Abstract
The improper or excessive use of herbicides has caused soil and water contamination, increasing the demand for techniques for environmental remediation. In this research, we assessed the effectiveness of nanostructured adsorbents in removing the herbicides mixtures of diuron, hexazinone, and sulfometuron-methyl, with the goal of applying this technique in environmental remediation efforts. The nanomaterials employed were microporous beta zeolite (BT), Al-MCM-41 mesoporous silica (AL), hydrotalcite clay (HT), and microporous carbonate biochar (BIO). The nanostructured adsorbents were characterized by X-ray diffraction, X-ray fluorescence, elemental analysis, and nitrogen adsorption and desorption at 77 K, point of zero charge, scanning electron microscope, and transmission electron microscopy. The descending adsorption values of nanostructured adsorbents: for diuron BT (99.35%) > BIO (96.36%) > AL (88.33%), hexazinone, BT (97.26%) > AL (83.75%) > BIO (14.22%), and sulfometuron-methyl, AL (95.73%) > BT (90.96%) > BIO (34.92%). The time required for the adsorption equilibrium was 1 hour, with greater adequacy to the pseudo-second-order kinetic model. The Langmuir and Freundlich isotherms showed the best fit of herbicide adsorption on the microporous BT and BIO, respectively. In the AL, the diuron and hexazinone experimental data fitted more precisely to Temkin's model and sulfometuron-methyl to Freundlich's model. Nanostructured adsorbents have different capacities to adsorb diuron, hexazinone, and sulfometuron-methyl herbicides present in aqueous media, with BT being the material with the highest adsorption capacity for the remediation of the mixture containing the three herbicides.
Introduction
In recent years, the class of pesticide most applied to crops has been herbicides, products used to control weeds and, consequently, avoid negative interference in the production and quality of agricultural products (Turek et al., 2022). Furthermore, applying herbicides in mixtures enhances both the effectiveness and the spectrum of weed control (Barbieri et al., 2022). However, the incorrect and excessive use of these products can cause soil and water contamination and harm human health (Sharma et al., 2019; Gawel et al., 2020; Alengebawy et al., 2021; Mielke et al., 2022).
The behavior of herbicides varies according to the physicochemical properties of the product used. These products can persist in the soil, generating a residual effect (carryover) by contaminating sensitive crops planted later (Milke et al., 2022). These products’ remarkable persistence can contaminate surface, groundwater, and nontarget organisms (Walker et al., 2005; Yavari et al., 2022).
In the study conducted by Carneiro et al. (2020), it was observed that the application of herbicides diuron, hexazinone, and sulfometuron-methyl in mixtures enhances their availability in soil solution and interactions may affect their environmental fate (Dos Reis et al., 2023; Mendes et al., 2016). This heightened availability substantially increases the risk of environmental contamination due to leaching. Groundwater contamination by herbicides can result from their ability to infiltrate the soil profile, particularly during prolonged periods of rainfall. This issue is exacerbated when the herbicide is highly soluble in water, such as hexazinone (solubility of 33,000 mg L−1 a 25 °C) (PPDB, 2021), as noted in prior research (Neto et al., 2017; Passos et al., 2019).Thus, using techniques to reduce the amount of these pesticides in the soil and water can reduce the impact caused.
The characteristics of the adsorption process and the choice of adsorbent is of fundamental importance for the efficient removal of organic contaminants, considering that the characteristics of adsorption capacity, specific surface area, pore volume, particle size, and distribution of pore size are essential for satisfactory results in the adsorptive capacity of the pollutant (Kaykhaii et al., 2018).
The efficiency of the technique chosen for herbicide remediation programs will depend on the interaction between the contaminant and the material used for adsorption. Some nanostructured adsorbents, such as zeolites, biochar, mesoporous silica, and hydrotalcite, have the potential for adsorbing herbicides because they have favorable properties for interaction with ionic and nonionic pesticides. Among these properties, the high surface area and porosity can be highlighted (Costa et al., 2017; Sikander et al., 2017; Zhang et al., 2018; Zhu et al., 2018; Dai et al., 2019; Zhu et al., 2020).
Unlike most articles that evaluate the removal of a single herbicide, the main objective of this article is to evaluate the adsorption capacity of different nanostructured adsorbents to remediate water contaminated with a mixture of herbicides (diuron, hexazinone, and sulfometuron-methyl). By comparing physicochemical characteristics of materials with kinetic study and adsorption balance we will increase the understanding of the interactions between herbicide mixtures and nanoadsorbents, thus developing more effective and sustainable remediation strategies to reduce the negative impacts of herbicide use in agriculture.
Material and methods
Synthesis of nanostructured adsorbents
Beta zeolite
Beta zeolite (BT) synthesis followed the procedure proposed by Camblor et al. (1997), with some adaptations. The following stoichiometric ratio of Si/Al equal to 30 (Al2O3: 60SiO2: 15.5 tetraethylammonium hydroxide (TEAOH): 1000 H2O) was used for this synthesis. Initially, two solutions were prepared: solution 1 was prepared by dissolving aluminum flakes (Sigma-Aldrich, 98%) in TEAOH (alpha solution 35%) and deionized water under constant stirring for 6 h at room temperature (25 ± 2°C). In parallel, solution 2 was prepared by dissolving the fumed silica in TEAOH and deionized water under constant stirring at room temperature until complete homogenization. Subsequently, solutions 1 and 2 were mixed and kept under stirring for 20 h at room temperature. The synthesis gel was transferred to a stainless steel-coated teflon autoclave at 135°C for 7 days under static conditions. After crystallization, the material was centrifuged at 11,000 rpm for 20 min three times, washed with deionized water, dried at 110 °C for 12 h, and calcined at 550 °C for 6 h with a heating ramp of 1.8 °C.min−1 in static air atmosphere.
Al-MCM-41 mesoporous silica
The synthesis of mesoporous silica Al-MCM-41 (AL) was carried out according to the method proposed in the literature (Matsumoto et al., 1999), with the following molar ratio Si/Al equal to 20 (1.5Al2O3: 60SiO2: 9CTAB: 165NH3: 8000H2O). Initially, two solutions were prepared: solution 1, formed by a suspension of aluminum isopropoxide (IPA—Aldrich, 98%) in isopropanol (2-propanol—Scharlau, 99.8%) and tetraethylorthosilicate (TEOS—Aldrich, 98%) was stirred in an ice bath for 1 h. Solution 2 was formed by a mixture of ammonium hydroxide (NH4OH—Scharlau, 32%) and cetyltrimethylammonium bromide (CTABr—Aldrich, 99%) and stirred for 30 min at room temperature. Then, solution 2 was added to solution 1 and kept under stirring for 30 min at room temperature. The synthesis gel was transfered to a Teflon-lined stainless steel autoclave under autogenous pressure and static conditions at 110°C for 2 days. Subsequently, the obtained material was cooled, washed, and filtered several times with deionized water, dried at 110 °C for 12 h, and calcined at 550 °C for 6 h with a heating ramp of 1.8 °C.min−1.
