Abstract
The adsorption of Rhodamine B onto CT269DR resin has been studied through static and dynamic experiments. The effects of shaking speed, resin dosage, and pH on adsorption were investigated by static experiments. The external mass transfer rate remains substantially unchanged when the shaking speed exceeds 160 r min−1. The optimal pH range is 5–8, and an increase of resin dosage can directly improve the percentage of removal of Rhodamine B. The equilibrium isotherm data of Rhodamine B on CT269DR resin fit the Langmuir adsorption isotherm well. The thermodynamics parameters, ΔH = 69.93 kJ mol−1, ΔS = 326.73 J mol−1 K−1, and ΔG < 0, demonstrate that the adsorption of Rhodamine B onto CT269DR resin is spontaneous and endothermic. The pseudo first-order kinetic model can be successfully used to represent the adsorption process and the activation energy is 25.7 kJ mol−1. The dynamic experiments show that the breakthrough point is advanced when the flow rate increases and the bed adsorption capacity increases with increasing temperature. Furthermore, the desorption using the solution of 2% NaOH is suitable for desorption and reusing process, and scanning electron microscope (SEM) and fourier transform infrared spectroscopy (FT-IR) analysis reveals that used resin has good wear resistance and chemical stability. The results confirm that CT269DR resin can be employed as an efficient adsorbent for the removal of Rhodamine B from wastewater.
Introduction
The rapid development of the modern printing and dyeing industry has produced a large amount of industrial wastewater which is difficult to be treated. One of the main pollutants is Rhodamine B, widely used in food, leather, textile, and cosmetic industries (Pourreza et al., 2008). Rhodamine B is a non-biodegradable organic substance with potential toxicity, mutagenicity, and carcinogenicity, therefore it is harmful to water quality and aquatic plants (Jain et al., 2007; Yu et al., 2013).
At present, the most commonly used methods for the dye wastewater treatment include electrochemical treatment (Baddouh et al., 2018; Parsa and Jafari, 2017), chemical oxidation (Ozcan et al., 2009; Sethi et al., 2019; Turhan and Turgut, 2009), photodegradation (Ma et al., 2016; Magdalane et al., 2017), adsorption (Gao et al., 2019; Lafi et al., 2019; Saruchi et al., 2019), and biological methods (Gan et al., 2016). Adsorption techniques have potential for removing organics from wastewater due to their high efficiency and ability to separate a wide range of chemical compounds (Adibmehr and Faghihian, 2018; Mittal et al., 2016; Petkovic et al., 2019). The ordinary used adsorbents are activated carbon, molecular sieves, metal organic frameworks, and other adsorbent materials (Dos Reis et al., 2011). The adsorption method has the characteristics of good adsorption capacity, simple operation, and wide application range which has been widely used in the practical work of dye wastewater pollution control. He et al. (2014) investigated the adsorption characteristics of UiO-66 (a Zr-containing metal-organic framework formed by terephthalate) for Rhodamine B, such as isotherms, kinetics, and thermodynamics. The thermodynamic analysis shows that the adsorption process of Rhodamine B on UiO-66 is more favorable at higher temperatures. UiO-66 can be regenerated by desorbing in DMF solution with ultrasonic for 1 h. UiO-66 can keep good performance for at least six cycles of adsorption/desorption. Jain et al. (2007) utilized activated carbon and rice husk as potential adsorbents to remove Rhodamine B from wastewater. The adsorption studies were carried out at 40, 50, and 60°C, and the effects of pH, temperature, amount of adsorbents, and concentration of adsorbate on the adsorption were measured. The adsorption isotherm constants were employed to calculate thermodynamic parameters like Gibb’s free energy, change in enthalpy and entropy.
