Abstract
Nowadays, phenol and chlorophenols give rise to many organic pollutants. Concurrently, the occurring of phenolic compounds in aquatic ambit creates potential human health and environmental problems. There are a lot of technologies available for the treatment of organic pollutants. To remove phenol and chlorophenols, adsorption is a wastewater purification method. In this study, natural and activated clinoptilolite were carried out for the removal of phenol, ortho-chlorophenol, para-chlorophenol, and meta-chlorophenol. Activated clinoptilolite was prepared via acid activation and evaluated by means of Fourier transform infrared, scanning electron microscope, X-ray diffraction, X-ray fluorescence, and Brunauer–Emmett–Teller analysis. Phenol and chlorophenols have high adsorption rate, and equilibrium was reached in approximately 30 min. The maximum adsorptions of phenol, ortho-chlorophenol, meta-chlorophenol, and para-chlorophenols on the activated clinoptilolite were 6.7386, 8.6300, 9.5787, and 7.3758 mg g−1, respectively. The affinity order was in the following order: m-CP > o-CP > p-CP > Ph. The adsorption of phenol and chlorophenols decreased after pH = 6.25. Using the thermodynamic data, Gibbs free energy of the activated clinoptilolite was evaluated. The effects of different conditions such as pH, adsorption time, and initial concentration were analyzed. The adsorption rates of phenol and chlorophenols were constituted to be the maximum at approximately pH 6.5. Desorption of phenol and chlorophenols was carried out using ethyl alcohol solution (30%, v/v). The adsorption isotherms of phenol and chlorophenols on activated clinoptilolite were evaluated. The equilibrium adsorption data were the best suited to Freundlich adsorption isotherm pattern.
Introduction
Phenolic compounds are the vital pollutants because of their toxicity in the environment (Lin and Juang, 2009). They cause negative effects such as bad odor and taste to water. At the same time, they can consist of carcinogens (Baker and Ghanem, 2009). Chronic toxicities are usually due to the effect on heart, blood vessels, lung, kidneys, and other mental disturbances (Kamble et al., 2008). CPs are designated as priority pollutant materials by the Environmental Protection Agency (Denizli et al., 2001). Phenols (Ph) are brought out into surface water from many industrial wastes. Many techniques have been suggested for the management of wastewaters carrying hazardous materials (Lin and Juang, 2009). It is essential to determine a remarkable technique to eliminate these substances from wastewaters. The most common techniques found are chemical, biological treatment and solvent extrication. The important restriction of the above techniques is their weak capability in the removing of parts per million degree of phenolic compounds. Adsorption process has presently been utilized widely for the removal of trace level hazardous compounds (Ahmaruzzaman, 2008). Many sorbents, such as silica gel, activated-alumina/carbon, zeolites, polymers, ion-exchange resins, metal oxides, and clays, have been used (Furuya et al., 1997). These low cost adsorbents (LCAs) have been researched, and the removal of phenolic compounds and other substituted Ph was discoursed (Denizli et al., 2001, 2002; Uçar, 2009; Uçar et al., 2015).
Zeolites are microporous crystalline-hydrated alumino silicate solid structures that form a framework with cavities and channels. Zeolites have high surface areas and cation exchange capacities (CEC) (Evcin and Tutkun, 2009; Kuleyin, 2007). They are used for the elimination of some pollutants from the wastewater (Ghiaci et al., 2004). There are two types of zeolites: natural and synthetic zeolites. They have been utilized as membranes, adsorbents, ion exchanger, catalysts, and molecular sieves in industrial and municipal pollution control (Baker and Ghanem, 2009). Properties of zeolites can be modified by several chemical treatments (acid/base treatment and surfactant impregnation) (Wang and Peng, 2010). Many countries such as UK, Turkey, Mexico, Greece, Italy, etc. have large reserves of natural zeolites. (Lin and Juang, 2009). Chiefly, large deposits of clinoptilolite can be established in Bigadiç-Balıkesir, Gördes-Manisa, Sivas-Divrigi (approximately 2.2 billion tones), and deposits (Anonymous, 2010; Uçar, 2009; Uçar et al., 2015).
Sprynskyy et al. (2009) have studied on adsorption of the raw, HDTMA-, and NaOH-modified clinoptilolite. They have used Langmuir, Freundlich, and Dubinin–Radushkevich equations to evaluate the thermodynamic properties. Finally, the Freundlich isotherm fit better to sorption in low Ph concentrations (Sprynskyy et al., 2009). Ortega et al. (2016) have evaluated on Ph removal from industrial effluents through evolving polymeric resins by thermodynamic studies. They found that equilibrium data fitted to the Langmuir, Freundlich, and Temkin isotherms. But Langmuir isotherm provided the best correlation. The Ph uptake was a spontaneous process (ΔG° = –1.55 kJ mol−1) (Ortega et al., 2016). The adsorption of Ph and p-CPs with surfactant-modified zeolite was studied by Kuleyin (2007). The adsorption kinetics were evaluated by intraparticle diffusion, Elovich equation, and the pseudo-second order (PSO) reaction. The adsorption model for Ph and p-CP is well-matched with Freundlich isotherm. She found that the thermodynamic parameter free energy of sorption (ΔG°) changed with temperature. ΔG° increased when the temperature increased. For 20°, 40°, and 60°C, its values were 2.8214, 2.9310, and 3.0405, respectively (Kuleyin, 2007).
