Abstract
The adsorption of phosphate ion onto natural reed (
Introduction
The intensive use of fertilizers, pesticides, as well as the discharge of incompletely treated wastewater has become a great environmental concern. Nutrients contained in the aforementioned sources, namely phosphorous (P) and nitrogen loaded into the aquatic environment. This accumulation poses an ecological problem, as phosphorous is a key factor that stimulates significant water eutrophication (Hong-Bing et al., 2012). A variety of phosphate ion (
Many adsorbents prepared from agricultural residues have been tested and investigated such as apple pomace (Robinson et al., 2002), coconut husk (Manju et al., 1998), and sawdust (Ajmal et al., 1998). However, the majority of tested adsorbents are not good options for large scale use due to their low phosphate removal capacity and high cost. Nowack and Stone (2006) reported a maximum phosphate adsorption capacity of 5.7 mg
The objective of the current work is to study the adsorption of phosphate ion onto
Experimental analysis
Preparation of Arundo donax reeds
The The reeds were collected, dried naturally under the sunshine, and then crushed into small pieces. They were ground in order to get smaller sizes. Afterward, the reeds were sieved in order to obtain particles with diameters in the range of 0.15 to 0.85 mm. The particles with the appropriate size were washed with distilled water, and then they were dried at 108℃ for 8 h.
The phosphate aqueous concentration (20, 40, 60, and 100 mg/L) was determined by the Vanadomolybdo-phosphoric acid colorimetric method, using a Hach DR/4000 spectrophotometer (APHA, 1989). The phosphate solution containing reeds was shaken with a shaking water bath. The pH of all samples was measured using a pH meter.
Phosphate removal
Effect of adsorbent and adsorbate dosage on phosphate removal
A stock solution of 1000 mg/L of phosphate was prepared by dissolving potassium dihydrogen phosphate (KH2PO4) in distilled water. The stock solution of the adsorbate was diluted to an initial concentration of 100 mg/L. The pH of the standard solution was adjusted to 6.5. The following masses: 0.5, 1, 1.5, and 2 g of adsorbent were added to 20 mL of the standard in 100 mL glass bottles, and the mixtures were shaken using a rotary shaker at 100 rpm and allowed to equilibrate over 24 h at 25℃. However, the minimum time needed to reach equilibrium is about 1 h. The bottles were then removed from the shaker and sent for analysis.
The previous procedure was repeated using different initial phosphate concentrations (20, 40, and 60 mg/L), respectively. Each run was made in duplicate.
Effect of pH on phosphate removal
The effect of pH on phosphate removal was investigated in a range from 2 to 10. The pH was varied using hydrochloric acid (HCl) and sodium hydroxide (NaOH). The experiment was run at 25℃ using an initial phosphate concentration of 100 mg/L with 5 mg/mL of reeds. Mixtures were shaken at 100 rpm and allowed to equilibrate over a period of 24 h. Each run was made in triplicate.
Effect of temperature on phosphate removal
Batch adsorption experiments were carried out to study the effect of temperature on phosphate ion removal. The uptake of phosphate ion was measured using 5 mg/mL of reeds and at different initial phosphate concentrations (20, 40, 60, and 100 mg/L). These experiments were performed at various temperatures namely 25℃, 35℃, and 45℃.
Effect of salt addition on phosphate removal
This part was devoted to examine the effect of the presence of different salts on phosphate removal. Specifically, the effect of sodium chloride and potassium chloride was tested using salt concentrations ranging from 0 to 0.5 M. Each run was made in triplicate.
Kinetic study
Twenty milliliters of the stock solution (diluted to initial concentration of 100 mg/L) was placed into glass bottles containing 0.1 g of the adsorbent (reeds). The bottles were placed and agitated using isothermal bath shaker adjusted to the required value. Samples were taken out at different time intervals (15, 30, 45, 60, 180, and 300 min). Each run was made in triplicate.
The adsorbent was separated from the samples by filtration using a 0.1 µm filter paper.
The amount of phosphate ion adsorbed at equilibrium (
Three models (pseudo-first order, pseudo-second order, and the intra-particle diffusion model) were investigated in this part in an attempt to identify the adsorption rate and mechanism.
