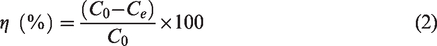Abstract
Reducing input of phosphorus is the key step for control of eutrophication and algal blooming in freshwater lakes. Adsorption technology is a cost-effective technology for phosphate removal in water for the purpose. Thus, in this study, a novel Fe–Mn–La tri-metal composite sorbent was developed, and then evaluated for phosphate removal. The results showed that the maximum adsorption capacity could be approached to 61.80 mg g−1 at 25°C under pH of 6.03. Adsorption of phosphate by Fe–Mn–La tri-metal composite adsorbent fitted better by pseudo-second-order kinetic equation and Langmuir model, which suggested that the adsorption process was surface chemical reactions and mainly in a monolayer coverage manner. The thermodynamic study indicated that the adsorption reaction was an endothermic process. The phosphate removal gradually decreased with the increasing of pH from 3.02 to 11.00. The sequence of coexisting anions competing with phosphates was that CO32− > Cl− > SO42− > NO3−. Dissolved organic matter, fulvic acid as a representative, would also decrease adsorption capacities of phosphate by Fe–Mn–La tri-metal composite adsorbents. Adsorption capacity would be decreased with increasing addition of adsorbents, while removal efficiency would be increased in this process. The Fe–Mn–La tri-metal composite adsorbent showed a good reusability when applied to removal of dissolved phosphate from aqueous solutions. The Fourier transform infrared spectrometer and X-ray photoelectron spectroscopy analyses indicated that some hydroxyl groups (–OH) on the surface of adsorbent were replaced by the adsorbed PO43−, HPO42−, or H2PO4−. Aggregative results showed that the novel Fe–Mn–La tri-mental composite sorbent is a very promising adsorbent for the removal of phosphate from aqueous solutions.
Keywords
Introduction
Phosphorus (P) is an important nutrient that restricts life such as microbial production in many freshwater and marine environments (Hudson et al., 2000). However, excessive discharge of phosphorus into water bodies such rivers, lakes, and inland seas has resulted in eutrophication and degradation of water quality (Correll, 1998; Smith et al., 2014; Smith and Schindler, 2009).Cultural eutrophication even has become the primary water quality issue for most of the freshwater and coastal marine in recent decades (Smith and Schindler, 2009). Thus, it is significant to reduce anthropogenic inputs of P into aquatic ecosystems. For example, emission standards of total phosphorus (TP) is generally controlled to ≤1.0 mg l−1 (Class I A emission standard) for waste water in China. And water quality standard of TP for river or stream is 0.2 mg l−1 at level of Class III in China. Additionally, it is also important to reduce P from water bodies directly to control algal blooming.
Adsorption technology has been widely developed for removal of P in water (Lǚ et al., 2013; Reitzel et al., 2013; Yu and Paul Chen, 2015). This is due to several advantages of adsorption technology, which would be easily and flexibly applied in industrial operations. Adsorption technology is a cost-effective technology for treatment of that water containing phosphate, even at low phosphate concentrations (Ou et al., 2007; Yu and Paul Chen, 2015). Additionally, concentrated phosphates could be recovered from the spent sorbent, which could be reused. Also, the spent sorbent itself can be easily regenerated and recycled for several times. Several sorbents have been developed for phosphate removal such as multivalent metal (e.g. Fe3+, Al3+, Mg2+) oxides or hydroxides (El Bouraie and Masoud, 2017; Gerke, 1993; Li et al., 2016; Tanada et al., 2003), clay minerals (e.g. montmorillonite) (Edzwald et al., 1976), slag (Xue et al., 2009), fly ash (Lu et al., 2009), and red mud (Yue et al., 2010).However, some adsorbents were first limited by low adsorption capacity, low mechanical intensity, and second, by pollution. Thus, some new adsorbents have been investigated continuously.
Lanthanum (La) is known to have a high affinity for phosphate, which is also relatively abundant in the earth’s crust and environmental friendliness (Xie et al., 2014b). For example, the maximum capacity could be approach to 107.53 mg g−1 for synthesized La hydroxide (Xie et al., 2014b). Other La-typed sorbents have also been developed recently, which showed high adsorption capability for phosphate (Wang et al., 2018a; Xie et al., 2015, 2014b; Yu and Paul Chen, 2015). For example, a novel Fe–Mg–La tri-metal composite sorbent was developed that maximum adsorption capacity of phosphate could be approached to 415.2 mg g−1 under pH 6.0, whichwas much higher than most of other sorbents' adsorption capacity reported previously (Yu and Paul Chen, 2015).
It is well known that iron and manganese (hydr) oxides exist widely in natural aquatic system, which play an important role in geochemical cycling of P (Zhu et al., 2018, 2015). Thus, removal of phosphate in water or control of phosphate release from sediments by Fe or Mn (hydr) oxides would be safe for aquatic system. Actually, a Fe–Mn binary oxide has been developed, whose adsorption capacity (48.3 mg−1 at 25°C) was higher than their reported single component oxide (Lǚ et al., 2013). However, adsorption capacity of Fe–Mn binary oxide was not excellent. Thus, a novel Fe–Mn–La tri-metal composite sorbent was developed and first reported in this study, which would be less costly than La (hydr) oxide only, and also environmental friendly. Adsorption experiments including adsorption kinetics, adsorption isotherms, as well as thermodynamic parameters, and the influence of pH, anions, and dissolved organic matter (DOM) were studied. The characteristics of the sorbent before and after phosphate adsorption were analyzed, thus the mechanisms of phosphate adsorbed by Fe–Mn–La sorbent were discussed.
Material and methods
Materials
