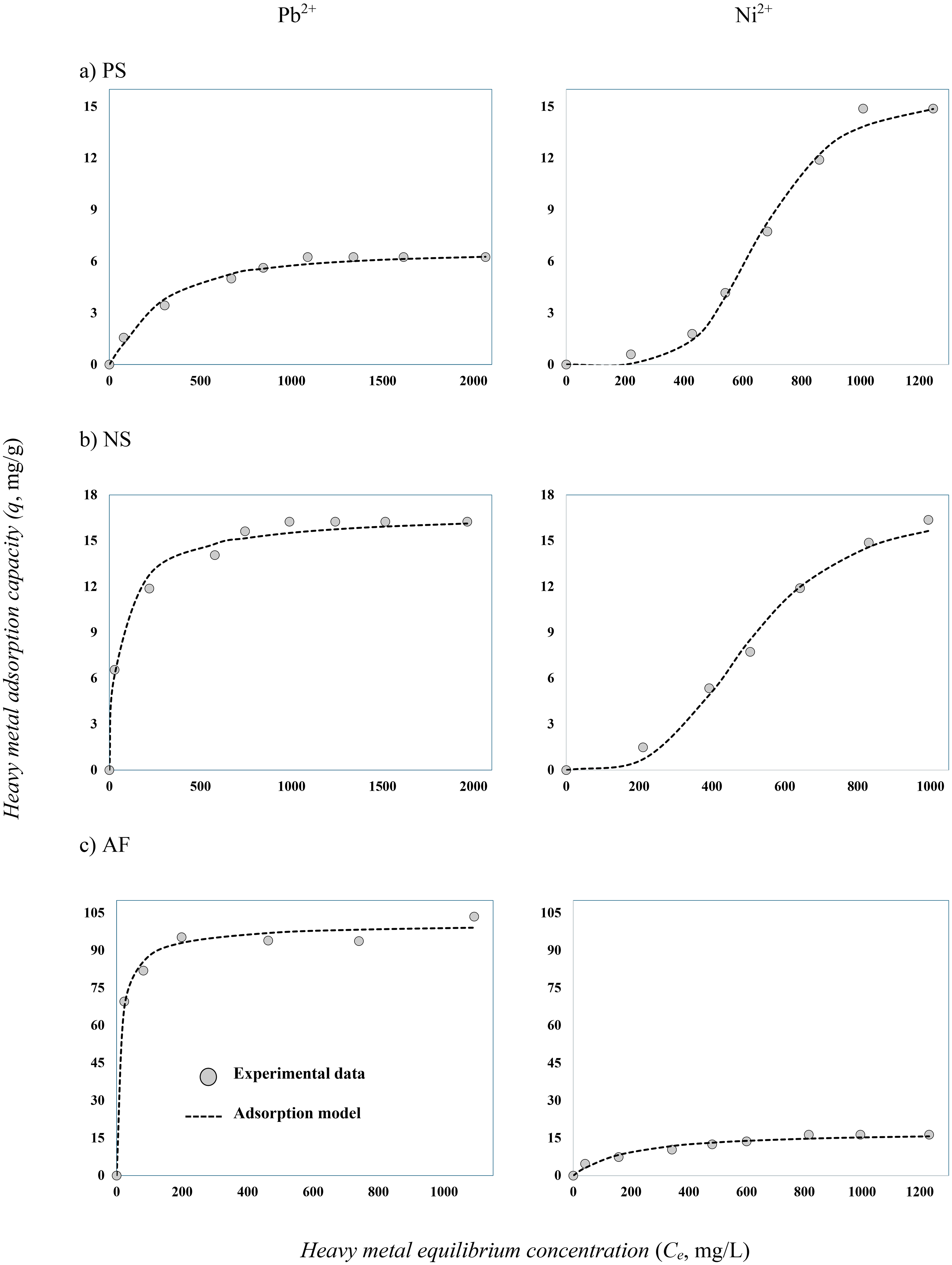
Adsorption isotherms for the removal of lead and nickel using pistachio shell (PS), nutshell (NS), and Agave fibers (AF) at pH 5 and 30°C. CaCO3 concentration of 5 × 10−5 M for Ni2+ and 1 × 10−4 M for Pb2+ and biomass dosage of 10 g/L.
Surface chemistry of tested agricultural residues was analyzed, and the results are reported in Figures 5 and 6. Fourier transform infrared spectra of agricultural residues were similar and contained the main absorption bands commonly identified for lignocellulosic materials, see Figure 5(a). The spectra of all the residues contained the characteristic absorption band of the hydroxyl group (O-H) at 3060–3700 cm−1, while the band observed at 2825–2970 cm−1 was attributed to the C-H bond stretching (Carvalho et al., 2023; Jennita Jacqueline et al., 2022). In the 1000−1800 cm−1 region of FTIR spectra, several absorption bands attributed to cellulosic compounds were identified. In particular, the absorption bands observed at 1050, 1245, and 1320 cm−1 were related to C-O, while the bands located at 1377, 1425, and 1462 cm−1 were attributed to C-H bond stretching (Chávez-Guerrero et al., 2017; Oudiani et al., 2017). The absorption bands identified at 1510 and 1630 cm−1 were associated with the C = C bond from the stretching of aromatic rings and the absorption band at 1735 cm−1 indicated the presence of carbonyl bond (C = O) from ketones (Chávez-Guerrero et al., 2017). For the case of agricultural wastes saturated with Ni2+ and Pb2+, it was observed some changes in the absorption bands of oxygenated functionalities from biomass surfaces indicating their participation in the heavy metal adsorption (Reynel Ávila et al. 2016).

