Abstract
During hydraulic fracturing in a high-methane coal seam, there is a water-displacing-methane effect. A pseudo triaxle experimental system, which is opposite to the name of true triaxial system, for the water-displacing-methane effect was created. First, cylindrical coal samples in a methane adsorption equilibrium state, spontaneously desorbed. And then water was injected into the coal samples. The following was shown: (1) The displacement methane volume gradually rises with an increase of injected water, while the displacement methane rate tends to rise at first before declining later. Simultaneously, the water-displacing-methane process is characterised by a time effect. The methane displacement lags behind water injection. (2) Competitive adsorption and displacement desorption between the water and methane will promote adsorption methane into free methane, while the pore pressure increase caused by water injection will turn free methane into adsorption methane. The net free methane of the combination action provides a methane source for the water-displacing-methane effect. (3) A pore pressure gradient, which provides a power source for the water-displacing-methane effect, is formed and reduces gradually at the front of the water seepage along the seepage direction. The increase in water pressure can rapidly improve the pore pressure gradient and boost the displacement methane volume as well as improve displacement methane efficiency. (4) A starting porosity pressure gradient and limit pore pressure exist in the process of water-displacing-methane. When the pore pressure gradient is less than the starting pore pressure gradient, there is free methane in the coal rock, but it cannot be displaced. When the pore pressure is between the starting pore pressure and the limit pore pressure, the free methane can be displaced. When the pore pressure is greater than the limit pore pressure, the methane is almost completely adsorption methane, and water cannot be used to displace the free methane.
Keywords
Introduction
Hydraulic fracturing not only has a role in the exploitation of coalbed methane but also has been more widely utilized to improve permeability and eliminate coal and methane outbursts in gassy coal seams (Lu et al., 2015; Ma et al., 2016; Wang et al., 2017; Xu et al., 2017; Zhai et al., 2015; Zhang et al., 2017). When water injection or hydraulic fracturing is conducted in high gassy coal seams, methane concentration of the air current in the air-return roadway will rise. This effect is termed the methane-displacement effect by water injection or hydraulic fracturing (Huang et al., 2016a, 2018). At the same time, the existing research results show that competitive adsorption between water and methane can lead to the effect of adsorption methane displacement by water (Abudour et al., 2012; Lee et al., 2013; Müller et al., 2000). In other words, free methane driven by water consists of two parts: the original free methane, and the free methane generated by the displacement desorption (Gensterblum et al., 2014; Liu et al., 2016; Xiang et al., 2014). Therefore, the concept of the displacement methane effect by water injection or hydraulic fracturing can more accurately and comprehensively represent the aforementioned phenomena. The methane-displacement effect by water injection or hydraulic fracturing has both advantages and disadvantages in production practice. On the one hand, it can provide new ideas and technology for the exploitation of coalbed methane and the hydraulic measures used to eliminate methane outbursts. On the other hand, it can increase the methane concentration of the air flow in the roadway in addition to the methane content and methane pressure of the local region in the coal seam, which is detrimental to the prevention of coal and methane outbursts and methane explosions (Lu et al., 2019; Ma et al., 2016; Huang et al., 2018). When the water-displacing-methane (W-D-M) effect is considered, its advantages should be enhanced and its disadvantages should be avoided. While the parameters of water injection or hydraulic fracturing can be optimised so as to avoid negative effects such as the over-limit of return airway methane concentration induced by the W-D-M effect, the W-D-M effect can be reached the level of methane displacement by hydraulic fracturing. A new concept and technological approach are provided for the extraction of coal bed methane and the elimination of outbursts in coal seams, especially coal seams with low permeability (Huang et al., 2016a; Lu and Huang, 2020).
At present, the existence of the W-D-M effect has been verified by both field tests and laboratory experiments (Huang et al., 2016a, 2016b). However, according to the basic theory of immiscible two-phase fluid in porous media, free methane in a position with high pore pressure migrates to the position with low pore pressure (Akhmetzyanov et al., 2016; Schlüter et al., 2016). When high pressure water is injected into a coal rock mass, an increase in the pore pressure will turn free methane into adsorption methane, resulting in consumption of the free methane (Lu et al., 2020). However, displacement desorption can generate free methane. There are two important questions to answer. One is the result of the both effects occurring at the same time, the other is the methane and driving power source of the W-D-M effect. To address these questions, the mechanism of the W-D-M effect should be studied.
Hydraulic fracturing can transform the structure of a coal rock mass, while water injection will not (Huang et al., 2016a, 2016b). In order to eliminate the influence of the structural transformation of the coal rock mass on the W-D-M effect, the mechanism of the W-D-M effect should be well understood beforehand. In this paper, using a self-developed triaxle experimental system for the W-D-M effect, the effect of an external water injection on the volume and rate of methane displacement was studied. The free methane and driving power source of the W-D-M effect is identified, and the mechanism of the W-D-M effect is revealed.