All chemicals were of analytical reagent grade and purchased from Sinopharm Chemical Reagent Company (China). Soil samples were collected from Jiufeng Forest Park of Beijing in October 2009. Fulvic acid (FA) was extracted from these soil samples. Extraction and purification of FA from these soil samples was performed according to the method recommended by the International Humic Substances Society (IHSS) (IHSS, 2017). The phosphate stock solution (500 mg l−1) was prepared from anhydrous potassium dihydrogen orthophosphate. All solutions were prepared using ultrapure water.
The Fe–Mn–La sorbent with Fe:Mn:La molar ratio of 1:1:1 was prepared by a co-precipitation method (Liu and Hesterberg, 2011; Yu and Paul Chen, 2015). A certain amount of Fe(NO3)3·9H2O, MnSO4, and La(NO3)3·6H2O was dissolved in ultrapure water. Sodium hydroxide solution (1 mol l−1) was then added into the solution until the mixed solution pH at 8.00 ± 0.20. The solution was continuously mixed for 1 h. The formed precipitate was aged at room temperature for 24 h, and then washed several times with ultrapure water. The precipitate was filtered and dried in the oven at 65°C for 24 h.Finally, the dried Fe–Mn–La tri-metal material (FML) was ground into fine powder for the sequence experiments.
Adsorption experiments
Adsorption kinetics
The adsorption kinetics experiments were carried out at an initial phosphate concentration of 20 mg l−1. And the pH of solution was controlled approaching to neutral. The FML adsorbents (20 mg) were added to a group of 50 ml centrifuge tubes. The prepared samples were taken under a present schedule (from 0.5 to 24 h). After adsorption experiments, the samples were centrifuged and the supernatants were filtered. The phosphate concentrations remaining in the filtered solutions were determined by a molybdate blue method (Murphy and Riley, 1962).
Adsorption isotherms
For the adsorption isotherm experiment, FML adsorbents (20 mg) were also added to a group of 50 ml centrifuge tubes. And then, 50 ml prepared phosphate solutions with concentrations varying from 20 to 100 mg l−1 were added to the tubes.These working solutions were shaken for 24 h at temperature of 289.15, 308.15, and 318.15 K for adsorption equilibrium testing. These solutions were then centrifuged and filtered, and phosphate concentrations remaining in the filters were determined by the molybdenum blue method. The amount of phosphate adsorbed was calculated by the difference in phosphate concentration measured before and after the adsorption equilibrium.
Influence factors
Influence factors including pH, anions (CO32−, SO42−, Cl−, and NO3−), DOM (FA, as a representative), and contents of FML adsorbent itself on adsorption were investigated. A series of phosphate solutions (25 mg l−1) with pH from 3.00 ± 0.10 to 11.00 ± 0.10 were prepared. The prepared phosphate solutions were added to a group of 50 ml centrifuge tubes. The FML adsorbents (20 mg) were added to these centrifuge tubes. These prepared samples were shaken for 24 h at room temperature. Also, phosphate solutions with different ionic strength (from 0.001 to 0.1 mol l−1) and concentrations of FA (from 10 to 120 mg l−1, calculated by C) were prepared. Other procedures were same as previous descriptions.
Finally, different contents of FML adsorbents (form 10 to 100 mg) were added to a group of 50 ml centrifuge tubes. And then, 50 ml phosphate solution (20 mg l−1) was added to the centrifuge tubes, respectively. Other procedures were the same as previous description.
Regeneration and recycle
The FML adsorbents (1.0 g) were weighted into a group of 50 ml centrifuge tube, and then 50 ml phosphate solutions (50 mg l−1) were added. The prepared samples were shaken for 24 h at 25°C and then centrifuged. The precipitate sorbents were dried at 65°C. After cooling, the sorbents were then washed by 0.5 M NaOH solution. The regenerated FML adsorbents would be used for adsorption experiments again. The regeneration and recycle experiments were conducted for four cycles to evaluate the reusability of the sorbent.
Adsorbent characterization
The prepared FML sorbent was characterized by SEM (JSM-7500F, JEOL Ltd Co., Tokyo, Japan) and XRD (Bruker D8 Advance, Bruker AXS, Germany). The zero point of charge (pHzpc) of the prepared FML sorbent was measured by using mass titrations (Xie et al., 2014a). In order to analyze the porosity and specific surface area of the prepared FML sorbent, nitrogen adsorption/desorption isotherms were measured using an ASAP 2020M surface area and porosity analyzer (Micromeritics Instruments Corp., USA).
The FML sorbents before and after adsorption of phosphate were analyzed by Fourier transform infrared (FT-IR) (Nicolet 6700, Thermo Scientific). X-ray photoelectron spectroscopy (XPS) analysis of the FML sorbent after adsorption of phosphate was performed by an XPS (ESCALAB 250, Thermo Fisher VG).
Data analysis
Adsorption capacity and removal efficiency
Adsorption capacity and removal efficiency were calculated by equations (1) and (2), respectively, as follows (Moharami and Jalali, 2013)
Adsorption isotherms
The isothermal adsorption curve was analyzed by Langmuir equation (3) and Freundlich equation (4) as follows (Wang et al., 2018b)
Adsorption kinetics
The pseudo-first-order kinetic equation (5), pseudo-second-order kinetic equation (6), Elovich kinetic equation (7), and intraparticle diffusion equation (8) were applied to analyze adsorption kinetics (Xie et al., 2019)
Thermodynamic data
Thermodynamic parameters of the adsorption process were analyzed according to the following equations (Tran et al., 2017)
Results and discussion
Characterization of Fe–Mn–La adsorbent
The SEM photograph of the prepared FML adsorbent shows that it has a rough and irregular surface (Figure 1(a)). The X-ray diffraction pattern of the FML adsorbent shows that it has some crystal structure peaks (Figure 1(b)). Thus, this composite sorbent was likely to be a mixture of crystal and amorphous structure. By contrast with previous report (Zhang et al., 2013), the peak present at 36.7° was δ-MnO2. The diffraction peaks at 35.6°, 43.2°, 57.3°, and 62.9° were thought to be Fe3O4 and/or Fe2O3 (Dong et al., 2010). The diffraction peak at 27.0°, 31.3°, 44.9°, and 53.2° was La2O3 (Wang et al., 2015).

