Abstract
The Fe3O4/Talc nanocomposite was synthesized by the coprecipitation-ultrasonication method. The reaction was carried out under a inert gas environment. The nanoparticles were characterized by X-ray diffraction (XRD), field-emission scanning electron microscopy (FESEM), fourier-transform infrared spectroscopy (FT-IR) and vibrating sample magnetometry techniques (VSM), the surface area of the nanoparticles was determined to be 77.92 m2/g by Brunauer-Emmett-Teller method (BET). The kinetic data showed that the adsorption process fitted with the pseudo-second order model. Batch experiments were carried out to determine the adsorption kinetics and mechanisms of Cr(VI) by Fe3O4/Talc nanocomposite. The adsorption process was found to be highly pH-dependent, which made the material selectively adsorb these metals from aqueous solution. The isotherms of adsorption were also studied using Langmuir and Freundlich equations in linear forms. It is found that the Langmuir equation showed better linear correlation with the experimental data than the Freundlich. The thermodynamics of Cr(VI) adsorption onto the Fe3O4/Talc nanocomposite indicated that the adsorption was exothermic. The reusability study has proven that Fe3O4/Talc nanocomposite can be employed as a low-cost and easy to separate.
Introduction
Chromium used for many applications in different industries, including alloy fabrication, corrosion protection of metals, paint, textile, tanning, electroplating (Sueker, 1964). Chromium discharged from these industries has caused serious pollution in the water environment. Chromium has five oxidation states (from +2 to +6) but three of them (Cr(0), Cr(III), Cr(VI)) are used commercially and present in the environment. The oxidation state of chromium affects its toxicity and mobility in aquatic media. In the aquatic environment, Chromium is found in predominantly two forms, trivalent chromium (Chromium (III) or Cr3+) which is nontoxic, less mobile and has a tendency to absorb to clays, sediments, organic matter, and hexavalent chromium, which is toxic, more mobile (Wilbur et al., 2000). According to WHO guidelines, the concentration of chromium in drinking water should not exceed 0.05 mg/l. IARC has classified chromium (VI) as carcinogenic (Group 1) and chromium (III) as noncarcinogen (Group 3) (WHO, 2003). There are many methods that used generally to remove chromium and other heavy metal from water (Mahmoodi et al., 2019; Mostafavi et al., 2019), such as adsorption (Chávez-Guajardo et al., 2015; Deveci and Kar, 2013), filtration (Gherasim et al., 2011; Jayalakshmi et al., 2012), precipitation [(Golbaz et al., 2014), ion exchang (Alvarado et al., 2013; Kalantari et al., 2014), electrochemical treatment (Malkoc et al., 2006). In recent years, nanotechnology is a practical approach in treating wastewaters, too. Among all these possible methods, those with cost-effective, environment-friendly and no further pollutant features are the favorites (Lofrano et al., 2017). The solid wastes could be used the development of new adsorbents, low – cost from biogenic sources (Badruddoza et al., 2013); waste materials from industry (Bhatnagar et al., 2011); locally available agricultural waste (Ligate and Mdoe, 2015). This adsorbent materials are capable of capturing heavy metal ions form aqueous solution. Such as nano Talc doped Fe3O4 materials, are many methods to produce the nano Fe3O4 materials such as coprecipitation (Jahanbakhsh et al., 2017; Wei et al., 2012), solvothermal reduction (Sun and Zeng, 2002; Vuong et al., 2015), polyol (Laurent et al., 2008), copolymer gels (Qi et al., 2011), electrochemical (Jouyandeh et al., 2019a; Jouyandeh et al., 2019b)),…. Maryam Jouyandeh (Jouyandeh et al., 2019c) et al. have electrochemically synthesized ZnxFe3-xO4 magnetic nanoparticles to apply in curing epoxy. Now, to stabilize liquid particles, increase the dispersity and increase the specific surface area, scientists use Talc [Mg3Si4O10(OH)2 coatings on the surface of Fe3O4 particles. Talc has a platy structure that consists of three layers, including a magnesium hydroxide layer sandwiched between two silicate layers. Adjacent layers are connected by weak van der Waals forces. The surface of talc powder has a large number of Mg-O, OH, Si-O-Si and O-Si-O bonds that will cooperate or complex with