Abstract
In this study, a magnetically separable Fe3O4/CeO2 (Fe/Ce) nanocomposite is synthesized by sol-precipitation method and characterized by field-emission scanning electron microscopy, transmission electron microscopy, X-ray diffraction, energy dispersive spectrometer , vibrating sample magnetometer, atomic absorption spectrometer, and zeta potential measurements. The Fe/Ce is used as sorbent to adsorb anionic dye of Acid Black 210 (AB210) from aqueous solutions, and the maximum adsorption capacity is about 90.50 mg/g, which is six times higher than that of the commercial CeO2. Dependence of absorption performance on essential factors, such as initial dye concentration, temperature and initial pH, are experimentally examined. The result shows that the adsorption kinetic of Fe/Ce follows pseudo-second-order model and the adsorption isotherm is well described by the Langmuir adsorption model. Furthermore, the thermodynamic analysis indicates that the adsorption of Fe/Ce for AB210 is spontaneous and endothermic.
Introduction
In recent years, organic dye contamination has attracted much attention due to their large amount of emissions and increasing damage to natural environment (Hao et al., 2014; Song et al., 2015). Even very low concentration of dyes in water could reduce the light penetration through the water surface, precluding photosynthesis of the aqueous flora (Aguayo-Villarreal et al., 2013). Additionally, some dyes may be highly toxic, potentially carcinogenic, allergenic, mutagenic, and teratogenic on human and aquatic life (Peng et al., 2016; Zhang et al., 2014). These molecules or their metabolites (i.e. aromatic amines) are chemically stable and poorly biodegraded in natural conditions (Ratna, 2012; Zahrim et al., 2011). Therefore, it is highly desirable to develop an economical solution to rapidly and efficiently dispose these dyes from aqueous solutions.
To date, various methods are available for the removal of these dyes from wastewater including adsorption, ultrafiltration, chemical oxidation, ion exchange, photocatalysis, electrodialysis, catalytic degradation and coagulation/flocculation (Boumaza et al., 2014; Fiorentin et al., 2010; Fu and Wang, 2011; Kasgöz and Durmus, 2008; Mane and Babu, 2011). Among these methods, adsorption is verified to be one of the most promising alternative techniques to remove the nonbiodegradable dyes from wastewater due to its simple operation, superior adsorption effectiveness, relatively low cost, and almost harmless byproducts (Maiti et al., 2017; Xiao et al., 2016). So, it is highly approving to design and fabricate superior adsorbents using an economic and facile approach. Until now, various adsorbents such as activated carbon (Tan et al., 2008), fly ash (Dizge et al., 2008), bentonite clay (Tahir and Rauf, 2006), mesoporous silica (Huang et al., 2011), zeolite (Meshko et al., 2001), carbon nanotubes (Machado et al., 2016), graphene oxide (Konicki et al., 2017), and magnetic nanomaterials (Li et al., 2017) have been adopted to remove dyes from aqueous solutions.
One of the promising and efficient adsorbents are magnetic nanocomposites because they can be easily separated from aqueous solutions by applying an external magnetic field (Xu et al., 2010). Therefore, many magnetic nanocomposites based on Fe3O4 have been tested for the possibility to reduce dye concentration in aqueous solutions, owing to Fe3O4 having strong ferromagnetic resonance property and easy recyclability (Kim and Song, 2014). Pan et al. (2016) have synthesized Fe3O4@HCP core–shell porous magnetic microspheres for Methyl Orange adsorption and fast separation from aqueous solutions. Shan et al. (2014) have studied the adsorption of dyes such as Reactive Red, Congo Red, and Acid Red 1 from aqueous solutions by magnetic Fe3O4/MgAl-layered double hydroxide composite. Yao et al. (2012) have prepared magnetic Fe3O4@graphene nanocomposite for the removal of methylene blue and Congo Red from water. Chong et al. (2016) have studied the removal of Methyl Orange, Methylene Blue, and Crystal Violet from aqueous solutions by magnetic Fe(0)/Fe3O4/graphene composite.
However, some magnetic nanocomposites are difficult to be reused because of their unstable performance and are easily affected by environmental conditions, which limit their actual application (Liu et al., 2008; Xu et al., 2010). Considering the factors mentioned above, CeO2 can be one of the efficient and easily reusable adsorbent due to its outstanding thermal stability (Ouyang et al., 2013; Zhang et al., 2016). Because Fe3O4 is easily aggregated leads to the decrease of surface area and affected by the environmental conditions, coated-CeO2 could improve the dispersion and stability of Fe3O4. (Chong et al., 2016; Yao et al., 2012). Additionally, CeO2 can be used not only as a adsorbent but also as a support in the Fe/Ce composite system to protect Fe3O4 against acid and alkaline environments due to the presence of the CeO2 in the outside (Xu and Wang, 2012). Therefore, the CeO2-coated Fe3O4 is a promising alternative adsorbent. It could be easily separated from the aqueous solutions, which is beneficial for multiple reuse, simplified post-processing steps, reduced costs, etc. (Xing et al., 2011; Yu et al., 2012). To date, only a few studies have been conducted on the adsorption of dyes by Fe3O4/CeO2 composites (Li et al., 2017). However, the scale productions of these adsorbents are also limited because of the tedious process of preparation, time-consumption, and costly production.
In the present work, we used the Fe/Ce as an adsorbent to remove anionic dye Acid Black 210 (AB210) from aqueous solutions. The effects of main parameters like initial dye concentration, temperature, and initial pH solution for the adsorption capacity of Fe/Ce have been investigated. Additionally, the kinetic data are analyzed by applying the pseudo-first-order, pseudo-second-order, and intra-particle diffusion kinetic models. The experimental equilibrium adsorption data are fitted to the Langmuir, Freundlich, and Temkin isotherm adsorption models. Thermodynamic parameters, such as ΔG°, ΔH°, and ΔS°, are also calculated.
Experiment
Materials
