Abstract
Textile industries use dyes that play a significant role in polluting water bodies. The current work examines the acid-modified palmyra palm male inflorescence activated carbon as a novel, environmentally feasible, and economical adsorbent to eliminate methylene blue in synthetic effluent. The adsorbent is chemically activated by nitric acid to enhance pores and active sites. The palmyra palm male inflorescence activated carbon adsorbent is characterized by some characterization techniques, including Fourier transform infrared spectroscopy, BET, and scanning electron microscope with energy dispersive X-ray, for analyzing its physio-chemical properties and nature. The prepared adsorbent has a large surface area of 343.781 m2/g. Batch adsorption tests were conducted to evaluate the efficacy of the palmyra palm male inflorescence activated carbon. The adsorbent capacity and the highest removal efficiency of the adsorbent are 38.82 mg/g and 98.5%, respectively. It is obtained by maintaining the batch study parameters at their optimum conditions. For this process, 5 g/L will be the suitable dosage for effectively eliminating methylene blue dye. The appropriate dye concentration and contact time for the maximum removal were determined as 100 mg/L and 50 min. Reusability evaluation of the adsorbent was done by utilizing 0.1 Mole of NaOH, and the adsorbent was successfully regenerated and used for up to four cycles.
Keywords
Introduction
Environmental pollution has always been a significant issue that has hampered the industrialization process and human health. For instance, many contaminants are released into the environment without being treated. These contaminants include heavy metal ions, antibiotics, disruptive compounds, and organic dyes (Tan et al., 2015). One contaminant that is simple to spot because it is unwelcome and very apparent in water is artificial color (or dye) (Aboua et al., 2015). For thousands of years, dyes have been widely utilized in fabric, paint, pigmentation, and several other uses. Approximately more than lakh varieties of dyes are currently available today. To meet industrial consumer demand, roughly ten lakh tonnes of colorants are generated yearly, and one-third of this amount is wasted as wastewater (Maneerung et al., 2016).
Most dyes are designed to resist environmental elements, including light, heat, and oxidizing chemicals. The presence of dyes in aquatic environments lowers light and oxygen penetration, affecting regular photosynthetic activity and deteriorating marine biota. Additionally, these harmful substances have an impact on human health, causing several diseases like cancer, jaundice, tumors, skin irritation, allergies, and heart problems. Moreover, even at low doses, their presence is in the water system (Hasanzadeh et al., 2020; Liu et al., 2019; Regti et al., 2017). Dye and dye-related items are harmful and can cause cancer, mutagenesis, and allergies. They might also hinder aquatic life's ability to photosynthesize. Given that complex aromatic textile dyes are highly stable compounds that withstand deteriorating environments, even a tiny proportion of their presence in wastewater raises serious problems (Li et al., 2018).
Additionally, dye wastewaters frequently have high salt contents and low biodegradation capability, complicating their removal by traditional effluent treatment techniques (Pérez-Calderón et al., 2018). The Advanced Oxidation Process (Gayathri et al., 2019), ion exchange (Joseph et al., 2020), photocatalytic degradation (Kheirabadi et al., 2019), membrane separation (Yang et al., 2020), electrochemical degradation (Samarghandi et al., 2020), and adsorption are a few of the methods that can be employed to handle dye wastewater (Muniyandi and Govindaraj, 2021). Methods like photocatalytic degradation, electrochemical degradation, and the advanced oxidation process were effectively utilized for degrading dyes, but their implementation was limited due to their being more expensive and technically challenging. Conversely, there may be restrictions on the volume and regeneration effectiveness of the ion exchange process. Compared with these methods, adsorption has become a more prominent treatment technique for treating aqueous environments because of its ease of application, simple layout, adaptability, and low energy requirement (Heidarinejad et al., 2018). One of the most influential and adaptable options for removing organic and inorganic contaminants from wastewater is adsorption on activated carbon (AC). An essential feature of AC is its greater surface area, high pore volume, more adsorption ability, physio-chemical nature, and high regeneration efficiency (de Souza et al., 2018). For the creation of inexpensive and environmentally beneficial AC, organic wastes like sunflower pith (Baysal et al., 2018), bamboo (Ademiluyi et al., 2009), coconut (Zhang et al., 2018), Tamarind seed (Ishak and Kumar, 2022), wall nutshell (Li et al., 2020), paper waste (Novais et al., 2018), and coffee husk (Ahmad and Rahman, 2011) are some of the raw materials employed. Borassus flabellifer, the scientific name for the palmyra tree, is a plant that may be found in several southeast Asian nations and semi-arid areas of India. Male flowers often have tiny sizes and are grouped inside an inflorescence. The sap is the main component used to produce jaggery from all the many parts used in product development. After sap removal, the palmyra palm male inflorescence (PPMI) is discarded as waste into the environment without considering its significance. Palmyra plam male inflorescence activated carbon (PPMIAC) has several beneficial compounds and functional groups that could be used to remediate effluents. Therefore, one can begin the process of producing AC using the male inflorescence of the Palmyra palm tree. Having AC from these natural sources will be the most economical, and it can then be applied to the ever-present issue of water pollution (Hevira et al., 2021; Jerry, 2018).