Hydrotalcite clay
Hydrotalcite clay (HT) was synthesized using the co-precipitation method with variable pH (between 10 and 11), followed by hydrothermal treatment. For this synthesis, a mixture of two cation solutions was used (solution 1): Magnesium nitrate hexahydrate (Mg(NO3)2.6H2O—Vetec, Duque de Caxias, Rio de Janeiro, Brazil) 1.6 mol L−1 and aluminum nitrate nonahydrate (Al(NO3)3.9H2O—Vetec)) 0.8 mol L−1, with a molar ratio Mg/Al equal to 2:1. A mixture formed from anhydrous sodium carbonate solution (Na2CO3)—(Vetec) 1.07 mol L−1 and a solution of sodium hydroxide (NaOH—obtained from Merck KGaA (Darmstadt, Hesse, Germany)) 2 mol L−1, formed solution 2. Solution 1 was solution 2, added by drop and under constant stirring at room temperature. Subsequently, heating between 60°C and 70°C was used under stirring for 24 h. The material obtained was filtered and washed with distilled water until reaching a pH between 10 and 11. Then the material was dried at 105°C for 2 h.
Biochar
Biochar (BIO) was produced from Eucalyptus sp. wood residues resulting from the clone of crossing Eucalyptus urophylla x Eucalyptus grandi. The material was crushed into small pieces using a Tecnal knife mill (Willey TE 340, Piracicaba, Brazil). Soon after, the crushed biomass was passed through a 2 mm sieve and dried in an oven with air circulation at 103 ± 2 °C. The pyrolysis of the biomass was carried out in a metallic cylinder inside a muffle (Q318 M, Quimis, Diadema, São Paulo, Brazil) at 950 °C, with a temperature increase of 1.7 °C min−1; after reaching the final temperature, it was maintained for 1 h under limited supply of air (Dias et al., 2016). After cooling, the BIO was ground, passed through a < 0.9 mm sieve, and stored in a desiccator. This material was selected for the experiment because it has a high adsorption capacity for the herbicide hexazinone (Fernandes et al., 2021).
Characterization of nanostructured adsorbents
Chemical composition and crystalline structure
The X-ray diffraction (XRD) patterns for all nanostructured adsorbents were obtained in a Rigaku diffractometer, model Mini Flex II using Cu Kα radiation (λ = 1.54056 Å) with a current of 15 mA, potential of 30 kV. For AL, a low angle 2θ sweep of 0.5° to 10° was used, using a step of 0.03 degrees s−1 and a time of 5 seconds. For the other materials, high-angle scans were used. For BT, a 2θ sweep from 5° to 50° was used, applying a step of 0.04 degrees s−1 and a time of 2 seconds. For HT and BIO, a 2θ angle sweep of 10° to 70° was applied using a step of 0.02 degrees s−1 with a time of 2 seconds.
The amount of aluminum and the Si/Al molar ratio for BT and AL were obtained through atomic emission spectroscopy analysis by induced plasma coupling (ICP-AES) in a VARIAN equipment, model VISTA AX CCD. Quantification of aluminum was performed from the emission line obtained at 396 nm. For HT, the Mg/Al molar ratio values were obtained by energy dispersive X-ray fluorescence (EDX) measurements in a Shimadzu EDX-7000 equipment with a Rh tube and a 10 mm diameter beam.
For the chemical determination of BIO by carbon (C), oxygen (O), hydrogen (H), and nitrogen (N), an elemental analyzer (Vario MACRO Cube, Elementar, Cheadle Hulme, UK) was used 100 mg of BIO were weighed in the tin capsule; then, the capsule was closed and placed in the elemental analyzer, which was operated by subjecting the samples to combustion in an atmosphere of pure oxygen (99.999%). The gases formed by this combustion were measured with a thermal conductivity detector. O content was estimated by mass balance: O = 100 − (C + H + N+ mineral).
Textural properties
The N2 adsorption and desorption isotherms at 77 K were obtained using the Micromeritics ASAP2020 equipment. To carry out the measurements, each sample was subjected to a degassing process at 573 K for 10 h, except for the HT that degassed was subjected to a heat treatment in an oven at 423 K for 4 h. Subsequently, the HT was degassed at 423 K for 10 h. The specific area (SBET) of all materials was obtained by applying the Brunauer–Emmett–Teller (BET) equation (Brunauer et al., 1938). The microporous volume (VμP) and the microporous (SμP) and external (SEXT) areas of BT and BIO were obtained by the αs-plot method (Villarroel et al., 2013). For the AL material, the mesoporous volume was determined by applying the BJH method (Barrett et al., 1951). The total volume of all nanostructured adsorbents was determined by the Gurvich method (Rouquerol et al., 2013).
The point of zero charge (PZC) was determined based on the methodology of Calvete et al. (2009), with some adaptations. First, a 0.05 mol/L NaCl solution was prepared in a 1000 mL volumetric flask. Then, 100 mL of this solution was distributed into several beakers, and the pH levels were adjusted by adding 0.1 mol/L NaOH and HCl, with tests conducted over a pH range of 2.0 to 10.0. Afterward, 30 mL of the 0.05 mol/L NaCl solution adjusted to each pH was added to Falcon tubes containing 200 mg of biochar. The solutions were stirred for 48 h at 25°C. The suspensions were centrifuged at 3600 r/min for 5 min, and the final pH of the supernatants was recorded. The analyses were carried out in triplicate. The PZC is determined by the point where the ΔpH (pHf − pHi) versus pHi curve crosses the zero line (Ofomaja and Ho, 2007).
Surface morphologies
Scanning electron microscopy (SEM) and transmission electron microscopy (TEM) were employed to examine the morphology of the materials. For SEM imaging, the materials were affixed onto a metal sample holder using conductive tape (PELCO Tabs™, Ted Pella, Inc., Redding, CA, USA) and coated with a 9 nm layer of gold using a sputter coater (Q150R ES, Quorum Technologies Ltd, Laughton, East Sussex, UK). SEM images were captured using an SEM (VEGA 3 LMU, Tescan, Czech Republic) operated at 30 kV with a secondary electron detector. TEM were collected on a microscope (Phillips TECHNAI 20, Móstoles, Madrid, Spain) equipped with LaB6 filament under an accelerating voltage of 200 kV. The samples were dispersed in acetone, stirred in an ultrasonic bath and finally deposited over a carbon-coated copper grid.
Reagents
Analytical grade and HPLC (high-performance liquid chromatography) reagents were used in all assays. Diuron (99.6%) and hexazinone (99.5%) standards were purchased from Sigma-Aldrich® (Saint Louis, MO, USA), sulfometuron-methyl (99.5%) from Supelco (Bellefonte, PA, USA). Acetonitrile and methanoic acid were obtained from Merck KGaA (Darmstadt, Hesse, Germany) and ultrapure water from Scharlau (Sentmenat, Barcelona, Spain). A stock solution was prepared for each herbicide at a concentration of 500 mg L−1 in acetonitrile and stored in amber bottles at −18 °C. The working solution was prepared by successive dilutions, with a concentration of 160, 70, and 4 µg L−1 of diuron, hexazinone, and sulfometuron-methyl, respectively, which corresponds to the highest recommended dose of the commercial product (FRONT®, Corteva, Barueri, São Paulo, Brazil). The physicochemical properties of the herbicides are shown in Table 1.
