Abstract
Hexavalent chromium (Cr(VI) is a toxic and persistent pollutant with serious environmental and health impacts. This study evaluated the biosorption potential of locally available
Keywords
Introduction
Chromium (Cr) is among the most abundant elements in the Earth's crust and it commonly exists in three oxidation states: Cr(0), Cr(II) and Cr(III). Of these, Cr(III) is the most stable and naturally prevalent form. However, industrial activities have increasingly contributed to the formation of Cr(0) and the more toxic hexavalent chromium species, Cr(VI). 1 Cr(VI) compounds are classified by the International Agency for Research on Cancer as Group 1 carcinogens due to their high toxicity and strong cancer-causing potential. 2
Chromium exposure arises from both natural and anthropogenic sources. Natural contributors include wind-blown dust, sea spray, volcanic eruptions and forest fires. In contrast, human activities – such as fossil-fuel combustion, sewage sludge disposal, phosphate fertiliser application, mining, chrome plating, pigment production, electroplating, textile processing and alloy manufacturing – are major sources of environmental chromium contamination. 3 Among these, leather tanneries are particularly significant producers of Cr(VI). 2 Due to its cost-effectiveness, rapid processing, vibrant leather colouration and enhanced durability, chromium-based tanning – especially using chromium sulphate (Cr2(SO4)3) – is widely adopted by the tanning industry.2,4 Although approximately 60–80% of the chromium is retained by the leather, 4 the remainder is discharged as wastewater, posing a serious environmental threat. This residual Cr(III) in tannery effluent can oxidise to Cr(VI) under conditions involving manganese dioxide (MnO2), 5 elevated temperatures, ultraviolet irradiation or the presence of dichromate impurities during Cr(III) synthesis.6,7
Cr(VI) is recognised as a potent carcinogen associated with severe health effects including diarrhoea, dermatitis, epigastric pain, gastrointestinal disturbances, nausea, vomiting and skin ulceration. 8 It has also been implicated in pulmonary and respiratory tract cancers 9 and presents a serious threat to aquatic ecosystems.10,11 The high toxicity of Cr(VI) is attributed to its ability to readily penetrate biological membranes and its strong oxidising potential. 12 Once inside cells, Cr(VI) undergoes a series of reductions to form Cr(III), generating reactive intermediates that damage vital organs such as the kidneys, liver and lungs. Its water solubility exacerbates these impacts by enabling systemic distribution. 2
As per the World Health Organization 13 and the United States Environmental Protection Agency, 14 permissible limits for Cr(III) and Cr(VI) in wastewater are set at 5.00 and 0.05 mg L−1, respectively. Exceeding these limits mandates remediation before effluent discharge, presenting a compliance challenge for industries, especially those in metal finishing and mineral processing.
Multiple techniques have been explored to remove Cr(VI) from aqueous media, including chemical precipitation,
15
coagulation,
16
electrodialysis,
17
membrane filtration,
18
ion exchange
19
and adsorption.
20
However, many of these methods are hindered by high-operational costs, excessive sludge generation and dependence on skilled labour. In contrast, adsorption emerges as an efficient, scalable and cost-effective approach capable of removing over 90% of metal ions.
21
Previous studies have demonstrated the potential of diverse agricultural biosorbents such as
To the best of our knowledge, this is the first study to investigate the biosorption potential of
Beyond introducing these novel biosorbents, our study systematically evaluates the influence of key operational parameters – including pH, adsorbent dose, contact time, initial Cr(VI) concentration and agitation speed – on Cr(VI) removal efficiency. We further apply adsorption isotherms and kinetic models to elucidate the underlying mechanisms, providing a comprehensive understanding of their adsorption behaviour. This dual focus on novel material selection and mechanistic insight distinguishes our work from prior studies and contributes meaningfully to the field of environmental nanotechnology and biosorption. By bridging material innovation with practical applicability, the study underscores the noble potential of indigenous plant resources in addressing pressing environmental challenges.
Experimental part
Materials and chemicals
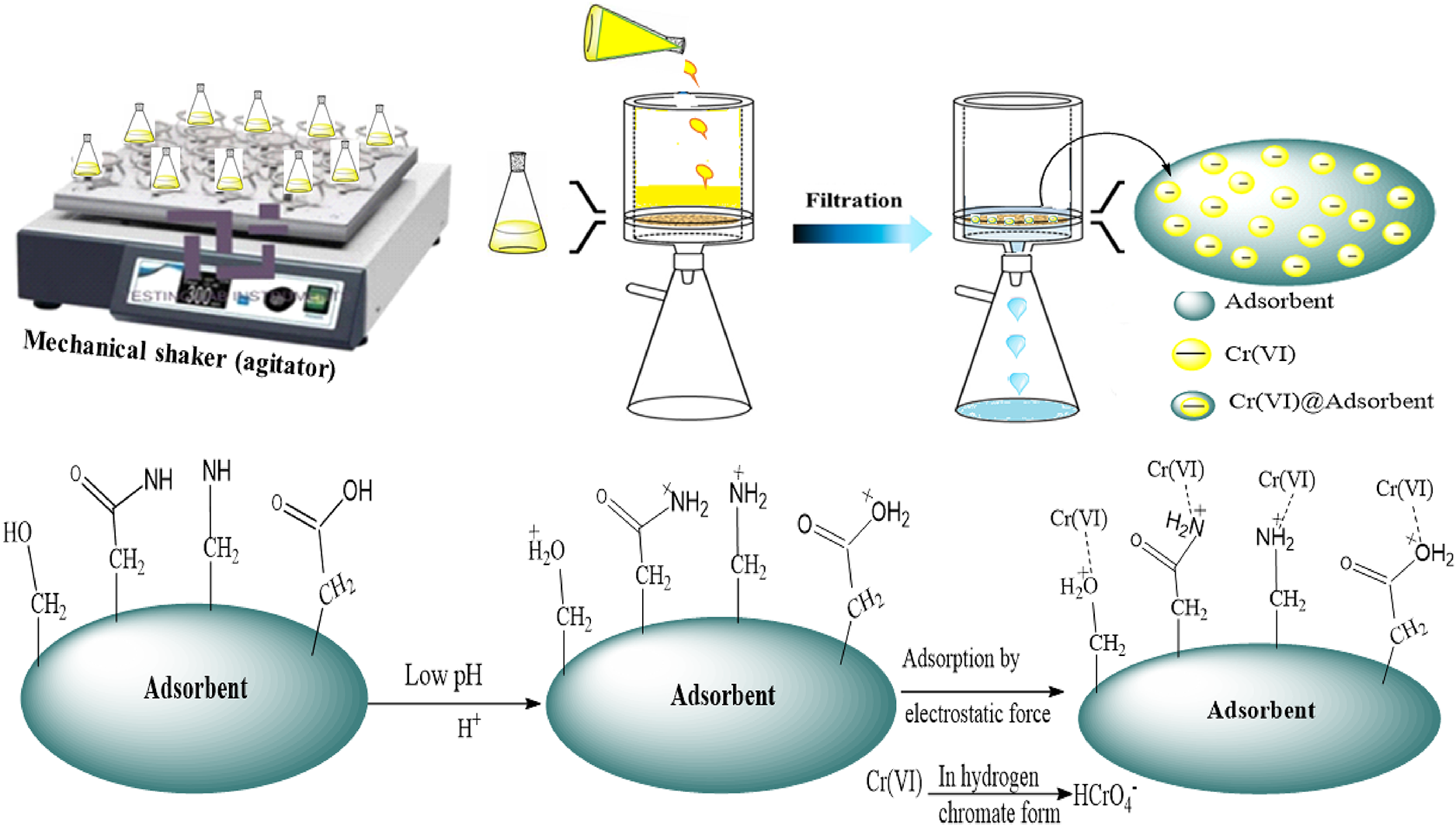
Potassium dichromate (K2Cr2O7, 99.9%), sodium acetate (CH3COONa), acetic acid (CH3COOH), hydrochloric acid (HCl) and sodium hydroxide (NaOH) were used for solution preparation and pH adjustment. pH measurements were recorded using a Model 300 pH meter (OHAUS, Zhengzhou, China). Biosorption experiments were conducted with continuous agitation using an HZ-300 rotary mechanical shaker (China), and particle size uniformity was maintained by passing the adsorbent through a 0.5 mm mesh sieve. All chemicals were of analytical reagent grade and sourced from BDH Chemicals (Mumbai, India). Distilled water was used throughout all experimental procedures to ensure consistency and purity.