heavy metal ions in water or soil to immobilize them on the surface of talc powder (Wang et al., 2018). Talc has been studied as an adsorbent for heavy metals in both forms: unmodified and modified talc. M.E. Ossman et al. used talc powder as an adsorbent for removing Cr(VI) from water. The results showed that the adsorption fitted well with the Freundlich model, optimization conditions include pH = 4, a contact time of 70 minutes to reach equilibrium (Ossman et al., 2014). Recently, some studies have been done on Fe3O4/Talc nanomaterial. Kamyar Shameli et al. (Kalantari et al., 2013) prepared Fe3O4 nanoparticles in talc as substrate using an environmentally friendly process. Fe3O4 MNPs have a mean size of 1.95–2.59 nm on the talc layers. K. Kalantari et al. (Kalantari et al., 2014) prepared Fe3O4/talc nanocomposites and used them as adsorbents for removing Cu(II), Ni(II), Pb(II) ions in the aqueous system. In optimal conditions, removal time of 120 s, removal efficiency for Cu(II), Ni(II), Pb(II) are 72.15%, 50.23%, 91.35%, respectively. The adsorption kinetics comply with the pseudo-second order kinetic equation and the Langmuir isotherm fitted well with data of the adsorption process. Shalini Rajput et al. (Rajput et al., 2016) synthesized Fe3O4 nanoparticles and used them for aqueous Cr(VI) and Pb(II) removal. Sorption data abided by pseudo-second order kinetics. The adsorptions of Cr(VI) and Pb(II) are in good agreement with the Sips and Langmuir models, respectively. The maximum adsorption capacities were 34.87 mg/g for Cr(VI) ions and 53.11 mg/g for Pb2+ ions at 45°C.
In Ultrasonic-assisted synthetic (Sonochemical synthesis) method, chemical reactions are facility due to the application of powerful ultrasound radiation (20 kHz–10 MHz). Sonochemistry generates small, hot bubbles that can archieve high temperature (5000–25,000 K), pressure of more than 1000 atmospheres and rate of cooling/heating that can exceed 10−11 K/s. These can break chemical bonds or produces chemical and physical effects that can be used for the production or modification of nanostructured materials (Qiao et al., 2011). Untrasonic-assisted reaction produced the smaller size, purer and more dispersible nanomaterials (Jiang et al., 2012) .
In this work, The Fe3O4/Talc nanocomposites were synthesized on the surface of talc layers in aqueous solution using Ferric chloride hexahydrate (FeCl3.6H2O), frerrous chloride tetrahydrate (FeCl2.4H2O), NH4OH as the iron precursor and reduction agent and Talcum powder substrate (Mg3Si4O10(OH)2). Talcum powder (30–50 µm) for preparing the nanocomposite was supplied by Talcum powder Phu Tho-Viet Nam. The adsorption isotherm, kinetic of Cr(VI) ion onto Fe3O4/Talc nanocomposite produced from Talc (Phu Tho – Viet Nam) were studied.
Materials and methods
Materials
Ferric chloride hexahydrate (FeCl3.6H2O), ferrous chloride tetrahydrate (FeCl2.4H2O) and potassium dichromate (K2Cr2O7) was purchased from Merck. The Talc – VietNam (particle size ≤50 μm; density 2.4 g/cm3; main components include: SiO2: 56.8%; MgO: 31.5%; Fe2O3: 3.5%). NH4OH were obtained from Macklin-China, India. A stock solution of 1000 mgl−1 Cr(VI) ions was made by dissolving 2.8289 g of K2Cr2O7 in distilled water. The working solutions of desired concentrations were made by suitable dilution of the stock solution with distilled water. Deionized water was used for the preparation of all aqueous solutions.
Synthesis of Fe3O4/Talc nanocomposite
The material is synthesized by the coprecipitation-ultrasonication method on the talc layers in an inert atmosphere, with the 1:2 ratio of Fe2+/Fe3+. The Fe3O4/Talc nanocomposite was prepared by mixing FeCl3.6H2O and FeCl2.4H2O with a ratio of 2:1 into 100 ml of distilled water. Adding Talc to the solution and vigorously stirring by a magnetic stirrer for 120 minutes. The sonication of the reaction mixture was then performed by an ultrasonic probe (Sonics & materials – VCX500; 500 w, 20 kHz), 20 mL of 25–28% NH3 solution was dropped gradually.
These procedures resulted in the formation of black precipitates of Fe3O4/Talc nanoparticles that was washed with distilled water several times until it reached the neutral pH, followed by washing twice with ethanol before dried in an oven at a temperature of 343 K. Figure 1 describes preparation process of the nanocomposite.