Physicochemical properties of diuron, hexazinone, and sulfometuron-methyl.

PubChem (2022) and PPDB (2022).
ASP: polar surface area; Kow: octanol/water partition coefficient; LH: hydrogen bond donor/acceptor; MM: molecular mass; pKa: logarithm of acid constant; PV: vapor pressure; SW: water solubility.
Chromatographic and mass spectrometry conditions
The LC-MS/MS is an ultra-HPLC (UHPLC) system coupled to a Shimadzu triple quadrupole mass spectrometer (LCMS-8040). The UHPLC is equipped with a Restek column (Pinnacle DB AQ C18 size 50 × 2.1 mm, with 1.9 μm particles), including two LC-30AD pumps, a DGU-20A5R degasser, a Sil- 30AC, a CTO−30AC column furnace and a CBM−20A controller. The operating conditions of the chromatographic system to obtain the best resolution during the analysis were through isocratic elution with a flow of 0.3 mL min−1, injection volume of 5 μL and the temperature of the automatic sampler at 15 °C. Mobile phase A is constituted by water with 0.1% formic acid, and mobile phase B by acetonitrile, in the proportion of 30% to 70%, respectively. The column oven temperature was set at 40 °C.
The electrospray ionization (ESI) source mass spectrometer was operated in positive ionization mode. Parameters for monitoring multiple reactions (MRM) are summarized in Table 2. Interface voltage was set to 4.5 kV, desolvation line temperature was 250 °C, nebulizing nitrogen gas flow was 3 L min−1, block temperature of 400°C, drying nitrogen gas flow with 15 L min−1, and collision argon gas with a pressure of 230 kPa.
Optimized parameters.

CE: collision energy; DP: decomposition potential; MRM: multiple reaction monitoring.
Maximum adsorption capacity
A preliminary trial was conducted to determine the feasibility of using the four nanomaterials to decontaminate water containing diuron, hexazinone, and sulfometuron-methyl. In Falcon tubes, 20 mg of the nanomaterial was added with 2 mL of the working solution; the tubes were subjected to vertical agitation (Wagner shaker, Model MA 160, Marconi, Piracicaba, São Paulo , Brazil) for 2 hours with rotation of 12 rpm, then, they were centrifuged (Centrifuge Excelsa II, 206 II, FANEM, São Paulo, Brazil) at 3600 rpm for 7 min. A 1 mL aliquot was filtered through a 0.22 µm nylon membrane into 1.5 mL vials for LC-MS/MS quantification. After analyzing the results, it was found that HT did not adsorb the evaluated herbicides, unlike HT, BT, AL, and BIO adsorbents were potentially promising in the adsorption of herbicides. The removal values found for AL were within the limits of detection (LD) and quantification (LQ), whereas the values obtained for BT and BIO were below LD and LQ.
However, as previously mentioned, the experiment was repeated for BT and BIO adsorbents, using 10 and 20 mL of the working solution. It was verified that the BT and BIO nanomaterials, in the volumes of 20 and 10 mL, presented results within LD and LQ, respectively. Consequently, volumes 2, 10, and 20 mL were standardized for the other AL, BIO, and BT assays. The study continued by testing the maximum adsorption capacity according to methodology adapted from Genskowsky et al. (2015). Masses of 10, 20, 30, 40, and 50 mg were defined for BT and BIO and AL, 10, 13, 17, and 20 mg. The assay was conducted in 50 mL Falcon tubes containing the nanomaterial mass plus 2, 10, or 20 mL of the working solution for AL, BIO, and BT nanomaterials. Then, they were subjected to vertical agitation for 2 hours at 12 rpm. After shaking, the tubes were centrifuged at 3600 rpm for 7 minutes. A 1 mL aliquot was taken and filtered through a 0.22 µm nylon membrane into 1.5 mL vials for quantification by LC-MS/MS. The amount of herbicides adsorbed on each material was determined by equation (1).
Kinetic study
The kinetic study was carried out using the “batch equilibrium” method (OECD, 2000). The amount of each material selected by the previous test was placed in different Falcon-type tubes, 20 mg for BT and AL, and 30 mg for BIO, then 20 mL of the working solution was added to the tubes with BT, 10 mL for BIO, and 2 mL for AL. The tubes were shaken vertically at time intervals (20; 30; 60; 90, and 120 minutes) at 12 rpm. After shaking, the samples were centrifuged at 3600 rpm for 7 minutes. A 1 mL aliquot was filtered through a 0.22 µm nylon membrane into 1.5 mL vials to quantify LC-MS/MS. The amount of herbicides adsorbed on each material was determined by equation (2). Pseudo-first-order (Lagergren, 1898) (equation (3)) and pseudo-second-order (Blanchard et al., 1984) (equation (4)) kinetic models were fitted to the experimental results.


In which Qt (mg g−1) is the amount adsorbed in time, Co (mg L−1) the initial concentration, Ct (mg L−1) the concentration at the evaluated instant, m (g) the mass, V (L) the volume, qe (mg g−1) the amount adsorbed at equilibrium, K1 (min−1) and K2 (g mg min−1) the rate constants, and t (min) the time.
Adsorption balance
The adsorption equilibrium test was also carried out using the “batch equilibrium” method (OECD, 2000). Four solutions containing 25%, 50%, 75%, and 100% of the concentration of the working solution were prepared for the test. A mass of BT (20 mg), AL (20 mg), or BIO (30 mg) nanomaterials was placed in 50 mL Falcon tubes, plus 20 mL of solution in the tubes with BT, 10 mL for BIO, or 2 mL for AL. The tubes were vertically shaken at 12 rpm for the period defined in the kinetic assay. After this time, the samples were centrifuged at 3600 rpm for 7 minutes. A 1 mL aliquot was filtered through a 0.22 µm nylon membrane into 1.5 mL vials for LC-MS/MS quantification. The amount of herbicides adsorbed on each material was determined by equation (1). The isothermal models of Freundlich (equation (5)), Langmuir (equation (6)), Temkin (equation (8)), and Dubini–Radushkevich (equation (9)) were adjusted to the test results. The average adsorption-free energy was calculated by equation (9).




Statistical analysis
Data from kinetic models and sorption isotherms were compared by determination coefficients (R2) and by the corrected Akaike criterion (AICC). The computation of the kinetic study and the adsorption mechanism was adjusted to the kinetic models of pseudo-first order (PPO), pseudo-second order (PSO), and the adsorption tests were adjusted to the isothermal models of Freundlich, Langmuir, Temkin, Dubinin–Radushkevish using the statistical program OriginPro® 2016 (Origin Lab Corporation, USA). The maximum adsorption capacity data were subjected to analysis of variance (ANOVA) and the means were compared using the Tukey test (p ≤ 0.05). The analyzes were carried out using the Sisvar software, version 5.7 (Ferreira, 2019).