Experimental method
Experimental system
According to the schematic diagram of the W-D-M principle (Figure 1(a)), a pseudo triaxle experimental system of methane displacement by (water) fracture (Figure 1(b)) was independently designed and developed. The experimental system can apply to variable axial and confining pressures, and it is characterized by fracturing of the coal rock mass, a permeability characteristic test, water-displacing methane, methane displacement by hydraulic fracturing, and so on.

Pseudo triaxle experimental system of methane displacement by (water) fracturing. (a) Schematic diagram of water-displacing-methane principle (b) System diagram.
The experimental system consisted of a pseudo triaxle fracturing displacement frame, an axial and confining pressure loading system, a water injection system, a methane injection system and a displacement methane collection and measurement device. In Figure 1(b), the principle and object of the fracturing displacement frame are in the dotted box of the blue rectangle. The axial pressure is applied by a hydraulic jack at the bottom of the W-D-M device. The confining pressure is applied by injecting hydraulic oil into a circular cavity of the W-D-M device. The maximum axial and confining pressure can reach 30 MPa. A loading system controlled by a four-channel electro-hydraulic servo based on a MOOG valve is adopted to control the load of the axial, confining, and hydraulic pressure on the sample.
A seepage experimental system is equipped with two seepage plates respectively placed at the top and bottom of the coal sample. The cross-section of the seepage plate has the same area as the cross-section of the cylinder sample. Eyelets 1 mm in diameter and with 2 mm spacing are distributed along the peripheral and radial direction of the seepage plate. The upper and lower seepage plates are assembled as a whole, which is sealed with a heat-shrinkable tube to form a seepage unit. An eyelet is reserved for hydraulic fracturing at the centre of the top seepage plate. When there is only seepage without hydraulic fracturing, the eyelet for hydraulic fracturing is sealed with an airproof bolt. A seepage experiment of different pore pressure and pore pressure gradient can be conducted by controlling the water pressure of the upper and lower seepage plates and the central fracturing eyelet with an electro-hydraulic servo.
The pressure of the methane circuit system is adjusted by reducing the valve at the outlet of the methane cylinder. The methane circuit system, which is characterised by good air-tightness, can be utilized to conduct an experiment of methane adsorption and desorption in a coal-rock mass and W-D-M.
Preparation of coal sample
First of all, the drill machine is utilized to take a preliminary coal sample from the original fat coal, which is from the coal mine. And then coal samples taken out from the drill machine are polished into coal samples, whose size meets the needs of the experiment. Three cylindrical coal samples with a diameter of 50 mm and a height of 100 mm are prepared for this experiment. In the process of the coal sample, both ends of the sample should be smoothed to guarantee equal force in the experiment.
The uniaxial compressive strength, tensile strength, cohesion force, internal friction angle, porosity and permeability of the coal sample were tested, and the results were shown in Table 1.
Physical parameters of coal sample.

Experimental process
Assemble the system and place the coal sample into the semmble. Assemble the experimental system according to Figure 1(b). Before placing the coal sample into the W-D-M device, a layer of gauze filtration should be initially placed between the coal sample and the bottom seepage plate. As a result, tiny particles generated by the crushed coal sample can be prevented from blocking the channel of the seepage plate.
Apply axial and confining pressure. Apply 1.1 MPa confining pressure and 2 MPa axial pressure at a loading rate of 0.11 MPa/s and 0.2 MPa/s, respectively. Then maintain the pressure for 5 minutes.
Check the airtightness. The output pressure of the methane cylinder should be maintained at 1.0 MPa by adjusting the reducing valve of the methane cylinder. Keep the methane inlet valve open until the methane outlet pressure reaches 1.0 MPa, and then close the methane inlet valve. If the outlet pressure remains unchanged at 1.0 MPa for at least 10 minutes, the airtightness of the experimental system is good. Otherwise, there are some airtightness problems in the experimental system. The methane leakage position can be detected by a portable methane detection alarm with a monitoring range of from 0 to 4%. After addressing the air leakage position, check the airtightness of the experimental system again until there are no methane leakage positions.
Expel all the air in the system. The output pressure of the methane cylinder should be maintained at 0.2 MPa by adjusting the reducing valve of the methane cylinder. Open the methane inlet and outlet valves, and collect the vented methane. Test the concentration of the collected methane with optical methane concentration detector and methane sampler (Figure 2). When the methane concentration is near 100%, it is considered that the air in the experimental system has been fully removed. Then close the methane outlet valve.
Methane adsorption and desorption equilibrium by methane injection. Open the methane cylinder, and control the output pressure by adjusting the reducing valve of the methane cylinder. Open the methane inlet valve and close the methane outlet valve. Methane continuously adsorbs onto the coal sample until the methane adsorption and desorption equilibrium is reached.
Free emission of the methane. After the methane adsorption and desorption equilibrium is reached, close the methane cylinder valve and methane inlet valve, and open the methane outlet valve. Collect the pipeline methane, original free methane in the coal sample and free methane from spontaneous desorption of the adsorption methane, until there is no methane releasing any longer.
Methane displacement by water injection. Close the methane inlet valve and open the methane outlet valve. Inject water from the water inlet according to the designed rate or pressure of water injection. Collect the released methane until there is no methane releasing any longer.