The procedure for preparation of Fe3O4/Talc nanocomposite.
The structure of materials was characterized by X-ray diffraction patterns of the solid powders on a P’Pret Pro – PANalytical X-ray diffractometer at 1,8kW (40 mA/45 KV) using Cu Kα (λ=1.5406 Å) radiation. FTIR spectra of the solid samples were recorded by KBr pellet method using a Bruker FTIR spectrometer. FESEM measurements were carried out using a Hitachi S-4800, EMAX operating at 20 keV. The minimal amount of solid sample was dispersed in ethanol and small drops were placed on an aluminium grid. The grid was dried from 1–2 h in a vacuum over at 40°C prior to the FESEM studies. Magnetic measurements of the solid samples were performed at room temperature (25°C) using a Magnet B-10 Vibrating sample magnetometer (VSM).
Nitrogen adsorption-desorption isotherms were performed at 77 K in Tristar 3000-Micromeritics equipment, USA, using the static adsorption procedure. Samples were degassed at 80°C and 10−6 Torr for minimum 12 h prior to analysis. BET surface areas were calculated from the linear part of BET plot according to IUPAC recommendation, pore size distributions of the samples were calculated via the conventional BJD model.
Batch adsorption procedure
Hexavalent chromium (Cr(VI)) adsorption was evaluated by the batch method, which permits to determinate of influence of parameters on the adsorption process easily. The batch adsorption experiments were carried out to investigate the influence of major affecting parameters like adsorbent dosage, pH, contact time, initial hexavalent chromium concentration and temperature on the amount of adsorption capacity. In the batch method, a fixed quantity of Fe3O4/Talc nanocomposite is mixed continuously with a specific volume of Cr(VI) solution and constantly shakened at 303 ± 1 K, until the equilibrium was reached. The contents in the flask were shaken in incubator Orbital shaker (OXYOS) at 120 r.min−1. After shaking, the solutions were filtered through a Whatman No. 42 filter paper. The filtrate was collected in polyethylene tubes and diluted before analysis. During the study, the adsorbent dosage varied from 0.5 to 6.0 g.l−1, pH from 4.0 to 9.0, the initial Cr(VI) concentration from 10 to 30 mg.l−1, the temperature from 293 to 313 K and the contact time from 0 to 120 min. The pH of Cr(VI) solution was adjusted using either 0.1 N HCl or 0.1 N NaOH solutions to the required value.
For adsorption isotherms, a series of 100-ml flask glass were filled with 50 mL Cr(VI) solution of varying concentrations (10–30 mg.l−1), maintained at the desired temperature and pH. After the amount of Fe3O4/Talc nanocomposite used for Cr(VI) solutions was 2.0 g.l−1. The optimum uptake time 60 min the concentrations of chromium ion was calculated by taking the difference in their initial and final concentrations. In each experiment constant shaking of the solution was performed by using a shaker. All the experiments were repeated three times and average values were reported. The standard deviation was found to be ±2.0%.
Thermodynamic studies of Cr(VI) adsorption onto the Fe3O4/Talc nanocomposite were also carried out at three temperatures (297, 303 and 313 K). Kinetic studies of adsorption by Fe3O4/Talc nanocomposite were also carried out at four initial chromium concentrations from 10 to 25 mg.l−1 at 303 K where in the extent of adsorption was analyzed at regular time intervals.
Analysis of Cr(VI) ions
The concentration of Cr(VI) ions in the sample was determined by UV visible spectrophotometer with GENESYS 10S UV-Vis using 1,5-Diphenylcarbazide in acidic solution (H3PO4). The absorbance of the purple colored complex was measured at 540 nm wavelength (Clesceri et al., 1998).
Adsorption capacity of Fe3O4/Talc nanocomposite was calculated by the formula (Ngah and Fatinathan, 2010):