We performed quantitative analysis of hexavalent chromium (Cr(VI)) concentrations using a BUCK Scientific 210VGP Flame Atomic Absorption Spectrometer (FAAS; USA). The instrument was equipped with an air-acetylene flame and operated at a wavelength of 357.9 nm, corresponding to the resonance absorption line specific to chromium.
Functional group analysis was conducted using a PerkinElmer Spectrum 65™ FT-IR spectrophotometer to identify chemical moieties involved in Cr(VI) binding. Surface morphology and microstructural features of the biosorbents prior to Cr(VI) adsorption were examined using a LEO 1525 Scanning Electron Microscope (SEM). Specific surface area and porosity characteristics were evaluated through Brunauer–Emmett–Teller (BET) analysis using a Quantachrome TouchWin™ surface area analyzer (Quantachrome Instruments, USA).
Adsorbent selection, collection and preparation
The selection of
Leaves of
Preparation of solutions
An acetate buffer solution (1.0 M, pH 4.75) was prepared by mixing 50.0 mL of 5.0 M acetic acid with 83.33 mL of 3.0 M sodium acetate in a 500 mL volumetric flask, followed by dilution to the mark with distilled water. A standard stock solution of Cr(VI) (1000 mg L−1) was prepared by dissolving 2.28 g of potassium dichromate (K2Cr2O7), equivalent to 1.0 g of Cr(VI), in distilled water and diluting to 1000 mL in a volumetric flask. An intermediate solution containing 100 mg L−1 Cr(VI) was subsequently prepared by diluting 50 mL of the stock solution to 500 mL with distilled water in a separate volumetric flask.
Standard solutions and measurements
Working standard solutions of Cr(VI) at concentrations of 0.5, 1.0, 2.0, 4.0, 6.0, 8.0 and 10 mg L−1 were prepared for method calibration (Figure 1). Batch adsorption experiments were carried out in 100 mL volumetric flasks to evaluate the effects of pH, contact time, initial metal ion concentration and adsorbent dosage. Each Cr(VI) solution was mixed with a pre-determined mass of 
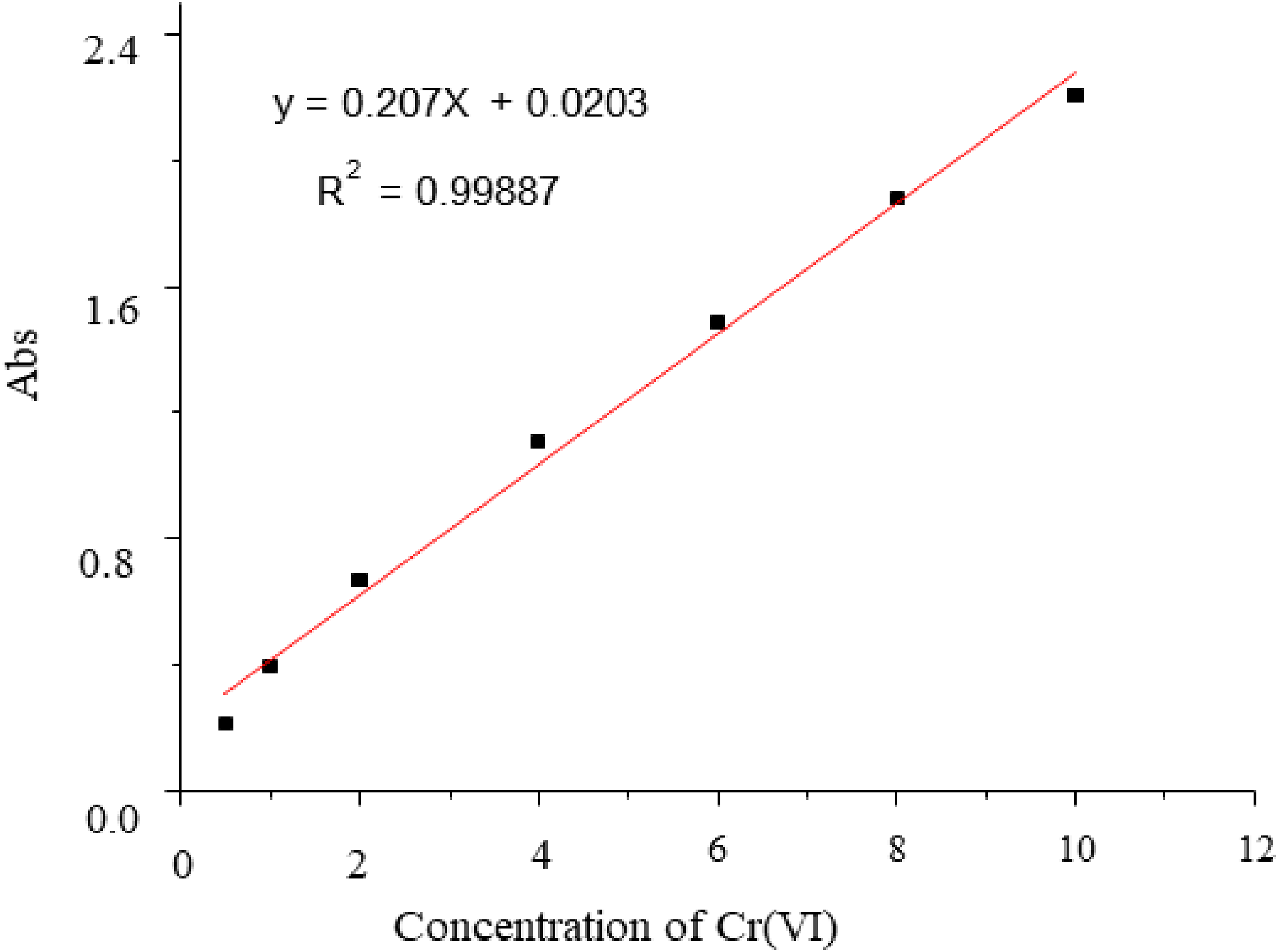
Calibration curve of Cr(VI).