Test the methane concentration and the moisture content of the coal sample surface. Test the methane concentration of the collected methane, and calculate the content of the collected methane. At the same time, test the moisture of the coal sample surface with a moisture meter.

Methane concentration test.
Experimental results and analysis
Test the concentration of the collected methane
The experiment lasted 155 minutes. A total of 4544 mL of water was injected, and 520 mL of methane was collected. In this experiment, methane with a high-purity of 99.999% was used, and 70 mL of air volume was used to zero the geological exploration methane desorptioner. Consequently, the collected methane concentration in theory was 520 mL/(520 + 70) mL = 88.136%. The result obtained by the methane concentration detector (Figure 2) is 88%, so the 520 mL of collected methane was all methane.
Test the moisture content of the coal sample surface
By observing the degree of wetness of the coal sample surface (Figure 3) at the end of the experiment, it was found that the water permeated through the coal sample. This is because (1) after the experiment, there was not any continuous trace wetted by water on the coal sample surface from all angles of observation, and (2) there are local water-wetting traces, which are shown as the dotted rectangle in Figure 3, appearing near the fractures of the coal sample surface. This just demonstrates that the water seepage occurred along the pore-fracture channel inside the coal sample, and it could also flow to the coal sample surface through the interconnected pore-fracture channels inside the coal sample. This also indicates that the methane displaced by the water is all from the interior of the coal sample.

Water-wetting status of coal sample surface after methane displacement by water.
To obtain the degree of wetness of the coal sample surface, a moisture meter was used to test the moisture content of the wet and dry parts of the coal sample surface. The measured results of the wet and dry locations are 9% and 3% respectively, as shown in Figure 4(a) and (b). The 3% moisture content of the dry location after the experiment is consistent with the 3% of the moisture content of the original coal sample surface, indicating that the dry position of the coal sample surface has never been in contact with water during the process of water injection. In other words, the result obtained using the instrument is identical with that of human observation, and it is fully proved that the water penetrated through the interior of the coal sample.

Moisture content of coal sample surface after methane displacement by water. (a) Wet location (b) Dry location.
Basic rules of methane displacement by water
Because of the W-D-M effect, the adsorption methane in the coal sample can be displaced by water. As is shown in Figure 5, (1) as the injection volume increases, the displacement methane volume increases accordingly. The slope of the displacement methane volume curve first increases, as shown in the change of the slope of curve segment 1, curve segment 2 and curve segment 3 in Figure 5, and then decreases, as shown in the change of the slope of curve segment 3, curve segment 4 and curve segment 5 in Figure 5. This shows that the displacement rate first increases, as shown in the change in displacement rate 1, displacement rate 2 and displacement rate 3 in Figure 5, and then decreases, as shown in the change in displacement rate 4 and displacement rate 5 in Figure 5. (2) At the initial time of the water injection, the methane was not immediately driven out. When the water injection stops, the W-D-M effect does not stop immediately as well. This indicates that the W-D-M process has a time effect, and the displacement methane lags behind the water injection. The time effect could not only be used to explain why the mining methane emission content of the working face in the field during the water injection was greater than that before the water injection but also could be used to explain why the mining methane emission content was still continuously rising after the water injection had stopped for a period of time.

The change of displacement methane content by water with water injection volume.
The process of methane displacement by water includes the process of free methane generated by competitive adsorption and displacement desorption between water and methane, the process of free methane consumed by pore pressure increase caused by water injection and the process of methane displacement by water, all of which are shown in Figure 6(a). When the pressure water enters into the pore-fracture channel system of the coal-rock mass, the original mechanical equilibrium is broken and the stress is redistributed. At the forefront of the seepage water, pore water moves forward by overcoming the resistance of the pore channel in the rock mass. As a result, free methane within a certain range of the seepage water forefront is compressed, which is shown in Figure 6(b), and the methane pressure is increased. At the forefront of the seepage water, the pore pressure gradient from high to low, as shown in Figure 6(b), is distributed along the direction of pore water migration (Huang et al., 2016a). Under the effect of the pore pressure gradient, a net increase in free methane is generated by the combined action of the displacement desorption and pore pressure increase.