Results and discussion
Characterization of nanomaterials
The AL XRD low-angle region pattern in the range of 0.5°–10° 2θ (Figure 1a) showed reflections corresponding to the (1 0 0), (1 1 0), and (2 0 0) Miller indices, indicating the formation of an ordered hexagonal structure, according to the literature (Matsumoto et al., 1999). The XRD patterns of the materials at wide-angle are shown in Figure 1b; the BT showed high crystallinity and direct reflections at positions 7.62° and 22.28° 2θ, characteristics of the *BEA phase, according to the database of the International Zeolite Association—IZA (2021). The prominent peaks observed in the HT diffractogram occurred at values of 2θ (2θ = 11.6°, 23.2°, 34.8°), corresponding to the reflections of (0 0 3), (0 0 6), and (0 1 2). For reflections (0 1 5), (0 1 8), (1 1 0), and (1 1 3), the equivalent angles are 39.38°, 46.45°, 60.64°, and 61.64°, respectively. The HT showed characteristics of a single phase with high crystallinity and followed the literature (Chagas et al., 2012; Park et al., 2018). The peak with the best definition in the reflection (0 0 3), 11.6°, consisted of layers of SO4-2 anions since the lamellar double hydroxides (LDHs) present sulfates in the basal spacing of up to 11 Å, occurring due to the swelling of the intermediate layers by water (Bernard et al., 2022). In smaller peaks, the presence of aluminum causes narrow alterations in the crystalline lattice, usually this diffraction is seen between angles 30° to 50°, this fact can be clarified by the appearance of Al-O interactions (Sampieri et al., 2011).
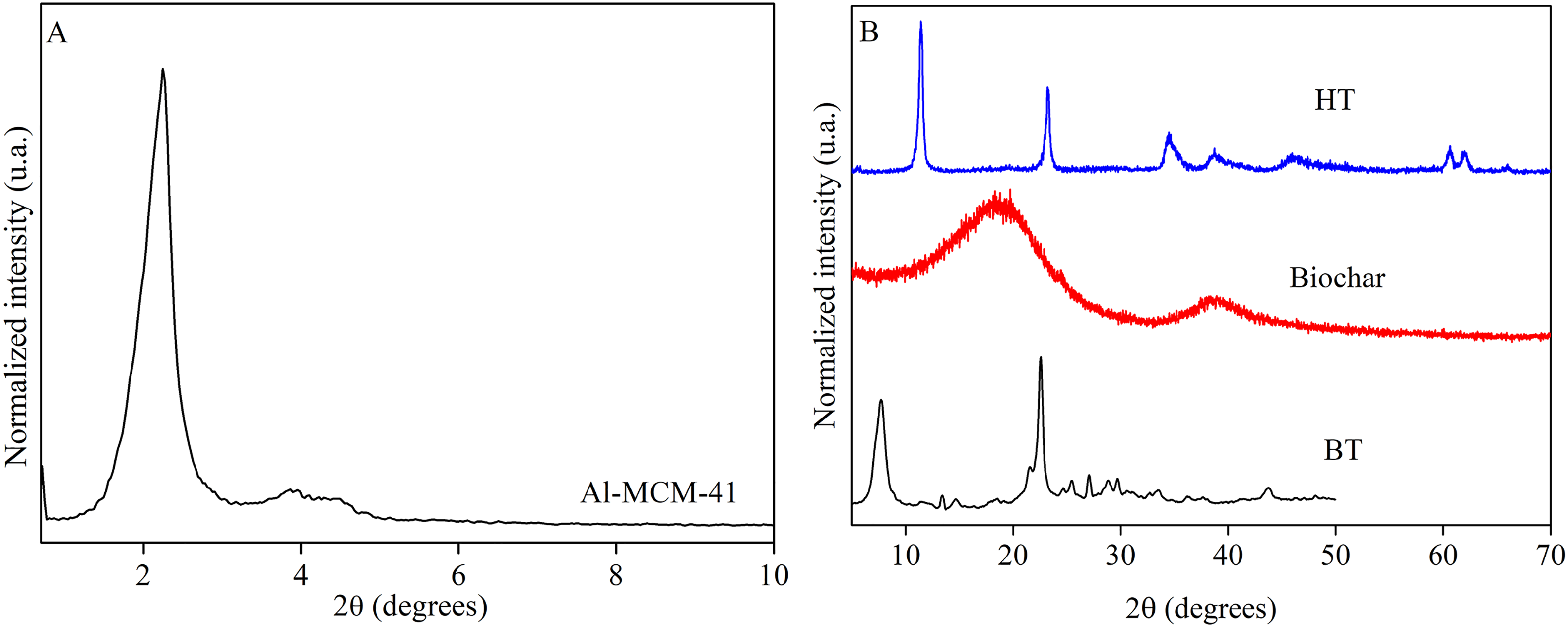
Diffractograms of nanostructured adsorbents: (a) Al-MCM-41 low-angle region pattern and (b) BT (beta zeolite), BIO (biochar), and HT (hydrotalcite) wide-angle region pattern.
In BIO, the reflection of approximately 20° 2θ was attributed to the crystallographic plane hkl (1 0 1) of the crystalline region of cellulose (Biswas et al., 2017) from the original biomass, not thoroughly degraded. The reflection at 40° 2θ indicates materials produced at high pyrolysis temperatures formed due to more stable graphite aromatic structures (Paris et al., 2005).
The analysis of the chemical properties of BT, AL, BIO, and HT adsorbents shows a Si/Al molar ratio of 24 and 22 for BT and AL adsorbents, respectively. The approximate values demonstrate the relative similarity of aluminum incorporation in the structural network of these adsorbents. The slight divergence between the theoretical and actual values for the Si/Al molar ratio can be explained by the partial hydrolysis of the silica used in the synthesis. The HT presented Mg/Al ratio of 0.97, lower than the theoretical value; possibly, there was a greater incorporation of Al + 3 ions in the centers of the octahedrons due to its more excellent solubility when compared to magnesium (Pedrotti, 2010). BIO presented 86.91% of carbon, 0.90% of hydrogen, 10.60% of oxygen, and 1.34% of nitrogen, amounts that make this nanomaterial with a polarity index of 0.13 and aromaticity of 0.01, indicating that this adsorbent has low polarity and high aromaticity, common in pyrolyzed nanomaterials at high temperatures (Fernandes et al., 2021).
