Abstract
In the textile industry, various acidic, basic and reactive dyes are used for different applications; the aim of this study is to eliminate Methylene blue (MB) a dangerous dye by a zeolithe produced at low economic cost by adsorption in batch mode. The adsorbent was characterized by the FTIR spectroscopy, X-ray diffraction (XRD), and point of zero charge (pHpzc = 10.42). Some examined factors were found to have a significant impact on the adsorption capacity of the zeolithe like the initial dye concentration (5–25 mg/L), solution pH (2–14), adsorbent dose (0.1–2 g/L), agitation speed (150–500 rpm), particles size (100–500 µm) and temperature (298–333 K). The best capacity was found at pH 6 with an adsorbent dose 0.2 g/L, an agitation speed 200 rpm and a contact time of 40 min. Modeling the Kinetics and Isotherms data shows respectively that the pseudo-second-order kinetic model and Langmuir isotherm provide better fitness to the experimental data with the maximum adsorption capacity of 12.50 mg/g at 25°C. The adsorption isotherms at different temperatures have been used for the determination of the free energy (ΔG°); enthalpy (ΔH°) and entropy (ΔS°) to predict the nature of MB adsorption. The positive values of ΔG° and ΔH° indicate a non-spontaneous and endothermic MB adsorption with a physisorption process. The adsorbent elaborated from the zeolithe was found to efficient and suitable for the removal of reactive dyes from aqueous solutions, due to its, low cost preparation and good adsorption capacity. The photocatalysis of MB in the presence of hybrid semiconductor (TiO2/Zeolithe) constitutes the logical continuation of this study.
Introduction
The effluents from the textile, leather, food processing, dyeing, cosmetics, paper, and dye manufacturing industries are among the most important sources of the aquatic pollution.1 –6 Many dyes and their break down products are highly hazardous for living organisms, among which the Methylene Blue (MB).The presence of dyes in water increase the chemical and biochemical oxygen demand, oxygen demand as well as high-suspended solids. As the global demand for water augments because of growing population, the amount of wastewater produced is continuously increasing around the world, thus requiring urgent needs for purification methods. Wastewater is often discharged into rivers and lakes or infiltrates into aquifers, and therefore affects dramatically the quality of freshwater. Globally, more than 80% of wastewater returned to the environment untreated and has negative consequences for the marine environment. 7 About 600 organic and inorganic pollutants have been listed in water along with biological species. 8 The presence of dyes even at small concentrations weakens the light penetration through the water surface, thus inhibiting considerably the photosynthetic process of the aqueous flora. Currently, there are nearly 10,000 types of synthetic dyes with a worldwide production of 7 × 105 tons per year.9,10 Methylene Blue is a cationic dye appears as a green crystalline powder, highly soluble in water. It is employed for the dyeing of cotton, paper, silk, wood, and leather products, anti-parasitical, antibacterial and antifungal in aquaculture and commercial fish hatchery industries. It is also used as a food coloring agent, food additive and medical disinfectant. MB has caused several health hazards such as damage to nervous system, brain and liver when ingested, eye burns, fast breathing, profuse sweating and cancer of different parts of the body. 11 Consequently, many treatments have been applied to limit its concentration in aqueous medium such as adsorption12–28, biological treatment 29 , discolorationand detoxification, 25 photocatalysis26,27 and electrochemical degradation. 