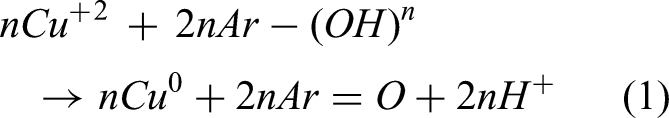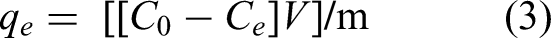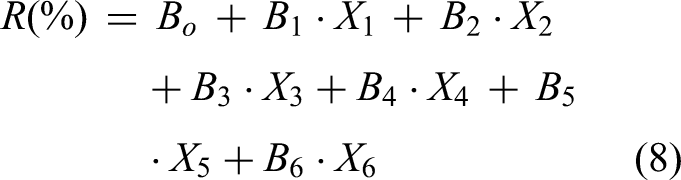Abstract
In this regard, green preparation of nanoscale-zero-valent copper was accomplished using Ficus benjamina leaves successfully. Ficus nano zero valent copper (Ficus-nZVCu) was characterized utilized scanning electron microscopy, and Fourier transform infrared spectroscopy. The results revealed that 63% of DR31 dye and 56% of DO26 dye were removed within the optimum conditions (0.3 g L-1, 30 min, 50 mg L-1, and pH 6). A Langmuir model was found to be the most fitting system for the removal process for DR31 and DO26. A Pseudo-second order kinetics model is the fit model of the removal processes for DR31 and DO26. The regenerated adsorbent still retained good adsorption capability after the fifth round of recycles. Ficus-nZVCu adsorbent is intensely recommended as a confident material for removing DR31 and DO26 anionic dyes from both prepared and actual wastewater samples. The co-presence of DR31 and DO26 did not affect each other's.
Introduction
The development of prepared dyes has led to a chemical revolution (Pereira and Alves, 2012; Wu et al., 2021), resulting in a more colorful world. Synthetic dye has gradually replaced natural dye and dominated the dye industry due to advantages such as simple structure, superior coloration, non-fading properties, and wide application (Tkaczyk et al., 2020). As production scales and application fields have expanded, synthetic dyes have gradually permeated every aspect of life (Kümmerer, 2010). Synthetic dye has a vibrant color but has also caused severe water pollution. Dyes wastewater from the textile, paper, and other industries have become contributors to water pollution. The most popular organic dyes among different dye wastes are azo dyes. Azo compounds account for more than 50% of all dyes. Despite their widespread use, they are regarded as persistent, and recalcitrant in natural environments due to the precision in which their structures are designed to withstand optical and chemical processes (Selvaraj et al., 2021; Wang et al., 2022). Azo dyes are challenging to degrade under normal conditions because of their stable complex aromatic structures (Munagapati and Kim, 2016; Schmidt et al., 2019). Azo dyes have a dark color that reduces water transparency, upsets the ecological balance of the water, and even stunts the development of aquatic life (Lau et al., 2014). The detrimental effects of azo dyes or their degradation byproducts on ecosystems and human health have also been established. In addition to increasing the risk of birth defects and cancer; they could also cause high blood pressure and other random illnesses.
Numerous studies have been conducted to combat environmental damage caused by dyes. For the treatment of azo dyes wastewater, electrochemical, chemical coagulation, and biological methods are currently employed. These removal methods failed to obtain a simple and effective treatment of dye-polluted water and they could not be reliable for industrial applications, and they were also expensive (Abdel-Gawad and Abd El-Aziz, 2019; Gholami et al., 2022; Xue et al., 2023). Adsorption has become the most promising water treatment method, with a large supply of adsorbents and a simple operation (Hossam and Abdel-Aziz, 2020; Abdel-Aziz et al., 2019). To improve adsorption performance, new eco-friendly adsorbents are being produced from nanomaterials (Abdel-Aziz et al., 2021; Khan et al., 2021).
Nanotechnology offers new opportunities for advanced wastewater treatment, which is a cost-effective, and reliable way to improve the quantity and consistency of water through nano-based materials (Abdulhameed et al., 2019; Tambe Patil, 2015). Nanomaterials have recently been recognized as a wide technique of wastewater treatment, due to their unique material goods, large surface area, high reactivity, mobility solution (Mohammad et al., 2019; Wu et al., 2019), good mechanical properties, hydrophilicity, and dispersibility (Khan et al., 2018; Yaqoob and Ibrahim, 2019).
Copper nanoparticles (CuNPs) acquired a lot of attention because of their outstanding optical, electrical, catalytic, mechanical, antifungal, and antibacterial characteristics (Abdel-Aziz et al., 2021; Haider et al., 2016; Ismail et al., 2019). CuNPs offer benefits compared to other metal nanoparticles (NPs) in that they are inexpensive, have extensive availability, and in comparison to the conventional adsorbent, provide superior yields and shorter reaction times under moderate reaction conditions (Abdel-Aziz et al., 2021; Kamal et al., 2016). Consequently, this is the primary motivation to develop CuNPs.
Green synthesis is a new nanotechnology field that investigates the synthesis of nanomaterials from natural biomaterials such as plants, flowers, and microorganisms. Plant leaf extract is the most significant source of NP synthesis. In the comparison of other chemical and physical methods, green biosynthesis of NPs provides an environmentally friendly, low-cost, and safe option (Abd El-Aziz et al., 2020; Zayed et al., 2023). Recently, many techniques for the preparation of CuNPs by the leaves extract, and their applications in the removal of contaminants have been uncovered through research (Chandra and Khan, 2020). Nanoscale zero-valent copper (nZVCu) was formed from the fruit leaves extract of mature Duranta erecta to reduce azo dyes such as Congo red and methyl orange (Ismail et al., 2019). To interact with organic dyes, CuNPs were made from the extract of Nerium leaves (Sebeia et al., 2019). To cut down dyes, Ahmed et al. (2019) produced CuNPs from Camelia sinensis leaf extract. In this paper, novel zero-valent copper (Cu0) NPs were created utilizing an innovative green technique (Das and Debnath, 2023; Deb et al., 2023) using extracts from Ficus benjamina (FB) leaves, as shown in equation (1):
Direct Red 31 (DR31) and Direct Orange 26 (DO26) (Figure 1) were selected as the study's focus among the various anionic azo dyes; because they are widely used for dyeing polyester fabrics, and printing, making them a common industrial pollutant.