The primary goal of this research work is to use the adsorption method to eliminate the surplus methylene blue (MB) dye from the aqueous solution. For this adsorption process, Nitric acid-activated PPMI was used as an adsorbent, termed PPMIAC. PPMI waste biomass was collected from the forests of Srivilliputhur in Tamil Nadu, India, Virudhunagar district. This adsorbent has substantial specific surface areas that effectively eliminate the basic dye (MB) from the wastewater. This specific adsorbent was chosen due to its cost-effectiveness, eco-friendliness, abundant availability, biodegradability, bioactive compounds, and sustainability compared to conventional ACs and other biomass sources. The primitive novelty of this proposed work is to utilize the waste materials into a value-added product to eradicate water pollutants for the benefit of environmental conservation. This work discusses in detail the adsorption ability of the PPMIAC to eliminate MB dye from the aqueous solution. Where batch mode adsorption studies are carried out to optimize the adsorption parameters. Various characterization techniques, including Fourier transform infrared spectroscopy (FTIR), scanning electron microscope (SEM), EDS, and BET analysis characterized the nature adsorbent. Adsorption kinetics, Isotherm, and thermodynamic studies were also carried out. The regeneration efficiency of the adsorbent was also discussed in detail. According to our knowledge and from the literature review, it is the first attempt to use PPMI to effectively remove MB dye by Nitric Acid (HNO3) activation.
Materials and methods
Materials
AC from PPMI is the adsorbent used in this study. It is acquired from the forests of Srivilliputhur in Tamil Nadu, India, Virudhunagar district. Annular-grade MB dye was utilized to produce synthetic textile effluents. Analar grade of HNO3 was purchased from the RFCL industry in New Delhi, India. The double distilled (DD) water of grade II is used for washing purposes.
Preparation of the adsorbent and adsorbate
The collected PPMI solid material is cleaned by DD water again and again to eliminate dissolved impurities and dried using an oven for two hours at 120 °C.
For chemical activation, 4 g of PPMI solid material is blended with 15 mL of 1 M of HNO3 and placed inside the muffle furnace for 12 h (Li et al., 2019). After the stipulated time interval, the sample was cleaned with distilled water several times to return the adsorbent to pH 7. The final powder was called PPMI and screened into 85 mm. Then, the dried powder (PPMIAC) was sealed in a Hermetic container and utilized for future use. For the experimental work, 1000 mg/L−1 of stock solution was prepared, and the desired MB dye concentrations could then be synthesized from the stock solution via consecutive dilutions. This outcome is termed an adsorbate.
Characterization of the adsorbent
The PPMIAC adsorbent's morphological features were analyzed using an EVO18 SEM (Carl Zeiss). The acceleration voltage was 20 kV, and the magnification range was adjusted to 2500 X and 5000 X. The composition of the adsorbents and the concentration of the accessible elements was investigated using the Quantax 200 and X Flash 6130 energy dispersive X-ray (EDX) analyzers. On an IR Tracer 100, the functional groups of PPMIAC were displayed using FTIR, and a JASCO V-630 evaluated the analytical measurement of dye. The surface area and porosity of the material were analyzed by using Quantachrome TouchWin v1.2 4 instrument.
Adsorption experiments
Dampness in the PPMIAC is removed using the forced air circulating oven by running it at 100 °C for two hours before adding it to the adsorbate. After adding an appropriate amount of PPMIAC to a titration flask containing MB dye mixture, the mixture was shaken at 140 r/min for optimal interaction to achieve the needed adsorption. The solid adsorbent was then removed from the adsorbate using No. 42 filtrate. Using a UV Spectrophotometer, the final concentration of the adsorbate can be evaluated. Studies on batch adsorption were undertaken by altering the adsorption-influencing factors, including pH (1–9), contact time (10–60 min), dose rate (1–6 g/L), and baseline dye concentration (50–250 mg/L). Adding 0.1 M of NaOH and HCl can alter the solution pH. The sorption experiment was triplicated to ensure consistent results. The entire sorption experiment was carried out to guarantee reliable findings. Co and Ce are the measurements of the dye concentration before and following adsorption. The equation below was used to calculate the process's adsorption efficiency: The equation, in which the components Co and Ce denote the initial and final dye concentration of MB dye (mg/L), was utilized to evaluate the maximum adsorption capacity. Here, qe represents the equilibrium adsorption capacity of the PPMIAC (mg/g), while V (L) and m (g) denote the MB dye solution's volume and the mass of the adsorbent.
Results and discussions
Scanning electron microscope evaluation
SEM analysis is an effective tool for analyzing the surface characteristics of the PPMIAC. The presence of pores and cavities on the surfaces of the PPMIAC will determine how well MB dye is absorbed through adsorption. As can be observed in Figure 1, chemical activation with Nitric acid resulted in the production of many active sites on PPMIAC before adsorption (Demiral et al., 2021). The structure of the pores and cavities was flaky and irregular. Post-adsorption, the MB dyes were captured in the PPMI pores, as shown in Figure 2, demonstrating the effectiveness of the dye removal. The elimination of MB dye from sewage using surface-modified neem leaves followed the same pattern where HCl acid significantly enhances the surface area and pore solidity (Mathivanan et al., 2021).