Nevertheless, the limited selectivity and adsorption capacity of the above adsorbents hamper their industrial use. Ion exchange resin has such benefits as reuse, no secondary pollution, and high adsorption capacity. Therefore, in this paper we use ion exchange resin to adsorb Rhodamine B from wastewater which has rarely been reported. Through static and dynamic experiments, the optimum parameters that influence the adsorption and desorption were determined. The adsorption thermodynamics and kinetics of Rhodamine B were also investigated to provide theoretical support for solid–liquid adsorption research and dye wastewater treatment.
Experimental
Materials and methods
Materials: Resin KIP210 (Kairui Chemical Co., Ltd); resin D201 (Shaanxi Lanshen Special Resin Co., Ltd); resin CT269DR (Purolite Co., Ltd); resin IRA402, IRA-900, 719, 717, and Rhodamine B (Shanghai Aladdin Co., Ltd). Sodium hydroxide and hydrochloric acid (Sinopharm Chemical Reagent Co., Ltd).
Equipments: Electric thermostatic water bath (BSG-24, Shanghai Yiheng Scientific Instrument Co., Ltd); high pressure constant current pump (P230II, Dalian Elite Analytical Instrument Co., Ltd); water bath constant temperature oscillator (SHA-B, Guohua Instrument Factory); Visible spectrophotometer(752PRO, Shanghai Lingguang Technology Co., Ltd); pH acidity meter (pHS-25, Shanghai Tianda Instrument Co., Ltd).
Analysis method
Rhodamine B solution at concentrations of 2, 4, 6, 8, and 10 mg l−1 was prepared with deionized water; the absorbance of the Rhodamine B solution was measured with a visible spectrophotometer at a wavelength of 554 nm and zeroed with deionized water. The standard curve of Rhodamine B absorbance (y) to concentration (x, mg l−1) is as follows: y = 0.0544x + 0.0076; the determination coefficient of the curves is 0.9991.
Selection of the resins
Different kinds of resin, 0.1 g each, and 100 ml of 100 mg l−1 Rhodamine B solution were added in 250 ml flasks. The flasks were sealed and shaken at 160 r min−1 and 298.15 K in a water bath constant temperature oscillator, and the pH of this solution was neutral. The experiment was carried out for 5 h to ensure that equilibrium was reached. Samples were analyzed and the equilibrium adsorption capacity of Rhodamine B per unit mass of adsorbent, qe (mg g−1) was calculated using equation (1) as follows
Static adsorption
All static adsorption experiments were carried out by mixing the different mass of resin with 100 ml of Rhodamine B solution at the desired concentration and pH in the 250 ml flasks. The flasks were then sealed and shaken at constant temperatures and shaking speeds in a water bath constant temperature oscillator. The samples were taken and analyzed by visible spectrophotometer as described, and the adsorption capacity of Rhodamine B per unit mass of adsorbent, qt (mg g−1), was calculated according to equation (2). The percentage of removal of Rhodamine B, Re, was calculated by the following equation
Dynamic adsorption
The glass columns were used for dynamic studies. The column height is 100 mm, and the diameter is 5.0 mm. The Rhodamine B solutions were passed through the column filled with the resin at different flow rates and temperatures to preconcentrate Rhodamine B. Then, the retained Rhodamine B was eluted with different concentrations of NaOH. The bed adsorption capacity, q (mg g−1), can be calculated from the integration of the breakthrough curve as follows (Rao et al., 2011)
Results and discussion
Selection of resin
The experimental results exhibit that the sequence of the equilibrium adsorption capacity of Rhodamine B on the seven resins is as follows: CT269DR > IRA402 > IRA-900 > 717 > D201> KIP210 > 719 and the values are 93.28, 20.00, 19.18, 15.61, 15.41, 14.83, and 4.33 mg g−1, respectively. CT269DR resin has the maximum adsorption capacity of the selected resin for Rhodamine B, which was used to adsorb Rhodamine B from the aqueous solution in this experimental study. The properties of the resins are listed in Table 1.
Typical physical and chemical characteristics of the used resin. a