Where, q is the amount of Cr(VI) ion adsorbed per unit mass of adsorbent (mg.g−1), V is the volume of solution (l); m is the mass of adsorbent (g); C0, Ct are Cr(VI) concentrations in the initial solution and at time t respectively (mg.l−1).
The Cr(VI) ion removed by adsorbent was calculated by the formula:
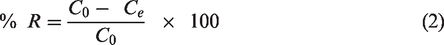
R is the percentage of Cr(VI) adsorbed by Fe3O4/Talc nanocomposite and Ce is Cr(VI) equilibrium concentrations (mg.l−1).
Results and discussion
Characterization of Fe3O4/Talc nanocomposite
The resulted brown-black precipitate can be attracted by a permanent magnet (Figure 1) that proved the formation of Fe3O4 crystals onto Talc. Characterization of the material reveals X-ray, FESEM-EDX, BET, FT-IR and VSM measurements. The X-ray patterns (a) and FT-IR (b) of synthesized material are shown in Figure 2.

X-ray patterns (a) and FT-IR (b) of Talc and Fe3O4/Talc nanocomposite.
Figure 2(a) depicts the powder X-ray diffraction (XRD) patterns of the Talc substrates and Fe3O4/Talc nanocomposite samples. The position and relative intensity of all diffraction peaks in both XRD patterns are fully matched. The peaks related to talc are observed at 2θ = 34.41°, 36.10°, 40.53°, 42.69°, 48.61°, 54.52°, 60.46°, 66.48°, 70.04° and 72.39°.
The Fe3O4/Talc nanocomposite samples, all diffraction peaks were in agreement with the standard card of Fe3O4 (00–072-2303). It is worth noting that the main peak (311) of Fe3O4/Talc nanocomposite samples is clearly the characteristic peak of magnetite (Daou et al., 2006). However, all the peak positions at 18.2 (111), 35.4 (311), 57.2 (511) and 62.6 (440) are consistent with the standard X-ray data for the magnetite phase of the Fe3O4/Talc nanocomposite. The change in the d spacing of Talc after interaction with Fe3O4 nanoparticles is negligible and these signify that the nanoparticles are on the external surface of the Talc (Kalantari et al., 2013). Figure 2 showed that, the original d-spacing (ds) of Talc at 2θ 9.35 degrees was 0.95 nm, which gradually decreased to 0.91 nm at 2θ 9.50 degrees by the formation of Fe3O4 nanoparticles on the surface of Talc layers.
Figure 2(b) shows the FT-IR spectra of Talc substrate and Fe3O4/Talc nanocomposite samples. There are three obvious peaks at 3633, 3429–34,271,630 cm−1 and 450–669 cm−1 in the spectrum of Talc and the synthesized nanoparticles. The peaks basically represent the vibrations of Mg3-OH, O-H stretching, H-O-H bending and Mg-O, Si-O-Mg, Si-O, respectively (Kalantari et al., 2013; Li et al., 2015). However, the peak areas of Mg-O bonds (451 cm−1), Si-O-Mg bonds (529 cm−1) and Si-O (669 cm−1) were all decreased in the spectrum of Fe3O4/Talc nanocomposite. It is because the ordering and bonding effects were influenced in the layer structure, since some of the Mg-O bonds were broken with radiant energy from the microwave synthesis system, making the binding force weaken in the interlayer. Finally, the Mg+ and Si-O− were exhibited [Li S-F et al., 2015]. The peak at 580 cm−1 in the spectrum of Fe3O4/Talc nanocomposite was attributed to Fe-O stretch vibrations of nano Fe3O4 (Willis et al., 2005), confirming the nature of the iron oxide nanoparticles. These FT-IR and X-ray results imply that the nanoparticles were synthesis successful.
The surface properties, porosity or pore size is one of the factors to evaluate the adsorption capacity of the material. Figure 3 is the FESEM image, BET and VMS of Fe3O4/Talc nanocomposite.