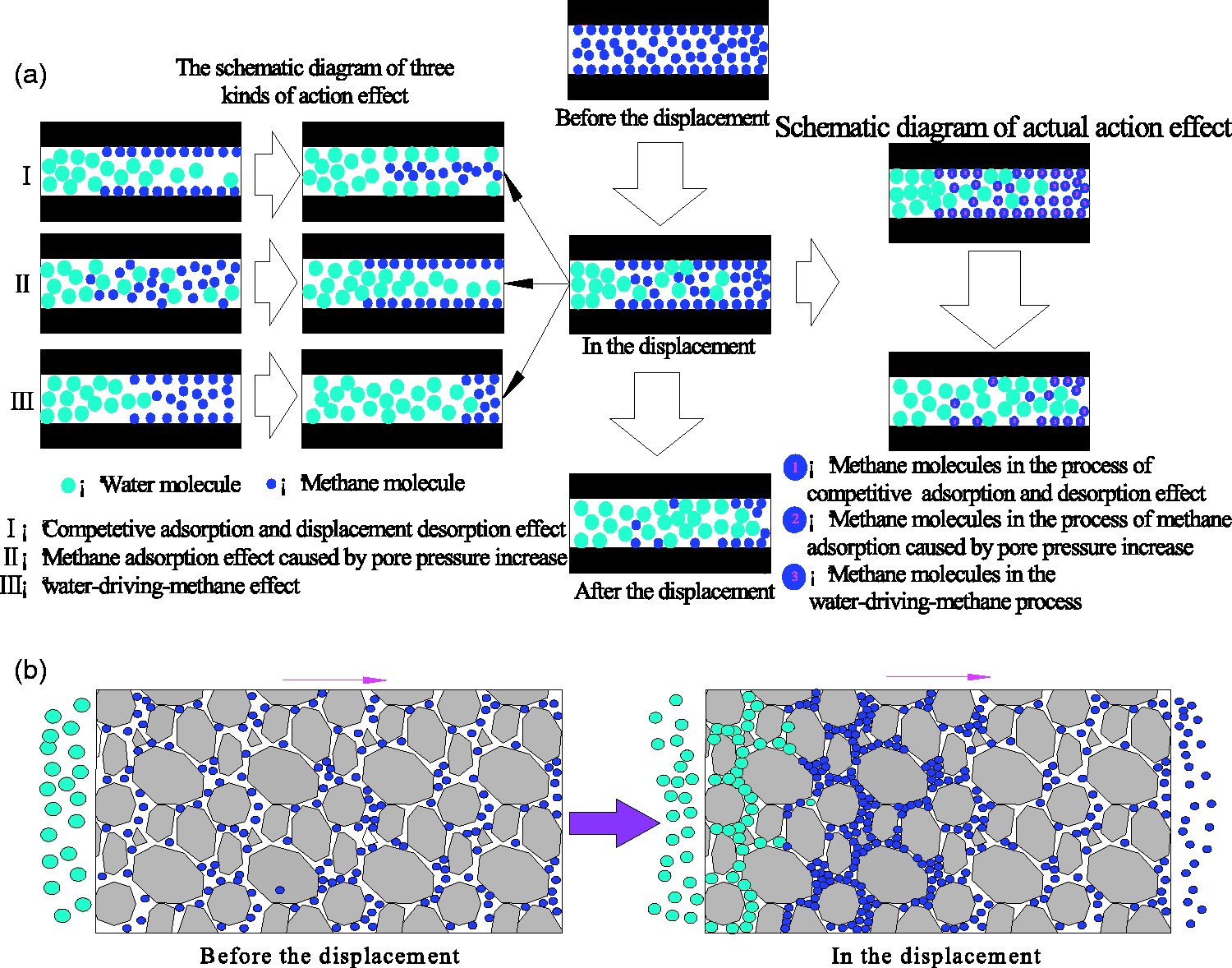
The schematic diagram of methane displacement by water. (a) A single pore channel(b) Porous coal.
Methane source of the water-displacing-methane effect
The adsorption of coal to methane and water is physical adsorption. Because the adsorption sites on the surface of the coal are limited, there is a competition between the water and methane molecules in capturing the adsorption sites. Because the force and interaction potential between the coal and water molecules is greater than that between the coal and methane molecules, the water molecules have an advantage over the methane molecules in capturing the adsorption sites on the internal surface of the coal (Meng et al., 2016; Tang et al., 2015; Wu et al., 2015). Under the effect of competitive adsorption between water and methane molecules, methane molecules on the adsorption sites will be displaced by water molecules, which is equivalent to promoting adsorption methane to free methane. This is the competitive adsorption and displacement desorption effect of water molecules to methane molecules. After pressure water enters into the coal-rock mass, it will enter into the pore channel system, composed of cleats, microcracks and pores in the coal seam (Huang et al., 2016b). As a result, the water pressure of the cleats and pores increases. According to the principle of Langmuir adsorption, free methane will turn into adsorption methane with an increase in pore pressure, resulting in a reduction of the free methane content. However, the displacement desorption effect generates free methane which increases the content of free methane. Therefore, the combined action of the two effects provides a methane source for W-D-M process.