(a) Molecular structure of DR31 and (b) of DO26.
However, some studies have been reported on using extracts from FB leaves to prepare nano zero valent particles as suitable adsorbents for commercial applications to remove dyes by using batch adsorption.
The main objective of this paper is to eliminate DR31 and DO26 from real samples of textile wastewater.
Materials and methods
Materials
All substances utilized were of the highest quality and analytical grade. Textile dyes DR31 (C
Synthesis of Ficus CuNPs (nZVCu)
Ficus leaves were washed, and dried at 105 °C; then crushed into small bits, and sieved through the sieve (2.5 mm). Plant extract of 50 g of leaves was added to 500 mL of bi-distilled water in a flask, heated (70 °C to 80 °C) for 60 min, and filtered. The solution of copper sulfate was prepared by dissolving 1.25 g of CuSO

Preparation of Ficus nanoscale-zero valent copper (Ficus-nZVCu) particles.
Characterization of Ficus-nZVCu
A scanning electron microscope was used to observe the morphology of the Ficus-zero valent copper (Ficus-ZVCu). Before scanning, Ficus-ZVCu nanoscale particles were coated with gold, which increases electron scatter to give high contrast measuring the particle size. Energy dispersive X-ray analysis (EDAX) was used to determine the composition of the sample. FT-IR was conducted to detect the vibrational groups present. These techniques help in identifying the chemical composition of prepared Ficus-ZVCu nanoscale particles.
Point of zero charge
Approximately 18.65 g of anhydrous potassium chloride was dissolved in 2.5 L of bi-distilled H2O. In Six 250 mL beakers, 200 mL of 0.1 M KCl solution was placed, and drop by drop the initial solution pH was adjusted with a pH meter (3510, Jenway) to solution pH 2 and 4 by 0.5 N HCl, solution pH 6 by 0.1 M HCl, solution pH 8 by 0.1 M NaOH, solution pH 10 and 12 by 0.5 N NaOH, then take 100 mL of adjusted 0.1 M KCl in a series of conical flask (solution pHi). Approximately 50 mg of Ficus-nZVCu particles were added to each flask (solution pH 2, 4, 6, 8, 10, and 12), then the flasks were closed, and the samples were left at room temperature for 24 h. Then, the final solution pH of each suspension was measured (solution pHf). Plotting Δsolution pH (final solution pHf − initial solution pHi) versus the initial solution pH value (solution pHi) was used to find the point of zero charge (Abdel-Aziz and Fayyadh, 2020).
Batch adsorption
The stock solution of 100 mg L
Adsorption equilibrium models
The adsorption equilibrium models are important to calculate and evaluate the various possible interactions between the DR31 and DO26 molecules with the adsorbent that occur in the adsorption process. These suggested models involved the linear Dubinin–Radushkevich, Elovich, Flory–Huggins, Fowler, Freundlich, Halsey, Harkins–Jura, Henry, Hill–Deboer, Jovanovic, Langmuir, Redlich–Peterson, Sips, Temkin, and Toth isotherm models, respectively. The equations used for different models that are used in the calculations of different adsorption parameters are depicted in Table S1 (Supplemental material).
Kinetic equilibrium models
The kinetic equilibrium isotherm models are important to investigate the mechanism of adsorption and the reaction processes that occur in the DR31 and DO26 removal processes using different possible orders of that reaction, such as pseudo-first-order, pseudo-second-order, Elovich, Fowler, intra-particle-diffusion, Avrami, Bangham, Boyd, and liquid film diffusion linear isotherm models. The equations used in different kinetic equilibrium models for studying adsorption processes in this text are shown in Table S2 (Supplemental material).
Thermodynamic study
To explore how temperature affects the removal of DR31 and DO26 dyes by Ficus-nZVCu materials, thermodynamic calculations were done to quantify the difficulty level and spontaneity of reaction processes. To examine the thermodynamics of the adsorption reaction, the experiment was done at different temperatures. The values of Gibbs free energy (ΔG), enthalpy (ΔH), and entropy (ΔS) were calculated (Atiya et al., 2022; Abdel-Aziz et al., 2020b):
Data analysis
The experimental settings changed throughout the present study, and all experiments were run separately in triplicate with appropriate controls running simultaneously. The collected data is given a mean (average) value, and Excel 2016 software is used to make a graph out of it. Also, equations are handled with the equation editor, and references are made with the Endnote program X7.2.1, numbered style.
Linear modeling algorithms for statistical data analysis
Linear regression analysis program (SPSS24
Response surface methodologies (RSMs)
The different operational factors’ impact on the batch was studied. If the P-value is <0.05, the effect is significant for the removal procedure. If the P-value is >0.05, the effect is not significant. Deduction of dyes in the adsorption process can be assumed to follow equation (8):
Artificial neural network (ANN)
An ANN with input, hidden, and output layers was developed using multilayer perceptron and “Multilayer Perceptron Backpropagation” to predict the efficiencies of wastewater contaminants removal. The input layer received the data from five experimental factors (dose, solution pH, time, stirring rate, initial concentration, and temperature), and the hidden layer contained different neurons. ANN models using backpropagation give the relationship between trained and tested values; they also provide the importance; and normalized importance of each effect (Abd El-Aziz et al., 2020).
Results and discussion
Characterization of Ficus-nZVCu
Different analytical and morphological tools were used to characterize and analyze the Ficus-nZVCu nano adsorbent.
Scanning electron microscopy (SEM)
SEM results of the morphological test of the prepared Ficus-nZVCu are presented in Figure 3.