Clinoptilolite minerals which have selectivity to cations are not effective of removing anion and pollutants. However, the recent studies revealed if they are activated or modified, success can be achieved in this subject (Kuleyin, 2007; Yapar and Yılmaz, 2004). For this reason, acid and thermal activation is applied for the preparation of clinoptilolite. Acid treatment of natural zeolite may get rid of impurities in pores, increasingly remove cations to exchange into H-type, and de-aluminate the crystal form. There are two primary types of the modification process: ammonium change followed by thermal treatment and direct ion-exchange reaction with dilute acid solution (Wang and Peng, 2010). Ali et al. (2012) investigated LCAs for the elimination of organic substances. The study reports the conversion of waste yield into efficient adsorbents and their usage for water treatment (Ali et al., 2012). Delkash et al. (2015) investigated the most-recent accomplishments on zeolite-based processes for treating these wastewaters and discussed the availability of zeolites for wastewater improvement systems. Perego et al. (2013) described LCAs such as clinoptilolite, the most attractive of modified zeolites (ZSM-5) which is more effective than granular-activated carbons for the elimination of pollutants from wastewater. Şimsek et al. (2017) studied the derivatives of CPs that are stable organic substances in wastewater with cellulose-based microporous carbon fibers. Among the 2-Ph (CP), 2,4-dichlorophenol (DCP), and 2,4,6-trichlorophenol (TCP), TCP showed higher affinity and diffusion rate. Musa et al. (2012) evaluated the photocatalytic degradation of CP, DCP, and TCP by ZnO nano powders. They observed that CP had the least photocatalytic degradation efficiency. Ayala et al. (2014) investigated the synthesis of zeolites that were presented high CEC as compared to the others. Pal et al. (2015) studied the adsorption of Ph from aquatic media by silver nanoparticles and gold nanoparticles as adsorbents. They found that removing efficiency was 90% for Ph. Thue et al. (2017) used active carbon (AC) to eliminate o-cresol from aquatic media. The usability of synthetic resins, AC, and different LCAs for the removing Ph from contaminated water was reviewed by Lin and Juang (2009).
This study copes with the phenolic compounds from the aquatic media in a batch process by the utilization of ACL. The second purpose of this study is to examine the physical, chemical, and spectral properties of natural and activated clinoptilolite. Adsorption and removal of phenolic compounds by prepared ACL were studied.
Materials and methods
Materials
The zeolites adsorbents were received from Sivas-Divrigi of Turkey. Their chemical composition was characterized by X-ray fluorescence (XRF) spectrometry. Phenolic compounds delivered by Merck GmbH were utilized in the preparation of solutions.
Reagents
Concentrations of phenolic compounds were found by using a colorimetric assay (Bayramoğlu and Arıca, 2008; Uçar et al., 2015). For each adsorption isotherm, experiment was applied as described in our previous study (Uçar et al., 2015). The concentration of phenolic compounds was found by DR-5000 spectrophotometer at 510 nm.
Ph, CPs, 4-aminoantipirin, potassium ferricyanide (K3[Fe(CN)6)), hydrochloric acid (HCI), and sodium hydroxide (NaOH) were purchased from Merck AG (Darmstadt, Germany) and were used without further purification.
Acid and thermal modification of clinoptilolite
ACL was dried at 150°C for 6 h. Acid and thermal modification of NCL was applied elsewhere as described in our previous study (Uçar et al., 2015). After modification, the sorbents were well washed with deionized water and dried in air (consist of H+ cations) (Afanassyev and Moroz, 2003). ACL-A was used by treatment with ammonium chloride. De-aluminated sorbents were washed with boiling water and dried at 105°C (consist of NH4+ cations) (Grejtak et al., 1992).
NCL, ACL-H, and ACL-A were characterized by FT-IR (Perkin Elmer 460 Spectrum BXI spectrometer), Brunauer–Emmett–Teller (BET) surface area (Micromeritics Gemini 2360), scanning electron microscope (SEM) (LEO 1430VP), Zeta potential (MALVERN Nano-Z), X-ray diffraction (XRD) (Rigaku D-max 2200), and XRF methods (RIGAKU/ZSX PRIMUS 2).
Physical and textural properties of studied NCL were analyzed by the BET method (Micromeritics). The surface areas found for the ACL-H (H-clinoptilolite, 89.40 m2/g) sample were higher than this from NCL (18.99 m2/g).
Adsorption studies
The sorption capacity was calculated by equation (1)
NCL, ACL-H, and ACL-A were obtained from the solution by filtering at the end of each experiment, and their absorbances were measured three times and calculated average of data to reduce measurement errors by UV spectrophotometer. The amounts of adsorbed phenolic compounds qe (mg/g) were determined according to our previous study (Denizli et al., 2001; Uçar et al., 2015). Ph and CPs are vaporized at high temperature and, in the experimental part, was examined in three different temperatures (15, 25, and 35°C). Therefore, the ambient temperature was preferred in experimental studies because of optimum condition, easy, and simplicity. Then, other temperatures were not studied.
Results and discussion
Characterization of NCL
XRD diffractograms of NCL samples are shown in Figure 1. The crystallographic structures were dealt with X-ray diffractometer (Rikagu D-max 2200) to identify the dominant crystalline phases in the zeolite samples. XRD patterns were determined from 5° to 60° 2θ at a scan rate of 2°/min. Sample in Figure 1 comprises clinoptilolite and heulandite besides quartz and feldspar. The intensities of the peaks did not change after and before the adsorption of Ph. As a result of the activation, clinoptilolites have high substantial crystallinity degree and structural stability of the substances. Therefore, the employed treatments do not appear to force break down or important destruction of the clinoptilolite structure, at any rate, noticeable by this method (Rivera et al., 2011; Uçar, 2009; Uçar et al., 2015).