SAC: strong acid cation; SBA: strong base anion; S-DVB: styrene-divinylbenzene; TMA: trimethylammonium.
aInformation provided by the manufacturer.
Influence factors of static adsorption
Effect of shaking speed
The effect of shaking speed was investigated in the shaking speed value range 60–210 r min−1 with 100 ml of 100 mg l−1 Rhodamine B solution containing 0.1 g CT269DR resin at 298.15 K. The adsorption rate curves at different shaking speeds were plotted and depicted in Figure 1. As displayed in the figure, the adsorption rate increases with an increase in shaking speed from 60 to 160 r min−1 and then perform essentially constantly. The above effect may be ascribed to the decrease in the thickness of the boundary layer around the resin particles because of an increase in the degree of mixing (Albadarin et al., 2012). When the mixture was shaken, the resin solid particles move rapidly in the solution, which increases the concentration of Rhodamine B near the surface of the solid particles, possibly reaching levels near the total concentration (Argun et al., 2007). As the diffusion of Rhodamine B to the boundary layer between the resin particles and the surrounding solution increases with an increase in shaking speed from 60 to 160 r min−1, the external mass transfer rate of the Rhodamine B increases and the equilibrium reaches faster, whereas the external mass transfer rate remains substantially unchanged by further increasing the shaking speed, that is the external diffusion resistance is maintained constant. Thus, the shaking speed was fixed at 160 r min−1 for further studies.
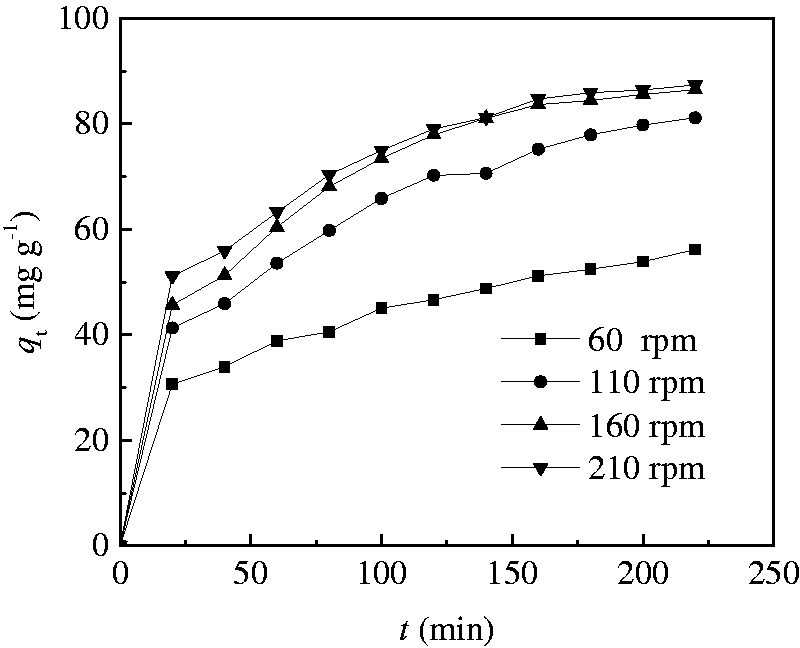
Effect of shaking speed on adsorption of Rhodamine B (C0 =100 mg l−1, resin quantity= 0.1 g, T = 298.15 K).
Effect of resin dosage
The effect of resin dosage was investigated by varying the amount of CT269DR resin from 0.05 to 0.8 g for adding to 100 ml of 100 mg l−1 Rhodamine B solutions shaken at 160 r min−1 and 298.15 K. The result is exhibited in Figure 2, which shows the removal of Rhodamine B as a function of resin dosage. It demonstrates that an initial increase of resin dosage can directly improve the percentage of removal of Rhodamine B because of the more active sites for adsorption. However, the percentage of removal tends to be consistently near to 99.2% when the resin dosage is beyond 0.3 g.