Fourier transform infrared spectroscopy (FTIR) spectra of Pistachio Shell (PS), Nutshell (NS), and Agave Fibers (AF) with and without loaded heavy metals.

XRD patterns of Pistachio Shell (PS), Nutshell (NS), and Agave Fibers (AF) with and without loaded heavy metals.
XRD patterns of waste samples are reported in Figure 6. Slight differences were observed in the diffraction patterns especially for AF and NS. It was identified the presence of hydrated calcium oxalates (ICDD: 00-001-0157 and 00-020-0231) for NS and AF via the diffraction signals at 14.9, 24.3, and 30.2 °2θ (Hernández-Meléndez et al., 2016; Chávez-Guerrero et al., 2017). The diffraction signals located at 16.8, 22.1, and 34.7 °2θ in the XRD patterns of three biomass residues were associated with the characteristic peaks of cellulose (Chávez-Guerrero et al., 2017). After the heavy metal adsorption, a slight change in sample crystallinity was observed but without identifying additional crystallographic phases related to the adsorbed heavy metal ions on biomass surfaces.
Based on these results of surface chemistry characterization, Pb2+ and Ni2+ adsorption isotherms were modeled assuming a monolayer process where oxygenated functional groups (i.e., carboxylic, hydroxylic, and phenolic) from residue surfaces were hypothesized to be the main active sites. Phenolic (OH) groups are mainly derived from lignin, while the carboxylic (COOH) groups are attributed to hemicellulosic compounds contained in the biomass residues (Zhang et al., 2019; Lindholm-Lehto, 2020). On the other hand, hydroxyl groups can be mainly derived from cellulose and hemicellulose (Singh et al., 2015). These oxygenated functionalities are deprotonated in the aqueous solution under tested pH condition (Singh et al., 2015; Lindholm-Lehto, 2020). Consequently, the metallic cations dissolved in the aqueous solution can be attracted by the negatively charged functional groups. Figure 7 illustrates the expected mechanism for the adsorption of Pb2+ and Ni2+ on lignocellulosic biomass waste (Singh et al., 2015; Zhang et al., 2019; Lindholm-Lehto, 2020).

Illustration of the adsorption mechanism of heavy metals (Ni2+ and Pb2+) on the main functional groups of cellulose, lignin, and hemicellulose contained in agricultural wastes.
For the modeling of adsorption isotherms, it was considered that nion heavy metal ions were adsorbed on the available oxygenated functionalities (DBio, mmol/g). Then, this monolayer adsorption model (Amrhar et al., 2021) is defined by equation (2):
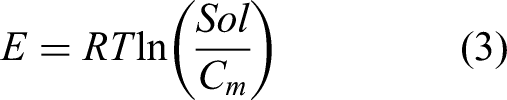
Calculated concentrations of oxygenated functional groups involved in Pb2+ and Ni2+ adsorption using different agricultural wastes.

PS, pistachio shell; NS, nutshell; AF, agave fiber.
Preparation of cement paste samples using heavy metal-loaded agricultural residues and characterization of the setting process
The results of the evaluation of the setting process using the cement paste samples prepared with the heavy-metal-saturated agricultural wastes, under the conditions of Table 2, are shown in Figure 8. It was observed that the setting was accelerated for the cement samples prepared with heavy metal-loaded NS and PS. This result can be explained by the fact that water, because of its solvent behavior, has an affinity for the Pb2+ and Ni2+ contained on the biomass surface allowing the hydration process to be more homogeneous between the biomass residue and the cement particles and also increases the water evaporation. Consequently, the setting process was accelerated. For the case of cement sample prepared with AF, a deceleration occurred in the setting process. Note that AF showed the best heavy metal adsorption properties and, consequently, the highest amount of heavy metal ions on its surface. The presence of this solute can contribute to modify the water vapor pressure and, therefore, the sample can require a greater amount of energy to be able to change phase (Zhang et al., 2021). Thus, the setting proceeded more slowly.

Setting curves of cement paste samples prepared with heavy metal-loaded agricultural residues.
Figure 9 shows the evolution of the compressive strength of samples obtained with heavy metal-loaded agricultural residues, as well as in their natural form, at 7, 14, and 28 days after their preparation. These results allowed to assess the effect of the presence of the metal on the mechanical property of cement paste samples containing biomass wastes. A clear trend was observed because the compressive strength of the samples prepared with biomass saturated with heavy metals increased in all cases. For the samples containing AF and PS, a higher resistance was reached at 28 days with the biomass saturated with Ni2+. In particular, the cement paste sample obtained with Pb2+-loaded NS exhibited the highest resistance especially at 7 and 14 days. This fact could be explained in terms of the better homogeneity and lower porosity of the cement paste from the microstructural point of view because the presence of metal on biomass surface allowed the development of reinforced hydration products, which translated into a better mechanical behavior (Gitanjali et al., 2024).