The methane released during the free emission stage of the methane included methane in the pipeline, original free methane and free methane released by spontaneous desorption of the adsorption methane of the coal sample. The methane free emission stage lasted for 44 minutes, and 380 mL of methane was released. The amount of methane released in the first nine minutes was 260 mL, and the methane emission from 9 to 44 minutes was 120 mL. It can be seen in Figure 7 that the methane emission content increased rapidly in the first nine minutes, and the average methane emission rate in the first nine minutes was significantly greater than that afterward. This is because the methane released in the first nine minutes is the compressed methane in the pipeline and original free methane in the coal samples, and the corresponding basis is as follows: (1) The average methane emission rate in the first nine minutes is 2.6 ∼ 60.4 times that of the rate afterward. (2) The volume of the methane inlet pipe with a length of 60 cm and an inner diameter of 0.6 cm is 16.9 mL, and the volume of the methane outlet pipe with a length of 118 cm and an inner diameter of 0.5 cm is 23.1 mL. Therefore, the total volume of the pipe is 40 mL. The temperature is constant during the experiment. According to the state equation of ideal methane, when it comes to the same quality of methane, methane volume is inversely proportional to methane pressure. As a result, the methane volume in the pipeline under 0.53 MPa pressure is 5.3 times as much as that under 0.1 MPa pressure in standard conditions, namely, 40 mL × 5.3 = 212 mL. In addition, when the methane outlet valve is opened, the methane pressure drops rapidly. The initial emission rate of the original free methane in the coal sample is high, and the corresponding volume of the original free methane is 260 mL - 212 mL = 48 mL. (3) During the actual methane collection process, it was found that the methane emission content after 9 minutes showed an obvious regularity, i.e., 10 mL of methane were released every 2 minutes from 9 to 20 minutes, 10 mL of methane were released every 3 minutes from 20 to 30 minutes, and during the last 14 minutes, 20 mL of methane were released and the methane emission increased slowly.

The change of average rate and content of methane emission through time in the stage of the methane free emission.
Desorption methane spontaneously desorbed into free methane with the decrease in methane pressure in the coal sample. As the spontaneous desorption rate of the adsorption methane decreased, the average methane emission rate gradually decreased as well. From 9 to 44 minutes, due to the decrease in the methane pressure, the volume of free methane desorbed spontaneously from the adsorption methane in the coal was 120 mL.
After the methane free emission ended, the W-D-M experiment was conducted. The first process of water injection lasted for 70 minutes. The water injection volume was 3566 mL, and the displacement methane volume was 110 mL. The first water injection lasted for 20 minutes, and the water injection volume was 978 mL and the displacement methane volume was 30 mL. This shows that the free methane of the coal sample increased on the whole, that is to say, the content of the free methane generated by displacement desorption was greater than the free methane consumed by the pore pressure increment. During the whole process of methane displacement by water, the methane in the pipeline is 212 mL, the original free methane in the coal sample is 48 mL, the spontaneous desorption methane of the adsorption methane is 120 mL, and the methane displacement by water is 140 mL. The percentage of the methane source in the W-D-M process is shown in Figure 8.
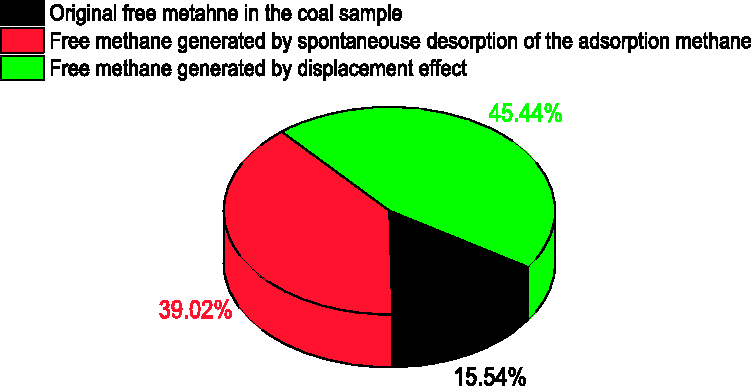
Percentage of the methane source.
The content of the adsorption methane can be calculated by the Langmuir equation, which is expressed as follows:

The adsorption constant a and b, determined by the laboratory test, of the original coal were 20.3065 m³/t and 0.6435 MPa−1, respectively. According to equation (1), it can be calculated that the adsorption methane content of the coal sample under the methane adsorption equilibrium pressure 0.53 MPa is 5.1643 m3/t, and there was 1239.4 mL of adsorption methane content in the coal sample with a weight of 250 g. Therefore, the displacement methane content was 11.3% of the adsorption methane content in the coal sample.
The functional mechanism of pore pressure and its gradient
The relation between pore pressure and methane displacement
When the water inlet valve is closed, water pressure in the pipeline rises with an increase in the water volume in the pipeline. When the water pressure rises to a peak number and the water inlet valve is opened, the methane in the coal sample is displaced by the pressure water, which enters into the coal sample. During the process of methane displacement by water, the curve of the displacement methane content, water injection volume and water injection pressure can be obtained, as is shown in Figure 9. As seen in Figure 9, with an increase in water injection volume, displacement methane content also increases.