Fourier transform infrared spectral analysis
Fourier transform infrared (FTIR) spectra were recorded using a PerkinElmer Spectrum 65™ FT-IR spectrophotometer over the range of 400–4000 cm−1. The analysis was performed to identify and compare functional groups present in
SEM analysis
The LEO 1525 SEM, equipped with a field emission gun and Gemini lens system, was utilised for high-resolution imaging and microstructural analysis. Imaging was conducted at an accelerating voltage of 15 kV, achieving a resolution of up to 1.5 nm, suitable for detailed surface morphology characterisation.
BET analysis
BET surface area analysis was performed using a Quantachrome TouchWin™ surface area analyzer (Quantachrome Instruments, USA), employing nitrogen adsorption at 77 K. Prior to analysis, samples were degassed under vacuum by heating to 150 °C at a rate of 10 °C min−1, followed by a 240-min hold to ensure complete removal of surface contaminants. Surface area was calculated using the standard multi-point BET method.
Batch adsorption experiments
The chromium concentration of the raw
Batch adsorption experiments were conducted at room temperature to evaluate the performance of
pH is a critical factor influencing the adsorption capacity of biosorbents for Cr(VI) removal from aqueous solutions. 30 To investigate its effect, the solution pH was systematically adjusted to 2, 3, 5, 7, 9 and 11. The impact of contact time on adsorption efficiency was examined by varying the duration from 10 to 120 min (10, 20, 40, 60, 80, 100 and 120 min), while keeping other parameters constant.
To assess the influence of initial Cr(VI) concentration, solutions were prepared with concentrations ranging from 5 to 30 mg L−1 in 5 mg L−1 increments. The effect of adsorbent dosage was evaluated by varying the mass from 1.5 to 4.0 g under constant experimental conditions. Additionally, the agitation speed of the mechanical shaker was optimised for both biosorbents to ensure uniform mixing and enhance mass transfer during the adsorption process.
Adsorption isotherms
To elucidate the interaction mechanisms and surface behaviour of the biosorbents during Cr(VI) removal, adsorption isotherm models were applied to equilibrium data obtained across a range of initial Cr(VI) concentrations. The Langmuir isotherm was used to assess mono-layer adsorption on a homogeneous surface, while the Freundlich isotherm provided insights into adsorption on heterogeneous surfaces. These models enabled the characterisation of adsorption capacity, intensity and site distribution, thereby revealing the nature of biosorbent–metal interactions and surface heterogeneity.
Langmuir isotherm