Effect of resin dosage on adsorption of Rhodamine B (C0 =100 mg l−1, T = 298.15 K, shaking speed = 160 r min−1).
Effect of pH
The pH value of the solution is a significant factor in the whole adsorption process, which is helpful to control the adsorption capacity according to the ionic form of dye solution (Singh et al., 2018). The adsorption experiments were carried out in the pH value range 1–14 with 100 ml of 100 mg l−1 of Rhodamine B solution containing 0.2 g CT269DR resin at 298.15 K, and the result is displayed in Figure 3. The adsorption capacity of Rhodamine B increases from 32.5 to 49.1 mg g−1 when the pH varied from 1 to 5 and then maintains almost unchanged with the pH in the range of 5–8. However, the adsorption capacity decreases with increasing the pH from 8 to 14. This is mainly due to the addition of NaOH in the solution to convert the resin used as adsorbent from hydrogen form to sodium form. Moreover, the zwitterionic form of Rhodamine B in aqueous solution can be induced by the addition of NaOH, which expands the aggregation of Rhodamine B to form a bulky molecule, and this form of Rhodamine B is not conducive to adsorption (Wang et al., 2018). Therefore, the optimal pH range is 5–8.

Effect of pH on adsorption of Rhodamine B (C0 =100 mg l−1, resin dosage = 0.2 g, T =298.15 K, shaking speed = 160 r min−1).
Adsorption isotherm
Adsorption equilibrium
Adsorption isotherms were carried out at the temperature of 298.15, 308.15, and 318.15 K, respectively. The CT269DR resin (0.1 g) and 100 ml of Rhodamine B solution with different initial concentrations (50, 100, 150, 200, 250, and 300 mg l−1) were put into each Erlenmeyer flask and shaken at 160 r min−1 for 5 h. The samples were analyzed by visible spectrophotometer as described in the “Analysis method” section. Each equilibrium concentration of Rhodamine B was calculated and the corresponding equilibrium adsorption capacity was obtained. Therefore, the adsorption equilibrium curves at different temperatures were acquired by plotting the data of qe versus Ce and displayed in Figure 4.
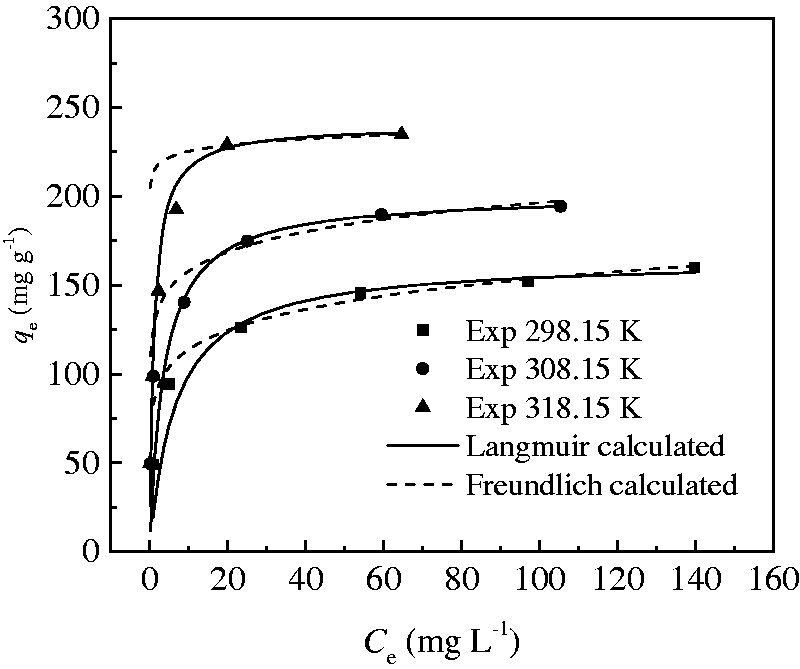
Experimental data of adsorption isotherm and non-linear fitting of Langmuir and Freundlich.
Adsorption isotherm is important to describe the ability of the adsorbent and how adsorbate interacts with absorbent. With the aim of optimizing the adsorption system to remove Rhodamine B from solution, it is critical to analyze the most appropriate correlations for equilibrium curve. There are many adsorption isotherm models available for describing experimental adsorption equilibrium data, such as Langmuir, Freundlich, Sips, Redlich–Peterson, Toth, Dubinin–Radushkevich, and Temkin isotherms. In this work, the equilibrium adsorption experimental data for removing Rhodamine B from solution on CT269DR resin were evaluated by the Langmuir and Freundlich isotherms, which are the two most commonly used isotherm models (Crini et al., 2007). The non-linear forms of two models were expressed by the following equations (5) and (6), respectively (Song et al., 2019; Yang et al., 2015)



Parameters of adsorption isotherm models.
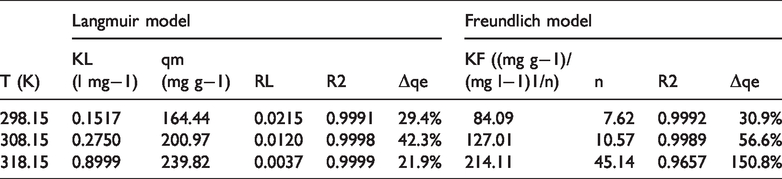
Figure 4 shows that Langmuir equation has the better consistency with the experimental data in this study. According to Table 2, the R2 values of the Langmuir model are all more than 0.999, and the normalized standard deviation (Δqe) of the Langmuir model at each temperature is lower than the value of the Freundlich model. Thus, the adsorption equilibrium data of Rhodamine B on CT269DR resin correctly fit the Langmuir relation. The Langmuir model is based on the assumption that the adsorbate molecules form a monomolecular layer on adsorbents with homogeneous surface and there is no interaction between the adsorbed molecules in the plane of the surface (Hu et al., 2011; Norouzi et al., 2018; Saruchi et al., 2019). Furthermore, the Langmuir model can be used to estimate the maximum adsorption capacity and it only corresponds to the monolayer surface of the adsorbent. Therefore, the adsorption isotherm modeling reveals the monolayer coverage of Rhodamine B at the outer surface of resin and the homogenous active sites within the adsorbent. The theoretical maximum adsorption capacities, qm, were correlated to be 164.44, 200.97, and 239.82 mg g−1 from the Langmuir model at 298.15, 308.15, and 318.15 K, respectively, which are in accordance with the experimental data.
The values of KL increase with the increase in temperature which demonstrates the endothermic adsorption process (Naushad et al., 2016). In addition, the fundamental characteristics of the Langmuir model can be expressed by the dimensionless separation factor RL as follows (Albadarin et al., 2012).