The N2 adsorption and desorption isotherms at 77 K for all adsorbents (Figure 2). The N2 adsorption and desorption isotherms (Figure 2) of the nanostructured adsorbents exhibited different behaviors about their accessibility. It was observed that BT and BIO adsorbents showed a type I (a) isotherm profile, characteristic of microporous materials, according to IUPAC (Thommes et al., 2015), where most of the adsorbed volume occurs at low relative pressures (p/p0 < 0.2). The AL presents a type IV (b) isotherm profile, corresponding to adsorbents with the presence of mesopores, according to IUPAC (Thommes et al., 2015), due to the adsorbed volume between intermediate relative pressures (0.2 < p/p0 < 0.8) referring to capillary condensation. This material presents a hysteresis cycle related to the evaporation of the condensed gas in the desorption stage. IUPAC classifies this hysteresis as type H3.

N2 adsorption and desorption isotherms at 77 K of nanostructured adsorbents.
The HT showed a type III isotherm profile with interparticulate spaces. This result is related to the relatively weak adsorbent-adsorbate interactions and adsorbed molecules clustered around the most favorable locations on the surface of a porous solid (Dantas et al., 2015; Thommes et al., 2015). The BT and BIO adsorbents, although presenting a type I (a) isotherm profile and the unique presence of micropores, such nanostructured adsorbents reveal distinct porous structures in terms of accessibility. It is essential to highlight the characteristic of adsorbents with micropore structure (BT and BIO), mesoporous (AL), and interparticulate porosity (HT).
The Table 3 presents the values of textural properties obtained from the N2 adsorption and desorption isotherms at 77 K of all adsorbents. It is observed that BT and AL adsorbents have high specific area values (SBET) concerning BIO and HT adsorbents Table 3). This fact is related to the high micropore and external areas (mesoporous area) of the BT and AL materials, respectively. Although the outer area (SEXT) of BIO is more significant than that of BT, the micropore area (SµP) of BT is larger than BIO, thus raising its SBET.
Texture properties of the nanostructured adsorbents.

AL: Al-MCM-41; BIO: biochar; BT: beta zeolite; HT: hydrotalcite; SμP: micropore area; SBET: specific area; SEXT: outer area; VμP: micropore volume; VMESO: mesopore volume; VTOT: total volume.
The absence of SµP in AL is compensated by the high SEXT value, which corresponds to its mesoporous structure, contributing to the high SBET value. Note that BT, AL, and HT adsorbents have the highest total volume values (VTOT); however, HT is the material that has the lowest SBET, a characteristic resulting from the absence of mesopore and micropore. Thus, the SBET values are in the following ascending order: HT < BIO < BT < AL, while the VTOT values follow the order BIO ˂ BT < HT < AL. BT has a higher adsorbed volume than BIO, indicating that this material has greater accessibility. Compared to other materials, the AL has the highest adsorbed volume, a characteristic provided by the presence of ordered mesopores in its nanostructure.
Determining the PZC is crucial because it indicates whether the surface of an adsorbent material predominantly carries positive or negative charges. This information enhances our understanding of its interactions with contaminants. If the PZC value is lower than the pH of the medium, the surface will be predominantly negative. Conversely, if the PZC is higher than the pH of the medium, the surface will be predominantly positive.
In our study, the working solution pH was adjusted to 5.5. The PZC values (Figure 3) for BT (3.18) and AL (3.82) indicate that their surfaces are predominantly negative. In contrast, the BIO material, with a PZC of 7.5, has a predominantly positive surface. Notably, in BIO-type adsorbents produced at high pyrolysis temperatures, there is an increase in the zero charge potential (Banik et al., 2018).

Point of zero charge (PZC) of nanoadsorbents BT, AL and BIO. AL: Al-MCM-41; BIO: biochar; BT: beta zeolite.
Surface morphologies
The morphology of the investigated of BT, AL, and BIO adsorbents were studied using SEM, and the micrographs are shown in Figure 4. From the results shown, it can be concluded that the BT material revealed the distinctive morphology of the particles synthesized. The images obtained Figure 4a demonstrated a uniform distribution of predominantly cubic and spherical particles, with sizes varying between 500 and 700 nm. The presence of these morphologies can be attributed to the nucleation and growth processes during material synthesis. Particle shape and size are crucial not only for understanding the crystal structure of BT but also for its potential applications in catalysis and adsorption. These observations are consistent with previous studies, as reported by Cabral et al. (2003) and Lujanienė et al. (2024).

Scanning electron microscopy of (a) BT, (b)AL, and (c) BIO. AL: Al-MCM-41; BIO: biochar; BT: beta zeolite.
The biochar produced at 950 °C reveals a surface characterized by a significant presence of macropores (Figure 4c). These macropores emerge both during the pyrolysis process, as a result of the volatilization of organic compounds, and through the formation of cavities and voids corresponding to the cell lumen, with remnants of the internal walls still visible. The biochar particles range in size from 40 to 200 µm and do not form aggregates.
The SEM analysis Figure 4b of Al-MCM-41 reveals a structure with regions lacking uniform shape and a porous surface. The particles range in size from 250 nm to 1 µm. SEM images display a pore distribution characteristic of MCM-41 materials, enhanced by the addition of aluminum, which contributes to structural stability. TEM analysis of the BT (Figure 5) material reveals predominantly spherical particles averaging approximately 200 nm in size, exhibiting a uniform particle size distribution. The particles exhibit a characteristic porous structure with homogeneously distributed pores. Electron diffraction patterns confirm the material's high crystallinity, highlighting distinct crystalline regions. In contrast, TEM imaging (Figure 5) of the AL adsorbent shows spherical particles averaging 20 nm in size, characterized by a mesoporous structure displaying typical hexagonal pore organization. High-resolution and phase-contrast images demonstrate homogeneous distribution of aluminum within the silica matrix. Unfortunately, no data could be obtained for the BIO material due to sample degradation at elevated temperatures.

Transmission electron microscopy of (a) BT and (b) AL. AL: Al-MCM-41; BT: beta zeolite.
Maximum adsorption capacity
In the preliminary test, it was found that the BT, AL, and BIO nanomaterials were able to adsorb diuron, hexazinone, and sulfometuron-methyl in an aqueous medium; however, HT did not show the same performance. According to Mishra et al. (2018), for HT to have the adsorptive capacity, it would be necessary for the layers of double hydroxides to be in the proportions of divalent/trivalent cations between 2:1 and 4:1, smaller or larger combinations, the layers are not formed properly. Despite using the preparation, the Mg/Al ratio of 2:1; in the final product, the ratio was 0.97; thus, there was no formation of hydroxide double layers. As the tested herbicides do not have adsorption capacity, the HT adsorbent was not used in the other tests.
The maximum adsorption capacity test was carried out to determine the minimum mass of nanostructured materials capable of adsorbing the maximum amount of diuron, hexazinone, and sulfometuron-methyl in water (Figure 6). For BT (Figure 6a–c, the amount of 20 mg was selected; in the case of AL (Figure 6d–f), the minimum mass would be 17 mg; however, as the plateau was only reached from the two last AL mass values, it was chosen to work with 20 mg. For BIO (Figure 6g–i) 30 mg was selected, and the masses of the chosen nanomaterials have the same adsorption efficiency as other larger ones.