28 However, the adsorption is attractive for the elimination of many kinds of pollutants because of its low cost, simplicity of design, removal efficiency and ease of use. In this respect, agricultural wastes have advantages as adsorbents, they are readily available, cost-effectiveness, renewable, require less processing time, offer suitable adsorption capability and can be easily regenerated. Moreover, they can be modified by treating them with chemical agents, for example, alkalis, acids, organic compounds, etc., or thermally. Such modifications could have beneficial effects on chemical/physical properties like increasing surface area, improved pore structure and functional group. Therefore, many studies have focused on the feasibility of abundant substances used for activated carbon. This has prompted a growing interest in the production of activated carbons from renewable and less expensive precursors which are by-products of industrial or agricultural origin, for water treatment. Modified adsorbents exhibit an adsorption capacity with selectivity and sensitivity. 29 In this optic, agricultural waste such as cotton stalk, 30 olive stones, 31 coconut shell, 32 rice husk, 33 orange peel, 34 pistachio shell, 35 and wheat bran 36 titanium dioxide 37 mesoporous silicate. 38 In view of the economic cost of industrial activated carbon, the objective of this study was oriented toward the preparation of an adsorbent from activated zeolithe to remove the methylene blue in aqueous solution, In addition, the equilibrium adsorption data were fitted to various equations in order to obtain the constants related to the adsorption phenomena.
The objectives of this study are (i) the preparation of an adsorbent at low economic cost, and increase its porosity by chemical and physical activation (ii) the regeneration of the adsorbent for other applications, reducing the pollution generated by textile discharges of Methylene blue using the adsorption technique (iii) the switching from batch mode to column mode and perform MB elimination tests by photocatalysis in the future studies
Materials and methods
FTIR spectroscopy
The FTIR spectroscopy was used to identify the characteristic functional groups of the zeolithe. 5 mg of zeolithe was mixed with dry KBr of spectroscopy quality and pressed under a pressure of 4500 psi to form thin disc. The spectra were plotted with a Perkin Elmer 2000 infrared spectrometer in the range (4000–400 cm−1) for 16 times to increase the signal to noise ratio.
X ray diffraction
The X-ray diffraction (XRD) patterns of the zeolithe was obtained with a Philips X-ray diffractometer (PW 1890 model) operating at 40 kV, 40 mA and equipped with CuKα radiation (λ = 1.54 Å). The patterns were obtained with CONIT T-2T scan mode at 0.17 deg/step of step width and 8°/min of scan speed.
Spectrophotometry
The spectrophotometry is a technique which owes its development to progress in the quantum mechanic, permitting to detect, identify and quantify the concentration of a solute in solution by using the Beer-Lambert’s law. 10 mm quartz cell was used with a reproducibility of 0.1 nm. Two light sources were used: a visible tungsten halogen and UV deuterium lamps; the scanning speed was set at 1200 nm/min. The blank tests were realized in a quartz cell identical to the previous one with ultra pure water. Measurements of the residual concentrations were obtained by linear interpolation from a calibration graph.
pH meter
The pH of the solutions was accurately measured using a microprocessor-based pH meter (Model HANNA HI 8521 type). The instrument was calibrated with commercial buffers of pH 4, pH 7, and pH 10. The pH was adjusted to by using H2SO4 and NaOH respectively for acidic and basic media.
The zero point charge
The Zero Point Charge pH(zpc) of the zeolite, that is, the pH for which the surface charge is zero, was reliably determined using a procedure similar to that reported elsewhere. 39 Briefly, 20 mL of of KNO3 solutions (0.01 M) were placed in closed conical flasks; the pH of each solution was adjusted between 2 and 14 by addition of HCl or NaOH solution. Then, 0.1 g of zeolite was poured in solution and the final pH was measured after 24 h under magnetic agitation at ambient temperature; pH(zpc) is the final pH versus initial pH crosses the line at final pH = initial pH.
Methylene blue adsorbate
Methylene Blue ( MB, purity ⩾ 82% ) used as adsorbate, is a Basic Blue 9 dye, whose IUPAC name is 3,7-bis (dimethylamino) phenothiazin-5-ium chloride (C.I. 52015). It was purchased from BLANC-LABO SA with a chemical formula C16H18ClN3S, and a molecular weight equal to 319.85 g/mol; the absorbance peaks at of 662 nm (λmax). Chemical structure and properties of the Methylene blue are gathered in Table 1.
The properties of Methylene blue.