SEM of a prepared Ficus-nZVCu sample.
The SEM image refers to the semi-spherical form of Ficus-nZVCu with nanoscale particles. In some cases, nano agglomerations are detected and linked to the sample centrifugation procedure. Several pores are also involved in the spread and transportation of organic dye to the inner Ficus-nZVCu.
EDAX results are depicted in Figure 4, which confirms the successful synthesizing of Ficus-nZVCu particles. Cu peak point to the occurrence of nano zero-valent particles. Other peaks include Ficus extract C, O, Si, and S (Abdel-Aziz et al., 2019).

EDAX of the prepared Ficus-nZVCu sample.
FT-IR measurements
The FT-IR measurements at 400 to 4000 cm

FT-IR spectrum of Ficus-nZVCu.
Polyphenol's broadband peak, which appeared at 3400 to 3100 cm
Point of zero charge (PZC)
The measurement of the PZC was done, and the results are shown in Figure 6.

Point of zero charge of Ficus nano zero valent copper (Ficus-ZVCu) nanoparticles.
The results in Figure 6 of Ficus-nZVCu PZC values are found to be ∼6.9 for FB.
Spectrum of dyes
The absorption spectra of both DR31 and DO26 dyes (50 mg L
Effect of operating parameters
Effect of adsorbent dose
The adsorbent dose effect of both DR31 and DO26 dye on the removal process was studied, and the obtained results, are presented in Figure 7.

The optimum effective dose and uptake on the removal.
The efficiency of DR31 and DO26 dyes removal was investigated when the doses of these dyes were altered from 0.2 to 0.5 g L
The influence of solution pH on the dye adsorption process
The effect of solution pH on the removal of dyes by the tested adsorbent is investigated, and the results are presented in Figure 8.