Thermodynamic parameters
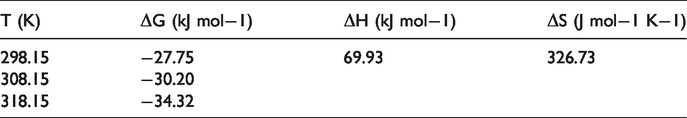
Adsorption thermodynamics is able to illustrate the in-depth message in terms of structural and inherent energy change of adsorbents after adsorption. It also describes the mechanism involved in the adsorption process (Chen et al., 2014). The values of thermodynamic parameters for the adsorption of Rhodamine B on CT269DR resin, such as enthalpy change (ΔH), entropy change (ΔS), and Gibbs free energy change (ΔG), can be calculated by the Van’t Hoff equation as follows (An et al., 2017; Lin et al., 2012)



The values of

Plot of
Thermodynamic parameters for the adsorption of Rhodamine B on CT269DR resin.
Adsorption kinetics
The study of adsorption kinetics is significant to clarify the adsorption dynamics in the field of the order of rate constant (Cazetta et al., 2011; Norouzi et al., 2018). The adsorption kinetics experiments were studied as follows: The CT269DR resin (0.2 g) and 100 ml of Rhodamine B solution with initial concentration 100 mg l−1 were added in the flask. The flask was shaken at different times and temperatures of 298.15, 308.15, and 318.15 K, and the shaking speed was fixed at 160 r/min. The samples were taken and analyzed at approximate intervals. The adsorption capacity of the CT269DR resin, qt (mg g−1), was calculated by equation (2). The experimental data of qt versus t were plotted to obtain the adsorption rate curves at different temperatures and displayed in Figure 6. As can be seen in the figure, the adsorption rate and equilibrium adsorption capacity both increase with the increase of temperature, which also indicates that the adsorption of Rhodamine B on CT269DR resin is endothermic.
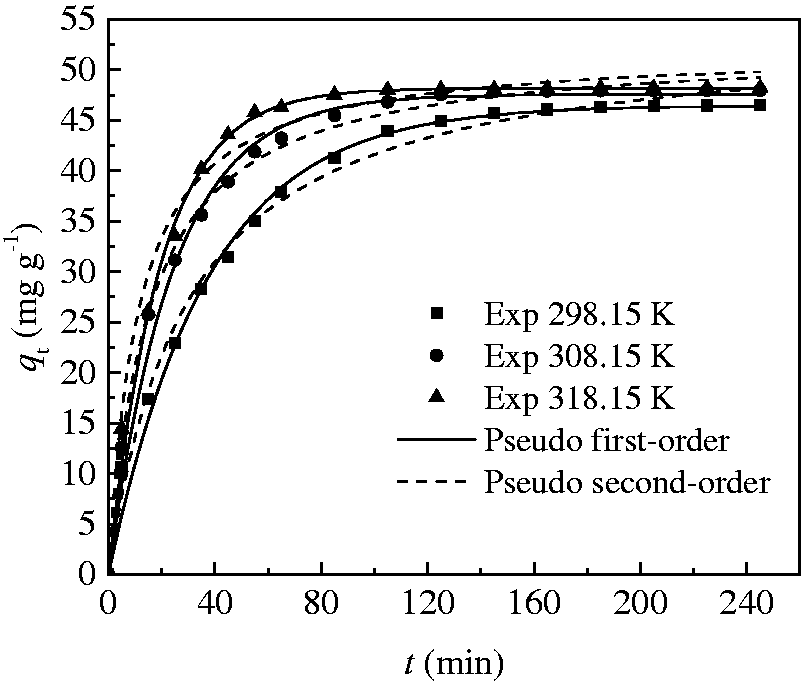
Experimental data of adsorption kinetics and non-linear fitting of kinetics model for Rhodamine B adsorption. (C0 =100 mg l−1, resin dosage = 0.2 g, shaking speed = 160 r min−1).
In order to determine the rate controlling step as well as the kinetics mechanism associated with adsorption process, the adsorption kinetics of Rhodamine B on CT269DR resin were assessed by using two kinetics models, pseudo first-order model (equation (12)) and pseudo second-order model (equation (13)) (Ho, 2006; Tran et al., 2016)


Parameters of adsorption kinetics models.

