Abstract
Biochar adsorbents used to treat different heavy metals in water are efficient and low-cost. Appropriate raw materials, excellent selectivity and detailed adsorption mechanism are of important for research on biochar adsorbents. In this work, konjac starch was dispersed in polyvinylpyrrolidone (PVP) solution to prepare different sizes hydrophilic carbon spheres (HCSs) by hydrothermal synthesis method. Adsorption kinetics of the HCSs towards Pb2+ is described perfectly by the pseudo-second-order equation. With the temperature increasing, adsorption thermodynamics are more consistent with the Freundlich model. The calculated ΔG, ΔH and ΔS shows the adsorption of the HCSs towards Pb2+ is a spontaneous, endothermic and entropy increase process. In addition, HCSs have excellent selectivity for the adsorption of Pb2+ and Cu2+. HCSs prepared from konjac starch make full use of natural biomass resources, they can be used as a potential adsorbent material in treatment on heavy metal ion from water field.
Introduction
In recent years, with the increase of water pollution, biochar adsorbents have attracted more and more attentions due to their outstanding advantages in environmental protection (Wang and Wang, 2018). Biochar is effective for wastewater treatment and soil improvement (Lu et al., 2012). It is called “charred organic matter” which can be prepared by a variety of organic matter (Mohan et al., 2014). The main source of biochar raw materials is agricultural biomass (Tan et al., 2015). Banana peel (Anwar et al., 2010), chestnut shell (Hong et al., 2017), corn straw (Yang et al., 2018) and other agricultural wastes were used to prepare for biochar which haves good adsorption effect on heavy metal ions. Many studies have shown that different biochar has a high adsorption capacity for Pb2+, and the adsorption performance of biochar is closely related to their structure and composition (Jia et al.2019; Li et al., 2020; Niu et al., 2020). In addition, researchers have also contributed to some research on the mechanism of biochar adsorption (Liu et al., 2019; Zhang et al. 2020a, 2020b; Zhao et al., 2019). The main interaction mechanism between the functional groups of biochar and heavy metal ions includes complexation, surface precipitation and cation exchange (Li et al., 2017; Qian and Chen, 2013). Ding et al. (2014) reported that the proportion of the cation exchange accounted for 62% in the adsorption mechanism of bagasse biochar adsorbents on Pb2+. Lu et al. used sludge-derived to prepare biochar adsorbents for Pb2+ adsorption, the proportion of the complexation with carboxyl and hydroxyl groups accounts for 38–42% in adsorption mechanism (Lu et al., 2012). However, the biochar adsorption mechanism is also affected by the raw materials and preparation methods (Li et al., 2017). Hydroxyl, carboxyl and other oxygen-containing functional groups which are used to adsorb heavy metal ions are on the surface of biochar (Qiu et al., 2008). Through physical and chemical methods, the amount and type of functional groups on the surface of biochar can be improved for increasing adsorption capacity on heavy metal ions. The downside is that these raw materials are not the mainly agricultural crops and the yield is very limited.
Starch is a polysaccharide that it can provide humans valuable energy (Zhang et al., 2017) and it is also one of the three major natural polymer (Espíndola-González et al., 2011). Starch has been widely used in various fields, such as papermaking (Tajik et al., 2018), drug synthesis (Chen et al., 2018), adsorbent preparation, etc. (Ma et al., 2018). Among them, the adsorbents prepared by starch have been widely concerned. A series of starch adsorbents are prepared by modifying (Liu et al., 2018), crosslinking (Prakash et al., 2016), and compounding (Yu et al., 2018) the starch. Starch has higher raw material yield and is relatively less affected by seasons and regions. It is a very good potential adsorbent raw material. However, there are few studies on starch biochar and its adsorption performance on heavy metal ions in water.
PVP is a polymer that is often used as a nonionic surfactant due to its non-toxic, biocompatible and good hydrophilicility (Huang et al., 2017a). Gouthaman et al. (2018) used PVP to fully disperse polyaniline and ZnO to prepare an adsorbent that can remove dyes in water. The PVP additive not only allows adsorbent to have a rich pore structure, but also enhances its hydrophilicity. In addition, PVP also contains a large number of amide group, which have a strong interaction with some heavy mental ions (Huang et al., 2017b). Hence, dispersing the konjac starch into the PVP solution can better adjust the size of the adsorbent and improve its adsorption performance during the process of the preparation for starch biochar.