Characterization of Fe–Mn–La adsorbent: (a) SEM photograph (5000×) and (b) XRD pattern.
The prepared FML adsorbent has a specific area of 89.36 m2 g−1, with the micro-pore volume (Vm) of 40.58 mm3 g−1 and total pore volume (VT) of 328.36 mm3 g−1. Additionally, the pHzpc of FML adsorbent is 3.55.
Adsorption kinetics
Amounts of phosphate adsorbed by the FML absorbents were increasing with time, and generally approached to dynamic equilibrium within 10 h (Figure 2). After 10 h, the adsorption capacity was increasing slowly, which is due to lack of available adsorption sites for phosphate in the surface of FML absorbents. Adsorption kinetics was fitted with pseudo-first-order kinetic equation, pseudo-second-order kinetic equation, Elovich kinetics model, and intraparticle diffusion equation, whose kinetic parameters are presented in Table 1. Based on the correlation coefficients (R2), the pseudo-second-order kinetic equation was the best model for description of adsorption of phosphate by the FML absorbent.These results suggest that surface chemical reaction between adsorbent active sites and phosphates is likely a dominant process for adsorption of phosphates by FML adsorbent (Lǚ et al., 2013).

Adsorption of phosphate on absorbent with time.
Kinetic parameters for the adsorption of phosphate on FML absorbent.