Compressive strength of cement paste samples using Pistachio Shell (PS), Nutshell (NS), and Agave Fibers (AF) as mineral admixtures in their natural form and saturated with heavy metals.
The utilization of NS for cement sample preparation provided greater resistance at 28 days when this biomass residue was saturated with Ni2+ and Pb2+. For these samples, increments of 7.2 and 10.7%, respectively, were observed in comparison to the cement sample obtained with the raw waste (i.e., without heavy metals). It was also possible to observe an improvement of the mechanical properties of cement paste samples prepared with PS saturated with heavy metals (i.e., 8.8 and 4.2% for Ni2+ and Pb2+, respectively). Finally, the compressive strength of cement samples containing AF increased up to 253.7 and 238.6% when the biomass was saturated with Ni2+ and Pb2+, respectively.
Therefore, it was clear that when NS loaded with Pb2+ was used as a mineral admixture in cement paste preparation, higher compressive strengths were observed. At the same time, the setting was lower, followed by the samples prepared with the agricultural residue saturated with Ni2+, where the compressive strengths increased with respect to the samples obtained with unsaturated waste. In the case of cement paste samples prepared with Ni2+-saturated AF, a higher setting time was obtained in comparison to the cement samples containing the unsaturated fiber. However, it should be mentioned that this mixture exhibited the best compressive strength especially at 7 and 14 days, followed by the sample loaded with Pb2+, which had a shorter setting time with respect to the sample prepared with the biomass residue saturated with Ni2+.
It is important to mention that the lignin content is related to the strength and stiffness of these biomass residues and, consequently, this parameter can be used to explain the trend of the mechanical strength of tested cement pastes. NS has 49 wt% of lignin (Barbu et al., 2020), whereas the lignin content of PS is between 6 and 15 wt% (Jadhav et al., 2022). Therefore, the cement paste samples prepared with NS showed the best mechanical properties.
Finally, the metals contained in the saturated adsorbent were stabilized via its incorporation into the cement matrix and, consequently, their leaching was significantly reduced. Previous studies proved that this approach is very effective for the immobilization of high concentrations of metal species due to the formation of phosphate salts and alkaline nature (Al-Kindi, 2019; Xia et al., 2019; Al Kindi and Al-Haidri, 2022). Herein, it is also important to remark that the recycling of adsorbents saturated with metals (after the treatment process to clean polluted water) in the manufacturing of cement-based specimens offers additional advantages over the adsorbent regeneration, avoiding the environmental pollution caused by the management of desorbing fluid that contains high concentrations of heavy metals. This approach can be applied in water treatment in vulnerable areas where the purification cost is a limitation.
Conclusions
This study reports a circular economy-based approach for the reuse and final disposal of agricultural residues applied in the heavy metal removal. First, the heavy metal adsorption properties of different lignocellulosic biomass residues from Mexico were evaluated under a variety of removal conditions. The results showed that the heavy metal adsorption properties of these residues depended on pH and temperature. The best removal conditions for Pb2+ and Ni2+ ions using these residues were identified via the S/N ratio analysis. NS, PS, and AF presented a similar performance in the removal of Ni2+, whereas Pb2+ removal using AF outperformed the results obtained with NS and PS. These biomass residues with loaded heavy metals were used to prepare cement paste samples to determine the impact of these materials as mineral admixtures on the mechanical properties of cement-based specimens. The setting process of cement paste samples was favored considerably using raw AF followed by NS and PS. This setting process was accelerated using NS and PS loaded with heavy metal ions, whereas it was decelerated for the cement specimens containing AF saturated with heavy metals. The lignin content of these biomass wastes affected the compressive strength of the cement paste samples. These results proved that it is possible to reuse pollutant-loaded adsorbents generated in a water purification process for the preparation of cement paste achieving a better performance under both fresh (setting) and hardened (compressive strength) conditions. This circular economy approach is attractive to face the waste management problem caused by agricultural residues and the secondary pollution problem generated by water treatment using adsorption processes.
Supplemental Material
sj-docx-1-adt-10.1177_02636174241298784 - Supplemental material for A circular economy-based valorization of agricultural residues to remove heavy metals from water: Adsorption and final disposal strategy
Supplemental material, sj-docx-1-adt-10.1177_02636174241298784 for A circular economy-based valorization of agricultural residues to remove heavy metals from water: Adsorption and final disposal strategy by M.R. Moreno-Virgen, K.A. Rodríguez-García, A. Bonilla-Petriciolet, H.E. Reynel-Ávila, F.J. Villalobos-Delgado, D.I. Mendoza-Castillo, O.F. González-Vázquez, H.E. Reynel-Ávila, D.I. Mendoza-Castillo and J.A. Ortiz-Lozano in Adsorption Science & Technology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Tecnológico Nacional de México, (grant number 14804.22-P).
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