Effect of solution pH on the removal of the dye (solution pH 4, 6, 8, and 10, dose 0.3 mg L−1, time 60 min, room temperature, stirring 100 rev/min, concentration 50 mg L−1).
The solution pH of the dyes plays the most significant part in the dye removal efficiency procedure. DR31 and DO26 dyes were studied at different solution pHs from 4 to 10, under the selected proper conditions of a dose of Ficus-nZVCu is 0.3 g L
Effect of contact time
The effect of contact time between dyes and adsorbents is very important in the adsorption process, and the results obtained are presented in Figure 9.
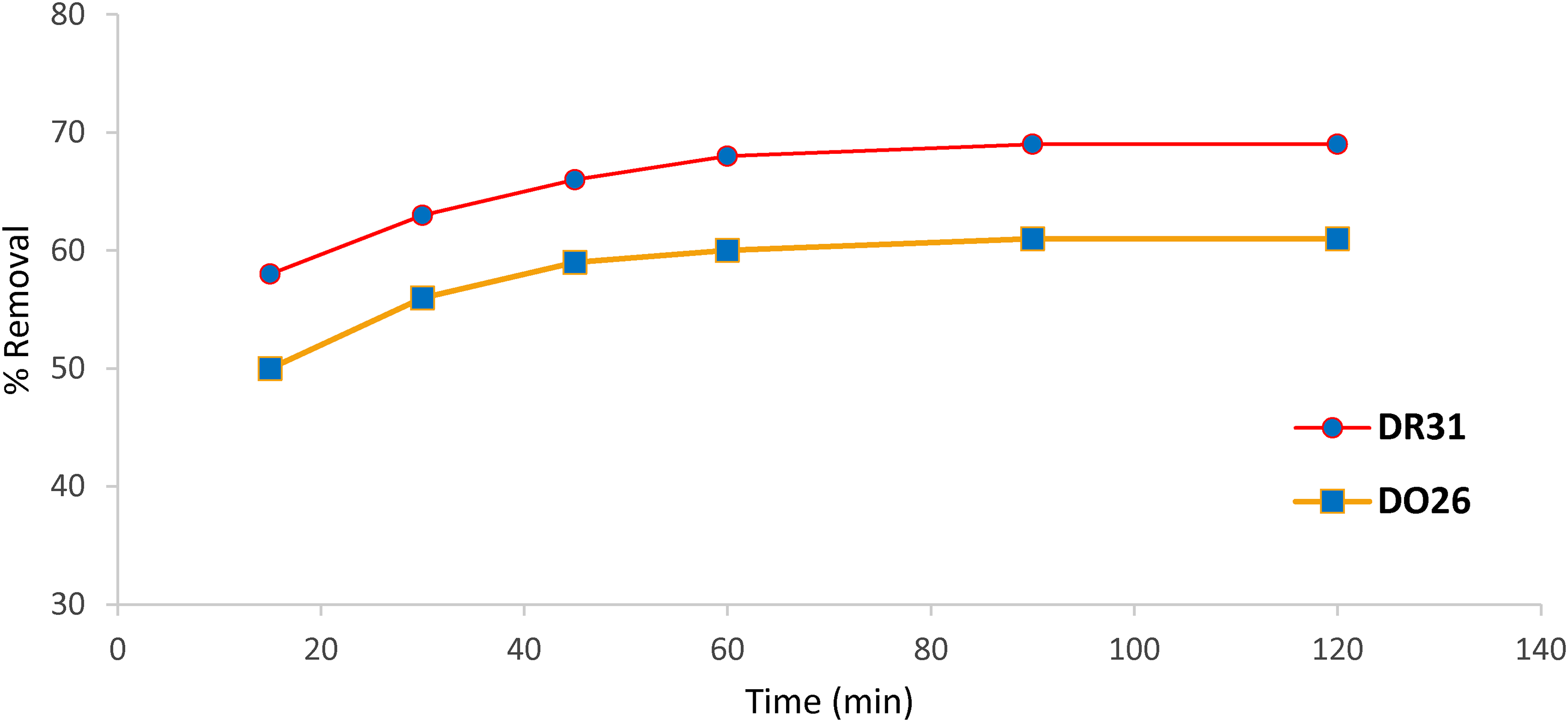
Effect of contact time on the removal of the dye (solution pH 6, dose 0.3 mg L−1, time 15, 30, 45, 60, 90, and 120 min, room temperature, stirring 100 rev/min, concentration 50 mg L−1).
At intervals of 15, 30, 45, 60, 90, and 120 min, the impact of time on effective removal was investigated. Using 0.3 g L
Effect of stirring rate on the dye removal
Figure 10 demonstrates DR31 and DO26 dye removal by Ficus-nZVCu at a varied stirring rate of 100 to 300 rev/min at solution pH 6, time 30 min, and a dye concentration of 50 mg L

Effect of stirring rate on the removal of the dye (solution pH 6, dose 0.3 mg L−1, time 60 min, room temperature, stirring 100, 150, 200, 250 rev/min, concentration 50 mg L−1).
The removal of DR31 and DO26 dye concentrations under these conditions is found to be (58%, 63%, 69%, and 69%) and (50%, 56%, 61%, and 61%), respectively. Therefore, the selected optimal stirring rate for the removal of both dyes is 100 rev/min.
Effect of the dye concentration
The removal experiments using Ficus-nZVCu particles were conducted on DR31 and DO26 dye solutions with different concentrations of 25, 50, 75, and 100 mg L