FML: Fe–Mn–La tri-metal material.
Adsorption isotherms and thermodynamics
The adsorption isotherms of phosphate on FML adsorbents at pH 6.03 under different temperatures are shown in Figure 3. The adsorption capacity increased significantly with equilibrium concentration of phosphate from 0 to 10 mg l−1. When the equilibrium concentration of phosphate increased above 30 mg l−1, the increase of adsorption capacity was less significant. The isotherms data are fitted by Langmuir model and Freundlich model (Figure 3 and Table 2). The calculated adsorption parameters and correlation coefficient (R2) for the two isotherm models by linear regression are listed in Table 2. Based on the correlation coefficient (R2) value for these isotherm models, it was found that the experimental data of phosphate adsorption on FML adsorbent could be described better by Langmuir at different temperatures (R2>0.990).This indicates that phosphate adsorption by FML adsorbents is mainly in monolayer coverage manner (Hu et al., 2018).

Equilibrium isotherms of phosphate on FML absorbent at different temperatures (a), line fitted to Langmuir model (b), and data fitted to Freundlich model (c).
Langmuir and Freundlich isotherm parameters for adsorption of phosphate on FML absorbent.

FML: Fe–Mn–La tri-metal material.
Based on Langmuir model, qm was 61.80, 65.83, and 71.68 mg g−1, respectively, at temperatures of 298.15, 308.15, and 318.15 K (Table 2). The adsorption capacity increased with temperature increasing. This indicates that the adsorption of phosphate by FML adsorbent is an endothermic process (Xie et al., 2019). The adsorption capacity of some different adsorbents including elements of Fe, Mn, or La is presented in Table 3. The adsorption capacity of phosphate by FML absorbent was significantly higher than that of Fe–Mn binary oxide adsorbent. Also, the adsorption capacity of FML absorbent was higher than that of magnetic Fe oxide, Mn oxide coated sand and crushed brick, commercial La hydroxide, and Fe–Al–Mn adsorbent. Comparatively, the FML adsorbent is a potential adsorbent for phosphate removal, which is also less costly than adsorbent containing element of La only.
Comparison of maximum adsorption capacities of phosphate by different adsorbents.

The thermodynamic parameters such as ΔG°, ΔH°, and ΔS° in the adsorption process were calculated out from adsorption isotherms of phosphate on FML adsorbent at different temperatures (Table 4). The ΔG° values varied from −19.71 to −18.21 kJ mol−1, which indicates that adsorption of phosphate by FML adsorbent is a spontaneous process. The increased negative values of ΔG° value with increasing temperature indicate that the adsorption reactions are thermodynamically more favorable at higher temperature. The positive ΔH° value (4.16 kJ mol−1) indicates that the adsorption is an endothermic process. Additionally, the positive ΔS° value (74.56 J mol−1 K−1) suggests that the degree of disorder increased when phosphate from the hydrous phase is adsorbed to the surface of the FML adsorbent (Lǚ et al., 2013; Wang et al., 2016).
Thermodynamic parameters for adsorption of phosphate on FML sorbent.
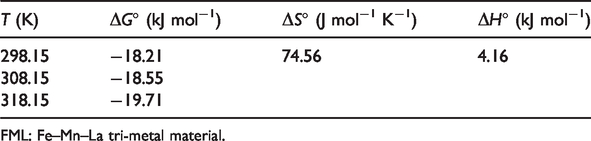
FML: Fe–Mn–La tri-metal material.
Effect of solution pH
The equilibrium adsorption capacities were decreased with pH varied from 3.02 to 11.00 (Figure 4). The FML adsorbent had relative high adsorption capacities within pH of 3.02–5.07. This is likely that the surface of adsorbent has been protonated, which increases the positive charge on the surface. This would be favorable to the adsorption of phosphate. Additionally, the positive groups dissociate more easily than hydroxyl groups in the surface of adsorbent, which would promote the exchange of ligands (Chubar et al., 2005). However, with the increase of pH, such as pH > pHZPC (the pH value of zero point of charge), an increased electrostatic repulsion of more negatively charged surface sites and electronegative phosphate species (from H2PO4− to HPO42−) resulted in the decrease of adsorption of phosphate (Liu et al., 2016; Lǚ et al., 2013; Zhou et al., 2005). The hydroxyl ions would compete with phosphate under alkaline condition, which also could reduce the adsorption of phosphate.