PPMIAC before the accumulation of MB dye. PPMIAC: palmyra palm male inflorescence activated carbon; MB: methylene blue.

PPMIAC after the accumulation of MB dye. PPMIAC: palmyra palm male inflorescence activated carbon; MB: methylene blue.
FTIR analysis
The FTIR analysis of PPMIAC for the elimination of MB is shown in Figures 3 and 4. The distinct chemical interactions in the sample are examined using FTIR. In the current study, ACs produced from PPMI using acid activation were employed to test its applicability. Figures 4 and 5 show the before and after adsorption of the PPMIAC sample for FTIR analysis. Peaks at 609.51, 1514.12, and 1622.13 occurred before and after the adsorption process. A broad peak at 609.51 indicates the existence of C-Br, a halo compound (Trivedi et al., 2015). The peak at 1514.12 suggests the presence of strong N-O that is asymmetrical nitro alkanes (Nitro compound) (Ak et al., 2008). A broad peak at 1622.13 indicates the existence of C-C (aromatic carbon). Sharp peaks shift at 1080–1035 and 1190–1371, suggesting the existence of C-O (Denis et al., 2016) and S

FTIR spectra of PPMIAC (before adsorption). FTIR: Fourier transform infrared spectroscopy; PPMIAC: palmyra palm male inflorescence activated carbon.

FTIR spectra of PPMIAC (After adsorption). FTIR: Fourier transform infrared spectroscopy; PPMIAC: palmyra palm male inflorescence activated carbon.

