Abstract
Adsorption of methylene blue dye over the adsorbent derived from
Introduction
The industrial effluent from textiles, paper, plastic, leather, etc. is reported to contain dyes that pollute water resources and environment. This pollution is harmful for living species (Doğan et al., 2009; Janet et al., 2015). One such dye is methylene blue which causes numerous health hazards to human health (Hameed et al., 2007b). This has led researchers to discover a new and cheap technique for the removal of dyes from the effluents. So far, the adsorption of pollutants on adsorbents has been found to be the most attractive method due to its simplicity. Commercially available adsorbents are very expensive as they are produced from natural resources like wood or coal (Hameed et al., 2007a). Therefore, there is a need to look for some cheap raw material to produce low-cost adsorbent.
Many research works have been reported on various agricultural byproducts or low-cost adsorbents such as olive stones, peach stones, pistachio nut shells, date pits, olive pomace,
Materials and methods
Preparation and characterization of the adsorbent
The surface morphology was determined using a ZEISS EVO Series Scanning Electron Microscope EVO 50. This microscope had the resolution of 2.0 nm at 30 kV with magnification range up to 1,000,000 and an acceleration voltage up to 30 kV. The sample was mounted on a circular metallic sample holder with the help of sticky carbon tape.
The Brunauer–Emmett–Teller (BET) surface area of the adsorbents was measured by using Micromeritics ASAP 2010 apparatus. Nitrogen adsorption/desorption isotherms were measured at −196℃. Prior to gas adsorption measurements, the adsorbent was degassed at 200℃ ± 1℃ under vacuum for a period of 30 min. The total pore volume was calculated at a (p/po) relative pressure 0.989.
The Fourier transform infrared spectroscopy (FT-IR) spectrum of adsorbents was generated using Perkin Elmer Frontier in the range of 400–4000 cm−1 at room temperature. The powdered sample of adsorbent mixed with 0.3 g of KBr in 2:100 ratio and pellet—1 mm thick—was prepared by pressing the mixture in a dye at a pressure of 7–8 ton through hydraulic press. The spectrum of the pellet was recorded with a resolution of 4 cm−1 and 16 scans per sample. Baseline correction was done with Spectrum 10 software.
Adsorbate: Methylene blue
The dye solution was prepared using distilled water and methylene blue (Merck), without further treatment. Distilled water was used for the preparation of solutions. Initially, a stock solution of 100 mg l−1 was prepared and the working solutions were prepared by the dilution of the prepared stock solution.
Adsorption studies
The adsorption study was done in phases. Equilibrium study was conducted by taking 50 ml of four different concentrations of methylene blue solution in the range of 10–100 mg l−1 at pH 6. To each solution, 100 mg of adsorbent was added. The mixture was then stirred for 360 min at 120 r/min to ensure equilibrium. The adsorption mixture was separated through centrifugation process and the supernatant was collected. The concentration of the supernatant was analyzed using ultraviolet–visible spectrophotometer (Varian Carry-100) at 668 nm. The amount of adsorption at equilibrium, qe in mg g−1, was calculated (Benadjemia et al., 2011; El Khames Saad et al., 2014) using equation (1)