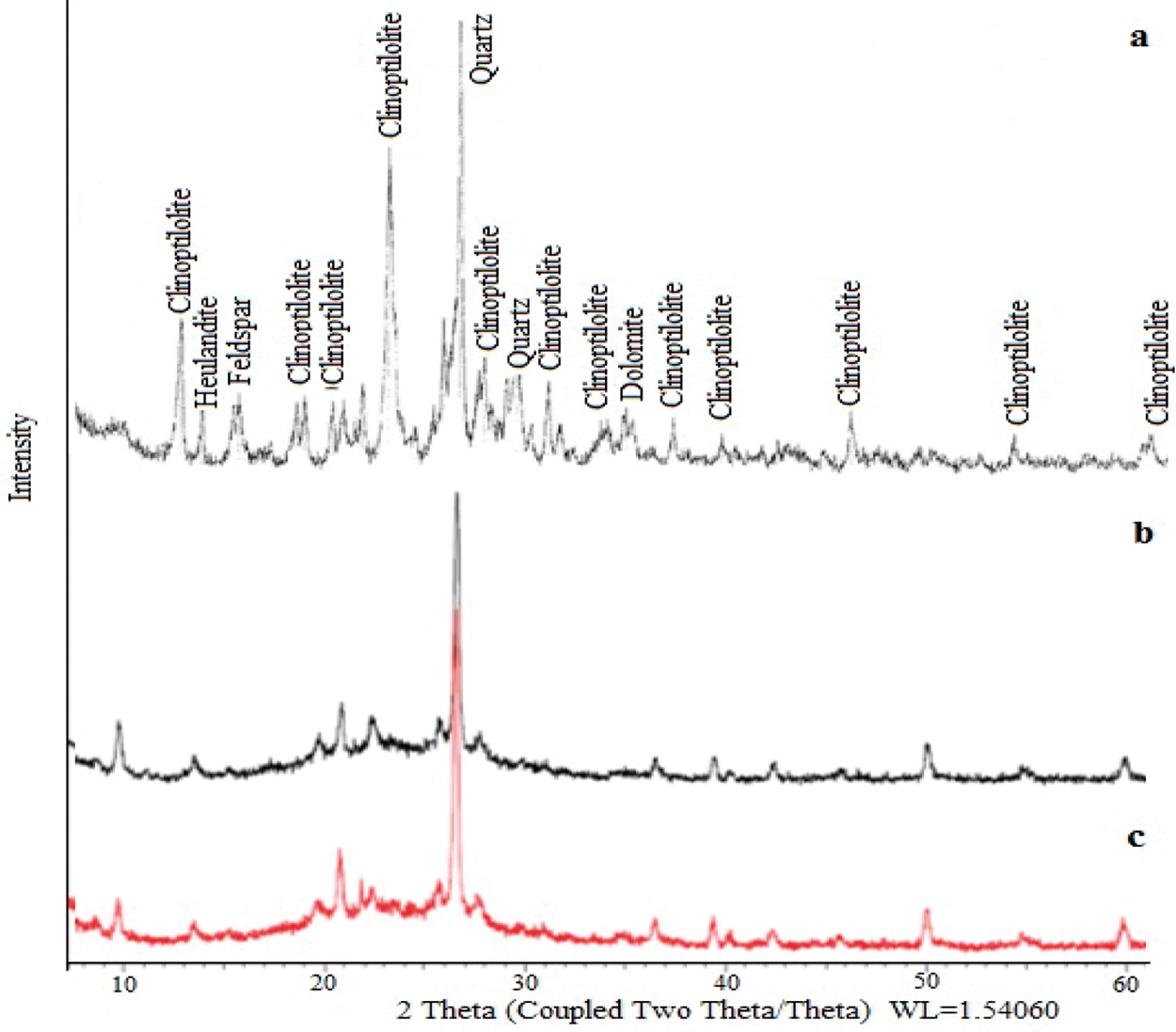
XRD spectrum of samples: (a) NCL, (b) ACL-A before adsorption, and (c) ACL-A after adsorption (m-CP).
XRF results of NCL are given in Table 1. ACL sample is characterized by high Ca, K and low Mg, and Na contents. Their chemical compositions have described that the main cations are Na+, K+, and Ca2+ by Ming and Dixon (Denizli et al., 2001; Douglas et al., 1987; Ersoy and Çelik, 2002; Saltali et al., 2007). Besides, iron element is present in the chemical composition. Iron oxide presents in zeolitic tuffs as an impurity. ACL has alumina, silica, magnesium oxide, and sodium oxide contents (Table 1). The SiO2/Al2O3 ratio estimated from these data was calculated as 4.81.
XRF results of NCL.

LOI: loss on ignition.
SEM–EDX study
The morphological properties of the NCL were characterized in scanning electron microscope (LEO 1430VP). SEM–EDX analyses of before and after adsorptions of ACL-A are shown in Figures 3 and 4. The zeolites have a structure with very high microporosity. The average silicone concentrations of ACL-A are 19.35%. The wide distribution of clinoptilolite is indicative of a glassy structure. Spherical structure can be seen because of heulandite in Figure 3.
The EDX data show the elemental composition of ACL-A. The chemical composition of pure clinoptilolite has a Si/Al ratio of 3.69 before adsorption and 5.45 after adsorption. Figure 2 shows that atomic ratios of the elements have changed before and after adsorption. Amount of sodium, magnesium, and aluminum have decreased, whereas that of silicone, potassium, and calcium have increased in these samples. Additionally, iron has not found in ACL-A after adsorption.