Effect of concentration on the removal of the dye (solution pH 6, dose 0.3 mg L−1, time 60 min, room temperature, stirring 100 rev/min, concentration: 25, 50, 75, and 100 mg L−1).
The removal efficiencies for DR31 and DO26 dyes at concentrations of (25, 50, 75, and 100 mg L−1), are found to be (94%, 63%, 41%, and 32%) and (89%, 56%, 38%, and 29%), respectively. Due to the abundance of empty adsorption sites at low concentrations at the start of the study, the removal ratio is high; however, as dye concentration increases gradually, this ratio decreases.
The temperature influence on the dye removal process
The impact of temperature on the rate of DR31 and DO26 dye removal from adsorption on Ficus-nZVCu particles was investigated at four different temperatures: 298, 303, 308, and 313 K, at solution pH 6, time of 30 min, concentration of 50 mg L
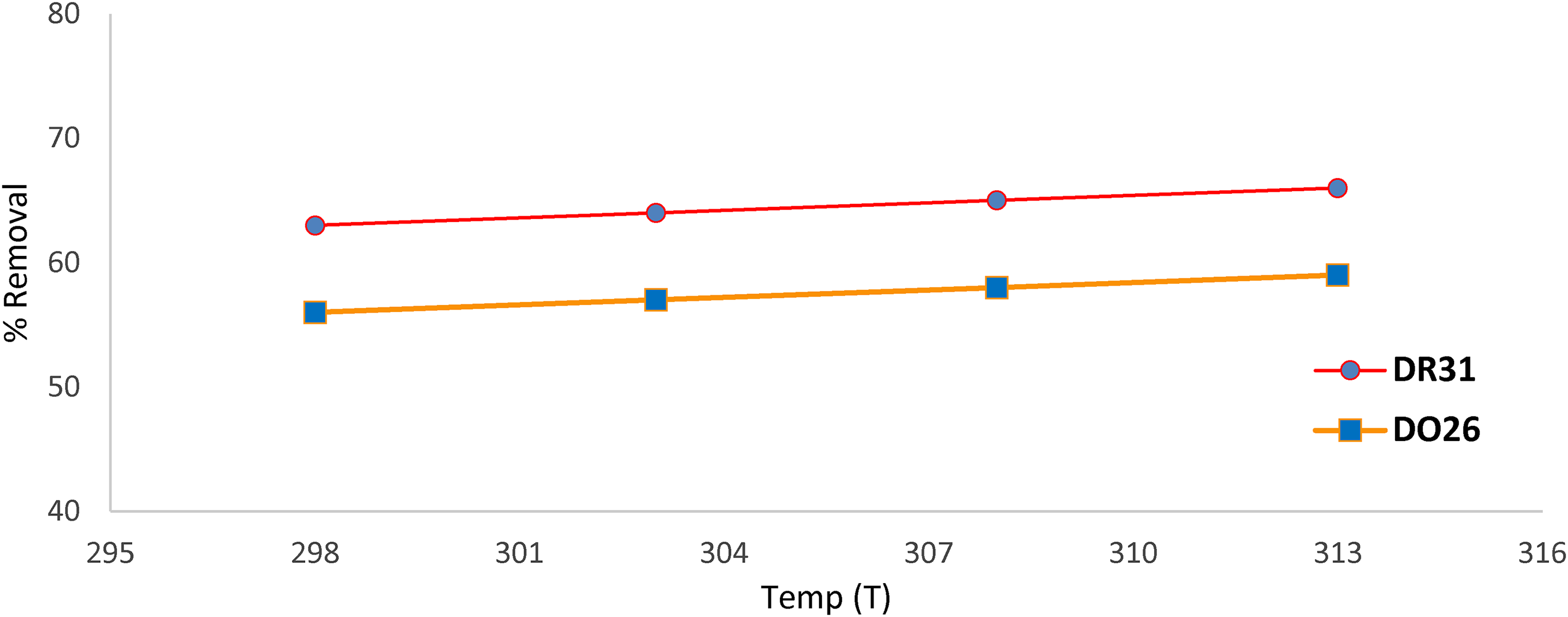
Effect of temperature on the removal of the dye (solution pH 6, dose 0.3 mg L−1, time 60 min, temperature (298, 303, 308, and 313), stirring 100 rev/min, concentration 50 mg L−1).
The removal efficiencies are found to be for DR31 (63%, 64%, 65%, and 66%), and for DO26 (56%, 57%, 58%, and 59%), as revealed in Figure 12. As predicted, raising the temperature causes a little bit of removal efficiency; which means the adsorption of dyes on Ficus-nZVCu particles is to some extent endothermic in nature. The reason for increasing removal efficiency with increasing temperature is probably due to the strong adsorptive powers between the molecules of the adsorbed phases as well as between the DR31 and DO26 dyes molecules and the Ficus-nZVCu active sites (chemical adsorption). Additionally, in some circumstances, the temperature increase affects the solubility of the dye molecules; which consequently has a substantial impact on the removal efficiency (Albroomi et al., 2017; Zhu et al., 2017).
Adsorption isotherm investigations
Linear isotherm models
The results of the study of the adsorption isotherm models are shown in Table 1 and represented graphically in Figure S1 (Supplemental material). As shown in Figure 13, the Langmuir model for DR31 and DO26 dyes fits the isotherm well by having higher correlation coefficients (R2) than other linear adsorption isotherm models and a higher maximum adsorption capability.

Langmuir isotherm studies for linear adsorption models (solution pH 6, dose 0.3 mg L−1, time 60 min, room temperature, stirring 100 rev/min, concentration (25, 50, 75, and 100 mg L−1)).
Linear adsorption isotherm models.

DR31: Direct Red 31; DO26: Direct Orange 26.
Non-linear isotherm models
The obtained results show that the Langmuir model fits the isotherm well by a low error sum compared to other non-linear adsorption isotherm models, as shown in Table 2 and graphically represented in Figure 14.

Langmuir isotherm studies for non-linear adsorption models (solution pH 6, dose 0.3 mg L−1, time 60 min, room temperature, stirring 100 rev/min, concentration (25, 50, 75, and 100 mg L−1)).
Parameters for different non-linear isotherm models.