Materials were purchased from Shanghai Titan Scientific Co., Ltd (China): HNO3 (AR, 65–68%), C2H5OH (AR), NaOH (AR), Fe3O4 (AR), Ce(NO3)3•6H2O (AR), HCl (AR, 36–38%), NH3•H2O (AR, 25–28%), commercial CeO2 (AR). AB210 was supplied by Alibaba.com. Deionized water (DI) supplied by our university was used in all the experiments. All chemicals used were of analytical grade and without further purification.
Preparation of Fe/Ce
Fe/Ce was prepared via sol-precipitation method (Gao et al., 2018). The specific experimental process is as follows. Firstly, 0.0020 mol Ce(NO3)3•6H2O and 20.0 mL of deionized water (DT) were added to a three-necked flask and stirred evenly at 65 °C. Then, 20.0 mL of ammonia solution (0.40 mL NH3•H2O) was added dropwise to the reaction solution to form Ce(OH)3 sol with the injection pump (20.0 mL/h). Subsequently, pretreated Fe3O4 (0.0007 mol) was added to the reaction and 20.0 mL ammonia solution (0.40 mL NH3•H2O) was also added dropwise into the above reaction solution. Thereafter, the reaction was stirred for 5 h at 65 °C in order to prompt Ce(OH)3 sol formed to completely precipitate on the surface of Fe3O4. Then heating was stopped and cooled to room temperature naturally. Finally, the resulting sediment was collected with a magnet and washed alternately with water and ethanol several times, and the Fe/Ce was obtained after drying at 70 °C overnight. The preparation conditions of CeO2 are the same as that of Fe/Ce except for the addition of Fe3O4.
Adsorption experiments
The as-obtained Fe/Ce was employed as an adsorbent for the removal of AB210 from the mimic waste water. Adsorption experiments were carried out in a flask, where the dye solution (50.0 mL) with initial dye concentration was placed. Initial concentrations of dye were varied from 20 mg/L to 80 mg/L for AB210. The flask with dye solution was sealed and placed in a temperature-controlled shaking water bath and agitated at a constant speed of 180 r/min. To observe the effect of temperature, the experiments were carried out at five different temperatures, i.e. 20 °C, 30 °C, 40 °C, 50 °C, and 60 °C. Before mixing with the adsorbent, the pH levels of various dye solutions were adjusted by adding hydrochloric acid (0.1 M HCl) or sodium hydroxide (0.1 M NaOH). When the desired temperature was reached, about 50.0 mg of Fe/Ce was added into the flask. At the end of the equilibrium period, aqueous sample was taken out from the flask with a syringe and the liquid was separated by the external magnetic field. The dye concentration was calculated by UV–visible spectrophotometer (λ = 461 nm).
The amount of dye adsorbed at equilibrium qe (mg/g) was calculated applying the following equation (Radia et al., 2018)
The procedures of kinetic experiments were identical with those of the equilibrium tests. At predetermined moment, aqueous sample was taken from the solution, and a UV–visible spectrophotometer was used to determine the liquid, which was separated from the adsorbent and dye. The adsorption capacity of Fe/Ce at time (t) qt (mg/g) was calculated by the following equation
Characterization
The morphology and microstructure of the sample were captured by a field-emission scanning electron microscopy (FE-SEM, JSM 7401F, JEOL, Japan) at 3.0 kV and transmission electron microscopy (TEM, Tecnai F30). The crystal structure and phase of the as-obtained sample were identified by an X-ray diffractometer (XRD, Mini Flex600, Rigaku, Japan). The elementary compositions of the sample were captured by energy dispersive spectrometer (EDS) (EDS:IE 300 X, Oxford, UK). The zeta potentials of Fe/Ce were measured at different pH values on zeta potential and particle size analyzer (90plus PALS, US) at room temperature. The magnetic hysteresis loops were measured by a vibrating-sample magnetometer (VSM, 7407, Quantum Design Company, USA). The released iron concentration of the sample was measured by double-beam flame atomic absorption spectrometer (AAS) (AA240, Agilent, USA). The adsorption experiments were carried out at the electric agitation condition (SHA-C, Sai De Li Si Experimental Analytical Instrument Company, Tianjin, China). The absorption spectra of the initial AB210 solution as well as the filtrate after adsorption were monitored by a UV–visible spectrophotometer (Beijing Purkinje General Instrument production of TU-1900) with a characteristic absorption at the wavelength of 461 nm to quantify the concentration of AB210 dye.
Results and discussion
Characterization of Fe/Ce
Figure 1 shows the high-magnification FE-SEM images (a, b) of Fe3O4 and Fe/Ce and TEM images (c, d) of Fe/Ce and CeO2. The morphology of Fe3O4 is irregular and some approximate spherical particles can be found in Figure 1(a). Owing to the strong interaction among magnetic particles, such as the magnetic dipolar interaction and van der Waals force, an obvious agglomeration and sticking together of irregular Fe3O4 nanoparticles also can be observed (Wang et al., 2015). Compared with Fe3O4, the morphology and size of Fe/Ce have no obvious change after coating of CeO2 (Figure 1(b)), which can be ascribed to the poor uniform size of Fe3O4 nanoparticles and uneven CeO2 coated on the surface of Fe3O4 (Figure 1(c)). Besides, the Fe/Ce particles still remain aggregate, which reveals that the sample still maintains the characteristic of magnetism. In Figure 1(c), obvious delamination of Fe/Ce particles can be found, where the black area in the middle is Fe3O4, and the light-colored area on the edge is the coated CeO2. Moreover, the particle size of the coated CeO2 is substantially the same as that of the CeO2 particles (prepared under the same conditions) (Figure 1(d)). It further confirms that CeO2 is successfully loaded onto Fe3O4 surface. Figure 1(e) and (f) shows the EDS element mapping images of Fe3O4 and Fe/Ce. In Figure 1(e), pure Fe3O4 contains iron and oxygen and it can be concluded from the table inside the figure that the atomic ratio of iron and oxygen is in accordance with that of Fe3O4. In Figure 1(f), Fe/Ce contains cerium apart from iron and oxygen, and the positions of iron and oxygen are basically the same as that of Fe3O4 (Figure 1(e)). According to the table inside the figure, the molar ratio of CeO2 to Fe3O4 can be roughly calculated to be about 3:1. This indicates that Fe/Ce material is successfully prepared.