The maximum adsorption capacity of diuron, hexazinone, and sulfometuron-methyl at different concentrations of BT ((a)–(c)), AL ((d)–(f)), and BIO ((g)–(I)) adsorbents. All comparisons were performed using Tukey's test (p ≤ 0.05). AL: Al-MCM-41; BIO: biochar; BT: beta zeolite.
The nanostructured materials showed different adsorptive capacities for the herbicides in this study (Figure 6). The BT adsorbed 99.35% (270.12 μg.g−1) of diuron, 97.26% (101.51 μg.g−1) of hexazinone, and 90.96% (2.61 μg.g−1) of sulfometuron-methyl. The AL showed the following adsorption rates 88.33% (12.26 μg.g−1), 83.75% (4.36 μg.g−1), and 95.73% (0.19 μg.g−1), respectively, for diuron, hexazinone, and sulfometuron-methyl. The BIO removed 96.36% (78.18 μg.g−1) of diuron, 14.22% (2.76 μg.g−1) of hexazinone, and 34.92% (9.83 μg.g−1) of sulfometuron-methyl.
The results demonstrate that BT is the most promising adsorbent for decontaminating diuron, hexazinone, and sulfometuron-methyl present in water, among the others tested. The fast and efficient adsorption of BT is due to the large surface area that increases the availability of adsorption sites; another essential feature is the high hydrophobicity that favors the retention of nonpolar molecules (Rad and Anbia, 2021). The structure of this adsorbent presents a high empty area of pores, highly ordered and parts resembling cages or channels (Janićijević et al., 2020), a characteristic that enables adsorption by intraparticle diffusion. BT allows ion exchange through unique network stability, trapping water molecules, cations, and organic molecules by electrostatic attraction (Andrunik and Bajda, 2021).
Kinetic study
The adsorption kinetics of diuron, hexazinone, and sulfometuron-methyl for each adsorbent is shown in Figure 7. The results show rapid adsorption of herbicides by the BT, AL, and BIO nanomaterials, with equilibrium reached after 1 hour of initial contact. At equilibrium, a high decontamination rate is achieved, removing diuron, hexazinone, and sulfometuron-methyl, respectively, 143.48, 56.74, and 0.75 µg g−1 by BT; 6.16, 2.18, and 0, 10 µg g−1 by AL; and 36.99, 2.80, and 6.10 µg g−1 by BIO. The rapid adsorption of herbicides by adsorbents in the initial moments is attributed to the abundance of available adsorption sites and the high affinity between adsorbents and adsorbates (Salomón et al., 2022).

Kinetic study of nanostructured adsorbents, BT ((a)–(c)), AL ((d)–(f)), and BIO ((g)–(i)) for diuron, hexazinone, and sulfometuron-methyl herbicides. AL: Al-MCM-41; BIO: biochar; BT: beta zeolite.
The experimental data were adjusted to the PPO and PSO kinetic models (Figure 7), and the values of the determination coefficients (R2) ranged from 0.84 to 0.97 for the PPO and from 0.89 to 0.97 for the PSO (Table 4); these results show that the two kinetic models fit the experimental data well. However, in order to verify which kinetic model best describes the adsorbent–adsorbate interactions closer to whole reality, the Corrected Akaike Informative Criterion (AICC) was adopted, which is based on the information and maximum likelihood theories (El-Khaiary and Malash, 2011). The lowest values found for AICC (Table 4) confirm that PSO can better describe the interactions between herbicide molecules and nanomaterials. Gikas found contrary results; Gikas et al. (2022) studied the adsorption of fluometuron in zeolites and verified that the PPO was the model that best fits the experimental results.
Parameters of adsorption isotherms.
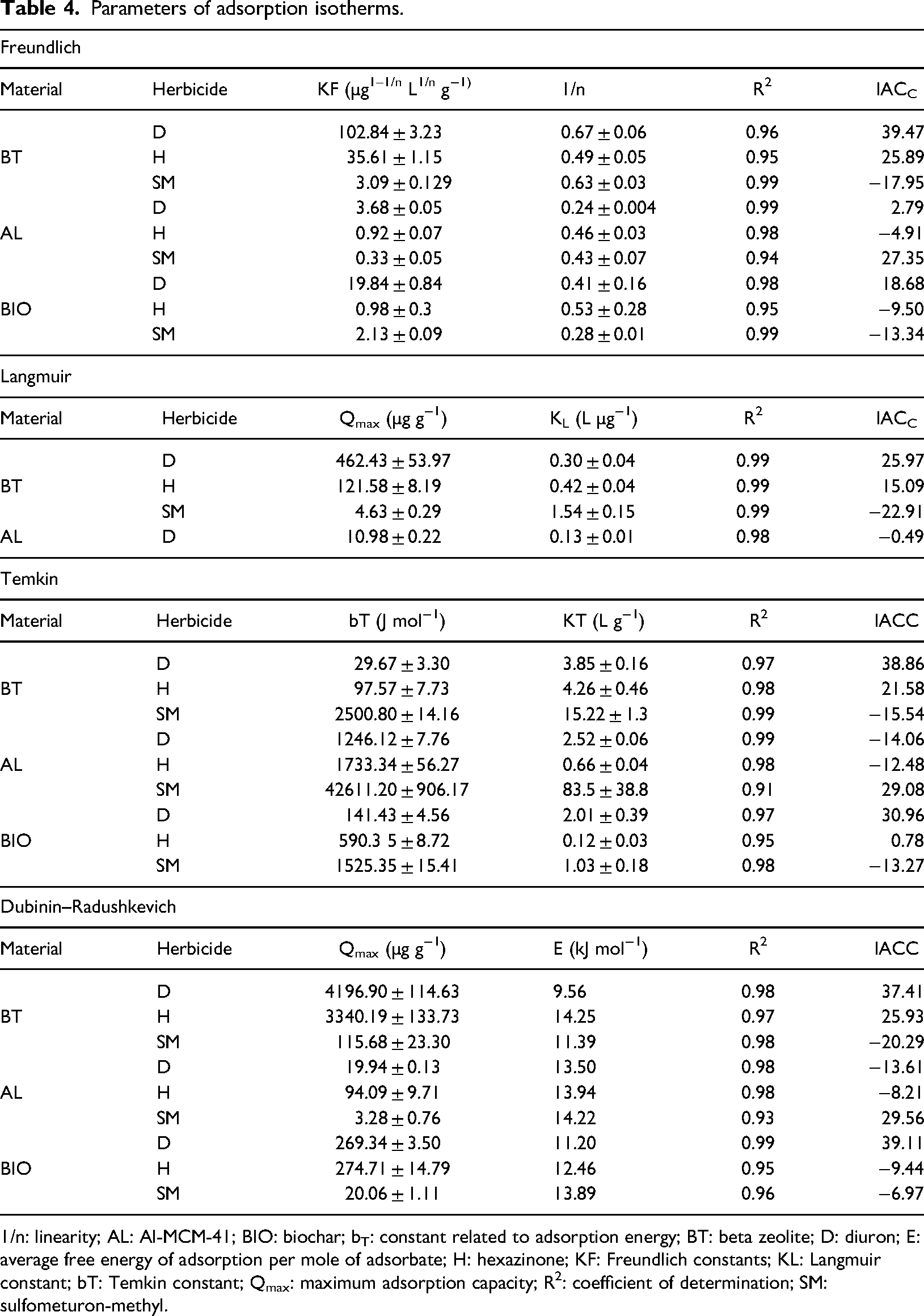
1/n: linearity; AL: Al-MCM-41; BIO: biochar; bT: constant related to adsorption energy; BT: beta zeolite; D: diuron; E: average free energy of adsorption per mole of adsorbate; H: hexazinone; KF: Freundlich constants; KL: Langmuir constant; bT: Temkin constant; Qmax: maximum adsorption capacity; R2: coefficient of determination; SM: sulfometuron-methyl.