EDX analysis of PPMIAC before adsorption. EDX: energy dispersive X-ray; PPMIAC: palmyra palm male inflorescence activated carbon.
EDX analysis
EDX ranges of the PPMIAC are shown in Figures 5 and 6. The figure lists the components of the synthesized PPMIAC adsorbent, which includes C, O, Al, Si, Mo, and S. Similar elements except for K and Ca were also visible in the dyes’ post-adsorption and may be present in the water that was used for the batch adsorption process. EDX was used to confirm the presence of Si, which impurities like sand particles could cause. The presence of Aluminum indicates that the sample was well-coated during inspection. The PPMIAC sample's mass percentage determines the dye removal's effectiveness. The increased mass percentage in the after-adsorption results shows the effectiveness of the adsorption process. Similar to this, the spectrum graphs were displayed following adsorption.
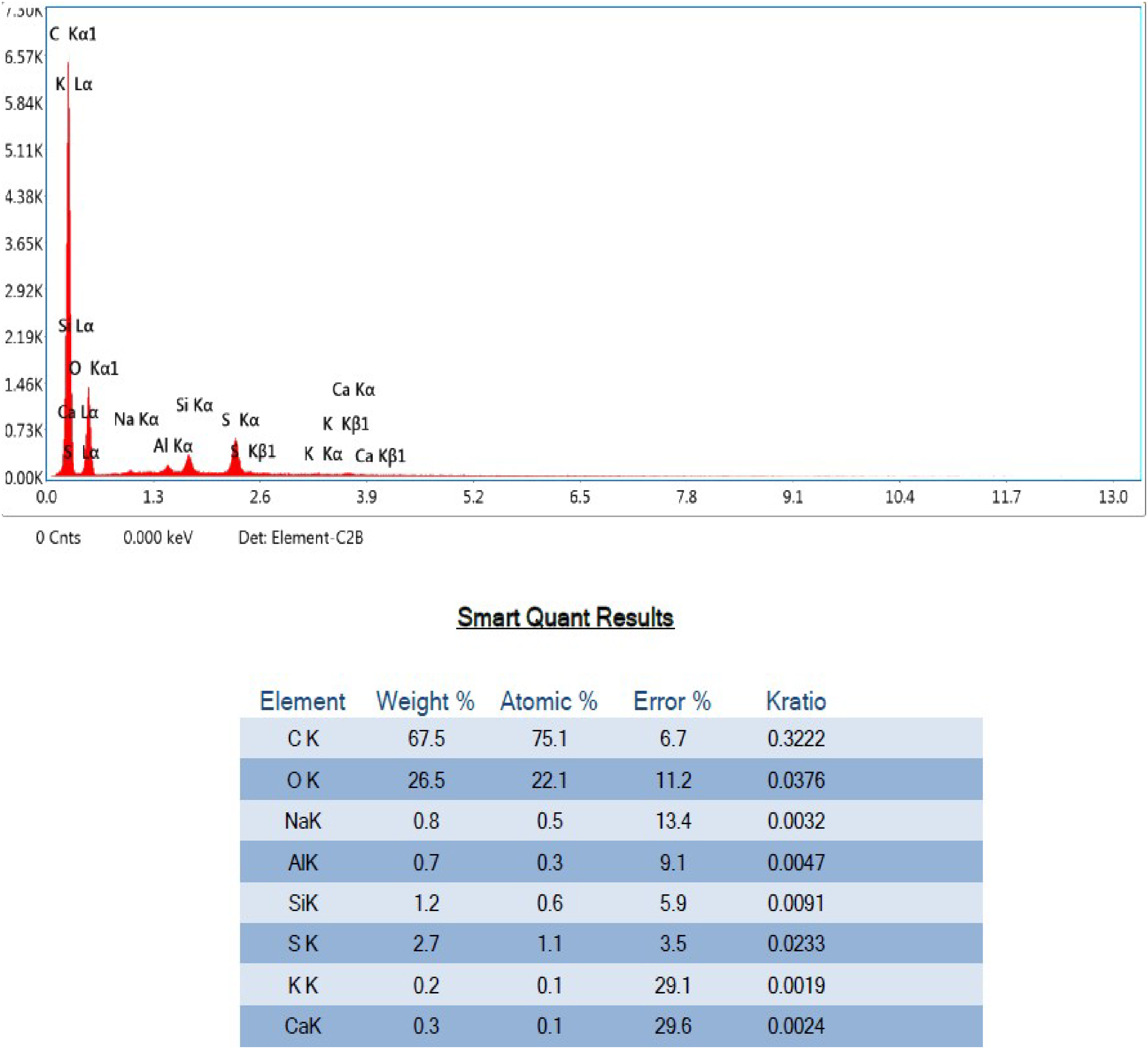
EDX analysis of PPMIAC after adsorption. EDX: energy dispersive X-ray; PPMIAC: palmyra palm male inflorescence activated carbon.
BET analysis
The surface area, pore volume, and pore width of the PPMIAC adsorbent can be evaluated using the Breuner-Emmer Teller analysis. For this analysis, the sample was first degassed at 200 °C for 4 hours to eliminate all adsorbed impurities. High-purity nitrogen gas was added to the sample at various pressures and temperatures of 77.35 k, and the equilibrium adsorption that occurred at those pressures was recorded. After that, the desorption investigation was completed by lowering the pressure used to remove the nitrogen gas that had been adsorbed. The pore volume and pore diameter of the recommended adsorbent were determined using the volume of nitrogen gas adsorbed and desorbed at different pressures. The surface parameters are listed in Table 1 and the adsorption and desorption graph is shown in Figure 7.

Nitrogen adsorption/desorption isotherm for palmyra palm male inflorescence activated carbon (PPMIAC).
BET analysis parameters.