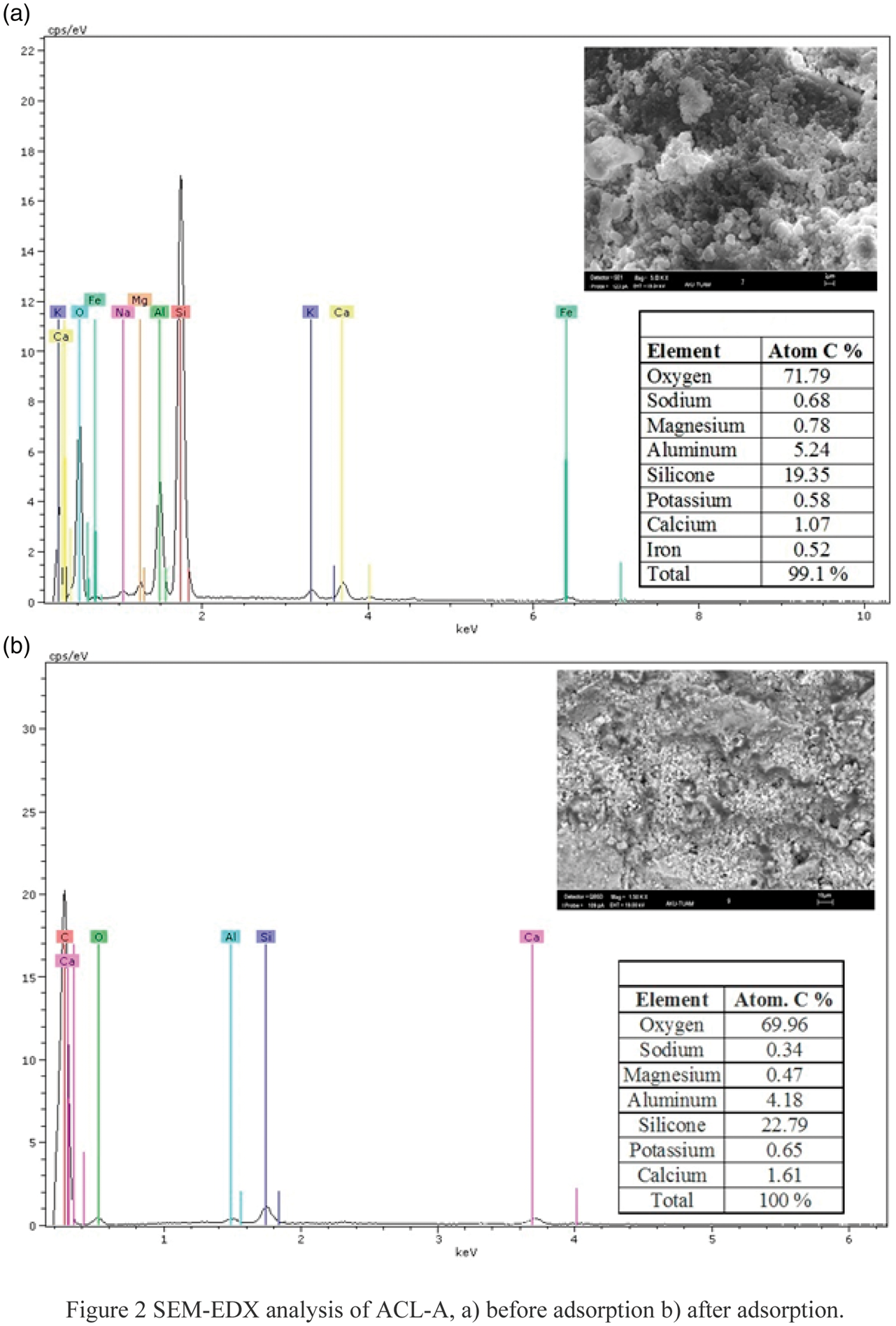
SEM–EDX analysis of ACL-A: (a) before adsorption and (b) after adsorption.
FT-IR study
FT-IR analysis of NCL and ACL-A is shown in Table 2 and Figures 3 and 4. It demonstrates the occurrence of similar bands after and before adsorption.
Molecular vibration by type.