The Langmuir isotherm model assumes mono-layer adsorption on a homogeneously distributed surface with uniform adsorption energies, where each adsorption site accommodates only one molecule of adsorbate. This model is based on the premise that once a site is occupied, no further adsorption can occur at that location and there are no interactions between adsorbed species. It is widely used to describe ideal adsorption behaviour in systems with well-defined surface properties. 31
The linearised form of the Langmuir equation is expressed as: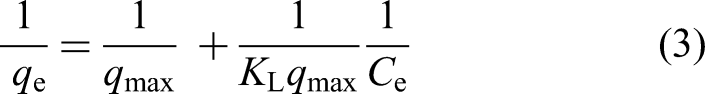
Values of
Freundlich isotherm
The Freundlich isotherm model describes adsorption on heterogeneous surfaces, where multi-layer formation and site–site interactions are possible. Unlike the Langmuir model, which assumes uniform adsorption energies, the Freundlich model accounts for surface heterogeneity and variable affinities across adsorption sites. This empirical model is particularly suitable for systems where the adsorbent surface exhibits a non-uniform distribution of active sites, making it widely applicable in environmental remediation studies involving biosorbents. 34
The linearised form of the Freundlich equation is expressed as: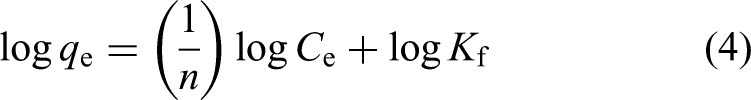
Constants 1/
Adsorption kinetics
Adsorption kinetics explains the solute's rate of adsorption and the adsorbates’ residence period on the solid–liquid interface. 35 It also provides the performance of the adsorbent used and the mass transfer mechanisms. When designing adsorption systems, it is helpful to understand the adsorption kinetics. 35 There are three steps in the adsorption mass transfer kinetic. The first step is the external diffusion which the adsorbate moves through the liquid layer and surrounding the adsorbent surface. The external diffusion is driven by the concentration difference between the adsorbent surface and the bulk solution. The second step is the internal diffusion which the adsorbate's diffusion takes place within the adsorbent's pores is referred to as internal diffusion. The third step is the adsorption of the adsorbate's in the adsorbent's active sites. 35
Pseudo-first-order and pseudo-second-order are two basic kinetic models that were used to assess the kinetic parameters of the adsorption process from variations in constant time. Equation (5) shows the expression for the pseudo-first-order rate
11
: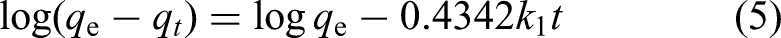
The adsorption kinetics rate equation for pseudo-second-order is expressed in the below equation
11
: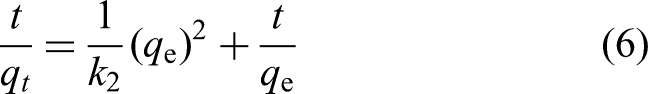
The plot of
Results and discussion
FTIR spectra analysis of adsorbents
Figure 2 presents the FTIR spectra of
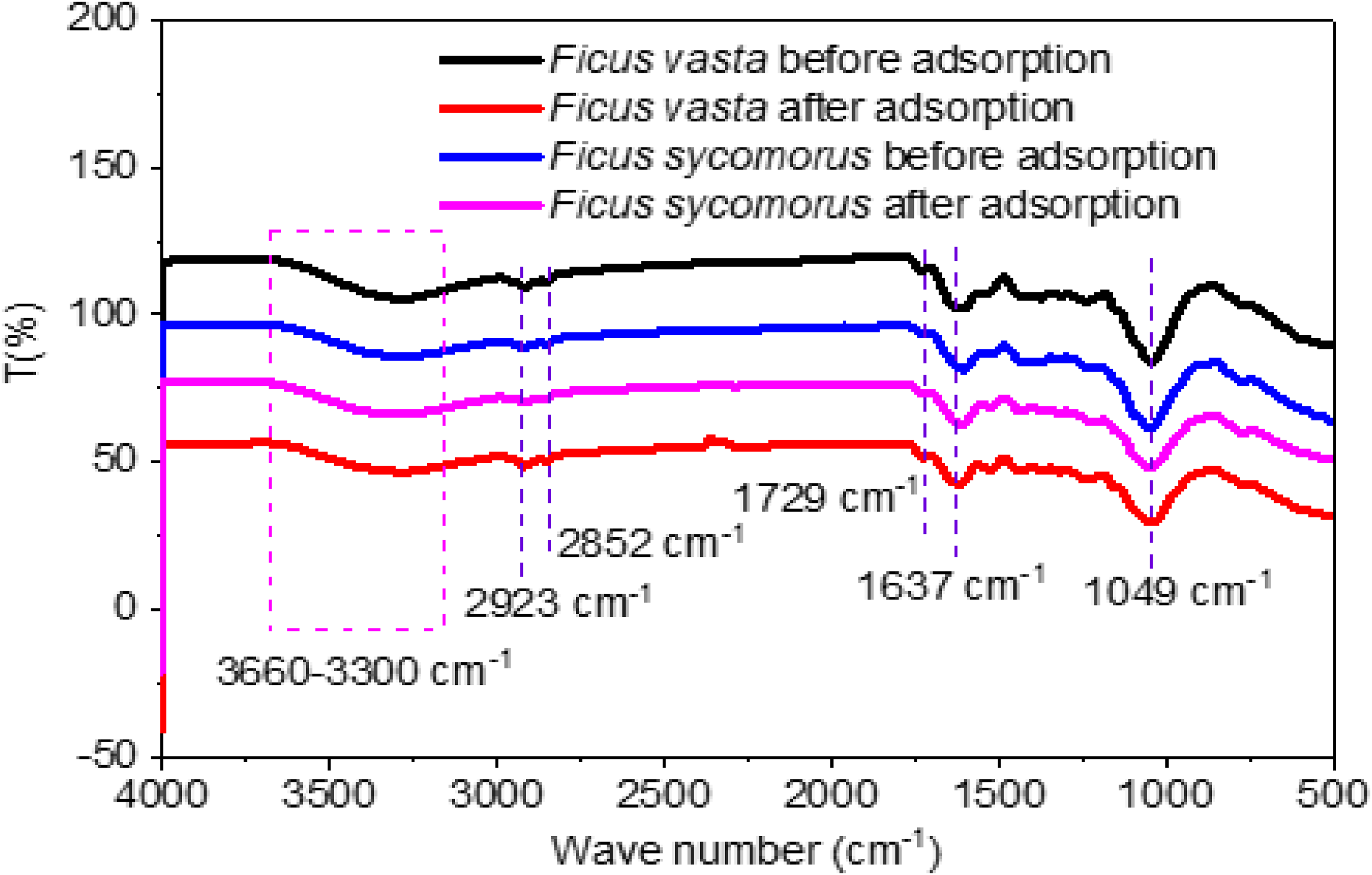
FTIR spectra of
These findings align with previous studies on natural biosorbents, where Cr(VI) reduction and subsequent binding to oxygen-rich sites occur without pronounced M–O spectral signatures.
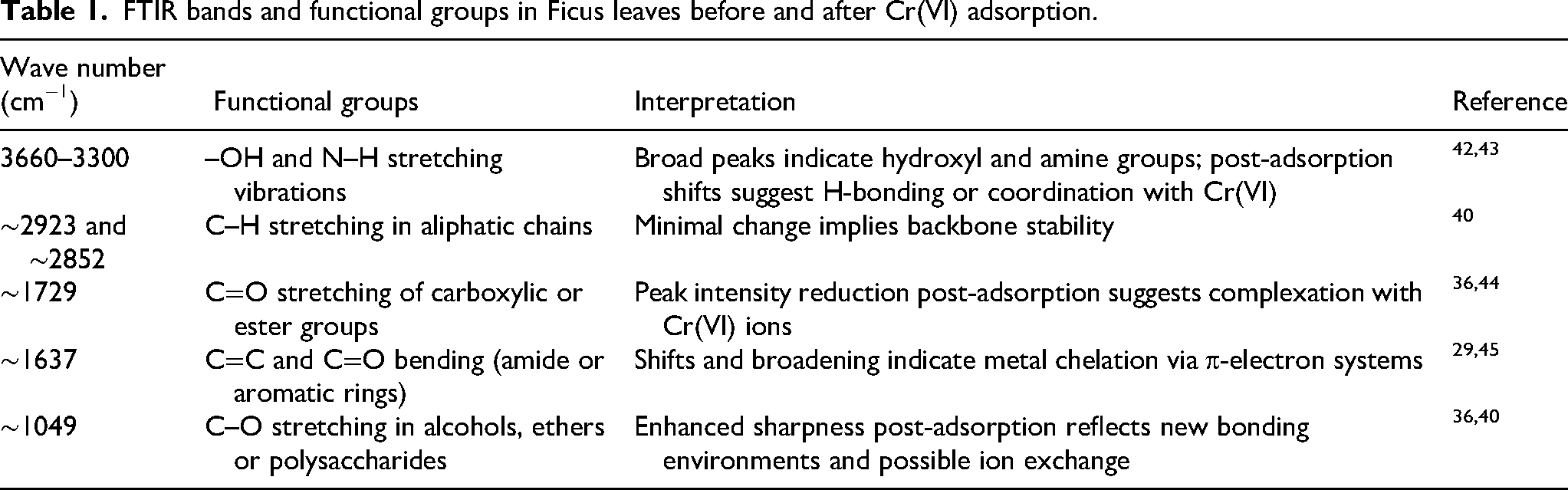
Key FTIR absorption bands and their functional group assignments for
FTIR bands and functional groups in Ficus leaves before and after Cr(VI) adsorption.