DR31: Direct Red 31; DO26: Direct Orange 26.
Kinetics studies
Linear kinetics studies
The kinetics data obtained by applying different kinetic isotherm models are shown in Table 4 and graphically presented in Figure S2 (Supplemental material). According to Table 3, the particle swarm optimization (PSO) model for DR31 and DO26 dyes fits the data better than other kinetic isotherm models, as illustrated in Figure 15.

Pseudo-second-order kinetic studies for linear models (solution pH 6, dose 0.3 mg L−1, time (15, 30, 45, 60, 90, and 120 min), room temperature, stirring 100 rev/min, concentration 50 mg L−1).
Parameters for different kinetic isotherm models.

DR31: Direct Red 31; DO26: Direct Orange 26.
Parameters for different kinetic isotherm models.

DR31: Direct Red 31; DO26: Direct Orange 26.
Linear kinetics studies
According to Table 4, the PSO model fits the data better than other kinetic isotherm models by a low error sum, as illustrated in Figure 16.
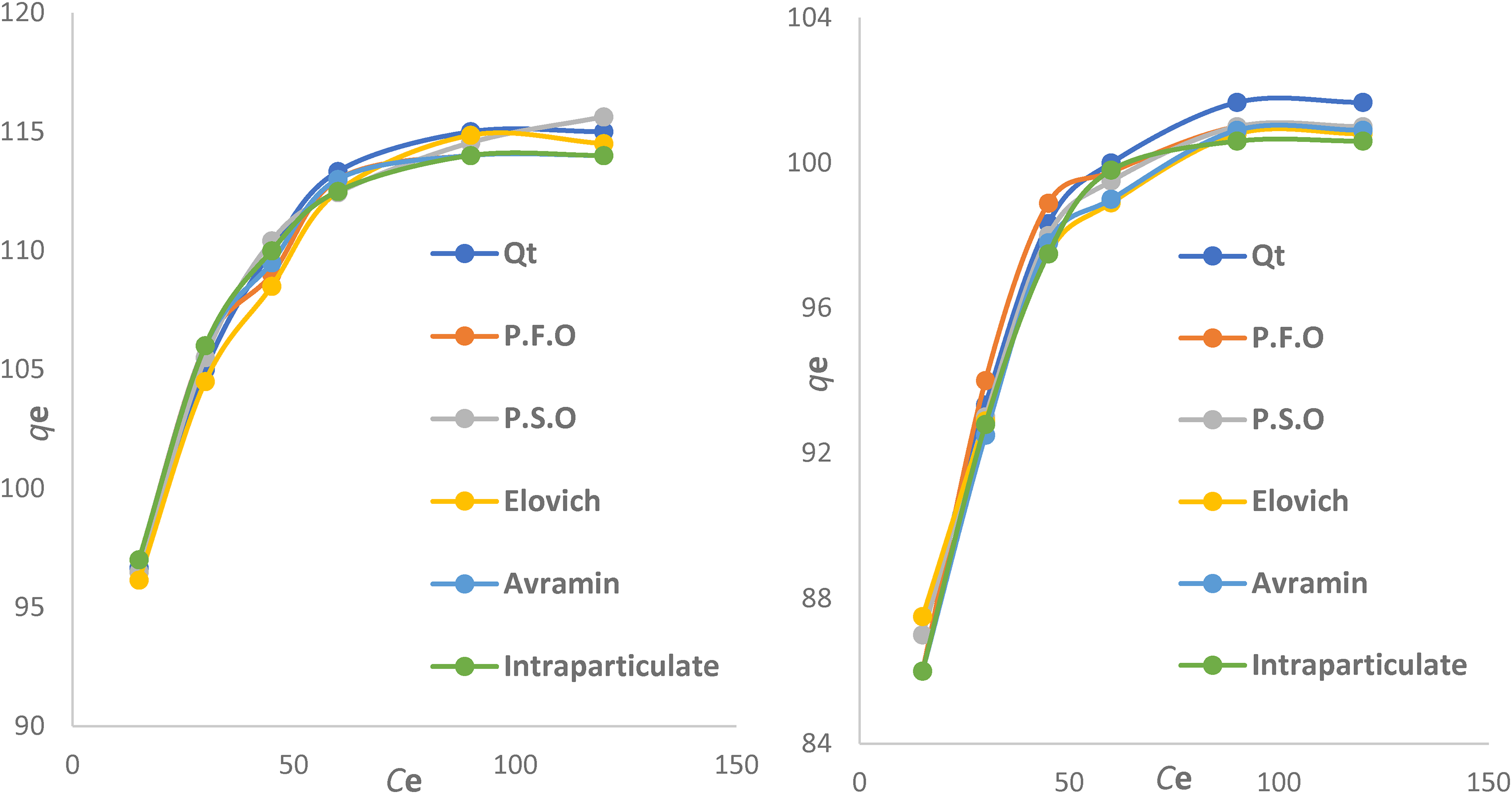
Pseudo-second-order kinetic studies for non-linear models (solution pH 6, dose 0.3 mg L−1, time (15, 30, 45, 60, 90, and 120 min), room temperature, stirring 100 rev/min, concentration 50 mg L−1).
Thermodynamic study
Figure 17 shows the thermodynamic parameters that were calculated for the different adsorption processes and how they affect their rates