In this work, Using PVP as dispersant and stabilizer and konjac starch as carbon source, hydrophilic carbon spheres was prepared by a simple hydrothermal synthesis and the size of the HCSs was suitable. After preparing HCSs, we carried out a series of experiments and made a discussion and explanation on the Pb2+ absorption mechanism.
Experimental sections
Materials
Konjac starch was purchased from supermarket. Polyvinylpyrrolidone (PVP, AR, K30), lead nitrate (Pb(NO3)2, >99.0%), copper chloride dehydrate (CuCl2•2H2O, >99.0%), magnesium sulfate (MgSO4, >99.0%), calcium chloride (CaCl2, >96.0%) and cadmium chloride (CdCl2•2.5H2O, >99.0%) were of analytical grade without further purification. Water was repeatedly distilled with purification system.
Methods
Preparation of HCSs
In a typical experiment, firstly, 0.043 g of PVP was stirred and completely dissolved in 80.0 mL of distilled water at room temperature for 0.5 h, and then a transparent solution was obtained. Nextly, 2.160 g of konjac starch was added into the transparent solution for dissolving and obtained a transparent konjac starch gel (The weight percentage of PVP and konjac starch is 2 wt.%). Then, the as-prepared gel was transferred into a small Teflon-sealed autoclave and kept at 453 ± 1 K for 10 h to yield brown precipitates. The brown precipitates were washed with distilled water for several times, repeatedly. Finally, the brown precipitates were placed in an oven and dried at 373 K for 10 h and the HCSs were obtained. (According to the mass percentage of PVP and konjac starch, the resulting products were labeled as HCSs-0, HCSs-0.05, HCSs-2 and HCSs-10.)
Adsorption experiments
0.050 g of the HCSs was added into 50.0 mL, 10 mg/L of Pb2+ solution. The suspension was sucked by the syringe and filtered by a filter which was equipped with a 0.45-μm membrane (Polytetrafluoroethylene, Shanghai xinya purification equipment factory, China.) at set intervals. Atomic Absorption Spectrometry (TAS-990F, Beijing Pui General Instrument Co. Ltd., China) was used to detect Pb2+ solution concentration at different moment. Adsorption kinetics experiments were performed at room temperature (292 K). Adsorption isotherms were obtained by varying initial Pb2+ concentration from 5 mg/L to 25 mg/L. Among them, a case study of the adsorption of 10 mg/L of Pb2+ solution, different adsorption isotherms were obtained by changing adsorption temperature (such as 292 K, 313 K, 323 K and 333 K). In addition, the adsorption thermodynamic parameters were calculated.
The amounts of Pb2+ adsorbed onto the surface of HCSs at equilibrium time and at any time are calculated by the following formula:
Where Qe,t (mg/g) is the adsorption amount of HCSs towards Pb2+ at equilibrium (Qe) and at a certain time (Qt), V(L) is the volume of Pb2+ solution, C0 (mg/L) is Pb2+ starting solution concentration, Ce,t (mg/L) is the Pb2+ concentrations at equilibrium (Ce) and at some point (Ct) and m (g) is the mass of the HCSs.
Other metal ion adsorption experiment
0.050 g of HCSs-2 was added to 50.0 mL, 15 mg/L of divalent metal ions (Cu2+/Mg2+/Ca2+/Cd2+) solution under stirring condition. The suspension is sucked by the syringe and filtered by a filter that was equipped with a 0.45 μm membrane (PTFE, Shanghai xinya purification equipment factory, China.) at fixed intervals. Atomic Absorption Spectrometry (TAS-990F, Beijin Pui General Instrument Co. Ltd., China) was used to determine the Cu2+/Mg2+/Ca2+/Cd2+ concentration at different time.
Characterization
The structures and morphology of the HCSs were observed with scanning electron microscope (SEM, FEI, NanoSEM450, 15 kV, America). The crystal structure of the HCSs was studied using X-ray diffraction (XRD, Bruker, D8-Advance X-Ray diffractometer, CuKα, λ = 1.5406 Å, Germany). The contact angle of HCSs film was measured by a contact angle meter (CAM, Krüss, DSA100, Germany). Chemical bonds of the HCSs were measured by Fourier transform infrared spectroscopy (FT-IR spectrum, Bruker, TENSOR 27, Germany).