Surface area and morphology analysis
Figure 3 presents SEM micrographs of
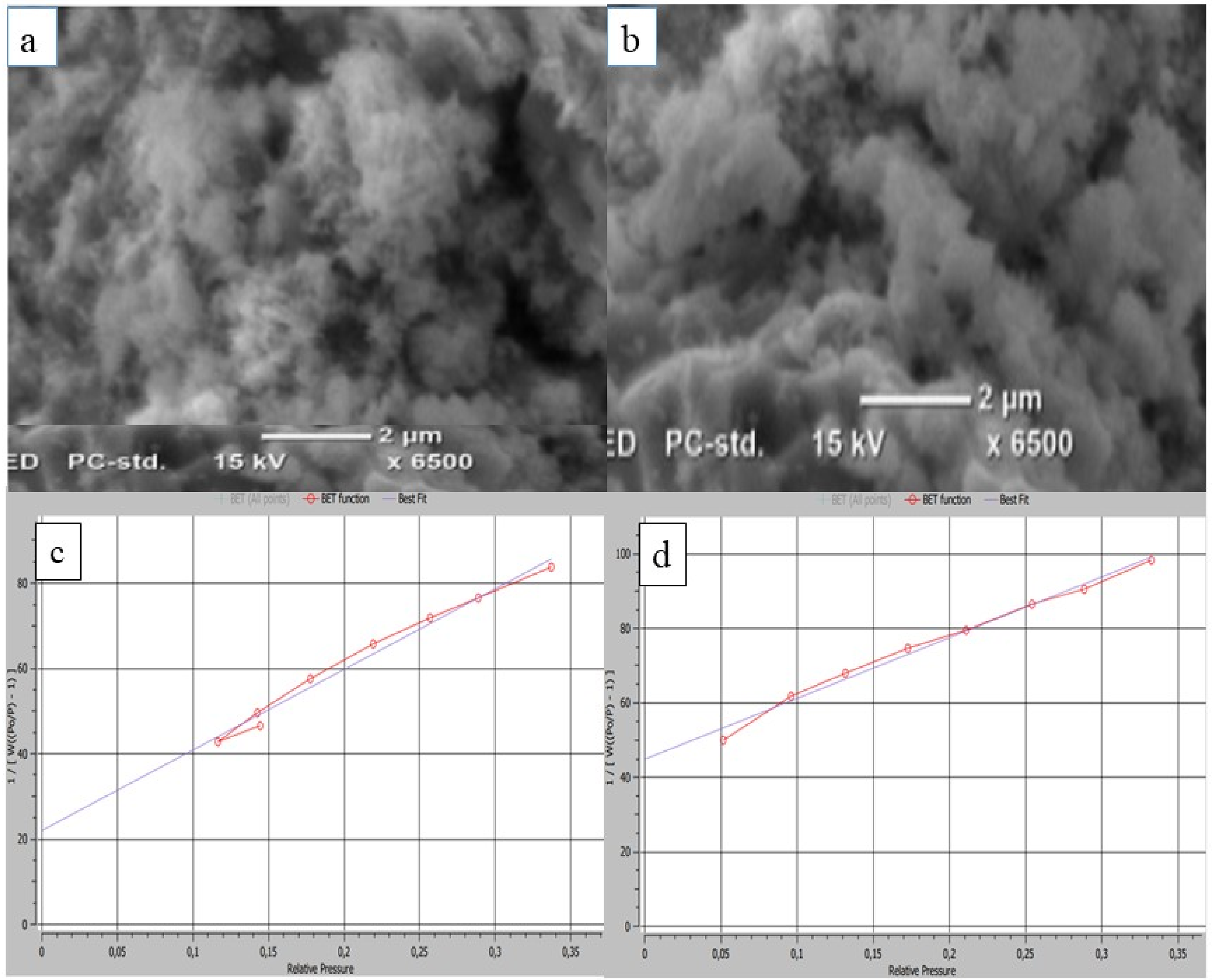
Surface characterisation of
Effects of dosage on adsorption
In this experiment, the dosages of both
As shown in Figure 4, the Cr(VI) removal efficiency increased markedly with adsorbent dosage – from 57.32 to 82.14% for
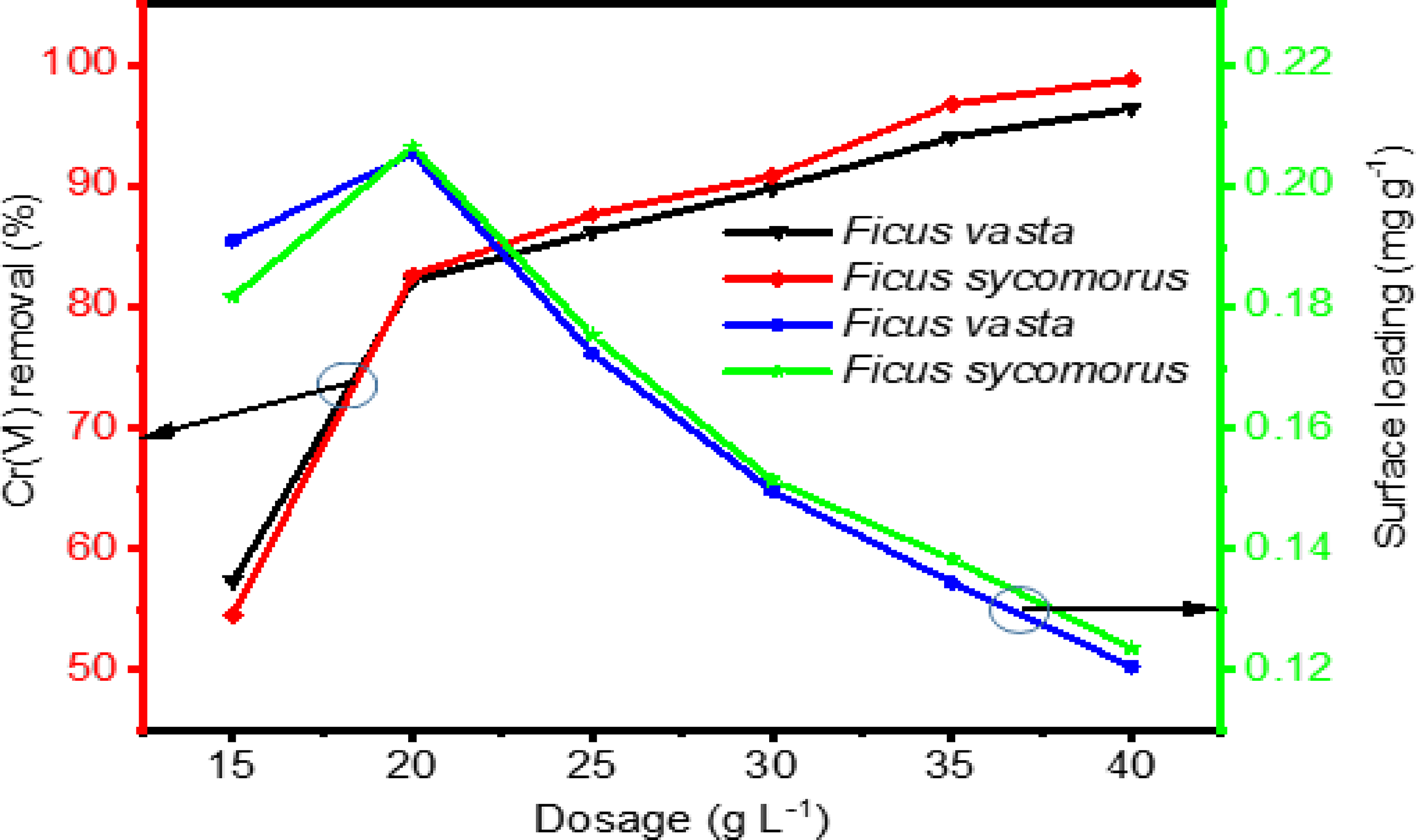
Effect of adsorbents dosage on removal of Cr (VI). Original pH 5.8, contact time (60 min), initial concentration 5.0 mg L−1, agitation speed (180 rpm).