The kinetic studies of adsorption were conducted out in the similar manner as adsorption at equilibrium. However, the samples for kinetic studies of adsorption were taken at different time intervals. The effect of parameters such as pH (2, 4, 6, 8 and 10), initial concentration of methylene blue dye (10, 40, 70 and 100 mg l−1) and temperature (30℃, 40℃ and 50℃) on the adsorption of dye for CAd and SAd were also studied.
Results and discussions
Scanning electron microscopy of C and S (shown in Figure 1(a) and (c), respectively) and CAd and SAd (shown in Figure 1(b) and (d), respectively), represents surface structure of the materials before and after chemical action. It is observed that C contains insignificant closed holes; however, after chemical treatment, CAd exhibits remarkable number of lengthy openings on the surface, constituting channels for the flow of fluids through them. It signifies that enormous volatiles have been removed from the base material, leaving behind the vacant space. The surface of SAd carries fewer pores mouth; yet precursor (S) highlighted complete absence of the pores in SEM image as presented in Figure 1(c). SEM images of (a) C, (b) CAd, (c) S and (d) SAd.
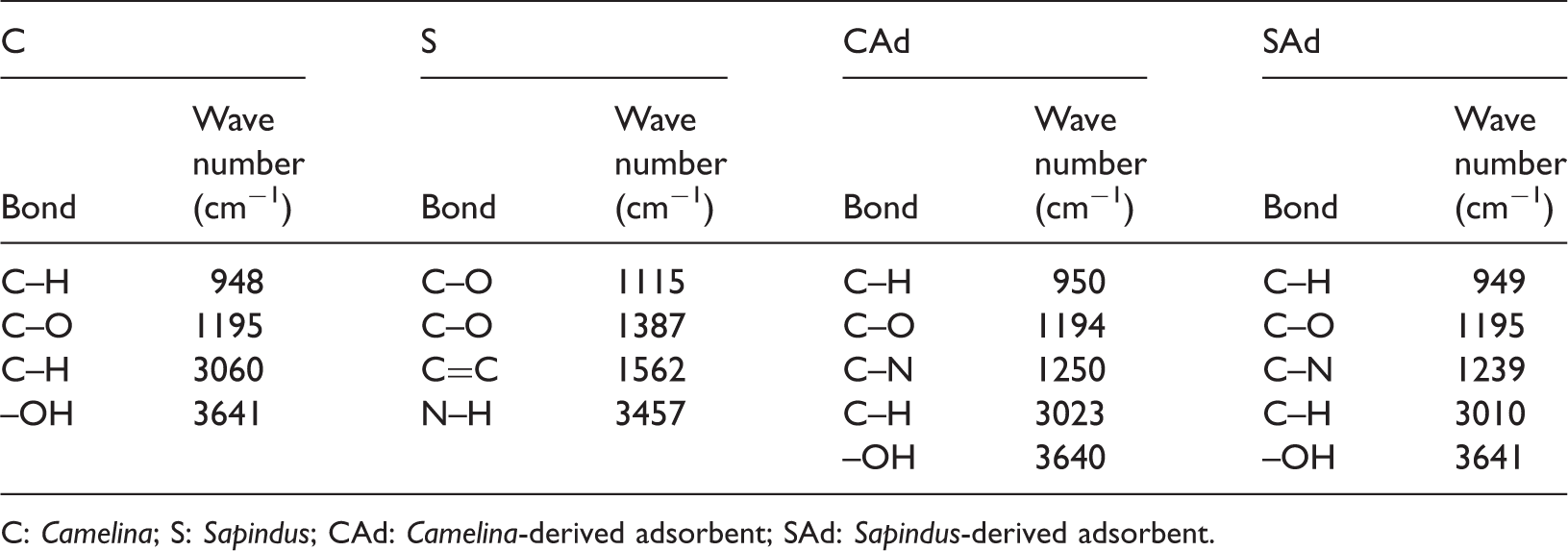
The FT-IR spectrum of both the precursors and adsorbents as shown in Figure 2 facilitates to understand the affinity between the adsorbent and the adsorbate. The existence of similar functional groups on the surface of CAd and SAd due to the employment of common activating agent for their production is given in Table 1. As it is apparent from Table 1, native bonds are retained by both the adsorbents with some improvement. Because dye molecules contain polar groups, these functional groups play a significant role in the new bond formation.
FT-IR spectrum of (a) CAd, (b) SAd, (c) S and (d) C. FT-IR spectra depicting various bonds on the surface of C, S, CAd and SAd. C: 
It is also important to examine the influence of the pH of the solution as it affects the extent of adsorption. Figure 3 represents the adsorption on CAd and SAd at different pHs (2–10). It is concluded from the data that the neutral pH favors maximum adsorption up to 12% and 99.38% for SAd and CAd, respectively. The reason behind this is that the acidic and basic nature of solution results in the protonation and deprotonation of the adsorbents’ surface charge, which was otherwise responsible for the uptake of dye molecules as already reported (Wang et al., 2008).