Results and discussion
Characterization of HCSs
Figure 1 shows XRD pattern of HCSs-2, FT-IR spectrum of HCSs-2, SEM image of HCSs-2 film and contact angle image of HCSs-2 film. The XRD pattern of HCSs-2 can clearly show a broad peak with low peak intensity in Figure 1(a). The result proves that the as-prepared HCSs-2 is a typical disordered glassy polymers of carbon (Yang et al., 2018). The FT-IR spectrum is shown in Figure 1(b), the existence of hydroxyl group was confirmed by the broad and strong absorption peak of the O-H stretching vibration at 3420 cm−1 (Tran et al., 2017). Hydroxyl groups can increase the hydrophilicity of carbon spheres which are beneficial to Pb2+adsorption. The peaks at 1650 cm−1, 1460 cm−1, 1420 cm−1 and 1290 cm−1 are the C = O stretching vibration, the -CH2- torsion out of plane, the -CH2- bending vibration and the -C-N- stretching vibration, respectively (Kaur et al., 2018). The three groups belong to the PVP on the surface of HCSs-2. Figure 1(c) and (d) display that HCSs-2 is evenly dispersed on the coverslip, its contact angle is 32° (<90°), which means that HCSs-2 exhibits good hydrophilicity.

Characterization of HCSs-2 and HCSs-2 film (a) XRD pattern of HCSs-2 (b) FT-IR spectrum of HCSs-2 (c) SEM image of HCSs-2 film (d) contact angle image of HCSs-2 film.
The SEM images of HCSs including different PVP amount are shown in Figure 2. The average particle sizes of these HCSs are analyzed by Image pro plus (IPP, version 6.0). The average diameters of HCSs-0, HCSs-0.05, HCSs-2 and HCS-10 were calculated as 3.22 ± 2.03 μm, 3.22 ± 1.87 μm, 3.37 ± 1.67 μm and 5.35 ± 1.27 μm, respectively. These data indicate that PVP affects the size distribution of HCSs. The diameters of the HCSs increase with the increasing addition of PVP. It is possible that konjac starch gel is uniformly dispersed in water by PVP, in the process of hydrothermal synthesis, PVP acts as dispersant and stabilizer, which can effectively prevent the aggregation of carbon spheres during carbonization, after PVP was added, the diameters of HCSs are affected by the size of the PVP micelles due to the konjac starch gel encapsulated by PVP micelles.

SEM images of HCSs (a) HCSs-0 (b) HCSs-0.05 (c) HCSs-2 (d) HCSs-10.
Adsorption of Pb2+
Effect of PVP
The adsorption capacities of HCSs including different PVP amount towards Pb2+ are shown in Figure 3. Among the HCSs with different PVP amount, the adsorption capacity of HCSs-2 is the highest up to 10.826 mg/L. It is probably that the adsorption capacity is inversely proportional to the size of the average diameter of HCSs with different PVP amount while HCSs-2 has a smaller average diameter, meaning it has a larger surface area and can provide more active sites.

Adsorption capacity of HCSs including different PVP amount towards Pb2+.
Adsorption kinetics
Pseudo-first-order equation (Ragren, 1898) and pseudo-second-order equation (Ho and Mckay, 1999; Li et al., 2019) are the most commonly used linear equations to describe adsorption kinetics (Javed et al., 2018). In this work, the two equations will be used to describe the adsorption kinetics of Pb2+ absorbed on the surface of HCSs. The pseudo-first-order equation (equation (2)) and the pseudo-second-order equation (equation (3)) are calculated using the following formula:
Where K1 (min−1) and K2 are the equilibrium constant of the pseudo-first-order equation and the pseudo-second-order equation, and t (min) is the adsorption time. Kinetic parameters of HCSs-2 are listed in Table 1.
Kinetic parameters for Pb2+ adsorbed onto the surface of HCSs-2.