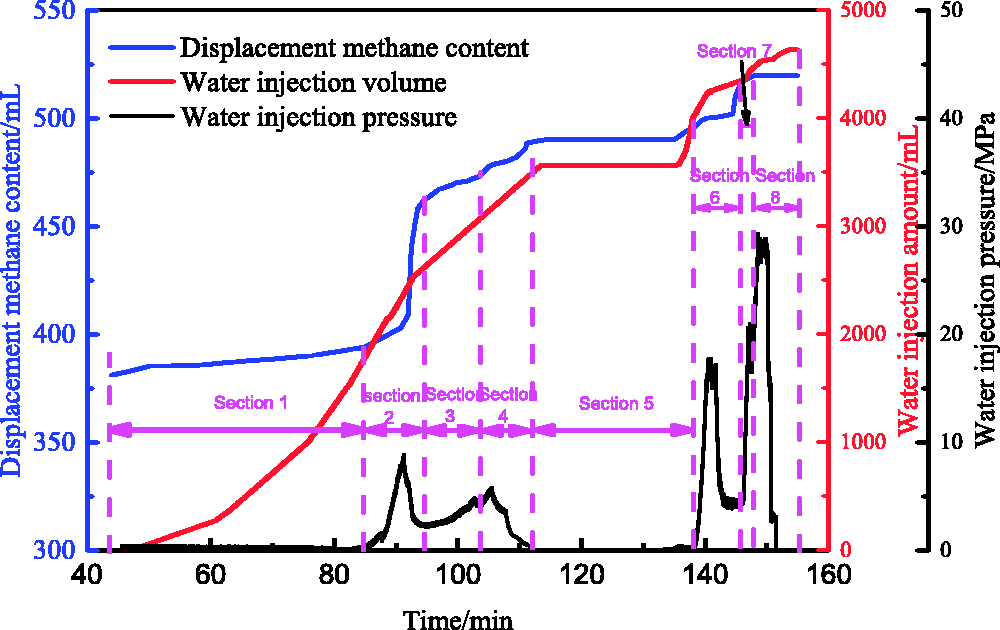
The change of displacement methane content, water injection volume and water injection pressure.
In the process of methane displacement by water, the methane content in a coal sample decreases gradually. It can be seen from Table 2 that with an increase in the peak of the water drive pressure in section 1 to section 6, both displacement methane volume and displacement methane efficiency show an increasing tendency. This is because the increase in water drive pressure can increase the pore pressure gradient rapidly and improve the driving force of displacement methane by water in the coal sample.
The content and efficiency of displacement methane under different displacement conditions.
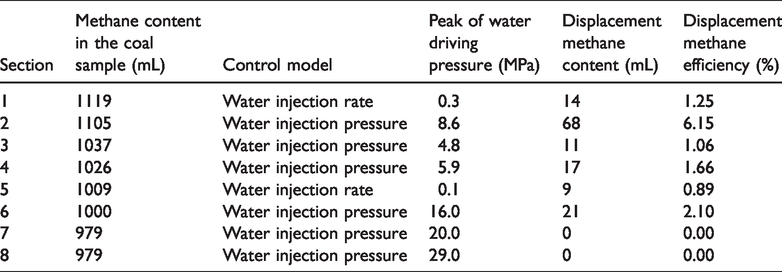
During the process of the second methane displacement by water, when it came to section 7 and section 8, there was still adsorbed methane left in the coal sample. However, during the process of a pore pressure increase from 4 MPa to 20 MPa, and up to 29 MPa, there was no longer any free methane displaced by water. This indicated that an increase in pore pressure and its gradient could not displace methane any longer. This can be explained by the fact that, on the one hand, the methane displacement efficiency of the same pore pressure (gradient) also decreased due to the decrease in the methane content in the coal sample. On the other hand, free methane consumption caused by the pore pressure increase was greater than the free methane generated by the displacement desorption methane. In other words, there was no longer free methane in the coal sample. As a result, there was no longer methane displacement by water. Accordingly, it was speculated that under a given methane content, there was a limit on the displacement pressure in the coal. When the pore pressure is greater than the limit displacement pressure, methane can no longer be displaced by water.
The relation between pore pressure gradient and methane displacement
Because of the water pressure, there was a pressure difference between the top and the bottom end of the coal sample. As a result, a pore pressure gradient, the ratio of the pressure difference to the height of the coal sample, is generated in the coal sample. When the pore pressure gradient exceeds the methane starting pressure gradient, free methane in the coal sample will be driven out by water. It can be seen in Figure 10 that the rate of displacement methane increases with an increase in the pore pressure gradient, which indicates that there is a positive correlation between the rate of displacement methane and the pore pressure gradient.

The change of the rate of displacement methane with the pore pressure gradient.