The values of qe (quantity adsorbed at equilibrium) calculated by the PSO (Table 5) estimate that BT is the most effective adsorbent in extracting diuron and hexazinone present in water bodies. At the same time, BIO is more efficient for removing sulfometuron-methyl. The high cation exchange capacity of BT favors the adsorption of diuron and hexazinone by electrostatic attraction, which under working conditions (pH = 5.5), have most of its molecules positively correlated. Hydrogen bonds are also meaningful interactions that may favor the adsorption of these herbicides by BT. Another essential characteristic of this adsorbent is that it acts as a Lewis acid, being able to receive pairs of electrons provided by the oxygen and nitrogen atoms present in the structure of these herbicides (Nucleophile) and make covalent bonds from the sharing of electrons. The most efficient removal of sulfometuron-methyl by BIO is due to the greater possibility of Van der Waals bonds and π–π interaction due to the high aromaticity of this adsorbent (Xiao and Pignatello, 2015a; Sarker et al., 2017).
Parameters of kinetic models for the adsorption of diuron, hexazinone, and sulfometuron-methyl herbicides on BT, AL, and BIO adsorbents.

AL: Al-MCM-41; BIO: biochar; BT: beta zeolite; D: diuron; E: average free energy of adsorption per mole of adsorbate; H: hexazinone; K1 and K2: the velocity constants for the pseudo-first-order and pseudo-second-order kinetic models, respectively; qe: amount of herbicide adsorbed at equilibrium; R2: coefficient of determination; SM: sulfometuron-methyl.
The rate constant (K2) estimated by PSO (Table 5) predicts a lower adsorption rate of hexazinone in BT and AL adsorbents, a phenomenon attributed to the more excellent solubility of this herbicide in aqueous media (Table 1), BIO is verified that the sulfometuron-methyl has the lowest K2 value (8.03 g mg−1 min−1), this lower adsorption rate is due to the more incredible difficulty for sulfometuron-methyl molecules to approach the adsorbent, because of its greater polar surface area (136 Å2) which encounters more excellent resistance to adsorption on the hydrophobic surface of BIO.
Adsorption balance
The adsorption isotherms describe the phenomenon of sorption under certain experimental conditions, being able to explain the interactions between adsorbent and adsorbate close to actual conditions. The Freundlich, Langmuir, Temkin, and Dubinin–Raduchkevich isothermal models were fitted to the experimental data to verify which isotherm best describes the adsorption equilibrium (Figure 8). The R2 values (Table 4) indicate that the kinetic models used present suitable adjustments to the experimental results, and the AICC was applied to select the isotherm capable of better explaining the phenomena that occurred during the adsorption. The Langmuir and Freundlich model better explains the interactions between herbicides, BT, and BIO nanomaterials. For AL, the Temkin isotherm can better describe the diuron and hexazinone adsorption; sulfometuron-methyl was the Freundlich isotherm.

Adsorption equilibrium plots of nanostructured adsorbents BT, AL, and BIO for diuron, hexazinone, and sulfometuron-methyl herbicides. AL: Al-MCM-41; BIO: biochar; BT: beta zeolite.
The 1/n values (linearity constant) of the Freundlich isotherm were less than 1 (Table 4), indicating an L-type isotherm. In this type of isotherm, at low concentrations of the adsorbent, the adsorption happens more quickly; as the concentration increases, it is verified that the increment in the adsorption decreases (Figure 8), indicating occupation of the binding sites (Giles et al., 1974). A similar behavior was observed by Fernandes et al. (2021) when evaluating the adsorption of hexazinone in eucalyptus wood biochar produced at different pyrolysis temperatures.
The values of the Freundlich constant (KF) and the maximum adsorption capacity (Qmax) (Table 4) show that BT is the adsorbent with the highest adsorptive capacity, followed by BIO and finally AL. BT characteristics such as textural ones, mainly VµP (0.21 cm3 g−1) and SBET (458 m2 g−1), together with the high Si/Al molar ratio, favor adsorption due to the high hydrophobicity and the greater availability of adsorption sites (Yonli et al., 2012; Jiang et al., 2020a). In this way, the synergistic effect between the hydrophobic surface and the high micropore area enhances the adsorptive capacity of BT about BIO and AL.
The size of the adsorbate molecule can also act as a limiting factor to access to the microporous structure of the adsorbent; the smaller the surface area of the adsorbate, the lower the steric hindrance, and consequently, the greater access to the adsorption sites (Moliner et al., 2015; Dai et al., 2020). The narrated fact is observed in the experiment, diuron having a smaller polar surface area (32.3 Å2), presented a Qmax estimated by the Langmuir isotherm about 3.80 and 99.87 times greater for hexazinone (56.2 Å2) and sulfometuron-methyl (136 Å2) (Table 1), respectively. Two other characteristics that possibly influenced the more significant adsorption of diuron in BT were kow and pKa. According to Table 1, the diuron molecule presents a greater cationic possibility due to its high pKa value, and high affinity for the nonpolar phase concerning hexazinone and sulfometuron-methyl.
The hydrophobicity of zeolites with a high Si/Al ratio is one of the main characteristics that favor the adsorption of organic contaminants present in water bodies (Jiang et al., 2020b), with Van der Walls forces being one of the main interactions responsible for the adsorption (Blasioli et al., 2014). A study by Koubaissy et al. (2008) observed that the higher the kow, the greater the adsorption of the contaminant in zeolites with a high Si/Al molar ratio. As can be seen in Table 1, the kow values 2.68, 1.85, and 1.2 for diuron, hexazinone, and sulfometuron-methyl herbicides, respectively, show a direct correlation with the kF and Qmax values. Behavior similar to the experiment by Fukahori et al. (2011), among the analyzed sulfa drugs (sulfathiazole, sulfamerazine, sulfamethizole, sulfadimidine, and sulfamethoxazole), that is, those with higher kow showed more significant adsorption on zeolites with high Si/Al molar ratio.