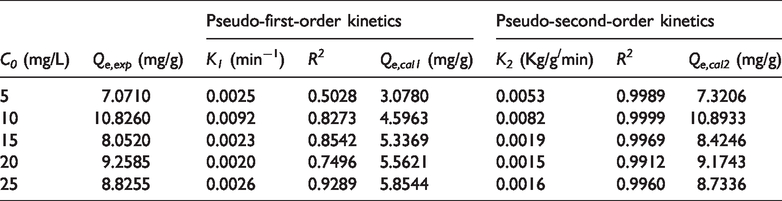
Where Qe,exp is the actual saturated adsorption capacity, Qe,cal1 is the saturation adsorption capacity of the pseudo-first-order equation, Qe,cal2 is the saturation adsorption capacity of the pseudo-second-order equation, R2 is correlation coefficients.
As shown in Table 1, the calculated value of R2 (>0.99) is more close to 1. The results demonstrate that the adsorption kinetics of HCSs-2 can be described well by the pseudo-second-order equation. In addition, all the value of Qe,cal2 are closer to Qe,exp than that of Qe,cal1, which fully demonstrates that the pseudo-second-order equation is more consistent with adsorption kinetics. The pseudo-first-order equation holds that the physical adsorption is the control step of reaction rate and can accurately describe the adsorption process for the initial 20–30 min stage. The equation only describes part of the adsorption process instead of the entire process (Ajmal and Piergiovanni, 2018). The pseudo-second-order equation is very suitable to describing the entire adsorption process. At C0=10 mg/L, Qe,exp reached a maximum of 10.826 mg/g. This phenomenon may explained that when C0 (<10 mg/L) is little, the adsorption driving force is insufficient and Qe is less, while in an opposite picture (C0>10 mg/L), Qe decreased due to competitive adsorption.
The surface of HCSs-2 is rich in hydroxyl groups and the hydroxyl group has two pairs of lone pair electrons. Pb2+ is positively charged and can be adsorbed by these pairs of electrons. In addition, -O− can be formed by ionization of hydroxyl groups and has stronger electronegativity to adsorb more Pb2+. Therefore, Pb2+ adsorbed onto HCSs-2 should be chemical adsorption, which is consistent with the fitting results in Table 1.