The relevant equations are (Ho, 2004; Hong-Bing et al., 2012):
Pseudo-first order model:
Pseudo-second-order model (Ho, 2004; Hong-Bing et al., 2012):
Intra-particle diffusion model



Validity and significance of each model can be verified using a normalized standard deviation Δq%, which can be calculated as follows:
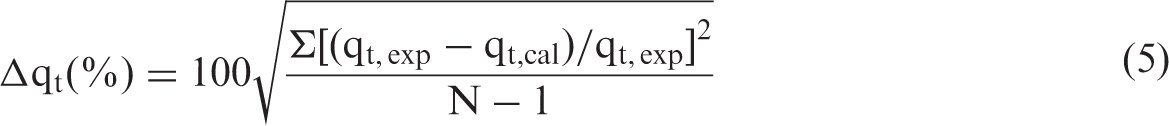
Adsorption isotherms
For the adsorption isotherm study, 0.1 g of reeds was added into glass bottles containing 20 ml of different phosphate initial concentrations (20, 40, 60, and 100 mg/L) at three temperatures (25℃, 35℃, and 45℃). The bottle-point technique was used to conduct the adsorption equilibrium test. The mixture was allowed to equilibrate over 5 h in a shaking water bath.
Results and discussion
Effect of adsorbent and adsorbate concentration
The effect of adsorbent concentration on phosphate adsorption onto reeds was investigated. Batch experiments were carried out with four different adsorbent amounts (5, 10, 15, and 20 mg/mL) at a fixed temperature, pH, contact time, and initial adsorbate concentration. This procedure was repeated with different initial phosphate concentrations (20, 40, 60, and 100 mg/L).
Figure 1 depicts the variation of phosphate ion uptake with the concentration of adsorbent in addition to the initial concentration of phosphate ion. It can be clearly seen that the phosphate ion removal process is monotonically increasing with phosphate ion initial concentration and monotonically decreasing with the amount of adsorbent. A maximum phosphate ion uptake of 16.2 mg/g was obtained at 100 mg/L and 5 mg/mL as phosphate initial concentration and reeds concentration, respectively. The decrease in the amount of phosphate adsorbed with increasing the adsorbent mass is due to the concentration gradient between the phosphate concentration in the bulk solution and the liquid phosphate concentration on the surface of the adsorbent. Therefore, with increasing the mass of adsorbent, the mass of phosphate adsorbed per unit weight of adsorbent get reduced (Ammari, 2014). The same concept is valid when increasing the initial concentration of phosphate in solution as this means altering the equilibrium and increasing the driving force of adsorption, which is the concentration gradient between the solution and the surface of adsorbent.
Effect of reeds concentration (ms) and initial phosphate concentration on phosphate (PO4−3) uptake (q) (rpm: 100; pH: 6.5; T: 25℃).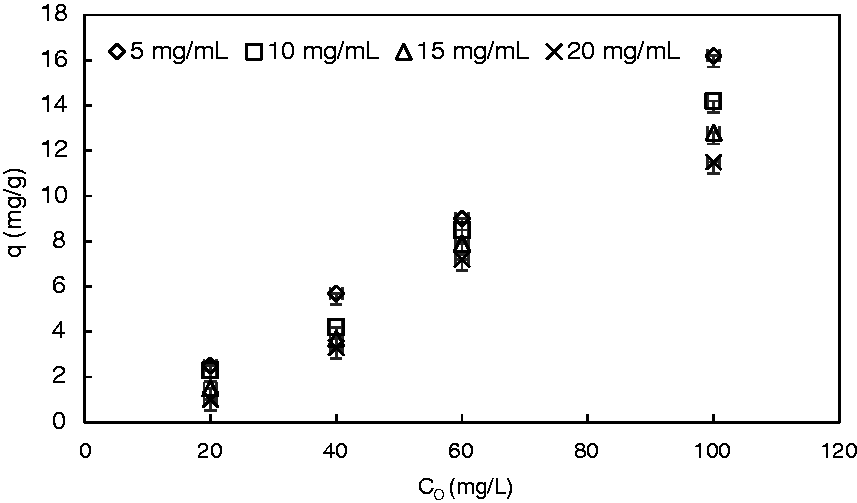
Effect of contact time
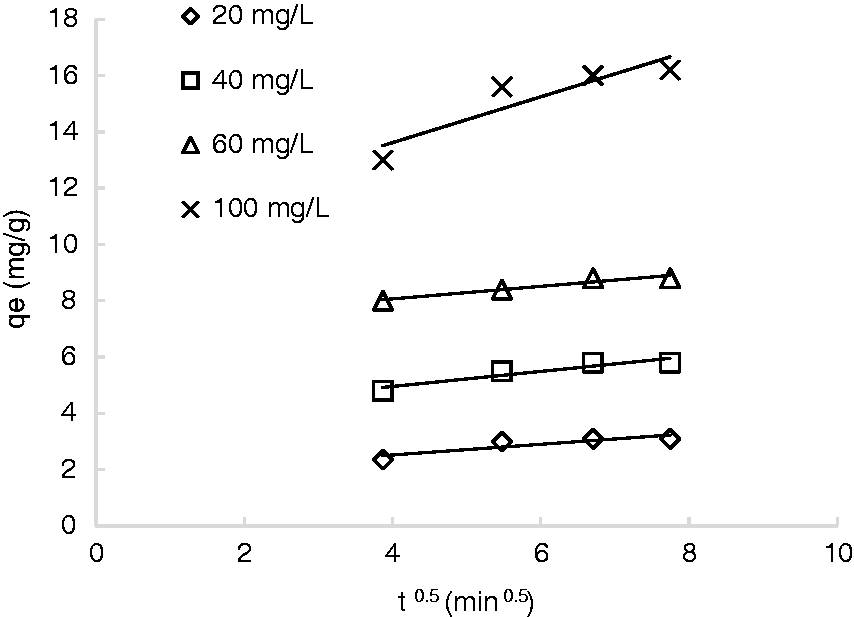
Batch assays were conducted to examine the phosphate uptake onto reeds during a contact time of 300 min using different initial phosphate ion concentrations (20, 40, 60, and 100 mg/L). The time course of the phosphate uptake in these batch tests is presented in Figure 2. As it is illustrated in Figure 2, for initial phosphate concentrations of 20, 40, and 60 mg/L, most of the phosphate ion is adsorbed during the first 15 min. After that, the rate of phosphate uptake started to stabilize with no further uptake noticed after 45 min. At this point, it was presumed that equilibrium was reached. At the initial phosphate ion concentration of 100 mg/L, the phosphate ion removal was rapid during the first 30 min, then proceeded at a slower rate until it reached equilibrium after 60 min. Hence, it was noticed that as the initial concentration increased, longer time may be required to attain equilibrium. This result is consistent with what was previously reported in literature (Kumar et al., 2010).