Adsorption of phosphate by FML adsorbent at different pHs.
Effect of coexisting anions and DOM
Coexisting anions such as Cl−, NO3−, SO42−, and CO32− are commonly distributed in waste water and natural water, which would compete with phosphate for active sites on the surface of adsorbents (Dai et al., 2014; Lǚ et al., 2013). Thus, the competition effects of some coexisting anions including Cl−, NO3−, SO42−, and CO32− on adsorption of phosphates by FML adsorbents were investigated (Figure 5). Generally, adsorption capacities (i.e. removal efficiencies) of phosphate by the FML adsorbents were decreased with increase of ionic strength (from 0 to 0.1 mol l−1) of Cl−, NO3−, SO42−, and CO32−. These four anions competing with phosphates on the FML adsorbents were in the order CO32− > Cl− >SO42− > NO3−. The presence of CO32− significantly reduced the removal efficiencies of phosphate by FML adsorbent, whose removal efficiencies were only 36.68% at the ionic strength of 0.1 mol l−1.This indicates that the adsorption of phosphate by FML adsorbent is strong effect by competition of CO32− with phosphate in aqueous solutions. The coexisting anion of CO32− also has a strong effect on adsorption of phosphate by other adsorbent such as Fe–Mg–La tri-metal composite sorbent (Yu and Paul Chen, 2015).

Effect of coexisting anions on adsorption of phosphate by FML adsorbent.
DOM is one of the most important colloids in natural water or waste water (Wu and Xing, 2009), which would influence the adsorption of phosphate by adsorbents such as Fe–Mn binary oxide (Lu et al., 2014) and sediments (Xie et al., 2019). FA is a representative DOM in nature (Wu and Xing, 2009). The effect of FA on adsorption of phosphate by FML adsorbent is shown in Figure 6. The removal efficiencies of phosphates by FML adsorbent were decreased with increase of FA concentrations in the prepared solutions. In this process, adsorption capacities of FA by FML adsorbents were also increased. This indicates that the competition adsorption between phosphate and FA has occurred on the surface of FML adsorbent. The DOM such as FA contains a large amount of carboxylic, phenolic, alcoholic, and amino groups, which would be competitive adsorption with phosphates (Wu and Xing, 2009; Xie et al., 2019).

Effects of FA on adsorption of phosphate by FML adsorbent. FA: fulvic acid.
Effect of adsorbent dosage and regeneration
With increase of adsorbent from 0.2 to 0.8 g l−1, the equilibrium adsorption capacity (qe) was decreased (Figure 7). However, the removal efficiency (η) was increased from 57.39 to 97.82%. Generally, the removal efficiency and adsorption capacity tended to be stable with the increase of adsorbent. When the content of adsorbent is low, the active sites would be fully exposed to the interactions with phosphate and the sites would be also saturated rapidly (Hosni and Srasra, 2010), while high contents of adsorbent would provide enough sites for adsorption of phosphate. The optimum adsorbent dosage could be checked by concentration of phosphate when FML adsorbent is applied.