Impact of PPMIAC dose rate
The result of PPMIAC on the elimination of MB dye is studied by altering the dosage of PPMIAC from 1 to 6 g/L and by retaining the concentration, shaking time, temperature, and pH (100 mg/L, 60 min, 303 K, and 8 pH) of the adsorbate. The effectiveness of MB dye at varied adsorbent dosages is given in Figure 8

The outcome of palmyra palm male inflorescence activated carbon (PPMIAC) dose rate.
From the obtained results, the effectiveness of the MB dye elimination rises with a rise in the adsorbent dosage. Figure 8 shows that the effective elimination of MB dye increased from 57.69% to 98.5%. The primary reason behind the better removal efficiency is increasing the PPMIAC dose rate between 1 and 6 g/L. We know that an increase in adsorbent dosage will also increase the removal effectiveness because more surface area will be available for the dye molecule to incorporate into the adsorbent surface. After 5 g/L of dosage, adsorption will not occur because the system has attained equilibrium. After the optimum dosage, there will be a slight increase in the effectiveness of the adsorption. So, 5 g/L will be the optimal dosage for eliminating MB.
A similar output trend was noticed in eliminating Black 5 and Congo red from synthetic effluent using coffee waste as a biosorbent (Wong et al., 2020).
Influence of pH
The pH mainly influences the elimination of MB dye by PPMIAC. By keeping the dose rate of PPMIAC at 0.5 g, 100 mg/L of MB concentration, and shaking time of 60 min as constant, the solution's initial pH is altered from 2 to 9. The removal efficiency for MB dye removal on the effect of pH is shown in Figure 9. Generally, MB is an aromatic dye with an amino group exhibiting positive charge ions. The adsorbent surface contains acidic groups and attracts the MB dye by electrostatic attractive force. While altering the pH value, the adsorption efficiency will increase from 35.57% to 98.5%. The greater removal efficiency of 98.5% for PPMIAC is obtained at pH 8. It is noticed that the adsorption effectiveness of the dye is increased with a rise in pH. After pH 8, the solution becomes more turbid. So, pH 8 is set as the optimum condition for this process. A similar result pattern was noticed in the elimination of MB by using two kinds of hydro chars prepared from the orange peels and D-Glucose as feedstocks (Nguyen et al., 2019).
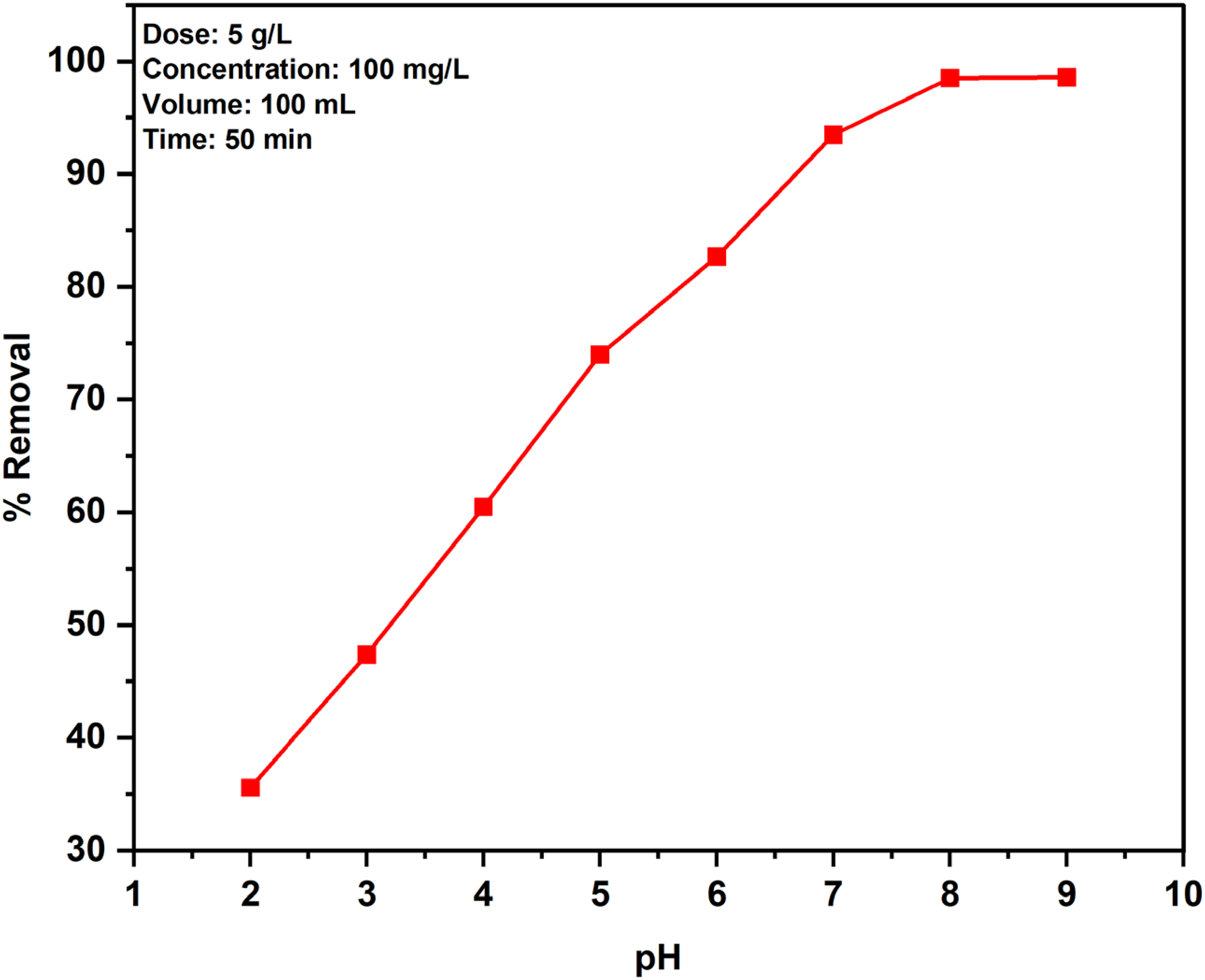
The outcome of pH of the adsorbate.
Outcome of initial dye concentration
The MB dye was effectively eliminated by altering adsorbate concentration varying from 50 to 250 mg/L and maintaining the other parameters as constant as pH 8, contact time of 60 min, and dosage of 5 g/L. Figure 10 displays the efficiency of MB dye removal by PPMIAC, and from the figure, it is clear that the removal effectiveness of the MB decreases with the rise in the dye concentration. It is lowered from 96.4% to 64.7% while altering the concentration. It results from the dye molecules on the adsorbent occupying the pores. After 100 mg/L, there will be no more space for the other dye molecule on the adsorbent surface. So, the equilibrium is attained at 100 mg/L. After equilibrium, there won't be any adsorption. Besides that, the speed of initial adsorption was higher for the lower concentration, and the resistance for the adsorption will be increased after the adsorbate concentration is increased. A similar result pattern was obtained in eliminating MB using corn straw and GO as an adsorbent (Liu et al., 2018).