Preparation of adsorbent
The crushed and sieved samples were subjected to several washes. The first wash was done with distilled water which consists of removing all the large particles of the sand as well as other impurities. This procedure was followed by a second wash with H2O2 to eliminate the organic matter. 100 g of crude zeolithe 4A (China, company) were put in 900 mL of distilled water to recover a large amount of purified zeolithe, then the mixture was left under stirring for 2 h, the purpose of which was to eliminate large particles by decantation. Then, the suspension containing fine particles was poured into crystallizers, and dried at of 80°C in an oven for 3 days until complete drying; the obtained solid was ground and sifted. To remove the organic matter present in the powder after sieving, the solution was treated with H2O2. 85 g of the zeolithe obtained, dissolved in 300 mL of H2O2 10% was heated at 60°C, the zeolithe sample was placed in a 1 L Erlenmeyer filled with distilled water, heated at 70°C and left under gentle stirring for 1 h.After decantation, the suspension containing the fine particles was poured into a crystallizer, and dried at 80°C for 2 days until complete drying. The obtained solid was crushed, sieved and hermetically stored in glass bottles.
Adsorption experiment
The effects of the initial MB concentration (Co: 5–25 mg/L), pH (2–12), adsorbent dose (0.1–2 g/L), stirring speed (100–500 rpm) and temperature (25–60°C) on the MB adsorption was investigated in a batch mode for a contact time (0–60 min). The MB solutions were prepared by dissolving the accurate amount MB (99%) in distilled water, used as a stock solution and diluted to the required initial concentration. Adsorption was carried out at room temperature and under atmospheric pressure in batch conditions. It was performed by introducing a mass of zeolithe into a glass Erlenmeyer of 100 mL, containing 50 mL of an aqueous Methylene blue solution of determined concentration. The mixture was magnetically stirred at 150 rpm for the desired period. Then, the separation was effectuated by centrifugation at 6000 rpm during 5 min and the residual MB concentration was analyzed by UV–Vis spectroscopy at the maximal wavelength (=662 nm).
The amount qt (mg/g) yield R (%) of MB adsorbed by Zeolithe (qt) was calculated from the relations:


Where Co is the initial MB concentration and Ct the concentrations (mg/L) at time t, V is the volume of solution (L) and m is the mass of zeolithe (g).
Results and discussion
Characterization of adsorbent
The FTIR spectroscopy analysis (Figure 1) shows that after modification, new peaks appear. The first one at 669.93 cm−1 corresponding to stretching vibration of
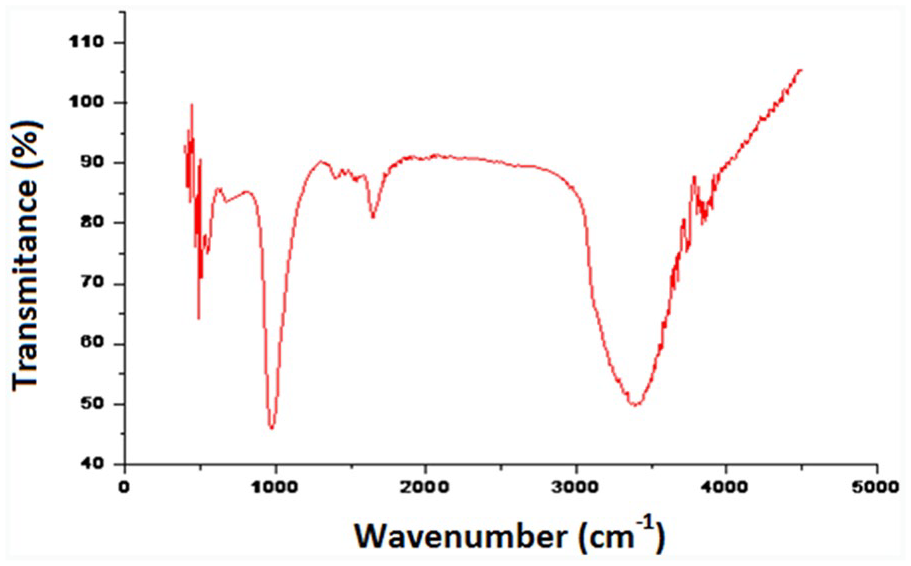
FTIR spectroscopy of the zeolithe 4A.
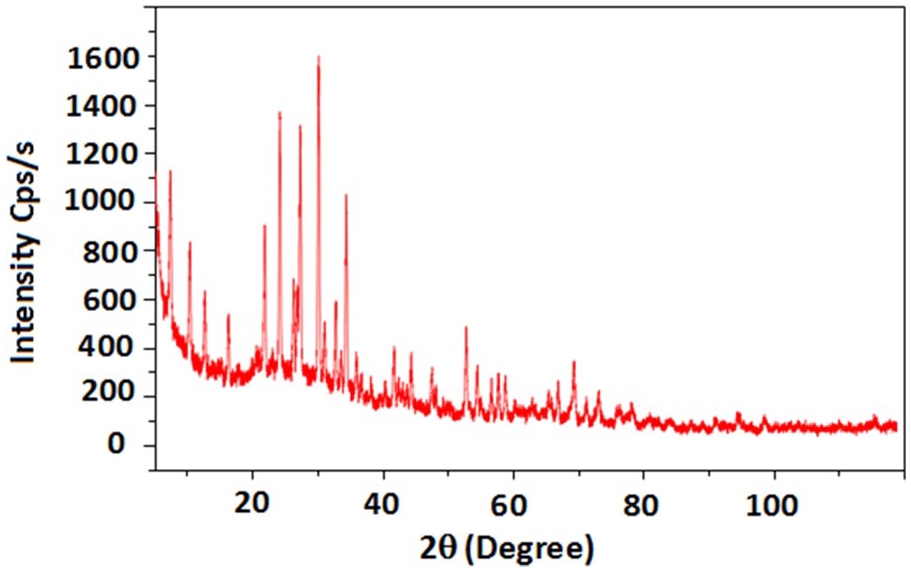
DRX spectrum of zeolithe 4A.
Influence of time and concentration of MB on the adsorption capacity
The MB concentration and the mass of the adsorbent are two important factors for the adsorption. To demonstrate the effect of the initial dye concentration (Co) on the adsorption, experiments were performed at different MB concentrations Co. We performed the experiments for Co valises (5–25 mg/L) and a mass of the zeolithe of 0.1 g. The temperature is maintained constant at 25°C and the mixture was stirred at 200 rpm. The samples were taken at regular time intervals, vigorously centrifuged for 10 min and finally analyzed by UV-visible spectrophotometry.
We notice from a very rapid adsorption (Figure 3), since a saturation level is reached after only 40 min; the latter indicates the formation of a monolayer. Note that the adsorption capacity increases with augmenting the MB concentration; this is due to the fact that the diffusion of the MB molecules from the solution to the surface of the adsorbent is accelerated by the increase in the dye concentration; similar result was already obtained for the adsorption of MB on apricot stones. 40 With raising the MB concentration (5–25 mg/L), the adsorbed amount increases from 4.7 to 10.3 mg/g, and we can deduce that the MB adsorption onto the zeolithe occurs in three stages:
Fast MB adsorption due to the presence of free sites on the adsorbent surface, which translates the linear increase of the adsorption capacity over time.
Reduction of the adsorption rate, reflected by a small increase in the adsorption capacity attributed to the decrease in the quantity of MB in solution and the number of available unoccupied sites.