According to Figure 6 and Table 4, the values of R2 obtained for the pseudo first-order kinetics model are all 0.9999 and better than the pseudo second-order kinetics model, and there is a good relationship between qt and t at each temperature by the pseudo first-order kinetics model. Moreover, the experimental data of qe (qe,exp) agree with the calculated qe (qe,cal) by the pseudo first-order better than the pseudo second-order. Therefore, the adsorption process of Rhodamine B by CT269DR resin obeys pseudo first-order model instead of the pseudo second-order model.
The adsorption activation energy was determined by the following logarithmic form of the Arrhenius equation

Adsorption mechanism
In order to identify the mechanisms and rate controlling steps in the adsorption process, the adsorption kinetics data qt versus t1/2 were further fitted according to the Weber’s intraparticle diffusion model (equation (15)), which considers that the adsorption mechanism occurs through the diffusion of adsorbate molecules into the pores of adsorbent material (Martins et al., 2015)


Intraparticle diffusion plots for Rhodamine B adsorption at different temperatures (C0 =100 mg l−1, resin dosage = 0.2 g, shaking speed = 160 r min−1).
Parameters of intraparticle diffusion model.

As shown in Figure 7, there are three different stages of linearity for the adsorption of Rhodamine B on CT269DR resin at different temperatures, which indicate that the adsorption process is controlled by a multi-step process (Weng et al., 2009). The first stage, starting from the origin, is the external mass transfer (film diffusion) of Rhodamine B to the surface of CT269DR resin, or the boundary layer diffusion of Rhodamine B. The high values of kid in the first stage indicate that adsorption rate is rapid. The second stage is related to the intraparticle diffusion, which is the rate-limiting step. The third stage indicates that adsorption reaches equilibrium, where the intraparticle diffusion begins to slow down because of the extremely low concentration of Rhodamine B in solution. The intercept value, C, of the plot characterizes the thickness of the boundary layer (Dotto and Pinto, 2011). The values of C at different temperatures are all in order of C3NC2NC1 (see Table 5), which exhibits the lower external diffusion resistance at the first stage because of more vacant adsorption sites. Moreover, the plots of the latter two stages deviate from the origin, which reveals that the rate-limiting step is not only due to the intraparticle diffusion but also other diffusion (Liu et al., 2019).
The Boyd equation (equation (16)) can be further used to clarify if the rate-limiting step of adsorption is intraparticle diffusion or film diffusion by plotting Bt versus t which is displayed in Figure 8. As exhibited in Figure 8, we note that the plots of Bt versus t present a certain linearity, but do not completely pass through the origin. The above results verify the involvement of film diffusion along with intraparticle diffusion in the adsorption mechanism (Hameed et al., 2008)


Boyd plots for Rhodamine B adsorption at different temperatures (C0 =100 mg l−1, resin dosage = 0.2 g, shaking speed = 160 r min−1).
Influence factors of dynamic adsorption
Effect of feed flow rate
The position of the breakthrough curve along the axis depends on the capacity of the column with respect to the flow rate, feed concentration, and bed height. In a fixed-bed adsorption column, the adsorbent was placed near to raw solution approaches saturation more rapidly where the equilibrium adsorption capacity occurs initially. Then, the adsorption zone transfers further as time goes on and reaches the exit zone of the column. When the adsorption zone passes through the column, the adsorbate concentration at the inlet and outlet of column becomes equal (Ghorai and Pant, 2005).
The flow rate of the feed solution through the column is an important parameter, since it not only affects the recovery of Rhodamine B, but also controls the time of analysis. In order to evaluate the effect of flow rate, a set of solutions containing Rhodamine B 400 mg l−1 were passed through the glass column containing 1.2 g of CT269DR resin at a flow rate that varied from 4 to 8 ml min−1 at 318.15 K, and the results are displayed in Figure 9. As shown in the figure, the breakthrough point is advanced when the flow rate increases. The bed adsorption capacities, q, were obtained to be 334.87, 518.98, and 582.10 mg g−1 via the integration of the breakthrough curve at the flow rate of 4, 6, and 8 ml min−1, respectively. The increase in bed adsorption capacity of the resin is due to the fact that high flow rate provides greater driving force for the mass transfer to reduce the transfer resistance. Therefore, the flow rate of 6 ml min−1 was used for further studies.