The effect of methylene blue (MB) dye concentration.
Impact of temperature and thermodynamic studies
Temperature is also an important parameter to determine the removal efficiency of an adsorbent. For this work, the impact of the temperature is evaluated by altering the temperature from 303 to 333 K, thereby maintaining the other batch study parameters as constant. Figure 11 displays that the highest removal effectiveness of 98.54% was attained at 303 K, and then by increasing the temperature, the removal efficiency decreased to 92.56 at 333 K. Thus, it is clear that the removal effectiveness was lowered with a rise in the temperature. The primary reason behind the decrease in efficiency was that the adsorbent surface may undergo structural changes (shrink or have altered pores) when the system's temperature increases. Therefore, the MB dye molecules will start to desorb from the already adsorbed active sites, reducing the PPMIAC adsorbent's adsorption efficiency. Reduced surfaced activity and surface property while increasing the system's temperature indicated that the adsorption between PPMIAC and MB was exothermic. A similar trend was observed in eliminating Removal Brillant Violet- 5R Textile dye using eggshell as a biosorbent (Rápó et al., 2020).

The effect of temperature.
Thermodynamic parameters, including ΔG (Gibbs free energy), ΔS (Entropy), and ΔH (Enthalpy), were used to determine the nature and the spontaneity of the adsorption process (Figure 12). The parameters for thermodynamic studies like ΔH, ΔG, and ΔS can be estimated using the following formulas (Hussain et al., 2021).