Zeolites with high molar Si/Al have a high ion exchange capacity (Andrunik and Bajda, 2021). Studies have shown more significant adsorption of cationic contaminants compared to anionic compounds due to the repulsion of the net negative charge present in aluminum (Koubaissy et al., 2008; Fukahori et al., 2011). As the pH of the working solution was adjusted to 5.5 and the PZC of BT was 3.18, according to Das Chagas et al. (2020), diuron molecules have a positive charge
The values of Temkin's constant related to the adsorption energy (bT) estimated for BT reaffirm the more remarkable ability to adsorb diuron since the bonds involved in the adsorption of hexazinone and sulfometuron-methyl, respectively, involve about 3.29, and 84.29 times more energy than interactions with diuron. According to Monjezi and Javanbakht (2022), lower the value of this variable, greater the affinity between the herbicide and the adsorbent.
According to AICC, the Freundlich isotherm best explains the adsorption of herbicides by BIO (Table 4). kF values assume diuron (19.84 µg1–1/nL1/ng−1) as being more adsorbed by BIO when compared to hexazinone (0.98 µg1–1/nL1/ng−1) and sulfometuron-methyl (2.13 µg1–1/nL1/ng−1). For Hassan et al. (2020), the main mechanisms of adsorption of organic contaminants in biochar produced at high temperatures (>500°C) are hydrophobic interactions, π–π bonding and pore filling, due to the high aromaticity and low polarity. As observed for BT, the more significant adsorption of diuron by BIO may also be related to the higher kow value and low solubility. According to Tong et al. (2019), evaluating the adsorption of micropollutants in biochar produced at high temperatures, observed a positive correlation between adsorption and log kow, the neutral and more hydrophobic contaminant, presenting the highest adsorption capacity, with the hydrophobic interaction being the dominant mechanism.
Due to aromaticity, the BIO adsorbent surface is rich in π electrons, which can interact with electron-poor molecules to form π–π electron donor and acceptor complexes (Xiao and Pignatello, 2015a). Sulfometuron-methyl contains in its structure deactivating groups, which make the aromatic ring poor in electrons, starting to act as receptors in π–π complexes; this characteristic boosts the adsorption of sulfometuron-methyl by BIO (Lattao et al., 2014). It was demonstrated by Nguyen et al. (2007) that this type of interaction occurs firstly on external surfaces and then by a pore-filling mechanism. Xiao and Pignatello, (2015b) observed that the π–π bond is essential to the adsorption of triazine herbicides and other nitroaromatic compounds in BIO.
Another factor that may have influenced its adsorption efficiency was its PZC of 7.5. When the pH of the medium is lower than the PCZ, protonation of the functional groups occurs, forming positive charges on the functional groups on the BIO surface, thus favoring the adsorption of anionic herbicides, such as sulfometuron-methyl (Hassan et al., 2020). In addition, such adsorbent has a more significant amount of O and N in its structure, which can form hydrogen bonds with H present on the surface of the BIO (Sun et al., 2012).
Although the BIO adsorbent has a low polarity, being composed chiefly of carbon, it still has 10.6% oxygen and 1.34% nitrogen; atoms that enable the formation of polar regions in the BIO structure (Fernandes et al., 2021). Sulfometuron-methyl showed a higher kF value than hexazinone; its larger polar surface area makes its molecules more attracted to BIO. The high solubility of hexazinone (33000 mg L−1) reduces adsorption by BIO, as it has excellent affinity for the aqueous phase.
Although the hydrophobicity of BIO was probably one of the essential factors for adsorption, this may be responsible for the lower adsorption of herbicides when compared to BT adsorbent. According to Gray et al. (2014), hydrophobicity can sometimes prevent water entry into the porous structure of biochar. Thus, reducing the access of herbicide molecules dissolved in the aqueous CaCl2 solution to the biochar binding sites.
The kF and Qmax values estimate that the adsorption order in AL is diuron > hexazinone > sulfometuron-methyl. Adding aluminum (Al) to the MCM-41 adsorbent promotes the formation of a negative charge, requiring a cation to stabilize the charges (Andrunik and Bajda, 2021). This balancing cation is from the absorbent structure and can be exchanged for positively charged herbicides. Under working conditions (pH = 5.5), most diuron and hexazinone molecules will be positively charged, favoring the adsorption of these two herbicides by electrostatic attraction, as the AL surface is negatively charged in solutions with a pH greater than 3 (Zhou et al., 2015). A high density of silanol groups (Si-OH) on the surface may have been the reason for the lower adsorption efficiency of herbicides compared to the other adsorbents in this study. The preferential adsorption of water molecules by Si-OH groups limits access to organic contaminants (Trouvé et al., 2012).
The Temkin isotherm predicts that the adsorption of sulfometuron-methyl in AL requires 34.19 and 24.58 times more energy than the adsorption of diuron and hexazinone, respectively, a fact that makes the adsorption of sulfometuron-methyl less favorable and energetically more unstable. The average free energy of adsorption (E), a parameter estimated by the Dubinin–Radushkevich model, is used to predict whether adsorption occurs through chemical or physical interactions (AKÇAY et al., 2009). For all evaluated nanostructured adsorbents, there was a predominance of chemical adsorption (E > 8 kJ mol−1) in all tested herbicides.
Conclusions
This study demonstrates the remarkable efficacy of nanostructured adsorbents beta zeolite (BT), Al-MCM-41 (AL), and biochar (BIO) in removing diuron, hexazinone, and sulfometuron-methyl from aqueous solutions. Among these, BT exhibited the highest adsorptive capacity, while AL and BIO also showed significant potential. The scalability of these findings suggests that these nanomaterials can be effectively used in large-scale water treatment and soil remediation. Future research should focus on practical applications, such as filters or biofilms, and explore the recyclability of these materials to enhance their sustainability and efficiency in mitigating herbicide contamination.
Footnotes
Acknowledgment
All authors are very grateful for the collaboration of Laboratory of Catalysis, Environment and Materials (LACAM/UERN/BRAZIL), Laboratory of Magnetic and Optical Analysis (LAMOP/UERN/BRAZIL), Centro de Pesquisa em Ciências Vegetais do Semi-Árido (CPVSA/UFERSA/BRAZIL). This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brazil (CAPES) – Finance Code 001.
Authorship
FDdS contributed to conceptualization, formal analysis, investigation, writing—original draft, and writing—review and editing. DVS was involved in resources, writing—original draft, writing—review and editing, and supervision. PSFdC was involved in formal analysis, methodology, resources, investigation, writing—original draft, and writing—review and editing. BCCF was involved in methodology, formal analysis, investigation, writing—original draft, and writing—review and editing. LLLdSR, CCdS, and MCRH were involved in writing—original draft, and writing—review and editing. TMdST was involved in methodology, formal analysis, and investigation. RBdL was involved in formal analysis, methodology, investigation, writing—original draft, and writing—review and editing. VPSC was involved in conceptualization, methodology, resources, formal analysis, investigation, writing—original draft, and writing—review and editing.
Data availability statement
Data is contained within the article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