Effect of pH
As displayed in Figure 5, the effect of pH was evaluated while keeping the other parameters – contact time, starting concentration, adsorbent dosage and agitation speed – constant at each pH value (2, 3, 5, 7, 9 and 11). The prepared acetate buffer was utilised for pH adjustment, and 0.1 M solutions of HCl and NaOH were employed to reach the desired pH level. The per cent removal of Cr(VI) ion was highest at pH 2 (88.99%) and pH 3 (91.97%) for
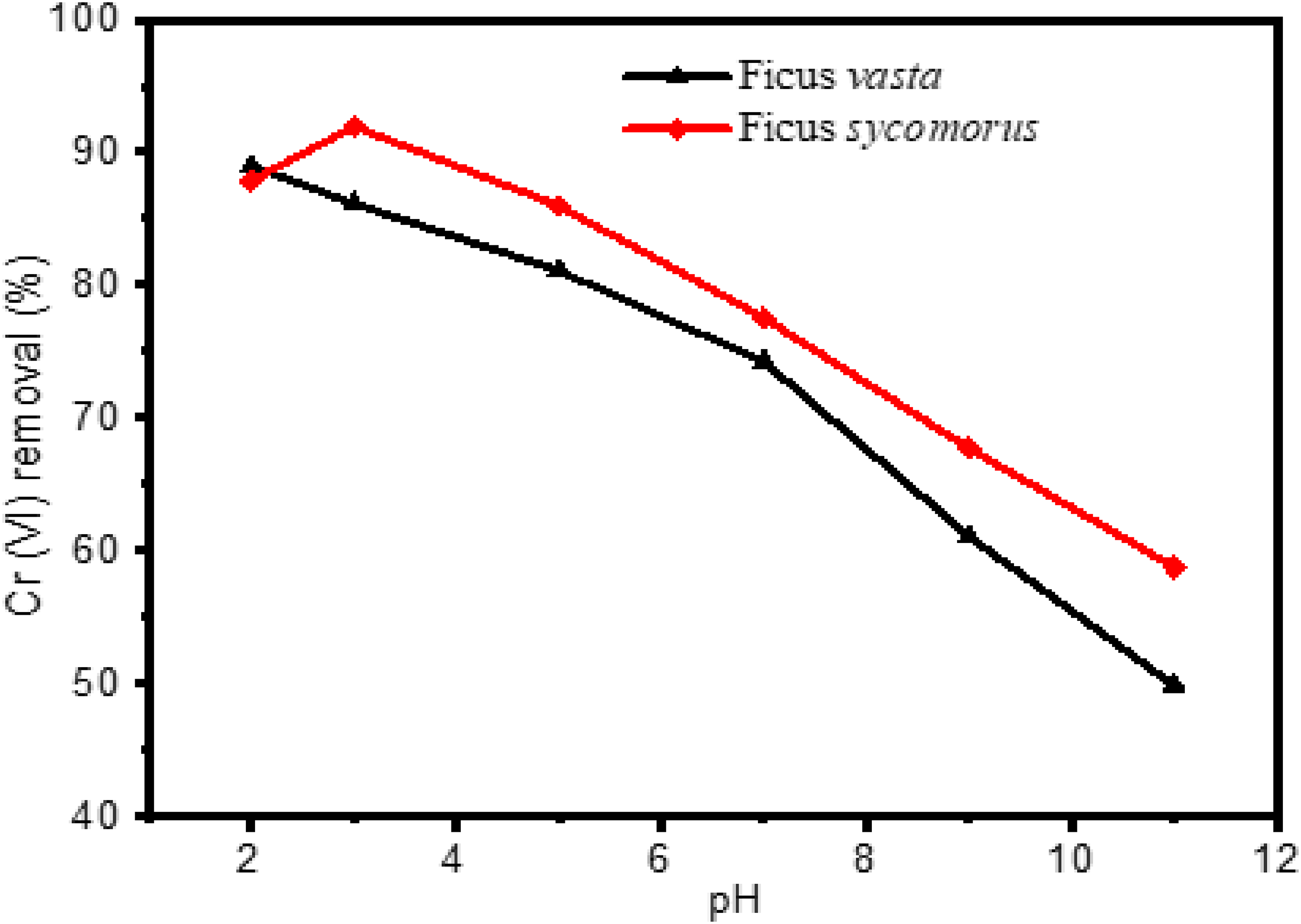
Effect of pH on removal of Cr (VI). Dose (20 g L−1), contact time (60 min), initial concentration (5 mg L−1) and agitation speed (180 rpm).
In aqueous solutions, chromium(VI) can appear as chromate (CrO42−), dichromate (Cr2O72−), hydrogen chromate (HCrO4−), hydrogen dichromate (HCr2O7−), chromic acid (H2CrO4) and dichromic acid (H2Cr2O7), depending on the pH and redox potential of the medium.54,55 HCrO4− is predominantly present at pH levels below 6, while H2CrO4 dominates below pH 1. 54 Therefore, when the pH of the solution becomes very low (pH ≤ 1), adsorption is expected to decrease due to the decline in electrostatic attraction between the positively charged adsorbent surface and the predominant neutral species of chromium (H2CrO4). 54 CrO42− becomes the predominant species at pH levels above 7.28,54 When the pH increases to higher levels (e.g. pH 11), the surface of the adsorbent acquires a net negative charge, resulting in reduced Cr(VI) adsorption due to electrostatic repulsion.