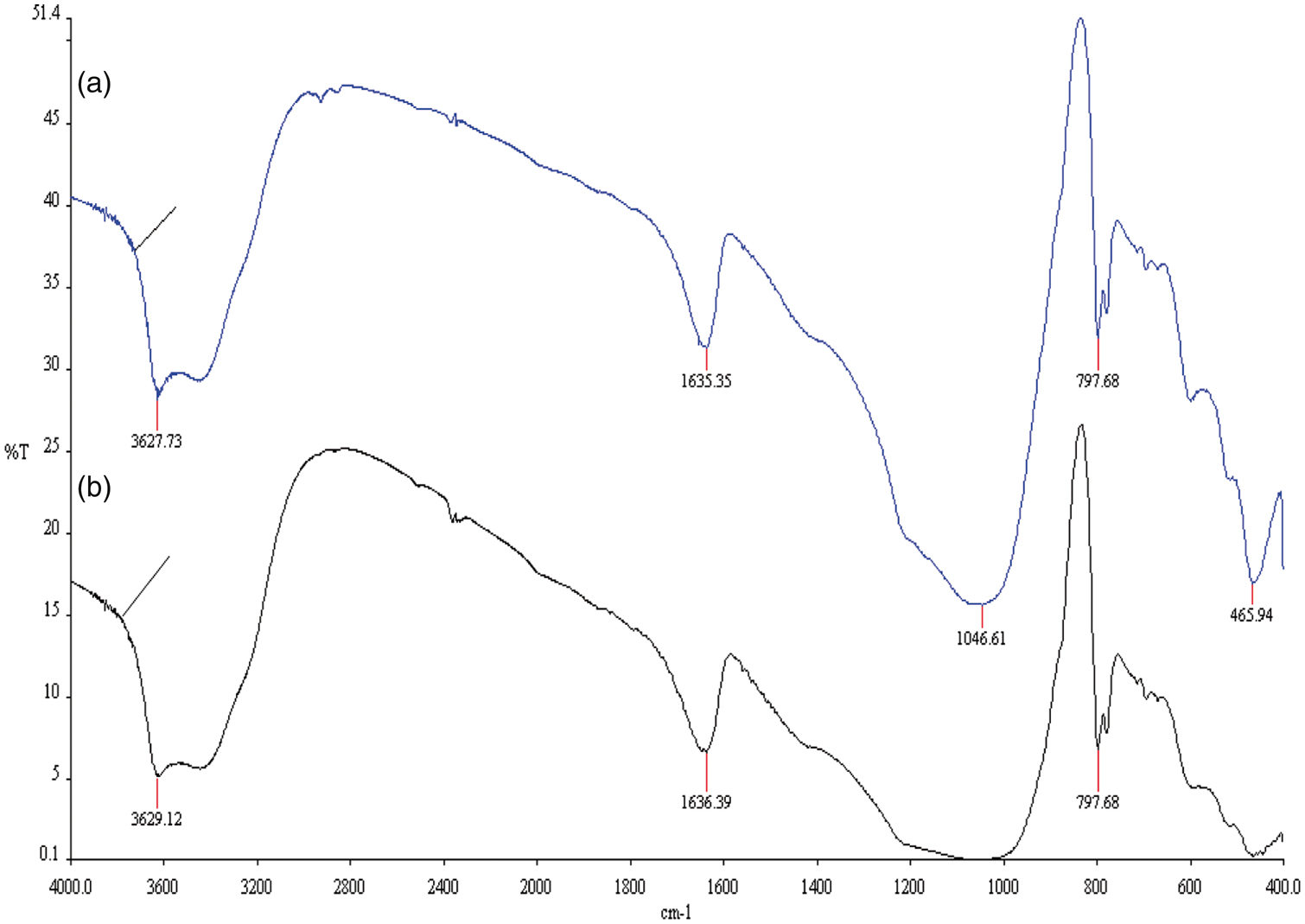
IR spectrum of NCL: (a) after p-CP adsorption and (b) before p-CP adsorption.

IR spectrum of ACL-A: (a) before p-CP adsorption and (b) after p-CP adsorption.
There are internal and external vibrations (Rodriguez-Fuentes et al., 1998; Zhi et al., 1992), Si–O(Al) and Si–O(Si) (Shindo et al., 2011; Spiridon et al., 2013), OH stretching vibrations (Bardakçı, 2007), and pseudolattice vibrations in all clinoptilolite (Bardakçı, 2007; Korkuna et al., 2006). Results of IR analysis are in agreement with the literature (Kussainova et al., 2019; Yazdi et al., 2019).
The zeta potential measurements
The zeta potential measurements of the NCL and ACL-A (NH4+ forms of clinoptilolite) particles were analyzed by Zeta-Meter 3.0. The Helmholtz–Smoluchowski equation can be used to underestimate zeta potentials. The zeta potential measurements were made as described in our previous study (Uçar et al., 2015). Determination of electrostatic interactions between adsorbate and adsorbent in solution will be realized with magnitude of charge repulsion/attraction between particles. Zeta potential is a scientific issue and fundamental parameter for electro kinetic potential in colloidal systems (Ersoy and Çelik, 2002).
Zeta potential values obtained for the NCL and ACL-A are shown in Figure 5. It reveals that the NCL and ACL-A surfaces assume a negative charge in water. It is due to the replacements of Al3+ for Si4+ in the clinoptilolite structure (Figure 5).
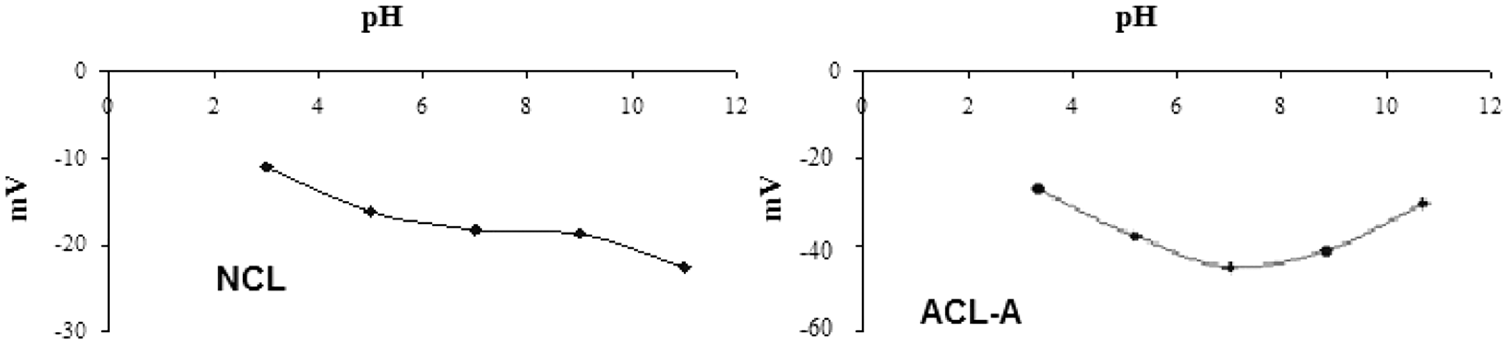
Zeta potential of NCL and ACL-A.