FE-SEM images of (a) Fe3O4, (b) Fe/Ce. TEM images of (c) Fe/Ce, (d) CeO2. EDS elemental patterns of (e) Fe3O4, and (f) Fe/Ce.
Figure 2 shows the XRD pattern of the pure CeO2, single Fe3O4, and Fe/Ce. For single Fe3O4, the stronger diffraction peaks and the corresponding crystal plane of the single Fe3O4 at 30.10°(220), 35.42°(311), 56.94°(511), 62.52°(440) are assigned to Fe3O4 (JCPDS No. 19–0629). The stronger diffraction peaks and the corresponding crystal plane of pure CeO2 at 28.55°(111), 33.08°(200), 47.48°(220), 56.33°(311) match well with those of CeO2 (JCPDS No. 34–0394). As a contrast, the XRD pattern of Fe/Ce contains obvious diffraction peaks of pure CeO2 and single Fe3O4. This indicates that these substances of Fe3O4 and CeO2 are included in the as-obtained material. Moreover, it is worth noting that the decrease in the intensity of diffraction peaks corresponds to Fe3O4 and CeO2, respectively, which reveals reduced crystallinity of as-prepared Fe/Ce due to the interaction between CeO2 and Fe3O4 and agglomeration of particles. In addition, no other peaks could be detected, which indicates that there is no impurity in Fe/Ce. The results obtained are in good agreement with that of EDS element mapping images and TEM images, which further confirms that the as-prepared sample is the CeO2-coated Fe3O4.