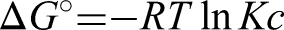
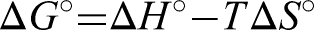
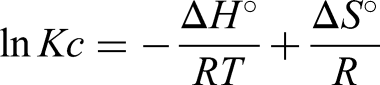
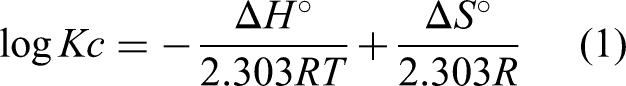

Thermodynamic evaluation.
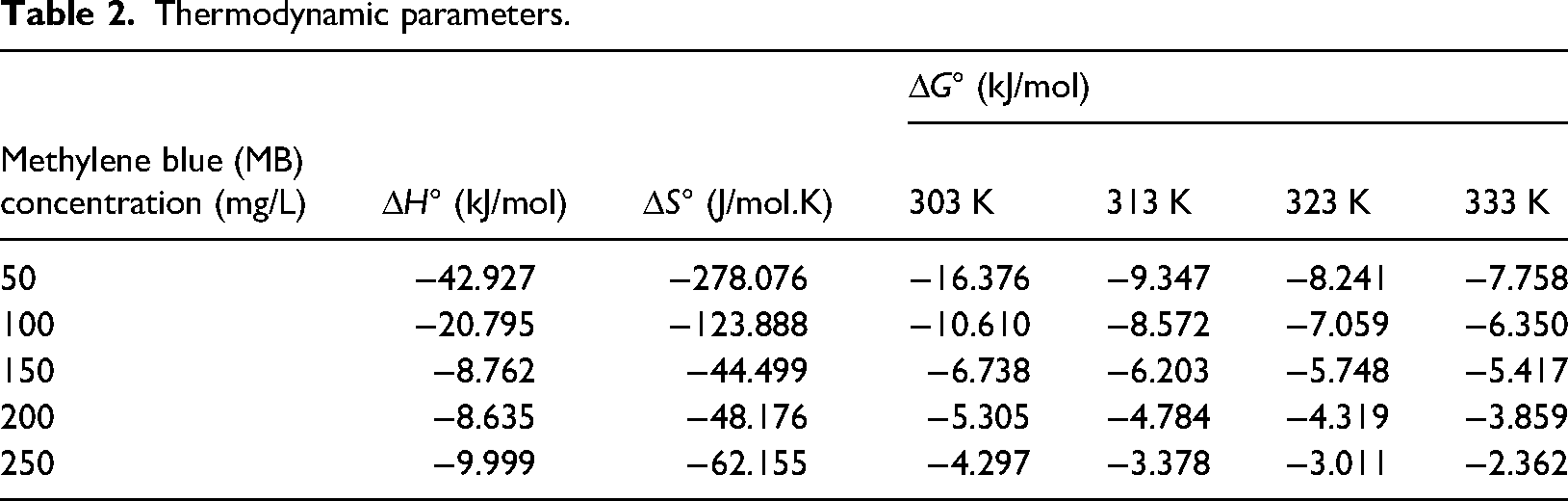
Gibbs's free energy (ΔG) values can be determined using Van't Hoff's equation, where the values of entropy ΔS and enthalpy ΔH were selected from the slope and intercept values obtained by plotting the graph between 1/T vs log Kc. The values obtained Δ for G, ΔH, and ΔS are tabulated in Table 2. The negative reading of ΔG indicates the feasibility and spontaneity of the adsorption process. The negative value of ΔH indicates the exothermic nature of the adsorption process. That is, heat energy is released during the adsorption. The negative value of ΔS describes the decreased randomness of the solid (PPMIAC)—solute interface. The decrease in randomness also indicates a strong association between the adsorbent and the adsorbate molecule.
Thermodynamic parameters.
Impact of shaking time
The effect of shaking time is also a critical criterion that enhances the system's effectiveness. The impact of shaking time for the effective elimination of MB dye is graphically represented in Figure 13. The interaction time varies from 10 to 60 min. The concentration of the MB dye is 100 mg/L; the dose rate of PPMIAC is 5 g/L, and the pH of the solution is set to 8. While using PPMIAC adsorbent, the equilibrium is attained more quickly within the first 50 min, and after that, there is no change in the removal efficiency. Due to that, most of the pores and active sites on the adsorbent will be occupied by dye molecules, and only a few will be left. Once the equilibrium is attained, this will significantly reduce the uptake of dyes. For PPMIAC, the equilibrium is achieved within 50 min, and for further understanding of the adsorption mechanism, the obtained data is utilized for kinetic studies. A similar trend was obtained in eliminating MB and Methyl Orange dye by oxidized chitosan (León et al., 2018).