Thermodynamic linear study for the removal of DR31 and DO26 (solution pH 6, dose 0.3 mg L−1, time 60 min, temperature (298, 303, 308, and 313 K), stirring 100 rev/min, concentration 50 mg L−1).
As seen, all of the values for the change in Gibbs free energy ΔG (−4.302, −4.483, −4.669, and −4.859) and (−3.581, −3.743, −3.909, and −4.081) were negative at various temperatures of 298, 303, 308, and 313 K, providing evidence that Ficus-nZVCu materials may spontaneously react with DR31 and DO26 dyes, respectively. The absolute value of ΔG increased as the temperature increased, indicating that the reaction was becoming more spontaneous and that high temperatures were favorable for the reaction, which points to a chemisorption mechanism. For DR31 and DO26 dyes, the enthalpy changed in a positive (6.77, 6.35), indicating an endothermic reaction. As a result, as the temperature goes up, the rate of both DR31 and DO26 dye reduction at equilibrium increases. In the system, the entropy ΔS change was also >0 (0.04, 0.03), showing that entropy increased during the adsorption process. When DR31 and DO26 dyes exchanged on the surface of Ficus-nZVCu materials, the entropy increased because solid–liquid interactions during the adsorption process made the system more random (Batool et al., 2018; Sahmoune, 2019).
Statistical analysis
Using the entered technique, the effect of variables on the removal processes of both DR31 and DO26 dyes was investigated and found that R
Response surface methodologies
Using linear regression analysis (IBM-SPSS Statistics), the impact of various studied parameters on the adsorption processes of both dyes was investigated, and the results corroborated the practical findings. The information obtained demonstrated that all factors had an impact on the removal process except time, stirring rate, and temperature, which can be disregarded because their P values are >0.05, as shown in Table 5.
Statistical analysis.

DR31: Direct Red 31; DO26: Direct Orange 26.
The B values are used for DR31 and DO26 in equations (9) and (10) to figure out the removal equation, while:
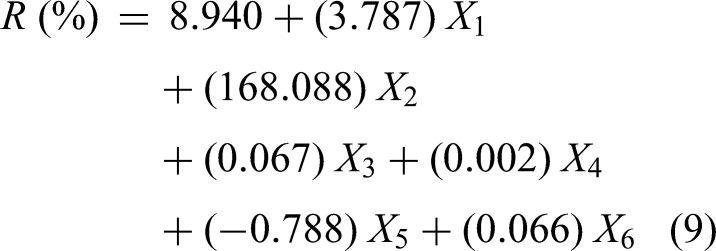

Artificial neural network
The multilayer perceptron neural network models 6-1-1 and 5-1-3 were used to train the ANN model for DR31 and DO26 dye removal using sample training and testing without excluding any results. Table 6 shows the sum of squared errors for testing and training, as well as the relative errors, for a total of 26 runs.
Model summary.

DR31: Direct Red 31; DO26: Direct Orange 26.
Figures S2 and S5 (Supplemental material) demonstrate that there is a slight difference between the normalized value and the predicted value. Figures S3 and S6 (Supplemental material) demonstrate that there is a negligible difference between the residual value and the predicted value, demonstrating the accuracy of the model's predictions for the adsorption of two dyes onto the Ficus-nZVCu surface. Figures 18 and 19 illustrate the significance of each co-variable in the removal efficiency, with dose for DR31; concentration for DO26 being the most useful parameters, stirring rate for DR31; and temperature for DO26 being the least useful. The ANN results were in agreement with the response surface methodology findings and the experimental data.