Effect of the feed flow rate on the adsorption.
Effect of temperature
Figure 10 shows the breakthrough curves of Rhodamine B at different temperatures. The flow rate is 6 ml min−1, and the concentration of Rhodamine B is 400 mg l−1. The glass column was filled with 1.2 g of CT269DR resin. As the temperature rises, the penetration curve is advanced. The bed adsorption capacities increase with increasing temperature, which are 343.03, 435.19, and 518.98 mg g−1 at the temperature of 298.15, 308.15, and 318.15 K, respectively. The results indicated that the process of adsorption is endothermic, which is the same result as it is described in the “Static adsorption” section.

Effect of temperature on the adsorption.
Dynamic desorption
In this experiment, sodium hydroxide solution was used as a desorbent. At 298.15 K, a sodium hydroxide solution having a mass fraction of 1 and 2%, respectively, was passed through the saturating 1.2 g of CT269DR resin in the column at the flow rate of 2 ml min−1. Figure 11 shows that the higher desorbent concentration is, the better desorption effect is. The desorption rate of the 1 and 2% NaOH solution was calculated to be 11.7 and 20.1%, respectively.
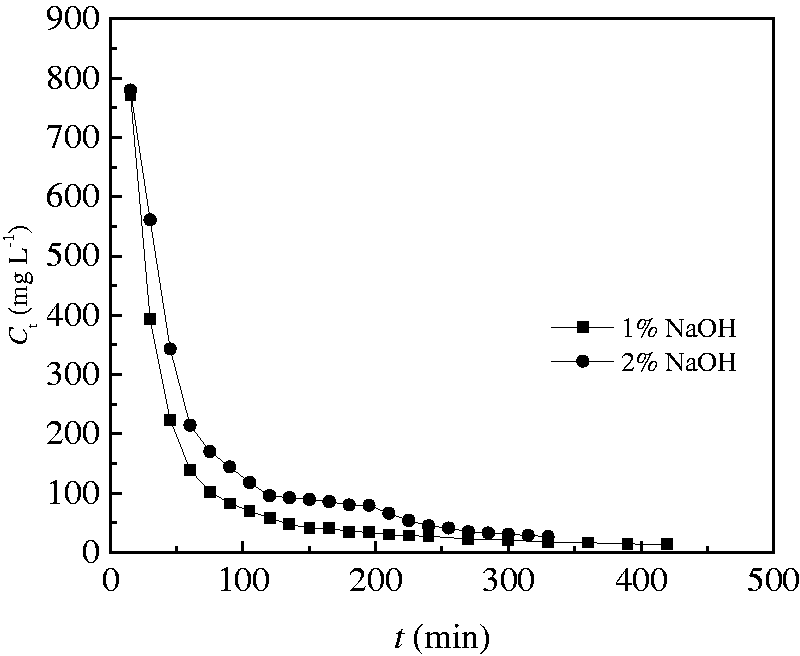
Effect of desorbent concentration on resin desorption.
Characterization of the resin
SEM
The SEM images of the fresh and used CT269DR resin are displayed in Figure 12, which vividly illustrates the alteration of their physical structure. As shown in Figure 12, the surface of the fresh CT269DR resin is relatively smooth and even. After adsorption, the surface texture of the used resin is slightly affected. Slight scratches and roughness were observed only on the partial surface due to the mechanical wear. It can be concluded that the surface appearance of the resin after repeated use is hardly changed under strong agitation. Therefore, this type of resin has good wear resistance.