The reduction of the negative potential for NCL between pH 7 and 11 is owing to the H3O+ adsorption on the negative charge regions. The increase of the suspension pH leads to an increase in the negative charge of ACL-A. Increasing the pH to 11 reaches the zeta potential to approximately –30 mV. This can be described to either the adsorption of OH– ions onto the positive charge centers of ACL-A or the deprotonation of surface OH– groups (Ersoy and Çelik, 2002).
The cation-exchange capacities for NCL and ACL-A were 24.457 and 21.739 mg/100 g, respectively. Cation exchange capacity (CEC) of clinoptilolites depends on the nature of the cation species, temperature, concentration of cation, anion species, solvent, and their chemical compositions (Gregg and Sing, 1981). The Si/Al ratio of clinoptilolite structure has an essential parameter. If the Si/Al ratio is lower, clinoptilolite loading capacity is higher. They regulate the ion change sites and the CEC.
Ph and CPs adsorption
Maximum adsorptions of Ph and CPs on the ACL-A were 6.7386 mg g−1 for Ph, 8.6300 mg g−1 for o-CP, 9.5787 mg g−1 for m-CP, and 7.3758 mg g−1 for p-CP. As shown in data, there is a consistency within the literature data (Denizli et al., 2001; Uçar et al., 2015).
The alteration in adsorptive activity of the CP types related to one another can be described by the dissimilar interaction between CP types and functional groups in ACL. A variation in crystal frame is most likely the circumstance for the ACL-A, bringing about a notably high adsorption of m-CP (Denizli et al., 2001).
Recent studies of the methods of exclusion of phenolic species from wastewater have focused on adsorption and other methods. For example, the adsorption properties of Ph and 4-CP by zeolite were studied by Kuleyin (2007). Elimination of CPs (Ph, o-CP, m-CP, p-CP) from aquatic media was studied by Uçar et al. (2015) and Denizli et al. (2001).
Adsorption time
In the adsorption studies, equilibrium time was studied (Figure 6). It is noteworthy that these batch processes were completed by single solutions of the relevant phenolic compounds. The initial concentrations of the phenolic compounds in the aquatic media were preserved constant at 200 mg/L. These kinetic experiments show that the adsorption quickly copes within the first 30 min (Shindo et al., 2011; Uçar et al., 2015). Saturation levels are step by step achieved within 30 min for all phenolic compounds.
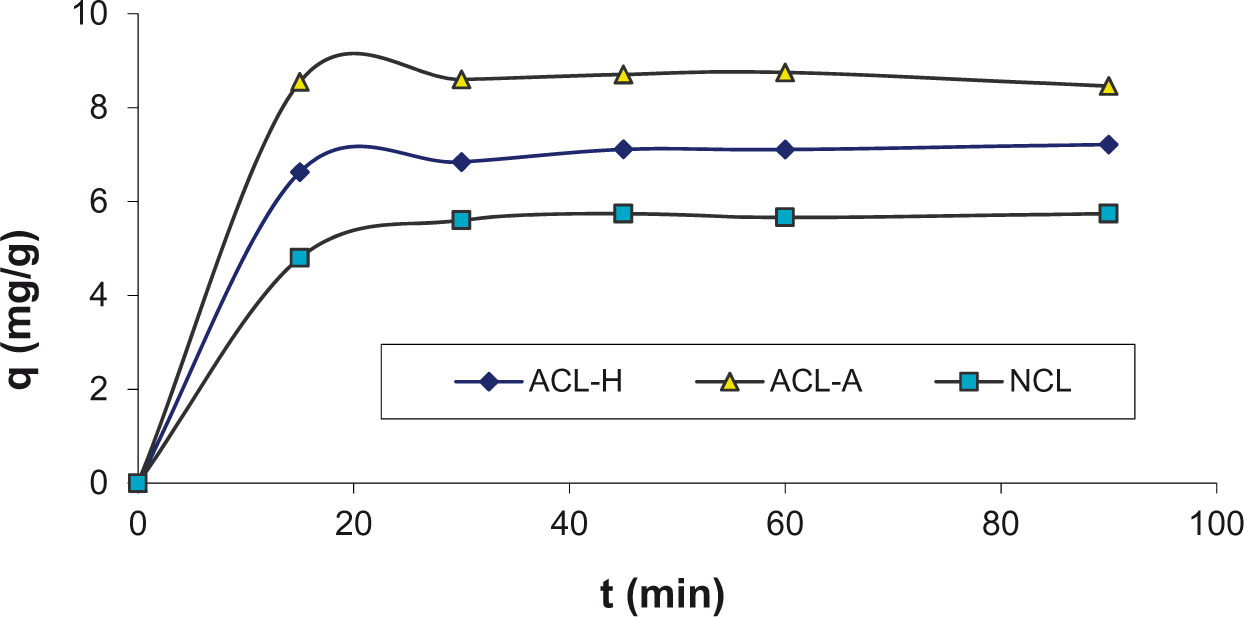
Adsorption rates of phenol on the samples at 25°C (initial concentration: 200 mg/L; s/l = 5 g/L; T = 25°C; pH = 6.25; 400 r/min).
Adsorption rate depends on some parameters such as mixing rate, physical properties of the sorbent, the amount of sorbent, adsorbate molecular weight, etc. Therefore, it is very difficult to make a comparison for that adsorption rates (Denizli et al., 2001; Kuleyin, 2007). Adsorbent dosage was examined such as 10, 50, 100, 200, 400, 800, and 1000 mg/L. Then, minimum adsorbent dosage was preferred to supply economical advantage.