The XRD of CeO2, Fe3O4, and Fe/Ce.
Magnetic properties of bare Fe3O4 and Fe/Ce are investigated via VSM at room temperature. Figure 3 shows that Fe/Ce has near-zero remanence and coercivity at room temperature, which reveals a super-paramagnetic characteristic (Wu et al., 2016). The saturation magnetization values of bare Fe3O4 and Fe/Ce are 73.58 emu/g and 28.06 emu/g, respectively. Compared with bare Fe3O4, the decreased 45.52 emu/g of saturation magnetization value of Fe/Ce is attributed to the existence of loaded CeO2, which weakens the magnetic property of bare Fe3O4 (Xu and Wang, 2012). Simultaneously, it illustrates the successful synthesis of Fe/Ce. So, the result of the VSM is consistent with the TEM, EDS, and XRD data. Moreover, the as-prepared Fe/Ce still shows excellent magnetic sensitivity, as shown in illustration 2, and the dye-loaded adsorbent can be rapidly separated from the mixture (illustration 1) within 10 s with the help of the magnet. This excellent magnetic response behavior of Fe/Ce contributes to a rapid, easy, and high throughput way to enrich and separate adsorbents (Xu et al., 2014).

Magnetic hysteresis loops of Fe3O4 and Fe/Ce. Photographs (inset) (1-AB210 and Fe/Ce mixing, 2-the magnet collects Fe/Ce after adsorption of AB210).
In order to evaluate the potential application in dye wastewater treatment, the Fe/Ce is employed as the adsorbent for the removal of AB210 from mimic waste water. As can be seen in Figure 4, the adsorption capacity (qt) of CeO2 and Fe/Ce rapidly increases to 38.64 mg/g and 34.34 mg/g within the first 2 min and continues to increase in the next 10 min to 30 min, respectively, then further increases at a relatively minor rate till 120 min. Nevertheless, qt of Fe/Ce is 41.18 mg/g in 120 min, which is 2.89 mg/g lower than that of CeO2. Since the CeO2 particles is coated on the surface of the Fe3O4, its specific surface area is reduced, which leads to the decrease in the adsorption performance. Simultaneously, we can clearly see that qt of commercial Fe3O4 is only 12.18 mg/g in 120 min, which is 29.00 mg/g lower than that of Fe/Ce. This demonstrates that Fe/Ce still maintains good adsorption property in addition to the magnetic property. Additionally, the Fe/Ce (illustration 3) and CeO2 (illustration 4) intuitively show that the dye solution is almost colorless after adsorption 120 min, respectively. However, the Fe3O4 (illustration 2) is not significantly different from the color of the initial concentration of the dye (illustration 1) within 120 min. This also indicates that the Fe/Ce still has a better adsorption performance.