There was a significant linear correlation between displacement methane content and time in section 2, section 3 and section 4 as seen in Figure 11. The displacement methane rates of section 2 were 48.61 mL/min, 15.77 mL/min, 3.92 mL/min and 1.64 mL/min, respectively, while the linear displacement methane rate of section 3 was 2.84 mL/min, and the linear displacement methane rates of section 4 were 2.28 mL/min, 1.60 mL/min, 1.0 mL/min, respectively, and the linear displacement rate in interval 4 was 2.28 mL/min, 1.60 mL/min and 1.0 mL/min. The following can be seen in Figure 10 and Figure 11(a) and (c): (1) In each section, with the extension of time, the displacement methane content increased linearly, but its increasing amplitude gradually became low, showing that the displacement methane rate decreased gradually over time. This was because the newly free methane was generated in the coal sample by the W-D-M effect. The free methane was promoted to be driven out by the pore pressure gradient generated from the water driving pressure in the coal sample. However, because of the continuous decrease in water driving pressure, the pore pressure gradient and the displacement power is also decreasing. As a result, the displacement methane rate decreases as well. (2) The average displacement methane rate in section 2, section 3, and section 4 was 9.33 mL/min, 2.84 mL/min, and 1.56 mL/min, respectively. This can be explained by the fact that, on the one hand, with the conduction of the W-D-M effect, the methane content in the coal sample was decreasing continuously. As a result, the methane displaced by water decreased, and the displacement methane rate decreased as well. On the other hand, the peak of the pore pressure gradient in section 2, section 3 and section 4 was 86 MPa/m, 48 MPa/m, and 59 MPa/m, respectively, and the greater the pore pressure gradient, the greater the content and rate of the displacement methane (Huang et al., 2016b; Li et al.,2016; Liu et al., 2012).

The change of displacement methane content, water injection pressure and pore pressure gradient through time. (a) Displacement process of section 2 (b) Displacement process of section 3(c) Displacement process of section 4 (d) Displacement process of section 6.
The following can be seen from the displacement process of section 6 in Figure 11(d): (1) With the extent of time, the increase in the content and rate of the displacement methane showed a linear tendency, but the displacement methane rate slowed gradually. The same change rules of the content and rate of displacement methane applied between Figure 11(a) to (d). (2) While the pore pressure gradient was maintained at 50 MPa/m, the displacement methane rate was nearly constant, and the displacement methane content increased linearly. This agreed with the change laws of content and rate of displacement methane in Figure 11(c).
The displacement methane rate (5.5 mL/min) of section 6 in Figure 11(d) was 4.23 times of that of section 5 (1.3 mL/min) as seen in Figure 9. This was because the 160 MPa/m of pore pressure gradient of section 6 provided enough power for the W-D-M effect, but the pore pressure gradient could not be generated in section 5. Therefore, the content and rate of displacement methane in section 6, which was in the stage of a water pressure decrease, were significantly greater than those in section 5 with the low pore pressure gradient.
Time effect of methane displacement by water
When the water was continuously injected into the top end face of the coal sample, there was a pressure difference between the top and bottom end face. With the impact of the pressure difference, a pore pressure gradient was generated in the coal sample. When the pore pressure gradient was greater than the starting methane pressure gradient, the free methane in the coal sample started to flow. The following can be seen in Figure 12: (1) At a certain time after the end of the W-D-M process, along the seepage direction from the water inlet end to the methane outlet end, the pore pressure water increased from 0 to the peak of the water pressure and then decreased from the peak of the water pressure to 0. (2) With the extent of time, the peak of the pore water pressure increased, and the corresponding position of the peak point was also continuously moving to the methane outlet side of the coal sample. This showed that when there was no water injected into the coal sample, the water pressure of the top end of the coal sample was zero. In other words, at the end of the water injection, the water pressure at the top end face became zero. However, along the seepage direction, all the pore pressures at each point of the coal sample would not immediately vanish, and this state would last for a period of time, and then the pore pressure became zero. During this period, net free methane was generated by the combined action of displacement desorption and the methane adsorption effect caused by the increase in the pore pressure. Under the action of the pore pressure gradient, the free methane was driven out by water. The external expression of the aforementioned process was that the water injection stopped at the end of the water inlet end, but there was still methane releasing from the coal sample. It has been shown that there was a time effect during the process of methane displacement by water. During the stage of a water pressure increase in each section as seen in Figure 11, no water was injected into the coal sample, but the displacement methane content was still increasing, which is the external expression of the displacement methane lagging behind the action of water injection. This experimental phenomenon proved that the time effect of methane displacement by water objectively existed.
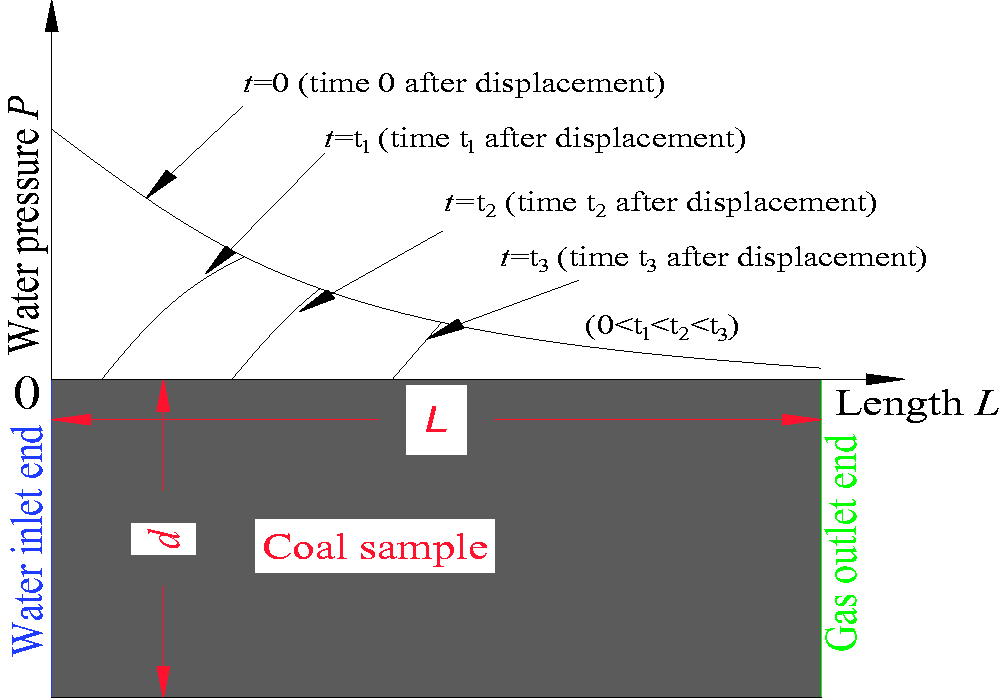
The change of water pressure in the coal sample at different time after displacement.