Effect of concentration
In Figure 7, removal percent of the ACL-A is shown as a function of the first concentration of phenolic compounds within the aquatic media. The percent of removal was importantly raised with the initial concentration.
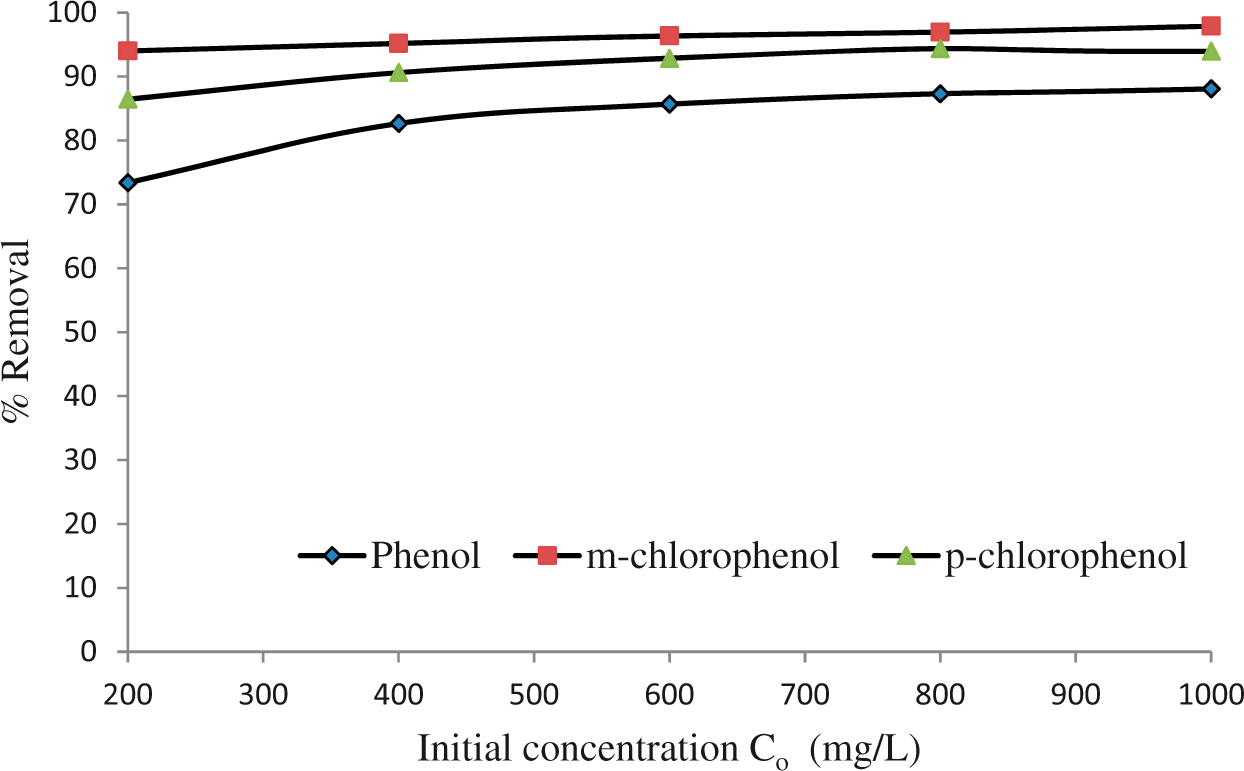
Comparison of phenolic compounds removal by ACL-A at 25°C (pH = 6.25, s/l: 5 g/L, 400 r/min, t = 45 min).
Effect of pH
pH is one of the most vital parameters affecting the adsorption. The effect of pH is shown in Figure 8. pH was adjusted by diluted NaOH solution. After pH = 5.0 was investigated in this study, clinoptilolite is not stable at below pH = 5.0 (Kamble et al., 2008). The maximum adsorption of phenolic compounds was at approximately pH = 6.25 for all types (Denizli et al., 2001; Ersoy and Çelik, 2002). The affinity order was m-CP, o-CP, p-CP, and Ph. Adsorption of phenolic compounds was fast at high initial solution concentration.