Instantaneous adsorption capacity of different materials within 120 min (the amount of all the materials are the same: 50.0 mg 50.0 mg/L). Photographs (inset) (1-AB210, 2, 3, 4 is the corresponding colors of the commercial Fe3O4, Fe/Ce, and CeO2 in 120 min, respectively).
Effect of initial dye concentration on adsorption kinetics
To determine the effect of the initial concentration of AB210 on the adsorption capacity of Fe/Ce, the adsorption experiments are performed in several initial concentrations ranging from 20 mg/L to 80 mg/L at 30 °C and pH = 7.0. As seen in Figure 5, the adsorption capacity of Fe/Ce increases with increasing AB210 initial concentration. And the adsorption capacity growth trends of different initial concentrations are basically the same. This indicates that the initial AB210 concentration plays a vital role in the adsorption capacity of Fe/Ce because the initial dye concentration provides a momentous driving force to overcome the mass transfer resistance of the dye between solid and aqueous phases. Hence, the adsorption capacity increases with increasing initial dye concentration.
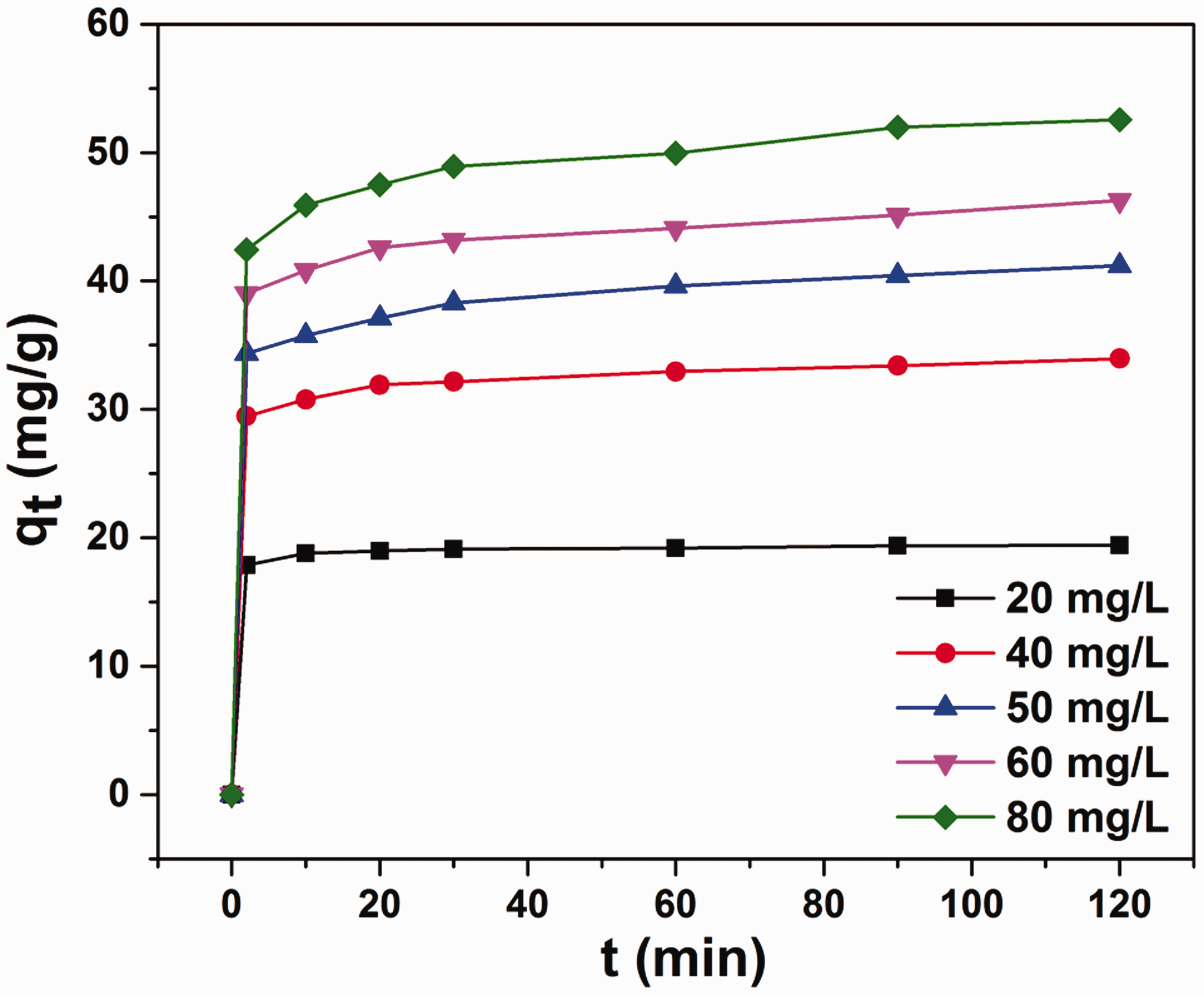
Effect of initial dye concentration on adsorption capacity of the AB210 onto Fe/Ce. Experimental conditions: T = 30 °C, pH = 7.0.
In this study, the experimental data for the adsorption of AB210 onto Fe/Ce are tested with three different adsorption kinetic models: pseudo-first-order model, pseudo-second-order kinetic model, and intra-particle diffusion model. The pseudo-first-order kinetic model presumes that the ratio of occupation of adsorption sites is proportional to the number of unoccupied sites. The pseudo-second-order kinetic model assumes that the adsorption mechanism depends on the adsorbent and adsorbate, and the rate-limiting step may be the chemical adsorption involving valence forces through the exchanging or sharing of electrons between adsorbent and adsorbate. The intra-particle diffusion model is used to research whether the intra-particle diffusion is the rate controlling step in the process of adsorption of AB210 dye onto Fe/Ce.
The pseudo-first-order kinetic model is shown as the following (Mellah et al., 2006)