Effect of pH on adsorption by CAd and SAd at 30℃.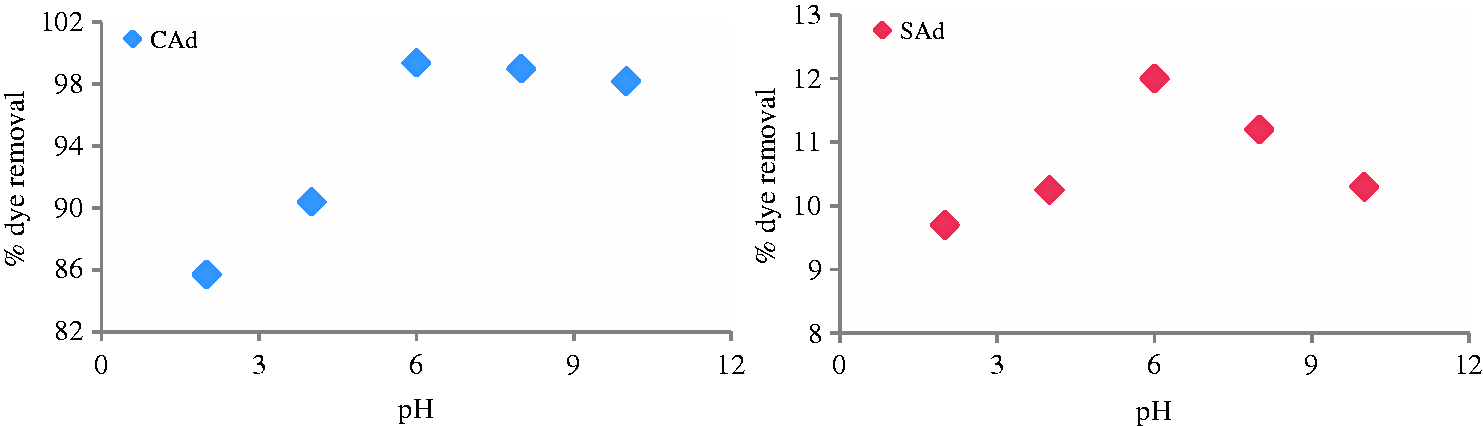
Figure 4 presents a study of removal percent of dye molecules with respect to temperature which further helps in the investigation of several thermodynamic parameters like entropy, enthalpy and Gibbs free energy. As evident from the individual curve, the slope increases as the temperature of the process is increased from 30℃ to 50℃, indicating soaring recovery. This may be because the high temperature excites the dye molecules which aid in combating mass transfer resistance. Also, the boundary layer becomes thinner at elevated temperature and this enables the molecules to reach adsorption sites within the pores. A similar trend has been reported in the literature for the temperature-dependent study of methylene blue dye adsorption on natural zeolite (Han et al., 2009).
Removal percentage of CAd and SAd at initial concentrations—40, 70 and 100 mg l−1.
Figure 4 also indicates that lesser adsorption takes place at higher initial concentration which is in line with the result reported for PP and APP (Tiwari et al., 2014). At the initial dye concentration of 40 mg l−1, CAd proved to have much higher removal capacity of 99% at 50℃ in comparison to 14.45% for SAd for the same conditions. The repulsion among the molecules could have resulted in the drop of percent removal with increase in initial concentration. The percent removal of CAd and SAd at 30℃ for 40 mg l−1 is 99.39 and 12.02, respectively. Again, the huge difference in uptake of dye molecules is due to the highly porous surface of CAd in comparison to SAd.
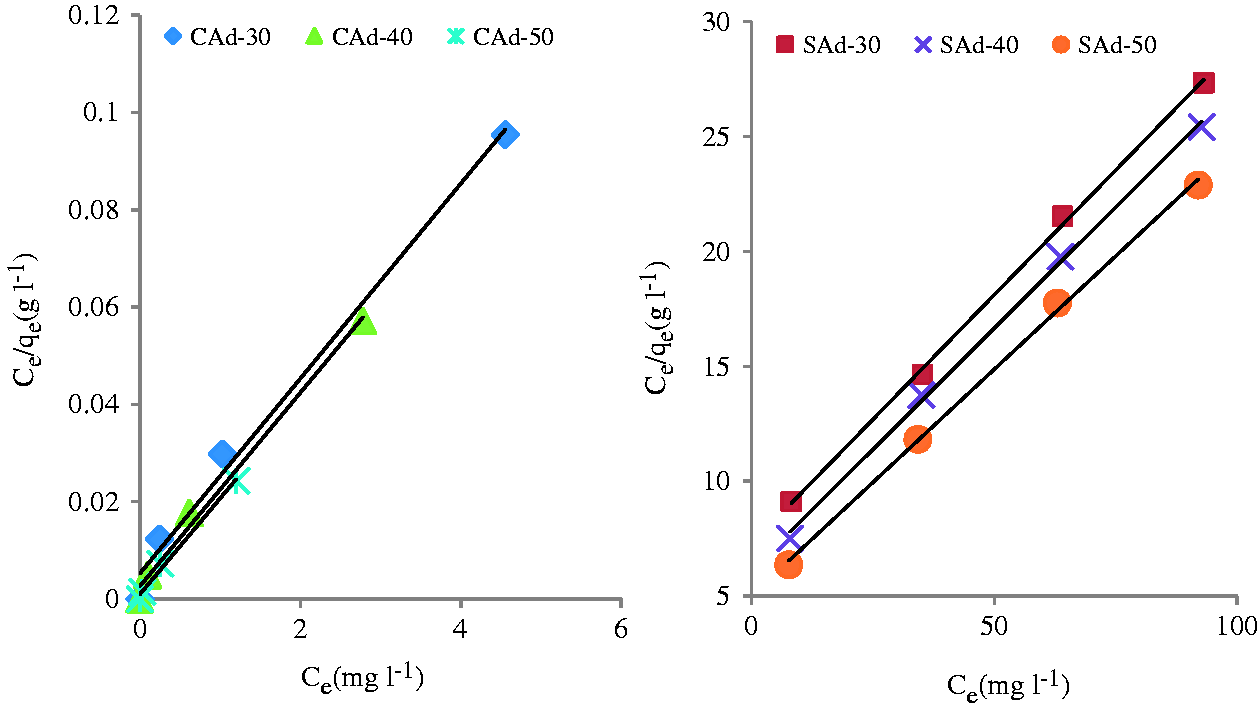
The adsorption isotherm gives the distribution of adsorbate in solid to liquid phase and hence presents a relation among the quantity adsorbed, qe, to the concentration in the solution at equilibrium, Ce (Tan et al., 2008). Langmuir and Freundlich isotherms are most commonly employed for the equilibrium study of adsorption (Hameed et al., 2007a).
Langmuir isotherm is used to understand the equilibrium with an important assumption of surface homogeneity (Hameed and Daud, 2008). The mathematical expression of Langmuir isotherm is presented in equation (2) (Ahmad et al., 2014)
Langmuir isotherms of methylene adsorption on CAd and SAd at different temperatures (30℃, 40℃ and 50℃). Equilibrium models constants for the adsorption of methylene blue on CAd and SAd. CAd: 