Equilibrium isotherms
Adsorption isotherms are fitted by fourth-order polynomial fitting lines. Adsorption isotherms of HCSs-2 at different temperatures are shown in Figure 4. With the temperature increases, the adsorption isotherms change gradually type IV into type II. This may be single layer chemisorption at first stage during the adsorption process. After single layer adsorption of Pb2+ on the surface of HCSs-2, Qe decreased slightly due to competitive adsorption. With the temperature increases, hydroxyl ionization forms a large amount of -O−. Since -O− has strong electronegative, it still has the ability to attract Pb2+ after single layer adsorption of Pb2+ on the surface of HCSs-2, which forms multilayer adsorption. The adsorption process is shown in Figure 5. In theory, the value of Qe at certain temperature should go up again significantly. The conjecture is well confirmed in the 323 K and 333 K adsorption isotherms.
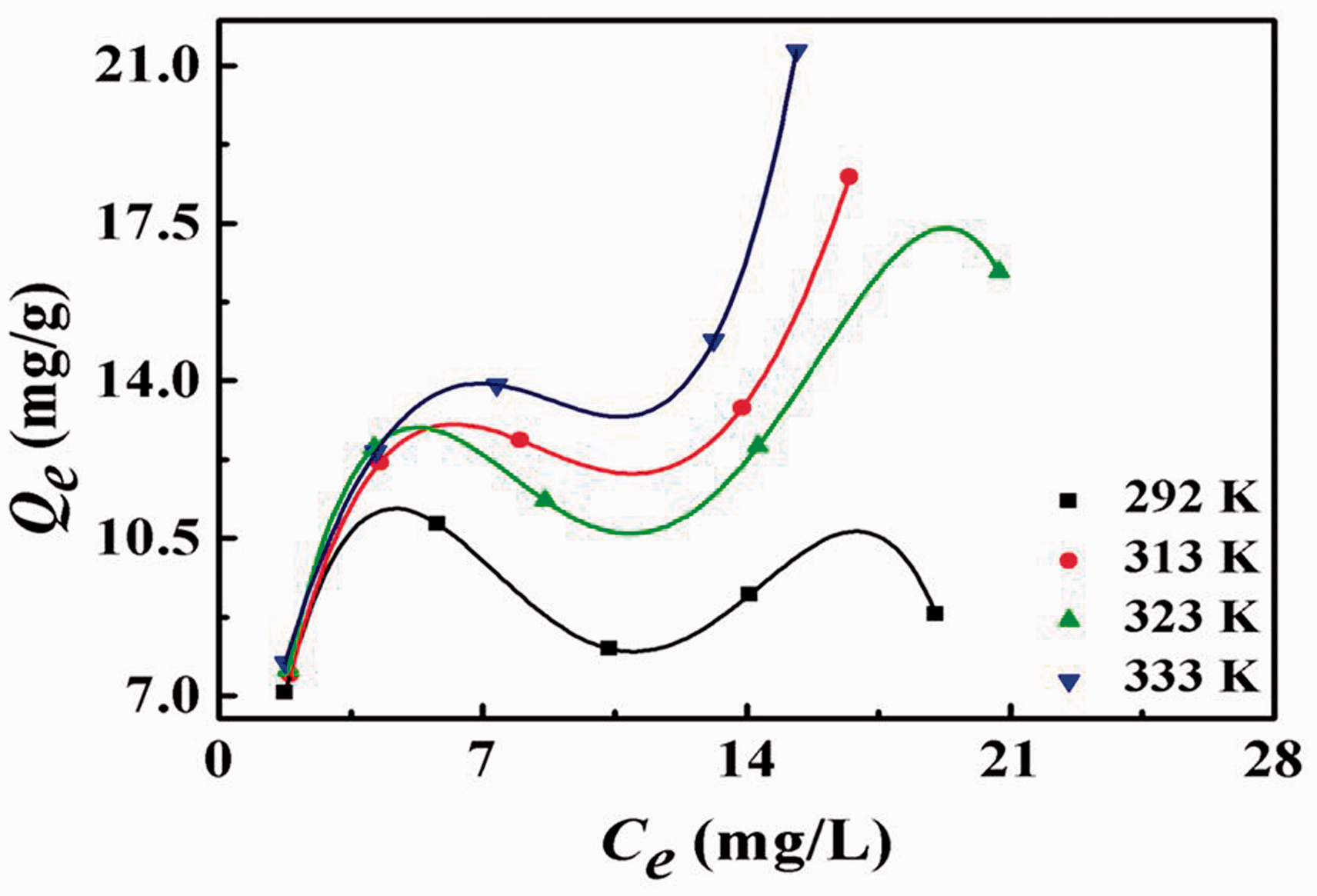
Adsorption isotherm of Pb2+ adsorbed onto HCSs-2 at different temperatures.
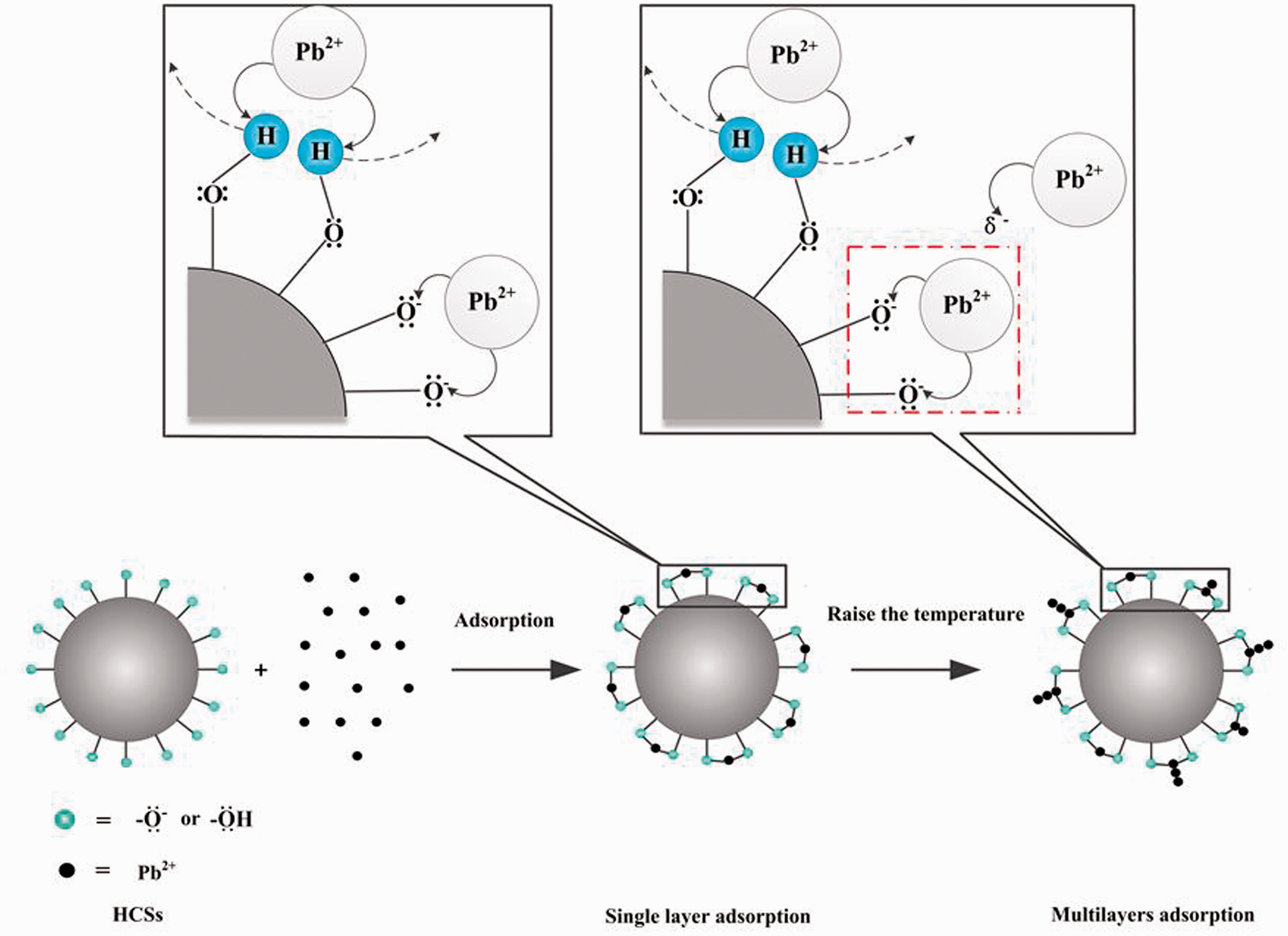
Adsorption mechanism of Pb2+ adsorbed onto HCSs-2.
Since the temperature is not high enough, the hydroxyl group dissociates less -O− and the effect of competitive adsorption. Therefore, Qe does not continue to rise in the high Ce/C0 region at 292 K. The adsorption isotherm at 313 K is in the transitional stage. Qe increases in the higher Ce/C0 interval, but the fitted line shows a decreasing trend.
The Langmuir model and Freundlich model are used to describe adsorption isotherms. Their expressions are as follows:


Where Qm represents the maximum adsorption capacity, KL and n are empirical coefficient in adsorption process, KF is a constant indicating the adsorption capacity of the adsorbent.
The fitting results are shown in Table 2. As the temperature increases, the R2 of the Langmuir model decreases, while the R2 of the Freundlich model increases. At 292 K, the R2 of the Freundlich model is only 0.2066 and that of Langmuir model is 0.9802. Adsorption isotherms can be better described by the Langmuir model. The Langmuir model assumes that the adsorption process is monolayer and uniformly adsorbed (Sahnoun and Boutahala, 2018), which is consistent with the adsorption kinetic results. However, when the temperature rises to 333 K, the R2 of the Freundlich model increases to 0.8600 and the R2 of the Langmuir model drops to 0.8362. The adsorption form is mainly monolayer adsorption at a lower temperature of 292 K and is mostly multilayer adsorbed at a higher temperature of 333 K, which is proved by the fitting data. The adsorption isotherms change gradually type IV into type II and Qe also improves more than double because of the existance of multilayer adsorption.
Equilibrium parameters for Pb2+ adsorbed onto the HCSs-2.

In addition, the value of n in the Freundlich model can be used as an important reference for the degree of adsorption difficulty (Bazargan-Lari et al., 2014; Wang et al., 2018). At 292 K, the value of n is 13.0208, which is much greater than 2. As the temperature rises to 333 K, the value of n gradually decreases to 2.5742. The value of n is smaller, the adsorption is easier. The value of n decreases with the increase of the temperature, indicating that the adsorption is favorable at higher temperature.