Stability of the adsorption capacity, due to the total occupation of adsorption sites: the establishment of the stable state reflecting this stage. The adsorption capacity of MB increases over time to reach a maximum after 40 min. and thereafter tends toward a constant value indicating that no more MB ions are removed from the solution.
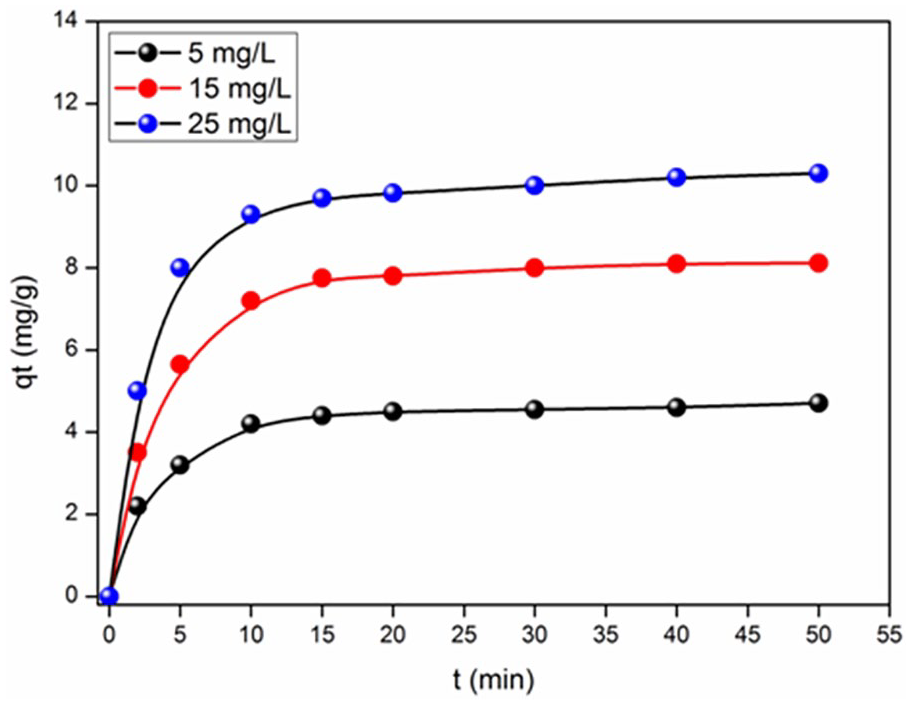
Influence of the MB concentration on the adsorption capacity.
Influence of adsorbent concentration
The effect of the adsorbent dose on the MB adsorption is crucial in determining the optimum value for the adsorption isotherm plot. This is the reason why we have started this step on the MB removal by the zeolithe. In order to evaluate the optimum content of the zeolithe for the MB adsorption, experiments were realized on MB solution (8 mg/L), to which different amounts of adsorbent were added in the interval (0.05–2 g/L).The changes in the adsorbed MB amount with the contact time, as a function of the zeolithe dose, are illustrated in the Figure 4. It is found that the adsorbed MB amount decreases with the addition of the adsorbent to the mass of 2 g/L. Beyond this dose, the adsorbed amount no longer changes. This may be due to the number of available sites which increases with the zeolithe amount up to the mass 2 g/L at which the number of sites becomes stable.41,42 Such behavior can be explained by: (i) as long as the amount of added adsorbent to the MB solution is small, the MB molecules can easily access the adsorption sites. The addition of adsorbent increases the number of adsorption sites but the charges of the dye, according to its functional groups, have difficulty approaching these sites because of the bulk (the electrical state of the surface semiconductor with dye); (ii) A large amount of adsorbent favors the agglomeration of particles. This results in a reduction in the total adsorption area and therefore a decrease in the amount of adsorbate per unit mass of adsorbent.