Adsorption kinetic for adsorption of AB210 onto Fe/Ce. Pseudo-first order (a) and Pseudo-second order (b). Experimental conditions: T = 30 °C, pH = 7.0.
The pseudo-second-order kinetic model can be represented in the following linear form equations (Ho and Mckay, 1999)
Comparison of the pseudo-first-order, pseudo-second-order, and the intra-particle diffusion models for different initial concentrations of AB210 at 30 °C and pH = 7.0.

The intra-particle diffusion model is expressed by the following equation (Wu et al., 2009)


Intra-particle diffusion model of adsorption the AB210 onto Fe/Ce. Experimental conditions: T = 30 °C, pH = 7.0.
Adsorption isotherm
The equilibrium adsorption data are analyzed using Langmuir, Freundlich, and Temkin isotherm models. The Langmuir isotherm model assumes that the adsorption occurs on the homogeneous surface of the adsorbent. The Freundlich isotherm model is commonly used to describe the surface heterogeneity. The Temkin isotherm presumes that the heat of adsorption of all molecules in the layer reduces linearly with coverage because of the adsorbate–adsorbent interactions and the adsorption is characterized by means of the uniform distribution of bonding energies, up to some maximum binding energy.
The Langmuir adsorption isotherm can be described by the following (Qiu et al., 2014)

The linear form of Freundlich isotherm is shown by the following (Zhou et al., 2015)

The linear form of Temkin isotherm equation can be described as the following (Mashhadi et al., 2016)

Figure 8(a) to (c) shows the linearized forms of the Langmuir, Freundlich, and Temkin isotherm models based on the experimental data, respectively. The values of these adsorption isotherm parameters are shown in Table 2. According to the table, Langmuir model is best fit as compared with the other two models, which suggests a homogeneous adsorption of Fe/Ce for AB210. This result also indicates the homogeneous nature of Fe/Ce surface, i.e. each AB210 dye molecule adsorption has equal adsorption activation energy. Based on the Langmuir equation, the value of qm is calculated as 90.50 mg/g, which is considerably higher than that of the commercial CeO2 and other CeO2 with different morphologies previously reported (Table 3). In particular, the qm of Fe/Ce is six times higher than that of the commercial CeO2. At the same time, this result also clearly shows the excellent adsorption performance of Fe/Ce for AB210 except for the magnetic property.