Comparison of maximum adsorption capacities, qe by several adsorbents for methylene blue dye.

Separation factor, RL, helps to identify the nature of adsorption process and is evaluated using the relation as given in equation (3) (Belhachemi and Addoun, 2011; Hameed and Daud, 2008)

The linear expression for Freundlich isotherm is shown in equation (4)
Freundlich isotherms of methylene adsorption on CAd and SAd at different temperatures (30℃, 40℃ and 50℃).

In order to understand the controlling step involved in the adsorption of methylene blue such as diffusion, adsorption and chemical reaction, various kinetic models have been applied to the experimental results. The pseudo-first-order (Kavitha and Namasivayam, 2007) expression is presented in equation (5)
Pseudo-first-order kinetics of methylene adsorption on CAd and SAd at different temperatures (30℃, 40℃ and 50℃). Kinetic models constants for adsorption of methylene blue on CAd and SAd at an initial concentration of 100 mg l−1. CAd: 


The pseudo-second-order kinetic model (Gerçel et al., 2007) is presented in equation (6)
Pseudo-second-order kinetics of methylene adsorption on CAd and SAd at different temperatures (30℃, 40℃ and 50℃).

Since the abovementioned two kinetic models are inadequate to explain intraparticle diffusion, diffusion mechanism is studied using the model proposed by Weber and Morris as presented in equation (7) (Gerçel et al., 2007)
Intraparticle diffusion plots of methylene adsorption on CAd and SAd at different temperatures (30℃, 40℃ and 50℃).

Several thermodynamic parameters are determined from the basic relations as given in equations (8) and (9)


In the above relations, R is the universal gas constant, T is the temperature in Kelvin and KL is the distribution coefficient (Tan et al., 2008) and considered Langmuir constant in this study (Ahmed et al., 2011; Geçgel et al., 2012; Khare and Kumar, 2012). The values for ΔH° and ΔS° are evaluated from slope and intercept, respectively, of graph between ln KL versus 1/T as shown in Figure 10.
Thermodynamic studies of methylene adsorption on CAd and SAd at different temperatures (30℃, 40℃ and 50℃).
Thermodynamic parameters for adsorption of methylene blue on CAd and SAd at initial concentration 100 mg l−1.

CAd:
Activation energy is calculated using the relation as presented in equation (10) (Gerçel et al., 2007)

Conclusion
The hardness of raw materials—
This study, therefore, proves the diverse potential of chemically treated
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