Influence of the adsorbent dose on the adsorption of MB onto zeolithe.
Influence of pH
The pH plays a crucial role in the adsorption process. It is evident that the percentage of MB elimination increases with augmenting the pH (Figure 5). The pH effect on the MB adsorption by can be explained by the pHzpc zeolithe, for which the charge of the adsorbent surface is neutral; the charge is positive below pHzpc (10.14). 43 When the pH is lower than 6.5, the number of positively charged sites increases and improves the MB adsorption by electrostatic attractions.

Determination of pH (zero point charge).
The pH is linked to the ionization state of the zeolithe surface 44 according to the following reactions:

At low pH: the zeolite is characterized by an anion exchange capacity: The addition of H+ cations generates a positive charge, and the anions in solution are more attracted to this surface, which hinders the adsorption of MB.
At high pH, a cation exchange capacity develops: the OH- binds more and a negative charge develops, so the cations in solution will in turn be attracted to the surface according to the above reactions.
Figure 6 reports the change in the adsorbed quantity (qads) of MB on the zeolithe as a function of the solution pH. For MB, the adsorbed quantity is dependent on pH in the range (2-8) and the adsorption capacity peaks at pH 6.
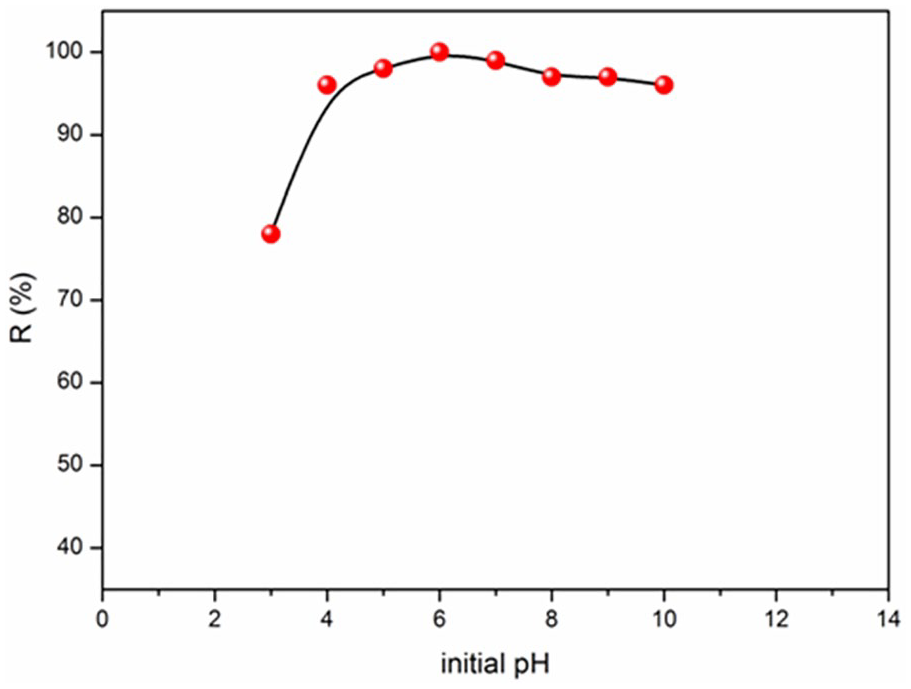
Influence of pH on the adsorption of MB onto the zeolite.
Effect of agitation speed on the adsorption of Methylene blue
The effect of the stirring speed is also investigatedin the range (100–500 rpm) on the adsorption capacity of MB onto the zeolithe. The optimal adsorption capacity is obtained for a speed of 200 rpm (Figure 7) which gives the best homogeneity of the mixture suspension above which the vortex phenomenon form in stirred fluids predominates. Therefore, a speed of 200 rpm was selected for further experiments.

Influence of the stirring speed on the adsorption of MB onto zeolithe.
Influence of particles size
In the first stage of the batch experiments, the effect of the particle size on the MB adsorption onto the zeolithe is examined. Significant variations in the MB removal efficiency are observed for different particles sizes (100–500 µm); Figure 8 shows that the best performance is obtained for smaller sizes (100 µm). In general, smaller particles provide large surface areas, resulting in high MB uptake capacity and removal efficiency. Therefore, the class 100 µm was subsequently used in all other adsorption experiments.

Influence of particles size on the adsorption of MB onto the zeolite.
Adsorption kinetic study
Many models were proposed to study the mechanisms controlling the adsorption. In this study, the experimental data of MB adsorption are examined using the pseudo-first kinetic, 45 pseudo-second order kinetic, 46 Elovich kinetic equation 47 intra-particle diffusion 48 whose equations are given respectively below
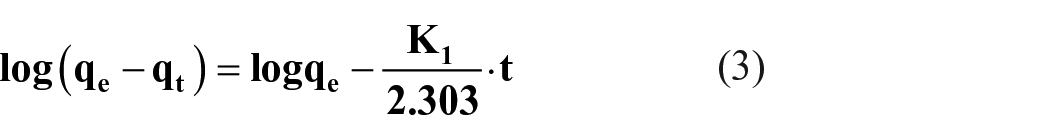



where
Constants of kinetic models.