Similar results were obtained by Kebede et al. 54 using biowastes for the removal of Cr(VI) from aqueous solution. In aqueous media, hydrolysis of Cr2O72− occurs via the reaction: Cr2O72− + H2O ↔ 2HCrO4−. 56 In this study, a synthetic solution of K2Cr2O7 was used as the Cr(VI) source. HCrO4− is more abundant at pH values below 6 and is the most commonly adsorbed form of Cr(VI) under acidic conditions. At low pH, the adsorbent surface becomes positively charged due to protonation with H+ ions, facilitated by functional groups such as amino, aromatic nitro, amide, carboxyl, carbonyl and hydroxyl. This promotes the binding of negatively charged HCrO4− ions, resulting in high Cr(VI) uptake. 57
Therefore, pH 2 and 3 were selected as the optimum values for
Effect of contact time
The influence of contact time between the adsorbent (leaf powder) and the adsorbate (metal ion), which is one of the factors influencing the proportion of the metal ion being adsorbed
28
is indicated in Figure 6. Having this in our mind, the contact time was varied from 10 to 120 min in 20 min interval keeping the other variables constant for each setting. The pH was adjusted to 2 and 3 for
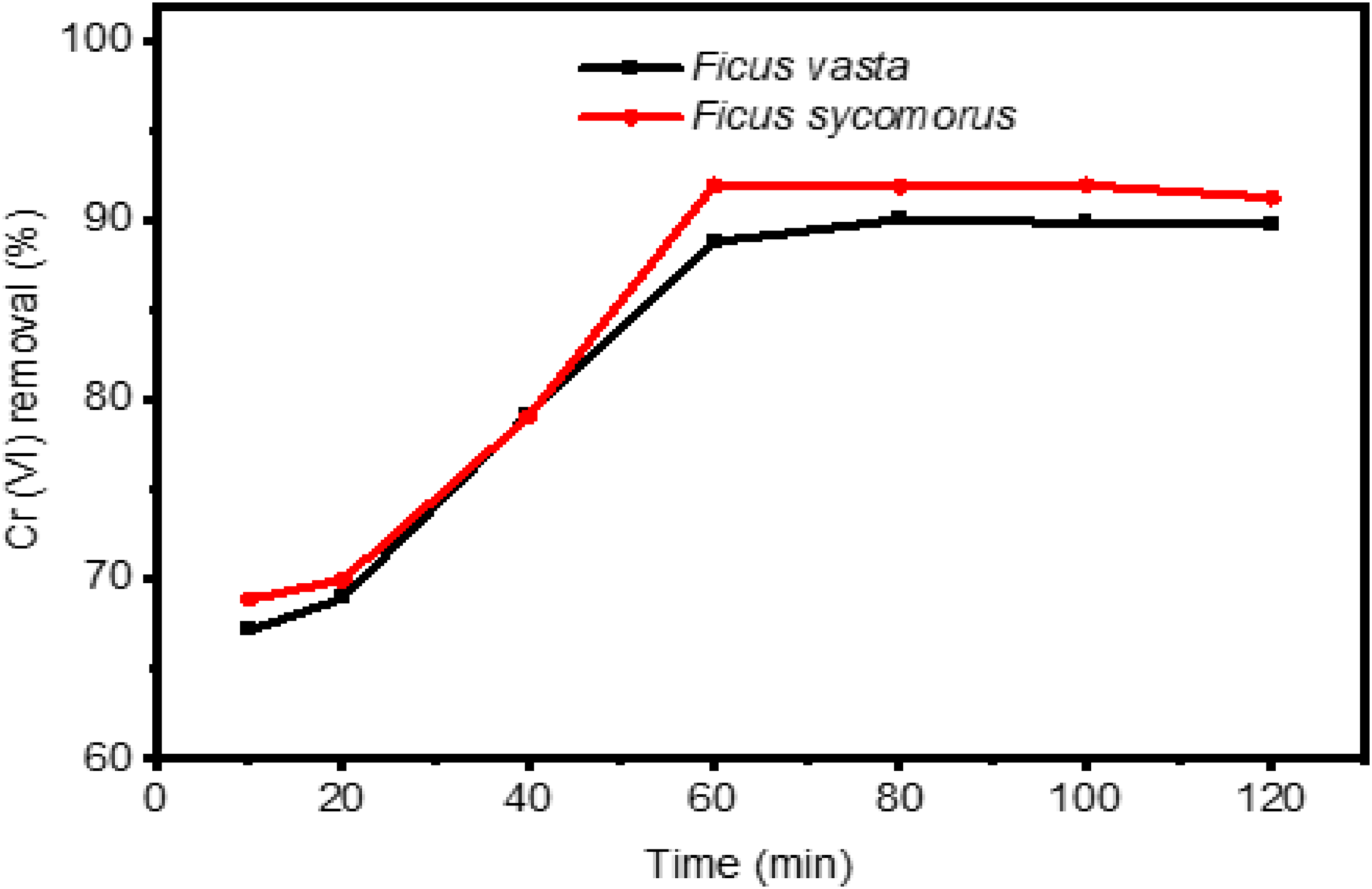
Effect contact time on Cr (VI) removal. Dose (20 g L−1), pH 2 and 3 for
Effect of initial concentration on adsorption
The effect of initial concentrations on the removal of Cr(VI) is displayed in Figure 7. The initial Cr(VI) concentration in the experiment ranged from 5.0 to 30 mg L−1 with 5 mg L−1 intervals keeping the other parameters constant for both adsorbents in each initial concentration setting. The pH was adjusted at 2 and 3 for
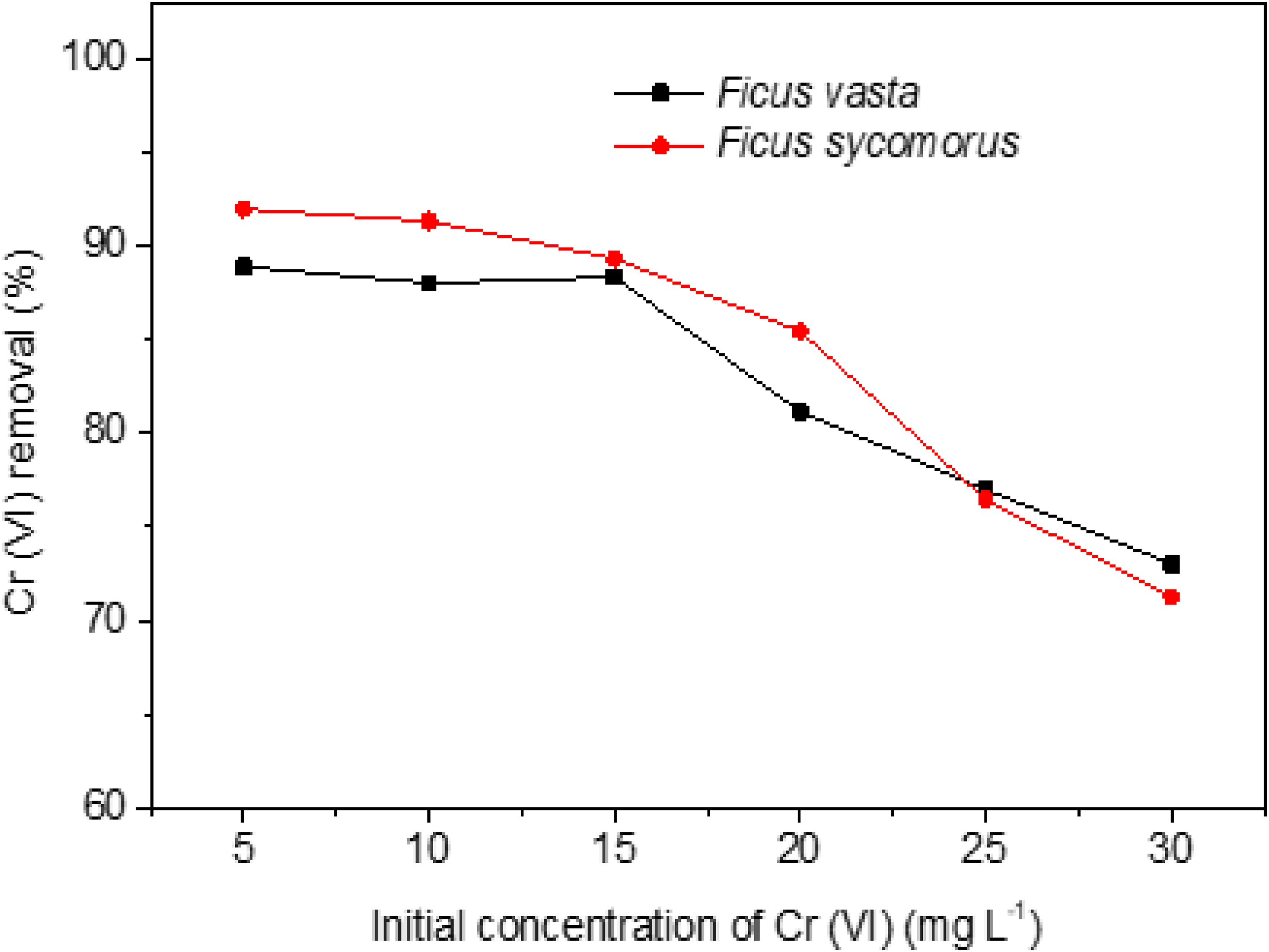
Effect of initial concentration on Cr(VI) removal. Dose (20 g L−1), pH 2 and 3 for
Although this study was conducted using Cr(VI) concentrations typical of contaminated groundwater (5.0 mg L−1), we recognise that industrial effluents often contain significantly higher levels, which can range from 250 to 1000 mg L−1.
62
The adsorption performance of
Adsorption isotherms
Figure S2 in the Supplemental materials illustrates Langmuir (a, b) and Freundlich (c, d) isotherm fits for
The isotherm parameters for Cr(VI) adsorption onto