The kinetic curves of the amount of phosphate adsorbed onto reeds at different initial phosphate concentrations (rpm: 100; pH: 6.5; T: 25℃, ms: 5 mg/mL).
The initial rapid adsorption could be attributed to the high affinity sites and adsorption. Then, as the reaction proceeds, the adsorption of phosphate ion increases the overall surface negative charge, causing the adsorption rate to slow down. This is also consistent with what was previously reported in literature (Fazhi et al., 2014; Luengo et al., 2006; Xiaoming et al., 2013; Yue et al., 2010). In these studies, the time required to reach equilibrium for phosphate ion adsorption on modified giant reed, Geothite and Hematite was about 25 min.
Effect of initial pH
The significant impact of the aqueous solution pH on the sorption process of phosphate ion was addressed in several studies (Biswas et al., 2008; Krishnan and Haridas, 2008). pH has an important effect in determining the phosphate ionic form. Hence, the effect of initial pH on phosphate ion adsorption was studied in this work. It was evident from Figure 3 that the pH had a significant influence on the amount of phosphates adsorbed onto reeds. In the pH range from 2 to 7, the removal efficiency of phosphate from the solution increased until it reached a peak (16.2 mg/g) at the neutral pH value and then descended as the solution had become more alkaline. These findings are similar to the results reported by Yue et al. (2010) and Xu et al. (2011). In these studies, it was shown that the maximum effective removal of phosphate ions by cotton stalk/wheat stalk and modified giant reed occurred in the pH range of 4–9. These results were explained by the presence of dominant dihydrogen and hydrogen phosphate Adsorption of phosphate onto reeds as a function of pH (rpm: 100; T: 25℃, Co: 100 mg/L; ms: 5 mg/mL).
Effect of NaCl and KCl addition
This part is devoted to investigate the effect of interfering ions on the adsorption of phosphate ion onto reeds. Different concentrations of NaCl and KCl salts were added to the phosphate-containing solution. It can be noticed from Figure 4 that both salts adversely affected the adsorption of phosphate. The latter observation could be ascribed to a competitive adsorption with the co-existing chloride ions especially at the high salt concentration used. Fazhi et al. (2014) demonstrated that Cl− ions had negative effect on PO4−3 adsorption on magnesium oxide nanoflake-modified diatomite adsorbent. The phosphate uptake from a solution containing 5 mg/L PO4−3 was decreased when chloride-containing salt concentration was increased from 1 to 10 mM. However, the effect of chloride ions is different when adding it as NaCl or KCl salts. This could be supported by the work of Razaq (1989) in which he mentioned that phosphate adsorption varies with the nature of the ionic composition and the ionic strength of the supporting matrix solution. In other words, it depends on the cations added to phosphate containing solutions. It was reported also that the effect of different salts on adsorption depends strongly on pH value. In the later work, phosphate adsorption capacity was different in the presence of potassium and sodium cations at pH of 5.5, which is in accordance with findings in our work. However, this effect of cations on PO4−3 adsorption cannot be generalized as it changes with pH (Razaq, 1989).