(a) Langmuir, (b) Freundlich, and (c) Temkin isotherms for AB210 adsorption onto Fe/Ce. Experimental conditions: T = 30 °C, pH = 7.0.
Langmuir, Freundlich, and Temkin parameters for the adsorption of AB210 onto Fe/Ce.

Maximum adsorption capacities of Ceria with different morphologies adsorbents to AB210 and other dyes.

The essential characteristics of Langmuir isotherm model can be described according to the dimensionless equilibrium parameter (RL), which is defined by the following (Kaur et al., 2008)

Effect of temperature
The effect of temperature on adsorption of AB210 onto Fe/Ce is shown in Figure 9. The experiments are implemented at five different temperatures (20 °C, 30 °C, 40 °C, 50 °C, and 60 °C) under a constant initial dye concentration of 50 mg/L and pH = 7.0. With the increasing of the temperature from 20 °C to 40 °C, adsorption capacity increases relatively significant from 41.18 mg/g to 43.90 mg/g. However, the rate of increase in the adsorption capacity decreases slowly from 43.90 mg/g to 44.78 mg/g with the temperature increasing from 40 °C to 60 °C. This phenomenon may be that the adsorption process is divided into two stages, which is consistent with the intra-particle diffusion model. The experimental results show that the adsorption capacity increases with temperature in the range of 20–60 °C, which indicates that the adsorption of AB210 onto Fe/Ce is endothermic in nature.

Effect of temperature on adsorption of the AB210 onto Fe/Ce. Experimental conditions: Co = 50 mg/L, pH = 7.0.
The values of thermodynamic parameters of change in Gibbs free energy (ΔG°), enthalpy (ΔH°), and entropy (ΔS°) are determined using the following equations (Gad and El-Sayed, 2009)




Van’t Hoff plot for the adsorption of the AB210 onto Fe/Ce. Experimental conditions: Co = 50 mg/L, pH = 7.0.
Thermodynamic parameters for the adsorption of AB210 onto Fe/Ce.