Effect of adsorbent dose on the adsorption capacity (the initial phosphate concentration was 20 mg l−1).
The adsorption capacities were decreased significantly with increase of pH, especially under alkaline pH conditions (Figure 4). Thus, a 0.5 M NaOH solution was used for regeneration of spent sorbent. Although the adsorption capacity (i.e. removal efficiency) was decreased with number of cycles, the removal efficiency still could be reached to 78.30% after four cycles (Figure 8). This indicates that the FML adsorbent has a good reusability in the removal of phosphate from aqueous solutions.
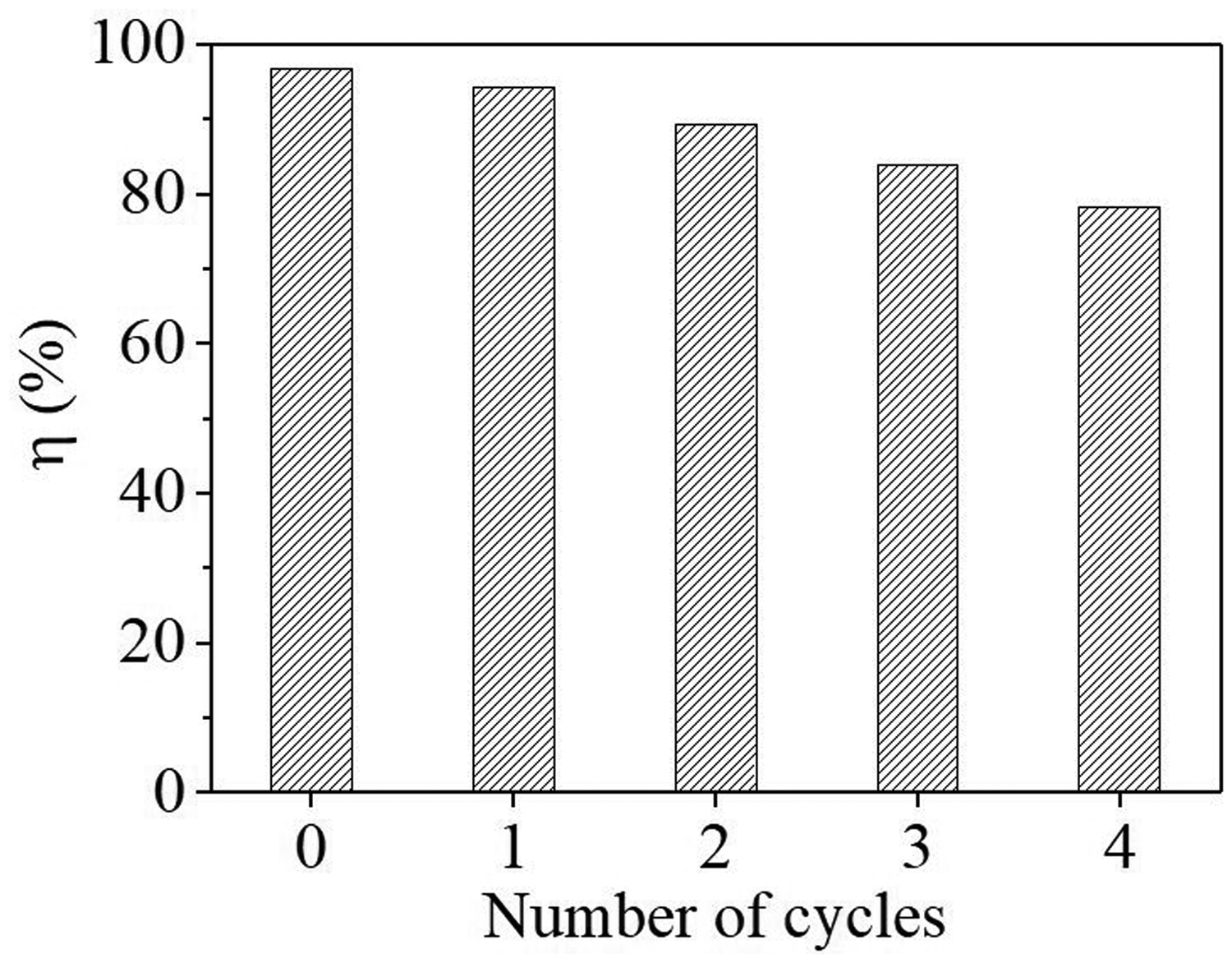
Regeneration/recycle of the spent sorbent.
Adsorption mechanisms
The FT-IR spectra of adsorbents before and after phosphate adsorption are shown in Figure 9. For these two samples, two peaks at 3600–3100 and 1625 cm−1 were assigned to stretching and bending vibrations of H–O–H, indicating that physisorbed H2O molecule on the surface of the FML adsorbents (Apte et al., 2007; Lǚ et al., 2013; Yu and Paul Chen, 2015). The appearance of the peak at 1120, 1072, and 979 cm−1 could be primarily attributed to the bending vibration of hydroxyl groups of iron (hydro)oxides (Fe-OH) (Zhang et al., 2009). The peaks of 1120, 1072, and 979 cm−1 disappeared on the surface of the FML adsorbent after adsorption of phosphate. While a new peak appeared at 1032 cm−1 in FT-IR spectra of adsorbents after phosphate adsorption, which could be assigned to the asymmetry vibration of P–O (Persson et al., 1996; Zhang et al., 2009). These results indicate that the group of –OH has been replaced by the adsorbed PO43−, HPO42−, or H2PO4−.