(a) FESEM images of the Talc and Fe3O4/Talc nanocomposite; (b) Nitrogen adsorption - desorption isotherms of Fe3O4/Talc nanocomposite; (c) M-H curves for Fe3O4/Talc nanocomposite.
Figure 3(a) showed the Talc surface material has particle sizes in the range of 30–50 µm, cubic particles are less uniform and have smooth surface. Nanocomposite materials at different magnifications, had distribution of Fe3O4 nanoparticles with dimensions less than 30 nm. Then, the synthetic nanocomposite material will give a larger surface area than the base material, which will be shown through the measurement of specific surface area by BET.
Figure 3(b) showed the Fe3O4/Talc nanocomposite samples which give adsorption-desorption isotherm of the intermediate form between III and IV with the appearance of H3 hysteresis loop, rod-shaped and letter-shaped according to IUPAC classification (Thommes et al., 2015). This allows the prediction that synthetic nanocomposite materials contain both macropores and mesopores, in which mesopores are more numerous. The pore size distribution obtained from the BJH analysis, the BJH curve shows narrow and intensity peaks in the pore size distribution curve. The hole size distribution curve shows that appear in a range 1 to 150 nm. The specific surface area of talc surface material is 3.45 m2.g−1 which is much smaller than that of the nanocomposite material, which reaches 77.92 m2.g−1. In particular, macropore area of the nanocomposite Smacro = SBET – SBJH,ads – Smicro = 8.27 m2.g−1; the pore diameter averages 11.8 nm. A similar types of Fe3O4 nanocomposite were also reported by some other workers (Chávez-Guajardo et al., 2015; Cui et al., 2015).
The magnetic properties of materials are assessed by the method of vibrating-sample magnetometry. The results of saturation magnetization of Fe3O4/Talc nanocomposite was 38.06 emu.g−1, this indicates that the survey sample is superparamagnetic, the iron oxide particles are distributed with nano size on the soluble surface. Therefore, the material is convenient to separate from the aqueous solution after adsorption with the help of external magnetic fields (Lv et al., 2014).
Adsorption studies

Effect of pH (adsorbent dosage = 0.1 g/100 ml; [Cr(VI)] = 10 and 20 mg/l; t = 60 min; agitation 120 rpm).

Effect of contact time (adsorbent dosage = 0.1 g/10 ml; pH = 5.0; agitation 120 rpm).

Effect of adsorbent dosage ([Cr(VI)] = 10 mg/l; t = 60 min; agitation 120 rpm, pH = 5.0, T = 303 K).

Effect of Cr(VI) concentration (t = 60 min; agitation 120 rpm, pH = 5.0, T = 303 K).
Figure 7 showed that, the qe increases from 4.45 to 7.17 mg.g−1 and removal efficiency decreases from 89.13 to 47.86%, respectively Cr(VI) initial concentration increases from 10 to 30 mg.l−1. When, higher initial concentrations, the active sites of prepared adsorbent would be surrounded with more Cr(VI) ions in the solution, the equilibrium adsorption capacity of Fe3O4/Talc nanocomposite increases with the increase of the Cr(VI) ion concentration which enhances the adsorption process. In addition, the removal percentage decreases by the increase in metal initial concentration. At low initial concentrations, the ratio of initial number of chromium ions to the accessible active sites of adsorbent is low; therefore, the removal efficiency of Cr(VI) is higher and at higher concentrations, further residual Cr(VI) ions remain in the aqueous solution (Radnia et al., 2012; Aydın et al., 2008).
Adsorption isotherm modeling
To understand the adsorption mechanism and surface characteristics of the Fe3O4/Talc nanocomposite, the mathematical models developed by Langmuir and Freundlich have been applied to the data. The linear equations of Langmuir and Freundlich isotherm models can be described as equation (3) (Ossman et al., 2014).


Where

Langmuir isotherm and Fruendlich isotherm (adsorbent dosage = 0.1 g/100 ml; pH 5.0; t = 60 min; agitation 120 rpm).
The related values of parameters and constants for these isotherms are presented in Table 1. The values of regression coefficient obtained from these models were used as the fitting criteria to find out these isotherms.
Langmuir and Freudlich isotherm constants for the adsorption of Cr(VI) on Fe3O4/Talc nanocomposit at different temperatures and pH 5.0.