The positive value of ΔH° (2.88 kJ/mol) indicates that the adsorption process of Fe/Ce for AB210 is endothermic in nature. The positive value of ΔS° (28.90 kJ/mol) suggests that the randomness increases at the solid–liquid interface during the adsorption of Fe/Ce for AB210. The negative values of ΔG° decrease from −5.20 kJ/mol to −5.53 kJ/mol with the increase in temperature, which indicates the feasibility and spontaneity of adsorption process. Moreover, this phenomenon also suggests that the adsorption process is more favorable at higher temperature. Similar result is also reported, e.g. Zheng et al. (2018) have synthesized shape-dependent CeO2 nanostructures to adsorb the anionic dye Congo Red.
Generally, the adsorption process is considered to be physisorption in nature when the ΔG° value is in the range of −20 kJ/mol to 0 kJ/mol, while the ΔG° value range from −400 kJ/mol to −80 kJ/mol implies a chemisorption process (Zeng et al., 2014). Furthermore, if the value of ΔH° is lower than 40 kJ/mol, then it is certified that it conforms to the physisorption process (Kara et al., 2003). Considering the obtained values of ΔH° and ΔG°, the adsorption process of Fe/Ce for AB210 is considered as physical adsorption.
Effect of initial pH
The pH of the aqueous solutions is a key parameter in the adsorption process. Figure 11(a) shows the effect of initial pH on the released iron concentration of Fe3O4 and Fe/Ce in treated solution, respectively. As the pH value increases from 3.0 to 9.0, the released iron concentration of Fe3O4 and Fe/Ce decreases from 2.04 × 10−1 mg/L to 6.46 × 10−4 mg/L, 9.05 × 10−3 mg/L to 2.58 × 10−4 mg/L−1, respectively. However, when pH = 9.0–11.0, the effect is almost nonexistent. When pH = 3.0, the released iron concentration of Fe/Ce is about 22.5 times lower than that of Fe3O4. Because the loaded CeO2 layer of Fe/Ce protects Fe3O4 inside from being affected by acidic condition. At the same time, it also indicates that CeO2 is successfully loaded on the surface of Fe3O4. Similarly, the released iron concentration of Fe/Ce (CFe=6.46 × 10−4 mg/L) is about 15 times lower than that of Fe3O4 (CFe=9.70 × 10−3 mg/L) at pH = 7.0 condition. The results obtained are consistent with those of TEM. The effect of initial pH on adsorption of anionic dye AB210 has been studied in the range 3.0–11.0 at 30 °C with the initial dye concentration always fixed at 50 mg/L. As shown in Figure 11(b), as the pH value increases from 3.0 to 11.0, the adsorption capacity at equilibrium decreases from 44.02 mg/g to 1.52 mg/g after 120 min. It is more important that the adsorption capacity declines dramatically from 41.18 mg/g to 3.05 mg/g with the increase in the pH value from 7.0 to 9.0. It can be explained by the electrostatic attractions (Wu et al., 2009). Meanwhile, the zeta potential of Fe/Ce also shows a decreasing trend with the effect of initial pH too. The value (pHpzc) of Fe/Ce is 7.7, at which the net surface charge on adsorbent is zero. The adsorbent surface has a net positive charge at pH < pHpzc, while has a net negative charge at pH > pHpzc. The AB210 is anionic dye, which dissociates into the cationic form (–OH2+, –NH3+) and the negatively charged dyes. At acidic pH, the presence of the hydroxyl (–OH) on the Fe/Ce surface is protonated to the cationic form (–OH2+) and generates electrostatic attraction force with dyes anions. Therefore, the adsorption of AB210 onto Fe/Ce surface is more favorable at lower pH value. However, at basic pH, the hydroxyl (–OH) is deprotonated to anionic form (–O−), causing a decrease in the number of positively charged sites, which in turn does not favor the adsorption of negatively charged dyes molecules. Thus, adsorption capacity of AB210 onto Fe/Ce tended to decrease with the increasing pH value, which can be attributed to the electrostatic repulsion between the negatively charged sites on the adsorbent surface and the negatively charged dye molecules. Therefore, the adsorption of AB210 is favorable when the pH value is smaller than pHpzc (Ahmad and Kumar, 2011; Konicki et al., 2017).

(a) The effect of initial pH on iron release from Fe/Ce, commercial Fe3O4, respectively. Iron standard curve (inset). (b) The effect of initial pH of dye solution on adsorption capacity of the AB210 (the black line) and the effect of initial pH on zeta potentials of Fe/Ce (the red line). Experimental conditions: Co=50.0 mg/L, V = 50.0 mL, T = 30 °C.
Conclusions
The removal of AB210 from aqueous solutions is studied by the Fe/Ce sorbent. Fe/Ce can be well dispersed in water and can be simply separated from water under external magnetic field. The adsorption process is investigated under key parameters such as initial dye concentration, temperature, and initial pH. The adsorption kinetic of Fe/Ce for AB210 follows the pseudo-second-order model well. The absorption isotherm of AB210 on Fe/Ce best fits the Langmuir adsorption model. The maximum adsorption capacity of Fe/Ce is 90.50 mg/g, which is six times higher than that of the commercial CeO2. The thermodynamic parameters indicate that the adsorption of AB210 onto Fe/Ce is spontaneous and endothermic process in the experimental conditions. The thermodynamic analysis data indicates that the adsorption of AB210 dye onto Fe/Ce is confirmed as a physisorption process. In addition, the effect of initial pH also shows that the adsorption of AB210 on Fe/Ce is favorable at acid condition. Therefore, the research on adsorption equilibrium, thermodynamics, and kinetics of Fe/Ce could enrich the adsorption mechanism research of adsorbents and Fe/Ce may be a convenient and efficient adsorbent for the treatment of dye wastewater.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.