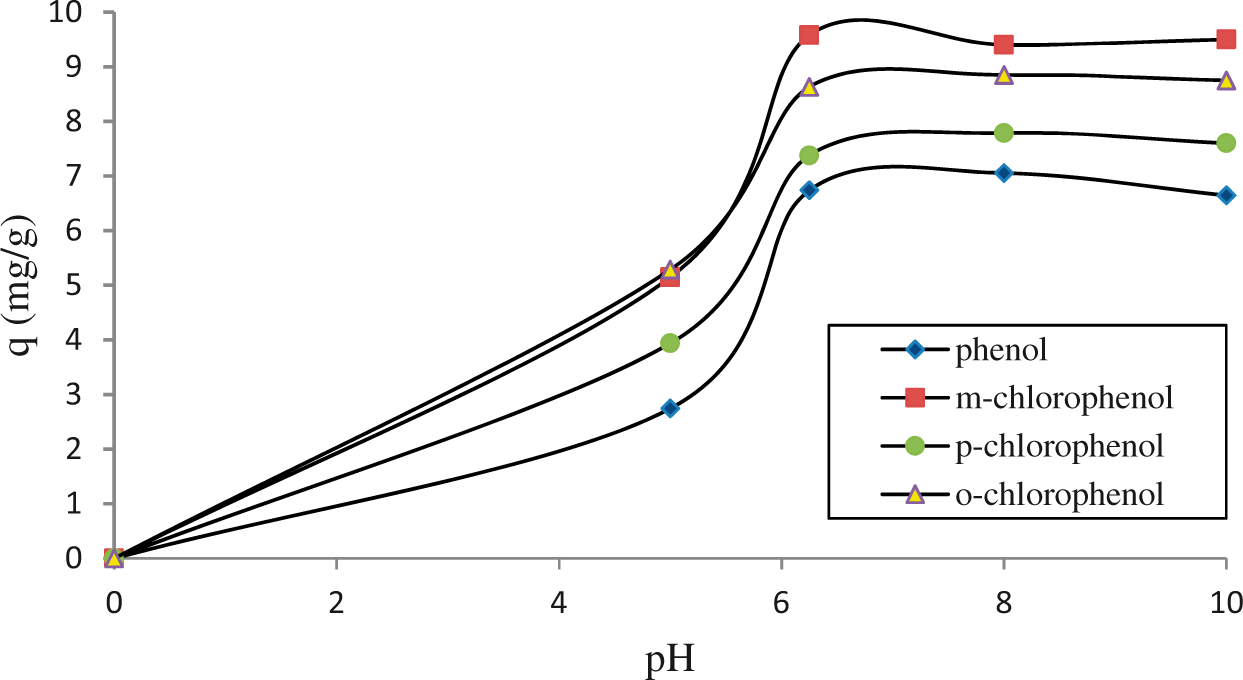
Effect of pH on adsorption of phenolic compounds on the ACL-A at 25°C (initial concentrations of CPs: 200 mg/L, s/l: 5 g/L, t = 45 min, 400 r/min).
Effect of thermodynamic parameters
ΔG° (Gibbs free energy) can be calculated with the following equations (Musa et al., 2012)


Thermodynamic parameters for phenol’s adsorption on NCL.

The negative values of free energy change (ΔG) indicate the spontaneous nature of the adsorption process. The negative values of ΔG0 confirm the feasibility of the process and the spontaneous nature of sorption with a high preference of Ph and CPs on NCL (Varank et al., 2012). However, the negative ΔG0 value decreased with an increase in temperature, indicating that the spontaneous nature of adsorption is inversely proportional to the temperature.
Freundlich adsorption isotherms
An adsorption isotherm was improved by H. Freundlich (1906) (Denizli et al., 2002; Kuleyin, 2007; Uçar et al., 2015). It defines the equilibrium on heterogeneous solid surfaces. It does not accept monolayer capacity. This isotherm is described by the following equation

Isotherm parameters for phenols (natural pH = 6.25, 400 r/min, s/l = 5 g L−1, 25°C).

The best fit isotherm was achieved with Freundlich as can be seen in literature (Kuleyin, 2007), and it was a physical adsorption.
Mechanism of adsorption
The adsorption kinetics followed the mechanism of PSO. Sorption equilibrium capacity for the PSO model can be given as

After integrating equation (7): t = 0, qt =0, t = t, qt = qt


The kinetic data are shown in Table 5. For PSO model, good correlation was obtained (Kuleyin, 2007). NCL are demonstrated by a Cl atom and an OH group, bound to ring of aromatic compound. The interactions between H and CI atoms are weaker than those between H and O atoms (Czaplicka and Kaczmarczyk, 2006).
Pseudo-second-order constants for the adsorption of phenolic compounds on ACL-A at 25°C (Co = 200 mg/L, natural pH = 6.25, 400 r/min, s/l = 5 g/L).

The adsorption model was mainly double molecule layer with adsorption isotherm fitting best with the Freundlich isotherm. In this study, according to the thermodynamic analysis, the adsorption nature is primarily physical which enhances with the increase of substitution degree, indicating that the more favorable adsorption of the substituted Ph could not be attributed to this mechanism. The hydrophobic interactions are believed to make contributions to the adsorption of Ph. In aqueous solution, the adsorbate with higher hydrophobicity has stronger tendency to be adsorbed and retain on the surface or in the pores. As suggested by the solubilities, the hydrophobicities of these Ph in study reinforce with the increase of the substitution degree, which is another reason for the enhanced adsorption of the substituted Ph. The molecular size also has some implications on the adsorption. The molecules with a suitable size would be adsorbed more favorably since they have more contact sites (Liu et al., 2010).
Conclusions
Wastewaters containing toxic organic substances are a crucial environmental trouble. Activated clinoptilolite is proving to be an effective new class of adsorbents for the adsorption of phenolic compounds. It has good physical as well chemical stability; hence it can be used for various environmental applications. The following conclusions are obtained from experimental studies: it is shown to be effective adsorbents in water treatment area. The stability of the clinoptilolite was not notably influenced by the acid treatment. The maximum adsorptions of Ph, o-CP, m-CP, and p-CPs on the ACL were 6.7386, 8.6300, 9.5787, and 7.3758 mg g−1, respectively. The affinity order was in the following order: m-CP > o-CP >p-CP > Ph. The adsorption of phenolic compounds reduced with increasing pH. The adsorption of CPs was high as compared to Ph, because CPs are more hydrophobic nature than Ph. It may be used at least three times.
In conclusion, the usage of NCL as an adsorbent could be an alternative for commercially available carbon due to its low cost and good efficiency.
Footnotes
Acknowledgements
The author thanks Dr Atilla EVCİN and Lecturer Songül UÇAR (chemistry engineer), researchers at Afyon Kocatepe University, for providing the necessary facilities.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by Afyon Kocatepe University via Scientific Research Projects (No: 07.MUH.09, 08.FENED.15 and 09.FENED.04).