Adsorption equilibrium isotherms
To assess the zeolithe performance, different isotherms existin the literature, among which those of Langmuir, 49 Freundlich, 50 Temkin, 51 and Elovich 52 isotherms were used (Figure 9). Besides, the isotherm models were applied under the optimal conditions. The Langmuir model is one of the widely applied, it is represented by the non linear and linear forms:




Where Ce is the equilibrium concentration (mg/L), qmaxthe monolayer adsorption capacity (mg/g) and KLthe constant related to the free adsorption energy. The constants L(/mg), AT, BT, KF, n, and KE(L/mg) are the Temkin, Freundlich, and Elovich constants,deduced after modeling of all models applied and grouped together in Table 3. Evaluating the fitness of experimental equilibrium data by various conventional isotherm models (based on considering

Adsorption isotherms under optimal conditions for the different models.
Constants of isotherms models.

Influence of temperature
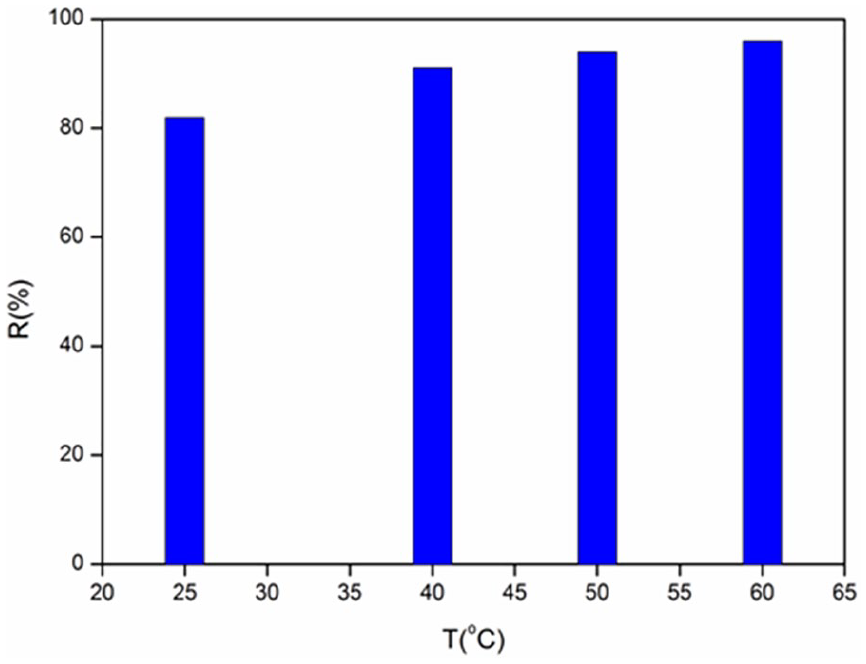
The temperature is an important factor that influences the adsorption of solutes such as dyes to the surface of zeolite. Its importance is not limited only to the need to understand how it affects adsorption/desorption, but the use of the experimental results obtained in the present case allow access to thermodynamic information, namely, equilibrium constants, enthalpy, and entropy. They can be evaluated from the modeling of the experimental results with adequate theoretical models. The variation of the adsorbed amount qads the MB equilibrium as a function of temperature (25–60°C), is illustrated in Figure 10.
Influence of temperature on adsorption capacity.
Thermodynamic characterization of adsorption
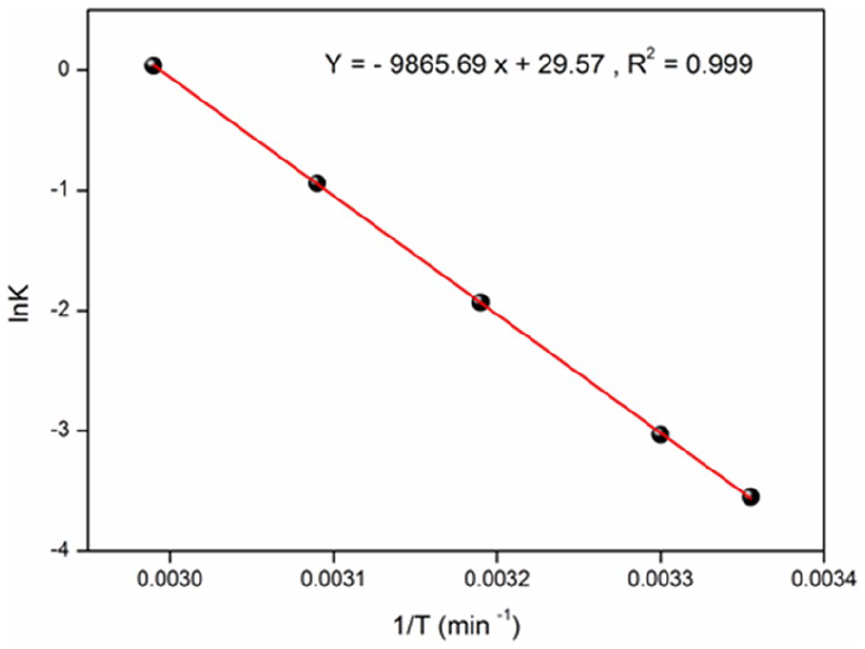
The adsorption capacity of MB increases with raising temperature over the range (20–60°C), above, the vaporization becomes problematic. The insights of the adsorption mechanism can be determined from the thermodynamic parameters: the free energy (ΔG°), the enthalpy (ΔH°) and the entropy (ΔS°). The equilibrium constant (K) is calculated by plotting ln(qe/Ce) versus qe and extrapolating to zero. The thermodynamic parameters are determined from the following equations.