Table 1 showed both plots depiced the linearized form of the isotherms at all temperatures and extremely high correlation coefficients (R2 = 0.97 to 0.99), thus indicating both monolayer and heterogeneous surface conditions. Also, from the Langmuir adsorption constant qe, the adsorption capacity of Cr(VI) on Fe3O4/Talc nanocomposite was observed as 7.17, 7.47 to 35.84 mg.g−1 with the rise in temperature from 293, 303 to 313 K, respectively. This result also showed that the value of RL, and the separation factor fell in the range of 0 to 1 which clearly showed that the adsorption process is favorable. Freundlich constant also varied from 1 to 10 which again proved that the adsorption is favorable. At pH = 5, the existence of Cr(VI) in solution was HCrO4− (Dubey and Gopal, 2007; Gupta et al., 2010). So, the formation and mechanism for the adsorption of Cr(VI) onto Fe3O4/Talc nanocomposite was described at Figure 9.

Illustration of the prepared Fe3O4/Talc nanocomposite and mechanism for the adsorption of Cr(VI) onto Fe3O4/Talc nanocomposite.
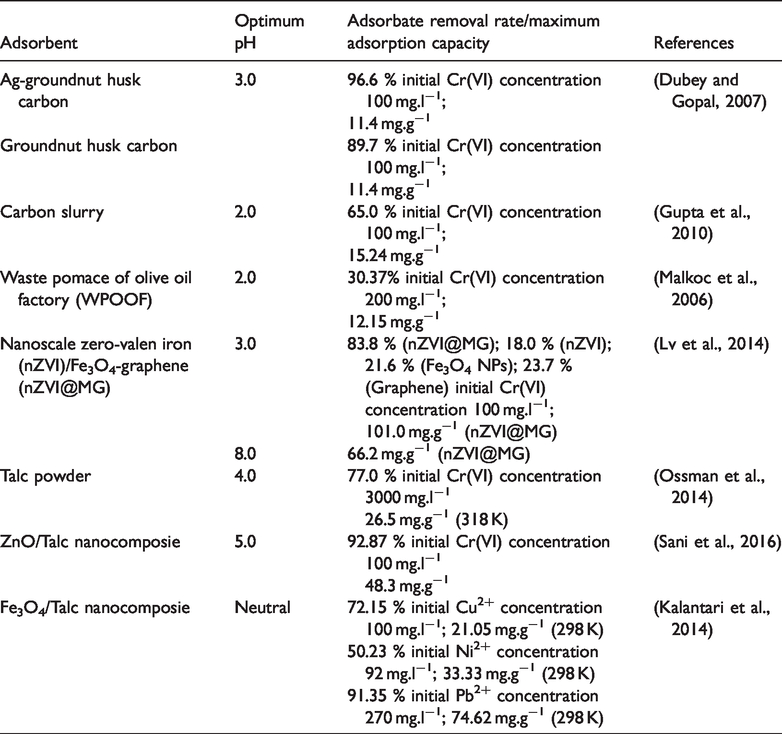
Table 2 compares maxium adsorption capacities and rate of removal obtained in this study with some other values reported in the article The adsorption capacity for Cr(VI) using Fe3O4/Talc nanocomposite, a low-cost adsorbent from the ferilizer industry. The adsorption capacity for Cr(VI) of Fe3O4/Talc nanocomposite is of the same order of magnitude has been found using similar adsorbents.
Removal rate/adsorption capacity of heavy metal ions onto various adsorbents.
Adsorption kinetics
Kinetics of the adsorption process is studied based on the pseudo-first-order adsorption kinetics equation (B1) in linear form:


Where: k2 (mg/g.min) is the rate constant of the adsorption kinetics process (Ho, 1995; Yuh-Shan, 2004). Intraparticle diffusion kinetic equation:
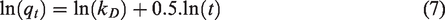