Direct Red 31 (DR31) importance for each co-variable.
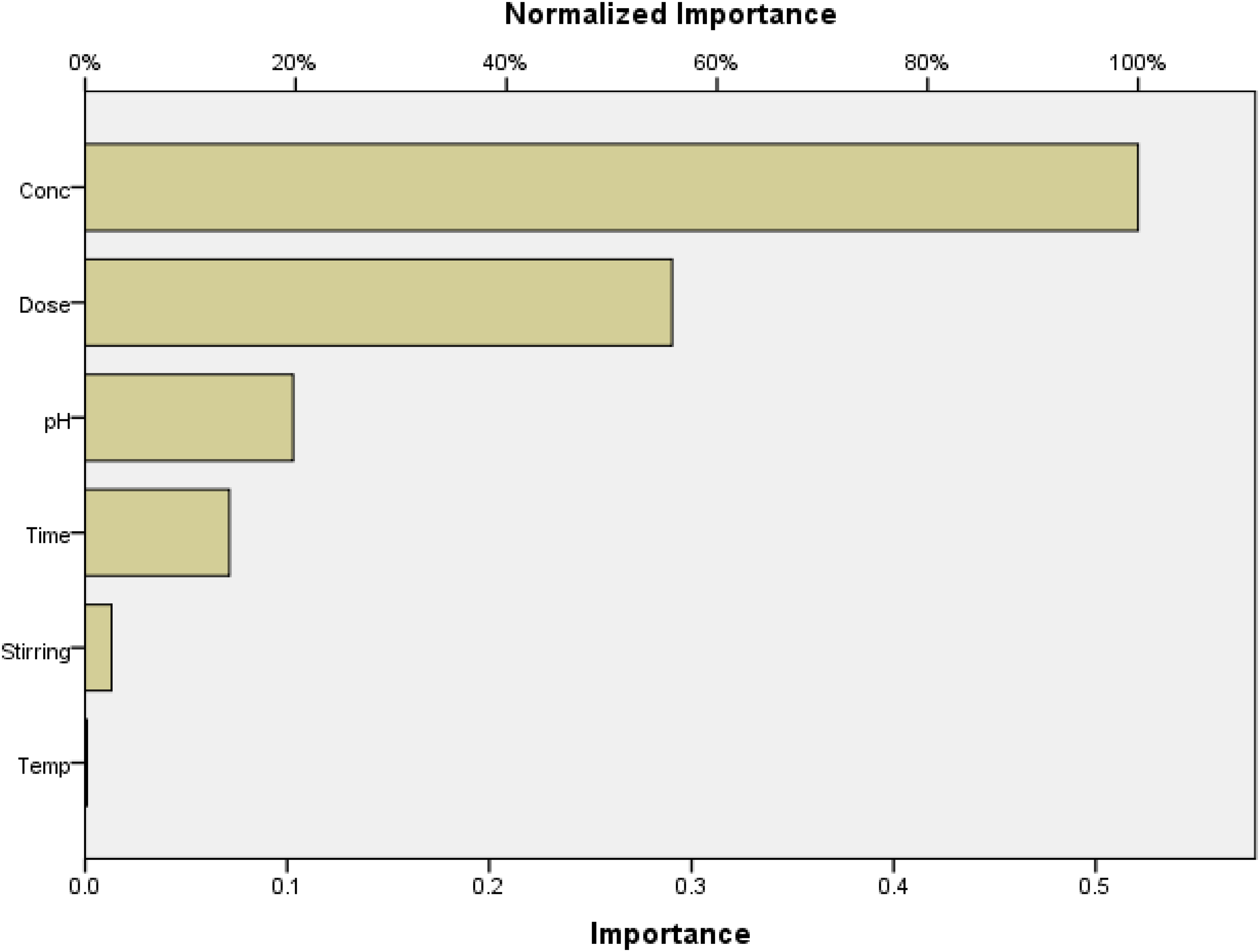
Direct Orange 26 (DO26) importance for each co-variable.
Reusability of FB-zero valent ion NPs
Figure 20 implies the removal efficiencies decreased with each reuse run of DR31 and DO26. Using 0.3 g L

Ficus nano zero valent copper (Ficus-nZVCu) particles reuse.
The co-presence of DR31 and DO26
Figure 21 depicts the effectiveness of nZVCu particles in removing the co-presence of DR31 and DO26. The absorption spectra of the mixed solution (50 mg L

The effect of co-present on the rate of the removal.
Finally, from the observed results obtained and the presented fruitful discussion, we can strongly say that Ficus-nZVCu is an eco-friendly adsorbent that is successful in the removal of both azo dyes DR31 and DO26 from textile wastewater and produces high-quality treated effluent.
Conclusion
Green synthesis of eco-friendly Ficus-ZVCuNPs was successful utilizing low-cost Ficus benjamina leaves.
SEM and FT-IR were used to confirm the production of porous NPs.
Under optimum conditions, the Ficus-nZVCu particles are efficient nano-adsorptive agents for removing DR31 and DO26 from wastewater.
The optimum removal efficiency of DR31 dye is 63%, and that of DO26 is 56% with the following parameters, dose 0.3 g L
The Langmuir isotherm and PSO kinetics models fit the adsorption data well with a high R2 value.
Langmuir and pseudo-second-order are more fitting in both linear and non-linear adsorption models.
The adsorption thermodynamic parameters showed the nature of spontaneity adsorption, chemisorption, endothermic processes, and random adsorption interference, which increased at the solid–solution interface and had a reasonable adsorption affinity of DR31 and DO26 dye molecules toward NPs.
The ANOVA software results revealed that nearly 95% and 93% of the variables influencing the DR31 and DO26 dye removal processes, respectively.
The co-presence of DR31 and DO26 had no impact on one another.
The regenerated adsorbent still retained fit adsorption capability after the fifth round of recycles.
Supplemental Material
sj-docx-1-adt-10.1177_02636174241256849 - Supplemental material for Adsorptive removal of Direct Red 31 and Direct Orange 26 azo dyes from aqueous solutions using Ficus nano zero valent copper: Linear, non-linear, response surface methodology (RSM), and artificial neural network (ANN) modeling
Supplemental material, sj-docx-1-adt-10.1177_02636174241256849 for Adsorptive removal of Direct Red 31 and Direct Orange 26 azo dyes from aqueous solutions using Ficus nano zero valent copper: Linear, non-linear, response surface methodology (RSM), and artificial neural network (ANN)
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.