Effect of salt addition on the uptake of phosphate by reeds (rpm: 100; pH: 6.5; T: 25℃; Co: 100 mg/L; ms: 5 mg/mL).
Effect of temperature
The effect of temperature on phosphate uptake by reeds was investigated using various phosphate initial concentrations. It is apparent from Figure 5 that the increase in temperature has a negative effect on the adsorption of phosphate, which leads to the conclusion that the sorption process of phosphate is of exothermic nature. This result was supported by a study conducted by Yue et al. (2010), using modified giant reed as a bio sorbent. They reported a similar temperature pattern as the phosphate adsorption capacity has decreased as the temperature increased from 20 to 60℃. This result was explained by the exothermic nature of the adsorption process.
Effect of solution temperature on the uptake of phosphate by reeds (rpm: 100; pH: 6.5; T: 25℃; ms: 5 mg/mL).
Thermodynamic studies


Thermodynamic parameters of phosphate onto reeds at different temperatures.

Kinetic studies
A kinetic study was performed to determine the rate at which the adsorption reaction is taking place as well as the adsorption mechanism. Three kinetic models were proposed in the analysis of the sorption data of phosphate onto reeds: pseudo-first order model, pseudo-second-order model, and the intra-particle diffusion model. The linearized form of the three models are depicted in Figures 6–8.
First-order adsorption plot for various phosphate concentrations onto activated reeds 25℃ and pH 6.5. Second-order adsorption plot for various phosphate concentrations onto activated reeds 25℃ and pH 6.5. Intra-particle diffusion model for adsorption of various phosphate concentrations onto activated reeds 25℃ and pH 6.5.

Kinetic parameters for adsorption rate of phosphate on activated reeds at different initial concentrations.
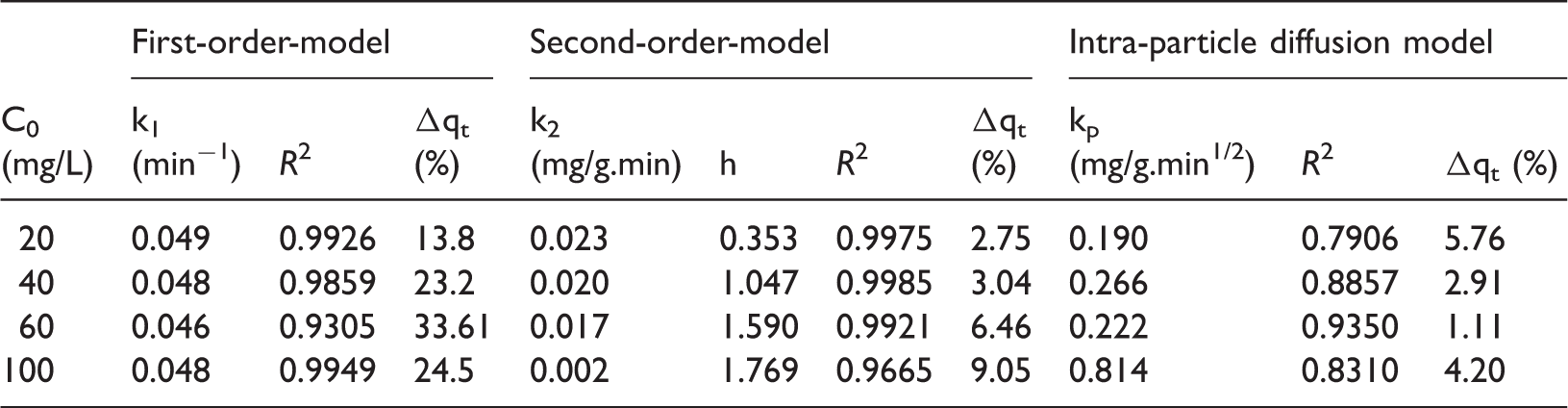
Based on the determination coefficients and normalized standard deviation, it was found that the adsorption data can be best fit by the second-order models. This model has shown (