FT-IR spectra of the FML sorbents before (a) and after (b) adsorption by phosphate.
The FML adsorbent after adsorption by phosphate was further analyzed by XPS spectra (Figure 10). The results show that the P2p core level peak appears in the XPS wide scan spectrum (Figure 10(a)), indicating that the phosphate is successfully adsorbed onto the sorbent. The XPS spectra of P2p could be divided into three component peaks with binding energy of 132.9, 133.95, and 134.65 eV (Figure 10(b)), which is assigned to PO43−, HPO42−, and H2PO4−, respectively. The XPS spectra of Fe2p show the presence of two photoelectron peaks at 711.1 and 724.9 eV (Figure 10(c)), corresponding to the binding energies of Fe 2P3/2 and Fe 2P1/2, respectively, whose shapes are characteristic of Fe(III) (Yamashita and Hayes, 2008; Zhang et al., 2007). The XPS spectra of Mn2p show the presence of two photoelectron peaks at 642.2 and 653.3 eV (Figure 10(d)), corresponding to the binding energies of Mn 2P3/2 and Mn 2P1/2 transitions of Mn(IV), respectively (Zhang et al., 2007). The results suggest that the Fe and Mn in the FML sorbent are in the oxidation states Fe3+ and Mn4+, respectively. Aggregately, the possible mechanisms of phosphate adsorbed by FML adsorbent could be described as Figure 11.

XPS spectra of the FML adsorbents after adsorption by phosphate: (a) wide scan XPS spectra, (b) P2p spectra, (c) Fe2p spectra, and (d) Mn2p spectra.

Schematic diagram of possible adsorption reaction of phosphate by the FML sorbents.
Generally, the possible adsorption mechanisms of phosphate by adsorbents including Fe–Mn–La (Figure 11), Fe–Mg–La (Yu and Paul Chen, 2015), Fe–Mn (Zhang et al., 2009), and Fe–Al–Mn (hydr)oxide (Lǚ et al., 2013) are similar. However, the adsorption capacities are different between these adsorbents (Table 3). Comparative studies of these adsorbents under the same conditions are needed for clarifying mechanisms for different adsorption capacities, which would be a favor for developing much more phosphate removal adsorbents further.
Conclusions
A novel Fe–Mn–La tri-mental composite sorbent was developed for phosphate removal by co-precipitation. The sorbent showed a rough and irregular surface. Adsorption of phosphate by FML adsorbent reached to the dynamic equilibrium within 10 h, which could be described by a pseudo-second-order kinetic equation. Compared with Freundlich equation, Langmuir model fitted better in describing the adsorption isotherms. The maximum adsorption capacity could be reached to 61.80 mg g−1 at 25°C under pH of 6.03. Phosphate could be well adsorbed by FML adsorbent, especially in low concentration.The dominant process for adsorption of phosphate by FML adsorbent was surface chemical reaction and mainly in monolayer coverage manner. The thermodynamic study indicates that adsorption reaction is favorable at higher temperature as an endothermic process.
The phosphate removal gradually decreased with the increasing of pH from 3.02 to 11.00. The sequence of coexisting anions competing with phosphate was CO32− > Cl− > SO42− >NO3−. DOM would also decrease adsorption capacities of phosphates by FML adsorbents. Adsorption capacities decreased with increasing addition of adsorbents, while removal efficiencies increased in this process. The optimum adsorbent dosage could be checked by concentration of phosphate. The FML adsorbent has a good reusability in the removal of phosphate. The FT-IR and XPS analyses indicate that the group of –OH on the surface of adsorbent has been replaced by the adsorbed PO43−, HPO42−, or H2PO4−. The novel Fe–Mn–La tri-mental composite sorbent is very promising for the removal of phosphate from aqueous solutions.
Footnotes
Acknowledgment
We thank Zhongqi He, USDA-ARS Southern Regional Research Center, for help with manuscript improvement.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported in part by the National Science Foundation of China (21777001, 41877380, 21107001, and 41630645), Natural Science Foundation of Anhui Province (1608085MB43), key Research and Development Program of Anhui Province(No. 202004i07020006), and Foundation of Anhui Provincial Depart of Education (No. KJ2018ZD049).