Adsorption thermodynamic
Gibbs free energy (ΔG) reflects the direction of adsorption, enthalpy change (ΔH) is determined to exothermic or endothermic of adsorption process and entropy change (ΔS) reflects degree of confusion on adsorption system. The thermodynamic parameters are calculated using the following equation:

The van't hoff equation is used to calculate the adsorption thermodynamic parameters. The values of ΔH and ΔS can be calculated from the intercept and the slope of the linear plot of lnKL versus 1/T. The linear expression is as follows:

Where R is a universal gas constant (8.314 J/mol/K) and T is adsorption temperature (K = 273+°C)
As shown in Table 3, the value of ΔG is negative (<0) at different adsorption temperatures and decreases with the increase of adsorption temperature. The result prove that the adsorption of HCSs-2 towards Pb2+ is a spontaneous process. As the adsorption temperature increases, the spontaneous process becomes easier and easier. The calculation result shows ΔH > 0, meaning that the adsorption process of HCSs-2 needs to absorb heat (Munagapati and Kim, 2017). Increasing the adsorption temperature is favorable to the adsorption process, which is consistent with the decrease of n and ΔG.
Adsorption thermodynamic parameters of Pb2+ absorbed onto HCSs-2.

The fitting result in Table 3 shows that ΔS is positive (>0), which proves that the adsorption of HCSs-2 is a process in which the confusion degree of the surface of HCSs-2 increases, if the surface sedimentation or complexation occurs alone, the confusion degree should be reduced, in other words, ΔS is negative (<0). Meanwhile, if surface precipitation and complexation occurs on the surface of HCSs-2, the surface will be covered by precipitates or complexes. Adsorption sites will be separated from Pb2+ by the cover layer. When the temperature rises, Qe should fall or be invariant rather than rise. However, as shown in Figure 5, Qe increases with enhancement of adsorption temperature. This fully shows that the main adsorption mechanism of HCSs-2 towards Pb2+ should be cation exchange. The proportion of surface precipitation and complexation is small or absent. This is consistent with the hypothesis that hydroxyl or -O- adsorbs Pb2+.
Other metal ion adsorption experiment
As shown Figure 6, HCSs-2 does not absorb Mg2+, Ca2+ and Cd2+, at the same time, HCSs-2 has higher adsorption capacity to Pb2+ than Cu2+. The results demonstrate that the HCSs-2 has selectivity to Pb2+ and Cu2+. Deng et al. (2017) consider that C = O (N-C = O and –COOH) is a key functional group for adsorbing Cu2+. The surface of HCSs-2 contains –CONH- (from PVP), so Cu2+ can be adsorbed by the HCPs-2. But on the surface of HCSs-2, no functional group has adsorption effect on Mg2+, Ca2+ and Cd2+. In addition, the order of the electronegativity of these elements is Pb>Cu>Cd>Mg>Ca in turn (2.33 for Pb, 1.90 for Cu, 1.69 for Cd, 1.31 for Mg and 1.00 for Ca) (Kinraide and Yermiyahu, 2007). Park et al. (2016) think that Pb2+ is a stronger Lewis acid while Cd2+ is a weaker Lewis acid. The ability of HCSs-2 providing electrons is not extremely strong and only corresponds to a weak base. Therefore, the more acidic Pb2+ and Cu2+ can be adsorbed by HCSs-2, but the weaker Mg2+, Ca2+ and Cd2+ cannot.

Adsorption capacity of HCSs-2 to different divalent metal ions.
Conclusions
HCSs were successfully prepared by hydrothermal synthesis method. The presence of numerous hydroxyl groups on the surface of HCSs might play a vital role in the adsorption process. These hydroxyl groups could be ionized to form O− with increasing adsorption temperature, which forms a multi-layered adsorption to improve the adsorption capacity. The adsorption isotherm gradually shifts type IV to the type II. HCSs exhibits a certain adsorption capacity for Pb2+ and Cu2+, but no adsorption ability for Mg2+, Ca2+ and Cd2+. HCSs can be used as an exclusive biochar adsorbent for the treatment on heavy metal ions from waste.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 51309074), the Natural Science Foundation of Hebei Province (No. B2016202271) and the “Thirteen Five” national key R & D project (No. 2016ZX05025-004–006).