Isotherm models and parameter values.
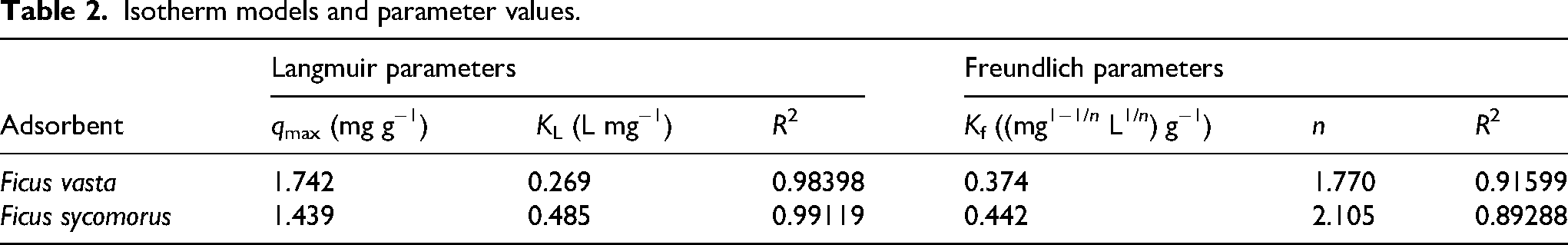
Taken together with the kinetic results, which showed excellent agreement with the pseudo-second-order model (
Adsorption kinetics
Two kinetic models – the pseudo-first order and pseudo-second order – were used for the analysis of the adsorption process's kinetic parameters, which were examined from variations in constant time (5, 10, 15, 20 and 25 min). The linear fitting plots of the kinetic models are shown in Figure S3 in the Supplemental materials. The kinetics parameters for
Using equation (6) and the linear plot of
Kinetic models and parameter values.

Conclusion and recommendation
Given their natural abundance, low cost and high removal efficiency, these leaf-based biosorbents offer a promising and sustainable alternative for scalable water purification technologies. However, while affordability is a key advantage, the long-term cost-effectiveness of the process depends on the reusability of the biosorbents. Future studies should incorporate regeneration and multi-cycle adsorption experiments to evaluate the stability, recovery potential and performance retention of
Although the chromium content of the raw
Supplemental Material
sj-docx-1-mpe-10.1177_25726641251398248 - Supplemental material for Locally accessible Ficus vasta and Ficus sycomorus leaves for adsorption
of Cr(VI) from aqueous solution
Supplemental material, sj-docx-1-mpe-10.1177_25726641251398248 for Locally accessible
Footnotes
Acknowledgements
The authors would like to acknowledge the Tshwane University of Technology, South Africa, Department of Chemical, Metallurgical and Materials Engineering, and Dilla University, Department of Chemistry, Ethiopia, for allowing them to access necessary materials and laboratory rooms.
Author contributions
EB Chemere carried out formal analysis, writing – first draft and editing, conceptualisation and designing. ML Mavhungu involved in conceptualisation and designing of the methodology. W Mhike carried out data curation, editing and offering resources. All authors gave final approval for publication and agreed to be held accountable for the work performed herein.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