Where Ce (mg/L) is the concentration at equilibrium and T the absolute temperature (K). The plot of LnK versus 1/T (Figure 11) is linear; the values of ΔH and ΔS are obtained from the slope and intercept of equation (14) while the free enthalpy (ΔG) at various temperatures are given in Table 4. The positive values of ΔG° and ΔH° indicate the non-spontaneous and endothermic MB adsorption with a physisorption process.
Linear form of Ln (
Determination of the thermodynamic parameters.

Performance of the zeolithe as adsorbent
It is interesting for a comparative purpose to give the adsorption capacity of some adsorbents available in the open literature. The values of the maximum adsorption capacity qmax of various adsorbent are gathered in Table 5. We can see that the MB adsorption observed in this work is well classified compared to other researches with a maximum adsorption capacity
Comparative study of the maximum adsorption capacity of MB compared to the bibliography.

Conclusion
The present study has shown that the prepared zeolithe has been successfully employed as an effective adsorbent for the removal of Methylene blue from aqueous solution. The Langmuir model provided a better fit of the equilibrium adsorption data. They gave a maximum adsorption capacity of 12.50 mg/gat 25°C. The positive freeen ergy ΔG° and positive enthalpy ΔH° showed that the MB adsorption onto zeolithe is endothermic and not spontaneous over the studied temperature range. The positive entropy ΔS° states that the randomness augments at the solid-solution interface during the MB adsorption onto the zeolithe, implying that some structural exchange may occur among the active sites of the adsorbent and MB ions. The MB adsorption follows a pseudo-second order kinetic model, which relies on the assumption that chemisorption is the rate-limiting step. The MB ions are attached to the adsorbent surface by forming a chemical bond and tend to find sites that maximize their coordination number with the surface. The kinetics and thermodynamic data can be further explored for the design for the treatment of real effluents.
This study in tiny batch showed encouraging results, and we hope to achieve the adsorption in column mode under with semi pilot scale. The present investigation showed also that the zeolithe is a potentially useful adsorbent for the basic dyes and metals. The elimination of MB in column mode and the degradation of dye by heterogeneous photocatalysis is our next step.
Footnotes
Acknowledgements
The authors gratefully acknowledge support from University M’hamed Bougara of Boumerdes, Laboratory of Soft Technologies and Biodiversity, Faculty of Sciences.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