The impact of contact time on the percentage removal of methylene blue by adsorption on palmyra palm male inflorescence activated carbon (PPMIAC).
Adsorption isotherm
Adsorption isotherm is a significant tool for estimating the adsorbents’ removal ability and providing basic details regarding the surface interaction between the pollutant and the adsorbent (Balji et al., 2022; Muniyandi and Govindaraj, 2021). Langmuir and Freundlich adsorption isotherm models were utilized to analyze the effectiveness of the removal and the nature of the mechanism in which the adsorption was carried out (Figure 14). In the Langmuir isotherm model, the surface of the adsorbent was assumed to be monolayer and homogeneous. In Freundlich isotherm studies, the surface nature of the adsorbent was considered to be multilayer with heterogeneity. The equation for the two isotherms is listed below (Freundlich, 1906; Langmuir, 1918).


Nonlinear adsorption Isotherm for the removal of MB dye by PPMIAC adsorbent. PPMIAC: palmyra palm male inflorescence activated carbon; MB: methylene blue.
Isotherm parameters for langmuir and freundlich.

SSE: sum of square error; RMSE: root mean square error.
Table 4 compares the adsorption capacity of already available adsorbents with the PPMIAC. The results show that the PPMIAC has a higher ability to remove MB from the aqueous solution.
Comparison of palmyra palm male inflorescence activated carbon (PPMIAC) adsorption ability with existing adsorbents.

Kinetics for the elimination of methylene blue (MB) dye.

SSE: sum of square error; RMSE: root mean square error; PFO: pseudo first-order model; PSO: pseudo-second order.
Kinetics of adsorption
The kinetics of adsorption is utilized to examine stability and the rate at which the adsorption occurs. Adsorption kinetics, which can depict the progression of an adsorbate as it migrates from solution onto the adsorbent, can offer basic details to comprehend the many sorts of adsorption processes and can also be used to investigate the rate-limiting step (Song et al., 2017). Pseudo-first-order (PFO), pseudo-second order (PSO), and intra-particle diffusion (IPD) models were used to analyze the uptake speed of MB dye by PPMIAC adsorbent. The equations for the kinetic models are listed below (Bharath Balji and Senthil Kumar, 2022),



Kinetic study for the removal of MB dye by PPMIAC adsorbent. PPMIAC: palmyra palm male inflorescence activated carbon; MB: methylene blue.
Regeneration
To assess the adsorbent's economic effectiveness, the spent adsorbent's regeneration plays a significant role. 0.1 M of NaOH was used as an eluting agent for the regeneration studies. Before regeneration, 0.5 g of the PPMIAC surface was fully saturated by agitating 100 mL of 50 mg/L MB solution for 24 h. After that, 0.1 M of NaOH was added to the solution, which was concerned for 180 min to desorb the MB dye molecules. After the filtration, the residual concentration of the solution was measured to determine its efficiency. The adsorbent was washed, dried, and used repeatedly for successive regeneration cycles. Figure 16 shows the regeneration efficiency of the PPMIAC adsorbent. The adsorbent can be regenerated up to four times, with the removal efficiency decreased to 92.5%–60.45%. The decrease in the efficiency of the regenerated carbon may be due to the physical or chemical changes in the surface of the adsorbent. These changes will diminish the available active sites and surface area of the adsorbent, thereby reducing the adsorption capacity of the regenerated material.

Regeneration of palmyra palm male inflorescence activated carbon (PPMIAC) adsorbent.
Conclusion
This study evaluated the successful preparation and evaluation of PPMIAC as an effective, environmentally friendly Neville material for efficiently eliminating MB dye as a synthetic effluent. The characterizing part of the adsorbent was done using a pair of techniques, including FTIR, BET, and SEM with EDX. From the experimental results, it is clear that the solution's pH considerably impacts the adsorption process. For the effective adsorption process, the pH range of 2–9 was set up, and the trials and the results show that the adsorption process was at its maximum at pH 8. At pH 8, the OH−ion concentration will be high, and an electrostatic force of attraction will occur between the positively charged MB and OH-ions. For the adsorbent dosage, the adsorption effectiveness will increase with a rise in adsorbent dosage because the surface area of the adsorbent will increase when the dosage of the adsorbent increases. For this process, 5 g/L will be the perfect dosage for effectively eliminating MB. The initial dye concentration and time for the maximum removal were found at 100 mg/L and 50 min. The PPMIAC adsorbent's maximum adsorption ability was determined as 38.82 mg/g. The Thermodynamic studies show that the adsorption process was exothermic. Kinetics and isotherm models show that the system follows PSOand Freundlich's adsorption isotherm.
Footnotes
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article