Equilibrium isotherms
Various adsorption models have been used in literature to describe the equilibrium relationship between the liquid phase concentration and the surface concentration of the adsorbate. Two different models, Langmuir and Freundlich are used in this section to simulate the adsorption isotherms of phosphate ion onto reeds. The linear equation for Langmuir isotherm is as follows:
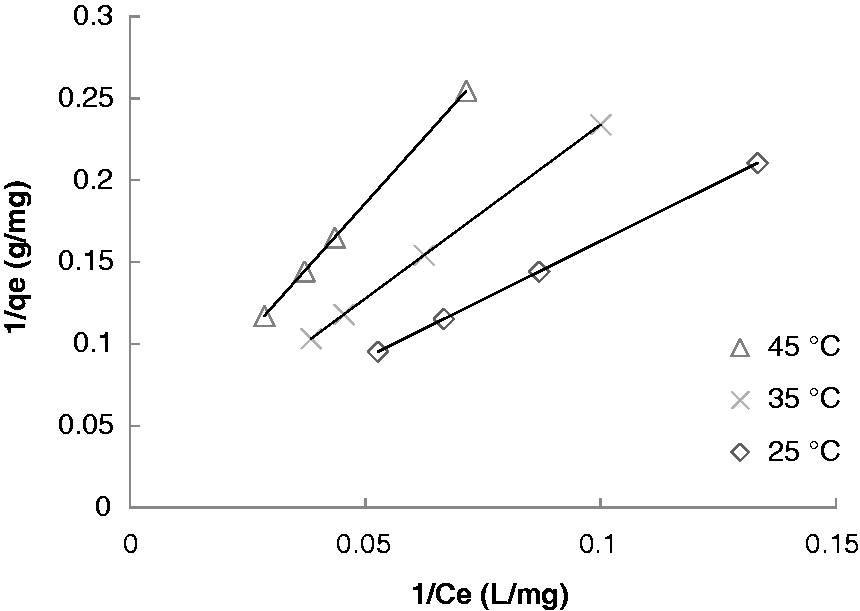
Langmuir isotherm for the adsorption of phosphate on reeds (rpm: 100; pH: 6.5; ms: 5 mg/mL).
The Freudlich isotherm theory states that the ratio of qe to Ce is not constant at different concentrations (Aksu, 2002). The isotherm is described by the following equation:

The linearized form of equation (9) is:

KF is the Freundlich constant related to the capacity of adsorption, 1/n is the Freundlich constant related to the intensity of adsorption. The Freundlich linear relations was used to fit the equilibrium data as shown in Figure 10.
Freundlich isotherm for the adsorption of phosphate on reeds (rpm: 100; pH: 6.5; ms: 5 mg/mL).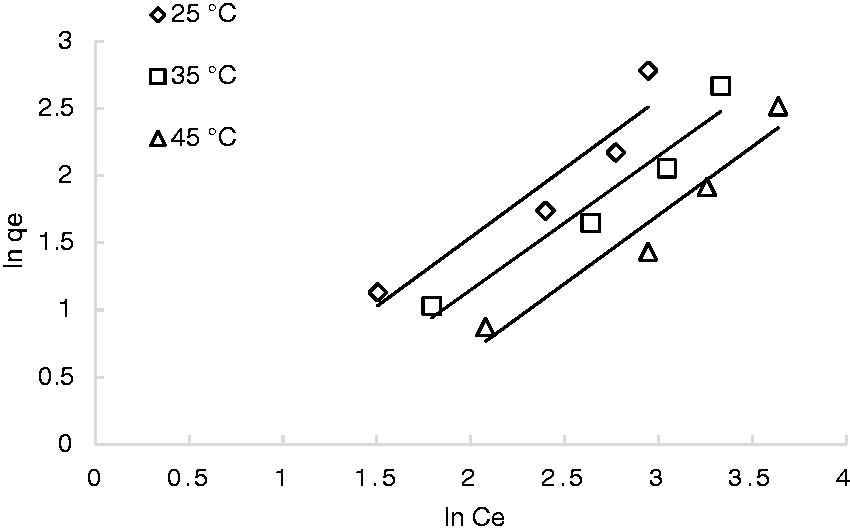
Langmuir and Freundlich's constants related to adsorption of phosphate from aqueous solution by onto reeds.

Based on the determination coefficients, it can be concluded that the data were best represented by Langmuir isotherm. Consequently, it can be suggested that the adsorption took place in the form of monolayer on a homogeneous surface in terms of energies of sorption. It assumes also that no transmigration of sorbate occurs on the plane of the surface (Wu, 2007). This result is in good agreement with the results reported by Yue et al. (2010). As it is also indicated in Table 3, the adsorption capacity Qm decreases with temperature, which is in agreement with the exothermic nature of the sorption process previously mentioned.
Conclusion
This work investigated the possibility of using
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