Mechanism of methane displacement by water
After pressure water enters into the coal sample, the pore pressure of the coal sample will be changed. As a result, the free methane content, adsorption methane content and displacement methane rate will be changed as well, which is shown in Figure 13. Considering the starting pore pressure and limit pore pressure as the dividing points, the process of methane displacement by water can be dived into three stages: (1) when the pore pressure gradient is smaller than the starting pore pressure gradient, free methane in the coal-rock mass will not migrate, so the corresponding displacement methane rate is zero. During this moment, the adsorption methane and free methane in the coal-rock mass are in a state of adsorption equilibrium, and the corresponding methane content is almost invariant. (2) When the pore pressure gradient is greater than the starting pore pressure gradient, the free methane in the coal sample begins to migrate, whose external expression is the W-D-M effect. Initially, the free methane generated by the competitive adsorption and desorption displacement effect is greater than that consumed into adsorption methane caused by the increase in pore pressure, resulting in an increase in free methane content and a decrease in adsorption methane content. However, with an increase in pore pressure, the free methane generated by the competitive adsorption and displacement desorption effect is less than the free methane consumed by the increase in the pore pressure, resulting in a decrease in free methane and an increase in adsorption methane. The external expression of the whole process can be described as follows: initially, the free methane source is sufficient, resulting in the increase in the displacement methane rate; then, the free methane source decreases, resulting in a decrease in the displacement methane rate, until the displacement methane rate becomes zero. The peak of the displacement methane rate slightly lagged behind the peak of the free methane content. This is because there is a phenomenon of the methane displacement lagging behind the water injection in the process of methane displacement by water. (3) When the pore pressure is greater than the limit pore pressure, all the methane in the coal is nearly adsorption methane, and the free methane is nearly zero. At this stage, even if water driving pressure is increased, no methane will be released.

The change of methane content and displacement methane rate with pore pressure in the process of methane displacement by water.
Conclusions
The self-developed pseudo triaxle experimental for methane displacement by water is characterised by good airtightness, and it can be utilized to investigate the laws of methane desorption under different stress load, different water injection volume and different water injection pressure.
Because of the limited adsorption sites on the inner surface of the coal, there is a competitive effect between water and methane for the adsorption sites. The physical adsorption of water on the inner surface of the coal has an advantage over that of methane on the inner surface of the coal, resulting in methane displacement by water in the coal-rock mass. Therefore, during the process of the methane displacement by water, because of this competitive adsorption effect between water and methane, adsorption methane will be transformed into free methane, causing an increase in free methane. After the pressure water enters the coal-rock mass, a pore pressure increase will convert the free methane into adsorption methane, causing the consumption of and decrease in free methane. The free methane generated by the displacement desorption is greater than the free methane consumed by the increase in methane pressure, and the net free methane generated by the combined action of the two effects serves as a methane source for the W-D-M effect.
After pressure water enters into the coal-rock mass, pore pressure is redistributed in the coal-rock mass. At the front of the water seepage, the pore pressure gradient from high to low is distributed along the migration direction of the pore water. The net free methane is driven out because of the pore pressure gradient.
During the process of methane displacement by water, while the water injection volume increases, the displacement methane content increases gradually, but the displacement methane rate firstly increases and then decreases. The process of methane displacement by water is characterized by a time effect, and the process of methane displacement lags behind the water injection process.
There is a starting pore pressure gradient and limit pore pressure in the process of methane displacement by water. When the pore pressure gradient is less than the starting pressure gradient, there is free methane in the coal-rock mass, but it cannot be driven out. When the pore pressure is between the starting pore pressure and limit pore pressure, the free methane can be driven out. When the pore pressure is greater than the limit pore pressure, the methane in the coal rock is almost completely adsorption methane, and free methane cannot be driven out.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by the National Natural Science Foundation of China (No. 51704285 and 51774272) and Transformation of Scientific and Technological Innovation Programs of Higher Education Institutions in Shanxi (TSTAP) (No. 2020CG050), which is gratefully acknowledged. acknowledged.