The kinetic models of adsorption of Cr(VI) ion on the Fe3O4/Talc nanocomposite (adsorbent dosage = 0.1 g/100 ml; pH 5.0; T = 303 K; agitation 120 rpm).
Kinetic parameters for the removal of Cr(VI) onto Fe3O4/Talc nanocomposite.
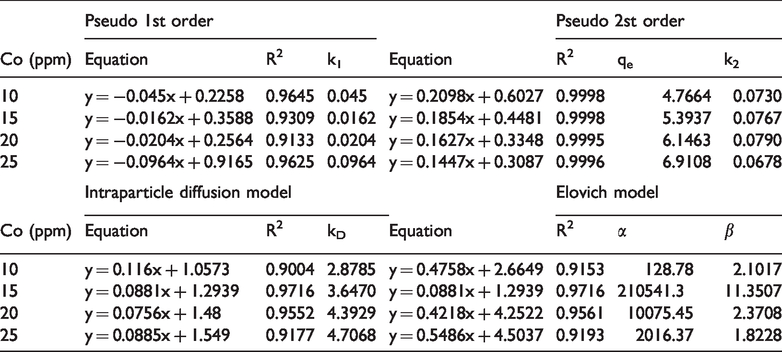
Table 3 showed the values of correlation coefficient R2 for the Pseudo-second-order adsorption model which is relatively high (R2 > 0.999), furthermore, the values of the k constant are almost unchanged, which indicates that the speed constant does not depend on concentration. This proves that the adsorption process depends on the number of adsorption centers on the surface and the adsorbate is the Cr(VI) ion.
Thermodynamic study
In this study, the thermodynamics of adsorption of Cr(VI) onto Fe3O4/Talc nanocomposite, thermodynamic constants such as: enthalpy change ΔHo, free energy change ΔGo and entropy change ΔS can be related to the Langmuir isotherm constant by the following equations (Gupta et al., 2004).


Thus, a plot of ln(KL) versus 1/T should be a straight line. ΔHo and ΔSo values could be obtained from the slope and intercept of this plot. The values of these parameters are given in Table 4. Figure 11 show thermodynamic data of adsorption of Cr(VI) onto Fe3O4/Talc nanocomposite.
Thermodynamic parameters for the adsorption of Cr(VI) on Fe3O4/Talc nanocomposite at different temperatures.

aMeasured between 293 K and 303 K.

Thermodynamic analysis (adsorbent dosage = 0.1 g/100 ml; pH 5.0; t = 60 minutes; agitation 120 rpm).
Table 4 showed that the values of
Conclusion
The Fe3O4/Talc nanocomposite was synthesized with the Talcum powder Phu Tho – Viet Nam by the coprecipitation-ultrasonication method. X-ray, FTIR, FESEM, BET and VSM analyses were performed for characterizing the synthesized nanocomposite. The surface area of the nanoparticles was determined to be 77.92 m2.g−1 with an average diameter of 11.8 nm and the saturation magnetization of 38.06 emu.g−1. The peak positions at 18.2 (111), 35.4 (311), 57.2 (511) and 62.6 (440) are consistent with the standard X-ray data for the magnetite phase of the Fe3O4/Talc nanocomposite. The original d-spacing (ds) of talc at 2θ 9.35 degrees was 0.95 nm, and 0.91 nm at 2θ 9.50 degrees by the formation of Fe3O4 nanoparticles on the surface of Talc layers. The peak at 580 cm−1 in the spectrum of Fe3O4/Talc nanocomposite was attributed to Fe-O stretch vibrations, confirming the nature of the iron oxide nanoparticles. These FT-IR and X-ray results imply that the nanoparticles were synthesis successful.
The batch studies experimental results indicate that the Fe3O4/Talc nanocomposite is an effective adsorbent of Cr(VI) ion from the aqueous solution. The adsorption equilibrium data fitted very well to the Langmuir and Freundlich adsorption isotherm models. The kinetic data showed that the adsorption process followed the pseudo-second order kinetic model. The maximum adsorption capacities of 35.84 mg/g occurred at pH 5.0 and 313 K, with adsortion dosage is 2 g.l−1. The adsorption is the process of chemical and exothermic. These results permit us to conclude that Fe3O4/Talc nanocomposite is a promising low-cost adsorbent for Cr(VI) removal from wastewater and can be applied in a magnetically-assisted water treatment technology.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