SEM images of (a) the fresh resin and (b) the four times reused resin.
FT-IR
The FT-IR spectrum of the fresh and four times reused CT269DR resin is shown in Figure 13. As can be seen in the figure, the absorption peaks of the fresh resin at 1598 and 1493 cm−1 are the stretching vibration of the benzene ring, and 2926 and 2852 cm−1 are the –CH2 group absorption peaks (Miao et al., 2011). The characteristic peaks of the sulfonic group are observed at 1125, 1032, and 1004 cm−1, and 3405 cm−1 is characteristic of the –OH stretching vibrations of the sulfonic group. In addition, the recovered CT269DR resin reused for four times has no obvious change in structure, referring to the FT-IR spectrum in comparison with fresh resin. Hence, the CT269DR resin as the adsorbent is recyclable and stable for adsorption of Rhodamine B.
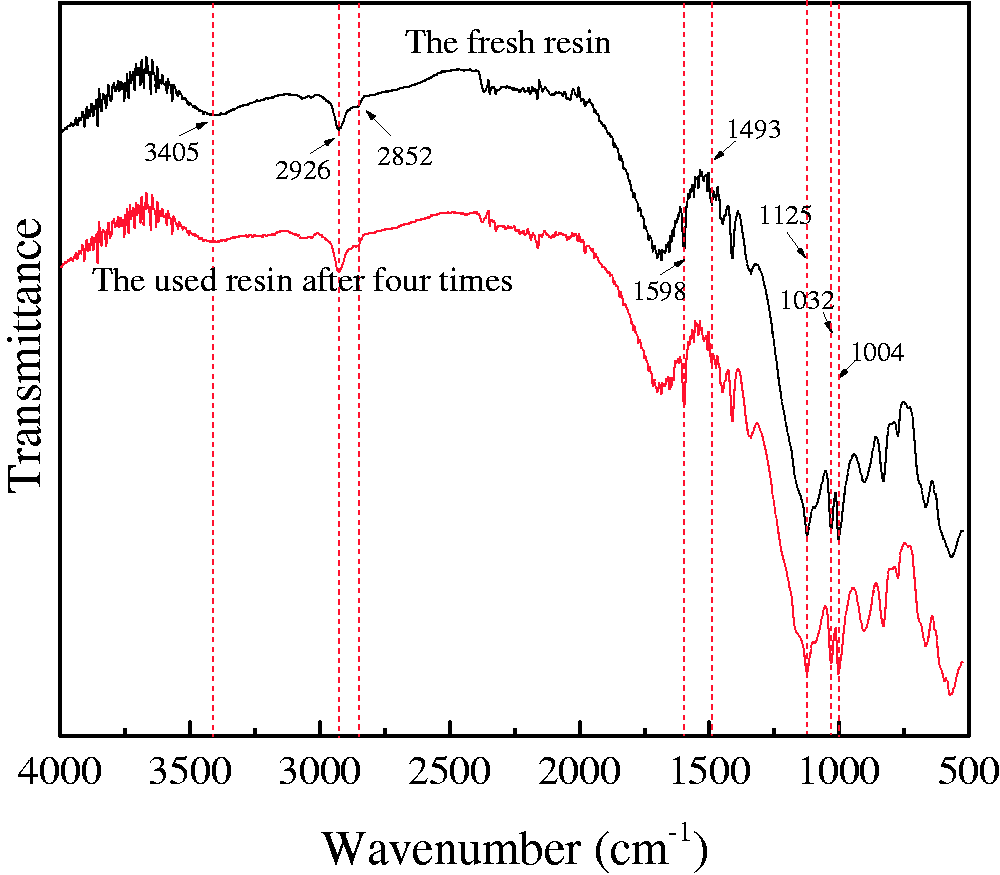
FT-IR spectra of the fresh and four times reused CT269DR resin.
Conclusions
In this work, seven kinds of resins were used as adsorbent for the removal of Rhodamine B from aqueous solution, and the CT269DR resin has the maximum adsorption capacity. The static experiments indicate that the external diffusion resistance is maintained constant when the shaking speed exceeds 160 r min−1, the optimal pH range is 5–8, and an increase of resin dosage can directly improve the percentage of removal of Rhodamine B. The equilibrium isotherm data of Rhodamine B on CT269DR resin were best fitted by the Langmuir model. The negative ΔG demonstrates the spontaneous nature of adsorption and confirms affinity of the adsorption, and the positive value of ΔH confirms the endothermic nature of the adsorption. The adsorption process of Rhodamine B by CT269DR resin obeys pseudo first-order model instead of the pseudo second-order model, and the activation energy is 25.7 kJ mol−1. Furthermore, the dynamic experiments show that the breakthrough point is advanced when the flow rate increases and the bed adsorption capacities increase with increasing temperature. The desorption using the solution of 2% NaOH is suitable for desorption and reusing process. In addition, SEM and FT-IR analysis reveals that the used resin has good wear resistance and chemical stability. Overall, CT269DR resin can be employed as an effective adsorbent for the removal of Rhodamine B from wastewater.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge the financial support for this work from the Natural Science Foundation of Fujian Province (No. 2019J05127), the Educational Research Project of Young and Middle-aged Teacher of Fujian Province (No. JT180625), the China Scholarship Council (No. 201908350057), the Fujian Provincial Collaborative Innovation Center for Clean Coal Gasification Technology (No. XK1705), the School-level Scientific Research Project (No. KY2017NS01), and the Training Program for Distinguished Young Scholars in Fujian Province University (No. Fujian Education Science [2017]52), the central government guides local science and technology development project (2019L3013).




